Abstract
Diabetes triples the risk for active tuberculosis, thus the increasing burden of type 2 diabetes will help to sustain the present tuberculosis epidemic. Recommendations have been made for bidirectional screening, but evidence is scarce about the performance of specific tuberculosis tests in individuals with diabetes, specific diabetes tests in patients with tuberculosis, and screening and preventive therapy for latent tuberculosis infections in individuals with diabetes. Clinical management of patients with both diseases can be difficult. Tuberculosis patients with diabetes have a lower concentration of tuberculosis drugs and a higher risk of drug toxicity than tuberculosis patients without diabetes. Good glycaemic control, which reduces long-term diabetes complications and could also improve tuberculosis treatment outcomes, is hampered by chronic inflammation, drug-drug interactions, suboptimum adherence to drug treatments, and other factors. Besides drug treatments for tuberculosis and diabetes, other interventions, such as education, intensive monitoring, and lifestyle interventions, might be needed, especially for patients with newly diagnosed diabetes or those who need insulin. From a health systems point of view, delivery of optimum care and integration of services for tuberculosis and diabetes is a huge challenge in many countries. Experience from the combined tuberculosis and HIV/AIDS epidemic could serve as an example, but more studies are needed that include economic assessments of recommended screening and systems to manage concurrent tuberculosis and diabetes.
Introduction
Tuberculosis is a major global health problem. In 2012, 8·6 million people developed tuberculosis and 1·3 million died from the disease.1 Diabetes increases the risk of developing active tuberculosis2,3 and is associated with worse tuberculosis treatment outcomes.4 Globally, 15% of tuberculosis cases are estimated to be attributable to diabetes.5 The International Diabetes Federation predicts that the number of people worldwide with diabetes will increase by 55% during the next 20 years, with the largest increase expected in Africa (+109%).6 Therefore, diabetes will make an increasingly important contribution to the tuberculosis epidemic. Whether screening for diabetes in patients with tuberculosis (and the converse) is feasible and cost effective, and how those identified with concurrent tuberculosis and diabetes can be best managed, are key research questions (panel 1).
Panel 1: Example case history.
A 45-year-old man presents with a 2 month history of a cough and night sweats to a lung clinic in Jakarta, Indonesia. He has no relevant medical history, has been a smoker for 25 years, and is not taking drugs. The patient’s physical examination is unremarkable, but his chest radiograph shows abnormalities suggestive of tuberculosis and his sputum is positive for acid-fast bacilli. His HIV test is negative. Standard treatment for pulmonary tuberculosis is started. When he returns after 2 weeks he reports longstanding thirst and polyuria. An endocrinologist is consulted and type 2 diabetes is diagnosed, even though the patient has a healthy weight (BMI of 20·1 kg/m²). The patient is given oral diabetes drugs but his blood glucose concentrations remain high. He is referred for insulin treatment but does not attend the diabetes clinic. His sputum smear remains positive after 2 months of therapy, calling into question compliance to treatment. After a full 6 month course of tuberculosis treatment, he remains sputum smear positive and still has hyperglycaemia.
In this Series paper, we focus on tuberculosis-endemic settings. We assess the epidemiology of tuberculosis and diabetes, screening for tuberculosis in patients with diabetes and the converse, and issues related to clinical management of concurrent tuberculosis and diabetes. We also discuss implications of the joint epidemic for health services and public health, and outline key questions for future research.
Epidemiology
Diabetes, tuberculosis, and evidence of an association between both diseases
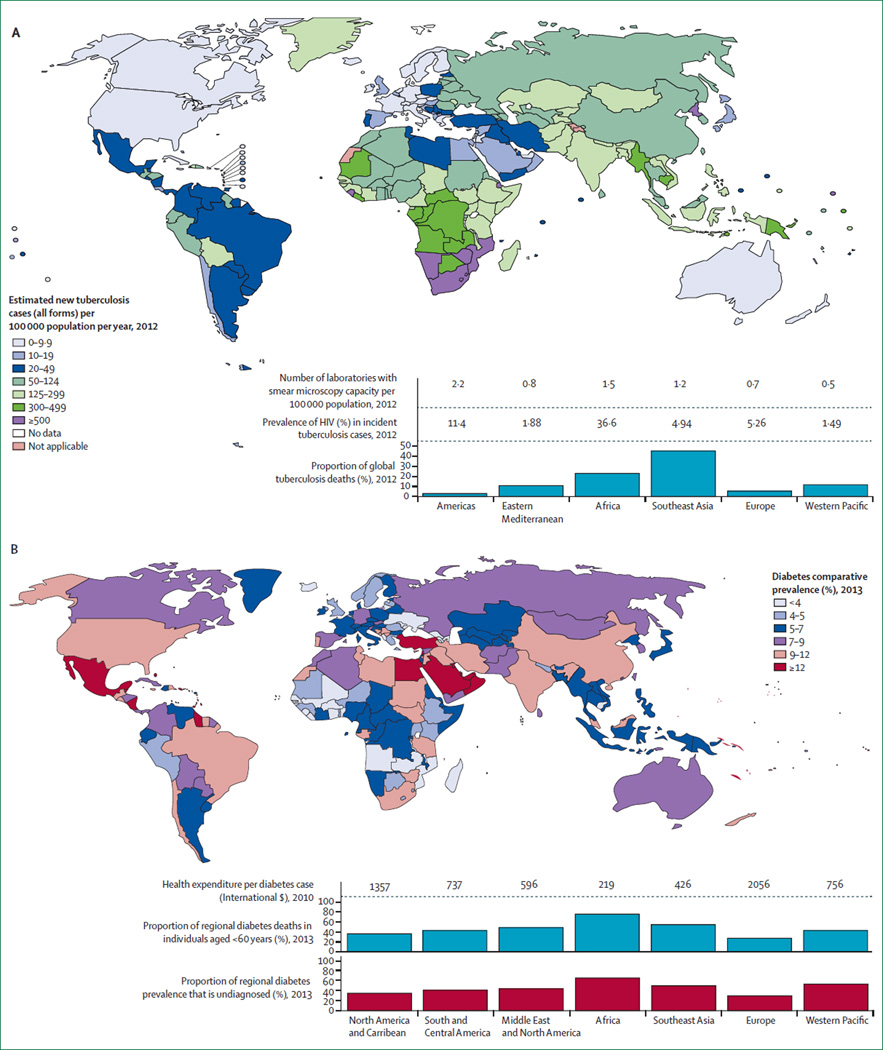
Tuberculosis is caused by Mycobacterium tuberculosis, which is thought to latently infect a third of the global population, placing such individuals at a 5–10% average lifetime risk of progressing to the active form of the disease. Individuals with HIV are at a 12–20 times increased risk of developing tuberculosis. The prevalence and annual incidence of tuberculosis varies greatly geographically, as does laboratory capacity to test for this disease (figure 1). Tuberculosis requires treatment with multiple antituberculosis drugs for at least 6 months. Around 500 000 cases globally are of multidrug-resistant tuberculosis, which need to be treated with expensive second-line drugs and for long durations.7
Figure 1. Diabetes and tuberculosis.
(A) Tuberculosis incidence in 2012 for each WHO region; reproduced from WHO’s Global tuberculosis report 2013.1 (B) Comparative prevalence of diabetes in 2013 for adults aged between 20 and 79 years within each International Diabetes Federation (IDF) region; reproduced from Diabetes Atlas, 2013, and with permission by IDF.6
Evidence shows that both type 1 and type 2 diabetes can increase tuberculosis risk. However, as type 2 diabetes accounts for 85–95% of global cases of diabetes the burden of comorbid disease from type 2 diabetes is much greater and thus the main focus of this paper. Diabetes prevalence is increasing substantially; from an estimated 151 million adults in 2000,8 to 382 million in 2013, which is predicted to increase to 592 million in 2035.6 Prevalence, number of undiagnosed cases, expenditure per case, and mortality, across the world varies greatly (figure 1).6 Ongoing globalisation, urbanisation, population growth, and rural to urban migration (which might contribute to diabetes burden because of changes in environment and resulting putative risk factors such as obesity) in low-income and middle-income countries all contribute to increases in the burden of type 2 diabetes.9,10 The thrifty phenotype hypothesis, whereby sparse nutrition and fetal growth leads to metabolic adaptation, might account for how such changes rapidly increase diabetes prevalence in regions with undernutrition;11,12 fetal growth patterns and programming might be unable to biologically cope in new obesogenic environments.11 Type 2 diabetes has historically been perceived as a disease of affluence that is strongly associated with obesity and physical inactivity, but in low-income countries diabetes is also prevalent in populations who live in poor urban settings, with up to 15% of adults affected.13 Diabetes prevalence is around 15–30% in patients with tuberculosis in some settings. As this figure is likely to increase, management of diabetes in patients with tuberculosis is becoming an inevitable part of clinical practice.
Two systematic reviews2,3 that together included 15 studies showed the risk of developing tuberculosis is at least tripled in individuals with diabetes, consistent across geographical regions, and that the increase in risk is highest in young people. However, few studies adjusted for confounders other than age and sex, there was variation in the quality of testing for both diseases, and the data from low-income and middle-income countries were scarce (figure 1).2,3 A meta-analysis2 of data from three prospective studies quantified tuberculosis risk in people with diabetes compared to those without diabetes (relative risk 3·11, 95% CI 2·27–4·26). However, two of the included studies were done in individuals with renal failure, which is a risk factor for tuberculosis, and all were done in Asia. Other studies14–16 have provided more recent data from low-income and middle-income countries and have used better case-ascertainment methods. HIV negative, but not HIV positive, individuals with diabetes in Tanzania had an increased risk of tuberculosis, which suggests that the effect of HIV overrides the risk conferred by diabetes.14 Another study17 showed the risk of tuberculosis was increased at levels of hyperglycaemia of less than present diabetes diagnostic cutoff s. A large, multicountry, longitudinal analysis18 from low-income countries reported an increased tuberculosis risk in people with diabetes. Additionally, tuberculosis prevalence (odds ratio 4·7, 95% CI 1·0–22·5) and incidence (8·6, 1·9–40·4) were more likely to increase in countries where diabetes prevalence had increased during the past 10 years (between 1990–95 and 2003–04), and where per-capita base year gross domestic product was low (highest gross domestic product quintile against lowest quintile for tuberulcosis prevalence [0·09, 0·02–0·42] and tuberulcosis incidence [0·03, 0·01–0·14]).
