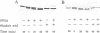Abstract
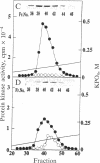
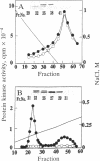
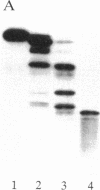
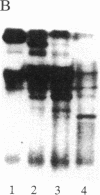
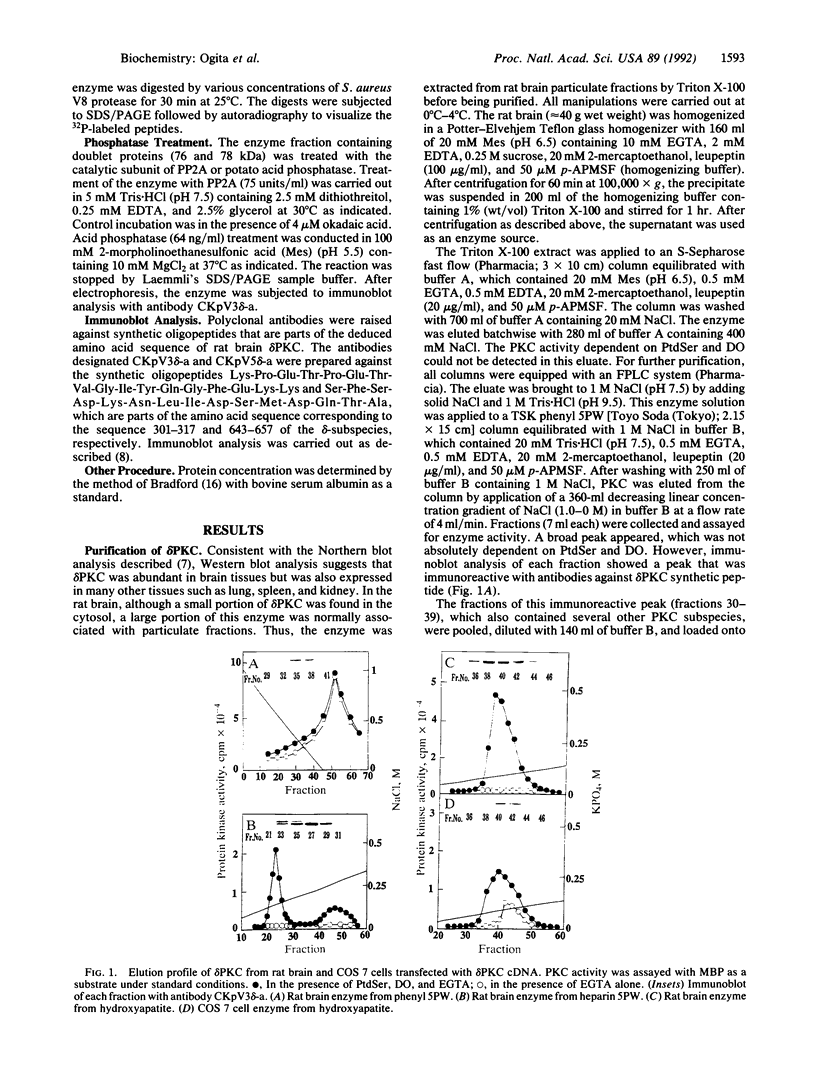
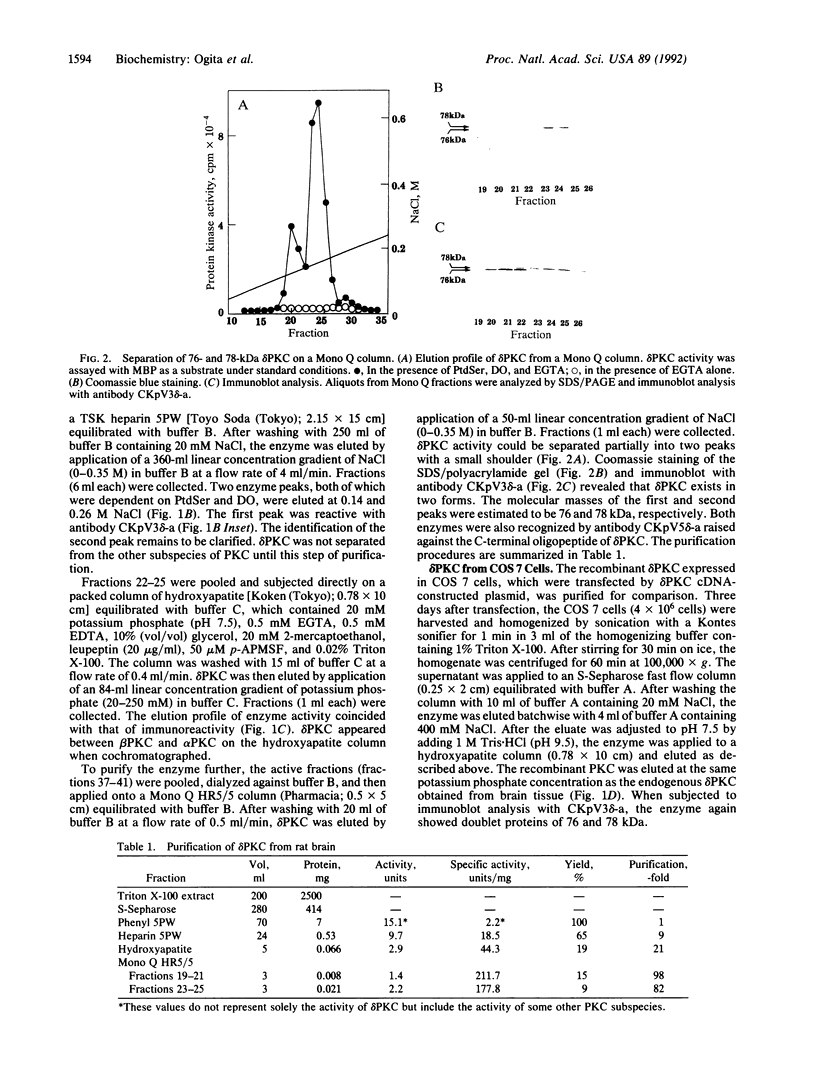
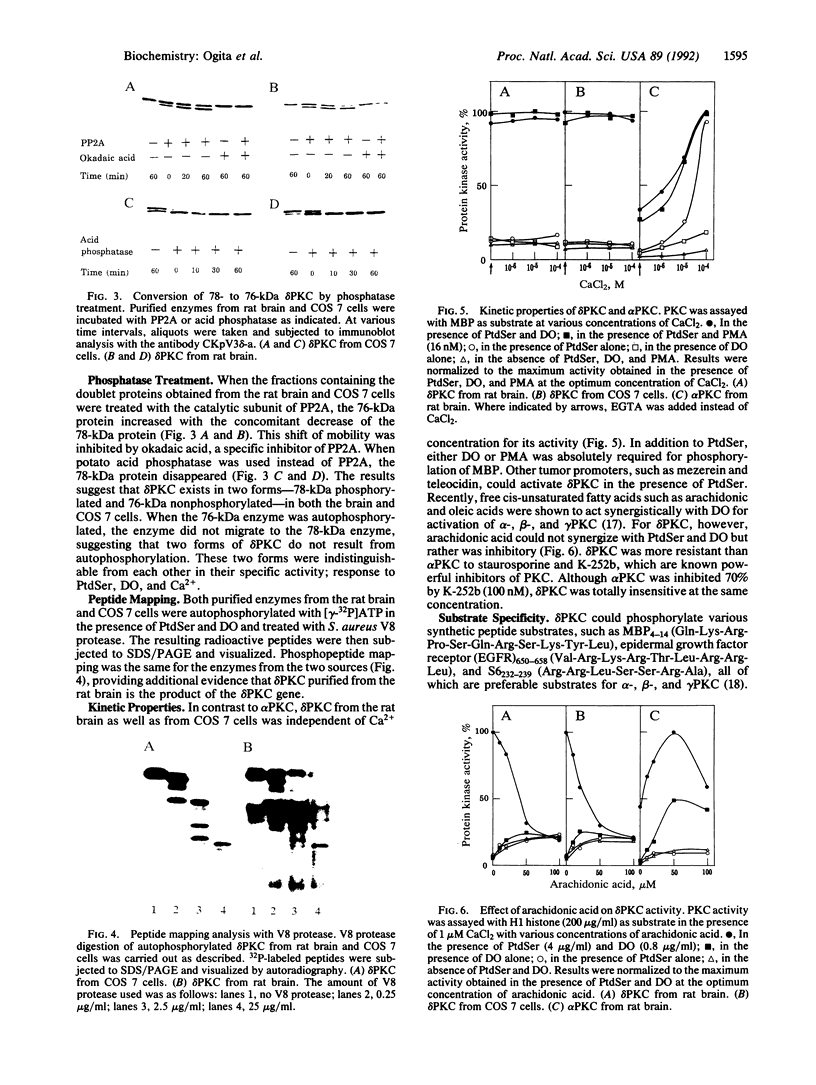
The delta-subspecies of protein kinase C (delta PKC) was purified to near homogeneity from the Triton X-100 extract of the rat brain particulate fraction by successive chromatographies on S-Sepharose fast flow, phenyl 5PW, heparin 5PW, hydroxyapatite, and Mono Q columns. The purified enzyme was a doublet with molecular masses of 78 and 76 kDa on SDS/PAGE. The doublet proteins were separated partially by Mono Q column chromatography; both were recognized by the antibodies raised against synthetic oligopeptides, parts of the deduced amino acid sequence of the rat delta PKC. Protein phosphatase 2A treatment suggested that the 78-kDa protein was a phosphorylated form of the 76-kDa protein. To confirm the structural and genetic identity of the doublet proteins, delta PKC was expressed in COS 7 cells by transfecting its cDNA-constructed plasmid and was purified for comparison. This recombinant enzyme was also a doublet. The enzymes isolated from the brain and COS 7 cells showed identical reactivities with delta PKC-specific antibodies, chromatographic behaviors, and V8 protease peptide mappings. In addition, these two enzyme preparations were indistinguishable from each other in their responses to phosphatidylserine, diacylglycerol, phorbol esters, free fatty acids, Ca2+, and enzyme inhibitors. Comparison was also made between the enzymologic properties of delta PKC and alpha PKC, which were distinctly different from each other.
Full text
PDF

Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Aihara H., Asaoka Y., Yoshida K., Nishizuka Y. Sustained activation of protein kinase C is essential to HL-60 cell differentiation to macrophage. Proc Natl Acad Sci U S A. 1991 Dec 15;88(24):11062–11066. doi: 10.1073/pnas.88.24.11062. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Asaoka Y., Oka M., Yoshida K., Nishizuka Y. Metabolic rate of membrane-permeant diacylglycerol and its relation to human resting T-lymphocyte activation. Proc Natl Acad Sci U S A. 1991 Oct 1;88(19):8681–8685. doi: 10.1073/pnas.88.19.8681. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bacher N., Zisman Y., Berent E., Livneh E. Isolation and characterization of PKC-L, a new member of the protein kinase C-related gene family specifically expressed in lung, skin, and heart. Mol Cell Biol. 1991 Jan;11(1):126–133. doi: 10.1128/mcb.11.1.126. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bradford M. M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem. 1976 May 7;72:248–254. doi: 10.1016/0003-2697(76)90527-3. [DOI] [PubMed] [Google Scholar]
- Exton J. H. Signaling through phosphatidylcholine breakdown. J Biol Chem. 1990 Jan 5;265(1):1–4. [PubMed] [Google Scholar]
- Graff J. M., Stumpo D. J., Blackshear P. J. Characterization of the phosphorylation sites in the chicken and bovine myristoylated alanine-rich C kinase substrate protein, a prominent cellular substrate for protein kinase C. J Biol Chem. 1989 Jul 15;264(20):11912–11919. [PubMed] [Google Scholar]
- Huang K. P., Nakabayashi H., Huang F. L. Isozymic forms of rat brain Ca2+-activated and phospholipid-dependent protein kinase. Proc Natl Acad Sci U S A. 1986 Nov;83(22):8535–8539. doi: 10.1073/pnas.83.22.8535. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kikkawa U., Ono Y., Ogita K., Fujii T., Asaoka Y., Sekiguchi K., Kosaka Y., Igarashi K., Nishizuka Y. Identification of the structures of multiple subspecies of protein kinase C expressed in rat brain. FEBS Lett. 1987 Jun 15;217(2):227–231. doi: 10.1016/0014-5793(87)80668-3. [DOI] [PubMed] [Google Scholar]
