Abstract
Oxidized LDL induces changes in several facets of the immune system, although the relationships between these facets and their contributions to atherogenesis have yet to be fully elucidated. A report in this issue of the JCI provides a novel demonstration of the adaptive immune system influencing the production of natural antibodies. The results demonstrate that injection of malondialdehyde-modified LDL promotes a Th2 response that in turn increases the titers of the natural antibody T15/EO6, which recognizes the oxidized phospholipid POVPC. Atherosclerotic lesion size in LDL receptor–deficient mice is reduced as a consequence of the increase in natural antibody titers, and IL-5 is identified as the link between the adaptive and natural immune systems.
Several decades ago it was proposed that oxidative modification of LDL promoted the formation of atherosclerotic lesions (1). Early studies focused on the ability of oxidized LDL (OxLDL) to bind macrophage scavenger receptors as a mechanism for the formation of lipid-laden foam cells that are present throughout the evolution of atherosclerotic lesions. Subsequent studies have demonstrated that oxidatively modified LDL is a complex entity with a range of atherogenic properties that affect macrophages. These include promotion of macrophage recruitment and retention, lipid loading via multiple receptor types, induction of inflammatory genes, and cell viability (2).
Although the role of macrophages initially took center stage in atherosclerosis research, there is a growing literature on the interaction of OxLDL with both the cell-mediated and humoral components of the adaptive immune system. Cell-mediated responses were demonstrated following OxLDL stimulation of T lymphocyte clones isolated from atherosclerotic lesions (3). In addition, many studies have demonstrated humoral immune responses in reaction to the presence of autoantibodies to specific forms of OxLDL, particularly malondialdehyde-modified LDL (MDA-LDL) (4). Although autoantibodies to OxLDL are consistently found in hyperlipidemic animals and humans, their impact on the atherosclerotic process is unclear. Higher titers of these autoantibodies often positively correlate with severity of atherosclerosis, which led to conjecture regarding their involvement in the disease process. For example, titers of autoantibodies to selected forms of OxLDL have been shown to predict the severity of human carotid atherosclerosis (5). To determine whether these correlations were causal, autoantibody titers were increased by immunizing hyperlipidemic rabbits and mice with different forms of modified LDL, which produced an unanticipated result: immunization with MDA-LDL actually decreased the size of atherosclerotic lesions in Watanabe heritable hyperlipidemic rabbits (6). Subsequent studies in rabbits and mice have shown some anomalies in the antibody isotypes developed during immunization but have consistently demonstrated a decrease in atherosclerotic lesion size in response to immunization with different forms of OxLDL (7, 8).
OxLDL autoantibodies in apoE–/– mice
apoE–/– mice represent a widely used animal model of atherosclerosis, and they develop autoantibody titers to modified LDL independent of immunization (9). Witztum and colleagues previously capitalized on this observation by creating a panel of monoclonal antibodies from the spleens of aged apoE–/– mice. This panel has proven to be invaluable for the identification of oxidative products formed in these hyperlipidemic mice (10). All of the characterized monoclonal antibodies are IgMs. The most extensively characterized IgM anti-OxLDL antibody, EO6, reacts against an oxidized phospholipid in modified LDL that has been identified as 1-palmitoyl-2-(5-oxovaleroyl)-sn-glycero-3-phosphorylcholine (POVPC). EO6 exerts functional effects by binding selectively to oxidized LDL and blocking macrophage lipid accumulation (11). An interesting extension of this story is the finding that EO6 is indistinguishable from the natural antibody T15, which is secreted by a specialized subclass of B lymphocytes termed B-1 cells (12). Hence, this antibody is now commonly referred to as T15/EO6. The function of natural antibodies has not been defined, but they are thought to represent part of an innate system that affords protection from microorganisms. For example, the T15 antibody confers protection from Streptococcus pneumoniae infection in mice. Binder et al. have previously demonstrated a link between the T15 antibody and atherosclerosis by increasing T15 antibody titers by S. pneumoniae immunization and observing a concomitant reduction in atherosclerotic lesion size in LDL receptor–deficient mice (13).