Few studies explicitly assess whether tuberculosis increases the risk of diabetes.19 Induced hyperglycaemia or impaired glucose tolerance has been noted during the early phase of tuberculosis, but whether these symptoms are newly established, previously undiagnosed, or intermittent is unknown.20–24
Diabetes and the risk of poor tuberculosis outcomes
Diabetes is not only a risk factor for active tuberculosis, but also for poor tuberculosis treatment outcomes. A meta-analysis4 showed that the combined outcome of tuberculosis treatment failure and death was significantly higher in patients with diabetes than in those without diabetes (relative risk 1·69, 95% CI 1·36–2·12). Similarly, the risk of death during tuberculosis treatment (1·89, 1·52–2·36) and relapse following treatment (3·89, 2·43–6·23) were higher in patients with diabetes compared to those without diabetes. Six of nine studies that assessed sputum conversion reported that patients with tuberculosis and diabetes were more likely to remain sputum smear positive at 2–3 months after starting treatment for tuberculosis.4 Severity of hyperglycaemia (as measured by HbA1c) did not seem to increase the risk of a poor tuberculosis outcome.25 The effects of diabetes on outcomes of tuberculosis treatment are unlikely to be from differences in drug resistance; a systematic review4 of 33 studies showed no association between drug-resistant tuberculosis and diabetes. However, many of these studies did not adjust for other key confounders4 and only reported all-cause mortality. Therefore, a high proportion of deaths in patients with diabetes presenting with tuberculosis could be because of poor underlying health or advanced age.4
An increasing number of studies have reported data from low-income and middle-income countries.4,26–29 A prospective study28 from Tanzania noted an increased risk of early death among HIV-negative individuals with tuberculosis and diabetes, compared to those without diabetes (relative risk of dying in first 100 days 5·09, 95% CI 2·36–11·02), but no increased long-term risk, suggesting comorbidity is the key determinant. Another prospective study30 from South Korea showed the combined effect of diabetes and smoking increased the risk of tuberculosis mortality by almost six times in the first year (hazard ratio 5·78, 95% CI 1·09–30·56) after enrollmnet and tuberculosis treatment.
Population effect of diabetes on tuberculosis
Evidence on the wider population health and economic effects of the joint tuberculosis and diabetes epidemic is scarce. Use of static models such as population attributable risk, which do not account for onward transmission of tuberculosis in those with tuberculosis and diabetes, are likely to yield conservative estimates. Odone and colleagues31 estimate that the present upward trend in diabetes prevalence (projected by the International Diabetes Federation to be a 55% rise in the number of people with diabetes by 2035) would off set the worldwide decreases in tuberculosis incidence by 3% by 2035, and by 8% in a pessimistic scenario (an 80% rise in the number of people with diabetes),31 which might be realistic in regions where diabetes risk factors are increasing fastest.32 The analysis did not account for onward tuberculosis transmission, and thereby underestimates the potential effect of diabetes. Clearly, further dynamic models with an economic investigative component are needed.
Patient management and screening
Screening for active tuberculosis in individuals with diabetes
Up to 2011, 12 studies had been undertaken for tuberculosis screening in individuals with diabetes.33 Half these studies used radiography whereas the other half used radiography plus microbiological testing to identify tuberculosis in patients.33 Only half the studies compared the results from people with diabetes with those in individuals without diabetes.33 Unsurprisingly, a large variance was noted in the estimates of tuberculosis prevalence (from 1·7% in Sweden to 36% in South Korea); and in the numbers needed to be screened to detect one previously undetected case of tuberculosis (4–442, respectively). Table 1 contains the different methods for tuberculosis screening, diagnosis, and potential pitfalls with their use in patients with diabetes. The yield (number needed to screen) and cost-effectiveness of different tuberculosis tests mainly depends on the tuberculosis prevalence in any given setting and the sensitivity and cost of tests used.33
Table 1.
Tests for screening and diagnosis of active tuberculosis and potential pitfalls with their use in individuals with diabetes38
| Mode of action | Comparative direct cost |
Time | Sensitivity and specificity | Restrictions | Issues of use in patients with diabetes |
|
|---|---|---|---|---|---|---|
|
Screening tests | ||||||
| Clinical assessment |
Symptomatic screen and clinical exam |
Low | Hours | Sensitivity: 77%; specificity: 67% (pooled data from eight screening studies for any tuberculosis symptoms*†)34 |
Low sensitivity and specificity |
Presentation of clinical characteristics might differ in patients with diabetes; evidence base is weak |
| Chest radiography |
Enables detection of tuberculosis- suggestive lesions, which can be present in asymptomatic individuals |
Medium | Hours | Sensitivity: 98%; specificity: 75% (pooled data from three screening studies for any abnormalities on chest radiography*†)34 |
High rates of non-tuberculosis abnormalities in some settings |
Radiographs might differ in patients with diabetes; weak evidence |
|
Diagnostic tests | ||||||
| Sputum microscopy |
Smear to identify acid fast bacilli |
Medium | Days | Sensitivity: 84%; specificity: 98% (LED microscopy vs culture; dependent on optimisation of microscopy, sensitivity can vary by around 20%)35 |
Less sensitive than culture |
No standard laboratory capacity for most diabetes clinics; high numbers of patients with diabetes unwilling to provide sputum sample36 |
| Sputum culture |
Mycobacterium tuberculosis culture (allows subsequent resistance test) |
High | Up to 8 weeks |
Gold standard | Time and skill required; throughput (difficult to do as many sputum cultures as there are patients with diabetes) |
No standard laboratory capacity for most diabetes clinics; high number of patients with diabetes unwilling to provide sputum sample36 |
| Xpert MTB/RIF |
M tuberculosis PCR; also detects rifampicin resistance |
Very high | Hours | Sensitivity: 88%; specificity: 98% (pooled data from 15 screening studies with Xpert MTB/RIF as an initial test replacing smear microscopy)37 |
High cost, low throughput |
No specific issues; automated, closed system; no laboratory or skills needed37 |
PCR=polymerase chain reaction. LED=light-emitting diode.
Screening completed in a general population.
Gold standard for any positive microbiological screen.
The overall evidence base identified by a systematic review33 was weak and exposed an urgent need for further research. Several subsequent studies from various different settings have been published since 2011,17,36,39–47 and have shown that symptom screening and chest radiography for tuberculosis are operationally feasible in diabetes clinics in India and China (countries with a high tuberculosis burden (provisionally defined as 100 cases per 100 000 people; appendix).40,45,46 Further studies are needed to provide reliable estimates of tuberculosis prevalence in individuals with diabetes, and whether any relation exists between duration of diabetes and glycaemic control. We did not find any studies that used alternative tests in individuals with diabetes, such as computer-aided radiography reading48 or Xpert MTB/RIF assay, a rapid automated molecular test that is being rolled out in settings with a high burden of tuberculosis.37
Overall, tuberculosis in individuals with diabetes is relatively uncommon and so selective tuberculosis screening might be indicated. Clearly a low index of suspicion is needed for when a patient with diabetes presents with suggestive symptoms of tuberculosis such as a cough, weight loss, or a history of exposure to a patient with tuberculosis.
Screening for latent tuberculosis infection in individuals with diabetes
Latent tuberculosis infection can be detected with the tuberculin skin test or interferon gamma release assay, both of which screen for immunological memory.49 Although the interferon gamma release assays can be slightly more specific and sensitive than a tuberculin skin test, this technique is more expensive and technically intricate. Tuberculin skin tests are disadvantageous because a follow-up visit is needed after 2–3 days (appendix). Few studies have addressed whether individuals with diabetes and latent tuberculosis infection are at an increased risk of progression to active tuberculosis.50–52 Screening for latent tuberculosis infection in individuals with diabetes, in particular those with poor glycaemic control, could help to identify a high-risk population that could be offered preventive therapy, which is not recommended but is a topic of debate within WHO.5 Whether chemoprophylaxis is equally effective in individuals with or without diabetes is unknown. Only two old and potentially underpowered studies with inadequate control groups have addressed this issue.33,53,54 Therefore, a randomised controlled trial of preventive tuberculosis therapy in individuals with diabetes comparative to without diabetes is a high priority.55
Screening for diabetes in patients with tuberculosis
Screening for diabetes in patients with tuberculosis is now widely recommended, especially where the prevalence of diabetes is high.55 Tuberculosis control programmes in India and the Pacific Islands region both advocate screening in this group.56,57 The Pacific Islands region has a particularly high prevalence of diabetes with up to a third of adults affected, and hence diabetes screening is recommended for all adults older than 18 years with tuberculosis. Both programmes recommend repeat testing for diabetes after 2–4 weeks of tuberculosis treatment or in patients who develop symptoms of hyperglycaemia during treatment.