- Konno Y., Ohno S., Akita Y., Kawasaki H., Suzuki K. Enzymatic properties of a novel phorbol ester receptor/protein kinase, nPKC. J Biochem. 1989 Oct;106(4):673–678. doi: 10.1093/oxfordjournals.jbchem.a122915. [DOI] [PubMed] [Google Scholar]
- Leibersperger H., Gschwendt M., Marks F. Purification and characterization of a calcium-unresponsive, phorbol ester/phospholipid-activated protein kinase from porcine spleen. J Biol Chem. 1990 Sep 25;265(27):16108–16115. [PubMed] [Google Scholar]
- Mizuno K., Kubo K., Saido T. C., Akita Y., Osada S., Kuroki T., Ohno S., Suzuki K. Structure and properties of a ubiquitously expressed protein kinase C, nPKC delta. Eur J Biochem. 1991 Dec 18;202(3):931–940. doi: 10.1111/j.1432-1033.1991.tb16453.x. [DOI] [PubMed] [Google Scholar]
- Nishizuka Y. The molecular heterogeneity of protein kinase C and its implications for cellular regulation. Nature. 1988 Aug 25;334(6184):661–665. doi: 10.1038/334661a0. [DOI] [PubMed] [Google Scholar]
- Nishizuka Y. Turnover of inositol phospholipids and signal transduction. Science. 1984 Sep 21;225(4668):1365–1370. doi: 10.1126/science.6147898. [DOI] [PubMed] [Google Scholar]
- Ogita K., Miyamoto S., Koide H., Iwai T., Oka M., Ando K., Kishimoto A., Ikeda K., Fukami Y., Nishizuka Y. Protein kinase C in Saccharomyces cerevisiae: comparison with the mammalian enzyme. Proc Natl Acad Sci U S A. 1990 Jul;87(13):5011–5015. doi: 10.1073/pnas.87.13.5011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ohno S., Akita Y., Konno Y., Imajoh S., Suzuki K. A novel phorbol ester receptor/protein kinase, nPKC, distantly related to the protein kinase C family. Cell. 1988 Jun 3;53(5):731–741. doi: 10.1016/0092-8674(88)90091-8. [DOI] [PubMed] [Google Scholar]
