Abstract
Background
Ginseng (Panax ginseng Meyer) is a well-characterized medicinal herb listed in the classic oriental herbal dictionary as “Shin-nong-bon-cho-kyung.” Ginseng has diverse pharmacologic and therapeutic properties. Black ginseng (BG, Ginseng Radix nigra) is produced by repeatedly steaming fresh ginseng nine times. Studies of BG have shown that prolonged heat treatment enhances the antioxidant activity with increased radical scavenging activity. Several recent studies have showed the effects of BG on increased lipid profiles in mice. In this study report the effects of water and ethanol extracts of BG on hypercholesterolemia in rats. To our knowledge, this is the first time such an effect has been reported.
Methods
Experiments were conducted on male Sprague Dawley rats fed with a high-cholesterol diet supplemented with the water and ethanol extracts of BG (200 mg/kg). Their blood cholesterol levels, serum white blood cell levels, and cholesterol-metabolizing marker genes messenger RNA (mRNA) expression were determined. Liver and adipose tissues were histologically analyzed.
Results
We found that BG extracts efficiently reduced the total serum cholesterol levels, low-density lipoprotein (LDL) levels with increased food efficiency ratio and increased number of neutrophil cells. It also attenuated the key genes responsible for lipogenesis, that is, acetyl-coenzyme A (CoA) acetyltransferase 2, 3-hydroxy-3-methyl-glutaryl-CoA reductase, and sterol regulatory element-binding protein 2, at the mRNA level inside liver cells. Furthermore, the BG extract also reduced the accumulation of fat in adipose tissues, and inhibited the neutral fat content in liver cells stained with hematoxylin and eosin and oil red O.
Conclusion
Administration of BG extracts to Sprague Dawley rats fed with high-cholesterol diet ameliorated hypercholesterolemia, which was mediated via modulation of cholesterol-metabolizing marker genes. This data throw a light on BG's cardioprotective effects.
Keywords: black ginseng, hypercholesterolemia, lipogenesis, messenger RNA
1. Introduction
Hypercholesterolemia is a serious problem faced by many countries. Hypercholesterolemia is a major concern for health professionals, because it is one of the primary risk factors for the development and growth of cardiovascular diseases such as atherosclerosis and its complications, acute infarction of the myocardium or hypertension, accumulation of low-density lipoproteins (LDLs), stroke, and cerebral infarctions [1], [2]. Diet-related chronic diseases such as type 2 diabetes, cardiovascular diseases, several types of cancer, and increased morbidity and mortality can be attributed to obesity and overweight. In general, obesity occurs due to an imbalance between energy intake and energy output. An imbalance in this ratio results in accumulation of fat inside adipose tissues and elevated LDLs inside blood and other fat-accumulation organs [3]. Although allopathic drugs provide a quick relief and show rapid effects for mitigating cardiovascular anomalies, their serious side effects cannot be ignored. Therefore, increasing attention is paid to utilizing natural products for alleviation of these chronic diseases.
Ginseng and ginsenosides have been studied for a long time for their therapeutic effects on body vitality, the immune system, various types of cancer, inflammatory anomalies, nervous system disorders, reproductive system functioning, and cardiovascular diseases. Studies in the past have demonstrated ginseng's potent suppressing effects on in vitro pancreatic lipase activity as well as its ability to reduce obesity and plasma lipid profile in rodents fed with high-fat diet [4], [5]. Here our compound of interest is black ginseng (BG). BG is prepared from raw ginseng by steaming it nine times at a high temperature for approximately 3 h up to the point where it becomes black. At this point, the BG possesses better qualities than red ginseng, including better anti-inflammatory, antimetastatic, and antioxidant effects [6]. This pharmacologically active agent has been studied for many aspects previously, however, its effects on hypercholesterolemia have not yet been studied in detail. We used both water and ethanol extracts of BG in this study. Because the ethanol extract has already been identified for its therapeutic effects, we aimed to investigate the effects of the water extract. Herein, we report for the first time the amelioration of hypercholesterolemia in high-cholesterol-fed rats by the water and ethanol extracts of BG. Our results show that the water and ethanol extracts of BG effectively lowered the total serum levels of cholesterol. It also increased the food efficiency ratio (FER) as well as the differential white blood cell (WBC) count. The key gene markers for fat metabolism such as acetyl-coenzyme A (CoA) acetyltransferase 2 (ACAT2), sterol regulatory element-binding protein 2 (SERBP2), and 3-hydroxy-3-methyl-glutaryl-CoA reductase (HMG-CoAr) were also reduced by the BG extract at the messenger RNA (mRNA) levels. Moreover, the histopathological images also show reduction in fat accumulation in liver and adipose tissues. Therefore, in a nutshell, BG appears to be a promising antihypercholesterolemic agent.
