Neuromyelitis optica spectrum disorder (NMOSD) is an autoimmune disease that usually presents with optic neuritis or severe myelitis. NMOSD can be diagnosed after only one typical clinical syndrome if immunoglobulin G (IgG) antibodies against the aquaporin-4 receptor (NMO-IgG) are present.1 Patients with NMOSD can develop marked spinal cord atrophy after myelitis,2,3 but it is unknown whether atrophy can occur without a history of myelitis. To address this question, we compared the mean upper cervical cross-sectional area (MUCCA) in patients with NMOSD without MRI cord lesions or history of myelitis, patients with NMOSD with MRI cord lesions or history of myelitis, patients with multiple sclerosis (MS), and neurologically intact controls.
Methods.
Participants.
We identified 33 NMO-IgG-positive patients with NMOSD, 33 age- and disease duration–matched patients with MS from the NYU MS Care Center, and 20 age-matched neurologically intact controls whose cervical spine and brain MRI scans, performed for headache or neck pain, were normal. All subjects were women. We correlated cervical spinal cord atrophy with disability via patient-derived disease steps (PDDS) score.4
Protocol approvals, registrations, and patient consents.
The study was approved by the local institutional review board.
Statistical analysis.
We used analysis of covariance to compare groups for MUCCA adjusted for age when the comparison involved the control group, and adjusted for age and disease duration when the 2 patient groups were compared. Spearman rank correlations were used to evaluate associations for MUCCA with disability score, age, disease duration, lesion length ratio, and episodes of myelitis.
MRI data.
We included cervical studies performed on 1.5T or 3T MRI scanners with slice thickness between 3 and 5 mm. Scans with gadolinium-enhancing lesions were excluded. A board-certified neuroradiologist grouped the patients with NMOSD into those with and without lesions in the cervicothoracic spinal cord. For the patients with NMOSD, lesion ratio was calculated as the total vertical span of lesions from C2/C3 to C6/C7 divided by the total length over this cervical region.
MRI postprocessing.
T2-weighted axial images were processed using PropSeg semiautomated segmentation software.5 We calculated the MUCCA by averaging the cross-sectional areas from sections between the C2/C3 intervertebral space and the C3/C4 intervertebral space.
Results.
Clinical data.
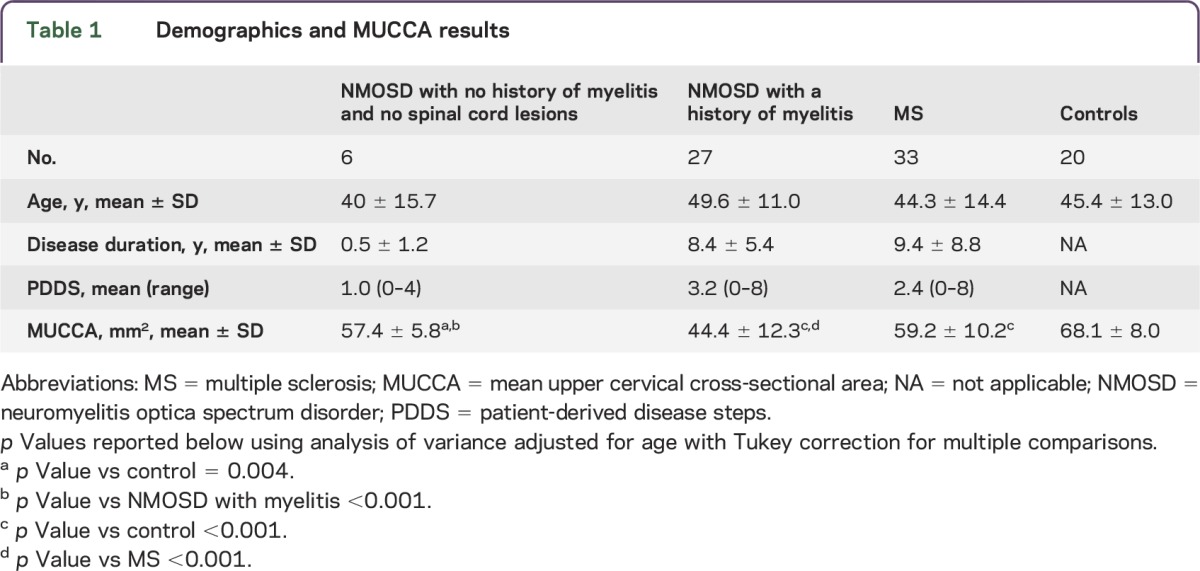
We identified 13 patients with NMOSD without cervical lesions, 6 of whom also had no history of myelitis and no visible lesions throughout the cord (table 1 and table e-1 at Neurology.org/nn).
Table 1.
Demographics and MUCCA results
Spinal cord cross-sectional area.
