Abstract
Objectives
Immune thrombocytopenia (ITP) is an autoimmune bleeding disorder caused by increased platelet destruction and impaired platelet production. Antibody binding to megakaryocytes may occur in ITP, but in vivo evidence of this phenomenon is lacking.
Methods
We determined the proportion of megakaryocytes bound with immunoglobulin G (IgG) in bone marrow samples from primary patients with ITP (n = 17), normal controls (n = 13) and thrombocytopenic patients with myelodysplastic syndrome (MDS; n = 10). Serial histological sections from archived bone marrow biopsies were stained for CD61 and IgG. IgG binding and the number of bone marrow megakaryocytes were determined morphologically by a hematopathologist with four assessors after a calibration exercise to ensure consistency.
Results
The proportion of ITP patients with high IgG binding (>50% of bone marrow megakaryocytes) was increased compared with normal controls [12/17 (71%) vs. 3/13 (23%), P = 0.03]. However, the proportion of ITP patients with high IgG binding was no different than thrombocytopenic patients with MDS [12/17 (71%) vs. 7/10 (70%), P = 1.00]. IgG binding was associated with increased megakaryocyte numbers. Like platelet-associated IgG, megakaryocyte-associated IgG is related to thrombocytopenia but may not be specific for ITP.
Conclusion
Mechanistic studies in ITP should focus on antibody specificity and include thrombocytopenic control patients.
Keywords: blood platelet disorders, autoimmune thrombocytopenia, megakaryocytes, antibody specificity
Immune thrombocytopenia (ITP) is a heterogeneous autoimmune disease characterized by low platelet counts and an increased risk of bleeding. Both clinical evidence and experimental evidence support a dual mechanism of platelet destruction and platelet underproduction in ITP (1–5); however, the mechanism of platelet underproduction remains uncertain.
Platelet destruction in ITP is mediated by autoantibodies against platelet surface glycoproteins (GP), particularly GPII-bIIIa and GPIbIX. These autoantibodies cause thrombocytopenia by accelerated Fcγ receptor-mediated platelet clearance in the reticuloendothelial system (6). Impaired platelet production in ITP is supported by evidence from radiolabelled autologous platelet survival studies demonstrating reduced platelet turnover (7–9), and by the success of thrombopoietin (TPO) receptor agonists (5, 10). One possible mechanism for platelet underproduction is autoantibody-mediated megakaryocyte inhibition (11). In support of this hypothesis, previous experiments have demonstrated that autoantibodies and isolated immunoglobulin G (IgG) fractions from some patients with ITP can inhibit megakaryocyte growth and maturation in vitro (12–14) and that antibodies from some patients with ITP bind to target bone marrow megakaryocytes ex vivo (15). However, in vivo studies investigating antibody binding in the bone marrow microenvironment are lacking. Thus, we designed a histological study of ITP bone marrow samples from patients to investigate antibody binding to megakaryocytes in vivo.
Materials and methods
ITP patients and controls
We identified patients with ITP who had a bone marrow biopsy performed at McMaster University Medical Centre as per clinical need. Patients had primary ITP based on established clinical criteria (16), platelet count levels below 100 × 109/L at the time of bone marrow sampling, and no other pathology identified on bone marrow examination. Patients were excluded if they had lymphoma; human immunodeficiency virus (HIV); hepatitis B or hepatitis C; an abnormal karyotype on cytogenetic evaluation; or if they had received treatment with a TPO receptor agonist. Normal controls were patients who were suspected of having a lymphoma or plasma cell dyscrasia but whose bone marrow biopsy was normal. They had normal platelet counts, hemoglobin levels, and leukocyte counts; did not have splenomegaly, HIV, hepatitis B, or hepatitis C; and had not received treatment with hematopoietic growth factors or antineoplastic medications. Thrombocytopenic controls were patients whose clinical presentation and bone marrow features were consistent with myelodysplastic syndrome (MDS). They had platelet counts below 100 × 109/L at the time of bone marrow sampling. Eligibility of patients and controls were verified by duplicate chart review. All bone marrow biopsy samples were collected, processed, and fixed in the same manner. This study was approved by the Hamilton Integrated Research Ethics Board, McMaster University.