However, the best time and methods to diagnose diabetes in patients with tuberculosis are unclear.40,46,55 The prevalence of diabetes rises steeply with age, but the most efficient age cut-off for screening is also unclear and varies between populations. Measurement of blood glucose concentrations at a single timepoint might lead to a false diagnosis of diabetes in patients with tuberculosis because they could have intermittent hyperglycaemia through induction of insulin resistance, mediated by inflammation, due to tuberculosis infection.20–24,58 Repeat testing could identify transient hyperglycaemia. Assessment of HbA1c concentration is the only diabetes test that shows average glycaemia over time and in a single study,59 was more sensitive than fasting blood glucose when used as a screening test for newly diagnosed diabetes in patients with tuberculosis. The point at which patients with tuberculosis are screened for diabetes is probably important. In studies identified by a systematic review,33 four described diabetes screening before tuberculosis treatment, seven after treatment initiation, four at several different follow-up times, and seven studies did not specify the screening timepoint. If screening at treatment initiation is done, a second test during tuberculosis treatment, or after treatment completion, seems logical.
In low-income and middle-income countries with a high tuberculosis burden, the tests used for diabetes screening are likely to be dictated by local circumstances such as health-care set-up, opportunities for patient follow-up, and costs associated with the tests.40,45,46,55 Large studies in India and China have shown that fasting blood glucose and random blood glucose measurements can be done in tuberculosis clinics (appendix).40,45,46 Various other tests are feasible for use, but their cost-effectiveness has not been investigated.17,36,40–47 Improved diabetes screening approaches in patients with tuberculosis might emerge as promising new techniques become available, including point-of-care HbA1c assays, non-invasive advanced glycation end product readers, and sudomotor function-based screening.60 Different methods used for diabetes screening might have potential pitfalls associated with their use in patients with tuberculosis (table 2).
Table 2.
Screening tests for diabetes and potential pitfalls with their use in patients with tuberculosis34,61,63
| Mode of action | Comparative direct costs, and perceived benefits |
Sensitivity and specificity* | Restrictions | Issues of use in patients with tuberculosis |
|
|---|---|---|---|---|---|
|
Screening tests | |||||
| Clinical assessment |
Classic signs and symptoms of diabetes |
Low; fast; easy to obtain |
Comparatively very low sensitivity and specificity |
Very low sensitivity | Overlap with tuberculosis symptoms, diagnostic for diabetes when plasma glucose concentrations raised |
| Risk scores | Questionnaire to compute a score for diabetes based on clinical or socio- demographic characteristics |
Low; non-invasive; easy to implement |
Sensitivity 35–72%; specificity 77–83%; many alternate risk scores exist, broad ranges of sensitivity and specificity show the heterogeneity between scores available; Finnish best validated and most commonly used61 |
Fewer risk scores validated in low-income and middle-income countries |
Diabetes risk markers might differ in patients with tuberculosis (weak evidence) |
| Urinary dipstick | Point-of-care test with urine sample |
Low; fast; less invasive than blood tests |
Sensitivity 16–64%; specificity >98%; figures are ranges across studies; broad ranges show the heterogeneity in test reading and interpretation61,62 |
Low sensitivity, particularly after eating |
None noted |
| Capillary glucose (fasting or non- fasting) |
Finger-prick blood test | Low; straightforward; widely available, no need for laboratory capacity |
Sensitivity 40–75%; specificity 66–96%61,62 |
Point-of-care meters affected by calibration, heat, humidity, and other factors |
All single timepoint tests might give false positive results because of intermittant hyperglycaemia caused by tuberculosis associated inflammation |
|
Diagnostic tests | |||||
| Random (non-fasting) plasma glucose |
Blood sample, taken at any time |
Medium; widely available through laboratory or point- of-care tests |
Sensitivity 40–65%; specificity 90–93%61,62† |
Must process in <2 h; difficult to interpret; affected by short-term lifestyle changes and diet |
All single timepoint tests might give false positive results because of intermittant hyperglycaemia caused by tuberculosis associated inflammation |
| Fasting plasma glucose |
Blood test taken in morning after 8 h fast |
Medium; most laboratories can do this point-of-care test |
Sensitivity 66–85%; specificity 98%61,62† |
Must process in <30 min; affected by short-term lifestyle changes; not all patients fast fully before test; needs return appointment |
Not practical in tuberculosis clinics; fasting might be contraindicated in active tuberculosis; all single timepoint tests might give false positive results because of intermittent hyperglycaemia caused by tuberculosis associated inflammation |
| HbA1c | Blood test; requires NGSP- or IFCC- certified laboratory, or point-of-care meter |
High; can provide rapid result at point of care; no fasting required |
Sensitivity 44–66%; specificity 79–98%62 † |
High stability but less sensitive as a diagnostic test than fasting plasma glucose or oral glucose tolerance test; assays can be affected by haemoglobinopathies and anaemias |
A measure of average blood glucose concentration during previous 8–12 weeks might miss new onset cases of diabetes |
| Oral glucose tolerance test |
Blood sample after 8 h fast plus repeat testing (2 h) after oral glucose challenge |
High; most sensitive test for diabetes |
Gold standard† | Needs to be processed within 30 min; time consuming and complex |
Not practical in tuberculosis clinics; all single timepoint tests might give false positive results because of intermittant hyperglycaemia cause by tuberculosis associated inflammation |
NGSP=National Glycohemoglobin Standardisation Programme. IFCC=International Federation of Clinical Chemistry.
Substantial heterogeneity exists between individual studies on diabetes screening test performance in general, and although commonly used, no robust evidence was reported on screening via clinical assessment; cited test performance estimates should thus be interpreted with caution.
Variation occurs dependent on cut-point and gold standard used (eg, prevalence of complication or oral glucose tolerance test).
Clinical presentation
Two systematic reviews64,65 reported no strong evidence of differences in radiographic presentation of tuberculosis in patients with or without diabetes. However, in 2014, a large case series66 showed that tuberculosis patients with diabetes had more lung cavities and parenchymal lesions than tuberculosis patients without diabetes; among patients with diabetes these radiographic abnormalities were more common in those with poorer glycaemic control. Evidence with respect to the clinical presentation of tuberculosis in individuals with diabetes is inconsistent, although patients with tuberculosis and diabetes are usually older and heavier than the general tuberculosis population and more likely to be male.14,28,64,65 Less is known about whether diabetes phenotype is different in patients with concurrent tuberculosis, but those patients with tuberculosis might be more likely to smoke, because smoking is a risk factor for tuberculosis.67
Treatment for concurrent tuberculosis and diabetes
Treatment of tuberculosis in patients with diabetes
The optimum treatment strategy for concurrent tuberculosis and diabetes is not known. Diabetes is associated with increased risk of tuberculosis treatment failure, death, and relapse, but whether optimum glucose control can partly or fully mitigate these negative effects, and whether tuberculosis treatment should be adjusted in patients with diabetes is uncertain. Treatment of drug-susceptible tuberculosis is highly standardised and usually consists of two drugs (rifampicin and isoniazid) that are taken for 6 months, combined with another two drugs (pyrazinamide and ethambutol) for the first 2 months of treatment. In general, patients with tuberculosis and diabetes are not treated differently than patients with only tuberculosis. However, this approach might need to be reconsidered. Increases in rates of tuberculosis treatment failure in patients with diabetes are unlikely to be due to high rates of M tuberculosis drug resistance or low treatment compliance.65 Alternative causes of treatment failure could be more extensive tuberculosis disease,66 an altered immune response in people with diabetes (although this theory is largely speculative), or reduced concentrations of antituberculosis drugs in patients with diabetes.
In Indonesia, exposure to rifampicin (area under the curve) and maximum rifampicin plasma concentrations during the continuation phase of tuberculosis treatment were about 50% lower in tuberculosis patients with diabetes than they were in age-matched and sex-matched patients with tuberculosis without diabetes.68 After correction for bodyweight, which was 20% higher in individuals with diabetes and tuberculosis, diabetes was still associated with reduced exposure to rifampicin and reduced plasma rifampicin concentrations were associated with more profound hyperglycaemia in patients.68 In contrast, no differences were evident in full curve pharmacokinetics of rifampicin, pyrazinamide, and ethambutol during the intensive phase of tuberculosis treatment of patients with diabetes and weight-matched patients with pulmonary tuberculosis without diabetes, and no evidence for decreased bioavailability of oral rifampicin in patients with diabetes.69 In Peru, no differences were recorded for 2 h and 6 h rifampicin plasma concentrations during the intensive phase of tuberculosis treatment between patients with or without diabetes.70 Overall, these and other studies suggest that diabetes could affect the pharmacokinetics of tuberculosis drugs. Additionally, a heavier bodyweight might decrease rifampicin exposure, especially during the continuation phase of tuberculosis treatment. Therefore, doses of tuberculosis drugs should be adjusted according to a patient’s weight, although this strategy might be difficult when fixed drug combinations are used. Even if diabetes is not associated with reduced drug concentrations, an increased rifampicin dose could improve tuberculosis treatment outcomes.71,72
Another option to improve tuberculosis treatment outcomes in patients with diabetes might be to extend treatment duration, a possibility that is advocated in some guidelines but has yet to be formally studied. However, toxic effects of first-line antituberculous drugs, especially peripheral neuropathy with respect to isoniazid and ocular toxic effects with respect to ethambutol, should be considered as these could be more common because of, or add to, complications of diabetes.73 Ethambutol dosing frequency should be decreased when patients with diabetes have reduced kidney function.
