Significance
Studies in transfected cells have established that G protein-coupled receptors (GPCRs) activate a number of intracellular signaling pathways; however, which of these pathways are physiologically important is unclear. Here, we use a genetically engineered mouse to demonstrate a novel role for M3-muscarinic acetylcholine receptor (M3-mAChR) phosphorylation in airway constriction, with implications for human respiratory disease, including asthma and chronic obstructive pulmonary disease. Combining this finding with other M3-mAChR physiological responses, we generate a map of responses that are downstream of G protein-dependent signaling or receptor phosphorylation-dependent signaling. Such a map predicts the outcome of biased GPCR drugs designed to drive receptor signaling preferentially toward pathways that improve therapeutic efficacy while minimizing toxic/adverse outcomes and provides a fundamental approach to the rational design of next-generation GPCR-based therapies.
Keywords: G protein-coupled receptor, asthma, muscarinic, ligand bias, signaling
Abstract
G protein-coupled receptors (GPCRs) are known to initiate a plethora of signaling pathways in vitro. However, it is unclear which of these pathways are engaged to mediate physiological responses. Here, we examine the distinct roles of Gq/11-dependent signaling and receptor phosphorylation-dependent signaling in bronchial airway contraction and lung function regulated through the M3-muscarinic acetylcholine receptor (M3-mAChR). By using a genetically engineered mouse expressing a G protein-biased M3-mAChR mutant, we reveal the first evidence, to our knowledge, of a role for M3-mAChR phosphorylation in bronchial smooth muscle contraction in health and in a disease state with relevance to human asthma. Furthermore, this mouse model can be used to distinguish the physiological responses that are regulated by M3-mAChR phosphorylation (which include control of lung function) from those responses that are downstream of G protein signaling. In this way, we present an approach by which to predict the physiological/therapeutic outcome of M3-mAChR–biased ligands with important implications for drug discovery.
Airflow in the lung is limited by vagally derived ACh that mediates airway smooth muscle (ASM) contraction primarily via activation of M3-muscarinic acetylcholine receptor (M3-mAChRs) (1). In both asthma and chronic obstructive pulmonary disease, this activity can be up-regulated, leading to bronchoconstriction and impaired lung function (1), thus explaining the therapeutic benefits provided by anticholinergics, such as tiotropium bromide (2).
M3-mAChRs can mediate ASM contraction through both calcium-dependent and calcium-independent mechanisms (1). The calcium-dependent mechanism is centered on Gq/11-mediated phospholipase C activation generating inositol 1,4,5-trisphosphate, which mobilizes calcium from the sarcoplasmic reticulum together with promoting extracellular calcium entry that ultimately results in a rise in intracellular calcium, activation of myosin-light chain kinase, and the phosphorylation of myosin leading to contraction (3). In contrast, the mechanism by which the M3-mAChR mediates calcium-independent ASM contraction is via activation of the small GTPase RhoA, which, in turn, activates Rho-kinase. The phosphorylation of myosin light chain (MLC) phosphatase by Rho-kinase decreases enzymatic activity, which results in higher levels of phosphorylated myosin and smooth muscle contraction (3–5). Although G protein-coupled receptor (GPCR) activation of RhoA is generally considered to be via Gq/11 and G12/13 heterotrimeric G proteins (6), the mechanism by which the M3-mAChR activates the RhoA pathway is unclear, with some researchers indicating that activation might proceed in a G protein-independent manner and possibly involve a direct interaction between the receptor and RhoA (7). The prominence of the calcium-independent pathway in the excessive airway narrowing (i.e., bronchial hyperresponsiveness) previously observed in animal models of allergic bronchial asthma has prompted the suggestion that inhibiting the M3-mAChR pathways leading to RhoA activation might be of therapeutic potential (8, 9).
GPCRs become rapidly phosphorylated following agonist stimulation, and this phosphorylation results in the recruitment of adaptor arrestin proteins that (i) uncouple receptors from their cognate G proteins and (ii) act as signaling scaffolds to drive G protein-independent signaling (10). The activation of RhoA signaling has been identified as one of those pathways regulated by receptor phosphorylation/arrestin-dependent signaling (11, 12). Because we have shown previously that the M3-mAChR is multiply phosphorylated following agonist stimulation (13) and that removal of the phosphorylation sites on the receptor results in the uncoupling of the M3-mAChR from arrestin, as well as arrestin-dependent processes such as receptor internalization (14, 15), we wanted to determine if M3-mAChR phosphorylation might play a role in calcium-independent ASM contraction and regulate lung function and airway hyperresponsiveness through this mechanism.
In addition, we aimed to establish an experimental system whereby we might distinguish between GPCR-mediated physiological responses that are downstream of G protein-dependent signaling and those responses that are downstream of receptor phosphorylation/arrestin-dependent signaling processes (16, 17). In this way, we might develop an approach by which to predict the physiological action of biased GPCR ligands, with important implications for drug discovery.
To achieve these dual aims, we use here a mouse model in which the M3-mAChR gene locus has been targeted to express a form of the M3-mAChR that is mutated in its phosphorylation sites (14, 15). This mutant receptor is uncoupled from phosphorylation-dependent pathways, including the recruitment of arrestin (14), but maintains coupling to Gq/11-dependent signaling (14, 15). In this regard, this mutant receptor can be considered as G protein-biased. Using this model, we determined a role for M3-mAChR phosphorylation in the regulation of lung function and in driving allergen-induced airway hyperresponsiveness. In addition, we were able to include this analysis in a map of M3-mAChR physiological responses that lay downstream of receptor phosphorylation/arrestin-dependent signaling and, in this way, provide a predictive model for the action of biased ligands to the M3-mAChR.
Results
Phosphorylation-Deficient M3-mAChR Mutant Is Expressed in ASM and Coupled Normally to Gq Signaling.
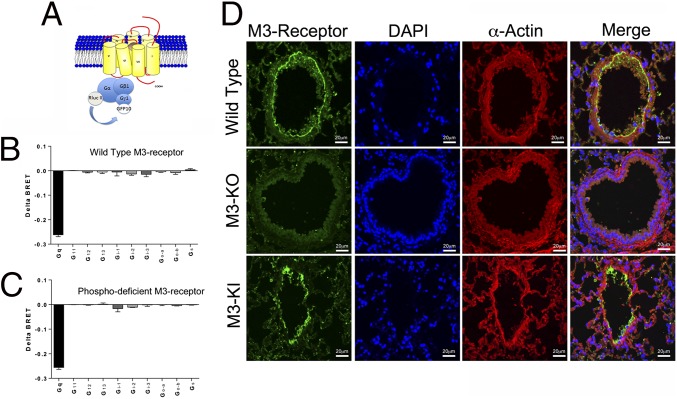
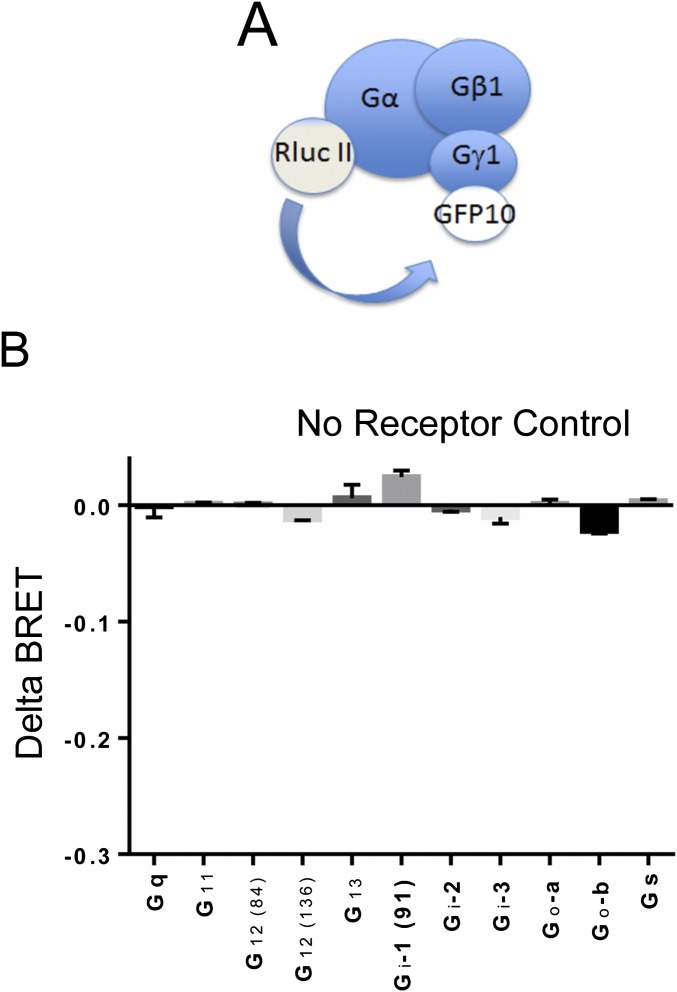
We have previously established that a mutant of the M3-mAChR, where 15 Ser phosphoacceptor sites in the third intracellular loop were mutated to Ala, showed reduced levels of agonist-mediated phosphorylation and significantly attenuated coupling to arrestin and arrestin-dependent signaling while maintaining near-normal coupling to Gq/11-dependent signaling (14, 15). Here, we present further evidence that G protein coupling of the phosphodeficient M3-mAChR mutant is normal by evaluating the coupling of both WT and mutant receptors to 10 different Gα-subunits in a bioluminescence resonance energy transfer (BRET)-based live cell assay (18, 19) (Fig. 1A). These experiments demonstrated that the profile of G protein activation between the WT M3-mAChR and the phosphodeficient mutant was very similar, with both receptor types predominantly coupling to Gq (Fig. 1 B and C). Importantly, there appeared to be no significant coupling of the M3-mAChR to G12/13 proteins (Fig. 1 B and C). Control experiments in which the receptor was not transfected showed no significant activation of G proteins, including Gq (Fig. S1 A and B).