2. Materials and methods
2.1. Sample preparation
Black ginseng (BG) was prepared according to the procedures described in previous reports, but with some minor modifications [7], [8]. In brief, the BG sample was ground in a cutting mill to pass through a 50-mesh sieve to obtain a fine powder and then extracted in 10-times volume of distilled water or 50% ethanol (V/W) at 80°C for 8 h in a water bath. It was then extracted again in seven-times volume of distilled water at 80°C for 8 h, which was repeated once more (third-time extraction). The total extract solution was filtered through a filter paper (medium fast: CHM F1001, CHMLAB GROUP, Barcelona, Spain). The filtrate solution was then concentrated in a low-vacuum evaporator at 60°C, and the water extract (water content 34.45%) and ethanol extract (35.55%) were prepared as the test samples.
2.2. Animals and experimental diets
Male Sprague Dawley rats, 8-wk-old, were obtained from Central Lab Animal Inc. (Seoul, Korea) and housed in standard conditions with free access to chow and water. All animals were acclimated for 1 wk before use. All in vivo experiments were conducted in accordance with internationally accepted guidelines in a specific pathogen-free facility, and the protocols were approved by the Institutional Animal Care and Use Committee of Daejeon University (Daejeon, Korea). Rats were fed a pelletized chow diet for 1 wk and according to diet and treatment with BG extracts, they were randomly divided into five groups (n = 5 in each group). The first group was given normal chow diet with only vehicle treatment. The second group was considered the negative control group and was given only cholesterol diet. The remaining three groups were given high-cholesterol diet and treated with lovastatin (20 mg/kg; LOVA), water extract of BG (200 mg/kg, BG_water), and ethanol extract of BG (200 mg/kg, BG_ethanol). The efficacious dose of water and ethanol extracts of BG (i.e., 200 mg/kg) was chosen based on the reports of previous studies indicating this dosage exerts sufficient anti-inflammatory and antioxidant effects [6]. The composition of the experimental diet was according to Paigen atherogenic rodent diet (D12336; Research Diets, Inc., NJ, USA) [9]. Rats were provided access to food and water ad libitum during the 4-wk study period.
2.3. Blood biochemical analysis
At the end of 4 wk, all rats were killed and serum samples were collected after overnight fasting. Fresh whole blood was taken directly from the heart of animals into tubes containing 18 mg of EDTA (for whole-blood hematology) and sodium heparin (for plasma fraction). An automatic hematology analyzer (Sysmex XE-2100D; Sysmex Corporation, Kobe, Japan) was used to perform a complete blood cell count on each blood sample, which included obtaining platelet counts, WBC counts, and WBC differential counts. Total cholesterol (TC), high-density lipoproteins (HDLs), LDLs, triglycerides, and creatinine levels were analyzed using the enzymatic method (FUJI DRI-CHEM 4000i, FUJIFILM, Tokyo, Japan).
2.4. Histological analysis
The liver, kidneys, and adipose tissues were fixed overnight in 10% formalin solution, dehydrated, embedded in paraffin, and cut into 5-μm sections. Cross sections of these tissues were stained with hematoxylin and eosin (H&E) and oil red O.
2.5. RNA extraction and real-time polymerase chain reaction for liver tissues
For the mRNA expression of ACAT2, SERBP2, and HMG-CoA, total RNA was extracted from the liver tissues using TRIzol reagent (Invitrogen, Carlsbad, CA, USA) following the manufacturer's instructions and as previously described, but with little modifications [10]. In brief, 1 mL TRIzol reagent was added to 100 mg of the liver sample and the tissues were homogenized using a power homogenizer. The samples were then incubated at room temperature for 5 min to permit complete disassociation of nuclear proteins. The remaining steps were carried out as described previously [10].
2.6. Statistical analysis
The results were presented as the mean ± standard error of the mean. One-way analysis of variance followed by Dunnett t test was used for statistical analysis. SAS 9.3 (SAS institute Inc, Cary, NC, USA) was used for statistical analysis. All p values < 0.01 and < 0.05 were considered statistically significant.
3. Results
3.1. Effects of BG extract on body weight, FER, and organ weights
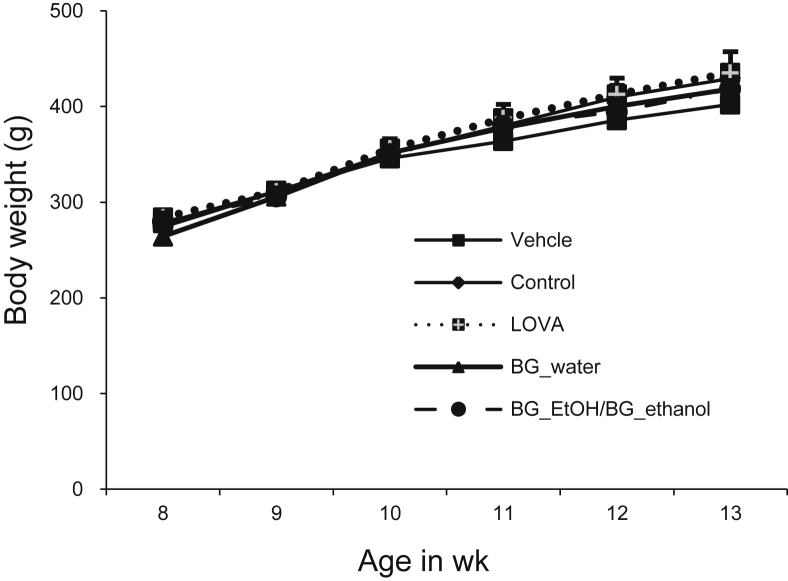
The effects of BG during the experimental procedure are shown in Fig. 1 and Table 1, Table 2. Upon supplementing high-cholesterol diet to the animals, there is an increase in body weight from approximately 250 g to 400 g, which decreases following treatment with lovastatin and water extract of BG. The FER increases significantly when using the water extract of BG up to 23%, indicating that BG extracts have positively increased the energy output in accordance with the input content of feed. The mean liver and kidney weights are significantly reduced following treatment with BG extracts.