We first evaluated patients with NMOSD without lesions in the region of the MUCCA analysis in order to avoid focal effects of prior inflammation on the cord cross-sectional area. We found that patients with NMOSD without upper cervical lesions had a 16% decrease in MUCCA vs neurologically intact controls (57.0 ± 4.2 mm2 and 68.1 ± 8.0 mm2, respectively; p < 0.001). We then examined the subset of 6 patients without either a clinical history of myelitis or MRI lesions to avoid potential anterograde/retrograde effects of inflammatory damage elsewhere in the spinal cord and found that MUCCA in this group (57.4 ± 5.8 mm2) was also decreased 16% compared to controls (p = 0.004; table 1, figures e-1 and e-2). MUCCA of the patients with NMOSD without MRI lesions or myelitis was larger (57.5 ± 5.4 mm2) than in patients with NMOSD with a history of myelitis (44.4 ± 12.3 mm2), but similar to that of patients with MS (59.2 ± 10.2 mm2).
Correlations between cervical cross-sectional area and disability.
For the patients with NMOSD but no upper cervical lesions (n = 9 with PDDS disability scores available), MUCCA correlated with the disability score (R = −0.86, p = 0.007; adjusted for age), but not with disease duration, episodes of myelitis, or age. For the entire NMOSD cohort (n = 22 with PDDS available), MUCCA correlated well with disability (R = −0.72; p < 0.001; adjusted for age, episodes of myelitis, and lesion length ratio).
Discussion.
We found cervical spinal cord atrophy in patients with NMOSD without a clinical history of myelitis or any spinal cord lesions on MRI, suggesting that cervical atrophy may occur without prior destructive attacks or degeneration related to lesions elsewhere in the spinal cord. A number of studies have documented diffuse brain atrophy in patients with NMOSD,6,7 and it is possible that cord atrophy seen in our patients also is due to a diffuse underlying process that occurs without myelitis. Alternatively, or perhaps co-contributory, the patients with NMOSD without visible lesions may have experienced transient or subclinical inflammatory events that resulted in only subtle spinal cord pathology not visible on conventional MRI.
Similar to previous reports,2 we found a correlation between MUCCA and general disability, suggesting a potential role for MUCCA as a biomarker for disease progression in clinical trials for NMOSD. Whether lower MUCCA has diagnostic and prognostic value in NMOSD requires further investigation. Longitudinal studies on patients with NMOSD without myelitis are necessary to validate our findings and better characterize the atrophy process involving the cord.
Supplementary Material
Footnotes
Supplemental data at Neurology.org/nn
Author contributions: Dr. Ventura: study concept and design, analysis and interpretation, data acquisition, critical revision of the manuscript for important intellectual content. Dr. Kister: study concept, analysis and interpretation, critical revision of the manuscript for important intellectual content. Dr. Chung: data acquisition, analysis and interpretation. Dr. Babb: statistical design, analysis and interpretation. Dr. Shepherd: study concept and design, analysis and interpretation, critical revision of the manuscript for important intellectual content.
Study funding: This research was supported in part by the Center for Advanced Imaging Innovation and Research, a National Institute for Biomedical Imaging and Bioengineering Biomedical Technology Resource Center, for grant number P41EB017183.
Disclosure: R.E. Ventura reports no disclosures. I. Kister is on the scientific advisory board for Biogen Idec, MS Franchise Data Generation; has consulted for Biogen Idec; and received research support from Biogen Idec, Serono, Novartis, Guthy-Jackson Charitable Foundation, and National Multiple Sclerosis Society. S. Chung reports no disclosures. J.S. Babb is deputy editor for Journal of Magnetic Resonance Imaging. T.M. Shepherd has been a speaker for Siemens Healthcare (without travel funding/speaker honoraria), holds a patent for a device for image-guided procedures, MRI method for selecting irradiation dosage during tumor treatment, spends 25% of his time reading spine and brain MRIs, and received research support from NIH. Go to Neurology.org/nn for full disclosure forms. The Article Processing Charge was paid by the authors.
References
- 1.Wingerchuk DM, Banwell B, Bennett JL, et al. International consensus diagnostic criteria for neuromyelitis optica spectrum disorders. Neurology 2015;85:177–189. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Liu Y, Wang J, Daams M, et al. Differential patterns of spinal cord and brain atrophy in NMO and MS. Neurology 2015;84:1465–1472. [DOI] [PubMed] [Google Scholar]
- 3.Asgari N, Skejoe HP, Lennon VA. Evolution of longitudinally extensive transverse myelitis in an aquaporin-4 IgG-positive patient. Neurology 2013;81:95–96. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Learmonth YC, Motl RW, Sandroff BM, Pula JH, Cadavid D. Validation of patient determined disease steps (PDDS) scale scores in persons with multiple sclerosis. BMC Neurol 2013;13:37. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.De Leener B, Kadoury S, Cohen-Adad J. Robust, accurate and fast automatic segmentation of the spinal cord. Neuroimage 2014;98:528–536. [DOI] [PubMed] [Google Scholar]
- 6.Chanson JB, Lamy J, Rousseau F, et al. White matter volume is decreased in the brain of patients with neuromyelitis optica. Eur J Neurol 2013;20:361–367. [DOI] [PubMed] [Google Scholar]
- 7.Zhao DD, Zhou HY, Wu QZ, et al. Diffusion tensor imaging characterization of occult brain damage in relapsing neuromyelitis optica using 3.0T magnetic resonance imaging techniques. Neuroimage 2012;59:3173–3177. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.