Fig. 1.
Characterization of M3-mAChR expression and G protein coupling in the airways of WT and M3-KI mice. (A) Illustration of the BRET-based biosensor to measure G protein coupling, where various donor Rluc II Gα-subunits can be transfected with an acceptor GFP10-Gγ1 subunit together with the M3-mAChR. (B and C) Cells were transfected with the BRET biosensor consisting of various Rluc Gα-subunits. The change in BRET signal following receptor stimulation with carbachol (100 μM) in cells expressing the WT (B) or phosphodeficient M3-mAChR (C) is shown. Data represent the mean ± SEM of three independent experiments. (D) M3-mAChR (green) and smooth muscle α-actin immunoreactivity (red) in the airways of WT, M3-KO, and M3-KI mice. DAPI (blue) was used to stain nuclei. (Right) Overlaid images are shown.
Fig. S1.
Control for G protein BRET biosensor. (A) Illustration of the BRET-based biosensor to measure G protein coupling where various donor Rluc II Gα-subunits can be transfected together with an acceptor GFP10-Gγ1 subunit. The illustration is of the biosensor transfected without the receptor as a control. (B) Change in BRET signal following stimulation with CCh (100 μM) in cells transfected with the BRET biosensor consisting of various Rluc Gα-subunits without cotransfection with the M3-mAChR. Data represent the mean ± SEM of three independent experiments.
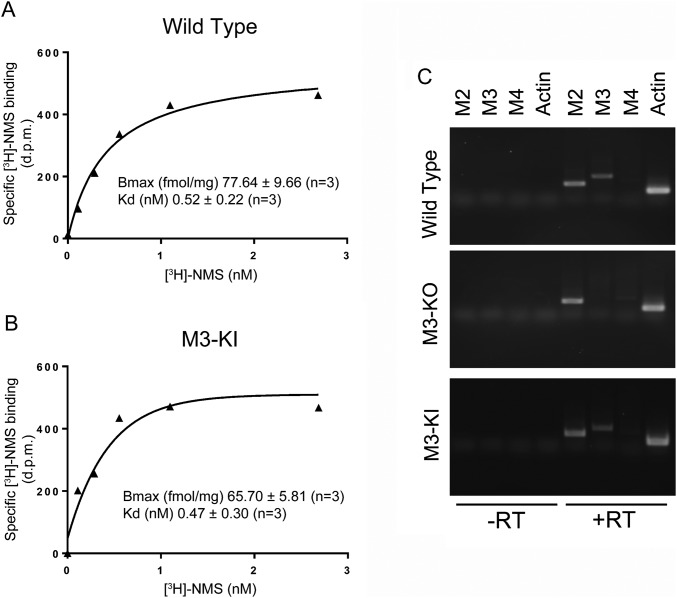
A mutant mouse strain, termed M3-knockin (M3-KI), was generated, where the M3-mAChR gene locus was modified to express the phosphodeficient M3-mAChR mutant in place of the WT receptor (14, 15). The muscarinic receptor expression levels in lungs from M3-KI mice (determined by radioligand binding) were not significantly different from the muscarinic receptor expression levels in lungs from WT mice (Fig. S2 A and B). Furthermore, the levels of the mutant receptor transcript in the lungs from M3-KI mice were similar to the levels of the WT receptor in C57BL/6J mice as determined by RT-PCR (Fig. S2C). Using an in-house Ab to the M3-mAChR, we found a normal expression profile of the mutant M3-mAChR in ASM of M3-KI mice, as indicated by colocalization of immunostaining of the receptor and α-actin in airways (Fig. 1E).
Fig. S2.
mAChR expression is unaltered in the lung of M3-KI mice relative to WT. Saturation binding of the muscarinic receptor antagonist [3H]-NMS to membranes prepared from the lung of WT (A) or M3-KI (B) mice. Membranes were incubated with increasing concentrations of [3H]-NMS in the absence or presence of 10 μM atropine to generate values for specific [3H]-NMS binding. Single representative experiments are shown, with similar data being obtained on two additional occasions (mean Bmax and Kd values are also shown). (C) RT-PCR showing the expression profile of M2-, M3- and M4-mAChR RNA in the lungs of WT, M3-KO, and M3-KI mice. Expression of actin RNA and omission of reverse transcriptase (RT) were used as a control.
M3-mAChR Mediates ASM Contraction in a Receptor Phosphorylation-Dependent Manner.
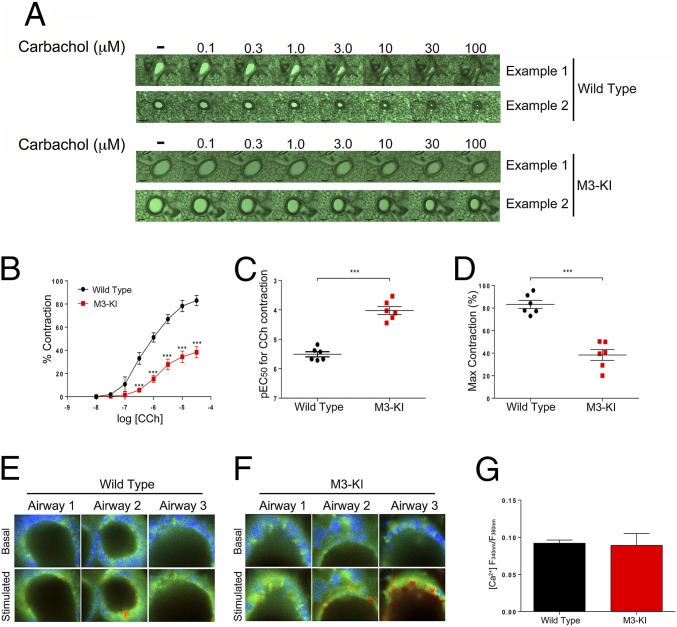
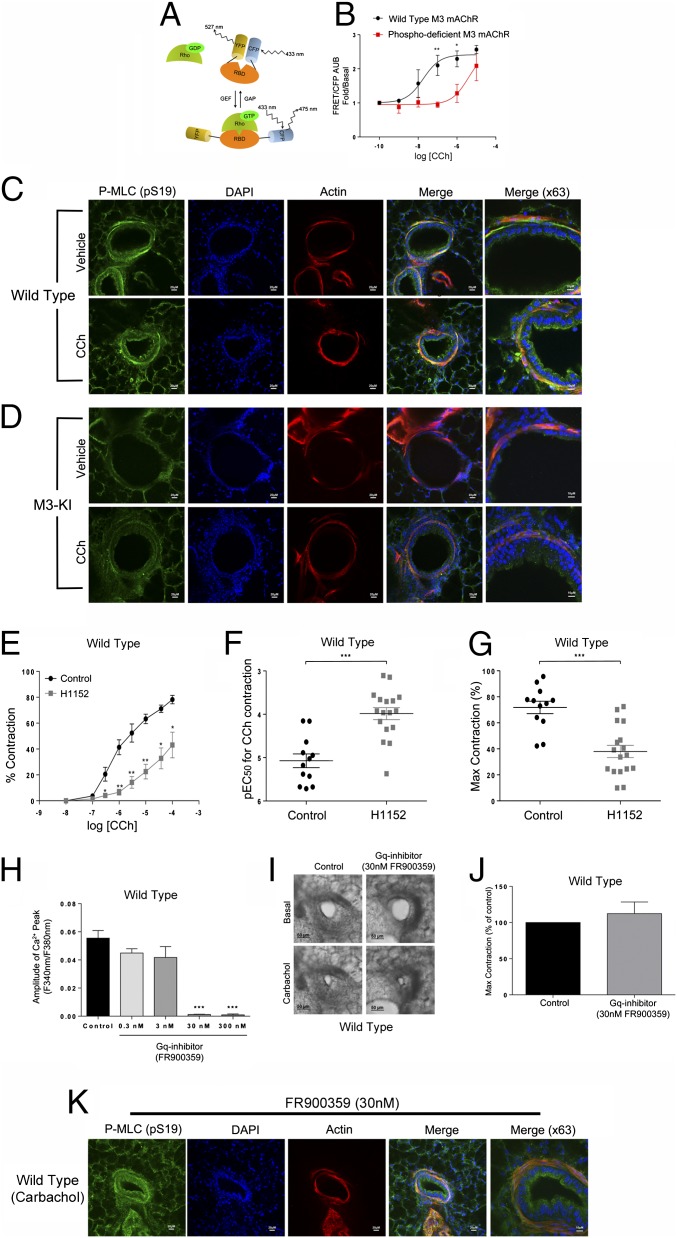
To determine the impact of mutating phosphorylation sites on the M3-mAChR in the lung, we used precision cut lung slices (PCLS) to monitor bronchoconstriction (20). In agreement with previous reports (21), we showed that muscarinic receptor stimulation of PCLS from WT mice resulted in a concentration-dependent narrowing of the airways [half-maximum effective concentration (pEC50) = 5.47 ± 0.06, maximum drug effect (Emax) = 82.47 ± 5.82] (Fig. 2 A–D). In comparison, the responsiveness of PCLS from M3-KI mice was significantly reduced in both maximal response and potency (pEC50 = 4.01 ± 0.06, Emax = 40.79 ± 5.73) (Fig. 2 A–D and Movies S1 and S2.). Importantly, the coupling of the phosphodeficient M3-mAChR mutant to Gq/calcium signaling in PCLS derived from M3-KI mice was seen to be very similar to the coupling observed in WT PCLS cultures (Fig. 2 E–G). Thus, despite normal coupling to Gq/calcium mobilization, the mutant receptor expressed in M3-KI mouse lung showed reduced airway contraction, indicating an important role for receptor phosphorylation-dependent signaling in M3-mAChR–mediated ASM contraction.
Fig. 2.