Immunohistochemical stains
Formalin-fixed bone marrow tissue blocks were retrieved from hospital archives, deparaffinized, washed in xylene, and rehydrated with graded washes of ethanol in water. Serial sections (2–4 μm) were pretreated with 0.1 M citrate buffer in a steamer for 45 min. Slides were washed and blocked in 1% normal goat serum for 20 min at room temperature. After washing, slides were incubated with mouse anti-human IgG or anti-CD61 for 60 min. Following incubation, goat anti-mouse IgG was added to slides for 10 min, slides were washed and incubated with 3,3-diaminobenzidine substrate for 20 min, and counterstained with hematoxylin.
Bone marrow assessments
Histological slides were coded, arranged in random order, and evaluated by an experienced hematopathologist blinded to the diagnosis. An initial calibration exercise was performed in duplicate, and four of the authors (DMA, IN, NI, and JWS) participated in the slide review to ensure consistency of reporting. The number of bone marrow megakaryocytes was determined by counting cells in 10 high powered fields (HPF; 400× magnification). The proportion of megakaryocyte-associated IgG was classified as high if greater than 50% of all megakaryocytes demonstrated IgG binding.
Platelet autoantibody quantification
Testing for glycoprotein-specific antibodies on platelets was performed when possible using the direct antigen capture assay (17). Briefly, platelets were harvested from blood collected into acid citrate dextrose (ACD) by differential centrifugation. Platelets were washed and solubilized (20 mmol/L Tris pH 7.4, 150 mmol/L NaCl containing 1% Triton X-100, 0.1% SDS), and platelet lysates were incubated in wells containing monoclonal antibodies against GPIIbIIIa or GPIbIX. Bound human antibody was detected using alkaline phosphatase conjugated to Fcγ-specific goat anti-human IgG, followed by p-nitrophenylphosphate substrate. The optical density (OD405 nm) was determined by absorbance spectrophotometry.
Statistical analysis
The mean number of megakaryocytes in bone marrow specimens was compared between groups using a two-sample t-test. The proportion of megakaryocytes with high IgG binding was compared using Fisher’s exact test (two-sided). Tests were considered significant at the 5% level.
Results
We evaluated freshly stained bone marrow biopsy specimens from patients with ITP (n = 17), age-matched controls with normal platelet counts (n = 13), and MDS controls matched for platelet count levels (n = 10). For patients with ITP, normal controls and MDS controls, median age was 57, 58, and 75 years; 59%, 31%, and 30% were female; and median platelet count at the time of bone marrow sampling was 10 × 109/L [interquartile range (IQR), 4–20], 280 × 109/L (IQR, 206–290), and 27 × 109/L (IQR, 12–35), respectively. Patients with ITP had chronic (n = 14) or persistent (n = 3) disease, with a median disease duration of 2 years (IQR, 1–15). They had received a median of 2 (IQR, 1–3) prior treatments which included corticosteroids, intravenous immunoglobulin, immunosuppressant medications, and danazol. None of the patients had received myelotoxic treatments, rituximab, or TPO receptor agonists in the year prior to bone marrow procurement.
