Abstract
Objective
Many patients with irritable bowel syndrome IBS not only have abdominal pain but also may suffer from visceral hypersensitivity and heighted visceral nociception. Moreover, IBS has few effective therapeutic agents and mechanisms of disease are unclear. Our goals were to (i) identify microRNA (miRNA) expression, signalling and targets in human colon (controls; patients with IBS); (ii) verify in vitro, IBS-associated changes in miRNAs, especially miR-199, which is complementary to the transient receptor potential vanilloid type 1 (TRPV1) gene; and (iii) determine whether modulating the expression of miRNAs in vivo, especially miR-199, reverses associated changes and pathological hallmarks of visceral hypersensitivity via TRPV1 signalling.
Design
We evaluated 45 patients with diarrhoea-predominant IBS (IBS-D) and 40 controls with (1) visceral pain severity score and (2) colonoscopy with biopsies. miRNA expression was evaluated in human colon following miRNA array analysis. Luciferase assays were done to confirm relationships between miR-199 and TRPV1 expression. A rat model of visceral hypersensitivity was used to study miR-199 and its target gene (TRPV1) expression in dorsal root ganglion (DRG) and colon in vivo.
Results
Gut miR-199a/b expression in IBS-D was significantly decreased, which correlated directly with both increased visceral pain scores and TRPV1 expression. In vivo upregulation of miR-199a by intraperitoneal injection of lenti-miR-199a precursors decreased visceral hypersensitivity via diminished TRPV1 signalling.
Conclusions
Decreased colonic miR-199a/b correlates with visceral pain in patients with IBS-D. Similarly, reduced miR-199a expression in rat DRG and colon tissue is associated with heightened visceral hypersensitivity. In vivo upregulation of miR-199a decreases visceral pain via inhibition of TRPV1 signalling. Thus, miR-199 precursors may be promising therapeutic candidates for the treatment in patients with visceral pain.
Keywords: IRRITABLE BOWEL SYNDROME
Significance of this study.
What is already known on this subject?
Increased neuronal expression of transient receptor potential vanilloid type 1 (TRPV1) has been reported in colonic biopsies of patients with IBS.
TRPV1 modulates visceral sensitivity and mechanosensation in animal models of IBS following stress or colonic inflammation.
Aberrantly expressed microRNAs are present in colonic biopsies of several GI tract diseases and disorders such as IBD and IBS.
What are the new findings?
Colonic miR-199 is decreased in patients with diarrhoea-predominant IBS (IBS-D) with visceral pain and directly correlates with increased colonic TRPV1 expression and inversely with visceral pain scores.
TRPV1 is a bona fide target of miR-199.
Decreased miR-199 levels are present in the colon and dorsal root ganglion of an animal model of visceral hypersensitivity.
Administration of miR-199 (lenti-miR-199 precursor) reverses visceral nociception in an animal model of visceral hypersensitivity.
How might it impact on clinical practice in the foreseeable future?
This study provides evidence in patients with IBS-D that decreased colonic miR-199 expression contributes to chronic visceral pain and nociception via upregulation of TRPV1 expression.
Decreased miR-199 is a key factor that may drive increased TRPV1 expression and results in chronic visceral pain in patients with IBS.
There is a potential role for miR-199 precursors, which may ultimately be used in vivo to dampen TRPV1 expression and subsequently reduce symptoms in patients with IBS.
Introduction
IBS is one of the most frequent GI disorders characterised by chronic abdominal pain and an alteration in bowel habits with diarrhoea and/or constipation.1 Despite its high prevalence, the pathophysiological mechanisms that lead to chronic symptoms in IBS are poorly understood. Some patients with IBS demonstrate enhanced responses to distension of the gut lumen, that is, visceral hypersensitivity (VH), which may account for symptoms of urgency, bloating and abdominal pain.2 3 The factors that trigger abdominal pain and VH are uncertain, but several mechanisms have been postulated such as inflammation or postinjury sensitisation.4 5 In response to a triggering event such as inflammation, trauma or environmental stress, the primary visceral afferents to the gut become sensitised, leading to chronic visceral pain in patients with IBS.6
Recent evidence suggests the involvement of the transient receptor potential vanilloid type 1 (TRPV1) protein in visceral pain. It is a non-selective, ligand-gated, cation channel that is expressed on peripheral primary afferent sensory neurons. When activated by heat, capsaicin, acidic pH, resinferatoxin and/or inflammatory mediators, these channels produce a burning sensation and/or pain.7 TRPV1 has been shown to increase visceral sensitivity and mechanically induced sensations (mechanosensation) in animal models following stress or adult and neonatal colonic inflammation.8–12 Patients with IBS have increased TRPV1 expression in colonic biopsies, which correlates with abdominal pain.13 Thus, aberrant activity of the TRPV1 receptor may play an important role in the pathogenesis of abdominal pain and GI symptoms in IBS.13 14
Another factor that is likely involved in these mechanisms is microRNA (miRNA). Particular sets of miRNAs are key regulators of gene expression throughout the digestive tract.15 16 We reported aberrant miR-29a expression in the gut tissue of patients with IBS with increased intestinal permeability.17 Here we present evidence that miR-199 is involved in visceral pain modulation in IBS. We focused our studies on miR-199a/b for several reasons: (1) based on our TaqMan miRNA real-time PCR assay, it was the most downregulated miRNA among the group of miRNAs altered; (2) both miR-199a and miR-199b have the same predicted target (TRPV1), which is an important modulator of visceral pain/hypersensitivity; and (3) the miR-199 family has various levels of complementarity between the target sequence (TRPV1) and miR-199a and miR-199b. Although pathophysiological roles for aberrant miRNA expression in GI disorders have been postulated, how specific miRNAs modulate nociceptive neurotransmitters such as TRPV1 has not been previously reported. In this study, we examined the possible mechanisms for upregulation of TRPV1 signalling by miRNAs. Our study objectives were as follows: (i) to evaluate visceral pain and gut miRNA expression in patients with diarrhoea-predominant IBS (IBS-D); (ii) to use an animal model of VH to study molecular changes in the dorsal root ganglion (DRG) and the colon; (iii) to determine whether induction of miR-199 in vivo can reverse VH; and (iv) to evaluate predicted targets of miR-199a/b (TRPV1) as critical signalling molecules involved in the regulation of visceral pain/hypersensitivity. Achievement of these objectives will lead to a better understanding of the pathophysiological mechanisms underlying visceral pain in patients with IBS and provide a basis for future development of miRNA-based therapies for IBS.18
Materials and methods
Human subjects
Patients with IBS-D and healthy controls were recruited via posted advertisements. The diagnosis of IBS-D was made using the ROME III criteria and exclusion of organic disease.1 During the introductory session, participants underwent (1) physical examination, (2) lactulose breath test for bacterial overgrowth (see online supplementary methods)19 and (3) blood draw for tissue transglutaminase antibody to rule out coeliac sprue. Subjects who qualified completed a visceral pain questionnaire and underwent colonoscopy within 1 week of the introductory session. The visceral pain questionnaire was a standardised, 0–100 visual analogue scale that was used to have patients rate their level of abdominal pain on a continuous line anchored from 0 (no pain) to 100 (most severe pain). Random biopsies were obtained from the ascending, transverse and descending colon for H&E staining and RNA and molecular studies. Standard pathological criteria were used to determine the presence of microscopic inflammation in the colonic tissues.