Fig. 1.
Effects of black ginseng (BG) supplementation on body weight. Experimental rats were fed with normal chow rat diet, high-cholesterol diet, high-cholesterol diet supplemented with lovastatin (LOVA), high-cholesterol diet supplemented with water extract of black ginseng (200 mg/kg, BG_water), and ethanol extract of black ginseng (200 mg/kg, BG_ethanol) for 4 wk. Weight gain after 4 wk is expressed in grams. Values are expressed as mean ± standard deviation (n = 4). EtOH, ethanol.
Table 1.
Food efficiency ratio
| Group | Food intakes (g/d) | Body weight gain (g/d) | Food efficiency ratio (%) |
|---|---|---|---|
| Vehicle | 19.95 | 4.44 ± 0.15 | 22.23 ± 0.75 |
| Control | 23.51 | 5.55 ± 0.34 | 23.62 ± 1.43 |
| Lovastatin (20 mg/kg) | 23.58 | 5.40 ± 0.56 | 22.89 ± 2.38 |
| BG_water (200 mg/kg) | 23.38 | 5.51 ± 0.72 | 23.59 ± 3.07 |
| BG_EtOH (200 mg/kg) | 22.47 | 4.94 ± 0.24 | 21.98 ± 1.05 |
Sprague Dawley rats were supplemented with a high-fat diet and black ginseng (BG) extracts for 4 wk and after completion of the experimental period, the food efficiency ratio (FER) was calculated as follows: FER = (food intake/body weight gain) × 100.
EtOH, ethanol.
Table 2.
Mean liver and kidney weights
| Group | Liver (mean ± SEM) | Kidney (mean ± SEM) |
|---|---|---|
| Vehicle | 11.75 ± 0.48 | 2.80 ± 0.11 |
| Control | 22.06 ± 0.49 | 2.96 ± 0.12 |
| Lovastatin (20 mg/kg) | 22.07 ± 0.78 | 2.87 ± 0.04 |
| BG_water (200 mg/kg) | 20.40 ± 1.44 | 2.66 ± 0.11 |
| BG_EtOH (200 mg/kg) | 19.25 ± 0.36 * | 2.50 ± 0.07 * |
The values presented were obtained after the experimental 13-wk period. The dry weight of the liver and kidney is significantly (* p < 0.01) reduced by treatment with the ethanol (EtOH) extract of black ginseng (BG). Values are presented as mean ± standard deviation (n = 4).
SEM, standard error of the mean.
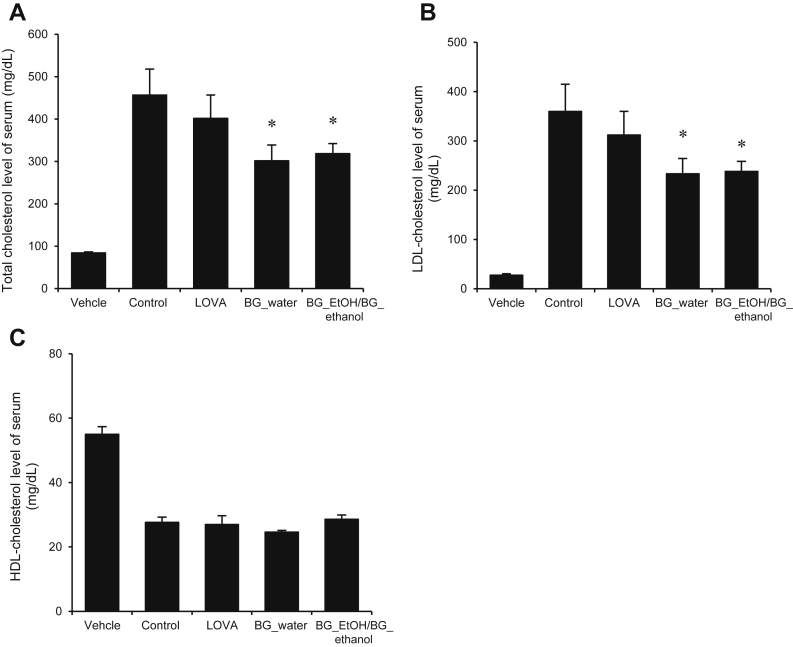
3.2. BG extracts lowered the levels of TC and LDL in serum
The TC and LDL levels are the markers for increased accumulation of harmful cholesterol in the body. As can be seen from Figs. 2A and 2B, BG extract has significantly lowered the levels of TC and LDL cholesterol from the serum, indicating that this extract exhibits outstanding antihypercholesterolemic properties. However, its effect on the HDL level was insignificant (Fig. 2C).