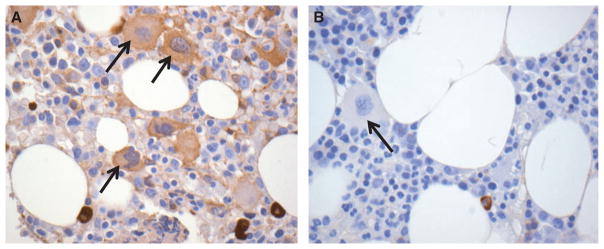
The proportion of ITP patients with high megakaryocyte-associated IgG was increased compared with normal controls [12/17 (71%) vs. 3/13 (23%), P = 0.03] (Table 1 and Fig. 1). However, the proportion of ITP patients with high IgG binding was no different than thrombocytopenic patients with MDS [12/17 (71%) vs. 7/10 (70%), P = 1.00] (Table 1). There was no correlation between megakaryocyte-associated IgG and platelet count or recent exposure to intravenous immune globulin in the ITP cohort (Table 2). IgG staining was mostly homogeneous throughout the cytoplasm of megakaryocytes in most patient samples (Fig. 2A); however, a distinct membrane pattern of staining was observed in three of 12 ITP patients with high megakaryocyte-associated IgG, one normal control patient, and none of the patients MDS (Fig. 2B).
Table 1.
Bone marrow megakaryocytes in patients with ITP. Evaluation of megakaryocyte number and proportion of megakaryocyte with IgG binding among patients with immune thrombocytopenia (ITP), normal controls, and thrombocytopenic controls
| ITP (n = 17) | Normal controls (n = 13) | Thrombocytopenic controls (n = 10) | |
|---|---|---|---|
| Increased IgG on megakaryocytes (n) | 12 (71%) | 3 (23%)1 | 7 (70%) |
| Number of megakaryocytes (Mean, SD) | 9.3 ± 6.0 | 5.2 ± 1.71 | 6.9 ± 4.9 |
Significant differences (P ≤ 0.05) were observed between ITP and normal controls, and between thrombocytopenic and normal controls, but not between ITP and thrombocytopenic controls.
Figure 1.
Immunohistochemical stains of bone marrow biopsy specimens from a patient with ITP (A) and a control patient with a normal platelet count (B). Serial sections were stained for IgG (brown stain). Arrows indicate megakaryocytes. Representative images are shown at 400× magnification.
Table 2.
Characteristics of ITP patients with high or low megakaryocyte-associated IgG as determined by semi-quantitative assessment
| Megakaryocyte-associated IgG | Gender | Age (years) | PLT count1 (×109/L) | ITP duration (years) | Number of prior ITP treatments | IVIG in the preceding 4 weeks |
|---|---|---|---|---|---|---|
| High | M | 26 | 4 | 20 | 6 | Yes |
| High | F | 23 | 4 | 0 | 1 | No |
| High | F | 26 | 5 | 1 | 2 | Yes |
| High | M | 57 | 20 | 1 | 1 | No |
| High | F | 27 | 2 | 1 | 3 | Yes |
| High | F | 29 | 26 | 23 | 3 | No |
| High | F | 31 | 2 | 0 | 1 | Yes |
| High | F | 41 | 4 | 15 | 2 | Yes |
| High | M | 83 | 29 | 1 | 1 | No |
| High | F | 53 | 4 | 25 | 6 | No |
| High | M | 87 | 76 | 2 | 0 | No |
| High | M | 76 | 15 | 0 | 1 | Yes |
| Low | F | 71 | 13 | 1 | 1 | No |
| Low | F | 61 | 65 | 17 | 1 | No |
| Low | M | 63 | 17 | 3 | 2 | Yes |
| Low | F | 73 | 8 | 2 | 4 | Yes |
| Low | M | 64 | 10 | 4 | 6 | No |
IgG, immunoglobulin G; IVIG, intravenous immune globulin.
Platelet count measured at the time of bone marrow sampling.
Figure 2.
Immunohistochemical stains of bone marrow biopsy specimens indicating a homogeneous IgG stain throughout the cytoplasm of megakaryocytes (A) and a IgG staining pattern that was mostly restricted to the megakaryocyte membrane (B). Arrows indicate megakaryocytes. Representative images are shown at 400× magnification.
Median number of bone marrow megakaryocytes was increased in ITP patients compared with normal controls (9.3 ± 6.0 vs. 5.2 ± 1.7 cells per HPF; P = 0.02); but the number of megakaryocytes was no different than patients with MDS (9.3 ± 6.0 vs. 6.9 ± 4.9 cells per HPF; P = 0.21). High megakaryocyte number was associated with increased megakaryocyte-associated IgG across all patients (P = 0.004; Fig. 3).