miRNA analysis
Total RNA was isolated from tissue biopsies by Trizol (Invitrogen, Carlsbad, California, USA) according to the manufacturer’s protocol. RNA was labelled and hybridised on miRNA microarray chips as previously described.17
TNBS animal model and visceral hypersensitivity testing
Male Fisher 344 rats received an intracolonic infusion (80 mg/kg) of 2,4,6-trinitrobenzene sulfonic acid (TNBS) in 50% ethanol to produce colitis.20 21 Thirty days after TNBS colitis was healed, colonic distension with a barostat was performed to measure the mean nociceptive threshold for each rat (see online supplementary methods).20
In vivo lentiviral delivery of miR-199a precursor
A lentivirus encoding the miR-199a precursor (miR-199a) and scrambled controls (scr-miR) were obtained from System Biosciences. For in vivo miRNA delivery, 18 male Fisher 344 rats with VH (weighing 100–150 g) received lentivirus containing either GFP and miR-199a expression vector (lenti-miR-199a) or GFP and control miRNA expression vector (lenti-miR-control). The lentivirus was introduced by two intraperitoneal injections daily; each containing 1 mL lentivirus solution containing 1×109 IFU in media, 4 µg/mL polybrene and 1× phosphate buffered saline per rat. The rats were sacrificed at 3 and 8 days (lentivirus-miR-control, each time point n=4; lenti-miR-199a-precusor, each time point n=5) following VH testing.
Quantitative real-time PCR
Reverse transcription was done using 0.5 µg of total RNA, and TaqMan MicroRNA Reverse Transcription Kits (Applied Biosystems, Foster City, California, USA). The primers and probes for real-time PCR were designed using Primer Express software and synthesised at Applied Biosystems (Foster City, California, USA).
Immunoblotting and ELISA assay
Protein extraction, western blot and ELISA assay were done on human colon tissue and rat DRG/colon tissue to detect TRPV1 expression.
Cell culture
Human neuronal cells (LUHMES, ATCC-CRL-2927, ATCC, Manassas, Virginia, USA) were used for this study. The cells were cultured as recommended by the suppliers, and the original growth protocols were followed.
Transductions and reagents
Human neuronal cells LUHMES (ATCC CRL-2927) were plated onto a 24-well plate, which were sequentially precoated with 50 µg/mL poly-L-ornithine (Sigma, cat. no. P-3655) and then with 1 µg/mL human fibronectin (Sigma, cat. no. F-0895) and cultured at 37°C overnight. Cells were transduced with 10 µL of 100× lentivirus encoding miR-199a/b precursors and control precursor using TransDux reagent (System Biosciences, Mountain View, California, USA).
Luciferase assay
Luciferase assays were done 48 h post-transfection following the manufacturer’s protocol (Dual-luciferase reporter assay system; Promega, Madison, Wisconsin, USA). Renilla luciferase activity was normalised to firefly luciferase activity for each sample (see online supplementary methods).
Confocal fluorescence microscopy
Human neuronal cells LUHMES (ATCC CRL-2927) were plated onto Nunc Lab-Tek II Chamber Slide System (Thermo Fisher Scientific, Waltham, Massachusetts, USA), which is sequentially precoated with 50 µg/mL poly-L-ornithine (Sigma, cat. no. P-3655) and then with 1 µg/mL human fibronectin (Sigma, cat. no. F-0895) and cultured in 37°C overnight. Cells were transduced with 10 µL of 100× lentivirus encoding miR-199a/b precursors or control precursors using TransDux reagent (System Biosciences, Mountain View, California, USA). Seventy-two hours post-transduction, cells were fixed and probed with anti-TRPV1 (Abcam, Cambridge, Massachusetts, USA). The primary antibody was detected using Alexa Fluor dye conjugated antibodies (Invitrogen, Carlsbad, California, USA) while the nuclei were stained with 4′,6-diamidino-2-phenylindole (Invitrogen, Carlsbad, California, USA). Cells were observed on a Zeiss LSM 510 META laser scanning confocal microscope. Images were analysed with LSM Image Browser software (V.4.2).
Fluorescence in situ RNA hybridisation
A locked nucleic acid (LNA) probe with complementarity to miR-199 was labelled with 5′ and 3′-digoxigenin and synthesised by Exiqon (Woburn, Massachusetts, USA). A scrambled LNA probe was used as a negative control. The slides were prehybridised for 30 min at 52°C, and then 10 pmol of the probe in 60–100 µL of hybridisation mixture was added to each slide and incubated for 1 h at 52°C. Slides were incubated with 3% (vol/vol) H2O2 for 10 min at room temperature (RT) to block endogenous peroxidases before applying horseradish peroxidase-conjugated antibodies. After washing, slides were incubated in blocking buffer for 30 min at RT, then 100–150 µL antibody was added and slides were incubated for 30 min at RT. The TSA Plus Fluorescein System (PerkinElmer, Waltham, Massachusetts, USA) was used for direct fluorescence detection according to the manufacturer's protocol. The slides were imaged using an epifluorescence microscope equipped with charge-coupled device camera and image analysis capabilities.
Northern blots
Samples containing 20–40 μg total RNA (in a volume of 10 μL or less) were mixed with Bartel Lab 8 M Urea Loading Buffer. The membrane was prehybridised in hybridisation buffer at 55°C for 5 h with rotation. A total of 1–20 nM biotin-labelled miRNA probe was added to the hybridisation buffer, which was incubated at 55°C overnight with rotation. The streptavidin-IRDye 680 was diluted at 1:10 000 in blocking buffer; the samples were then incubated for 30 min at RT with gentle shaking and scanned on an LI-COR Odyssey infrared imaging system.
