Abstract
Introduction
The risk of morbidity associated with venous thromboembolism (VTE) after pediatric orthopaedic surgery remains unclear despite increased use of thromboprophylaxis measures.
Methods
The American College of Surgeons National Surgical Quality Improvement Program, Pediatric database was queried for patients undergoing an orthopaedic surgical procedure between 2012 and 2013. Upper extremity and skin/subcutaneous surgeries were excluded. Associations between VTE and procedure, demographics, comorbidities, preoperative laboratory values, and 30-day postoperative outcomes were evaluated.
Results
Of 14,776 cases, 15 patients (0.10%) experienced postoperative VTE. Deep vein thrombosis (DVT) occurred in 13 patients (0.09%), and pulmonary embolism developed in 2 patients (0.01%). The procedure with the highest VTE rate was surgery for infection (1.2%). Patient factors associated with the development of VTE included hyponatremia (P = 0.003), abnormal partial thromboplastin time (P = 0.046), elevated aspartate transaminase level (P = 0.004), and gastrointestinal (P = 0.011), renal (P = 0.016), and hematologic (P = 0.019) disorders. Nearly half (46.2%) of DVTs occurred postdischarge. Complications associated with VTE included prolonged hospitalization (P = <0.001), pneumonia (P = <0.001), unplanned intubation (P = 0.003), urinary tract infection (P = 0.003), and central line-associated bloodstream infection (P = <0.001). Most of the postoperative complications (66.7%) occurred before VTE diagnosis, and no patients with VTE died.
Conclusion
In the absence of specified risk factors, thromboprophylaxis may be unnecessary for this population.
Keywords: venous thromboembolism, deep vein thrombosis, pulmonary embolism, pediatric orthopaedics, National Surgical Quality Improvement Program, morbidity
Venous thromboembolism (VTE) is a well-known complication of surgery and is associated with significant morbidity and mortality in adults and, to a lesser degree, in children.1–3 Rates of VTE are age-dependent, with persons aged <15 years experiencing a 100-fold reduction in risk for VTE compared with 80-year-old patients.1 However, children ≥13 years are at statistically significant greater risk for the development of VTE after trauma than are children aged birth to 12 years; the greatest increase in risk is seen after age 16 years.4 In pediatric patients, VTE is a rare event, occurring in 0.01% to 0.2% of pediatric hospitalizations.2,3,5 Identified risk factors for VTE in pediatric patients include trauma, increased age, central catheter line placement, and chronic conditions, such as malignancy, obesity, inflammatory bowel disease, and respiratory and cardiovascular comorbidities.3,6–9 Prophylaxis is recommended for certain at-risk pediatric patients; however, because of the rare incidence of VTE in children and the possible adverse events related to VTE prophylaxis (eg, bleeding), there is a need for further clarification of risk factors for the development of VTE and for establishing rates of VTE-associated morbidity and mortality.7,10 Improved identification of at-risk pediatric patients may help to refine guidelines for and implementation of VTE prophylaxis, especially because the developing trend is toward the use of widespread anticoagulation in adult patients.5
Despite the prevalent use of thromboprophylaxis in children, its benefits are not completely understood. Morbidity and mortality associated with VTE in children have not been well studied, further complicating the issue of prevalent use of thromboprophylaxis in this population. In general pediatrics, mortality following VTE varies from 1% to 8% (1% in a study examining death directly attributable to VTE, 8% in another study where any cause of mortality after VTE is included).2,3,5 VTE-associated mortality remains unknown in pediatric orthopaedic populations. Furthermore, the risk of morbidity as a result of serious VTE-associated postoperative complications is unclear.
Although the incidence of VTE and risk factors in pediatric surgical populations have been investigated,2,3,5 few studies have specifically focused on rates, risks factors, or VTE-associated complications in orthopaedic populations. In pediatric spine surgery, patients with congenital scoliosis, syndromic scoliosis, or thoracolumbar fractures are at an increased for the development of VTE complications compared with other spine surgery patients.11 In a retrospective review of 143,808 elective pediatric orthopaedic procedures, older age, admission type, metabolic conditions, obesity, and complications of implanted devices and/or surgical procedures were associated with deep vein thrombosis (DVT).12 A recall survey of 354 pediatric orthopaedic surgeons identified prior DVT, prior pulmonary embolism (PE), family history of DVT, oral contraceptive use, and current malignancy as perceived predisposing factors for DVT.13 In the spine study by Jain et al,11 the authors used the Nationwide Inpatient Sample; this is an inpatient-only database with no postdischarge follow-up, and it has a tendency to overestimate risk factors.14 In a large retrospective review by Georgopoulos et al,12 the authors analyzed elective orthopaedic procedures only and did not examine preoperative laboratory values, gastrointestinal (GI) comorbidities, pulmonary comorbidities, cardiovascular comorbidities, or renal comorbidities as risk factors for VTE, several of which have been previously identified as risk factors for VTE in children.3,6–9 In addition, procedures or diagnoses associated with trauma, infection, coagulopathies, or malignancy were excluded, as well as patients who had procedures performed after the day of admission. To our knowledge, none of these studies has examined adverse postoperative outcomes associated with VTE in the general pediatric orthopaedic population.
VTEs have been deemed by the Centers for Medicare and Medicaid Services as hospital-acquired conditions, or “never events,” in most instances,15 and recently instituted a policy of not reimbursing hospitals for such complications in adults and children. As hospitals seek to prevent these infrequent events, some institutions have established standard orders for thromboprophylaxis in children, despite a lack of evidence in favor of such a practice. The object of this study is to identify whether certain patient groups are at risk for the development of postoperative VTE complications, including DVT and PE, in the general pediatric orthopaedic patient population and to examine associated adverse postoperative outcomes using a large, nationally representative database.
Methods
The American College of Surgeons National Surgical Quality Improvement Program, Pediatric (ACS NSQIP Pediatric) is a prospectively collected nationwide cohort of pediatric surgical cases with >50 participating institutions.16 The program trains nurses and data abstractors at each participating institution to record variables, such as patient demographics, laboratory results, comorbidities, postoperative complications, readmissions, and mortality. Patients are contacted by telephone or letter after discharge to collect accurate follow-up data up to 30 days postdischarge. For this study, data from the 2012 and 2013 NSQIP Pediatric database were analyzed. The authors’ institution does not require Institutional Review Board approval because the NSQIP Pediatric data are de-identified and HIPAA-compliant.