Treatment of diabetes in patients with tuberculosis
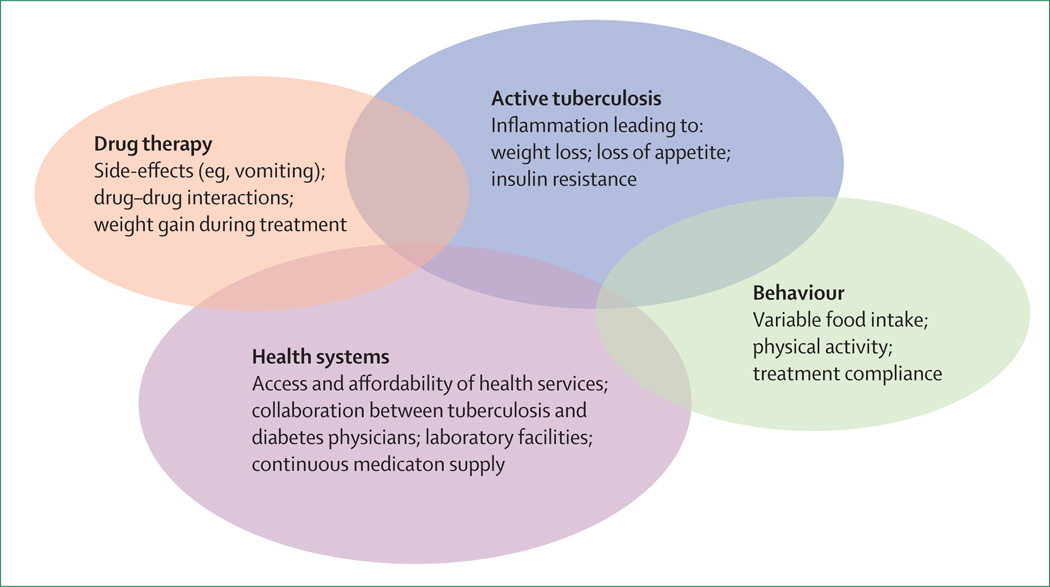
Optimal glycaemic control might improve outcomes of tuberculosis treatment and prevent many of the complications associated with diabetes. However, tuberculosis often leads to decreases in appetite, bodyweight, and physical activity (patients might be tired and therefore less active), all of which might affect glucose homoeostasis. Conversely, tuberculosis treatment can have opposite but unpredictable effects by decreasing inflammation, and increasing appetitie, body weight, and physical activity (figure 2). Although, some patients might have a paradoxical increase in inflammation or a small apetite as a side-effect of the tuberculosis drugs. Inflammation associated with active tuberculosis could cause insulin resistance, which is usually reversed once tuberculosis is treated. Additionally, good glycaemic control is dependent on the quality of the health-care system.
Figure 2.
Factors that affect glycaemic control for patients with diabetes during treatment for tuberculosis
To achieve good glycaemic control, an aggressive approach for diabetes management in patients with tuberculosis has been suggested.74 However, this approach should be balanced against the possible harm and additional efforts needed for implementation. Personalised pragmatic glycaemic targets might be needed that account for an array of factors, such as severity and prognosis of a given patient’s tuberculosis disease, risk of adverse events such as hypoglycaemia, duration of diabetes, comorbidities, age, patient capabilities and treatment preferences, and available resources.75
Frequent monitoring is needed to ensure good glycaemic control. Self-measurement of blood glucose is not feasible in many settings and for some patients. Clinic-based measurement of fasting blood glucose is not ideal for ambulatory care in busy clinics in tuberculosis-endemic settings, and random blood glucose and glycosuria measurements are less accurate than fasting blood glucose.76
With respect to the choice of diabetes drugs, possible drug–drug interactions should be taken into account, especially for rifampicin, the most important antituberculosis drug because it helped to shorten tuberculosis treatment time from 18 to 6 months. Drug resistance to rifampicin is associated with poorer treatment outcomes, worse than for any other tuberculosis drug. Rifampicin increases the hepatic metabolism of all sulphonylurea derivatives, the most widely used class of oral diabetes drugs worldwide. This effect on sulphonylurea derivatives has great inter-individual variation, which makes dose adjustments difficult and increases a patient’s risk of hyperglycaemia or hypoglycaemia. Inter-individual variation in the induction of the metabolism of diabetes drugs makes dose adjustment difficult when rifampicin treatment is interrupted or stopped; the same is true for most other oral antidiabetes drugs (appendix). Less is known about newer antidiabetes drug classes, and no data have been published from patients with tuberculosis treated with these drugs. Rifampicin probably has no effect on the exposure of glucagon-like peptide-1 receptor agonists and only a slight effect on dipeptidyl peptidase-4 inhibitors;77 however, poor availability and high costs of these drugs will restrict their use in tuberculosis-endemic settings.
Several other factors determine the choice of antidiabetes drugs to be used in patients with tuberculosis, such as availability, cost, ease of administration, and safety. Safety concerns include hypoglycaemia with sulphonylureas and insulin, lactic acidosis (especially under hypoxic conditions) with beguanides, and gastrointestinal complaints wih biguanides, meglitinides, and alpha-glucosidase inhibitors, and hypersensitivity to sulphonylureas (which might overlap with side-effects of antituberculous drugs). Insulin use at the start of tuberculosis treatment has been suggested;74 some national treatment guidelines (for instance in Indonesia) strongly suggest the use of insulin for diabetes in patients with tuberculosis even though no evidence base lends support to that approach. Because insulin is not metabolised, it has no pharmacokinetic interactions with rifampicin or other antituberculous drugs, but insulin has several potential drawbacks when used in under-resourced settings including cost, availability, storage, and delivery.
Metformin is a first-line drug for type 2 diabetes and does not usually lead to hypoglycaemia. Metformin is not metabolised by P450 enzymes,78 and thus its concentrations should not be decreased by an inductive effect of rifampicin on these metabolic enzymes. However, exposure to metformin could be affected because metformin is a substrate for human organic cation transporters and other transporters.77 Rifampicin increases the expression of organic cation transporter (OCT1) and hepatic uptake of metformin, leading to an enhanced glucose-lowering effect in healthy individuals.79 Possible disadvantages of metformin use in patients with tuberculosis include the risk of gastrointestinal side-effects and, very rarely, lactic acidosis.80,81
Drug–drug interactions are even more probable when patients with tuberculosis also have HIV, because many anti-HIV drugs are substrates of metabolic or transporter enzymes, or can change the activity of these enzymes. Similarly, toxicity profiles and side-effects of HIV, tuberculosis, and diabetes drugs might overlap. To our knowledge, no studies on the combined treatment of these three diseases have been published.
What is needed for patients with concurrent tuberculosis and diabetes?
Patients with tuberculosis and diabetes might need alternative interventions besides changes to their drug regimens. Counselling and education are advised for patients with tuberculosis and newly diagnosed diabetes. In an Indonesian study, 60% of cases of diabetes in patients with tuberculosis were newly diagnosed.82 To refer a patient with active tuberculosis to a separate diabetes clinic is not an option that appeals, both from a patient’s and an infectious control point of view. However, patients that need insulin, in particular, need to learn about glucose measurements and the symptoms and necessary action in case of hypoglycaemia. Intense engagement of patients with tuberculosis with the health system, especially early on in their treatment, provides an opportunity for repeated counselling including about lifestyle interventions (eg, nutrition, weight loss, smoking cessation, and physical activity).83 In the Pacific Islands region, extensive educational materials have been developed for patients with tuberculosis and diabetes.84
Patients with concurrent tuberculosis and diabetes might have specific needs during treatment follow-up. Monitoring of liver and kidney functions might need to be more intense than in patients with only tuberculosis. Although not part of routine practice in tuberculosis clinics, international guidelines recommend regular measurements of blood pressure and lipid profiles for individuals with diabetes, with low thresholds to start antihypertensive drugs or statin therapy. Aspirin is indicated for secondary prevention in patients with cardiovascular events and could be used as a general primary prevention strategy.83
To consider all of the aforementioned issues in some patients—such as those who are very unwell with tuberculosis or who have co-infection with HIV, drug-toxicity, substance misuse issues, psychiatric comorbidity, or poor compliance to tuberculosis treatment—is unrealistic. However, the long-term prognosis of patients with uncomplicated tuberculosis, or of those who quickly recover, might depend heavily on the management of their diabetes. A patient’s diabetes phenotype at the time of completion of their tuberculosis treatment—in terms of HbA1c concentrations, diabetes treatment, BMI, blood pressure, lipid profile, proteinuria, smoking, and alcohol consumption—will determine their long-term need for lifestyle interventions and antidiabetic, antihypertensive, lipid-lowering, anti platelet, and other drugs (panel 2). After completion of tuberculosis treatment, individuals with diabetes need continued diabetes management and a heightened alertness for tuberculosis symptoms because relapse is common in these patients.4
Panel 2: Clinical considerations for managing patients with concurrent tuberculosis and diabetes.
Infection control
Diabetes clinic might not be equipped or be poorly equipped for tuberculosis infection control
Tuberculosis treatment
Increased duration of treatment and weight-adjusted doses of antituberculous drugs might be necessary
Diabetes treatment
No evidence-based recommendations are available for diabetes therapy choice; different approaches might be needed for newly diagnosed and previously diagnosed diabetes
Drug–drug interactions
Rifampicin increases metabolism of antidiabetes drugs, except insulin and metformin; HIV treatment is likely to incur additional interactions
Treatment adherence
Disease symptoms, high pill load, and side-effects could compromise treatment adherence in patients with both diseases
Monitoring
Intense monitoring and follow-up might be needed because of increased likelihood of toxic effects of drugs, failure, and relapse rates; active tuberculosis screening might be worthwhile in patients with diabetes who previously had tuberculosis because of high recurrence rates
Glycaemic control
Potential logistical issues to measurement of glycaemic control in the tuberculosis clinic; decisions for adjustment of diabetes drug is dependent on glucose concentrations and various other factors
Coordination of care
Treatment delivery by separate clinics or different specialists could lead to conflicts in advice on treatment and safety issues
Counselling and education
Needs to address both tuberculosis and diabetes; intense counselling is needed for patients starting insulin therapy
Lifestyle and smoking
Patients should be actively engaged in adoption of healthy lifestyle changes to address issues such as smoking and obesity
Continuation of care
Patients with tuberculosis and newly diagnosed diabetes need to be referred for chronic diabetes care once tuberculosis treatment is completed
Implications for health service delivery and health economics
The integration of services for tuberculosis and diabetes should lead to better tuberculosis control and early diagnosis and treatment of diabetes, which will improve the alignment of treatment, monitoring, and counselling of patients with both diseases. Improved outcomes that result from such integration could lead to efficiency gains in terms of patient care. However, integration might overwhelm already overburdened and understaffed programmes.85 Unfortunately, diabetes management in low-income and middle-income countries rarely meets evidence-based standards.86 Very often the public’s awareness of both diseases is low, diagnostic services and trained specialists are scarce, and diabetes treatments are restricted because of the low supply and high cost of drugs.86,87 In many low-income and middle-income countries, diabetes monitoring and treatment are only partly covered by health insurance or government support. In Peru, for example, diabetes care for patients with tuberculosis is only covered for the duration of their tuberculosis treatment—after completion, the costs are the patient’s responsibility.