Data analysis
Statistical analyses were done using GeneSpring GX software V.7.3 (Agilent Technologies, Santa Clare, California, USA), Prism V.6 (Graph Pad, San Diego, California, USA) and ASA software V.9.1.3 of the ASA system. One-way analysis of variance was done followed by Tukey's comparison or by the Benjamini and Hochberg correction for false-positive reduction. t Tests were also used. Values are expressed as mean±SD. Tissue samples were paired for comparisons based on matching for age and sex. Spearman's correlation coefficients were used to examine the correlation between visceral pain scores and colonic miR-199a/b expression.
Results
There were 131 patients with IBS-D and 61 controls that were screened for the study. A total of 45 patients with IBS-D (mean age 29.4+3.2 years) and 40 controls (mean age 30.8+2.8 years) were enrolled in the study. All subjects completed the study without any dropouts. All patients with IBS-D had between 3 and 8 loose, watery stools (Bristol Stool Scale 6–7) per day associated with daily lower abdominal pain for at least 5 years. All participants had both a negative hydrogen breath test and tissue transglutaminase antibody. No participant ingested non-steroidal anti-inflammatory drugs or alcohol for at least 3 weeks prior to the study. Colonic biopsies did not reveal any evidence of intestinal inflammation.
Colonic miR-199a/b expression in patients with IBS-D
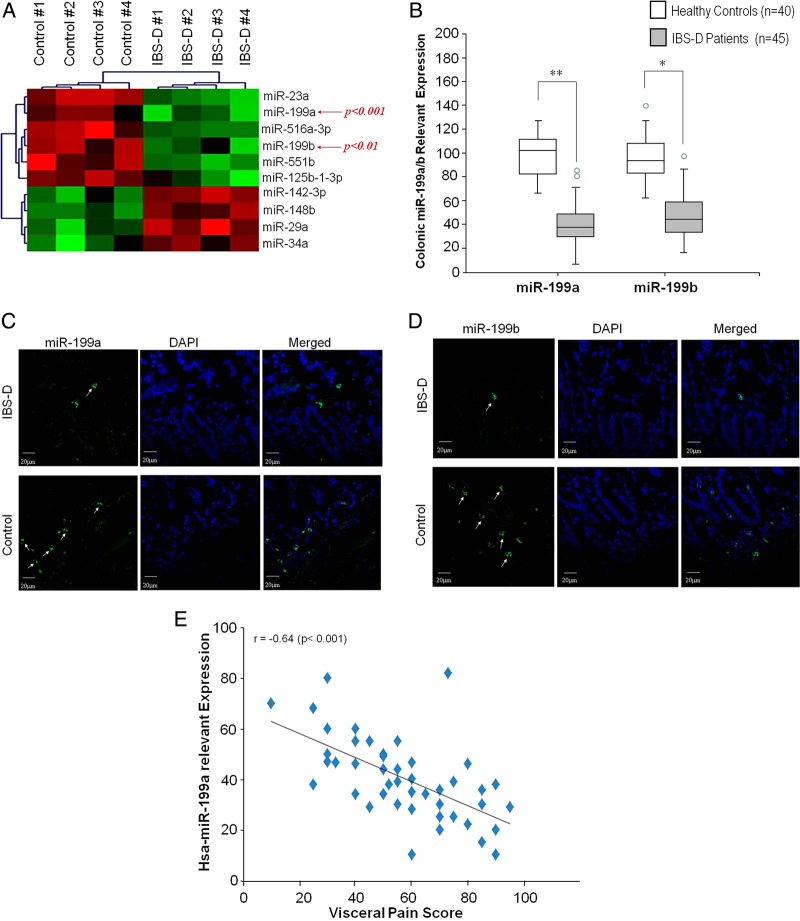
miRNA microarrays were used for miRNA profiling of colonic biopsies from patients with IBS-D and controls. Of the 460 human miRNAs represented in this microarray, aberrant expressions of selected miRNAs were observed in patients with IBS-D. This cluster analysis distinguished expression of specific colonic miRNAs that were either significantly decreased or increased in patients with IBS-D (figure 1A) compared with age-matched and sex-matched controls. Expression of miR-199 family members including miR-199a and miR-199b was significantly downregulated (p<0.001 and <0.01, respectively). Figure 1B illustrates miRNA expression analysis for miR-199a/b that was done using TaqMan microRNA qPCR Assay kits. Quantitative data from 45 patients with IBS-D and 40 controls further confirm a significant decrease in colonic miR-199a/b in patients with IBS-D compared with controls. Additional reproducibility studies were done by miRNA microarray and PCR array using 10 sample comparisons (see online supplementary figure S1A,B). Fluorescence in situ hybridisation (FISH) was done on colon tissue sections using a labelled, specific miR-199a/miR-199b probe. There were substantial decreases in miR-199a expression in the colon of patients with IBS-D (figure 1C, upper panels) compared with controls (figure 1C, lower panels). Colon miR-199b expression was also diminished in patients with IBS-D (figure 1D, upper panels) compared with controls (figure 1D, lower panels). We then correlated miR-199a/b expression in the colon of patients with IBS-D with the visceral pain scores. Figure 1E illustrates Spearman's correlation analysis that revealed an inverse correlation between miR-199a expression and visceral pain scores in patients with IBS-D (r=0.64 with p<0.001). Similarly, miR-199b expression also correlated with visceral pain scores (r=0.50 with p=0.001). These data strongly suggest that diminished miR-199a/b expression may augment visceral pain and GI symptoms in patients with IBS-D.
Figure 1.
Colonic miR-199a/b expression in patients with diarrhoea-predominant IBS (IBS-D). Microarray heat map depicting microRNA (miRNA) profiling of colonic biopsies from four patients with IBS-D and four controls. (A) Green colour illustrates decreased miRNAs expression, and red shows upregulation of miRNAs expression. Expression of miR-199a and miR-199b was significantly downregulated in patients with IBS-D compared with controls (p<0.001 and <0.01, respectively). (B) Box plots for the distribution of miR-199a and miR-199b. The bottom and top edges of the box indicate the intraquartile range, and the line inside the box indicates the mean. The circles outside the boxes are outliers (data points >1.5*IQR). Colonic miR-199a and miR-199b of IBS-D is decreased compared with controls (**p<0.01; *p<0.05). (C) Fluorescence in situ hybridisation (FISH) was used to identify the localisation of human colon tissue sections using a specific miR-199a probe. Substantial decreases in miR-199a expression in the colon of patients with IBS-D (upper panel) compared with controls are present (lower panel). (D) FISH on colon tissue sections using a labelled, specific miR-199b probe. Substantial decreases in miR-199b expression in the colon of patients with IBS-D (upper panel) compared with controls are present (lower panel). (E) Scatterplot of colonic miR-199a expression versus visceral pain scores in patients with IBS-D. The figure shows the regression line illustrating Spearman's correlation coefficients (r). DAPI, 4′,6-diamidino-2-phenylindole.