M3-mAChR–mediated ASM contraction is dependent on receptor phosphorylation-dependent signaling. (A) Four representative experiments, two from WT controls and two from M3-KI mice, showing the bronchoconstriction responses to increasing concentrations of carbachol in PCLS. (B–D) Contractile responses in PCLS derived from WT and M3-KI mice. (B) Mean concentration–response curves to carbachol (CCh) in WT and M3-KI PCLS. Mean pEC50 (C) and mean Emax (D) values calculated from the above PCLS concentration–response curves (n = 6 in each group). (E and F) In PCLS from WT and M3-KI mice, calcium responses following ACh (100 μM) stimulation were evaluated. Shown are three representative airway responses from PCLS derived from WT mice (E) and three representative airway responses from PCLS derived from M3-KI mice (F). (G) Average (±SEM) peak calcium responses from representative airways shown in E and F (all responsive single cells from each airway were included in the analysis). Data show the mean ± SEM and were analyzed using an unpaired t test (***P < 0.001).
Because previous studies had established that the RhoA pathway mediates calcium-independent ASM contraction (3–5), we investigated the role of RhoA signaling downstream of M3-mAChR phosphorylation. In vitro analysis of RhoA activation using a FRET-based biosensor (Fig. 3A) revealed that the phosphodeficient M3-mAChR mutant coupled with >200-fold lower potency to the RhoA pathway (pEC50 = 5.41 ± 0.63) than the WT receptor (pEC50 = 7.74 ± 0.17) (Fig. 3B). Activation of the RhoA pathway ultimately results in the phosphorylation of MLC and smooth muscle contraction (3–5). Staining for phosphorylation of MLC on Ser-19 showed pronounced MLC phosphorylation in the ASM in PCLS from WT animals in both the basal state and following carbachol stimulation (Fig. 3C). In contrast, ASM in PCLS from M3-KI mice showed substantially less MLC phosphorylation (Fig. 3D), a result consistent with reduced activation of RhoA signaling in M3-KI airways. Furthermore, the RhoA-kinase inhibitor H1152 reduced the maximal M3-mAChR contractile response in PCLS from WT mice by 43% and the potency of ACh-mediated contraction by >10-fold (pEC50 was reduced from 5.07 ± 0.15 to 3.98 ± 0.13 following H1152 treatment) (Fig. 3 E–G). These data indicate that a significant component of the M3-mAChR–mediated ASM contractile response was independent of Gq/calcium signaling but was dependent on M3-mAChR phosphorylation and the activation of the RhoA pathway.
Fig. 3.
M3-mAChR–mediated ASM contraction is dependent on receptor phosphorylation-dependent coupling to RhoA signaling. (A) Illustration of the FRET-based biosensor used to detect activated RhoA (GTP-bound). (B) Rho activity in CHO cells expressing the WT or the phosphodeficient M3-mAChR mutant in response to CCh. Data represent the mean ± SEM (n = 4). Data were analyzed using two-way ANOVA (*P < 0.05; **P < 0.01). Phospho-MLC2 (Ser-19, green) and smooth muscle α-actin (red) immunoreactivity is shown in the airways of WT (C) or M3-KI (D) mice stimulated with vehicle or CCh (100 μM,10 min). DAPI (blue) was used to stain nuclei. (Right) Overlaid images and images of the same airway using a 63× objective are shown. (E–G) Effect of Rho-kinase inhibitor H1152 on contractile responses in WT PCLS. (E) Mean concentration–response curves to CCh in WT PCLS pretreated with vehicle or H1152 (100 nM, 45 min of preincubation). Mean pEC50 (F) and mean Emax (G) values calculated from the above PCLS CCh concentration–response curves in the presence and absence of H1152 (n = 12 in vehicle-treated PCLS and n = 17 in H1152-treated PCLS) are shown. Data in E–G were analyzed using an unpaired t test (***P < 0.001). (H–J) Effects of the Gq inhibitor FR900359 on CCh (100 μM)-stimulated calcium responses and bronchoconstriction in PCLS from WT mice. (H) Average (±SEM) peak calcium responses stimulated by CCh after preincubation with increasing concentrations of FR900359 (0.3–300 nM, 30 min). (I) Representative examples of CCh-induced bronchoconstriction in PCLS from WT mice under control conditions (Left) or following preincubation with 30 nM FR900359 (Right). (J) Mean contractile response to CCh in WT PCLS following preincubation with FR900359 (30 nM, 30 min), expressed as a percentage of CCh-stimulated maximum contraction in control slices. Max, Maximum. (K) Phospho-MLC2 (Ser-19, green) and smooth muscle α-actin (red) immunoreactivity in PCLS from WT mice stimulated with CCh (100 μM, 10 min) after preincubation with 30 nM FR900359. DAPI (blue) was used to stain nuclei. (Right) Overlaid image and an image of the same airway using a 63× objective are shown.
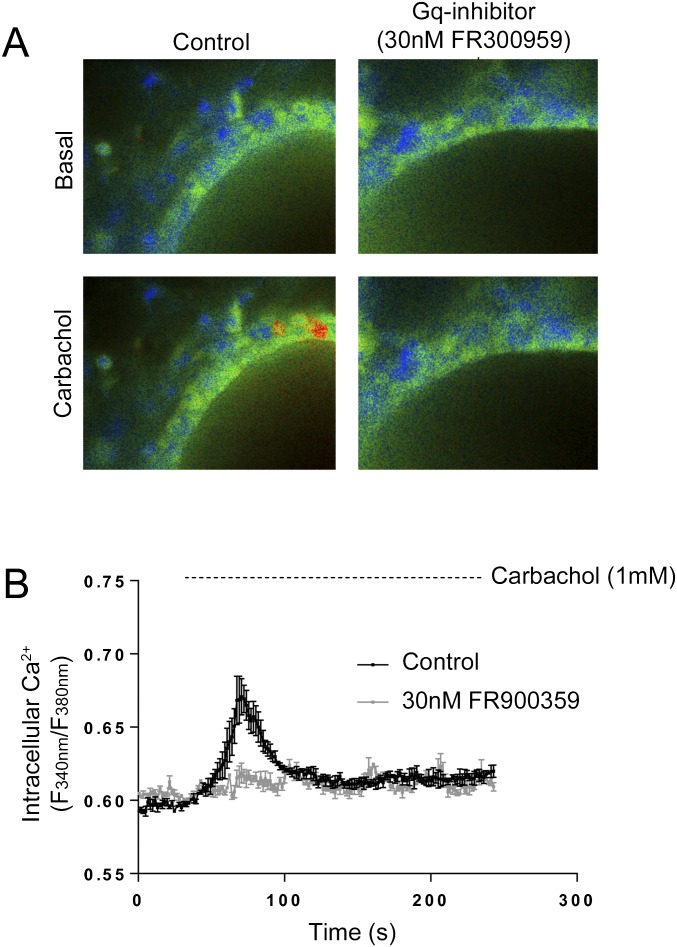
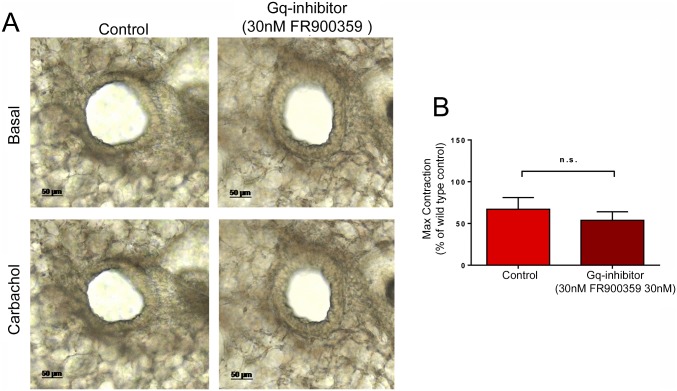
Consistent with this notion was the fact that inhibition of M3-mAChR–mediated Gq/calcium mobilization in PCLS from WT mice using the recently characterized Gq inhibitor FR900359 (22) (Fig. 3H and Fig. S3 A and B) did not affect the M3-mAChR–mediated contractile response (Fig. 3 H and J). The concentration of FR900359 used in these experiments (30 nM) completely prevented the carbachol-mediated calcium response in PCLS (Fig. 3H) but had no significant effect on carbachol-mediated smooth muscle contraction (Fig. 3 I and J). Importantly, inhibition of Gq/calcium signaling with FR900359 did not significantly affect the small contractile response seen in PCLS from M3-KI mice in response to carbachol, (Fig. S4 A and B) and FR900359 did not appear to affect the level of MLC phosphorylation in PCLS from WT mice stimulated with carbachol (Fig. 3K). These data point to RhoA-mediated signaling downstream of receptor phosphorylation having a prominent role in M3 receptor-mediated ASM contraction.
Fig. S3.
Inhibition of Gq abolishes the CCh-stimulated calcium mobilization in airways. (A) Representative examples of CCh (100 μM)-stimulated calcium release in PCLS from WT mice under control conditions or following preincubation with FR300959 (30 nM, 30 min) are shown. Images are 40× magnification. (B) Calcium response following carbachol (1 mM) stimulation under control conditions or following preincubation with FR900359 (30 nM, 30 min). The data are generated by analysis of fluorescent changes in 3–5 regions of interest.
Fig. S4.
FR900359 has no effect on the contractile response to CCh in PCLS from M3-KI mice. (A) Representative examples of CCh-induced bronchoconstriction in PCLS from M3-KI mice under control conditions (Left) or following preincubation with 30 nM FR900359 (Right). (B) Mean contractile response to CCh in M3-KI PCLS following preincubation with FR900359 (30 nM, 30 min), expressed as a percentage of CCh-stimulated maximum contraction in control WT PCLS. Max, Maximum; n.s., not significant.
Lung Function and Allergen-Associated Airway Hyperresponsiveness Are Dependent on M3-mAChR Phosphorylation.
