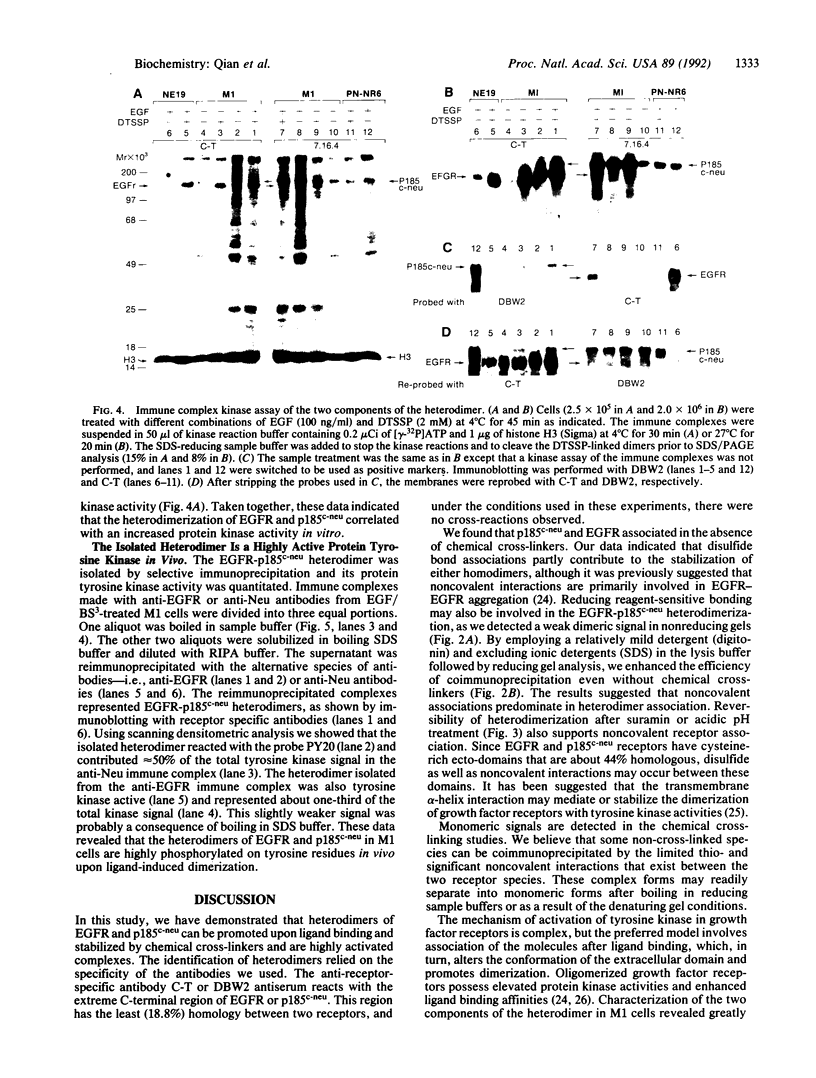
Abstract
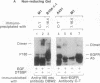
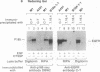
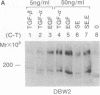
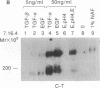
The protein product of the neu protooncogene, p185c-neu, is structurally similar to the epidermal growth factor receptor (EGFR). Overexpression of these two receptor tyrosine kinases, but not either separately, leads to transformation and tumorigenicity. Heterodimerization of p185c-neu and EGFR occurs in M1 cells, which express both receptors. We have individually identified the two components of the heterodimer as EGFR and p185c-neu. Analysis of this association with relatively nondenaturing detergents and in the absence of cross-linkers indicates that noncovalent interactions are primarily responsible for heterodimer formation. The rapid reversible heterodimerization was promoted by EGF binding to its receptor. Functionally, the heterodimer is a highly active protein kinase for receptor autophosphorylation and exogenous substrate phosphorylation in vitro. The isolated heterodimer was highly phosphorylated on tyrosine residues in vivo. These results indicate that the physical association between EGFR and p185c-neu is of functional significance and define enzymatic features of complex receptor formation.