Figure 3.
Megakaryocyte number [per high-powered field (HPF)] in patients with high (n = 22) or low (n = 18) megakaryocytes-associated IgG (*P = 0.004; bars represent standard error of the mean).
Autoantibodies against GPIIbIIIa or GPIbIX directly on platelets were found in 10 of 13 (69.2%) patients with ITP (four were untested); of those, 6 (60%) had high megakaryocyte-associated IgG.
Discussion
The aim of this study was to evaluate IgG binding to megakaryocytes in bone marrow specimens of patients with ITP as the first step toward determining whether megakaryocytes are targeted by autoantibodies in vivo. Our results indicate that high IgG binding on megakaryocytes occurs in immune (ITP), but also in non-immune thrombocytopenic conditions such as MDS. We did not find a correlation between megakaryocyte-associated IgG and the presence of direct, glycoprotein-specific autoantibodies on platelets. Our findings provide a cautionary note about the need to establish antibody specificity and the importance of appropriate thrombocytopenic controls in such mechanistic studies.
The downstream mechanism of impaired platelet production in ITP may be caused by upregulation of pro-(18) or anti-apoptotic (19) pathways in megakaryoctes; complement-mediated megakaryocyte destruction (20); direct cellular effects on de novo platelet synthesis (21); or impairment of proplatelet formation by ITP autoantibodies (14, 22). The latter may explain why the number of bone marrow megakaryocytes is normal or increased in patients with ITP despite severe thrombocytopenia (23). Upstream, these effects may be triggered by autoantibodies. The evidence for an antibody effect on platelet underproduction derives from in vitro studies using cultured megakaryocytes (12–14). While these experiments demonstrate proof of concept, our study is the first to address whether antibodies bind to megakaryocytes in vivo.
Non-specific IgG binding to megakaryocytes in patients with thrombocytopenia is reminiscent of previous investigations of platelet-associated IgG (PAIgG). PAIgG can be detected on platelets in both immune and non-immune thrombocytopenic conditions (24–26) as a result of increased adsorption of IgG and other proteins in equilibrium with plasma (27). Thus, the amount of IgG bound to platelets increased as platelet count levels decreased. Our results suggest that the same phenomenon of non-specific IgG binding appears to occur for megakaryocytes, but in the same direction as cell number: The amount of IgG bound to megakaryocytes increased as megakaryocyte numbers increased. These results are consistent with data demonstrating that IgG is taken up by maturing megakaryocytes and ultimately stored in platelet α-granules (28, 29). IgG uptake by megakaryocytes may be increased in conditions of thrombocytopenia due to the increased drive toward megakaryopoiesis.
Even though total IgG binding to megakaryocytes did not differentiate immune and non-immune thrombocytopenic conditions, our findings do not exclude the possibility that antibody binding to megakaryocytes is a mechanism of disease. Megakaryocyte-specific antibody binding, even in small amounts, may be enough to disrupt normal megakaryopoiesis and thrombopoiesis. Thus, direct glyco-protein-specific assays, which could detect specific anti-GPIIbIIIa, anti-GPIbIX, or other megakaryocyte antibodies are needed. Such investigations are challenging because of the need for fresh bone marrow samples and highly sensitive serological assays (30), and because of the relatively low numbers of megakaryocytes present in bone marrow samples.
A strength of this study was our careful selection of thrombocytopenic controls matched for platelet count levels, representing a comparable cohort with non-immune platelet underproduction. MDS is a bone marrow disorder characterized by decreased production of peripheral blood cells. Megakaryocytes may be morphologically abnormal, and their numbers may be increased, normal, or decreased in the context of a typically hypercellular marrow. In our study, the number of bone marrow megakaryocytes among patients with MDS was no different than ITP, but the variability around these estimates was wide owing to small sample size. Up to 10% of patients with MDS may have an associated autoimmune disorders (31); however, the patients with MDS in our cohort had typical disease manifestations and treatment courses suggesting that immune thrombocytopenia was not a feature. Another strength of our study was our process of blinded histological review and group calibration for slide reading. A limitation of this study was the use of semi-quantitative assessments of megakaryocyte-associated IgG staining. Further validation of this immunohistochemical stain is needed in larger populations to adjust for background staining and establish threshold levels for specific cells.