Fig. 2.
Attenuation in the levels of total cholesterol and low-density lipoproteins (LDLs) by the black ginseng extract. Rats were fed with normal chow diet (vehicle), high-cholesterol diet (negative control), high-cholesterol diet with lovastatin (LOVA), high-cholesterol diet with water extract of black ginseng (200 mg/kg, BG_water), and high-cholesterol diet with ethanol extract of black ginseng (200 mg/kg, BG_ethanol). (A, B) The total cholesterol levels and LDLs cholesterol levels of serum are significantly decreased with the water and ethanol extracts of BG. Bar graph is mean ± standard deviation. (C) The serum high-density lipoprotein (HDL) levels remain unchanged. In panels A and B, * p values < 0.05 are considered statistically significant when compared with control only. BG, black ginseng.
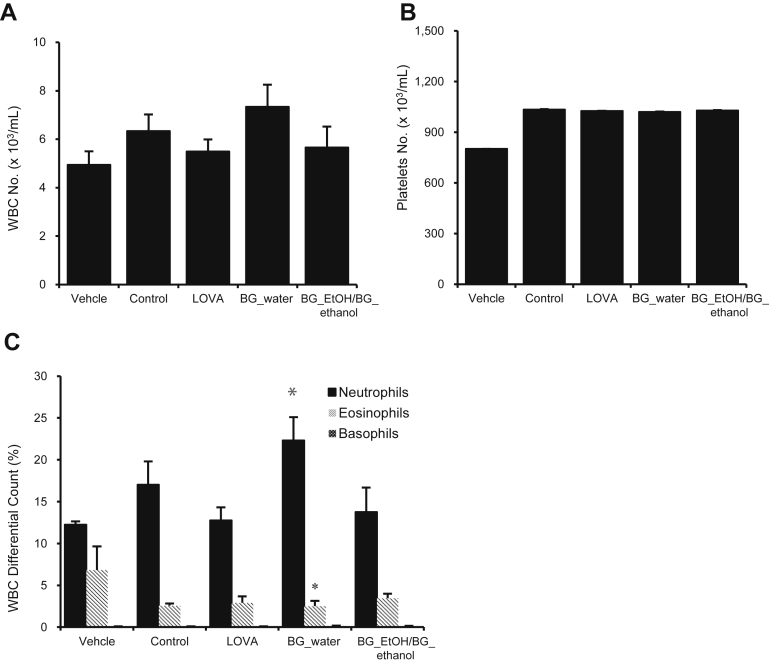
3.3. Blood analysis
As shown in Fig. 3A, the BG extract increased the levels of whole WBC counts, compared with the negative control. However, no change was observed in the levels of platelet count in any of the groups except the vehicle-treated group (Fig. 3B). In the case of differential WBC count (Fig. 3C), there was a remarkable increase in the levels of neutrophils in the group treated with the water extract of BG, compared with the negative control group.
Fig. 3.
Elevation of differential white blood cell (WBC) count by black ginseng (BG) extracts. Rats were experimentally fed a normal chow diet and high-cholesterol diet supplemented with lovastatin (LOVA), high-cholesterol diet with water extract of black ginseng (200 mg/kg, BG_water), and high-cholesterol diet with ethanol extract of black ginseng (200 mg/kg, BG_ethanol). On the 4th wk, the blood was withdrawn from rats and analyzed. (A, B) The number of platelets and whole WBC count showed no statistical significance. (C) WBC differential count, however, showed a significant increase in neutrophils by percentage. Bar diagram is mean ± standard deviation (n = 4). *p < 0.05 is considered statistically significant.
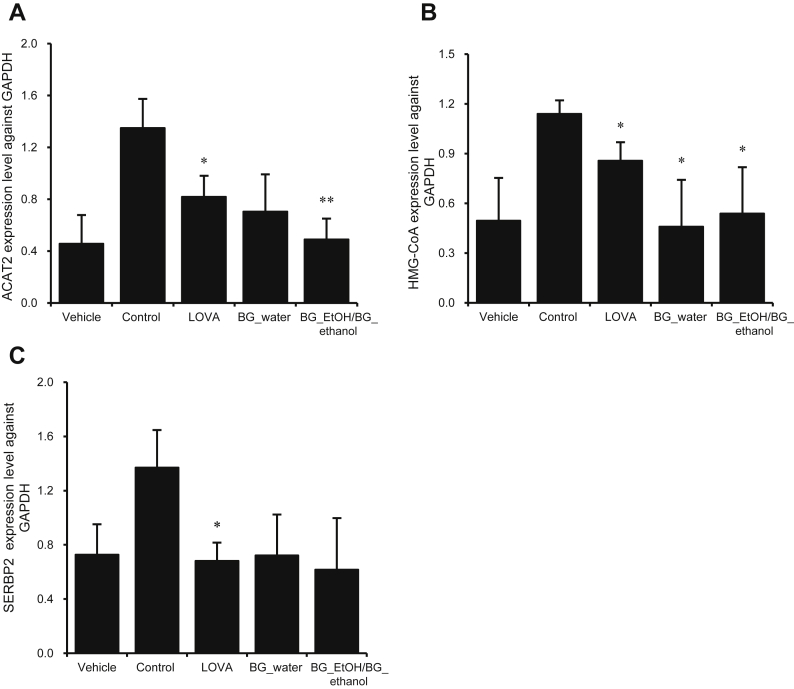
3.4. Attenuation of ACAT2, HMG-CoAr, and SERBP2 mRNA levels by BG extracts
ACAT2, SERBP2, and HMG-CoAr are considered the hallmark genes and regulators for cholesterol metabolism inside liver cells. Their respective levels indicate the amount of cholesterol being formed and stored inside hepatocytes. From Figs. 4A–4C, it can be seen that the BG extracts significantly diminished the levels of these aforementioned genes when compared with the housekeeping glyceraldehyde-3-phosphate dehydrogenase (GAPDH) gene.