Full text
PDF



Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Akiyama T., Saito T., Ogawara H., Toyoshima K., Yamamoto T. Tumor promoter and epidermal growth factor stimulate phosphorylation of the c-erbB-2 gene product in MKN-7 human adenocarcinoma cells. Mol Cell Biol. 1988 Mar;8(3):1019–1026. doi: 10.1128/mcb.8.3.1019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ascoli M. Internalization and degradation of receptor-bound human choriogonadotropin in Leydig tumor cells. Fate of the hormone subunits. J Biol Chem. 1982 Nov 25;257(22):13306–13311. [PubMed] [Google Scholar]
- Barber J. R., Verma I. M. Modification of fos proteins: phosphorylation of c-fos, but not v-fos, is stimulated by 12-tetradecanoyl-phorbol-13-acetate and serum. Mol Cell Biol. 1987 Jun;7(6):2201–2211. doi: 10.1128/mcb.7.6.2201. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bargmann C. I., Hung M. C., Weinberg R. A. Multiple independent activations of the neu oncogene by a point mutation altering the transmembrane domain of p185. Cell. 1986 Jun 6;45(5):649–657. doi: 10.1016/0092-8674(86)90779-8. [DOI] [PubMed] [Google Scholar]
- Bargmann C. I., Hung M. C., Weinberg R. A. The neu oncogene encodes an epidermal growth factor receptor-related protein. Nature. 1986 Jan 16;319(6050):226–230. doi: 10.1038/319226a0. [DOI] [PubMed] [Google Scholar]
- Böni-Schnetzler M., Pilch P. F. Mechanism of epidermal growth factor receptor autophosphorylation and high-affinity binding. Proc Natl Acad Sci U S A. 1987 Nov;84(22):7832–7836. doi: 10.1073/pnas.84.22.7832. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Decker S. J., Harris P. Effects of platelet-derived growth factor on phosphorylation of the epidermal growth factor receptor in human skin fibroblasts. J Biol Chem. 1989 Jun 5;264(16):9204–9209. [PubMed] [Google Scholar]
- Derynck R. Transforming growth factor alpha. Cell. 1988 Aug 26;54(5):593–595. doi: 10.1016/s0092-8674(88)80001-1. [DOI] [PubMed] [Google Scholar]
- Dobashi K., Davis J. G., Mikami Y., Freeman J. K., Hamuro J., Greene M. I. Characterization of a neu/c-erbB-2 protein-specific activating factor. Proc Natl Acad Sci U S A. 1991 Oct 1;88(19):8582–8586. doi: 10.1073/pnas.88.19.8582. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dobashi K., Weiner D. B., Greene M. I. Differential regulation of oncogenic and cellular p185 by serine/threonine kinases. DNA. 1989 Dec;8(10):723–732. doi: 10.1089/dna.1989.8.723. [DOI] [PubMed] [Google Scholar]
- Drebin J. A., Link V. C., Stern D. F., Weinberg R. A., Greene M. I. Down-modulation of an oncogene protein product and reversion of the transformed phenotype by monoclonal antibodies. Cell. 1985 Jul;41(3):697–706. doi: 10.1016/s0092-8674(85)80050-7. [DOI] [PubMed] [Google Scholar]
- Goldman R., Levy R. B., Peles E., Yarden Y. Heterodimerization of the erbB-1 and erbB-2 receptors in human breast carcinoma cells: a mechanism for receptor transregulation. Biochemistry. 1990 Dec 18;29(50):11024–11028. doi: 10.1021/bi00502a002. [DOI] [PubMed] [Google Scholar]
- Harris A. L., Nicholson S., Sainsbury J. R., Farndon J., Wright C. Epidermal growth factor receptors in breast cancer: association with early relapse and death, poor response to hormones and interactions with neu. J Steroid Biochem. 1989;34(1-6):123–131. doi: 10.1016/0022-4731(89)90072-1. [DOI] [PubMed] [Google Scholar]
- Honegger A. M., Schmidt A., Ullrich A., Schlessinger J. Evidence for epidermal growth factor (EGF)-induced intermolecular autophosphorylation of the EGF receptors in living cells. Mol Cell Biol. 1990 Aug;10(8):4035–4044. doi: 10.1128/mcb.10.8.4035. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kelly J. D., Haldeman B. A., Grant F. J., Murray M. J., Seifert R. A., Bowen-Pope D. F., Cooper J. A., Kazlauskas A. Platelet-derived growth factor (PDGF) stimulates PDGF receptor subunit dimerization and intersubunit trans-phosphorylation. J Biol Chem. 1991 May 15;266(14):8987–8992. [PubMed] [Google Scholar]
