Abstract
Objective
The objective of this study was to compare the potential Influence of imaging variables on surgical margins after preoperative radioactive seed localization (RSL) and wire localization (WL) techniques.
Materials and Methods
A total of 565 women with 660 breast lesions underwent RSL or WL between May 16, 2012, and May 30, 2013. Patient age, lesion type (mass, calcifications, mass with associated calcifications, other), lesion size, number of seeds or wires used, surgical margin status (close positive or negative margins), and reexcision and mastectomy rates were recorded.
Results
Of 660 lesions, 127 (19%) underwent RSL and 533 (81%) underwent WL pre-operatively. Mean lesion size was 1.8 cm in the RSL group and 1.8 cm in the WL group (p = 0.35). No difference in lesion type was identified in the RSL and WL groups (p = 0.63). RSL with a single seed was used in 105 of 127 (83%) RSLs compared with WL with a single wire in 349 of 533 (65%) WLs (p = 0.0003). The number of cases with a close positive margin was similar for RSLs (26/127, 20%) and WLs (104/533, 20%) (p = 0.81). There was no difference between the RSL group and the WL group in close positive margin status (20% each, p = 0.81), reexcision rates (20% vs 16%, respectively; p = 0.36), or mastectomy rates (6% each, p = 0.96). Lesions containing calcifications were more likely to require more than one wire (odds ratio [OR], 4.44; 95% CI, 2.8–7.0) or more than one seed (OR, 7.03; 95% CI, 1.6–30.0) when compared with masses alone (p < 0.0001). Increasing lesion size and the presence of calcifications were significant predictors of positive margins, whereas the use of more than one wire or seed was not (OR, 0.9; 95% CI, 0.5–1.5) (p = 0.75).
Conclusion
Close positive margin, reexcision, and mastectomy rates remained similar in the WL group and RSL group. The presence of calcifications and increasing lesion size increased the odds of a close positive margin in both the WL and RSL groups, whereas the use of one versus more than one seed or wire did not.
Keywords: 125I seed localization, breast cancer, mammography, ultrasound-guided wire localization, wire localization
The increased utilization of and improvement in screening mammography programs have led to an increase in the number of nonpalpable imaging-detected breast cancers [1, 2]. The standard technique for preoperative localization of nonpalpable lesions uses a needle-wire assembly (wire localization [WL]) under mammographic, stereotactic, sonographic, or MRI guidance. More recently, the technique of preoperative radioactive seed localization (RSL) as an alternative to WL of breast lesions has gained increased acceptance and frequency of use [3–5]. Although multiple studies have shown success rates using RSL at least equal to those of WL for achieving negative surgical margins, others have suggested improved negative margin and reexcision rates with RSL [3–5]. These latter studies have also reported improved scheduling, shorter localization procedure times, and shorter operative times with the use of RSL versus WL [3–9].
Prelocalization planning differs between the RSL and WL techniques, reflecting the different equipment and surgical techniques required [5, 10–12]. It is unclear whether factors such as lesion type (mass vs calcifications vs architectural distortion), lesion size, or method of preoperative localization influence the success of localization, with success determined by the incidence of single-step surgical excision of the targeted lesion [4, 6, 7, 10]. Therefore, we undertook this retrospective study to investigate whether lesion-specific and technical factors such as lesion type, lesion size, and the number of seeds or wires used in the localization procedure had an impact on the surgical margins.
Materials and Methods
Patient Demographics and Lesion Imaging Features
The study time period encompassed the initial time period of RSL implementation at the University of Texas M. D. Anderson Cancer Center. A waiver of informed consent was granted by the institutional review board for this HIPAA-compliant study. All mammography- and ultrasound-guided localizations performed at our institution using either RSL or WL that occurred between May 16, 2012, and May 30, 2013, were retrospectively reviewed. The decision for the patient to undergo RSL or WL was based on the operating surgeon's preference, which was also contingent on whether the surgeon had completed institutional radiation safety and RSL credentialing steps. Patient age, lesion type (mass, calcifications, mass with associated calcifications, other), lesion size based on imaging, and lesion histopathology were recorded using patients' electronic records. Both available imaging reports and images were reviewed for the image analysis by two dedicated fellowship-trained breast imaging radiologists with 13 and 15 years' breast imaging experience. The imaging reports were used to standardize lesion type. Lesion size was based on imaging reports when available; when lesion size differed between imaging modalities, a consensus agreement was reached for lesion size. Lesions were classified according to their mammographic appearance using the American College of Radiology BI-RADS lexicon [13]. Lesions were described as a mass, calcifications, a mass with associated calcifications, or other (i.e., focal asymmetry, architectural distortion, or postbiopsy clip; and intraductal filling defect for localizations performed under ductography guidance). The term “postbiopsy clip” was used to describe lesions that were totally removed at biopsy or for malignant lesions that showed a complete response to neoadjuvant chemotherapy with no residual lesion identified on imaging at the time of localization.