Identification of TRPV1 as a target gene for miR-199
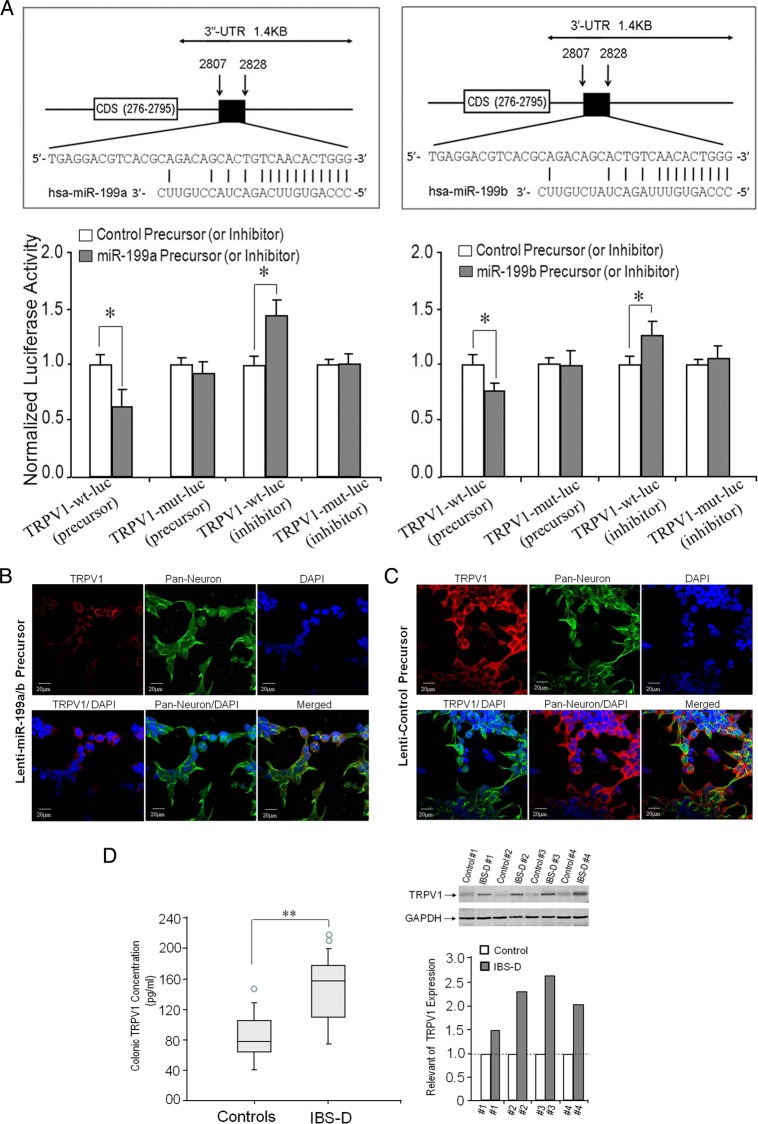
We next addressed the following question: What are the mechanisms underlying the link between visceral pain and colonic miR-199a/b and TRPV1 expression? Based on 3′-UTR sequence analysis and prediction algorithms, translation of several proteins could be potentially targeted by miR-199a/b. Interestingly, the target prediction program, Sanger miRBase database (http://microrna.sanger.ac.uk/), indicated the presence of a highly conserved binding site for miR-199a/b in the 3′-UTR region of TRPV1 mRNA, which codes for CDS 276-2795, a well-characterised capsaicin receptor, and is the vanilloid receptor gene (figure 2A, upper panel). To verify that TRPV1 is a bona fide target of miR-199a/b, luciferase reporter constructs containing the miR-199a and miR-199b recognition sequences from the 3′-UTR of TRPV1 inserted downstream of the luciferase gene were performed. Relative luciferase reporter activity of human 293T cell line, co-transfected TRPV1-wild type (wt) with miR-199a/b precursor was significantly lower than relative luciferase reporter activity of human 293T cell line transfected TRPV1-mut (mutant) with miR-199a/b precursor. In contrast, the cells co-transfected anti-miR-199a/b (inhibitor) with TRPV1-wt displayed increased luciferase activity compared with a TRPV1-mut with an miR-199a/b inhibitor (figure 2A, lower panel).
Figure 2.
Identification of transient receptor potential vanilloid type 1 (TRPV1) as a target gene for miR-199. (A) Upper panel: the location of the putative miR-199a/b target site in the TRPV1 3′-UTR is shown with highly conserved binding site for miR-199a/b in the 3′-UTR region of the TRPV1 mRNA, which codes for CDS 276–2795, a well-characterised capsaicin (vanilloid) receptor (upper panel left: miR-199a; upper panel right: miR-199b). (A) Lower panel: relative luciferase reporter co-transfected either TRPV1-wild type (wt) or TRPV1-mut (mutation) firefly luciferase expression construct, along with either miR-199a/b or control precursors (or inhibitors). Luciferase assays were done after 48 h using a dual luciferase reporter assay system. All the groups are normalised to the average of TRPV1 MUT group co-transfected either with control precursor or with control anti-microRNA (miRNA) (average value=1). A significant decrease (or increase) in relative firefly luciferase activity in the presence of wt miR-199a/b (lower left: miR-199a; lower right: miR-199b) with miR-199a/b precursors (or anti-miR-199a/b inhibitors) indicates the presence of a miR-199a/b modulated target sequence in the 3′-UTR of TRPV1. Bars show mean±SD from eight separate experiments done in triplicate (*p<0.05). (B and C) Confocal immunofluorescent staining of human neuron cells. TRPV1 was reduced after miR-199a/b overexpression (B) compared with control precursor (C). (D) Left panel: ELISA assay of colonic TRPV1 expression in patients with diarrhoea-predominant IBS (IBS-D). Higher levels of TRPV1 expression were found in colon biopsies from patients with IBS-D (n=45) compared with controls (n=40) (**p<0.01). The circles outside the boxes are outliers (data points >1.5*IQR). Right panel: Substantial increases in colonic TRPV1 expression in patients with IBS-D (n=4) compared with controls (n=4) on western blot analysis (right upper panel). We have quantitative bands from the western blots and analysed the data with an internal control of GAPDH (right lower panel). DAPI, 4′,6-diamidino-2-phenylindole; GAPDH, glyceraldehyde 3-phosphate dehydrogenase.