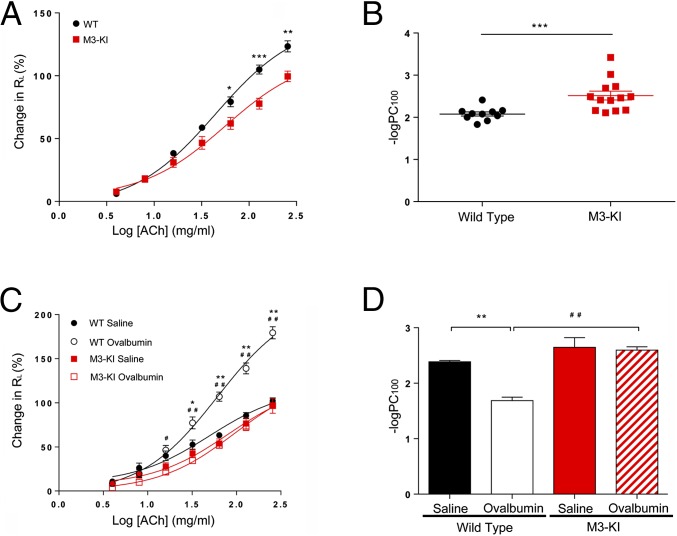
To test if the reduction in the contractile response of PCLS obtained from M3-KI mice translated to changes in in vivo lung function, lung resistance in response to ACh challenge in WT mice and M3-KI mice was monitored. These studies revealed a rightward shift in the potency and a decrease in the maximal response of the lung resistance induced by administration of ACh in M3-KI mice (Fig. 4A). The decrease in lung resistance in response to ACh in the M3-KI mice was also reflected in a significant increase in the concentration of ACh required to elevate lung resistance by 100% (−logPC100) (Fig. 4B).
Fig. 4.
Lung function and airway hyperresponsiveness are regulated by M3-mAChR phosphorylation/arrestin signaling. (A) Lung resistance (RL) was measured in WT and M3-KI mice at various ACh concentrations. Data represent the mean ± SEM (WT, n = 10; M3-KI, n = 13). (B) −LogPC100 was determined for WT and M3-KI mice. Data presented in A and B are the mean ± SEM (WT, n = 10; M3-KI, n = 13). Data were analyzed using Kruskal–Wallis and Mann–Whitney tests (WT vs. M3-KI: *P < 0.05; **P < 0.01; ***P < 0.001). (C) Airway hyperresponsiveness was induced using ovalbumin sensitization followed by ovalbumin challenge in WT and M3-KI mice. Controls were ovalbumin sensitization followed by saline challenge. RL was determined at various ACh concentrations. Data represent the mean ± SEM (WT, n = 6; M3-KI, n = 6). Data were analyzed using Kruskal–Wallis and Mann–Whitney tests (WT saline vs. WT ovalbumin: *P < 0.05; **P < 0.01 and WT ovalbumin vs. M3-KI ovalbumin: #P < 0.05; ##P < 0.01). (D) −LogPC100 was determined for WT and M3-KI mice sensitized with ovalbumin and challenged with either control saline or ovalbumin. Data present the mean ± SEM (WT, n = 6, M3-KI, n = 6). Data were analyzed using Kruskal–Wallis and Mann–Whitney tests (WT saline vs. WT ovalbumin: **P < 0.01 and WT ovalbumin vs. M3-KI ovalbumin: ##P < 0.01).
M3-mAChR signaling through RhoA in ASM has been closely linked with experimentally induced airway hyperresponsiveness in different studies using murine models of allergy (23, 24). We tested here if the reduced coupling of the phosphodeficient M3-mAChR mutant to RhoA signaling and the subsequent reduction in ASM contraction observed in M3-KI mice had an impact on allergen-induced airway hyperresponsiveness using a murine model of allergic inflammatory airway disease (25). In these experiments, WT mice sensitized with ovalbumin and challenged with saline (0.9% NaCl) showed an increase in lung resistance to ACh that was significantly augmented in mice sensitized with ovalbumin and challenged with ovalbumin (Fig. 4C), a response defined as classical airway hyperresponsiveness (24, 26, 27). In contrast, M3-KI mice sensitized with ovalbumin and challenged with ovalbumin showed no hyperresponsiveness in response to ACh administration (Fig. 4C). The lack of hyperresponsiveness following ovalbumin sensitization/challenge in M3-KI mice was also reflected by the lack of change in the −logPC100 value in M3-KI mice following ovalbumin sensitization/challenge (Fig. 4D). Hence, the −logPC100 value fell from 2.38 ± 0.02 in WT mice sensitized with ovalbumin and challenged with saline to 1.68 ± 0.06 after ovalbumin sensitization and ovalbumin challenge, indicative of airway hyperresponsiveness (Fig. 4D). In contrast, the −logPC100 value for the M3-KI mice that were ovalbumin-sensitized and saline-challenged (2.64 ± 0.17) was not significantly different from the −logPC100 value for M3-KI mice that had been ovalbumin-sensitized and ovalbumin-challenged (2.59 ± 0.06) (Fig. 4D). These data indicate that M3-mAChR phosphorylation was required in the development of allergen-induced airway hyperresponsiveness.
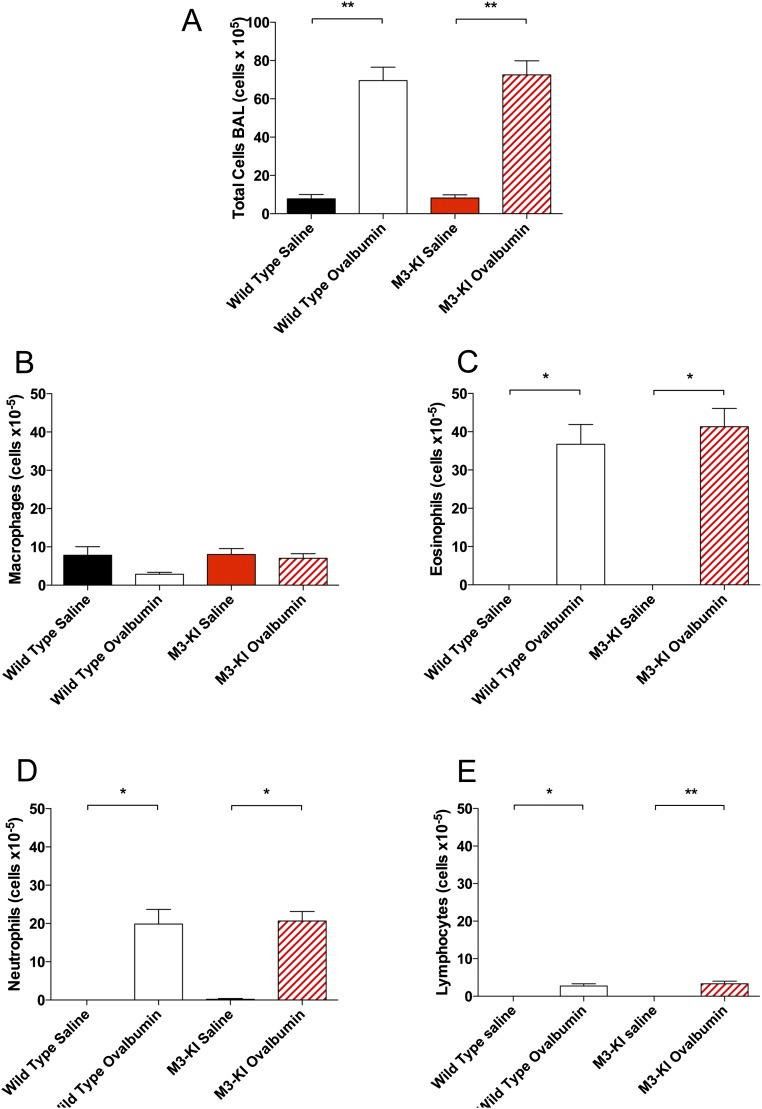
The acute ovalbumin-sensitization model used here is associated with an inflammatory response, as indicated by changes in cytokine levels and infiltration of immune cells into the lung. Analysis of cell infiltration by assessing total cells in the bronchoalveolar lavage (BAL) determined that the accumulation of inflammatory cells into the lung following ovalbumin sensitization and ovalbumin challenge was not significantly different between WT and M3-KI mice (Fig. S5A). Analysis of different inflammatory cell types in the BAL was carried out. Whereas the macrophage levels (Fig. S5B) were not seen to change following ovalbumin sensitization/challenge of WT and M3-KI mice, the numbers of eosinophils (Fig. S5C), neutrophils (Fig. S5D), and lymphocytes (Fig. S5E) were significantly increased, by the same extent, in WT and M3-KI mice sensitized with ovalbumin and challenged with ovalbumin.
Fig. S5.
Immune cell infiltration into the lung following allergen challenge is not dependent on M3-mAChR signaling via phosphorylation-dependent pathways. (A) Total cell count on BAL was counted using a trypan blue exclusion method. Cells isolated from BAL were centrifuged on glass slides and stained. Macrophages (B), eosinophils (C), neutrophils (D), and lymphocytes (E) were counted. Data represent the mean ± SEM (WT, n = 6; M3-KI, n = 6). Data were analyzed using Kruskal–Wallis and Mann–Whitney tests (*P < 0.05; **P < 0.01).
Phosphorylation of the M3-mAChR Regulates Specific Physiological Responses.
We show here how the G protein-biased M3-mAChR mutant receptor expressed in M3-KI mice can be used to define the role of receptor phosphorylation-dependent signaling in bronchial ASM. We reasoned that this approach could be extended to include other M3-mAChR–mediated responses and that a map of the physiological responses that lay downstream of the two fundamental signaling arms of the M3-mAChR, namely, G protein-dependent and phosphorylation/arrestin-dependent signaling, could be generated. To test this hypothesis, we compared the M3-mAChR–mediated contraction of bronchial smooth muscle with other M3-mAChR–mediated physiological responses, namely, salivary secretion and weight gain.