Case Selection and Statistical Analysis
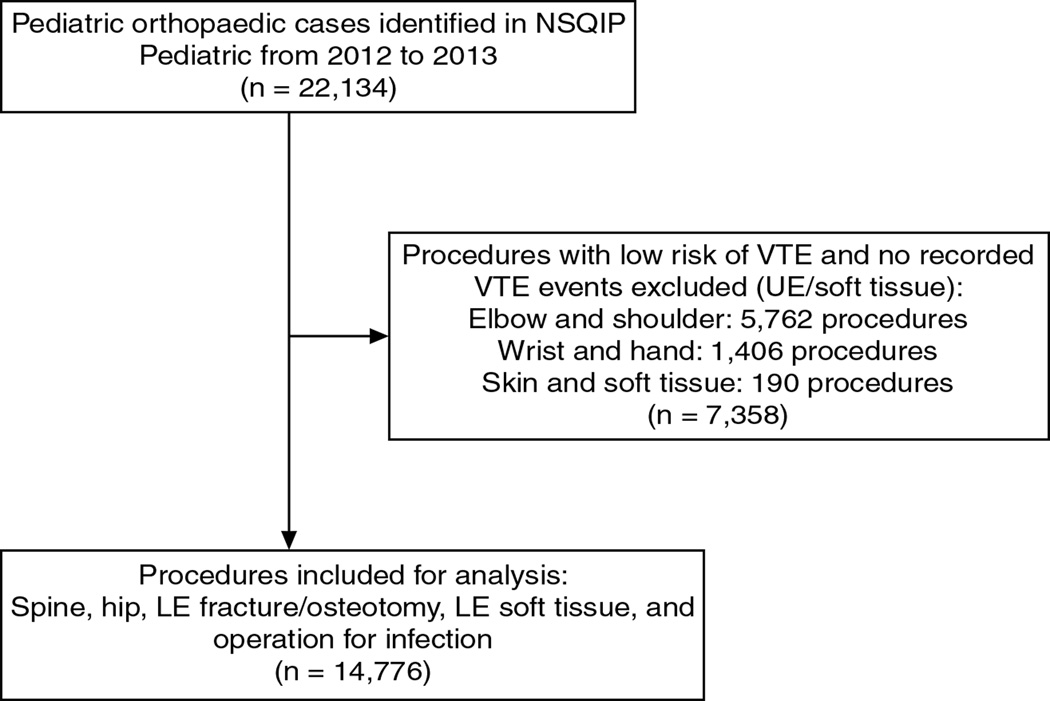
In this retrospective study, pediatric orthopaedic cases were filtered from the NSQIP Pediatric database according to orthopaedic surgical subspecialty by querying the surgical specialty variable for “Orthopaedics” and “Pediatric Orthopaedic Surgery.” Pediatric orthopaedic cases were then divided into categories by surgical procedure type using Current Procedural Terminology codes for classification. Categories for surgical procedure type included (in order of decreasing frequency): elbow, spine, lower extremity (LE) fracture/osteotomy, LE soft tissue, hip, hand/wrist, skin/subcutaneous, and infection. Upper extremity structures (ie, elbow, shoulder, wrist, and hand), skin, and subcutaneous procedures were excluded from the analysis because they had a low risk of VTE, had no recorded VTE events in the NSQIP, and typically would not be considered for prophylaxis. Spine, hip, LE soft tissue, LE fracture or osteotomy, and infection procedures were included for analysis (Figure 1).
Figure 1.
Case cohort selection from the National Surgical Quality Improvement Program (NSQIP) database by procedure type. LE = lower extremity, UE = upper extremity, VTE = venous thromboembolism
Demographics included age, gender, obesity, race, and the American Society of Anesthesiologists classification. Comorbidities included pulmonary, GI, renal, central nervous system, cardiac, cerebral palsy or other neuromuscular disorder, chronic steroid use, need for nutritional support, bleeding disorder, hematologic disorder, preoperative blood transfusion, inotropic support at the time of surgery, and childhood malignancy. A detailed list of the conditions included in each comorbidity categorical variable are shown in Table 1. Preoperative laboratory values included hypoalbuminemia, hyponatremia, elevated white blood cell count, thrombocytopenia, anemia, elevated aspartate transaminase (AST) level, abnormal prothrombin time, and abnormal partial thromboplastin time (PTT). Postoperative complications included length of stay, surgical site infections (ie, deep, incisional, organ/space), wound dehiscence, pneumonia, unplanned intubation, renal insufficiency, acute renal failure, urinary tract infection (UTI), coma, cerebrovascular accident, seizure, peripheral nerve injury, cardiac arrest, blood transfusion, graft/prosthesis failure, sepsis, and central line-associated bloodstream infection (CLABSI). The NSQIP provides different variables and/or codes for preoperative versus postoperative complications.16
Table 1.
Comorbidity Details for Conditions Included in ACS NSQIP-P Categorical Variables
| Comorbidity Categorical Variable | Conditions |
|---|---|
| Pulmonary | Asthma, ventilator dependency, oxygen support, bronchopulmonary dysplasia, pneumonia, cystic fibrosis, structural pulmonary abnormality |
| Gastrointestinal | Esophageal, gastric, intestinal, biliary, liver, pancreatic Disease |
| Renal | Acute renal failure, dialysis |
| Central nervous system | Coma >24 h, history of CVA or TBI, CNS tumor, developmental delay, seizure disorder, structural CNS abnormality |
| Cardiac | Previous cardiac surgery, congenital heart defects, aortic valve disease, cyanotic heart disease, hypertrophic cardiomyopathy |
| Bleeding disorder | Vitamin K deficiency, hemophilias, thrombocytopenia, chronic anticoagulation therapy that has not been discontinued before surgery |
| Hematologic disorder | Sickle cell disease, thalassemia, hereditary spherocytosis, thrombocytopenia, idiopathic thrombocytopenic purpura, neutropenia, Henoch- Schönlein disease, anemia (hemolytic, hypoproliferative, macrocytic, microcytic, normocytic, pernicious), basophilia, dysfibrinogenemia, eosinophilia |
ACS NSQIP-P = American College of Surgeons National Surgical Quality Improvement Program Pediatric database, CNS = central nervous system, CVA = cerebrovascular accident, TBI = traumatic brain injury
The association between VTE incidence and patient and surgical variables, including procedure type, patient demographics, comorbidities, preoperative laboratory values, surgical time, length of stay, and adverse postoperative outcomes, was evaluated using the Fisher exact test or the independent sample Student’s t-test, where appropriate. The small number of events precluded a multivariable analysis. Statistical analyses were performed using SPSS, version 22.0. Significance level was set when P = ≤0.05.