How does adaptive immunity affect natural antibodies?
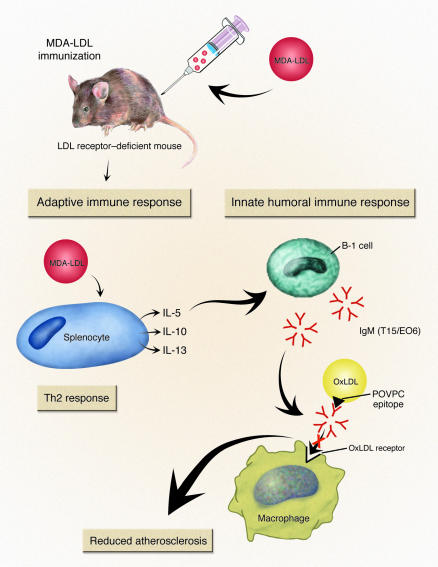
In this issue of the JCI, Binder et al. have elucidated a mechanism of the paradox of immunization with MDA-LDL producing a T lymphocyte–dependent increase in the titers of T15/EO6 antibodies and a reduction in the size of atherosclerotic lesions in LDL receptor–deficient mice. Thus, activation of the adaptive immune system led to a protective effect that was attributed to augmentation of natural immunity. Furthermore, they identify IL-5 as the critical link between these two distinct forms of immune responses (Figure 1) (14).
Figure 1.
The sequence of events elucidated by Binder et al. (14) define the link between adaptive and natural immunity in atherogenesis. MDA-LDL immunization of LDL receptor_deficient mice led to a Th2 immune response. This was demonstrated by the predominance of IgG1 isotype antibodies against MDA-LDL that were generated during immunization. Also, antigen challenge of splenocytes led to secretion of the Th2 cytokines: IL-5, IL-10, and IL-13. IL-5 secretion promoted an increase in titers of natural antibodies, termed T15/EO6. These antibodies recognize an oxidized phospholipid, POVPC, that is not present in the immunizing material. The T15/EO6 antibody has been previously shown to inhibit OxLDL recognition by macrophages, which may account for the reduction in atherosclerotic lesion size.
Binder et al. demonstrate that immunization of wild-type C57BL/6 mice with MDA-LDL led to a predominantly Th2 immune response (14). Antigen challenge resulted in the induction of MDA-LDL–specific IgG1 isotype autoantibodies and the release of IL-5 and IL-13 from splenocytes. Immunization of hypercholesterolemic, LDL receptor–deficient mice also led to enhanced IgG1 autoantibody titers. This production of IgG1 isotype autoantibodies against MDA-LDL was noted previously during severe hypercholesterolemia (15). Immunization also led to a decrease in atherosclerotic lesion size that was selective for the aortic root. Splenocytes from these mice preferentially secreted selected Th2 cytokines, including IL-5, IL-10, and IL-13, when exposed to MDA-LDL. These cytokines were secreted in equivalent mass from splenocytes when antigen challenged in the absence of costimulation with CD28. However, in the presence of CD28 stimulation, IL-5 was the most highly secreted cytokine from splenocytes in response to MDA-LDL challenge. In addition, MDA-LDL immunization also produced an increase in plasma concentrations of IL-5. Other Th2 cytokines were not measured.
This increased adaptive immune response to MDA-LDL was associated with elevated titers of the T15/EO6 natural antibody. This was unexpected since the POVPC epitope recognized by T15/EO6 was not present in the immunizing material (14). Basing their ideas in part on the preferential secretion of IL-5 promoted by MDA-LDL immunization, the authors speculated that the release of this cytokine augmented the production of natural antibodies. This notion was supported by several observations including: (a) IL-5 promoted T15/EO6 secretion from cultured B-1 cells; (b) intraperitoneal injections of IL-5 into naive C57BL/6 mice increased T15/EO6 titers; and (c) IL-5–deficient mice had reduced T15/EO6 titers. Furthermore, the IL-5 genotype had no effect on the titers of IgG1 and IgG2a raised following immunization with MDA-LDL, while an IL-5 deficiency ablated the increased IgM titers elicited in response to MDA-LDL immunization.