The method of localization (RSL vs WL) and the number of seeds or wires used were recorded for each procedure. Localization was used to describe procedures that used a single seed or wire preoperatively, and the term “bracketing” was reserved for cases that required the use of more than one seed or wire. The seed-to-target distance was measured on the final preoperative mammograms obtained immediately after RSL and was recorded to determine whether any significant migration of the seed had occurred. Seed migration was deemed significant if the seed-to-target distance was greater than 1 cm per our institutional protocol. Procedure reports routinely contain the distance from the seed or needle to the lesion and a graph that depicts the seed direction and distance. This information was recorded from the procedure reports; when the distance between the seed and target was more than 1 cm, this information was recorded from the post-procedure mammograms. At the time of surgical removal, specimen radiographs were reviewed to document the removal of the seed and the targeted lesion. The procedure time was calculated using the acquisition time of the first and final images obtained during each procedure.
Radioactive Seed Localization Planning and Technique
Before lesion localization, images were reviewed 1–8 days before surgery by one of 14 dedicated breast radiologists with 3–20 years' experience. The method of localization, length of the required needle-seed system, and number of seeds required to localize or bracket the lesion were determined, and a written directive was generated. All RSL procedures were performed 1–5 days before the day of surgery.
All needle localizations were performed on the same day as surgery. An identical process was used for needle localization except that there was no written directive. Mammography-guided localizations were performed on a Lorad M3 (Hologic) or DMR (GE Healthcare) unit. Ultrasound-guided procedures were performed using a 13-7–MHz or 10-5–MHz linear-array transducer connected to an Antares scanner (Siemens Healthcare) by one of 12 fellowship-trained dedicated breast radiologists with 3–35 years' experience in breast ultrasound-guided procedures. The operator, using sterile technique, used a preloaded and presterilized needle seed assembly (18-gauge) that included a single seed with radioactivity ranging from 0.100 to 0.200 mCi (3.7–7.4 MBq) and was occluded with bone wax (Advantage I-125 IsoAid) (Fig. 1). Seed placement was confirmed by a handheld Geiger counter and a two-view postprocedure mammogram that was then annotated for the surgeon's review. A schematic depicting the breast anatomy, distance from the seed to the targeted lesion, and distance from the seed to the overlying skin was provided for each patient.
Fig. 1.
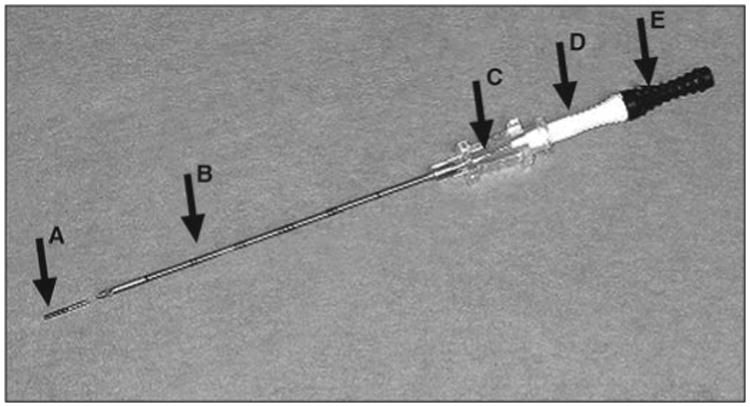
Photograph shows preloaded needle seed system used for radioactive seed localization. A = bioabsorbable glycolic lactide pellet interwoven with seed, B = 18-gauge needle, C = needle hub, D = security stopper, E = stylet.