Confocal immunofluorescent staining of human neuron cells revealed that expression of TRPV1 was reduced after lenti-miR-199a/b precursor transfection (overexpression of miR-199a/b) (figure 2B) compared with lenti-control precursor (figure 2C). Reduction in TRPV1 directly correlated with miR-199a/b expression and provides further evidence that (i) miR-199a/b may be a key contributing factor to visceral pain in patients with IBS-D and (ii) TRPV1 is a biologically relevant target of miR-199a/b.
Colonic TRPV1 expression in patients with IBS-D
Significantly higher levels of TRPV1 expression were found by ELISAs in colon biopsies from patients with IBS-D (figure 2D, left panel). We then selected four patients with IBS-D and four age-matched and sex-matched normal controls to further examine colonic TRPV1 expression using western blots. There were substantial increases in TRPV1 expression in patients with IBS-D compared with controls (figure 2D, right panel).
Visceral hypersensitivity and miR-199a expression in an animal model
We used a rat model of VH to further investigate the mechanisms underlying the effects of miR-199 modulation on TRPV1 expression and to see whether miR-199 precursors can reduce VH.20 21 There were significant decreases in miR-199a expression in both the colon (**p<0.001) and DRGs (*p<0.01) in VH rats compared with control rats (figure 3A). To confirm the decreases in miR-199a expression using an independent approach, northern blot analyses were performed. The results (figure 3B) showed that there was significantly diminished expression of miR-199a in the colon (upper panel) and DRGs (lower panel) of VH rats versus control rats.
Figure 3.
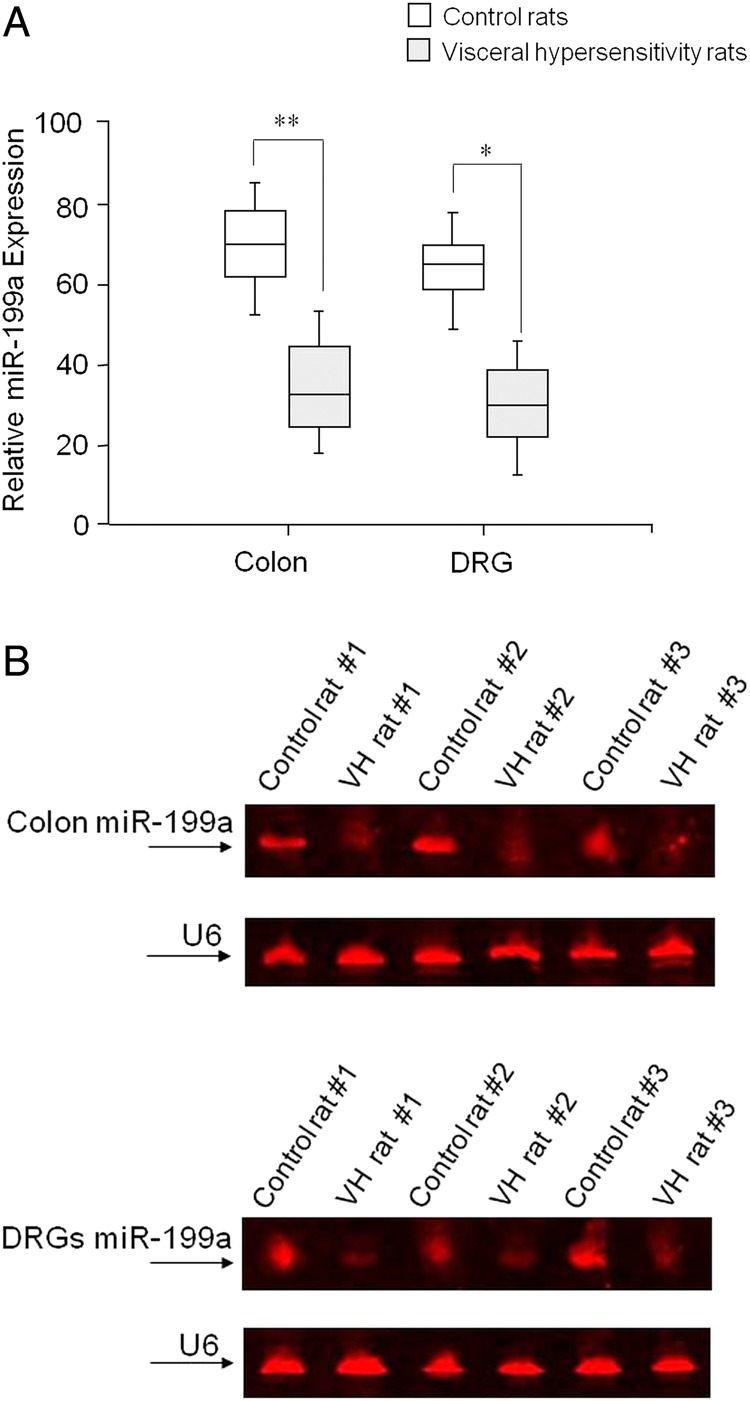
Visceral hypersensitivity (VH) and miR-199a expression in an animal model. (A) Significant decreases in miR-199a expression in both the colon (**p<0.001) and dorsal root ganglions (DRGs) (*p<0.01) from VH rats compared with control rats. (B) Northern blots that show diminished expression of miR-199a in the colon (upper panel) and DRGs (lower panel) of VH rats versus controls.