Results
A total of 14,776 cases were identified meeting the inclusion criteria, 15 of which experienced either a DVT or PE. A postoperative DVT occurred in 13 patients, and 2 patients had a postoperative PE. The most common procedures were for spine; the least common were for infection (Table 2). The procedure associated with the highest occurrence of VTE was operation for infection (1.2%).
Table 2.
Frequency of VTE Events per Orthopaedic Procedure Categorya
| Category | No. of Events | No. of Procedures |
% VTE |
||
|---|---|---|---|---|---|
| VTE | DVT | PE | |||
| Spine | 5 | 5 | 0 | 5,478 | 0.10 |
| Hip | 2 | 2 | 0 | 2,192 | 0.10 |
| Lower extremity soft tissue | 1 | 1 | 0 | 2,771 | 0.03 |
| Lower extremity fracture or osteotomy | 5 | 4 | 1 | 4,171 | 0.12 |
| Infection | 2 | 1 | 1 | 164 | 1.2 |
| Total | 15 | 13 | 2 | 14,776 | 0.10 |
Procedures were categorized by Current Procedural Terminology code. Data acquired from 2012 to 2013 from >50 participating National Surgical Quality Improvement Program, Pediatric database institutions.
DVT = deep vein thrombosis, PE = pulmonary embolism, VTE = venous thromboembolism
Patients who experienced a VTE had an average age of 12.0 ± 4.0 years (mean ± SD) compared with 11.3 ± 4.3 years for those who did not (P = 0.552). For the American Society of Anesthesiologists classification, 46.7% of patients who experienced a VTE had a classification of ≥3, compared with 25.5% of those who did not (P = 0.075). No difference was seen in age, gender, obesity, or race. Full demographic results are seen in Table 3.
Table 3.
Patient Demographic and Hospital Factor Association With VTEa
| Parameter | VTE | No VTE | P value |
|---|---|---|---|
|
Demographics Age (yr) |
12.0 ± 4.0 | 11.3 ± 4.3 | 0.552 |
| Male (%) | 40.0 | 47.8 | 0.612 |
| Female (%) | 60.0 | 52.2 | |
| Obese (%) | 20.0 | 23.9 | 1.000 |
| Racial minority (%) | 40.0 | 29.8 | 0.613 |
| ASA score ≥3 (%) | 46.7 | 25.5 | 0.075 |
| Hospital Factors | |||
| Length of stay (d) | 12.5 ± 16.3 | 3.2 ± 5.4 | <0.001 |
| Emergent/urgent case status (%) | 33.3 | 8.7 | 0.002 |
| Elective case status (%) | 66.7 | 91.3 | |
| Inpatient procedure (%) | 93.3 | 65.7 | 0.027 |
| Outpatient procedures (%) | 6.7 | 34.3 |
Statistical analysis was performed using the Fisher exact test or the independent sample Student’s t-test where appropriate. Continuous variables expressed as mean ± standard deviation of the mean.
ASA = American Society of Anesthesiologists, VTE = venous thromboembolism
Significant associations between VTE and comorbidities were seen for patients on oxygen support (13.3% versus 1.6%, P = 0.025), structural pulmonary abnormality (20.0% versus 4.4%, P = 0.026), GI comorbidity (33.0% versus 9.7%, P = 0.011), renal comorbidity (6.7% versus 0.1%, P = 0.016), and hematologic disorder (13.3% versus 1.4%, P = 0.019). Other comorbidities that demonstrated a trend toward association, but were not statistically significant, included pulmonary comorbidity and the need for nutritional support. Table 4 presents the relationship between VTE and comorbidities.
Table 4.
Patient Comorbidity Association With VTEa
| Parameter | VTE (%) |
No VTE (%) |
P value |
|---|---|---|---|
| Pulmonary comorbidity | 26.7 | 13.6 | 0.137 |
| Ventilator-dependent | 6.7 | 1.9 | 0.249 |
| Pneumonia | 0.0 | 0.0 | 1.000 |
| Asthma | 13.3 | 8.3 | 0.358 |
| Cystic fibrosis | 0.0 | 0.0 | 1.000 |
| Bronchopulmonary dysplasia | 6.7 | 2.9 | 0.353 |
| Oxygen support | 13.3 | 1.6 | 0.025b |
| Structural pulmonary abnormality | 20.0 | 4.4 | 0.026b |
| GI comorbidity | 33.3 | 9.7 | 0.011b |
| Esophageal/gastric/intestinal disease | 33.3 | 9.5 | 0.011b |
| Biliary/liver/pancreatic disease | 6.7 | 0.3 | 0.049b |
| Renal comorbidity | 6.7 | 0.1 | 0.016b |
| Renal failure | 0.0 | 0.0 | 1.000 |
| Dialysis | 6.7 | 0.1 | 0.015b |
| CNS comorbidity | 40.0 | 29.0 | 0.394 |
| Coma >24 h | 0.0 | 0.0 | 1.000 |
| History of CVA or TBI | 6.7 | 4.7 | 0.515 |
| CNS tumor | 0.0 | 0.0 | 1.000 |
| Developmental delay | 33.3 | 20.7 | 0.214 |
| Seizure disorder | 13.3 | 9.9 | 0.644 |
| Structural CNS abnormality | 6.7 | 13.1 | 0.709 |
| Cardiac comorbidity | 0.0 | 7.0 | 0.620 |
| Cerebral palsy or neuromuscular disorder | 33.3 | 25.1 | 0.550 |
| Chronic steroid use | 0.0 | 1.1 | 1.000 |
| Nutritional support | 20.0 | 6.8 | 0.079 |
| Bleeding disorder | 0.0 | 0.6 | 1.000 |
| Hematologic disorder | 13.3 | 1.4 | 0.019b |
| Preoperative blood transfusion | 0.0 | 0.9 | 1.000 |
| Inotropic support | 0.0 | 1.5 | 1.000 |
| Current or previous malignancy | 0.0 | 1.5 | 0.891 |
Statistical analysis was performed using the Fisher exact test.