In the operative suite, the operating surgeon used the Neoprobe and the Gamma Detection System 2300 (Devicor Medical Products) optimized to 27 keV to identify the seed and the targeted lesion. Because 125I and 99mTc have different kiloelectron volts, the Neoprobe 2300 was used to individually distinguish the two isotopes if a concomitant sentinel lymph node biopsy was also being performed. Lesion removal was confirmed by intraoperative specimen radiography documenting all deployed seeds within the specimen, the presence of counts within the specimen, and no residual counts in the breast.
Surgical Margins
For all lesions, an intraoperative margin assessment was performed using specimen radiography, as previously described [14, 15]. Final pathology reports were used to assess the surgical margin status. Margins described as a distance of 2 mm or greater to the inked specimen margin were considered negative (negative margin). A distance of less than 2 mm was classified as a close margin, and lesions touching the inked specimen margin were considered positive (positive margin), according to the published consensus guidelines on surgical margins for breast-conserving surgery by the Society of Surgical Oncology and the American Society for Radiation Oncology [16]. For the purpose of this study, close and positive margins were grouped together and are referred to as “close positive margin” to allow a binary outcome of margin status as either negative or positive.
The reexcision rate was recorded. The type of final surgery was recorded as segmental mastectomy or mastectomy.
Statistical Analysis
Lesions were treated independently if patients had more than one lesion because each lesion has its own independent margin status outcome. Frequencies and percentages are displayed for categoric variables, and means and SDs are provided for continuous variables. The chi-square test was used to test the univariate association between outcomes such as margin status by localization type. Multivariate logistic regression was used to predict margin status and the number of needles or seeds used. ROC curves were drawn to find the one optimal cut point in lesion size described by Youden [17] to predict the odds of using more than one needle or seed; 95% CIs were provided for the AUC under the ROC curve using the two exact method described by DeLong et al. [18]. All statistical analyses were performed using SAS (version 9.3, SAS Institute) for Microsoft Windows and R (version 3.1, The R Foundation).
Results
A total of 565 patients underwent preoperative imaging-guided WL or RSL of 660 breast lesions. The median patient age for the RSL group was 60 years (age range, 37–83 years). The median patient age for the WL group was 64 years (age range, 23–88 years). The mean lesion size was 1.8 cm (SD, 1.3 cm) for the RSL group and 1.8 cm (SD, 1.7 cm) for the WL group (p = 0.35). The distribution of the lesions by preoperative histopathology diagnosis is summarized in Table 1.
Table 1. Preoperative Histopathologic Subtypes of 660 Breast Lesions in 565 Patients.
| Lesion Type | No. (%) of Lesions | |
|---|---|---|
|
| ||
| Wire Localization | Radioactive Seed Localization | |
|
| ||
| IDC | 91 (17) | 24 (19) |
| IDC with associated DCIS | 187 (35) | 59 (46) |
| DCIS | 119 (22) | 22 (17) |
| Mixed invasive ductal and lobular carcinoma | 16 (3) | 4 (3) |
| IDC, ILC, and DCIS | 3 (1) | 1 (1) |
| ILC | 35 (7) | 6 (5) |
| ILC and DCIS | 4 (1) | 1 (1) |
| Other cancer typesa | 6 (1) | 1 (1) |
| High-risk lesionsb | 72 (14) | 9 (7) |
|
| ||
| Total | 533 (100c) | 127 (100) |
Note—IDC = invasive ductal carcinoma, DCIS = ductal carcinoma in situ, ILC = invasive lobular carcinoma.
Other cancer types are sarcoma, metaplastic carcinoma, adenoid cystic carcinoma, invasive papillary carcinoma, and malignant phyllodes tumor.
High-risk lesions are atypical ductal hyperplasia (ADH), atypical lobular hyperplasia, lobular carcinoma in situ, papillary neoplasm, and radial scar. No upgrades to breast cancer occurred in the radioactive seed localization group, whereas three high-risk lesions were upgraded to breast cancer in the wire localization group: Two atypical papillomas were upgraded to papillary DCIS, and one ADH was upgraded to DCIS.
Total percentage does not add up to 100 because of rounding.
Of the 660 lesions in our study, 127 (19%) were localized with RSL and 533 (81%) were localized with WL.
Localization Time
The median localization time was 15 minutes for both RSL and WL, with a range of 10–30 minutes for RSL and 12–30 minutes for WL.