In summary, our study demonstrates that non-specific binding of IgG to megakaryocytes – megakaryocyte-associated IgG – occurs in thrombocytopenic conditions, whether due to immune or non-immune causes. Testing for specific anti-megakaryocyte antibody binding with studies that incorporate thrombocytopenic controls is needed to determine whether or not antibody-mediated platelet underproduction is an important mechanism of ITP.
Acknowledgments
This study was funded by Canadian Institutes of Health Research and Amgen (#102446). L.J. Toltl was funded by a MITACS Industrial Postdoctoral Fellowship.
Footnotes
Authorship contributions
DM. Arnold takes primary responsibility for the manuscript. DM. Arnold, I. Nazi, LJ. Toltl, and JG. Kelton designed the research; DM. Arnold, I. Nazi, LJ. Toltl, C. Ross, N. Ivetic, and JW. Smith performed the research, contributed analytical tools, and collected the data; DM. Arnold, I. Nazi, LJ. Toltl, C. Ross, N. Ivetic, JW. Smith and JG. Kelton analyzed and interpreted the data; I. Nazi and Y. Liu performed statistical analysis; DM. Arnold, I. Nazi, LJ. Toltl, and JG. Kelton wrote the manuscript.
Disclosure of conflict of interests
None of the authors have any conflict of interests to report.
References
- 1.Kuter DJ, Bussel JB, Lyons RM, et al. Efficacy of romiplostim in patients with chronic immune thrombocytopenic pur-pura: a double-blind randomised controlled trial. Lancet. 2008;371:395–403. doi: 10.1016/S0140-6736(08)60203-2. [DOI] [PubMed] [Google Scholar]
- 2.Bussel JB, Kuter DJ, Pullarkat V, Lyons RM, Guo M, Nichol JL. Safety and efficacy of long-term treatment with romiplostim in thrombocytopenic patients with chronic ITP. Blood. 2009;113:2161–71. doi: 10.1182/blood-2008-04-150078. [DOI] [PubMed] [Google Scholar]
- 3.Kuter DJ, Bussel JB, Newland A, et al. Long-term treatment with romiplostim in patients with chronic immune thrombocytopenia: safety and efficacy. Br J Haematol. 2013;161:411–23. doi: 10.1111/bjh.12260. [DOI] [PubMed] [Google Scholar]
- 4.Saleh MN, Bussel JB, Cheng G, Meyer O, Bailey CK, Arning M, Brainsky A. Safety and efficacy of eltrombopag for treatment of chronic immune thrombocytopenia: results of the long-term, open-label EXTEND study. Blood. 2013;121:537–45. doi: 10.1182/blood-2012-04-425512. [DOI] [PubMed] [Google Scholar]
- 5.Cheng G, Saleh MN, Marcher C, Vasey S, Mayer B, Aivado M, Arning M, Stone NL, Bussel JB. Eltrombopag for management of chronic immune thrombocytopenia (RAISE): a 6-month, randomised, phase 3 study. Lancet. 2011;377:393–402. doi: 10.1016/S0140-6736(10)60959-2. [DOI] [PubMed] [Google Scholar]
- 6.Cines DB, Blanchette VS. Immune thrombocytopenic purpura. N Engl J Med. 2002;346:995–1008. doi: 10.1056/NEJMra010501. [DOI] [PubMed] [Google Scholar]
- 7.Stoll D, Cines DB, Aster RH, Murphy S. Platelet kinetics in patients with idiopathic thrombocytopenic purpura and moderate thrombocytopenia. Blood. 1985;65:584–8. [PubMed] [Google Scholar]