P value <0.05.
CNS = central nervous system, CVA = cerebrovascular accident, GI = gastrointestinal, TBI = traumatic brain injury, VTE = venous thromboembolism
With respect to laboratory values, hyponatremia (20.0% versus 2.1%, P = 0.003), elevated AST levels (13.3% versus 0.6%, P = 0.004), and abnormal PTT (13.3% versus 2.3%, P = 0.046) were significantly associated with VTE; whereas hypoalbuminemia (6.7% versus 1.5%, P = 0.208) and thrombocytopenia (6.7% versus 2.6%, P = 0.324) were not. Preoperative laboratory values are shown in Table 5.
Table 5.
Abnormal Preoperative Laboratory Value Association With VTEa
| Parameter | VTE (%) | No VTE (%) | P value |
|---|---|---|---|
| Hypoalbuminemia | 6.7 | 1.5 | 0.208 |
| Hyponatremia | 20.0 | 2.1 | 0.003b |
| Elevated WBC count | 6.7 | 1.6 | 0.216 |
| Thrombocytopenia | 6.7 | 2.6 | 0.324 |
| Elevated AST level | 13.3 | 0.6 | 0.004b |
| Elevated BUN level | 13.3 | 3.8 | 0.110 |
| Abnormal PT | 0.0 | 2.2 | 1.000 |
| Abnormal PTT | 13.3 | 2.3 | 0.046b |
| Anemia | 0.0 | 1.8 | 1.000 |
Statistical analysis was performed using the Fisher exact test.
P value <0.05.
AST = aspartate transaminase, BUN = blood urea nitrogen, PT = prothrombin time, PTT = partial thromboplastin time, VTE = venous thromboembolism, WBC = white blood cell
Individual case summaries are shown in Table 6, including data on the number of days after surgery to the VTE event and events occurring postdischarge. Nearly half of DVT events (46.2%) occurred postdischarge, whereas no PE events occurred postdischarge. The mean number of days postoperatively on which DVT events occurred was 9.4 ± 8.6 days, whereas the mean number of days postoperatively on which PE events occurred was 3.5 ± 2.1 days. Inpatient procedure type was associated with VTE development (93.3% versus 65.7%, P = 0.027). Case status (emergent or urgent versus elective) was significantly associated with VTE; 33.3% of patients with VTE underwent emergent or urgent surgery compared with 8.7% of cases without VTE (P = 0.002).
Table 6.
Case Summaries of Patients Experiencing VTE
| Patient (DVT or PE) |
Procedure | Comorbidities | Abnormal Laboratory Values |
Days to VTE (Postoperative Days to Discharge) |
Complications |
|---|---|---|---|---|---|
| 14 y/o Caucasian male (DVT) |
Arthrotomy, hip, with drainage for infection |
DD | Hyponatremia, ↑ BUN, ↑ AST, thrombocytopenia |
1 (18) | None |
| 10 y/o minority female (DVT) |
Arthrodesis, posterior; 13 or more vertebral segments |
DD, seizure disorder, CP, nutritional support |
None | 29 (39) | Blood transfusion, pneumonia, UINT (×2) |
| 10 y/o minority female (DVT) |
Arthrodesis, posterior; 13 or more vertebral segments |
GI, CVA, DD, CP, nutritional support |
↑ WBC, ↑ BUN | 6 (Not available) | CLABSI, blood transfusion, sepsis, UTI |
| 10 y/o Caucasian female (DVT) |
Arthrodesis, posterior; 13 or more vertebral segments |
Asthma, oxygen support, structural pulmonary abnormality, GI comorbidity, DD, seizure disorder, CP, nutritional support |
None | 3 (7) | Blood transfusion |
| 13 y/o Caucasian female (DVT) |
Arthrodesis, posterior; 13 or more vertebral segments |
Obesity | None | 14 (0) | None |
| 16 y/o minority female (DVT) |
Osteotomy, femur, shaft or supracondylar; with fixation |
Bronchopulmonary dysplasia, CP |
PTT, hyponatremia, hypoalbuminemia |
6 (54) | CLABSI, transfusion, pneumonia, UINT, UTI |
| 17 y/o Caucasian female (DVT) |
Arthrodesis, posterior; 7 to 12 vertebral segments |
Structural pulmonary, GI, structural CNS, neuromuscular disorder |
None | 18 (6) | Blood transfusion |
| 8 y/o minority male (DVT) |
Hemiepiphyseal arrest, distal femur or proximal tibia or fibula |
Obesity, ventilator, asthma, oxygen support, structural pulmonary, GI, dialysis, DD, organ transplant, hematologic |
PT, ↑ AST | 0 (10) | None |
| 17 y/o Caucasian female (DVT) |
Reconstruction of dislocating patella |
Obesity, hematologic disorder |
None | 6 (2) | None |
| 9 y/o minority female (DVT) |
In situ pinning of slipped femoral epiphysis |
None | None | 11 (5) | None |
| 8 y/o Caucasian male (DVT) |
Open treatment of femoral shaft fracture |
None | None | 4 (0) | None |
| 10 y/o minority male (DVT) |
Open treatment of femoral shaft fracture |
None | None | 4 (2) | None |
| 3 y/o Caucasian male (DVT) |
Osteotomy, iliac, acetabular, or innominate bone |
GI | None | 20 (Not available) | Blood transfusion |
| 16 y/o Caucasian male (PE) |
Open treatment of femoral shaft fracture |
None | PTT | 2 (3) | None |
| 10 y/o Caucasian female (PE) |
Arthrotomy, hip, with drainage for infection |
Preoperative SIRS/sepsis |
Hyponatremia | 5 (12) | Pneumonia |
elevated
AST = aspartate transaminase level, BUN = blood urea nitrogen, CLABSI = central line-associated bloodstream infection, CP = cerebral palsy, CNS = central nervous system, CVA = cerebrovascular accident, DD = developmental delay, DVT = deep vein thrombosis, GI = gastrointestinal, PE = pulmonary embolism, PT = prothrombin time, PTT = partial thromboplastin time, SIRS = systemic inflammatory response syndrome, UINT = unplanned intubation, UTI = urinary tract infection, VTE = venous thromboembolism, WBC = white blood cell
An analysis of associated postoperative complications is shown in Table 7. Length of stay in patients with VTE was significantly longer than that in patients without VTE (12.5 ± 16.3 versus 3.2 ± 5.4 days, P = <0.001). Significant associations with VTE were seen for prolonged length of stay (P = <0.001), postoperative pneumonia (P = <0.001), unplanned intubation (P = 0.003), UTI (P = 0.003), and CLABSI (P = <0.001). Most of the complications (66.7%) significantly associated with VTE occurred before or were concurrent with the diagnosis of VTE. All postoperative pneumonia and intubation events occurred before corresponding VTE events. UTIs occurred on the same day and 12 days after VTE. CLABSI occurred after DVT in both instances.