Fig. 4.
Inhibition of acetyl-coenzyme A (CoA) acetyltransferase 2 (ACAT2), 3-hydroxy-3-methyl-glutaryl-CoA (HMG-CoA), and sterol regulatory element-binding protein 2 (SERBP2) expressions at the messenger RNA level. For evaluating the expressions of these three cholesterol metabolic markers, total RNA was extracted from the liver tissue using TRIzol reagent following manufacturer's instructions. Reverse transcription was carried out in a thermocycler (Biometra). For evaluation of expression of genes, complementary DNA was analyzed by BIO-RAD CFX96 real-time system. (A–C) Significant decrease in these gene markers for cholesterol is observed. Bar graphs are representative of four independent experiments. Values are mean ± standard deviation. * p < 0.05 and ** p < 0.01 are considered statistically significant when compared with control only. GAPDH, glyceraldehyde-3-phosphate dehydrogenase.
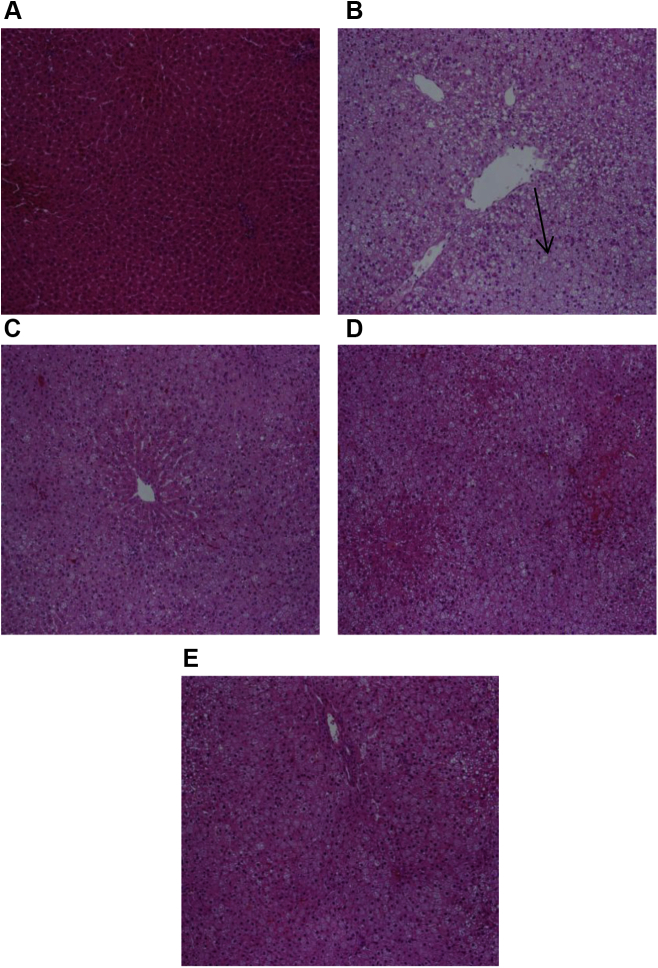
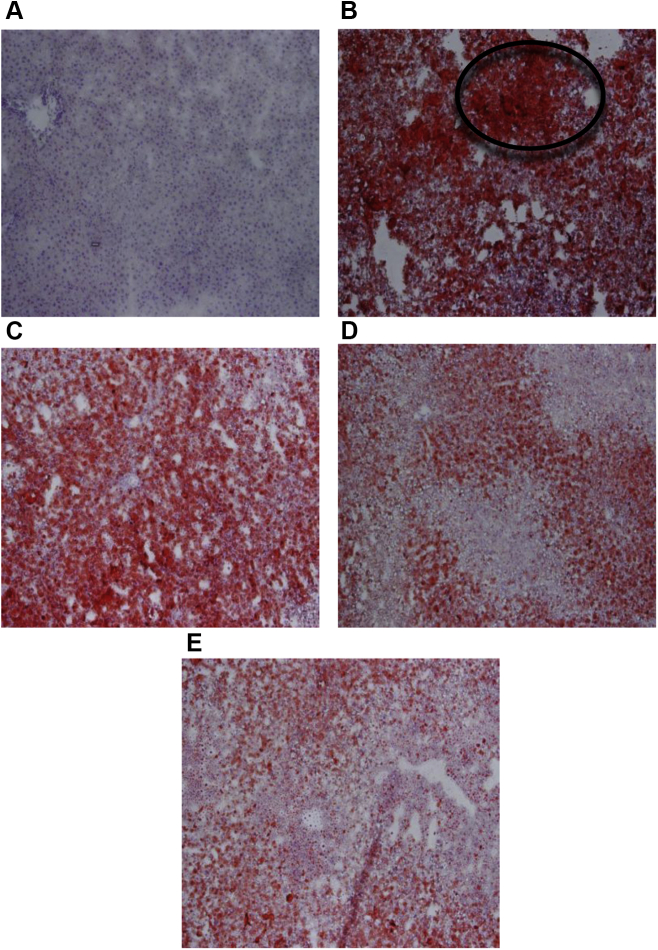
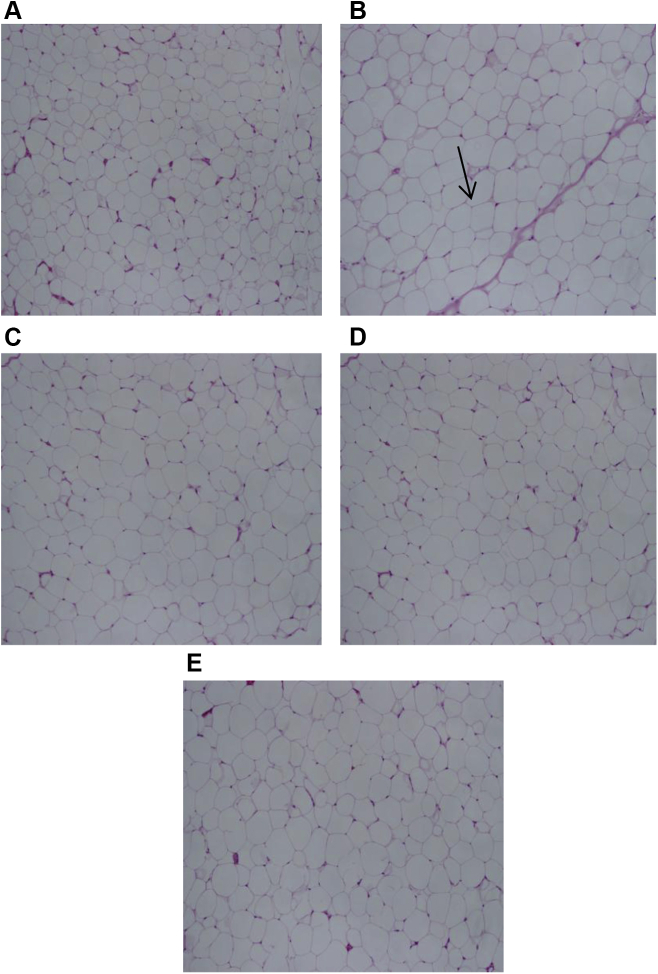
3.5. Histological effects of BG extracts on liver and adipose tissue
The BG extract attenuated the accumulation of cholesterol in hepatocytes, compared with negative control cells (Fig. 5A). Similarly, following oil red O staining for liver sections, we observed that the negative control showed intense excessive red staining, compared with that of the BG extracts-treated groups in which the staining intensity was more close to that of the vehicle-treated group (Fig. 6). Using H&E staining for adipose tissue (Fig. 7), there is an increase in the size of cells in the negative control group, compared with that of the BG extracts-treated group in which the size of cells and accumulation of cholesterol are close to the normal vehicle-treated group. These histological data are in close conformity with BG's antihypercholesterolemic effects.
Fig. 5.