Although similar problems once existed for tuberculosis and HIV in under-resourced settings, substantial progress occurred during the past 10–20 years. Integration of services has led to increased uptake of services by patients, improved treatment outcomes, and decreased mortality rates.88,89 The experience and methods used to address the burden of tuberculosis and HIV—such as evidence-based clinical algorithms, measures to control tuberculosis infections, point-of-care diagnostics, so-called task-shifting, and innovative health service delivery (panel 3)—could be adapted for integrated tuberculosis and diabetes care. For instance, through use of standardised cohort recording and reporting methods in the tuberculosis and HIV DOTs (directly observed therapy, short course)99 and of real-time electronic medical record systems at the point of care (so called E-health), the rates of diabetes, measures of diabetes service performance (eg, proportion of patients with blood pressure measurements), and treatment results were reported for thousands of Palestinian refugees.100,101 Tuberculosis treatment programmes can also learn from patient’s self-monitoring experiences, with or without peer support, for use in diabetes care.102
Panel 3: Implementation of integrated services for concurrent tuberculosis and diabetes: lessons learned from HIV and tuberculosis.
Clinical algorithms
Evidence-based algorithms for bidirectional screening and combined treatment of tuberculosis and HIV have been developed40,45,46
Tuberculosis chemoprophylaxis
Tuberculosis chemoprophylaxis (isoniazid preventive therapy) is indicated for all patients with HIV once active tuberculosis is excluded; implementation might be easier in patients with diabetes as active tuberculosis can more reliably be excluded in patients with diabetes than in those with HIV; however, the efficacy of such preventive treatment is unknown50–52
Health promotion
Training materials and other techniques used for HIV alone and combined HIV and tuberculosis can be adapted to create methods for lifestyle interventions for patients with concurrent tuberculosis and diabetes
Tuberculosis infection control
Evidence-based policies are available for implementation of sound tuberculosis infection control in health-care facilities, including use of available spaces, separation of infectious patients, tuberculosis surveillance and preventive therapy for health workers, and environmental controls such as ventilation systems90
Decentralisation of health services
Mobile units have a high yield for combined screening of HIV, tuberculosis, diabetes, and hypertension in South Africa;91 linking to chronic care is challenging; successful community engagement (eg, in work sites and households) used for tuberculosis and HIV could be adapted for tuberculosis and diabetes
Human resources, task shifting
Task shifting from physician to non-physician and to lay health worker to combat growing burden of disease is safe and cost-effective91–94
Drug delivery
Combination of treatments for several diseases leads to strengthening of health systems (eg, supply chain, laboratory services) and increases accessibility and availability of care; tuberculosis and HIV treatment is free, diabetes treatment often is not
Point-of-care diagnosis
Point-of-care diagnosis of HIV and tuberculosis has led to improved access to care and early treatment; point-of-care HbA1c testing might have similar effects to increase diagnosis and care of diabetes in under-resourced settings95
Standardised recording and reporting
Cohort records and reports from the DOTs (directly observed therapy, short course) framework were adapted and used to monitor individuals with diabetes in Malawi;96 tuberculosis treatment cards and registers might need modifications to capture information on diabetes screening and diagnosis, just as with HIV
Adherence and retention to care
Various strategies have helped sustain long-term adherence and retention to care, including empowered patients,97 who are actively invovled in their own medical care, and mobile phone technology98
Assessments of the cost-effectiveness of bidirectional screening for tuberculosis and diabetes, and the integration of treatments for both diseases have not been done. An early diagnosis of diabetes and intense monitoring of blood glucose concentrations is expected to result in earlier and improved glycaemic control, leading to improved health outcomes. However, the overall costs of treatment for diabetes and associated comorbidities could increase. Screening tests for diabetes vary substantially in cost and resources needed (table 2); evidence from low-income and middle-income countries suggests that the most cost-effective approach might include a risk score with confirmatory blood testing103 (recommended by the International Diabetes Federation and the UK Health Technology Assessment Programme63,104). The presence of tuberculosis could be a contributor to such risk scores, although this effect has not been investigated. Tighter glycaemic control is resource intensive and might place a burden on patients in terms of out-of-pocket costs for care, which could affect adherence to treatment. A less intense schedule of glucose monitoring, preferably following the established decision points in tuberculosis treatment after 2 and 6 months, would offer substantial advantages in terms of costs and burden on the patient. An ongoing pragmatic randomised clinical trial (NCT02106039)105 in patients with tuberculosis and diabetes in Peru, Romania, South Africa, and Indonesia will define the requirements for good glycaemic control by comparison with standard versus intensified glucose monitoring.
Evidence is scarce with respect to the cost-effectiveness of screening for tuberculosis in patients with diabetes. The yield will probably be very low, except in regions with a very high incidence of tuberculosis.5,33,40,45 However, about half of people with diabetes are undiagnosed globally, and many people with diabetes in low-income and middle-income countries are not on appropriate treatments. Moreover, about 30% of patients with tuberculosis are undiagnosed. Thus bidirectional screening would decrease the proportion of undiagnosed individuals with tuberculosis or diabetes, and integrated screening and care will enable patients with both diseases to receive appropriate treatments.
Conclusions
The prevalence of diabetes in patients with tuberculosis will continue to increase as a result of the rising global burden of type 2 diabetes. Although screening patients with tuberculosis for diabetes is recommended, many questions are unanswered with respect to the best way to implement screening in different settings. Screening patients with diabetes for active tuberculosis could be considered but the best clinical algorithm needs to be identified and cost-effectiveness remains to be established. Good glycaemic control might improve health outcomes when tuberculosis and diabetes are simultaneously treated. Tuberculosis treatment monitoring might need to be more intensive if patients with tuberculosis also have diabetes. Additionally, other interventions should be considered, especially for patients with newly diagnosed diabetes and for people who need insulin. More evidence is needed to support screening and subsequent treatment for latent tuberculosis infections in patients with diabetes. Integration of health services could result in better tuberculosis prevention, an early diagnosis and start of treatment for diabetes, and improved care for concomitant disease. However, the practical and economic implications need further assessment before they are implemented and many key research questions need to be answered (panel 4). Clearly, there is a call to clinicians and researchers to generate the necessary evidence for improvements to patient services and policies with respect to combined tuberculosis and diabetes.
Panel 4: Key questions for future research in tuberculosis and diabetes.
What is the effect of glycaemic control (both short term and long term) on tuberculosis infection, active tuberculosis, and tuberculosis treatment outcomes?
What are the most feasible techniques or strategies for screening for diabetes in patients with tuberculosis and the converse?
How cost effective are strategies for screening and clinical management?
Is screening and prophylactic treatment of latent tuberculosis infection indicated for people with diabetes?
What is the possible benefit and what are the operational issues related to intensified monitoring of diabetes and its treatment in patients with tuberculosis? And what is the respective role of insulin versus metformin or other antidiabetic drugs?
What models of health service delivery can contribute to integration and sustainability of care for diabetes and tuberculosis in low-income and middle-income countries?
Supplementary Material
Search strategy and selection criteria.
We searched Medline, Embase, and Web of Science between each database’s inception date and May 31, 2014, for articles in English using the search terms (alone or in combination) “diabetes”, “tuberculosis”, “risk factors”, “outcomes”, “epidemiology”, “survival analysis”, “low and middle income countries (LMIC)”, “low resource”; “screening”, “blood glucose”, “fasting blood glucose”, “HbA1c”, “tuberculin test”, “IGRA”, “Xpert TB”, “computer-aided”, “point-of care”; “diabetes guidelines”, “patient management”, “treatment”, “treatment adherence”, “radiographic manifestations”, “pharmacokinetics”, “rifampicin” and other first line tuberculosis drugs in combination with each of the diabetes drugs; “cost”, “(health)-economic”, “cost-effectiveness”, “cost analysis”; “smoking”, “non-communicable disease”, “communicable disease”, “comorbidity”, “HIV”, “health system”, “task shifting”, “infection control”, “(chemo) prophylaxis”. We manually searched the references of individual articles for additional publications that were not previously identified. A predefined maximum of 110 articles was selected based on relevance to a pre-specified review outline, scientific strength, and date of publication. We did not have exclusion criteria; however, if many references were available the papers of a higher scientific quality and of a more recent date were chosen.
Acknowledgments
ALR, FP, CU-G, BA, NMP, PCH, RR, RA, JAC, and RvC are supported by the TANDEM project, which is funded by the European Union’s Seventh Framework Programme (FP7/2007–2013) under Grant Agreement Number 305279.105 CU-G is supported by Peru International Clinical, Operational, and Health Services Research and Training Award Network for AIDS/TB Research Training (National Institutes of Health Grant 1U2RTW007368-01A1 Fogarty International Center, Lima, Peru); the National Institutes of Health Office of the Director, Fogarty International Center, Office of AIDS Research, National Cancer Center, National Heart, Blood, and Lung Institute, and the National Institutes of Health Office of Research for Women’s Health through the Fogarty Global Health Fellows Program Consortium comprised of the University of North Carolina, John Hopkins, Morehouse and Tulane (1R25TW009340-01) and the American Recovery and Reinvestment Act. JAC is supported by the Higher Education Funding Council for England. RvC is supported by a VIDI grant from the Netherlands Foundation for Scientific Research.