IL-5 regulates atherosclerotic lesion size
The final step in this study was to define the effect of IL-5 deficiency on the development of atherosclerosis. This was achieved by irradiating LDL receptor–deficient mice and reconstituting them with bone marrow–derived stem cells from either IL-5 wild-type or deficient mice (14). After an engraftment period, mice were fed a high-fat diet. The group reconstituted with bone marrow–derived stem cells harvested from IL-5 wild-type mice had decreased atherosclerotic lesion size. This decrease in lesion size in mice secreting IL-5 occurred in both the aortic root and the aortic intima in contrast to the site-selective effects of MDA-LDL immunization.
These studies establish a working paradigm for further research. This will include determining whether MDA-LDL immunization has any effect on the development of atherosclerosis in IL-5–deficient mice. It will also be important to establish whether IL-5 is the exclusive cytokine that links the effects of adaptive and natural immunity in atherogenesis. Other Th2 cytokines, particularly IL-10, have established antiatherogenic properties, although links to humoral responses have not been elucidated (16–18). Furthermore, the studies of Binder et al. (14) have demonstrated that the effects of MDA-LDL immunization are associated with increased titers of the IgM-antibody isotype. However, future studies will need to determine the relative contribution of T15/EO6 IgMs to the reduction in atherosclerosis versus that of other IgMs that are formed during MDA-LDL immunization. Finally, it will be important to determine whether the T15/EO6-mediated inhibition of OxLDL uptake by macrophages observed in cultured cells is the mechanism responsible for the reduced atherosclerotic lesion size.
Overall, these studies have mapped out an interesting and novel pathway linking the adaptive and natural arms of the immune system (14). These findings also highlight a practical issue that needs to be considered for future therapeutic strategies. Specifically, antagonists of IL-5 are being developed for asthma therapy. The studies of Binder et al. indicate that it will be important to determine the ramifications of such a therapy on vascular disease.
Footnotes
See the related article beginning on page 427.
Nonstandard abbreviations used: malondialdehyde-modified LDL (MDA-LDL); oxidized LDL (OxLDL); 1-palmitoyl-2-(5-oxovaleroyl)-sn-glycero-3-phosphorylcholine (POVPC).
Conflict of interest: The authors have declared that no conflict of interest exists.
References
- 1.Henriksen T, Mahoney EM, Steinberg D. Enhanced macrophage degradation of low density lipoprotein previously incubated with cultured endothelial cells: Recognition by receptors for acetylated low density lipoproteins. Proc. Natl. Acad. Sci. U. S. A. 1981;78:6499–6503. doi: 10.1073/pnas.78.10.6499. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Steinberg D, Parthasarathy S, Carew TE, Khoo JC, Witztum JL. Beyond cholesterol. Modifications of low density lipoprotein that increase its atherogenicity. N. Engl. J. Med. 1989;320:915–924. doi: 10.1056/NEJM198904063201407. [DOI] [PubMed] [Google Scholar]
- 3.Stemme S, et al. T lymphocytes from human atherosclerotic plaques recognize oxidized low density lipoprotein. Proc. Natl. Acad. Sci. U. S. A. 1995;92:3893–3897. doi: 10.1073/pnas.92.9.3893. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Palinski W, et al. Low density lipoprotein undergoes oxidative modification in vivo. Proc. Natl. Acad. Sci. U. S. A. 1989;86:1372–1376. doi: 10.1073/pnas.86.4.1372. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Salonen JT, et al. Autoantibody against oxidised LDL and progression of carotid atherosclerosis. Lancet. 1992;339:883–887. doi: 10.1016/0140-6736(92)90926-t. [DOI] [PubMed] [Google Scholar]