- 8.Ballem PJ, Segal GM, Stratton JR, Gernsheimer T, Adamson JW, Slichter SJ. Mechanisms of thrombocytopenia in chronic autoimmune thrombocytopenic purpura. Evidence of both impaired platelet production and increased platelet clearance. J Clin Invest. 1987;80:33–40. doi: 10.1172/JCI113060. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Louwes H, Zeinali Lathori OA, Vellenga E, de Wolf JT. Platelet kinetic studies in patients with idiopathic thrombocytopenic purpura. Am J Med. 1999;106:430–4. doi: 10.1016/s0002-9343(99)00054-6. [DOI] [PubMed] [Google Scholar]
- 10.Kuter DJ, Rummel M, Boccia R, Macik BG, Pabinger I, Selleslag D, Rodeghiero F, Chong BH, Wang X, Berger DP. Romiplostim or standard of care in patients with immune thrombocytopenia. N Engl J Med. 2010;363:1889–99. doi: 10.1056/NEJMoa1002625. [DOI] [PubMed] [Google Scholar]
- 11.Perdomo J, Yan F, Chong BH. A megakaryocyte with no platelets: anti-platelet antibodies, apoptosis, and platelet production. Platelets. 2013;24:98–106. doi: 10.3109/09537104.2012.669508. [DOI] [PubMed] [Google Scholar]
- 12.Chang M, Nakagawa PA, Williams SA, Schwartz MR, Imfeld KL, Buzby JS, Nugent DJ. Immune thrombocytopenic purpura (ITP) plasma and purified ITP monoclonal autoantibodies inhibit megakaryocytopoiesis in vitro. Blood. 2003;102:887–95. doi: 10.1182/blood-2002-05-1475. [DOI] [PubMed] [Google Scholar]
- 13.McMillan R, Wang L, Tomer A, Nichol J, Pistillo J. Suppression of in vitro megakaryocyte production by antiplatelet autoantibodies from adult patients with chronic ITP. Blood. 2004;103:1364–9. doi: 10.1182/blood-2003-08-2672. [DOI] [PubMed] [Google Scholar]
- 14.Takahashi R, Sekine N, Nakatake T. Influence of monoclonal antiplatelet glycoprotein antibodies on in vitro human megakaryocyte colony formation and proplatelet formation. Blood. 1999;93:1951–8. [PubMed] [Google Scholar]
- 15.McMillan R, Luiken GA, Levy R, Yelenosky R, Longmire RL. Antibody against megakaryocytes in idiopathic thrombocytopenic purpura. JAMA. 1978;239:2460–2. doi: 10.1001/jama.239.23.2460. [DOI] [PubMed] [Google Scholar]
- 16.Neunert C, Lim W, Crowther M, Cohen A, Solberg L, Jr, Crowther MA. The American Society of Hematology 2011 evidence-based practice guideline for immune thrombocytopenia. Blood. 2011;117:4190–207. doi: 10.1182/blood-2010-08-302984. [DOI] [PubMed] [Google Scholar]
- 17.Warner MN, Moore JC, Warkentin TE, Santos AV, Kelton JG. A prospective study of protein-specific assays used to investigate idiopathic thrombocytopenic purpura. Br J Haematol. 1999;104:442–7. doi: 10.1046/j.1365-2141.1999.01218.x. [DOI] [PubMed] [Google Scholar]
- 18.Houwerzijl EJ, Blom NR, van der Want JJ, Esselink MT, Koornstra JJ, Smit JW, Louwes H, Vellenga E, de Wolf JT. Ultrastructural study shows morphologic features of apoptosis and para-apoptosis in megakaryocytes from patients with idiopathic thrombocytopenic purpura. Blood. 2004;103:500–6. doi: 10.1182/blood-2003-01-0275. [DOI] [PubMed] [Google Scholar]
- 19.Yang L, Wang L, Zhao CH, Zhu XJ, Hou Y, Jun P, Hou M. Contributions of TRAIL-mediated megakaryocyte apoptosis to impaired megakaryocyte and platelet production in immune thrombocytopenia. Blood. 2010;116:4307–16. doi: 10.1182/blood-2010-02-267435. [DOI] [PubMed] [Google Scholar]