Histological changes in liver brought about following treatment with black ginseng (BG) extracts. “A” refers to normal vehicle-treated tissue, “B” refers to negative control tissue (high-cholesterol-treated rats), “C” refers to lovastatin with high-cholesterol-diet-treated rats (LOVA), “D” refers to water extract of black ginseng-treated and high-cholesterol diet rats (200 mg/kg, BG_water), and “E” refers to black ginseng ethanol-treated and high-cholesterol-fed diet rats (200 mg/kg, BG_ethanol). The fat accumulation inside the BG-treated groups is less than that of the negative control group. Arrow in image b indicates the accumulation of fat in hepatocytes.
Fig. 6.
Histological changes in liver brought about by Black Ginseng (BG) extracts. The liver parenchyma was stained with Oil Red O. A–E images refer to the above elaborated treatment groups in Fig. 5. The intensity of red color in BG treated groups is less as compared to negative control group. Encircled portion indicates the fat accumulated stained intensive red areas.
Fig. 7.
Histological changes in adipose tissue brought about by Black Ginseng (BG) extracts. The adipose tissue was stained with hematoxylin & eosin (H&E). A–E images correspond to the above mentioned description in Fig. 5. The increased size of adipose tissue corresponds to the increased fat accumulation due to high fat diet. Arrow indicates the increased cell size due to fat accumulation.
4. Discussion
The risk of accumulation of excessive cholesterol inside the body and cardiovascular diseases is a serious concern for health professionals all over the world in the 21st century [11]. Ginseng has long been used for the treatment of heart failure and to protect against tissue damage under cellular and environmental stress without any side effects [5]. Therefore, in nutshell, several studies were carried out on the cardioprotective effects of ginseng. However, little is known about the effect of BG on hypercholesterolemia.