Footnotes
See Online for appendix
Contributors
ALR, FP, JAC, and RvC did the literature review and wrote the first draft of the report. RA provided specific input for the section on pharmacokinetic interactions. All authors provided input to the report and approved the final version.
Declaration of interests
NMP reports grants from European Commission–Seventh Framework Programme (FP7), Romanian Executive Agency for higher education and university research, European Foundation for the Study of Diabetes, and Romanian Society of Diabetes Nutrition and Metabolic Diseases outside the submitted work. NMP received personal fees from MSD, Eli Lilly, Novo Nordisk, and Sanofioutside the submitted work. ALR, FP, CU-G, BA, SvdV, PCH, RR, DM, RA, JAC, and RvC declare no competing interests.
References
- 1.WHO. Global tuberculosis report 2013. Geneva: World Health Organization; 2013. [Google Scholar]
- 2.Jeon CY, Murray MB. Diabetes mellitus increases the risk of active tuberculosis: a systematic review of 13 observational studies. PLoS Med. 2008;5:e152. doi: 10.1371/journal.pmed.0050152. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Stevenson CR, Critchley JA, Forouhi NG, et al. Diabetes and the risk of tuberculosis: a neglected threat to public health? Chronic Illn. 2007;3:228–245. doi: 10.1177/1742395307081502. [DOI] [PubMed] [Google Scholar]
- 4.Baker MA, Harries AD, Jeon CY, et al. The impact of diabetes on tuberculosis treatment outcomes: a systematic review. BMC Med. 2011;9:81. doi: 10.1186/1741-7015-9-81. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Lönnroth K, Roglic G, Harries AD. Improving tuberculosis prevention and care through addressing the global diabetes epidemic: from evidence to policy and practice. Lancet Diabetes Endocrinol. 2014;2:730–739. doi: 10.1016/S2213-8587(14)70109-3. [DOI] [PubMed] [Google Scholar]
- 6.IDF. IDF Diabetes Atlas. sixth. Brussels: International Diabetes Federation; 2013. [Google Scholar]
- 7.Caminero JA, Sotgiu G, Zumla A, Migliori GB. Best drug treatment for multidrug-resistant and extensively drug-resistant tuberculosis. Lancet Infect Dis. 2010;10:621–629. doi: 10.1016/S1473-3099(10)70139-0. [DOI] [PubMed] [Google Scholar]
- 8.IDF. IDF Diabetes Atlas. first. Brussels: International Diabetes Federation; 2000. [Google Scholar]
- 9.Unwin NC, Alberti KG. Diabetes and the good, the bad and the ugly of globalization. Int Diabetes Monitor. 2007;19:6–11. [Google Scholar]
- 10.United Nations Population Division. An overview of urbanization, internal migration, population distribution and development in the world. New York: United Nations Population Division; 2008. [Google Scholar]
- 11.Hales CN, Barker DJP. Type 2 (non-insulin-dependent) diabetes mellitus: the thrifty phenotype hypothesis. Int J Epidemiol. 2013;42:1215–1222. doi: 10.1093/ije/dyt133. [DOI] [PubMed] [Google Scholar]
- 12.Wells JC. The thrifty phenotype: An adaptation in growth or metabolism? Am J Hum Biol. 2011;23:65–75. doi: 10.1002/ajhb.21100. [DOI] [PubMed] [Google Scholar]
- 13.Unwin N, Whiting D, Roglic G. Social determinants of diabetes and challenges of prevention. Lancet. 2010;375:2204–2205. doi: 10.1016/S0140-6736(10)60840-9. [DOI] [PubMed] [Google Scholar]
- 14.Faurholt-Jepsen D, Range N, Praygod G, et al. Diabetes is a risk factor for pulmonary tuberculosis: a case-control study from Mwanza, Tanzania. PLoS One. 2011;6:e24215. doi: 10.1371/journal.pone.0024215. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Alavi SM, Khoshkhoy MM. Pulmonary tuberculosis and diabetes mellitus: co-existence of both diseases in patients admitted in a teaching hospital in the southwest of Iran. Caspian J Intern Med. 2012;3:421–424. [PMC free article] [PubMed] [Google Scholar]
- 16.Balakrishnan S, Vijayan S, Nair S, et al. High diabetes prevalence among tuberculosis cases in Kerala, India. PLoS One. 2012;7:e46502. doi: 10.1371/journal.pone.0046502. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Viswanathan V, Kumpatla S, Aravindalochanan V, et al. Prevalence of diabetes and pre-diabetes and associated risk factors among tuberculosis patients in India. PLoS One. 2012;7:e41367. doi: 10.1371/journal.pone.0041367. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Goldhaber-Fiebert JD, Jeon CY, Cohen T, Murray MB. Diabetes mellitus and tuberculosis in countries with high tuberculosis burdens: individual risks and social determinants. Int J Epidemiol. 2011;40:417–428. doi: 10.1093/ije/dyq238. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Young F, Wotton CJ, Critchley JA, Unwin NC, Goldacre MJ. Increased risk of tuberculosis disease in people with diabetes mellitus: record-linkage study in a UK population. J Epidemiol Community Health. 2012;66:519–523. doi: 10.1136/jech.2010.114595. [DOI] [PubMed] [Google Scholar]
- 20.Basoğlu OK, Bacakoğlu F, Cok G, Sayiner A, Ates M. The oral glucose tolerance test in patients with respiratory infections. Monaldi Arch Chest Dis. 1999;54:307–310. [PubMed] [Google Scholar]
- 21.Oluboyo PO, Erasmus RT. The significance of glucose intolerance in pulmonary tuberculosis. Tubercle. 1990;71:135–138. doi: 10.1016/0041-3879(90)90010-6. [DOI] [PubMed] [Google Scholar]
- 22.Gearhart MM, Parbhoo SK. Hyperglycemia in the critically ill patient. AACN Clin Issues. 2006;17:50–55. doi: 10.1097/00044067-200601000-00007. [DOI] [PubMed] [Google Scholar]
- 23.Takasu N, Yamada T, Miura H, et al. Rifampicin-induced early phase hyperglycemia in humans. Am Rev Respir Dis. 1982;125:23–27. doi: 10.1164/arrd.1982.125.1.23. [DOI] [PubMed] [Google Scholar]
- 24.Niemi M, Backman JT, Neuvonen M, Neuvonen PJ, Kivistö KT. Effects of rifampin on the pharmacokinetics and pharmacodynamics of glyburide and glipizide. Clin Pharmacol Ther. 2001;69:400–406. doi: 10.1067/mcp.2001.115822. [DOI] [PubMed] [Google Scholar]
- 25.Chang JT, Dou HY, Yen CL, et al. Effect of type 2 diabetes mellitus on the clinical severity and treatment outcome in patients with pulmonary tuberculosis: a potential role in the emergence of multidrug-resistance. J Formos Med Assoc. 2011;110:372–381. doi: 10.1016/S0929-6646(11)60055-7. [DOI] [PubMed] [Google Scholar]
- 26.Jiménez-Corona ME, Cruz-Hervert LP, García-García L, et al. Association of diabetes and tuberculosis: impact on treatment and post-treatment outcomes. Thorax. 2013;68:214–220. doi: 10.1136/thoraxjnl-2012-201756. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Reis-Santos B, Locatelli R, Horta BL, et al. Socio-demographic and clinical differences in subjects with tuberculosis with and without diabetes mellitus in Brazil—a multivariate analysis. PLoS One. 2013;8:e62604. doi: 10.1371/journal.pone.0062604. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Faurholt-Jepsen D, Range N, PrayGod G, et al. Diabetes is a strong predictor of mortality during tuberculosis treatment: a prospective cohort study among tuberculosis patients from Mwanza, Tanzania. Trop Med Int Health. 2013;18:822–829. doi: 10.1111/tmi.12120. [DOI] [PubMed] [Google Scholar]
- 29.Viswanathan V, Vigneswari A, Selvan K, Satyavani K, Rajeswari R, Kapur A. Effect of diabetes on treatment outcome of smear-positive pulmonary tuberculosis—a report from South India. J Diabetes Complications. 2014;28:162–165. doi: 10.1016/j.jdiacomp.2013.12.003. [DOI] [PubMed] [Google Scholar]
- 30.Reed GW, Choi H, Lee SY, et al. Impact of diabetes and smoking on mortality in tuberculosis. PLoS One. 2013;8:e58044. doi: 10.1371/journal.pone.0058044. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Odone A, Houben RMGJ, Lönnroth K, White RG. The effect of diabetes and undernutrition trends on reaching 2035 global tuberculosis targets. Lancet Diabetes Endocrinol. 2014;2:754–764. doi: 10.1016/S2213-8587(14)70164-0. [DOI] [PubMed] [Google Scholar]
- 32.Al-Quwaidhi AJ, Pearce MS, Sobngwi E, Critchley JA, O’Flaherty M. Comparison of type 2 diabetes prevalence estimates in Saudi Arabia from a validated Markov model against the International Diabetes Federation Diabetes Atlas and other modelling studies. Diabetes Res Clin Pract. 2014;103:496–503. doi: 10.1016/j.diabres.2013.12.036. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Jeon CY, Harries AD, Baker MA, et al. Bi-directional screening for tuberculosis and diabetes: a systematic review. Trop Med Int Health. 2010;15:1300–1314. doi: 10.1111/j.1365-3156.2010.02632.x. [DOI] [PubMed] [Google Scholar]
- 34.Cox ME, Edelman D. Tests for screening and diagnosis of type 2 diabetes. Clin Diabetes. 2009;27:132–138. [Google Scholar]
- 35.WHO. Fluorescent light-emitting diode (LED) microscopy for diagnosis of tuberculosis. Geneva: World Health Organization Press; 2007. [PubMed] [Google Scholar]