miR-199 precursors reverse VH via downregulation of TRPV1
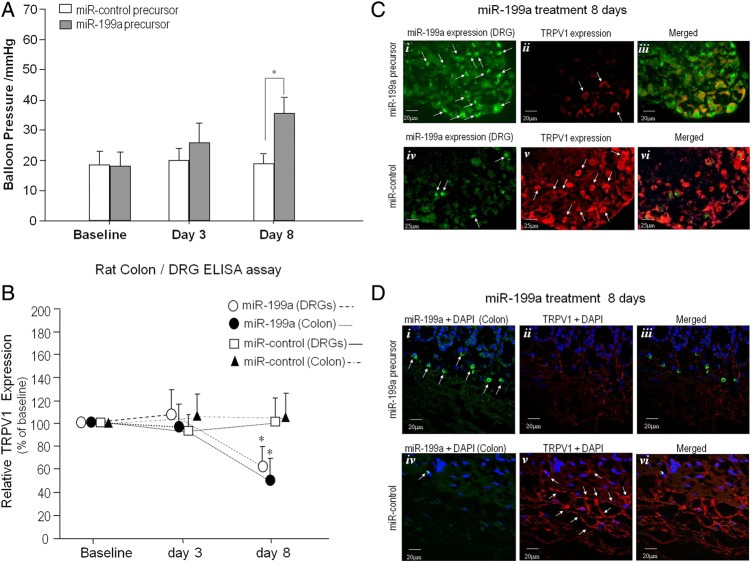
At 3 and 8 days following intraperitoneal injection of miRNA (lenti-miR-199a precursors) into VH rats, there were significant increases in the visceral response threshold to colonic distension, indicating a large reduction in VH at 8 days following miR-199a treatment versus treatment with miR-control precursor (figure 4A). To confirm that (i) TRPV1 is the miR-199 target and (ii) miR-199 regulates VH in VH rats via TRPV1 signalling, ELISA assays were performed. Figure 4B shows diminished TRPV1 expression in rat colon and DRGs 8 days following treatment with lenti-miR-199a precursor (n=5) compared with lenti-miR-controls (n=4) (*p<0.05). FISH was done with a labelled miR-199a specific probe to identify key target genes that modulate VH. Figure 4C shows FISH for miR-199a and its co-localisation with TRPV1 expression in the DRG of VH rats that received lenti-miR-199a precursor (figure 4C.i) compared with VH rats that received lenti-miR-control injection (figure 4C.iv). There was a significant decrease in DRG TRPV1 expression in lenti-miR-199a precursor-treated rats (figure 4C.ii) compared with lenti-miR-controls (figure 4C.v). We found similar results in rat colon tissue. Figure 4D shows enhanced colon miR-199a expression in VH rats (figure 4D.i) and diminished TRPV1 expression (figure 4D.ii) 8 days following lenti-miR-199a precursor treatment. Accordingly, there was increased TRPV1 expression (figure 4D.v) with diminished miR-199a (figure 4D.iv) in VH rat colon 8 days following lenti-miR-control injection.
Figure 4.
miR-199 precursors reverse visceral hypersensitivity (VH) via downregulation of transient receptor potential vanilloid type 1 (TRPV1). (A) Following intraperitoneal injection of microRNA (miRNA) (lenti-miR-199a precursors) into VH rats, there were significant increases in the visceral response threshold to colonic distension indicating a large reduction in VH (*p<0.01) at 8 days, but not at 3 days. (B) ELISA assay demonstrates diminished TRPV1 expression in rat colon and dorsal root ganglion (DRG) at 8 days following treatment with lenti-miR-199a precursor compared with lenti-miR-control (*p<0.05). (C and D) Fluorescence in situ hybridisation (FISH) with a labelled, specific miR-199a probe to identify key target genes that modulate VH. Increased miR-199a expression in DRG of VH rats that received lenti-miR-199a precursor treatment at 8 days (C.i) versus lenti-miR-control injections (C.iv). Decreased DRG TRPV1 expression is shown (C.ii) following lenti-miR-199a precursor treatment of VH rats compared with lenti-miR-controls (C.v). VH rats that received lenti-miR-199a precursor treatment exhibited increased miR-199a colonic expression at 8 days (D.i) versus lenti-miR-control injection (D.iv). Following lenti-miR-199a precursor treatment of VH rats, there was decreased TRPV1 expression (D.ii) compared with lenti-miR-controls (D.v). DAPI, 4′,6-diamidino-2-phenylindole.
Discussion
This translational study reveals a major mechanism for visceral pain in patients with IBS-D. Our results provide the first evidence in patients with IBS-D that decreased colonic miR-199a/b expression contributes to chronic visceral pain and nociception via upregulation of TRPV1 expression. Although a potential role for increased expression of TRPV1 in functional GI disorders has been previously described, the mechanisms by which TRPV1 expression is elevated and the identity of specific miRNAs such as miR-199 that may modulate intestinal TRPV1 signalling and visceral nociception have not been previously elucidated.13–14 22
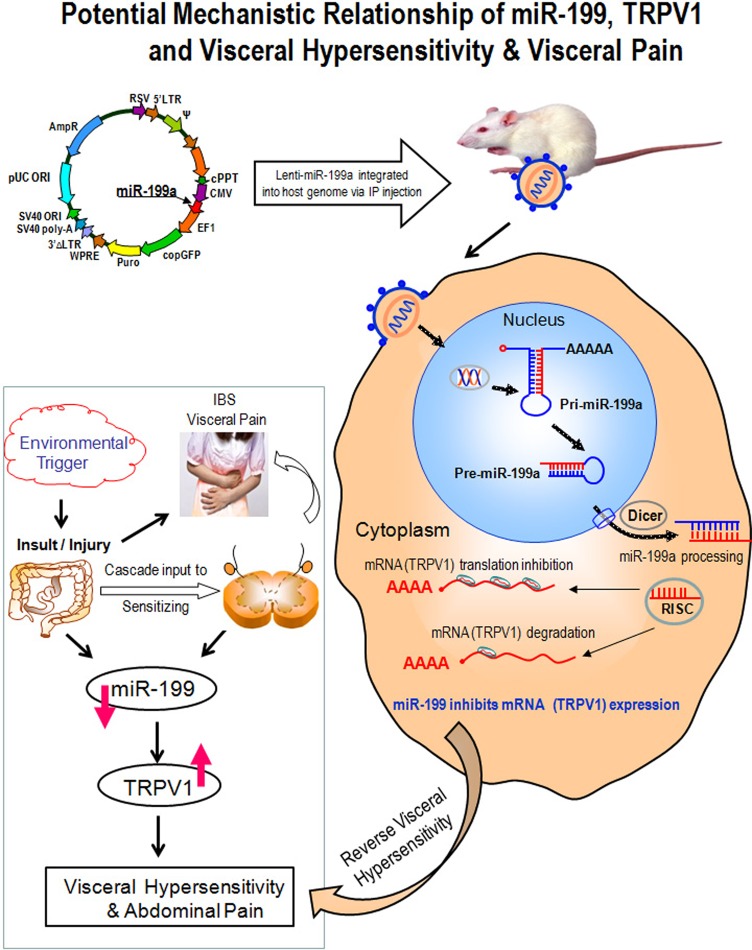
Our study using an animal model of VH provides the first in vivo evidence that miR-199 may modulate VH and TRPV1-associated peripheral nerve changes (figure 5). Our in vitro studies show that TRPV1 is an miR-199 target in both humans and rats. Furthermore, the results suggest a new potential strategy for treating visceral pain in patients with IBS-D, a concept that may also apply to other conditions with visceral pain. The results also show that in vivo introduction of an miR-199 precursor leads to decreased TRPV1 expression in rat colon and DRGs, which may be responsible for reversing VH. Overall we have presented unrecognised pathophysiological mechanisms underlying visceral pain in patients with IBS, which may have the potential for future development of miRNA-based therapies for IBS.