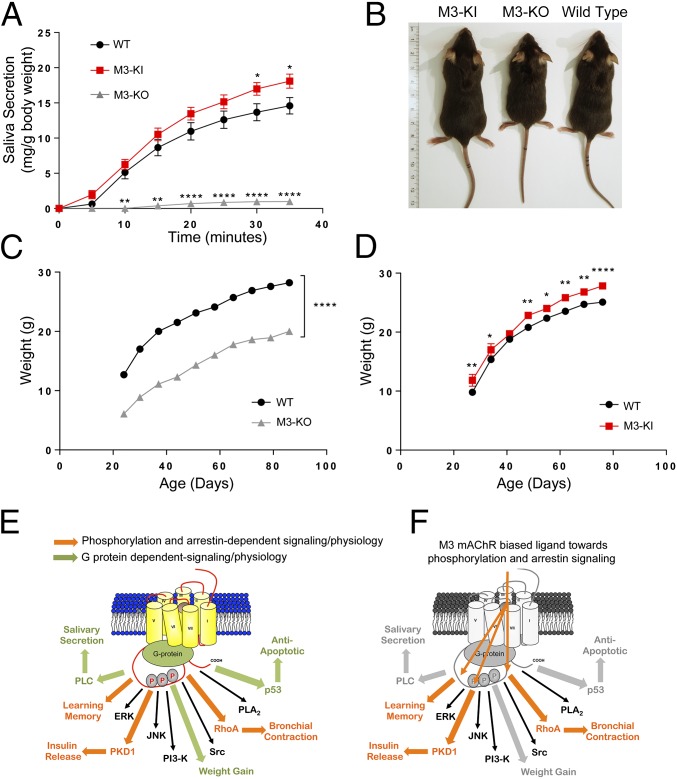
Previous gene KO studies established that salivary secretion in response to low doses of the muscarinic partial agonist pilocarpine was almost completely dependent on M3-mAChR (28). The results of these studies were confirmed here, where salivary secretion in response to pilocarpine was significantly reduced in M3-mAChR KO mice (Fig. 5A). In contrast, salivary secretion in response to pilocarpine in M3-KI mice was significantly enhanced compared with WT animals (Fig. 5A). These results indicated that in contrast to M3-mAChR–mediated contraction of ASM, M3-mAChR–mediated secretion of saliva was independent of receptor phosphorylation signaling but was dependent on signaling through Gq pathways. The slight increase in salivary secretion observed in M3-KI mice may reflect the fact that phosphorylation-dependent mechanisms, which are traditionally considered to desensitize G protein responses, are not in operation in M3-KI mice.
Fig. 5.
Mapping M3-mAChR physiological responses mediated by G protein- and receptor phosphorylation/arrestin-dependent signaling. (A) Salivary secretion in response to pilocarpine (1 mg/kg) administration was measured in WT, M3-KO, and M3-KI mice. The data represent the mean ± SEM of three to six mice. (B) Representative images of M3-KI, M3-KO, and WT mice demonstrating differences in weight. (C) Weight gain in WT and M3-KO mice. (D) Weight gain in WT and M3-KI mice. The data in A, C, and D represent the mean ± SEM of six to 11 mice and were analyzed using two-way ANOVA (*P < 0.05; **P < 0.01; ****P < 0.0001). (E) Illustration of the physiologically relevant signaling pathways downstream of the M3-mAChR. Some of the pathways activated by the M3-mAChR in heterologous systems are illustrated (i.e., PLC, ERK, PKD, JNK, PI3-K, Src, RhoA, PLA2, and p53). P13-K, phosphoinositide 3-kinase; PKD, protein kinase D; PLA2, phospholipase A2; PLC, phospholipase C. In this study, we used a mutant mouse strain (M3-KI) expressing a G protein-biased variant of the M3-mAChR to assign those physiological responses that were downstream of either G protein signaling (green arrows) or receptor phosphorylation and arrestin signaling (orange arrows). (F) Such a map also allows for the rational design of biased ligands because our studies present a model by which the physiological/therapeutic outcome of biased ligands can be predicted. Thus, for the M3-mAChR, a ligand that is biased toward receptor phosphorylation and arrestin signaling would preferentially affect insulin release, learning and memory, and bronchial contraction (orange arrows), although having potentially little impact on weight gain, salivary secretion, and cell death pathways (gray arrows).
M3-mAChR KO mice have also previously been reported to show reduced food intake and decreased body fat (29). This phenotype has been associated with the action of the M3-mAChR on the hypothalamic leptin/melanocortin system (29). In addition, the M3-mAChR KO mice showed an increase in both resting and total energy expenditure (oxygen consumption) as well as an increase in the rate of oxidation of fatty acids (30), phenotypes that have been linked with M3-mAChR regulation of sympathetic flow (30). Disruption of both of these physiological processes in M3-mAChR KO mice combines to generate an overall lean phenotype, where the mice show reduced total body weight (29, 31). We confirm these data here by demonstrating that M3-mAChR KO mice have reduced body weight compared with WT controls (Fig. 5 B and C). In contrast to the M3-mAChR KO mice, M3-KI mice were slightly, but significantly, heavier than the WT controls (Fig. 5 B and D). These data suggest that M3-mAChR signaling via Gq protein is the primary signaling pathway that regulates M3-mAChR metabolic and feeding responses responsible for weight gain.
Combining these data with previously published results (Discussion), a map of the physiological pathways downstream of phosphorylation/arrestin signaling and G protein-dependent signaling can be generated (Fig. 5E). This map can be used to predict the physiological/therapeutic outcome of biased M3-mAChR ligands (Fig. 5F).
Discussion
Our study demonstrates a previously unidentified role for M3-mAChR phosphorylation in the regulation of lung function and in driving airway hyperresponsiveness, one of the key features of allergic inflammatory airway disease. Our unique observation also highlights the importance of receptor phosphorylation in mediating M3-mAChR–induced ASM contraction via the activation of the RhoA pathway.
Because previous studies had demonstrated a role for RhoA signaling in calcium-independent ASM contraction (3–5), we used a genetically engineered mouse that expresses a phosphorylation-deficient variant of the M3-mAChR (M3-KI) to investigate the possibility that phosphorylation of the M3-mAChR was involved in coupling the receptor to RhoA signaling and, through this mechanism, to ASM contraction. That phosphorylation of the M3-mAChR plays a role in coupling to RhoA was indicated by (i) data showing that pharmacological inhibition of Rho-kinase in WT PCLS phenocopied the reduced airway contraction response observed in PCLS from M3-KI mice, (ii) that the phosphodeficient M3-mAChR showed markedly reduced coupling to RhoA, and (iii) that phosphorylation of MLC was significantly reduced in ASM of M3-KI mice. In addition, we used the recently characterized Gq inhibitor FR900359 (22) to demonstrate that M3-mAChR–induced calcium mobilization was inhibited by FR900359, whereas M3-induced airway contraction was not. Because our experimental design measured the overall calcium changes in whole PCLS, we cannot completely exclude a role of compartmentalized changes in Gq-driven calcium signals in ASM in the regulation of airway contraction, as shown by others (32, 33). Nonetheless, our data are consistent with a recent report demonstrating that muscarinic-mediated contractile responses in human PCLS were less sensitive to FR900359, and therefore less dependent on calcium, than contraction mediated by histamine receptors (34). Furthermore, knocking down β-arrestin-1 has recently been shown to impair muscarinic-mediated bronchoconstriction (33), demonstrating the complexity surrounding the procontractile pathways coupled to M3-mAChRs in ASM. These studies, coupled with our data, support the hypothesis that M3-mAChR phosphorylation mediates activation of RhoA signaling (possibly by arrestin recruitment) that results in increased MLC phosphorylation and ASM contraction. This phosphorylation-dependent calcium sensitization process plays a prominent role in M3-mAChR–dependent ASM contraction, with a minor role played by calcium transient-dependent processes.
It is known that vagally derived ACh limits airflow by bronchoconstriction mediated by the activation of the M3-mAChR on ASM (1). If the role for M3-mAChR phosphorylation in ASM contraction identified here is of physiological relevance, then it might be expected that the lung function of M3-KI mice would be different from the lung function of WT mice. We found (by assessing lung resistance) that airway obstruction in response to ACh was significantly reduced in M3-KI mice compared with WT mice, an observation consistent with the notion that the phosphorylation status of the M3-mAChR is important in regulating baseline cholinergic ASM tone. This observation is in line with studies conducted in healthy volunteers showing a significant improvement in lung function by anticholinergic drugs (2). Furthermore, in an acute murine model of allergic inflammatory airway disease, which mimics several of the central hallmarks of human bronchial asthma (35), including airway hyperresponsiveness and immune cell lung infiltration, we show that the development of allergen-induced airway hyperresponsiveness to ACh was dependent on M3-mAChR phosphorylation-dependent pathways because sensitized M3-KI mice failed to demonstrate the typically enhanced airway hyperresponsiveness. Thus, both normal and pathophysiological lung function in M3-KI mice was significantly different from WT mice, indicating an important role for M3-mAChR phosphorylation in the regulation of airway narrowing in health and disease states.
The mechanisms by which allergen-exposed M3-KI mice were protected against the development of abnormal bronchial responsiveness appeared to be unrelated to the number of inflammatory cells (eosinophils, lymphocytes, and neutrophils) recruited within the airways. This dissociation between airway inflammation and the degree of airway hyperresponsiveness has been previously reported in various studies, including observations in asthmatic patients (36) as well as in animal models of allergic inflammatory airway disease (34, 37, 38). Rather, we propose that in the lung, phosphorylation of the M3-mAChR is important in driving key asthmatic features, such as airway hyperresponsiveness, in part, via M3-mAChR phosphorylation and RhoA-dependent signaling.