Although one previous report has indicated the antiobesity effects of ethanol extract of BG in mice [7], we have demonstrated, in this study, the antihypercholesterolemic effects of water and ethanol extracts of BG in rats with particular explanation in relation to mRNA expressions of genes that are specific to cholesterol metabolism and histological staining of the adipose tissue. In this study, we found that both water and ethanol extracts of BG decreased the TC and LDL cholesterol levels of serum (Figs. 2A and 2B). However, the HDL cholesterol levels of serum remain unaffected. It indicates that BG extract can efficaciously reduce the levels of “bad cholesterol” (LDL cholesterol) inside a rat model of hypercholesterolemia. With the administration of BG extract at a dose of 200 mg/kg, there was an observable increase in body weight gain and FER (Fig. 1; Table 1). The increase in FER indicates that the BG extract helped in metabolizing energy and increasing the output. With regard to the white blood differential count, there was a remarkable increase in the number of neutrophils with BG extract (Fig. 3C). Rats treated with the ethanol extract of BG showed a significant decrease in mean liver and kidney weights (Table 2). The increase in weights of the liver and kidneys in this case would imply the accumulation of LDLs inside the fat-accumulating cells, which was ameliorated by the BG extract.
ACAT2 is the most preferred target for studies related to hypercholesterolemia and coronary heart diseases. ACAT2 deletion causes lowering of lipoproteins inside the body [12], [13]. In this study, we found the ACAT2 expression was significantly downregulated in murine hepatocytes when compared with GAPDH as a housekeeping gene (Fig. 4A). HMG-CoAr is a rate-limiting step for the cholesterol synthesis pathway [14], [15]. This enzyme is also a potent target for lovastatin, a predominant drug for atherosclerosis and hyperlipidemia; in fact, lovastatin is also called “HMG-CoA reductase inhibitor” [16]. The BG-treated groups in our experiments represent a significant lowering of the mRNA expressions of HMG-CoAr inside liver cells (Fig. 4B). There is a pair of transcription factors termed “SREBPs” that are important for biosynthesis of cholesterol and its uptake from plasma LDL [17]. Research has suggested that SREBP is increased in normal conditions; however, with lovastatin treatment the SERBP2 levels are degraded [18]. Similarly, our findings have shown that BG extracts reduced the levels of SREBP mRNA inside the liver (Fig. 4C).
Prolonged access of mice to a high-fat diet culminates in increased liver mass, hepatic steatosis, and hyperlipidemia [7], [19]. The H&E staining for liver parenchyma showed less alteration in the central vein and sinusoidal spaces compared with that of positive control hepatocytes where there is increase in the size of cells due to fat accumulation, indicating steatohepatitis (Fig. 5). The oil red O staining of adipose cells in the liver is also less intensified for positive control cells with maximal accumulation of cholesterol, indicating steatosis in liver cells (Fig. 6). For the H&E staining of adipose tissues, it is clearly demonstrated that the size of fat cells in the BG-treated groups is near to normal when compared with that of positive control cells in which fat cells are highly accumulated with cholesterol as depicted by increase in the size of cells (Fig. 7).
In conclusion, the administration of BG extracts in Sprague Dawley rats with high-cholesterol diet lowered the LDLs cholesterol levels, caused an elevation in the serum triglyceride levels, and increased the food efficiency. Moreover, with respect to the liver metabolic marker genes, the mRNA expressions of ACAT2, SREBP 2, and HMG-CoAr were inhibited by ethanol and water extracts of BG. The histological changes brought about by the high-cholesterol diet in liver and adipose tissues were reverted by the BG extracts, as confirmed by the H&E and oil red O staining results. From this first report on the hypercholesterolemic effect of ethanol and water extracts of BG in rats, it can be concluded that these extracts may be studied in the future as a functional food for various cardiovascular-related anomalies (Fig. 6, Fig. 7).
Conflicts of interest
The authors declare that they have no competing interests.
Acknowledgments
This work was supported by Development of High-Value-Added Products for IPET (Institute of Planning and Evaluation for Technology in Food, Agriculture Forestry and Fisheries), Korea in 2014.
Contributor Information
Hyun-Kyoung Kim, Email: kimhk4@empas.com.
Man-Hee Rhee, Email: rheemh@knu.ac.kr.