Figure 5.
Potential mechanistic relationship of miR-199, transient receptor potential vanilloid type 1 (TRPV1) and visceral hypersensitivity and visceral pain. Lentiviruses deliver microRNAs (miRNAs) in vivo and offer several potentially unique advantages over conventional retroviral gene delivery systems. Principal among these is the ability to provide a long-term and stable gene/miRNA expression and to infect non-dividing cells, such as dorsal root ganglion neuron cells. This study used lentiviruses to deliver and overexpress miR-199a to silence the TRPV1 expression. The related molecular mechanisms are illustrated. Left lower panel: environmental triggers lead to gut injury, which sets up a cascade of events that leads to a state of visceral hypersensitivity. This hypersensitivity state may be characterised by decreased miR-199 and increased TRPV1 expression. Right: after a host cell is infected by a RNA virus, the viral genome integrated into host genome in the nucleus to yield pri-miR-199a. The pri-miR-199a is processed by host factors in the nucleus to yield the pre-miRNA intermediate, which is then exported to the cytoplasm through exportin, where the mature miRNA (miR-199a) is generated via Dicer involvement and incorporated into the RISCs that are programmed by miRNA, and then inhibit expression of the host mRNAs (TRPV1) in the infected cell's cytoplasm. Decreased TRPV1 expression reverses visceral hypersensitivity. RISC, RNA-induced silencing complex.
During the last several years, it has been increasingly appreciated that alterations in the expression of genes coding for miRNAs contribute to the pathogenesis of human diseases.23 Moreover, non-coding DNA sequences account for >95% of the human genome, and their unique role in sustaining human health and disease is becoming better recognised.24 Large non-coding RNAs (ncRNAs) (>200 bp) (eg, ultraconserved regions) contain conserved sequences that are associated with distinct gene expression signatures in human disease states.23 Small ncRNAs (<200 bp) (eg, miRNAs) have also been identified and are being recognised as important in regulating gene expression in important cellular processes (differentiation, proliferation, apoptosis, development) through imperfect pairing with target mRNAs of protein-coding genes.25–28 The large number of miRNA platforms and analytical methods that have been developed in recent years has led to a challenge in cross-platform miRNA microarray comparison and reproducibility. In this study, following miRNA array analysis, we performed additional reproducibility studies by microarray and PCR array to confirm using 10 sample comparisons (see online supplementary figure S1).
It is becoming increasingly apparent that underlying pathophysiology of certain GI tract diseases and disorders may be related to aberrantly expressed miRNAs.15–18 One recent study found increased expression of miR-510 in patients with IBS-D in which miR-510 targets HTR3E, a serotonin receptor gene.29 Another study evaluated patients with both active or inactive UC and demonstrated differential expression of 11 miRNAs in active UC compared with inactive disease.16 Recently, we began to elucidate such miRNA-related mechanisms in the gut and found upregulated miR-29a profiles in patients with IBS-D along with decreased glutamine synthetase and increased intestinal permeability.17 Upregulated GI miR-29a expression increased intestinal permeability in patients with IBS directly through glutamine-dependent mechanisms and confirmed the previously reported functional interaction between miR-29a and the gene, GLUL, that codes for glutamine synthetase.30
TRPV1 is a non-selective ligand-gated cation channel that is expressed on peripheral primary afferent sensory neurons and, when activated (by heat temperature, capsaicin, acidic pH, resinferatoxin and/or inflammatory mediators), produces a burning sensation and/or pain.31 Recent evidence suggests a role for TRPV1 in functional GI disorders, and this idea is supported by animal models in which TRPV1 is elevated following stress and/or inflammatory events. TRPV1 has been shown to modulate visceral sensitivity and mechanosensation in animal models following stress or colonic inflammation (adult or neonatal).8–12 21–22 However, the mechanisms by which TRPV1 signalling is increased in GI tissue in animal models and patients with GI symptoms are unknown.13 14
This study revealed increased TRPV1 expression in colon tissue of patients with IBS-D, which was associated with visceral pain scores. These results support earlier findings by Akbar et al,13 who reported increased neuronal expression of TRPV1 in colonic biopsies from patients with IBS that correlated with abdominal pain severity. These findings suggest a potential role for TRPV1 in the pathogenesis of visceral nociception following triggering events such as inflammation with resultant activation of immune or mast cells that may lead to upregulation of TRPV1.32 The current work by our group extends these earlier findings and is the first to suggest a mechanistic and functional role for decreased colonic miR-199 expression in the potential regulation of chronic visceral pain and GI symptoms (via TRPV1 mechanisms) in both patients with IBS-D and in an animal model of VH. Thus, decreased miR-199 expression may be a key pathophysiological factor leading to increased TRPV1 levels and heightened GI nociception in humans.
We used an animal model of VH to elucidate the functional relationship between miR-199 and TRPV1 signalling in the gut and nervous system.20 Our results show that VH rats had a significant decrease in miR-199 levels and an increase in TRPV1 in the colon and DRGs that correlated with VH. These results support earlier studies in animal models following stress or colonic inflammation in which quantitative expression of TRPV1 was shown to modulate visceral sensitivity and mechanosensation.8–12 21–22 We then determined whether miR-199 could be an important driving mechanism of TRPV1 expression. Following an injection (intraperitoneal) of an miR-199 precursor, there was a decrease in VH in rats. The decrease in VH correlated with upregulation of miR-199 and decreased TRPV1 in the colon and DRGs 8 days after the miR-199 precursor was given. The response time of 8 days may be caused by the intraperitoneal delivery route of lentiviral particles that may take longer due to loss of targeting efficiency given the complexities of the abdominal cavity and dynamics of intraperitoneal fluid movement. In fact, delivery of lentiviral particles in vivo requires a very high-titre virus (>1×109 TUs/mL or greater) as adsorption and fusion in a dynamic environment is much more difficult than a ‘static’ environment such as cell culture, where fusion of virus particles into cells can occur unabated. These novel in vivo findings are the first in which an miRNA precursor was used to modulate a downstream target that may drive visceral nociception.