Comparison of Lesion Types and Number of Wires or Seeds
Of the 533 WL procedures, 405 (76%) were performed under mammography guidance and 128 (24%) with ultrasound guidance. Of the 127 RSLs, 57 (44.8%) were performed with mammographic guidance and 68 (53.5%) with ultrasound guidance. Two RSLs (1.6%) were performed using ductography guidance. The distribution of lesion types by mammographic imaging is summarized in Table 2. The proportions of lesion types between WL and RSL groups were similar: masses (48% vs 52%, respectively), calcifications and masses with associated calcifications (35% vs 34%), and other lesion types (17% vs 14%) (p = 0.6307).
Table 2. Distribution of Lesion Type by Mammography in Radioactive Seed Localization Group and Wire Localization Group.
| Lesion Type by Mammography | No. (%) of Lesions | pa | ||
|---|---|---|---|---|
|
| ||||
| Radioactive Seed Localization | Wire Localization | Total | ||
|
| ||||
| Mass | 66 (52) | 256 (48) | 322 (49) | |
| Calcification or mass with associated calcifications | 43 (34) | 187 (35) | 230 (35) | |
| Otherb | 18 (14) | 90 (17) | 108 (16) | |
|
| ||||
| Total | 127 (100) | 533 (100) | 660 (100) | 0.6307 |
Chi-square test.
Other includes focal asymmetry, architectural distortion, postbiopsy clip, and intraductal filling defect.
A single seed was used in 105 of 127 (83%) RSL procedures compared with the utilization of a single wire in 350 of 533 (66%) WL procedures (p = 0.0003). Two seeds were used in 14 of 127 (11%) RSL procedures compared with the use of two wires in 141 of 533 (26%) WL procedures. Three or more seeds were used in eight of 127 (6%) RSL procedures compared with the use of three or more wires in 42 of 533 (8%) WL procedures (Figs. 2 and 3). Overall, single-seed RSL was more commonly used in the absence of a significant difference of lesion size (p = 0.35).
Fig. 2.
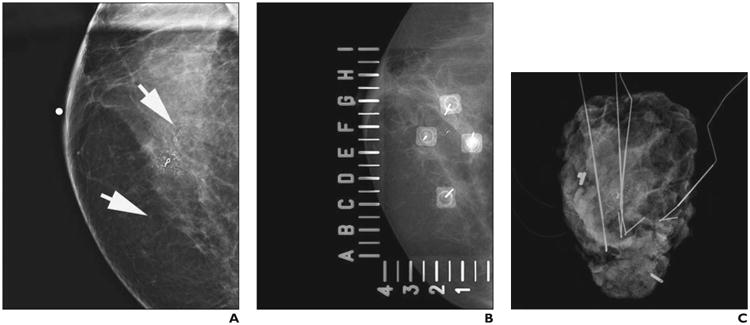
62-year-old woman with ductal carcinoma in situ (DCIS), high nuclear grade, and solid and cribriform subtype with comedonecrosis.
A, Craniocaudal magnification mammogram shows calcifications (arrows) span 12- to 2-o'clock positions in superior breast.
B, Craniocaudal spot mammographic view obtained during wire localization shows four Kopans localization needles used to bracket calcifications. Alphanumeric grid is used to perform localization.
C, En bloc specimen radiograph shows removal of all four needles and calcifications. Additional tissue was obtained intraoperatively on basis of interpretation of margins of sliced specimen (not shown). Surgical margins were focally positive for DCIS (i.e., close positive margin). Patient underwent reexcision segmental mastectomy with negative final margins.
Fig. 3.
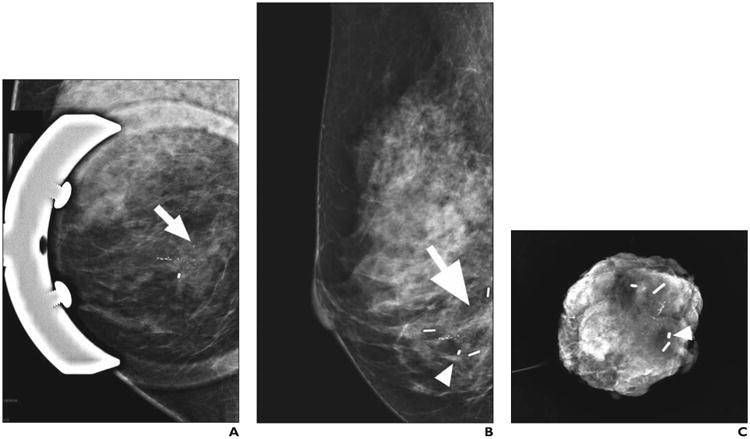
50-year-old woman with right breast ductal carcinoma in situ, high nuclear grade, with comedonecrosis and associated microcalcifications.