- 20.Najaoui A, Bakchoul T, Stoy J, Bein G, Rummel MJ, Santoso S, Sachs UJ. Autoantibody-mediated complement activation on platelets is a common finding in patients with immune thrombocytopenic purpura (ITP) Eur J Haematol. 2012;88:167–74. doi: 10.1111/j.1600-0609.2011.01718.x. [DOI] [PubMed] [Google Scholar]
- 21.Li S, Wang L, Zhao C, Li L, Peng J, Hou M. CD8+ T cells suppress autologous megakaryocyte apoptosis in idiopathic thrombocytopenic purpura. Br J Haematol. 2007;139:605–11. doi: 10.1111/j.1365-2141.2007.06737.x. [DOI] [PubMed] [Google Scholar]
- 22.Lev PR, Grodzielski M, Goette NP, et al. Impaired proplatelet formation in immune thrombocytopenia: a novel mechanism contributing to decreased platelet count. Br J Haematol. 2014;165(6):854–64. doi: 10.1111/bjh.12832. [DOI] [PubMed] [Google Scholar]
- 23.Mahabir VK, Ross C, Popovic S, et al. A blinded study of bone marrow examinations in patients with primary immune thrombocytopenia. Eur J Haematol. 2013;90:121–6. doi: 10.1111/ejh.12041. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Mueller-Eckhardt C, Kayser W, Mersch-Baumert K, Mueller-Eckhardt G, Breidenbach M, Kugel HG, Graubner M. The clinical significance of platelet-associated IgG: a study on 298 patients with various disorders. Br J Haematol. 1980;46:123–31. doi: 10.1111/j.1365-2141.1980.tb05942.x. [DOI] [PubMed] [Google Scholar]
- 25.Kelton JG, Powers PJ, Carter CJ. A prospective study of the usefulness of the measurement of platelet-associated IgG for the diagnosis of idiopathic thrombocytopenic purpura. Blood. 1982;60:1050–3. [PubMed] [Google Scholar]
- 26.Beardsley DS, Spiegel JE, Jacobs MM, Handin RI, Lux SE. Platelet membrane glycoprotein IIIa contains target antigens that bind anti-platelet antibodies in immune thrombocytopenias. J Clin Invest. 1984;74:1701–7. doi: 10.1172/JCI111587. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Kelton JG, Steeves K. The amount of platelet-bound albumin parallels the amount of IgG on washed platelets from patients with immune thrombocytopenia. Blood. 1983;62:924–7. [PubMed] [Google Scholar]
- 28.George JN. Platelet immunoglobulin G: its significance for the evaluation of thrombocytopenia and for understanding the origin of alpha-granule proteins. Blood. 1990;76:859–70. [PubMed] [Google Scholar]
- 29.George JN, Saucerman S. Platelet IgG, IgA, IgM, and albumin: correlation of platelet and plasma concentrations in normal subjects and in patients with ITP or dysproteinemia. Blood. 1988;72:362–5. [PubMed] [Google Scholar]
- 30.Huh HJ, Park CJ, Kim SW, Han SH, Jang S, Chi HS. Flow cytometric detection of platelet-associated immunoglobulin in patients with immune thrombocytopenic purpura and nonimmune thrombocytopenia. Ann Clin Lab Sci. 2009;39:283–8. [PubMed] [Google Scholar]
- 31.Saif MW, Hopkins JL, Gore SD. Autoimmune phenomena in patients with myelodysplastic syndromes and chronic myelomonocytic leukemia. Leuk Lymphoma. 2002;43:2083–92. doi: 10.1080/1042819021000016186. [DOI] [PubMed] [Google Scholar]