Because our M3-KI mouse line expressing G protein-biased M3-mAChR mutant defined a previously unidentified role for M3-mAChR phosphorylation-dependent signaling in ASM contraction, we reasoned that this mouse line could be used to investigate other M3-mAChR–mediated physiological responses and establish whether these responses were dependent on G protein- or receptor phosphorylation/arrestin-dependent signaling. We therefore examined M3-mAChR–mediated salivary secretion and weight gain and established that both of these responses were slightly up-regulated in M3-KI mice, indicating that they were not dependent on receptor phosphorylation/arrestin signaling, but more likely dependent on G protein coupling. The loss of the phosphorylation-dependent desensitization of G protein-mediated signaling may be responsible for the modest but significant increase in weight and salivary secretion observed in M3-KI mice.
These physiological responses could be added to our previous studies demonstrating a predominant role for M3-mAChR phosphorylation/arrestin coupling in the regulation of insulin secretion (14) and learning and memory (15), as well as in G protein coupling in M3-mAChR regulation of p53 subcellular localization and apoptosis (39, 40). By combining all these studies, we have generated a map of the physiological responses mediated by the M3-mAChR through either G protein- or receptor phosphorylation/arrestin-dependent signaling (Fig. 5E).
This map has allowed us, for the first time to our knowledge, to make a prediction of the likely physiological outcomes of a biased GPCR ligand. Thus, an M3-mAChR drug-like ligand showing stimulus bias toward receptor phosphorylation and arrestin signaling would preferentially engage physiological signaling pathways that would promote changes in insulin secretion, have an impact on learning and memory, and regulate bronchoconstriction (Fig. 5F). This same ligand would not be expected to have a substantial impact on salivary secretion, weight gain, and mechanisms associated with cell survival (Fig. 5F). This analysis has considerable potential application in drug discovery, where biased ligands can be designed to direct signaling toward those pathways that result in clinical efficacy and away from pathways that lead to adverse/toxic outcomes. Hence, by using a genetically modified mouse expressing a G protein-biased receptor, we are not only able to define the physiologically relevant signaling pathways acting downstream of a particular GPCR (in this case, the M3-mAChR) but also to determine what type of signaling bias to design into ligands targeting that receptor to promote a particular physiological/therapeutic outcome.
Experimental Procedures
Information on materials and methods used in this study is provided in SI Experimental Procedures. Animal experiments were conducted under the approval of the UK government home office licence PPL 802353 following ethic review by the University of Leicester.
SI Experimental Procedures
Animal Experimentation.
Experiments were performed under a project license from the British Home Office, United Kingdom, under the Animals (Scientific Procedures) Act of 1986.
Weight Gain.
Male C57BL/6NTac WT (n = 10) and M3-KO (n = 10) mice and C57BL/6J WT (n = 10) and M3-KI (n = 6) mice were weighed weekly after weaning (between day 24 and day 27 after birth). Mouse were housed in groups of three to five and fed with normal chow ad libitum.
Preparation of PCLS.
Precision cut lung slicing was performed as described previously (10, 41). WT C57BL/6J or M3-KI adult mice (8–10 wk old) were killed by cervical dislocation. The trachea was exposed, and a cannula was inserted. Lungs were inflated using 1.0 mL of a 2% (wt/vol) low-melting-point agarose solution, followed by a 0.1-mL air bolus to force the agarose out of the airways and into the alveoli. The inflated lungs were dissected from the thoracic cavity, mounted in agarose, and sliced using a Manual Vibroslice (World Precision Instruments) at a thickness of 250 μm and placed in ice-cold HBSS buffer [10 mM Hepes, 1 mM MgCl2, 2 mM CaCl2 (pH 7.4)]. Suitable airways on slices were selected on the basis of the following criteria: presence of a full smooth muscle wall (i.e., cut perpendicular to the direction of the airway), presence of beating cilia to eliminate blood vessels, and unshared muscle walls at airway branch points to eliminate possible counteracting contractile forces. Slices were then transferred to incubating buffer and incubated at 37 °C on a rotating platform. Trauma caused by tissue slicing contracts the airway, presumably by the release of mediators. The incubation buffer (DMEM supplemented with Glx and penicillin/streptomycin) was therefore changed every 30 min for 6 h to remove any constrictor mediators released from the tissue that would prevent the airway from relaxing to baseline and then every 24 h as indicated. The next day (18 h later), slices were washed again with fresh medium and used in experiments.
Contraction Responses of PCLS.
PCLS were placed in a 12-well plate in 1.0 mL of assay buffer. The airway was located using a microscope (Zeiss Axiovert 25), and the slice was held in place using a stainless-steel ring. Media were added for baseline measurements. The camera was positioned so that a live video feed (QICAM; QImaging) of the airway could be viewed. A baseline image was taken, followed by the administration of different concentrations of carbachol (CCh). Images were collected every 5 min after each concentration.
Airway lumen size was measured using a macro written within Image Pro-Plus (version 6.0) software (Media Cybernetics) and given in units of 2 μm. After functional studies, the area of each airway at baseline and at the end of each dose of agonist was calculated using the same macro written within Image Pro-Plus software. Data were plotted as a percentage of contractions of the initial (basal) airway size (expressed as 100%) for each concentration. Log pEC50 and Emax values for each airway were derived from a concentration–response curve. Data were expressed as the mean ± SEM. Statistical difference was shown by using paired and unpaired t tests.
Immunostaining for Ser-19 Phosphorylation of MLC2 in PCLS.
For phosphorylated Ser-19 MLC staining, PCLS were prepared as described above. Slices were incubated in HBSS and stimulated with vehicle or CCh (100 μM, 10 min). PCLS were fixed for 1 h in 4% (wt/vol) paraformaldehyde (PFA)/PBS and then washed in Tris-buffered saline (TBS) plus 0.1% Triton X-100. Free-floating PCLS were then blocked for 2 h at room temperature (RT) in TBS, 0.1% Triton X-100, 10% (vol/vol) goat serum, and 5% (wt/vol) BSA. Slices were incubated with an Ab to phospho-MLC2 (Ser-19) (1:100 in blocking buffer; Cell Signaling Technology) overnight at 4 °C. Slices were washed three times in TBS plus 0.1% Triton X-100 and then incubated for 2 h at RT with rabbit Alexa Fluor 488 (Molecular Probes, Inc.) and antiactin α-smooth muscle-Cy3 Ab (Sigma–Aldrich) (both 1:400 in blocking buffer). Slices were then washed three times, and mounted in Vectorshield hardset mounting medium (Vector Laboratories) with DAPI.
Radioligand Binding.
Crude lung membranes from WT C57BL/6J and M3-KI mice were prepared as follows. Mice were culled by cervical dislocation, and lungs were removed and homogenized in 19 vol of ice-cold assay buffer [30 mM Hepes, 100 mM NaCl, 0.5 mM EGTA (pH 7.5)]. The suspension was subsequently centrifuged at 40,000 × g for 20 min at 4 °C using an RC5 centrifuge (Sorvall) with an SS34 rotor. Supernatant was discarded, and the pellet was resuspended in assay buffer. The suspension was homogenized, the protein concentration was determined using a Bradford protein assay, and membranes were adjusted to a 0.5-mg/mL concentration to use in the binding assay.
Saturation binding experiments were performed in duplicate with increasing concentrations of [3H]-N-methyl scoploamine (NMS) (0.1–2.5 nM) added to each tube containing assay buffer (as above). Nonspecific binding for each concentration of [3H]-NMS was determined by the addition of 10 μM atropine. Membranes (50 μg) were added to each tube (to a total volume of 500 μL) for 2 h at RT. Membrane-bound radioligand was separated from free ligand by rapid filtration onto GF/B glass microfiber filters (GE Healthcare Life Sciences), followed by washing twice with ice-cold 0.9% NaCl. Membrane-bound radioactivity was extracted overnight and determined by liquid scintillation counting.
Reverse Transcriptase PCR.
WT C57BL/6J, M3-KO, or M3-KI adult mice were killed by cervical dislocation, and tissue from the hippocampus, heart, and lung was removed and immediately frozen on dry ice. Total RNA was prepared from 100 mg of each tissue using an RNeasy Lipid Tissue Mini Kit (Qiagen) according to the manufacturer’s instructions, including treatment with RNase-free DNase. First-strand synthesis was performed using SuperScript III First-Strand Synthesis SuperMix (Life Technologies) with 1 μg of total RNA template per reaction. Phusion DNA polymerase (New England Biolabs) was used in PCR reactions to amplify muscarinic receptor subtypes from the resulting cDNA using 5 μL of first-strand synthesis reaction products as a template. The following primer pairs were used:
M2 682 Forward (F) 5′ AAGAAAAGAAGGAACCAGTGGCCAACC
M2 1141 Reverse (R) 5′ TGTCCTGGTCACTTTCTTTTCCCG
M3 789 F 5′ ATAAGGAGACTGAGAAACGTACCAAAGAG
M3 1430 R 5′ TCCTTGATGAGCGACATCCTTTTCC
M4 681 F 5′ AGTCGCAGCCGAGTCCACAAG
M4 926 R 5′ CTCTGTGGTGGACAGCTCTGTGG
β-Actin 41 F 5′ GCTCG TCGTC GACAA CGGCT C
β-Actin 349 R 5′ CAAAC ATGAT CTGGG TCATC TTCTC
Immunostaining for the M3-mAChR in Mouse Lung.
Mice were killed by cervical dislocation, ensuring that the trachea remained intact. The chest cavity and trachea were exposed, and the trachea was cannulated. The lungs were gently overinflated with ice-cold 4% (wt/vol) PFA. Following fixation, lungs were immediately removed and further fixed overnight in 4% (wt/vol) PFA at 4 °C. Lungs were processed in paraffin wax, and sections from each lobe were taken using a microtome. Slices were either stained with H&E or underwent antigen retrieval. Following antigen retrieval, sections were washed in TBS plus 0.1% Triton X-100 and blocked for 2 h at RT in TBS, 0.1% Triton X-100, 10% (vol/vol) goat serum, and 5% (wt/vol) BSA. Sections were incubated with a mouse mAb (in-house) to the M3-mAChR (1:100 in blocking buffer) overnight at 4 °C. Following this incubation, sections were washed three times in TBS plus 0.1% Triton X-100 and incubated with a rabbit α-actin Ab (1:100 in blocking buffer; Abcam) for 1 h at RT. Sections were washed again three times, and then incubated with both fluorescent secondary Abs (1:400 in blocking buffer; mouse Alexa Fluor 488 and rabbit Alexa Fluor 594; both from Molecular Probes, Inc.) for 1 h at RT. Following three washes, slices were mounted in Vectorshield hardset mounting medium with DAPI. All images were taken using either a Zeiss Axiovert 200M microscope with a Colibri illumination system with Axiovision 4.8 software (Zeiss) or a Zeiss LSM 510 META NLO microscope with Zen 2009 software (Zeiss).