References
- 1.Gielen S., Landmesser U. The year in cardiology 2013: cardiovascular disease prevention. Eur Heart J. 2014;35:307–312. doi: 10.1093/eurheartj/eht551. [DOI] [PubMed] [Google Scholar]
- 2.Tunstall-Pedoe H., Vanuzzo D., Hobbs M., Mähönen M., Cepaitis Z., Kuulasmaa K., Keil U. Estimation of contribution of changes in coronary care to improving survival, event rates, and coronary heart disease mortality across the WHO MONICA Project populations. Lancet. 2000;355:688–700. doi: 10.1016/s0140-6736(99)11181-4. [DOI] [PubMed] [Google Scholar]
- 3.Hill J.O., Wyatt H.R., Peters J.C. Energy balance and obesity. Circulation. 2012;126:126–132. doi: 10.1161/CIRCULATIONAHA.111.087213. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Kim J.H. Cardiovascular diseases and Panax ginseng: a review on molecular mechanisms and medical applications. J Ginseng Res. 2012;36:16–26. doi: 10.5142/jgr.2012.36.1.16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Lee C.H., Kim J.H. A review on the medicinal potentials of ginseng and ginsenosides on cardiovascular diseases. J Ginseng Res. 2014;38:161–166. doi: 10.1016/j.jgr.2014.03.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Lee M.R., Yun B.S., In O.H., Sung C.K. Comparative study of Korean white, red, and black ginseng extract on cholinesterase inhibitory activity and cholinergic function. J Ginseng Res. 2011;35:421–428. doi: 10.5142/jgr.2011.35.4.421. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Lee M.R., Kim B.C., Kim R., Oh H.I., Kim H.K., Choi K.J., Sung C.K. Anti-obesity effects of black ginseng extract in high fat diet-fed mice. J Ginseng Res. 2013;37:308–349. doi: 10.5142/jgr.2013.37.308. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Lee M.R., Yun B.S., Sung C.K. Comparative study of white and steamed black Panax ginseng, P. quinquefolium, and P. notoginseng on cholinesterase inhibitory and antioxidative activity. J Ginseng Res. 2012;36:93–101. doi: 10.5142/jgr.2012.36.1.93. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Paigen B., Morrow A., Brandon C., Mitchell D., Holmes P. Variation in susceptibility to atherosclerosis among inbred strains of mice. Atherosclerosis. 1985;57:65–73. doi: 10.1016/0021-9150(85)90138-8. [DOI] [PubMed] [Google Scholar]
- 10.Im E.J., Yayeh T., Park S.J., Kim S.H., Goo Y.K., Hong S.B., Son Y.M., Kim S.D., Rhee M.H. Antiatherosclerotic effect of Korean red ginseng extract involves regulator of G-protein signaling 5. Evid Based Complement Alternat Med. 2014;2014:985174. doi: 10.1155/2014/985174. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Qi Y., Liu J., Ma C., Wang W., Liu X., Wang M., Lv Q., Sun J., Liu J., Li Y. Association between cholesterol synthesis/absorption markers and effects of cholesterol lowering by atorvastatin among patients with high risk of coronary heart disease. J Lipid Res. 2013;54:3189–3197. doi: 10.1194/jlr.P040360. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Parini P., Davis M., Lada A.T., Erickson S.K., Wright T.L., Gustafsson U., Sahlin S., Einarsson C., Eriksson M., Angelin B. ACAT2 is localized to hepatocytes and is the major cholesterol-esterifying enzyme in human liver. Circulation. 2004;110:2017–2023. doi: 10.1161/01.CIR.0000143163.76212.0B. [DOI] [PubMed] [Google Scholar]
- 13.Parini P., Jiang Z.Y., Einarsson C., Eggertsen G., Zhang S.D., Rudel L.L., Han T.Q., Eriksson M. ACAT2 and human hepatic cholesterol metabolism: identification of important gender-related differences in normolipidemic, non-obese Chinese patients. Atherosclerosis. 2009;207:266–271. doi: 10.1016/j.atherosclerosis.2009.04.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Haines B.E., Steussy C.N., Stauffacher C.V., Wiest O. Molecular modeling of the reaction pathway and hydride transfer reactions of HMG-CoA reductase. Biochemistry. 2012;51:7983–7995. doi: 10.1021/bi3008593. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Singh D.K., Banerjee S., Porter T.D. Green and black tea extracts inhibit HMG-CoA reductase and activate AMP kinase to decrease cholesterol synthesis in hepatoma cells. J Nutr Biochem. 2009;20:816–822. doi: 10.1016/j.jnutbio.2008.07.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Tobert J.A. Lovastatin and beyond: the history of the HMG-CoA reductase inhibitors. Nat Rev Drug Discov. 2003;2:517–526. doi: 10.1038/nrd1112. [DOI] [PubMed] [Google Scholar]
- 17.Shimano H. Sterol regulatory element-binding proteins (SREBPs): transcriptional regulators of lipid synthetic genes. Prog Lipid Res. 2001;40:439–452. doi: 10.1016/s0163-7827(01)00010-8. [DOI] [PubMed] [Google Scholar]
- 18.Shimano H., Shimomura I., Hammer R.E., Herz J., Goldstein J.L., Brown M.S., Horton J.D. Elevated levels of SREBP-2 and cholesterol synthesis in livers of mice homozygous for a targeted disruption of the SREBP-1 gene. J Clin Invest. 1997;100:2115–2124. doi: 10.1172/JCI119746. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.VanSaun M.N., Lee I.K., Washington M.K., Matrisian L., Gorden D.L. High fat diet induced hepatic steatosis establishes a permissive microenvironment for colorectal metastases and promotes primary dysplasia in a murine model. Am J Pathol. 2009;175:355–364. doi: 10.2353/ajpath.2009.080703. [DOI] [PMC free article] [PubMed] [Google Scholar]