A, Lateromedial magnification mammogram shows calcifications (arrow) in inferior breast.
B, Lateromedial mammogram confirms placement of three radioactive seeds to bracket calcifications (arrow) in posterior, inferior, and anterior directions and postbiopsy clip (arrowhead).
C, En bloc specimen radiograph shows that seeds, clip (arrowhead), and residual calcifications have been removed from patient. Surgical margins were wide and negative.
The presence of calcifications on mammography was a significant factor Influencing localization planning. Calcifications alone and masses with associated calcifications were significantly more likely to require more than one wire (odds ratio [OR], 4.44; 95% CI, 2.8–7.0) or more than one seed (OR, 7.03; 95% CI, 1.6–30.0) for localization when compared with masses alone (p < 0.0001).
In all patients, wires or seeds were placed within 1 cm of the targeted lesion or within 1 cm of the targeted lesion margins for bracketing. Significant seed migration of greater than 1 cm occurred in one of 127 RSLs (0.8%) in which a clip was being localized. The clip was removed at the time of surgery, and reexcision was not warranted.
Surgical Margin Results
The initial surgery was segmental mastectomy or excisional biopsy for all patients. In both the RSL and WL groups, close positive margin was observed in 20% of excisions (26/127 lesions in RSL group vs 104/533 in WL group) (p = 0.81). Subsequent reexcision was performed for 112 of 660 (17%) lesions. Reexcision was performed in 25 of 127 (20%) lesions in the RSL group and in 87 of 533 (16%) lesions in the WL group (p = 0.36). Mastectomy was performed in 30 of 533 lesions in the WL group and seven of 127 lesions in the RSL group (6% each, p = 0.96). Surgical margins for the RSL group versus the WL group are summarized in Table 3.
Table 3. Surgical Margins for Radioactive Seed Localization Group Versus Wire Localization Group.
| Preoperative Localization | Margins, No. (%) of Lesions | pa | Reexcision, No. (%) of Lesions | pa | Mastectomy, No. (%) of Lesions | pa | |||
|---|---|---|---|---|---|---|---|---|---|
|
|
|
|
|||||||
| Negative Margin | Close Positive Margin | No | Yes | No | Yes | ||||
|
| |||||||||
| Wire localization (n = 533) | 429 (80) | 104 (20) | 446 (84) | 87 (16) | 503 (94) | 30 (6) | |||
| Radioactive seed localization (n = 127) | 101 (80) | 26 (20) | 102 (80) | 25 (20) | 120 (94) | 7 (6) | |||
|
| |||||||||
| Total (n = 660) | 530 (80) | 130 (20) | 0.81 | 548 (83) | 112 (17) | 0.36 | 623 (94) | 37 (6) | 0.96 |
Chi-square test.
Multivariate logistic regression analysis was performed to evaluate imaging factors that may Influence surgical margin status (Table 4). Increasing lesion size and the presence of calcifications were significant predictors of close positive margin. The use of more than one wire or more than one seed was not found to decrease the probability of a close positive margin (OR, 0.9; 95% CI, 0.5–1.5) (p = 0.75).
Table 4. Multivariate Logistic Regression Analysis to Assess Factors Increasing the Odds of Close Positive Margin.
| Factors That May Influence Margin Status | OR for Close Positive Margin | 95% CI for OR | p |
|---|---|---|---|
|
| |||
| Radioactive seed localization vs wire localization (consistency) | 1.10 1 | 0.666–1.821 | 0.7074 |
| Lesion size per centimeter | 1.258 | 1.107–1.429 | 0.0004 |
| More than one wire or seed vs single wire or single seed | 0.924 | 0.565–1.512 | 0.7531 |
| Mammographic calcifications within lesiona | 2.476 | 1.575–3.893 | < 0.0001 |
Note—OR = odds ratio.
Mammographic lesion type was calcification or mass with associated calcifications.
Discussion
Our findings suggest that although RSL is equivalent to WL with regard to surgical margin status, reexcision rates, and final mastectomy rates, fewer single-seed RSLs were performed than single-wire WLs in an equivalent number of masses and calcified lesions. A possible explanation for this outcome is the technique used for excision. Surgeons had the capability to optimize the site of surgical incision. Furthermore, since they had the ability to have constant, real-time orientation to targeted lesion using Neoprobe, they could better tailor the excision procedure [3, 5].