Ca2+ Signaling in Lung Slices.
PCLS from WT or M3-KI mice were washed twice in Kreb’s Heinseleit buffer (KHB) and incubated in KHB containing 4 μM Fura-2:00 AM (Sigma-Aldrich), 1 mg/mL BSA, and probenecid for 1 h at 37 °C. After loading, the slice was placed in a prewarmed chamber containing KHB. Measurements were made on a Zeiss Axiovert 200 inverted epifluorescence microscope with a 40× oil-immersion objective. Loaded lung slices were excited at 340 nm and 380 nm at a sample rate of 0.67 Hz by means of an excitation wheel. Sequential fluorescent image pairs were recorded at wavelengths >510 nm via a cooled ORCA-ER CCD camera (Hamamatsu Photonics) and processed with MetaFluor software (Molecular Devices). Free intracellular Ca2+ signal was expressed at a ratio of 340:380 or change of the ratio of fluorescence.
Determination of RhoA Activation.
Polyornithine-coated 96-well plates were seeded with CHO cells (10,000 cells per well). After 24 h, cells were transfected with the Rho-binding-domain (Raichu-RBD) probe [described by Itoh et al. (42)] and with either M3-mAChR or the M3-receptor phosphonegative mutant, using Lipofectamine 2000. The media was changed 24 h posttransfection. On the next day, cells were starved for 1 h at RT in Tyrode buffer. FRET signal was assessed using an EnSpire reader (PerkinElmer), after cells were treated for 15 min with an increasing concentration of CCh diluted in Tyrode buffer or Tyrode buffer alone at 25 °C. Briefly, the Raichu-RBD sensor was excited at 405 nm, and FRET (520–540/4 nm) or CFP (460–480/4 nm) emission was recorded (500 flashes). Area-under-the-curve values for each emission were assessed, and the FRET values were calculated using a FRET/CFP ratio. Data are expressed as the mean ± SEM, represent four independent experiments, and were analyzed using two-way ANOVA followed by a Bonferroni correction (*P < 0.05; **P < 0.01).
Ovalbumin Sensitization of Mice.
Mice were sensitized on day 0 and day 10 with alum (diluted 1:1 in saline) and ovalbumin (10 μg per mouse, i.p.) for a period of 19 d. On days 19–24, mice were challenged with vehicle, saline intranasal (i.n.), or ovalbumin (50 μg per mouse, i.n.). Airway hyperresponsiveness (AHR) assessment, BAL, and tissue collection were performed 24 h after the last challenge on day 25.
AHR Assessment.
Resistance/compliance measurements were obtained from tracheotomized and ventilated mice using whole-body plethysmography (EMMS) as previously described (43). Lung resistance (RL) was expressed as a percentage of change from baseline RL with nebulized PBS (Sigma). The concentration of ACh required to increase RL by 100% from baseline was calculated (PC100), and −log PC100 was taken as a measure of AHR.
BAL Collection.
BAL samples and cytospins were obtained and analyzed as previously described (2). BAL fluid (BALF) was obtained by rinsing lungs three times with 0.8 mL of cold PBS via endotracheal tube. BALF samples were centrifuged to isolate cells from fluid and frozen at −80 °C until analysis. BAL cells were resuspended in 200 μL of PBS and centrifuged (30 × g for 6 min in a Shandon Cytospin 4; Thermo Scientific) onto glass slides. Cell slides were subsequently fixed and stained using a Diff-Quick Kit (Reagena) and counted under an optical microscope (Olympus BH2).
Cytokine Measurements in BALF.
BALF was used to measure IL-5, IL-6, keratinocyte-derived cytokine, IFN-γ–induced protein-10, eotaxin, and TNF-α concentrations using magnetic beads coated with capture Ab. Biotinylated detection Ab to each analyte was bound to the bead–cytokine complex, followed by the addition of phycoerythrin (PE)-conjugated streptavidin. Magnetic beads were read using a Luminex MAGPIX analyzer. Magnetic beads were illuminated, and two spectrally distinct light-emitting diodes illuminated the beads. The analyte of interest and the magnitude of the PE-derived signal were detected and recorded on xPONENT software (Luminex). The mean fluorescent intensity was analyzed using a five-parameter logistic method on XLfit software v.5.3.1.3.
Salivary Secretion.
Mice were anesthetized by i.p. injection with 100 mg/kg of ketamine/0.25 mg/kg of medetomidine. Following this procedure, mice were injected with pilocarpine (1 mg/mL, i.p.) and salivary secretion (in milligrams of saliva) onto GF/B filter paper (GE Healthcare Life Sciences) was recorded every 5 min over a 35-min period.
Plasmids Used for G Protein BRET Biosensor.
Rluc II Gα-subunit constructs were generated by insertion of Rluc II into the α-helical domain of the Gα-subunit. GFP10 was fused to the N-terminal domain of Gγ1 to generate GFP10-Gγ1 as described previously (19).
G Protein BRET Biosensor Assay.
To assess G protein coupling to the M3-mAChR, HEK-293 cells were seeded into six-well plates at 5 × 105 cells per well and allowed to settle. Cells were then cotransfected with 200 ng of receptor, 200 ng of Rluc II Gα-subunit, 100 ng of Gβ1, and 200 ng GFP10-Gγ1 subunits using PEI (Polysciences) at a ratio of 3 μg of PEI to 1 μg of cDNA. Twenty-four hours after transfection, cells were detached using PBS containing 1 mM EDTA and distributed into poly-d-lysine–coated white 96-well microplates and incubated for a further 24 h before the assay. Cells were then washed with HBSS and incubated in HBSS for 1 h at 37 °C. To initiate the assay, Coelenterazine 400a (Nanolight Technology) was added (2.5 μM) and incubated for 5 min; cells were then stimulated with CCh (100 μM) for 10 min before emissions at 400 nm and 510 nm were measured using a Tristar LB 941 multimode plate reader (Berthold Technologies) equipped with a bioluminescence resonance energy transfer 2 (BRET2) filter set (400 ± 70-nm and 515 ± 20-nm filters). The BRET signal was represented as the 510/400 ratio. The ligand-induced change in BRET signal was determined by subtracting the signal obtained by the addition of vehicle from the values obtained from the addition of CCh.
Statistics.
Where indicated, data were evaluated using two-way ANOVA, Kruskal–Wallis, and Mann–Whitney tests; paired and unpaired t tests; and two-way ANOVA with Dunnett’s multiple comparisons test.
Supplementary Material
Acknowledgments
We thank Dr. Christos Rossios for cytokine assay and Dr. Jurgen Wess (NIH) for providing the M3-KO mice and help with generating the M3-KI mice. This study is funded by the Medical Research Council (MRC) through funding of program leaders provided by the MRC Toxicology Unit (to A.B.T.). S.A.L. is a “Chercheur Senior” from the Fonds de recherche du Québec-Santé. M.B. holds a Canada Research Chair in Signal Transduction and Molecular Pharmacology. Part of the study (BRET-based profiling) was funded by Canadian Institutes of Health Research Grant 10501 (to M.B.). Funding for the isolation of FR900359 was provided by the Deutsche Forschungsgemeinschaft research unit FOR2372 (to G.M.K. and E.K.). This study was performed in laboratories partially funded by European Regional Development Fund Grant 05567 and supported by the National Institute for Health Research Leicester Respiratory Biomedical Research Unit.
Footnotes
The authors declare no conflict of interest.
This article is a PNAS Direct Submission.
This article contains supporting information online at www.pnas.org/lookup/suppl/doi:10.1073/pnas.1521706113/-/DCSupplemental.