Table 7.
Associated Postoperative Complications With VTEa
| Complication | VTE (%) | No VTE (%) | P value |
|---|---|---|---|
| Infection | |||
| Superficial SSI | 0.0 | 0.9 | 1.000 |
| Deep SSI | 0.0 | 0.5 | 1.000 |
| Organ space SSI | 0.0 | 0.2 | 1.000 |
| Postoperative pneumonia | 20.0 | 0.6 | <0.001b |
| Sepsis | 6.7 | 0.4 | 0.052 |
| CLABSI | 13.3 | 0.0 | <0.001b |
| UTI | 13.3 | 0.5 | 0.003b |
| Wound dehiscence | 0.0 | 0.5 | 1.000 |
| Unplanned intubation | 13.3 | 0.5 | 0.003b |
| Renal insufficiency | 0.0 | 0.0 | 1.000 |
| Acute renal failure | 0.0 | 0.0 | 1.000 |
| Coma | 0.0 | 0.0 | 1.000 |
| CVA | 0.0 | 0.0 | 1.000 |
| Seizure | 0.0 | 0.0 | 1.000 |
| Peripheral nerve injury | 0.0 | 0.2 | 1.000 |
| Cardiac arrest | 0.0 | 0.0 | 1.000 |
| Blood transfusion | 40.0 | 23.1 | 0.130 |
| Graft/prosthesis failure | 0.0 | 0.0 | 1.000 |
Statistical analysis was performed using the Fisher exact test
P value <0.05
CLABSI = central line-associated bloodstream infection, CVA = cerebrovascular accident, SSI = surgical site infection, UTI = urinary tract infection, VTE = venous thromboembolism
Discussion
VTE is a serious complication of surgery with well-documented adverse outcomes in the adult surgical population. However, its association with other complications in children is unclear. The purpose of this study was to use a high-quality, large, nationally representative database to determine the risk of VTE in the pediatric orthopaedic population and to identify other complications associated with its occurrence. The incidence was found to be 0.1% in this population. GI comorbidity, renal comorbidity, hematologic disorder, elevated AST level, abnormal PTT, and hyponatremia were patient factors associated with the development of VTE. Inpatient procedures and emergent or urgent surgery were more likely to result in VTE. Prolonged hospital stay, postoperative pneumonia, UTI, CLABSI, and unplanned intubation were complications associated with the development of VTE, with most complications occurring before or being concurrent with the diagnosis of VTE.
To our knowledge, this is the first study examining postoperative complications associated with VTE in the pediatric orthopaedic population. Despite the literature discussing the need to enumerate risks and benefits of thromboprophylaxis in children, few studies have investigated morbidity and mortality associated with VTE in children. We observed a substantially higher rate of infections (ie, pneumonia, UTI, and CLABSI) and unplanned intubation in patients with VTE. Most of these complications preceded or occurred concurrently with the diagnosis of VTE. These findings suggest that patients with a complicated course may need closer observation for, or prophylaxis for, VTE. In addition, those patients who have VTE will more likely have a longer length of stay, require use of a catheter or central line, and have a higher rate of preexisting comorbidities, thus increasing the risk of certain complications such as CLABSI (both instances of CLABSI in this cohort occurred after DVT events). However, no patients died in our patient cohort.
In a study of the pediatric orthopaedic population, Georgopoulos et al12 reported that the incidence of VTE in 1 in 1,000 patients differs greatly from the adult orthopaedic population and is nearly double the incidence reported in previous pediatric orthopaedic retrospective studies. However, the authors excluded pediatric orthopaedic patients with procedures or diagnoses related to trauma, infection, malignancy, or coagulopathies as well as patients admitted through the emergency department or whose orthopaedic procedure was not performed on the admission date, potentially contributing to differences in VTE rates between studies. An upward trend in pediatric postoperative VTE events from 2001 to 2007 has been reported and was attributed to improved recognition of previously undiagnosed DVT.2 It is unclear from our data at what age the risk of VTE approaches that of adult levels, but in a study of pediatric trauma patients conducted by Van Arendonk et al,4 the authors reported that the incidence of VTE increased from zero in patients younger than 12 years to 0.4% in those aged 13 to 15 years and to 0.8% in those aged 16 to 21 years. In adult total hip and knee arthroplasty, the rate of predischarge symptomatic VTE approaches 2% even after thromboprophylaxis, although many more VTEs occur postdischarge and are asymptomatic.17 In adult spine surgery, symptomatic VTE occurs in 1.5% of patients;18 this rate is □10 times the rate in our observed pediatric spine population (0.10%). Therefore, the low rate of occurrence in pediatric orthopaedic surgery patients, compared with adults, calls into question whether routine thromboprophylaxis should be indicated.
If thromboprophylaxis is indicated, limited data are available on the optimization of anticoagulant administration in children, with most recommendations extrapolated from the adult population.19 Similarly, the risks of anticoagulation in children are not completely understood; however, the frequency of major bleeding with the use of low-molecular-weight heparin in children has been reported to be as high as 5.6% and, with unfractionated heparin, as high as 18%.20,21 Although the mortality rate of VTE in adults is as high as 25%,22 rates may be as low as 2% in children.5,23 In our study , no patients with VTE or PE died, thus reinforcing the question whether the use of routine anticoagulation in a pediatric population without risk factors for VTE exposes those children to a greater probability of harm compared with a protocol of no anticoagulation prophylaxis. In a pediatric critical care study of 655 patients by Faustino et al,24 23.8% of those aged ≥8 years received mechanical prophylaxis; however, no data exist regarding the efficacy of mechanical prophylaxis in children, in part because of its very low rate of occurrence. Although the costs of VTE events in the pediatric population are similar to that of adults (ie, approximately $20,000 per episode),25,26 the cost of complications from anticoagulation remains unknown. We believe additional investigation is needed before endorsing recommendations for universal prophylactic anticoagulation for pediatric orthopaedic procedures.