Another possible explanation is that multiple seeds placed in close proximity (i.e., within 1 cm) may emit overlapping signals, thus hampering the surgeon's ability to differentiate their location and thus potentially complicating the operative procedure.
In our institution, radiologists routinely preview the breast imaging studies 1–8 days before the day of RSL or WL and formulate a plan of localization on the basis of the targeted lesion type, lesion size, presence of calcifications, and need for a single or multiple localization seeds or wires. We found a higher association of close positive margin in both groups with increasing lesion size and lesions with associated calcifications. These findings are in agreement with previous studies showing larger lesion size and the presence of ductal carcinoma in situ (DCIS) as factors for increasing numbers of cases with positive surgical margins [19]. In our study, despite using a single seed for similarly sized lesions for which more than one wire was used in the WL group, our results do not indicate an increase in the number of cases with close positive margin in the RSL group.
Although using fewer seeds for same-sized lesions would result in a slight decrease in cost, the true cost savings in the RSL procedures are the results of decreased delays from uncoupling of radiology and surgery department schedules and of shorter surgical procedure times due to the surgical technique used [20–22]. In a recent study, Sharek et al. [21] reported that the greatest benefit of RSL was eliminating the operating room delays and unnecessary operating room time caused by delays in radiology procedures. In addition, RSL has been noted to improve the effciency of scheduling radiology staff and to decrease interventional wait times for biopsy patients. A detailed cost analysis per patient was not part of our study in the auditing of this initial set of our cases. Prior published studies investigating the cost of WL versus RSL showed that, in a bundled payment system, RSL results in a modest reduction of cost per patient over WL and in a slightly increased margin [20] based on reported differences in the rates of positive surgical margins and the resulting differences in the rates of additional surgeries between the two forms of localization. Although a fee-for-service system showed a moderate loss of revenue per patient with RSL, this loss of revenue per patient was largely because of lower reexcision rates, which reflect overall better patient outcomes and decreased total health care cost.
The RSL technique in which a 125I radiopaque titanium seed with a half-life of 59 days was placed within a targeted lesion in a patient's breast in lieu of the traditional WL was first described by Gray et al. [22] in 2001. With activities ranging from 0.100 to 0.200 mCi (3.7–7.4 MBq), seeds can be placed up to 5 days before surgery, which greatly increases scheduling flexibility for both radiologists and patients. This increased flexibility of RSLs removes the dependency of the surgery schedule on the radiology schedule of WLs. Multiple studies have consistently shown at least equal success using RSL compared with WL for achieving negative surgical margins, and some studies actually suggest improved negative margins and reexcision rates with RSL [3–9].
Our results show similar procedure times for both RSL and WL (median, 15 minutes), whereas prior studies have shown shorter localization procedure times and operative times with the use of RSL versus WL [5–8]. A possible explanation for this difference in findings is that our patient cohort was composed of the initial 127 RSL procedures performed at our institution, most of which were training cases for the radiologists and surgeons to learn and perfect the technique. Therefore, the implantation times for these cases are slightly longer than for our current practice. In addition, preprocedure and postprocedure patient and needle surveys, which are performed according to our radiation safety guidelines using a Geiger counter, slightly increase the overall procedure or in room time for the patient. Multiple prior studies have listed advantages of RSL over WL for scheduling, operating theater time, and ease of the procedure [3–9]. Significant wire migration occurring between the time of insertion and the time of surgery has been reported [22–24]. To our knowledge, there have been no reports of significant radioactive seed migration. Because of these factors, after our initial training period, approximately 50% of our preoperative localizations are now being performed using RSL and 50% using WL depending on the operating surgeon's preference. In tandem, both the localization and surgical procedure times for RSL have decreased since the initial 100 cases.
Our study has limitations. First, the retrospective study design over a limited study period limits the power of the data presented. During this period, most patients undergoing localization for breast-conserving therapy were still undergoing traditional WL. In addition, both the radiologists and surgeons were undergoing training for RSL during the study period, which may have increased the relative percentage of patients who had a close positive margin using RSL and the seed insertion time in the radiology department. Finally, a greater percentage of patients in the WL group underwent mammography-guided localization compared with the RSL group. A possible reason for this difference may be because approximately 40% of patients in our practice receive preoperative neoadjuvant therapy. At surgery, the imaging footprint of residual disease in patients undergoing neoadjuvant therapy is frequently too vague to be localized by ultrasound. Therefore, mammography is used to preoperatively plan and perform the localization. Because the time period of this retrospective study reflects our initial implementation of RSL, more patients with a history of neoadjuvant chemotherapy were likely included in the WL group than in the RSL group, leading to more mammography-guided WLs.