- Kern J. A., Schwartz D. A., Nordberg J. E., Weiner D. B., Greene M. I., Torney L., Robinson R. A. p185neu expression in human lung adenocarcinomas predicts shortened survival. Cancer Res. 1990 Aug 15;50(16):5184–5187. [PubMed] [Google Scholar]
- King C. R., Borrello I., Bellot F., Comoglio P., Schlessinger J. Egf binding to its receptor triggers a rapid tyrosine phosphorylation of the erbB-2 protein in the mammary tumor cell line SK-BR-3. EMBO J. 1988 Jun;7(6):1647–1651. doi: 10.1002/j.1460-2075.1988.tb02991.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kokai Y., Dobashi K., Weiner D. B., Myers J. N., Nowell P. C., Greene M. I. Phosphorylation process induced by epidermal growth factor alters the oncogenic and cellular neu (NGL) gene products. Proc Natl Acad Sci U S A. 1988 Aug;85(15):5389–5393. doi: 10.1073/pnas.85.15.5389. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kokai Y., Myers J. N., Wada T., Brown V. I., LeVea C. M., Davis J. G., Dobashi K., Greene M. I. Synergistic interaction of p185c-neu and the EGF receptor leads to transformation of rodent fibroblasts. Cell. 1989 Jul 28;58(2):287–292. doi: 10.1016/0092-8674(89)90843-x. [DOI] [PubMed] [Google Scholar]
- Legocki R. P., Verma D. P. Multiple immunoreplica Technique: screening for specific proteins with a series of different antibodies using one polyacrylamide gel. Anal Biochem. 1981 Mar 1;111(2):385–392. doi: 10.1016/0003-2697(81)90577-7. [DOI] [PubMed] [Google Scholar]
- Schechter A. L., Hung M. C., Vaidyanathan L., Weinberg R. A., Yang-Feng T. L., Francke U., Ullrich A., Coussens L. The neu gene: an erbB-homologous gene distinct from and unlinked to the gene encoding the EGF receptor. Science. 1985 Sep 6;229(4717):976–978. doi: 10.1126/science.2992090. [DOI] [PubMed] [Google Scholar]
- Stern D. F., Kamps M. P. EGF-stimulated tyrosine phosphorylation of p185neu: a potential model for receptor interactions. EMBO J. 1988 Apr;7(4):995–1001. doi: 10.1002/j.1460-2075.1988.tb02906.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sternberg M. J., Gullick W. J. A sequence motif in the transmembrane region of growth factor receptors with tyrosine kinase activity mediates dimerization. Protein Eng. 1990 Mar;3(4):245–248. doi: 10.1093/protein/3.4.245. [DOI] [PubMed] [Google Scholar]
- Todderud G., Wahl M. I., Rhee S. G., Carpenter G. Stimulation of phospholipase C-gamma 1 membrane association by epidermal growth factor. Science. 1990 Jul 20;249(4966):296–298. doi: 10.1126/science.2374928. [DOI] [PubMed] [Google Scholar]
- Ullrich A., Schlessinger J. Signal transduction by receptors with tyrosine kinase activity. Cell. 1990 Apr 20;61(2):203–212. doi: 10.1016/0092-8674(90)90801-k. [DOI] [PubMed] [Google Scholar]
- Wada T., Qian X. L., Greene M. I. Intermolecular association of the p185neu protein and EGF receptor modulates EGF receptor function. Cell. 1990 Jun 29;61(7):1339–1347. doi: 10.1016/0092-8674(90)90697-d. [DOI] [PubMed] [Google Scholar]
- Weiner D. B., Liu J., Cohen J. A., Williams W. V., Greene M. I. A point mutation in the neu oncogene mimics ligand induction of receptor aggregation. Nature. 1989 May 18;339(6221):230–231. doi: 10.1038/339230a0. [DOI] [PubMed] [Google Scholar]
- Williams L. T., Tremble P. M., Lavin M. F., Sunday M. E. Platelet-derived growth factor receptors form a high affinity state in membrane preparations. Kinetics and affinity cross-linking studies. J Biol Chem. 1984 Apr 25;259(8):5287–5294. [PubMed] [Google Scholar]
- Yarden Y., Schlessinger J. Epidermal growth factor induces rapid, reversible aggregation of the purified epidermal growth factor receptor. Biochemistry. 1987 Mar 10;26(5):1443–1451. doi: 10.1021/bi00379a035. [DOI] [PubMed] [Google Scholar]
- Yarden Y., Weinberg R. A. Experimental approaches to hypothetical hormones: detection of a candidate ligand of the neu protooncogene. Proc Natl Acad Sci U S A. 1989 May;86(9):3179–3183. doi: 10.1073/pnas.86.9.3179. [DOI] [PMC free article] [PubMed] [Google Scholar]