Although the incidence of VTE in our study was low, certain patient characteristics emerged that may influence the surgeon’s decision to initiate anticoagulation. Renal, hematologic, and GI comorbidities were associated with VTE. Renal comorbidity, which included patients undergoing dialysis and those with renal failure, has previously been reported as a risk factor for catheter-associated venous thrombosis in children.27–29 GI comorbidities, which included esophageal/gastric/intestinal disease and biliary/liver/pancreatic disease, also increased the risk of VTE. Previously, only inflammatory bowel disease and short bowel syndrome were linked to VTE in children.27 An association with liver, biliary, and pancreatic disease is less clear, although elevated AST levels were found to be associated with VTE in this study. Hypercoagulable states have been observed in adults with liver disease;30 however, this effect has not been studied in children.
With regard to laboratory values, the cause for the association between VTE and hyponatremia in this cohort is unclear. To our knowledge, this is the first study showing preoperative hyponatremia as a risk factor for VTE in a pediatric population. Existing studies examining the link between the two conditions have come in different contexts. For example, hyponatremia has been shown to increase the risk for VTE in adults with non–small cell lung cancer.31 Hyponatremia has also been shown to be a predictor of a poor prognosis after PE.32 Postoperative pneumonia and other pulmonary disorders have also been shown to alter antidiuretic hormone levels, increasing the risk of hyponatremia;33 we observed a higher rate of pneumonia in patients experiencing VTE. Additionally, elevated AST levels were found to be a risk factor for VTE. Liver injury or disease resulting in elevated AST levels may lead to hypercoagulable states via increased clotting factor VIII or decreased protein C,30,34 although a direct association to VTE in pediatric populations remains unclear. Abnormal PTT was also found to be a risk factor for VTE in this study; this finding is not surprising because an abnormal PTT can indicate clotting dysfunction.35
Musculoskeletal infections have been shown to put patients at risk for thromboembolic complications.36,37 Our study included septic hip and knee conditions. Previous studies have observed DVT in children with septic arthritis.36 DVT has also been seen in the setting of osteomyelitis.37 Because the current evidence appears to be limited to case reports, the true rate of VTE in musculoskeletal infections is unknown, although a rate of 1.2% for septic arthritis of the hip or knee was observed in our study (compared with an overall VTE incidence of 0.10%). Furthermore, VTE may already be present at the time of surgical treatment of infection. Thus, screening and early recognition of VTE before surgery is of critical importance for children with septic arthritis, and thromboprophylaxis should be considered, especially for children in intensive care and in patients with markedly elevated C-reactive protein levels or methicillin-resistant Staphylococcus aureus.36,37
Several pertinent negatives that differ from previous literature were observed in this study. The first of these was obesity, which has been demonstrated to be a risk factor for VTE in both adult and pediatric populations.12,38 LE fracture and osteotomy procedures were also expected to be a risk for VTE based on previous literature, but no association was identified in this analysis.38 Older age did not emerge as a risk factor despite previous studies implicating older age as a risk factor for VTE in children.1–4 The reason for these negatives is unclear, but the rarity of VTE in the pediatric population may create type II error. Studies of future NSQIP Pediatric data may clarify these unexpected results.
Strengths of the study include a large number of patients from a reliable database. NSQIP has been shown to be highly consistent, with inter-rater disagreement rates of <2%.16 Hospitals using NSQIP are more likely to lower costs and meet better quality outcomes, and patient outcomes and readmission data have been shown to be highly consistent with hospital administrative and medical records.14,39,40 The use of the database also has some limitations. Patient comorbidities reported in the NSQIP Pediatric are limited to those predefined in the database. Although these comorbidities provide useful information, they are not sufficiently granular to distinguish between specific conditions (eg, Crohn disease versus ulcerative colitis, or Factor V Leiden thrombophilia versus antithrombin deficiency). Additional factors that have been shown to increase the risk for VTE, such as prolonged time in a cast, immobility, and a family history of clotting disorders, are also absent.38 Further, post-thrombotic syndrome, an important complication of VTE, cannot be identified by this database; therefore, the full impact of the VTE event is not known. This study also has the potential for sampling bias because participating hospitals have minimum case requirements to which they must adhere, but not every surgical case in the hospital is reported, and a minimum of only 80% of 30-day follow-up data are required.16 There is also the possibility for undiagnosed asymptomatic DVT or VTE occurring >30 days after surgery. Medications are also absent in the database, and these could potentially be confounding variables. It is unknown what percentage of patients in this cohort received anticoagulation therapy. Statistical analysis was limited by a very low number of VTE outcomes relative to the number of variables analyzed. Therefore, the variables associated with VTE in the bivariate analysis, although noteworthy, could not be evaluated as independent risk factors for VTE development.
Summary
Despite limitations, this study presents useful information for clinicians and policymakers. Patients with VTE should be monitored for infections and aggressive pulmonary support, which may be required to avoid unplanned intubation. These complications are more likely to be associated with mortality than the VTE event in pediatric populations. Thromboprophylaxis and VTE screening may be most appropriate in patients with GI or renal comorbidity; hematologic disorders; abnormal AST, PTT, or sodium laboratory values; and patients with musculoskeletal infection or those undergoing surgery for musculoskeletal infection. Overall, the frequency of VTE in the pediatric orthopaedic population is very low, and no evidence was found in support of routine thromboprophylaxis for patients without predisposing risk factors.
Footnotes
Dr. Ponce or an immediate family member is a member of a speakers’ bureau or has made paid presentations on behalf of Mitek and Tornier, serves as a paid consultant to Acumed and Tornier, has stock or stock options held in VIPAAR, and has received research or institutional support from Arthrex and Tornier. Dr. Gilbert or an immediate family member serves as a board member, owner, officer, or committee member of the American Academy of Orthopaedic Surgeons and the Pediatric Orthopaedic Society of North America. None of the following authors or any immediate family member has received anything of value from or has stock or stock options held in a commercial company or institution related directly or indirectly to the subject of this article: Mr. Baker, Mr. Sherrod, and Dr. McGwin,
The American College of Surgeons National Surgical Quality Improvement Program and the hospitals participating in the ACS NSQIP are the source of the data used herein; they have not verified and are not responsible for the statistical validity of the data analysis or the conclusions derived by the authors. Complications and 30-day Outcomes Associated With Venous Thromboembolism in the Pediatric Orthopaedic Surgical Population
References
Evidence-based Medicine: Levels of evidence are described in the table of contents. In this article, reference 20 is a level I study. References 6, 17, 24, and 25 are level II studies. References 2, 4, 5, 7–9, 11–13, 18, 23, 26–29, 31, 32, 34, 37, and 39 are level III studies. References 33 and 36 are level IV studies. Reference 14 is level V expert opinion.