In summary, in our study, single-seed RSL was more commonly performed for equivalent-sized lesions that required the use of multiple wires, whereas RSL and WL showed similar results for close positive margin status, reexcision rates, and mastectomy rates. These results show that RSL is as effective as WL in the localization of malignancy and may even impact procedure times with potential downstream cost reduction. The presence of calcifications increased the odds of a close positive margin. Bracketing with more than one seed may be required for larger lesions and for lesions associated with calcifications.
Acknowledgments
Supported in part by the Cancer Center Support Grant (NCI Grant P30 CA016672).
Footnotes
Based on a presentation at the Radiological Society of North America 2014 annual meeting, Chicago, IL.
References
- 1.Cady B, Stone MD, Schuler JG, Thakur R, Wanner MA, Lavin PT. The new era in breast cancer: invasion, size, and nodal involvement dramatically decreasing as a result of mammographic screening. Arch Surg. 1996;131:301–308. doi: 10.1001/archsurg.1996.01430150079015. [DOI] [PubMed] [Google Scholar]
- 2.Skinner KA, Silberman H, Sposto R, Silverstein MJ. Palpable breast cancers are inherently different from nonpalpable breast cancers. Ann Surg Oncol. 2001;8:705–710. doi: 10.1007/s10434-001-0705-1. [DOI] [PubMed] [Google Scholar]
- 3.Lovrics PJ, Goldsmith CH, Hodgson N, et al. A multicentered, randomized, controlled trial comparing radioguided seed localization to standard wire localization for nonpalpable, invasive and in situ breast carcinomas. Ann Surg Oncol. 2011;18:3407–3414. doi: 10.1245/s10434-011-1699-y. [DOI] [PubMed] [Google Scholar]
- 4.Lovrics PJ, Cornacchi SD, Vora R, Goldsmith CH, Kahnamoui K. Systematic review of radioguided surgery for non-palpable breast cancer. Eur J Surg Oncol. 2011;37:388–397. doi: 10.1016/j.ejso.2011.01.018. [DOI] [PubMed] [Google Scholar]
- 5.Jakub JW, Gray RJ, Degnim AC, Boughey JC, Gardner M, Cox CE. Current status of radioactive seed for localization of non palpable breast lesions. Am J Surg. 2010;199:522–528. doi: 10.1016/j.amjsurg.2009.05.019. [DOI] [PubMed] [Google Scholar]
- 6.Sung JS, King V, Thornton CM, et al. Safety and efficacy of radioactive seed localization with I-125 prior to lumpectomy and/or excisional biopsy. Eur J Radiol. 2013;82:1453–1457. doi: 10.1016/j.ejrad.2013.04.008. [DOI] [PubMed] [Google Scholar]
- 7.Barentsz MW, van den Bosch MA, Veldhuis WB, et al. Radioactive seed localization for non-palpable breast cancer. Br J Surg. 2013;100:582–588. doi: 10.1002/bjs.9068. [DOI] [PubMed] [Google Scholar]
- 8.Murphy JO, Moo TA, King TA, et al. Radioactive seed localization compared to wire localization in breast-conserving surgery: initial 6-month experience. Ann Surg Oncol. 2013;20:4121–4127. doi: 10.1245/s10434-013-3166-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Diego EJ, Soran A, McGuire KP, et al. Localizing high-risk lesions for excisional breast biopsy: a comparison between radioactive seed localization and wire localization. Ann Surg Oncol. 2014;21:3268–3272. doi: 10.1245/s10434-014-3912-2. [DOI] [PubMed] [Google Scholar]
- 10.Chadwick DR, Shorthouse AJ. Wire-directed localization biopsy of the breast: an audit of results and analysis of factors Influencing therapeutic value in the treatment of breast cancer. Eur J Surg Oncol. 1997;23:128–133. doi: 10.1016/s0748-7983(97)80005-4. [DOI] [PubMed] [Google Scholar]