- 6.Palinski W, Miller E, Witztum JL. Immunization of low density lipoprotein (LDL) receptor-deficient rabbits with homologous malondialdehyde-modified LDL reduces atherogenesis. Proc. Natl. Acad. Sci. U. S. A. 1995;92:821–825. doi: 10.1073/pnas.92.3.821. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Ameli S, et al. Effect of immunization with homologous LDL and oxidized LDL on early atherosclerosis in hypercholesterolemic rabbits. Arterioscler. Thromb. Vasc. Biol. 1996;16:1074–1079. doi: 10.1161/01.atv.16.8.1074. [DOI] [PubMed] [Google Scholar]
- 8.Freigang S, Horkko S, Miller E, Witztum JL, Palinski W. Immunization of LDL receptor-deficient mice with homologous malondialdehyde-modified and native LDL reduces progression of atherosclerosis by mechanisms other than induction of high titers of antibodies to oxidative neoepitopes. Arterioscler. Thromb. Vasc. Biol. 1998;18:1972–1982. doi: 10.1161/01.atv.18.12.1972. [DOI] [PubMed] [Google Scholar]
- 9.Palinski W, et al. ApoE-deficient mice are a model of lipoprotein oxidation in atherogenesis - Demonstration of oxidation-specific epitopes in lesions and high titers of autoantibodies to malondialdehyde-lysine in serum. Arterioscler. Thromb. 1994;14:605–616. doi: 10.1161/01.atv.14.4.605. [DOI] [PubMed] [Google Scholar]
- 10.Palinski W, et al. Cloning of monoclonal autoantibodies to epitopes of oxidized lipoproteins from apolipoprotein E-deficient mice - Demonstration of epitopes of oxidized low density lipoprotein in human plasma. J. Clin. Invest. 1996;98:800–814. doi: 10.1172/JCI118853. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Horkko S, et al. Monoclonal autoantibodies specific for oxidized phospholipids or oxidized phospholipid-protein adducts inhibit macrophage uptake of oxidized low-density lipoproteins. J. Clin. Invest. 1999;103:117–128. doi: 10.1172/JCI4533. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Shaw PX, et al. Natural antibodies with the T15 idiotype may act in atherosclerosis, apoptotic clearance, and protective immunity. J. Clin. Invest. 2000;105:1731–1740. doi: 10.1172/JCI8472. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Binder CJ, et al. Pneumococcal vaccination decreases atherosclerotic lesion formation: molecular mimicry between Streptococcus pneumoniae and oxidized LDL. Nat. Med. 2003;9:736–743. doi: 10.1038/nm876. [DOI] [PubMed] [Google Scholar]
- 14.Binder CJ, et al. IL-5 links adaptive and natural immunity specific for epitopes of oxidized LDL and protects from atherosclerosis. J. Clin. Invest. 2004;114:427–437. doi:10.1172/JCI200420479. doi: 10.1172/JCI20479. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Zhou XH, Paulsson G, Stemme S, Hansson GK. Hypercholesterolemia is associated with a T helper (Th) 1/Th2 switch of the autoimmune response in atherosclerotic apoE-knockout mice. J. Clin. Invest. 1998;101:1717–1725. doi: 10.1172/JCI1216. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Pinderski LJ, et al. Overexpression of interleukin-10 by activated T lymphocytes inhibits atherosclerosis in LDL receptor-deficient mice by altering lymphocyte and macrophage phenotypes. Circ. Res. 2002;90:1064–1071. doi: 10.1161/01.res.0000018941.10726.fa. [DOI] [PubMed] [Google Scholar]
- 17.Pinderski-Oslund LJ, et al. Interleukin-10 blocks atherosclerotic events in vitro and in vivo. Arterioscler. Thromb. Vasc. Biol. 1999;19:2847–2853. doi: 10.1161/01.atv.19.12.2847. [DOI] [PubMed] [Google Scholar]
- 18.Mallat Z, et al. Protective role of interleukin-10 in atherosclerosis. Circ. Res. 1999;85:E17–E24. doi: 10.1161/01.res.85.8.e17. [DOI] [PubMed] [Google Scholar]