References printed in bold type are those published within the past 5 years.
- 1.White RH. The epidemiology of venous thromboembolism. Circulation. 2003;107(23, Suppl 1):I4–I8. doi: 10.1161/01.CIR.0000078468.11849.66. [DOI] [PubMed] [Google Scholar]
- 2.Raffini L, Huang YS, Witmer C, Feudtner C. Dramatic increase in venous thromboembolism in children’s hospitals in the United States from 2001 to 2007. Pediatrics. 2009;124(4):1001–1008. doi: 10.1542/peds.2009-0768. [DOI] [PubMed] [Google Scholar]
- 3.Parasuraman S, Goldhaber SZ. Venous thromboembolism in children. Circulation. 2006;113(2):e12–e16. doi: 10.1161/CIRCULATIONAHA.105.583773. [DOI] [PubMed] [Google Scholar]
- 4.Van Arendonk KJ, Schneider EB, Haider AH, Colombani PM, Stewart FD, Haut ER. Venous thromboembolism after trauma: When do children become adults? JAMA Surg. 2013;148(12):1123–1130. doi: 10.1001/jamasurg.2013.3558. [DOI] [PubMed] [Google Scholar]
- 5.Setty BA, O’Brien SH, Kerlin BA. Pediatric venous thromboembolism in the United States: A tertiary care complication of chronic diseases. Pediatr Blood Cancer. 2012;59(2):258–264. doi: 10.1002/pbc.23388. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Hanson SJ, Punzalan RC, Arca MJ, et al. Effectiveness of clinical guidelines for deep vein thrombosis prophylaxis in reducing the incidence of venous thromboembolism in critically ill children after trauma. J Trauma Acute Care Surg. 2012;72(5):1292–1297. doi: 10.1097/TA.0b013e31824964d1. [DOI] [PubMed] [Google Scholar]
- 7.Hanson SJ, Punzalan RC, Greenup RA, Liu H, Sato TT, Havens PL. Incidence and risk factors for venous thromboembolism in critically ill children after trauma. J Trauma. 2010;68(1):52–56. doi: 10.1097/TA.0b013e3181a74652. [DOI] [PubMed] [Google Scholar]
- 8.O’Brien SH, Candrilli SD. In the absence of a central venous catheter, risk of venous thromboembolism is low in critically injured children, adolescents, and young adults: Evidence from the National Trauma Data Bank. Pediatr Crit Care Med. 2011;12(3):251–256. doi: 10.1097/PCC.0b013e3181f36bd9. [DOI] [PubMed] [Google Scholar]
- 9.Vu LT, Nobuhara KK, Lee H, Farmer DL. Determination of risk factors for deep venous thrombosis in hospitalized children. J Pediatr Surg. 2008;43(6):1095–1099. doi: 10.1016/j.jpedsurg.2008.02.036. [DOI] [PubMed] [Google Scholar]
- 10.Monagle P, Chan AK, Goldenberg NA, et al. American College of Chest Physicians: Antithrombotic therapy in neonates and children: Antithrombotic Therapy and Prevention of Thrombosis, 9th ed: American College of Chest Physicians Evidence-Based Clinical Practice Guidelines. Chest. 2012;141(2, Suppl):e737S–e801S. doi: 10.1378/chest.11-2308. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Jain A, Karas DJ, Skolasky RL, Sponseller PD. Thromboembolic complications in children after spinal fusion surgery. Spine (Phila Pa 1976) 2014;39(16):1325–1329. doi: 10.1097/BRS.0000000000000402. [DOI] [PubMed] [Google Scholar]
- 12.Georgopoulos G, Hotchkiss MS, McNair B, Siparsky G, Carry PM, Miller NH. Incidence of deep vein thrombosis and pulmonary embolism in the elective pediatric orthopaedic patient. J Pediatr Orthop. 2016;36(1):101–109. doi: 10.1097/BPO.0000000000000391. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Weiss A, Anderson JE, Chang DC. Comparing the National Surgical Quality Improvement Program With the Nationwide Inpatient Sample Database. JAMA Surg. 2015;150(8):815–816. doi: 10.1001/jamasurg.2015.0962. [DOI] [PubMed] [Google Scholar]
- 14.Sabharwal S, Passannante MR. Venous thromboembolism in children: Preliminary results of a survey of POSNA members. J Pediatr Orthop. 2013;33(8):852–856. doi: 10.1097/BPO.0b013e3182a35c7e. [DOI] [PubMed] [Google Scholar]
- 15.Centers for Medicare and Medicaid Services: Evidence-based guidelines for selected, candidate, and previously considered hospital-acquired conditions. Woodlawn, MD: [Accessed July 6, 2015]. Published May 2014. Available at: http://www.cms.gov/Medicare/Medicare-Fee-for-Service-Payment/HospitalAcqCond/Downloads/Evidence-Based-Guidelines.pdf. [Google Scholar]
- 16.American College of Surgeons: User Guide for the 2013 ACS NSQIP Pediatric Participant Use Data File. Chicago, IL: [Accessed April 27, 2015]. Published November 2014. Available at: https://www.facs.org/~/media/files/quality%20programs/nsqip/peds_puf_userguide_2013.ashx. [Google Scholar]
- 17.Januel JM, Chen G, Ruffieux C, et al. IMECCHI Group: Symptomatic in-hospital deep vein thrombosis and pulmonary embolism following hip and knee arthroplasty among patients receiving recommended prophylaxis: A systematic review. JAMA. 2012;307(3):294–303. doi: 10.1001/jama.2011.2029. [DOI] [PubMed] [Google Scholar]
- 18.Tominaga H, Setoguchi T, Tanabe F, et al. Risk factors for venous thromboembolism after spine surgery. Medicine (Baltimore) 2015;94(5):e466. doi: 10.1097/MD.0000000000000466. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Law C, Raffini L. A guide to the use of anticoagulant drugs in children. Paediatr Drugs. 2015;17(2):105–114. doi: 10.1007/s40272-015-0120-x. [DOI] [PubMed] [Google Scholar]