- 11.Kepple J, Van Zee KJ, Dowlatshahi K, Henry-Tillman RS, Israel PZ, Klimberg VS. Minimally invasive breast surgery. J Am Coll Surg. 2004;199:961–975. doi: 10.1016/j.jamcollsurg.2004.07.032. [DOI] [PubMed] [Google Scholar]
- 12.Staradub VL, Rademaker AW, Morrow M. Factors Influencing outcomes for breast conservation therapy of mammographically detected malignancies. J Am Coll Surg. 2003;196:518–524. doi: 10.1016/S1072-7515(02)01833-1. [DOI] [PubMed] [Google Scholar]
- 13.D'Orsi CJ, Sickles EA, Mendelson EB, et al. ACR BI-RADS Atlas, Breast Imaging Reporting and Data System. Reston, VA: American College of Radiology; 2013. [Google Scholar]
- 14.Erguvan-Dogan B, Whitman GJ, Nguyen VA, et al. Specimen radiography in confirmation of MRI-guided needle localization and surgical excision of breast lesions. AJR. 2006;187:339–344. doi: 10.2214/AJR.05.0422. [DOI] [PubMed] [Google Scholar]
- 15.Chagpar A, Yen T, Sahin A, et al. Intraoperative margin assessment reduces reexcision rates in patients with ductal carcinoma in situ treated with breast-conserving surgery. Am J Surg. 2003;186:371–377. doi: 10.1016/s0002-9610(03)00264-2. [DOI] [PubMed] [Google Scholar]
- 16.Moran MS, Schnitt SJ, Giuliano AE, et al. Society of Surgical Oncology; American Society for Radiation Oncology Society of Surgical Oncology-American Society for Radiation Oncology consensus guideline on margins for breast-conserving surgery with whole-breast irradiation in stages I and II invasive breast cancer. J Clin Oncol. 2014;32:1507–1515. doi: 10.1200/JCO.2013.53.3935. [DOI] [PubMed] [Google Scholar]
- 17.Youden WJ. Index for rating diagnostic tests. Cancer. 1950;3:32–35. doi: 10.1002/1097-0142(1950)3:1<32::aid-cncr2820030106>3.0.co;2-3. [DOI] [PubMed] [Google Scholar]
- 18.DeLong ER, DeLong DM, Clarke-Pearson DL. Comparing the areas under two or more correlated receiver operating characteristic curves: a nonpara-metric approach. Biometrics. 1988;44:837–845. [PubMed] [Google Scholar]
- 19.Cabioglu N, Hunt KK, Sahin AA, et al. Role for intraoperative margin assessment in patients undergoing breast-conserving surgery. Ann Surg Oncol. 2007;14:1458–1471. doi: 10.1245/s10434-006-9236-0. [DOI] [PubMed] [Google Scholar]
- 20.Loving VA, Edwards DB, Roche KT, et al. Monte Carlo simulation to analyze the cost-benefit of radioactive seed localization versus wire localization for breast-conserving surgery in fee-for-service health care systems compared with accountable care organizations. AJR. 2014;202:1383–1388. doi: 10.2214/AJR.13.11368. [DOI] [PubMed] [Google Scholar]
- 21.Sharek D, Zuley ML, Zhang JY, Soran A, Ahrendt GM, Ganott MA. Radioactive seed localization versus wire localization for lumpectomies: a comparison of outcomes. AJR. 2015;204:872–877. doi: 10.2214/AJR.14.12743. [DOI] [PubMed] [Google Scholar]
- 22.Gray RJ, Salud C, Nguyen K, et al. Randomized prospective evaluation of a novel technique for biopsy or lumpectomy of nonpalpable breast lesions: radioactive seed versus wire localization. Ann Surg Oncol. 2001;8:711–715. doi: 10.1007/s10434-001-0711-3. [DOI] [PubMed] [Google Scholar]
- 23.Davis PS, Wechsler RJ, Feig SA, March DE. Migration of breast biopsy localization wire. AJR. 1988;150:787–788. doi: 10.2214/ajr.150.4.787. [DOI] [PubMed] [Google Scholar]
- 24.Homer MJ, Pile-Spellman ER. Needle localization of occult breast lesions with a curved-end retractable wire: technique and pitfalls. Radiology. 1986;161:547–548. doi: 10.1148/radiology.161.2.3763930. [DOI] [PubMed] [Google Scholar]