References
- 1.Gosens R, Zaagsma J, Meurs H, Halayko AJ. Muscarinic receptor signaling in the pathophysiology of asthma and COPD. Respir Res. 2006;7:73. doi: 10.1186/1465-9921-7-73. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Singh D, et al. Plethysmography and impulse oscillometry assessment of tiotropium and ipratropium bromide; a randomized, double-blind, placebo-controlled, cross-over study in healthy subjects. Br J Clin Pharmacol. 2006;61(4):398–404. doi: 10.1111/j.1365-2125.2006.02594.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Somlyo AP, Somlyo AV. Ca2+ sensitivity of smooth muscle and nonmuscle myosin II: Modulated by G proteins, kinases, and myosin phosphatase. Physiol Rev. 2003;83(4):1325–1358. doi: 10.1152/physrev.00023.2003. [DOI] [PubMed] [Google Scholar]
- 4.Bai Y, Sanderson MJ. Modulation of the Ca2+ sensitivity of airway smooth muscle cells in murine lung slices. Am J Physiol Lung Cell Mol Physiol. 2006;291(2):L208–L221. doi: 10.1152/ajplung.00494.2005. [DOI] [PubMed] [Google Scholar]
- 5.Liu C, Zuo J, Janssen LJ. Regulation of airway smooth muscle RhoA/ROCK activities by cholinergic and bronchodilator stimuli. Eur Respir J. 2006;28(4):703–711. doi: 10.1183/09031936.06.00025506. [DOI] [PubMed] [Google Scholar]
- 6.Vogt S, Grosse R, Schultz G, Offermanns S. Receptor-dependent RhoA activation in G12/G13-deficient cells: Genetic evidence for an involvement of Gq/G11. J Biol Chem. 2003;278(31):28743–28749. doi: 10.1074/jbc.M304570200. [DOI] [PubMed] [Google Scholar]
- 7.Mitchell R, et al. Rhodopsin-family receptors associate with small G proteins to activate phospholipase D. Nature. 1998;392(6674):411–414. doi: 10.1038/32937. [DOI] [PubMed] [Google Scholar]
- 8.Chiba Y, Matsusue K, Misawa M. RhoA, a possible target for treatment of airway hyperresponsiveness in bronchial asthma. J Pharmacol Sci. 2010;114(3):239–247. doi: 10.1254/jphs.10r03cr. [DOI] [PubMed] [Google Scholar]
- 9.Kume H. RhoA/Rho-kinase as a therapeutic target in asthma. Curr Med Chem. 2008;15(27):2876–2885. doi: 10.2174/092986708786242831. [DOI] [PubMed] [Google Scholar]
- 10.Lefkowitz RJ. A brief history of G-protein coupled receptors (Nobel Lecture) Angew Chem Int Ed Engl. 2013;52(25):6366–6378. doi: 10.1002/anie.201301924. [DOI] [PubMed] [Google Scholar]
- 11.Anthony DF, et al. β-Arrestin 1 inhibits the GTPase-activating protein function of ARHGAP21, promoting activation of RhoA following angiotensin II type 1A receptor stimulation. Mol Cell Biol. 2011;31(5):1066–1075. doi: 10.1128/MCB.00883-10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Barnes WG, et al. beta-Arrestin 1 and Galphaq/11 coordinately activate RhoA and stress fiber formation following receptor stimulation. J Biol Chem. 2005;280(9):8041–8050. doi: 10.1074/jbc.M412924200. [DOI] [PubMed] [Google Scholar]
- 13.Butcher AJ, et al. Differential G-protein-coupled receptor phosphorylation provides evidence for a signaling bar code. J Biol Chem. 2011;286(13):11506–11518. doi: 10.1074/jbc.M110.154526. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Kong KC, et al. M3-muscarinic receptor promotes insulin release via receptor phosphorylation/arrestin-dependent activation of protein kinase D1. Proc Natl Acad Sci USA. 2010;107(49):21181–21186. doi: 10.1073/pnas.1011651107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Poulin B, et al. The M3-muscarinic receptor regulates learning and memory in a receptor phosphorylation/arrestin-dependent manner. Proc Natl Acad Sci USA. 2010;107(20):9440–9445. doi: 10.1073/pnas.0914801107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Galandrin S, Oligny-Longpré G, Bouvier M. The evasive nature of drug efficacy: Implications for drug discovery. Trends Pharmacol Sci. 2007;28(8):423–430. doi: 10.1016/j.tips.2007.06.005. [DOI] [PubMed] [Google Scholar]
- 17.Azzi M, et al. Beta-arrestin-mediated activation of MAPK by inverse agonists reveals distinct active conformations for G protein-coupled receptors. Proc Natl Acad Sci USA. 2003;100(20):11406–11411. doi: 10.1073/pnas.1936664100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Paradis JS, et al. Receptor sequestration in response to β-arrestin-2 phosphorylation by ERK1/2 governs steady-state levels of GPCR cell-surface expression. Proc Natl Acad Sci USA. 2015;112(37):E5160–E5168. doi: 10.1073/pnas.1508836112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Galés C, et al. Real-time monitoring of receptor and G-protein interactions in living cells. Nat Methods. 2005;2(3):177–184. doi: 10.1038/nmeth743. [DOI] [PubMed] [Google Scholar]
- 20.Sanderson MJ. Exploring lung physiology in health and disease with lung slices. Pulm Pharmacol Ther. 2011;24(5):452–465. doi: 10.1016/j.pupt.2011.05.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Amrani Y, et al. Expression and activation of the oxytocin receptor in airway smooth muscle cells: Regulation by TNFalpha and IL-13. Respir Res. 2010;11:104. doi: 10.1186/1465-9921-11-104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Schrage R, et al. The experimental power of FR900359 to study Gq-regulated biological processes. Nat Commun. 2015;6:10156. doi: 10.1038/ncomms10156. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Schaafsma D, et al. Differential Rho-kinase dependency of full and partial muscarinic receptor agonists in airway smooth muscle contraction. Br J Pharmacol. 2006;147(7):737–743. doi: 10.1038/sj.bjp.0706665. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Witzenrath M, et al. Rho-kinase and contractile apparatus proteins in murine airway hyperresponsiveness. Exp Toxicol Pathol. 2008;60(1):9–15. doi: 10.1016/j.etp.2008.03.002. [DOI] [PubMed] [Google Scholar]
- 25.Nath P, et al. Complete inhibition of allergic airway inflammation and remodelling in quadruple IL-4/5/9/13-/- mice. Clin Exp Allergy. 2007;37(10):1427–1435. doi: 10.1111/j.1365-2222.2007.02789.x. [DOI] [PubMed] [Google Scholar]
- 26.Aad G, et al. ATLAS Collaboration CMS Collaboration Combined Measurement of the Higgs Boson Mass in pp Collisions at sqrt[s]=7 and 8 TeV with the ATLAS and CMS Experiments. Phys Rev Lett. 2015;114(19):191803. doi: 10.1103/PhysRevLett.114.191803. [DOI] [PubMed] [Google Scholar]
- 27.Schaafsma D, Bos IS, Zuidhof AB, Zaagsma J, Meurs H. Inhalation of the Rho-kinase inhibitor Y-27632 reverses allergen-induced airway hyperresponsiveness after the early and late asthmatic reaction. Respir Res. 2006;7:121. doi: 10.1186/1465-9921-7-121. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Gautam D, et al. Cholinergic stimulation of salivary secretion studied with M1 and M3 muscarinic receptor single- and double-knockout mice. Mol Pharmacol. 2004;66(2):260–267. doi: 10.1124/mol.66.2.260. [DOI] [PubMed] [Google Scholar]
- 29.Yamada M, et al. Mice lacking the M3 muscarinic acetylcholine receptor are hypophagic and lean. Nature. 2001;410(6825):207–212. doi: 10.1038/35065604. [DOI] [PubMed] [Google Scholar]
- 30.Gautam D, et al. Metabolic roles of the M3 muscarinic acetylcholine receptor studied with M3 receptor mutant mice: A review. J Recept Signal Transduct Res. 2008;28(1-2):93–108. doi: 10.1080/10799890801942002. [DOI] [PubMed] [Google Scholar]
- 31.Gautam D, et al. A critical role for beta cell M3 muscarinic acetylcholine receptors in regulating insulin release and blood glucose homeostasis in vivo. Cell Metab. 2006;3(6):449–461. doi: 10.1016/j.cmet.2006.04.009. [DOI] [PubMed] [Google Scholar]
- 32.Borchers MT, et al. Methacholine-induced airway hyperresponsiveness is dependent on Galphaq signaling. Am J Physiol Lung Cell Mol Physiol. 2003;285(1):L114–L120. doi: 10.1152/ajplung.00322.2002. [DOI] [PubMed] [Google Scholar]
- 33.Carr R, 3rd, et al. Interdicting Gq Activation in Airway Disease by Receptor-Dependent and Receptor-Independent Mechanisms. Mol Pharmacol. 2016;89(1):94–104. doi: 10.1124/mol.115.100339. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Birrell MA, Battram CH, Woodman P, McCluskie K, Belvisi MG. Dissociation by steroids of eosinophilic inflammation from airway hyperresponsiveness in murine airways. Respir Res. 2003;4:3. doi: 10.1186/rr197. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.National Heart Lung, and Blood Institute and GINA Executive Committee . Global Strategy for Asthma Management and Prevention, NIH Publication No. 02-3659. Bethesda; 2004. [Google Scholar]
- 36.Crimi E, et al. Dissociation between airway inflammation and airway hyperresponsiveness in allergic asthma. Am J Respir Crit Care Med. 1998;157(1):4–9. doi: 10.1164/ajrccm.157.1.9703002. [DOI] [PubMed] [Google Scholar]
- 37.Heuer HO, Wenz B, Jennewein HM, Urich K. Dissociation of airway responsiveness and bronchoalveolar lavage (BAL) cell composition in sensitized guinea-pigs after daily inhalation of ovalbumin. Clin Exp Allergy. 1994;24(7):682–689. doi: 10.1111/j.1365-2222.1994.tb00973.x. [DOI] [PubMed] [Google Scholar]
- 38.Kelada SN, et al. Strain-dependent genomic factors affect allergen-induced airway hyperresponsiveness in mice. Am J Respir Cell Mol Biol. 2011;45(4):817–824. doi: 10.1165/rcmb.2010-0315OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Budd DC, McDonald J, Emsley N, Cain K, Tobin AB. The C-terminal tail of the M3-muscarinic receptor possesses anti-apoptotic properties. J Biol Chem. 2003;278(21):19565–19573. doi: 10.1074/jbc.M211670200. [DOI] [PubMed] [Google Scholar]
- 40.Tobin AB, Budd DC. The anti-apoptotic response of the Gq/11-coupled muscarinic receptor family. Biochem Soc Trans. 2003;31(Pt 6):1182–1185. doi: 10.1042/bst0311182. [DOI] [PubMed] [Google Scholar]
- 41.Cooper PR, et al. C-027 inhibits IgE-mediated passive sensitization bronchoconstriction and acts as a histamine and serotonin antagonist in human airways. Allergy Asthma Proc. 2011;32(5):359–365. doi: 10.2500/aap.2011.32.3460. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Itoh RE, et al. Activation of rac and cdc42 video imaged by fluorescent resonance energy transfer-based single-molecule probes in the membrane of living cells. Mol Cell Biol. 2002;22(18):6582–6591. doi: 10.1128/MCB.22.18.6582-6591.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Pinart M, et al. IL-17A modulates oxidant stress-induced airway hyperresponsiveness but not emphysema. PLoS One. 2013;8(3):e58452. doi: 10.1371/journal.pone.0058452. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.