- 36.Kumpatla S, Sekar A, Achanta S, et al. Characteristics of patients with diabetes screened for tuberculosis in a tertiary care hospital in South India. Public Health Action. 2013;3:23–28. doi: 10.5588/pha.13.0035. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Steingart KR, Sohn H, Schiller I, et al. Xpert MTB/RIF assay for pulmonary tuberculosis and rifampicin resistance in adults. Cochrane Database Syst Rev. 2013;1 doi: 10.1002/14651858.CD009593.pub2. CD009593. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Van’t Hoog AH, Langendam MW, Mitchell E, et al. A systematic review of the sensitivity and specificity of symptom- and chest-radiography screening for active pulmonary tuberculosis in HIV-negative persons and persons with unknown HIV status. Geneva: World Health Organisation; 2013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Li L, Lin Y, Mi F, et al. Screening of patients with tuberculosis for diabetes mellitus in China. Trop Med Int Health. 2012;17:1294–1301. doi: 10.1111/j.1365-3156.2012.03068.x. [DOI] [PubMed] [Google Scholar]
- 40.Lin Y, Li L, Mi F, et al. Screening patients with diabetes mellitus for tuberculosis in China. Trop Med Int Health. 2012;17:1302–1308. doi: 10.1111/j.1365-3156.2012.03069.x. [DOI] [PubMed] [Google Scholar]
- 41.Amin S, Khattak MI, Ghulam S, Wazir MN. Frequency of pulmonary tuberculosis in patients with diabetes mellitus. Gomal J Med Sci. 2011;9:163–165. [Google Scholar]
- 42.Gounder S, Harries AD. Screening tuberculosis patients for diabetes mellitus in Fiji: notes from the field. Public Health Action. 2012;2:145–147. doi: 10.5588/pha.12.0058. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Kibirige D, Ssekitoleko R, Mutebi E, Worodria W. Overt diabetes mellitus among newly diagnosed Ugandan tuberculosis patients: a cross sectional study. BMC Infect Dis. 2013;13:122. doi: 10.1186/1471-2334-13-122. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Olayinka A, Yetunde K, Anthonia O. Prevalence of diabetes mellitus in persons with tuberculosis in a tertiary health centre in Lagos, Nigeria. Indian J Endocrinol Metab. 2013;17:486–489. doi: 10.4103/2230-8210.111646. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.India Diabetes Mellitus-Tuberculosis Study Group. Screening of patients with diabetes mellitus for tuberculosis in India. Trop Med Int Health. 2013;18:646–654. doi: 10.1111/tmi.12083. [DOI] [PubMed] [Google Scholar]
- 46.India Tuberculosis-Diabetes Study Group. Screening of patients with tuberculosis for diabetes mellitus in India. Trop Med Int Health. 2013;18:636–645. doi: 10.1111/tmi.12084. [DOI] [PubMed] [Google Scholar]
- 47.Prakash BC, Ravish KS, Prabhakar B, et al. Tuberculosis-diabetes mellitus bidirectional screening at a tertiary care centre, south India. Public Health Action. 2013;3:18–22. doi: 10.5588/pha.13.0032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Jaeger S, Karargyris A, Candemir S, et al. Automatic screening for tuberculosis in chest radiographs: a survey. Quant Imaging Med Surg. 2013;3:89–99. doi: 10.3978/j.issn.2223-4292.2013.04.03. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Pai M, Zwerling A, Menzies D. Systematic review: T-cell-based assays for the diagnosis of latent tuberculosis infection: an update. Ann Intern Med. 2008;149:177–184. doi: 10.7326/0003-4819-149-3-200808050-00241. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Mansilla Bermejo MJ, Sanz Gil MJ, Moraleda Velasco P, Alvarez Prado A, Carbayo Garcia JJ, Mata Guijarro F. Tuberculin test in diabetic patients in a health center. Aten Primaria. 1995;16:154–57. (in Spanish) [PubMed] [Google Scholar]
- 51.Vega Torres RA, Conde JG, Díaz M. Prevalence of tuberculin reactivity and risk factors for the development of active tuberculosis upon admission to a nursing home. P R Health Sci J. 1996;15:275–277. [PubMed] [Google Scholar]
- 52.Nwabudike LC, Ionescu-Tirgoviste C. Intradermal reactions to purified protein derivative in patients with diabetes mellitus. Rom J Intern Med. 2005;43:127–132. [PubMed] [Google Scholar]
- 53.Lesnichii AV, Karpina LZ. Experience with the chemoprophylaxis of pulmonary tuberculosis in diabetes mellitus patients. Probl Tuberk. 1969;47:1–3. (in Russian) [PubMed] [Google Scholar]
- 54.Pfaffenberg R, Jahler H. Isoniazid & recurrence of tuberculosis in diabetics. Z Tuberk. 1958;111:167–173. (in German) [PubMed] [Google Scholar]
- 55.WHO and International Union against TB and Lung Disease. Collaborative framework for care and control of tuberculosis and diabetes. Geneva: World Health Organization; 2011. [PubMed] [Google Scholar]
- 56.Pacific Island TB Controllers Association. [accessed March 1, 2014];United States Affiliated Pacific Islands—standards for the management of tuberculosis and diabetes. 2012 http://www.spc.int/tb/component/content/article/75-pacific-standards-for-management-of-tb-and-diabetes.
- 57.International Union of Tuberculosis and Lung Disease. [accessed March 1, 2014];Screening all TB patients for diabetes mellitus: from pilot to policy change. http://old.theunion.org/index.php/en/newsroom/news/item/2310-screening-all-tb-patients-for-diabetes-mellitus-from-pilot-to-policy-change.
- 58.McCowen KC, Malhotra A, Bistrian BR. Stress-induced hyperglycemia. Crit Care Clin. 2001;17:107–124. doi: 10.1016/s0749-0704(05)70154-8. [DOI] [PubMed] [Google Scholar]
- 59.Kumpatla S, Aravindalochanan V, Rajan R, Viswanathan V, Kapur A. Evaluation of performance of A1c and FPG tests for screening newly diagnosed diabetes defined by an OGTT among tuberculosis patients-a study from India. Diabetes Res Clin Pract. 2013;102:60–64. doi: 10.1016/j.diabres.2013.08.007. [DOI] [PubMed] [Google Scholar]
- 60.Adepoyibi T, Weigl B, Greb H, Neogi T, McGuire H. New screening technologies for type 2 diabetes mellitus appropriate for use in tuberculosis patients. Public Health Action. 2103;3:10–17. doi: 10.5588/pha.13.0036. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.WHO and IDF. Screening for Type 2 Diabetes. Report of a World Health Organization and International Diabetes Federation meeting. Geneva: World Health Organization and International Diabetes Federation; 2003. [Google Scholar]
- 62.Echouff o-Tcheugui JB, Ali MK, Griffin SJ, Narayan KM. Screening for type 2 diabetes and dysglycemia. Epidemiol Rev. 2011;33:63–87. doi: 10.1093/epirev/mxq020. [DOI] [PubMed] [Google Scholar]
- 63.Waugh N, Scotland G, McNamee P, et al. Screening for type 2 diabetes: literature review and economic modelling. Health Technol Assess. 2007;11:1–125. doi: 10.3310/hta11170. [DOI] [PubMed] [Google Scholar]
- 64.Dooley KE, Tang T, Golub JE, Dorman SE, Cronin W. Impact of diabetes mellitus on treatment outcomes of patients with active tuberculosis. Am J Trop Med Hyg. 2009;80:634–639. [PMC free article] [PubMed] [Google Scholar]
- 65.Ruslami R, Aarnoutse RE, Alisjahbana B, van der Ven AJ, van Crevel R. Implications of the global increase of diabetes for tuberculosis control and patient care. Trop Med Int Health. 2010;15:1289–1299. doi: 10.1111/j.1365-3156.2010.02625.x. [DOI] [PubMed] [Google Scholar]
- 66.Chiang CY, Lee JJ, Chien ST, et al. Glycemic control and radiographic manifestations of tuberculosis in diabetic patients. PLoS One. 2014;9:e93397. doi: 10.1371/journal.pone.0093397. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Alisjahbana B, Sahiratmadja E, Nelwan EJ, et al. The effect of type 2 diabetes mellitus on the presentation and treatment response of pulmonary tuberculosis. Clin Infect Dis. 2007;45:428–435. doi: 10.1086/519841. [DOI] [PubMed] [Google Scholar]
- 68.Nijland HMJ, Ruslami R, Stalenhoef JE, et al. Exposure to rifampicin is strongly reduced in patients with tuberculosis and type 2 diabetes. Clin Infect Dis. 2006;43:848–854. doi: 10.1086/507543. [DOI] [PubMed] [Google Scholar]
- 69.Ruslami R, Nijland HMJ, Adhiarta IGN, et al. Pharmacokinetics of antituberculosis drugs in pulmonary tuberculosis patients with type 2 diabetes. Antimicrob Agents Chemother. 2010;54:1068–1074. doi: 10.1128/AAC.00447-09. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Requena-Méndez A, Davies G, Ardrey A, et al. Pharmacokinetics of rifampin in Peruvian tuberculosis patients with and without comorbid diabetes or HIV. Antimicrob Agents Chemother. 2012;56:2357–2363. doi: 10.1128/AAC.06059-11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.van Ingen J, Aarnoutse RE, Donald PR, et al. Why do we use 600 mg of Rifampicin in tuberculosis treatment? Clin Infect Dis. 2011;52:e194–e199. doi: 10.1093/cid/cir184. [DOI] [PubMed] [Google Scholar]