- 20.Massicotte P, Julian JA, Gent M, et al. REVIVE Study Group: An open-label randomized controlled trial of low molecular weight heparin compared to heparin and coumadin for the treatment of venous thromboembolic events in children: The REVIVE trial. Thromb Res. 2003;109(2–3):85–92. doi: 10.1016/s0049-3848(03)00059-8. [DOI] [PubMed] [Google Scholar]
- 21.Revel-Vilk S, Chan AK. Anticoagulation therapy in children. Semin Thromb Hemost. 2003;29(4):425–432. doi: 10.1055/s-2003-42592. [DOI] [PubMed] [Google Scholar]
- 22.Heit JA The epidemiology of venous thromboembolism in the community. Implications for prevention and management. J Thromb Thrombolysis. 2006;21(1):23–29. doi: 10.1007/s11239-006-5572-y. [DOI] [PubMed] [Google Scholar]
- 23.Monagle P, Adams M, Mahoney M, et al. Outcome of pediatric thromboembolic disease: A report from the Canadian Childhood Thrombophilia Registry. Pediatr Res. 2000;47(6):763–766. doi: 10.1203/00006450-200006000-00013. [DOI] [PubMed] [Google Scholar]
- 24.Faustino EV, Hanson S, Spinella PC, et al. PROphylaxis against ThRombosis prACTice (PROTRACT) Study Investigators of the PALISI BloodNet: A multinational study of thromboprophylaxis practice in critically ill children. Crit Care Med. 2014;42(5):1232–1240. doi: 10.1097/CCM.0000000000000147. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Mahan CE, Holdsworth MT, Welch SM, Borrego M, Spyropoulos AC. Deep-vein thrombosis: A United States cost model for a preventable and costly adverse event. Thromb Haemost. 2011;106(3):405–415. doi: 10.1160/TH11-02-0132. [DOI] [PubMed] [Google Scholar]
- 26.Kerlin BA. Current and future management of pediatric venous thromboembolism. Am J Hematol. 2012;87(Suppl 1):S68–S74. doi: 10.1002/ajh.23131. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Smitherman AB, Alexander T, Connelly M, et al. The incidence of catheter-associated venous thrombosis in noncritically ill children. Hosp Pediatr. 2015;5(2):59–66. doi: 10.1542/hpeds.2014-0041. [DOI] [PubMed] [Google Scholar]
- 28.Takemoto CM, Sohi S, Desai K, et al. Hospital-associated venous thromboembolism in children: Incidence and clinical characteristics. J Pediatr. 2014;164(2):332–338. doi: 10.1016/j.jpeds.2013.10.025. [DOI] [PubMed] [Google Scholar]
- 29.Kerlin BA, Smoyer WE, Tsai J, Boulet SL. Healthcare burden of venous thromboembolism in childhood chronic renal diseases. Pediatr Nephrol. 2015;30(5):829–837. doi: 10.1007/s00467-014-3008-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Lisman T, Caldwell SH, Burroughs AK, et al. Coagulation in Liver Disease Study Group: Hemostasis and thrombosis in patients with liver disease: The ups and downs. J Hepatol. 2010;53(2):362–371. doi: 10.1016/j.jhep.2010.01.042. [DOI] [PubMed] [Google Scholar]
- 31.Wang Z, Yan HH, Yang JJ, et al. Venous thromboembolism risk factors in Chinese non-small cell lung cancer patients. Support Care Cancer. 2015;23(3):635–641. doi: 10.1007/s00520-014-2405-y. [DOI] [PubMed] [Google Scholar]
- 32.Korkmaz I, Güven FM, Eren SH, et al. Baseline characteristics and the association between hyponatraemia and pulmonary embolism prognosis. J Pak Med Assoc. 2013;63(3):331–335. [PubMed] [Google Scholar]
- 33.Skippen P, Adderley R, Bennett M, et al. Iatrogenic hyponatremia in hospitalized children: Can it be avoided? Paediatr Child Health. 2008;13(6):502–506. doi: 10.1093/pch/13.6.502. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Tripodi A, Primignani M, Chantarangkul V, et al. An imbalance of pro- vs anti-coagulation factors in plasma from patients with cirrhosis. Gastroenterology. 2009;137(6):2105–2111. doi: 10.1053/j.gastro.2009.08.045. [DOI] [PubMed] [Google Scholar]
- 35.Ng VL. Prothrombin time and partial thromboplastin time assay considerations. Clin Lab Med. 2009;29(2):253–263. doi: 10.1016/j.cll.2009.05.002. [DOI] [PubMed] [Google Scholar]
- 36.Walsh S, Phillips F. Deep vein thrombosis associated with pediatric musculoskeletal sepsis. J Pediatr Orthop. 2002;22(3):329–332. [PubMed] [Google Scholar]
- 37.Hollmig ST, Copley LA, Browne RH, Grande LM, Wilson PL. Deep venous thrombosis associated with osteomyelitis in children. J Bone Joint Surg Am. 2007;89(7):1517–1523. doi: 10.2106/JBJS.F.01102. [DOI] [PubMed] [Google Scholar]
- 38.Buesing KL, Mullapudi B, Flowers KA. Deep venous thrombosis and venous thromboembolism prophylaxis. Surg Clin North Am. 2015;95(2):285–300. doi: 10.1016/j.suc.2014.11.005. [DOI] [PubMed] [Google Scholar]
- 39.Sellers MM, Merkow RP, Halverson A, et al. Validation of new readmission data in the American College of Surgeons National Surgical Quality Improvement Program. J Am Coll Surg. 2013;216(3):420–427. doi: 10.1016/j.jamcollsurg.2012.11.013. [DOI] [PubMed] [Google Scholar]
- 40.Brighton BK. National Surgical Quality Improvement Program-pediatric (NSQIP) and the quality of surgical care in pediatric orthopaedics. J Pediatr Orthop. 2015;35(5, Suppl 1):S48–S50. doi: 10.1097/BPO.0000000000000548. [DOI] [PubMed] [Google Scholar]