- Ono Y., Fujii T., Ogita K., Kikkawa U., Igarashi K., Nishizuka Y. Protein kinase C zeta subspecies from rat brain: its structure, expression, and properties. Proc Natl Acad Sci U S A. 1989 May;86(9):3099–3103. doi: 10.1073/pnas.86.9.3099. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ono Y., Fujii T., Ogita K., Kikkawa U., Igarashi K., Nishizuka Y. The structure, expression, and properties of additional members of the protein kinase C family. J Biol Chem. 1988 May 15;263(14):6927–6932. [PubMed] [Google Scholar]
- Osada S., Mizuno K., Saido T. C., Akita Y., Suzuki K., Kuroki T., Ohno S. A phorbol ester receptor/protein kinase, nPKC eta, a new member of the protein kinase C family predominantly expressed in lung and skin. J Biol Chem. 1990 Dec 25;265(36):22434–22440. [PubMed] [Google Scholar]
- Pelech S. L., Sanghera J. S., Daya-Makin M. Protein kinase cascades in meiotic and mitotic cell cycle control. Biochem Cell Biol. 1990 Dec;68(12):1297–1330. doi: 10.1139/o90-194. [DOI] [PubMed] [Google Scholar]
- Schaap D., Parker P. J., Bristol A., Kriz R., Knopf J. Unique substrate specificity and regulatory properties of PKC-epsilon: a rationale for diversity. FEBS Lett. 1989 Jan 30;243(2):351–357. doi: 10.1016/0014-5793(89)80160-7. [DOI] [PubMed] [Google Scholar]
- Sekiguchi K., Tsukuda M., Ase K., Kikkawa U., Nishizuka Y. Mode of activation and kinetic properties of three distinct forms of protein kinase C from rat brain. J Biochem. 1988 May;103(5):759–765. doi: 10.1093/oxfordjournals.jbchem.a122343. [DOI] [PubMed] [Google Scholar]
- Shinomura T., Asaoka Y., Oka M., Yoshida K., Nishizuka Y. Synergistic action of diacylglycerol and unsaturated fatty acid for protein kinase C activation: its possible implications. Proc Natl Acad Sci U S A. 1991 Jun 15;88(12):5149–5153. doi: 10.1073/pnas.88.12.5149. [DOI] [PMC free article] [PubMed] [Google Scholar]