- 72.Ruslami R, Ganiem AR, Dian S, et al. Intensified regimen containing rifampicin and moxifloxacin for tuberculous meningitis: an open-label, randomised controlled phase 2 trial. Lancet Infect Dis. 2013;13:27–35. doi: 10.1016/S1473-3099(12)70264-5. [DOI] [PubMed] [Google Scholar]
- 73.Hall RG, Leff RD, Gumbo T and the Insights from the Society of Infectious Diseases Pharmacists. Treatment of active pulmonary tuberculosis in adults: current standards and recent advances. Pharmacotherapy. 2009;29:1468–1481. doi: 10.1592/phco.29.12.1468. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Niazi AK, Kalra S. Diabetes and tuberculosis: a review of the role of optimal glycemic control. J Diabetes Metab Disord. 2012;11:28. doi: 10.1186/2251-6581-11-28. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Inzucchi SE, Bergenstal RM, Buse JB, et al. and the American Diabetes Association (ADA), and the European Association for the Study of Diabetes (EASD) Management of hyperglycemia in type 2 diabetes: a patient-centered approach: position statement of the American Diabetes Association (ADA) and the European Association for the Study of Diabetes (EASD) Diabetes Care. 2012;35:1364–1379. doi: 10.2337/dc12-0413. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Rotchford AP, Rotchford KM, Machattie T, Gill GV. Assessing diabetic control—reliability of methods available in resource poor settings. Diabet Med. 2002;19:195–200. doi: 10.1046/j.1464-5491.2002.00601.x. [DOI] [PubMed] [Google Scholar]
- 77.Tornio A, Niemi M, Neuvonen PJ, Backman JT. Drug interactions with oral antidiabetic agents: pharmacokinetic mechanisms and clinical implications. Trends Pharmacol Sci. 2012;33:312–322. doi: 10.1016/j.tips.2012.03.001. [DOI] [PubMed] [Google Scholar]
- 78.Sun H, Scott DO. Impact of genetic polymorphisms of cytochrome P450 2 C (CYP2C) enzymes on the drug metabolism and design of antidiabetics. Chem Biol Interact. 2011;194:159–167. doi: 10.1016/j.cbi.2011.08.011. [DOI] [PubMed] [Google Scholar]
- 79.Cho SK, Yoon JS, Lee MG, et al. Rifampin enhances the glucose-lowering effect of metformin and increases OCT1 mRNA levels in healthy participants. Clin Pharmacol Ther. 2011;89:416–421. doi: 10.1038/clpt.2010.266. [DOI] [PubMed] [Google Scholar]
- 80.Salpeter SR, Greyber E, Pasternak GA, Salpeter EE. Posthumous. Risk of fatal and nonfatal lactic acidosis with metformin use in type 2 diabetes mellitus. Cochrane Database Syst Rev. 2010;4 doi: 10.1002/14651858.CD002967.pub4. CD002967. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Scheen AJ, Paquot N. Metformin revisited: a critical review of the benefit-risk balance in at-risk patients with type 2 diabetes. Diabetes Metab. 2013;39:179–190. doi: 10.1016/j.diabet.2013.02.006. [DOI] [PubMed] [Google Scholar]
- 82.Alisjahbana B, van Crevel R, Sahiratmadja E, et al. Diabetes mellitus is strongly associated with tuberculosis in Indonesia. Int J Tuberc Lung Dis. 2006;10:696–700. [PubMed] [Google Scholar]
- 83.American Diabetes Association. Executive summary: standards of medical care in diabetes—2014. Diabetes Care. 2014;37(suppl 1):S5–S13. doi: 10.2337/dc14-S005. [DOI] [PubMed] [Google Scholar]
- 84.Australian Respiratory Council. [accessed March 1, 2014];Key Messages for TB DM. 2012 http://www.thearc.org.au/TBandDiabetes.aspx.
- 85.Marais BJ, Lönnroth K, Lawn SD, et al. Tuberculosis comorbidity with communicable and non-communicable diseases: integrating health services and control efforts. Lancet Infect Dis. 2013;13:436–448. doi: 10.1016/S1473-3099(13)70015-X. [DOI] [PubMed] [Google Scholar]
- 86.Grant P. Management of diabetes in resource-poor settings. Clin Med. 2013;13:27–31. doi: 10.7861/clinmedicine.13-1-27. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Beran D, Yudkin JS. Looking beyond the issue of access to insulin: what is needed for proper diabetes care in resource poor settings. Diabetes Res Clin Pract. 2010;88:217–221. doi: 10.1016/j.diabres.2010.03.029. [DOI] [PubMed] [Google Scholar]
- 88.Lawn SD, Campbell L, Kaplan R, Little F, Morrow C, Wood R and the IeDEA-Southern Africa. Delays in starting antiretroviral therapy in patients with HIV-associated tuberculosis accessing non-integrated clinical services in a South African township. BMC Infect Dis. 2011;11:258. doi: 10.1186/1471-2334-11-258. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Hermans SM, Castelnuovo B, Katabira C, et al. Integration of HIV and TB services results in improved TB treatment outcomes and earlier prioritized ART initiation in a large urban HIV clinic in Uganda. J Acquir Immune Defic Syndr. 2012;60:e29–e35. doi: 10.1097/QAI.0b013e318251aeb4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.WHO. WHO policy on TB infection control in health-care facilities, congregate settings and households. Geneva: World Health Organization; 2009. [PubMed] [Google Scholar]
- 91.Govindasamy D, Kranzer K, van Schaik N, et al. Linkage to HIV, TB and non-communicable disease care from a mobile testing unit in Cape Town, South Africa. PLoS One. 2013;8:e80017. doi: 10.1371/journal.pone.0080017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Van Rie A, Patel MR, Nana M, et al. Integration and task shifting for TB/HIV care and treatment in highly resource-scarce settings: one size may not fit all. J Acquir Immune Defic Syndr. 2014;65:e110–e117. doi: 10.1097/01.qai.0000434954.65620.f3. [DOI] [PubMed] [Google Scholar]
- 93.Mdege ND, Chindove S, Ali S. The effectiveness and cost implications of task-shifting in the delivery of antiretroviral therapy to HIV-infected patients: a systematic review. Health Policy Plan. 2013;28:223–236. doi: 10.1093/heapol/czs058. [DOI] [PubMed] [Google Scholar]
- 94.Callaghan M, Ford N, Schneider H. A systematic review of task-shifting for HIV treatment and care in Africa. Hum Resour Health. 2010;8:8. doi: 10.1186/1478-4491-8-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Hirst J, Farmer A, Stevens R, Price C. Systematic review and meta-analysis of the accuracy of point-of-care HbA1c tests. [accessed March 1, 2014];PROSPERO: CRD42013006678. 2013 http://www.crd.york.ac.uk/PROSPERO/display_record.asp?ID=CRD42013006678.
- 96.Allain TJ, van Oosterhout JJ, Douglas GP, et al. Applying lessons learnt from the ‘DOTS’ tuberculosis model to monitoring and evaluating persons with diabetes mellitus in Blantyre, Malawi. Trop Med Int Health. 2011;16:1077–1084. doi: 10.1111/j.1365-3156.2011.02808.x. [DOI] [PubMed] [Google Scholar]
- 97.Behforouz HL, Farmer PE, Mukherjee JS. From directly observed therapy to accompagnateurs: enhancing AIDS treatment outcomes in Haiti and in Boston. Clin Infect Dis. 2004;38(suppl 5):S429–S436. doi: 10.1086/421408. [DOI] [PubMed] [Google Scholar]
- 98.Lester RT, Ritvo P, Mills EJ, et al. Effects of a mobile phone short message service on antiretroviral treatment adherence in Kenya (WelTel Kenya1): a randomised trial. Lancet. 2010;376:1838–1845. doi: 10.1016/S0140-6736(10)61997-6. [DOI] [PubMed] [Google Scholar]
- 99.Sullivan T, Ben Amor Y. The co-management of tuberculosis and diabetes: challenges and opportunities in the developing world. PLoS Med. 2012;9:e1001269. doi: 10.1371/journal.pmed.1001269. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Khader A, Ballout G, Shahin Y, et al. Treatment outcomes in a cohort of Palestine refugees with diabetes mellitus followed through use of E-Health over 3 years in Jordan. Trop Med Int Health. 2014;19:219–223. doi: 10.1111/tmi.12241. [DOI] [PubMed] [Google Scholar]
- 101.Khader A, Farajallah L, Shahin Y, et al. Cohort monitoring of persons with diabetes mellitus in a primary healthcare clinic for Palestine refugees in Jordan. Trop Med Int Health. 2012;17:1569–1576. doi: 10.1111/j.1365-3156.2012.03097.x. [DOI] [PubMed] [Google Scholar]
- 102.Fisher EB, Boothroyd RI, Coufal MM, et al. Peer support for self-management of diabetes improved outcomes in international settings. Health Aff (Millwood) 2012;31:130–139. doi: 10.1377/hlthaff.2011.0914. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Mohan V, Deepa R, Deepa M, Somannavar S, Datta M. A simplified Indian diabetes risk score for screening for undiagnosed diabetic subjects. J Assoc Physicians India. 2005;53:759–763. [PubMed] [Google Scholar]
- 104.International Diabetes Federation. [accessed March 1, 2014];Clinical guidelines task force global guideline for type 2 diabetes. 2012 http://www.idf.org/webdata/docs/IDF%20GGT2D.pdf.
- 105.van Crevel R, Dockrell HM for the TANDEM Consortium. TANDEM: understanding diabetes and tuberculosis. Lancet Diabetes Endocrinol. 2014;2:270–272. doi: 10.1016/S2213-8587(14)70011-7. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.