In conclusion, this study suggests a role for TRPV1 in the generation and modulation of chronic visceral nociception in both patients with IBS and in an animal model of VH. Our results also extend previous knowledge related to increased colonic TRPV1 in that decreased miR-199 may be a potential factor that drives increased TRPV1 expression and results in increased abdominal pain and other GI symptoms in patients with IBS. We also discovered a potential role for miR-199 precursors, which may be ultimately used in vivo to dampen TRPV1 expression and reduce symptoms in patients with IBS. Future studies are now needed in patients with IBS that use miRNA precursors to reverse key nociceptive signalling processes that may be the underlying mechanisms that lead to visceral pain.17–18 33
Supplementary Material
Footnotes
Contributors: Study concept and design: QZ and GNV. Acquisition of data: LY, SL, SB, SM and QZ. Analysis and interpretation of data: QZ, CC and GNV. Drafting of the manuscript: QZ and GNV. Critical revision of the manuscript for important intellectual content: CC and GNV. Statistical analysis: QZ, LY and AT. Obtained funding: GNV and QZ.
Funding: This work was supported by grants from the National Institutes of Diabetes and Digestive and Kidneys Diseases (DK099052), National Center for Complementary and Alternative Medicine (AT005291) and from the Department of Veteran Affairs.
Competing interests: None.
Ethics approval: All human and animal studies were approved by the University Institutional Review Board and Institutional Animal Care and Use Committee, respectively.
Provenance and peer review: Not commissioned; externally peer reviewed.
References
- 1.Longstreth GF, Thompson WG, Chey WD, et al. Functional bowel disorders. Gastroenterology 2006;130:1480–91. 10.1053/j.gastro.2005.11.061 [DOI] [PubMed] [Google Scholar]
- 2.Mayer EA, Bradesi S, Chang L, et al. Functional GI disorders: from animal models to drug development. Gut 2008;57:384–04. 10.1136/gut.2006.101675 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Zhou Q, Verne GN. New insights into visceral hypersensitivity-clinical implications in IBS. Nat Rev Gastroenterol Hepatol 2011;8:349–55. 10.1038/nrgastro.2011.83 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Chaloner A, Rao A, Al-Chaer ED, et al. Importance of neural mechanisms in colonic mucosal and muscular dysfunction in adult rats following neonatal colonic irritation. Int J Dev Neurosci 2010;28:99–103. 10.1016/j.ijdevneu.2009.09.001 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Mayer EA. Gut feelings: the emerging biology of gut-brain communication. Nat Rev Neurosci 2011;12:453–66. 10.1038/nrn3071 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Ohman L, Simren M. Pathogenesis of IBS: role of inflammation, immunity, and neuroimmune interactions. Nat Rev Gastroenterol Hepatol 2010;7:163–73. 10.1038/nrgastro.2010.4 [DOI] [PubMed] [Google Scholar]
- 7.Dhaka A, Uzzell V, Dubin AE, et al. TRPV1 is activated by both acidic and basic pH. J Neuroscience 2009;29:153–8. 10.1523/JNEUROSCI.4901-08.2009 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Van Den Wijngaard RM, Klooker TK, Welting O, et al. Essential role for TRPV1 in stress-induced (mast cell-dependent) colonic hypersensitivity in maternally separated rats. Neurogastroenterol Motil 2009;21:1107–e94. 10.1111/j.1365-2982.2009.01339.x [DOI] [PubMed] [Google Scholar]
- 9.Winston J, Shenoy M, Medley D, et al. The vanilloid receptor initiates and maintains colonic hypersensitivity induced by neonatal colon irritation in rats. Gastroenterology 2007;132:615–27. 10.1053/j.gastro.2006.11.014 [DOI] [PubMed] [Google Scholar]
- 10.Hong S, Fan J, Kemmerer ES, et al. Reciprocal changes in villoid (TRPV1) and endocannabinoid (CB1) receptors contribute to visceral hyperalgesia in the water avoidance stressed rat. Gut 2009;58:202–10. 10.1136/gut.2008.157594 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Jones RCW III, Xu L, Gebhart GF. The mechanosensitivity of mouse colon afferent fibers and their sensitization by inflammatory mediators require transient receptor potential vanilloid 1 and acid-sensing ion channel 3. J Neurosci 2005;25:10981–9. 10.1523/JNEUROSCI.0703-05.2005 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Phillis BD, Martin CM, Kang D, et al. Role of TRPV1 in high-threshold rat colonic splanchnic afferents is revealed by inflammation. Neuroscience Letters 2009;459:57–61. 10.1016/j.neulet.2009.04.051 [DOI] [PubMed] [Google Scholar]
- 13.Akbar A, Yiangou Y, Facer P, et al. Increased capsaicin receptor TRPV1-expressing sensory fibres in irritable bowel syndrome and their correlation with abdominal pain. Gut 2008;57:923–9. 10.1136/gut.2007.138982 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Holzer P. TRPV1: a new target for treatment of visceral pain in IBS? Gut 2008;57:882–4. 10.1136/gut.2008.149724 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Bakirtzi K, Hatziapostolou M, Karagiannides, et al. Neurotensin signaling activates microRNAs-21 and -155 and Akt, promotes tumor growth in mice, and is increased in human colon tumors. Gastroenterology 2011;141:1749–61. 10.1053/j.gastro.2011.07.038 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Wu F, Zikusoka M, Trindade A, et al. MicroRNAs are differentially expressed in ulcerative colitis and alter expression of macrophage inflammatory peptide-2 alpha. Gastroenterology 2008;135:1624–35. 10.1053/j.gastro.2008.07.068 [DOI] [PubMed] [Google Scholar]
- 17.Zhou Q, Souba WW, Croce C, et al. MicroRNA-29a regulates intestinal membrane permeability in patients with irritable bowel syndrome. Gut 2010;59:775–84. 10.1136/gut.2009.181834 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Zhou Q, Verne GN. miRNA-based therapies for the Irritable Bowel Syndrome. Expert Opin Biol Ther 2011;11:991–5. 10.1517/14712598.2011.577060 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Pimentel M, Chow EJ, Lin HC. Eradication of small intestinal bacterial overgrowth reduces symptoms of irritable bowel syndrome. Am J Gastroenterol 2000;95:3503–6. 10.1111/j.1572-0241.2000.03368.x [DOI] [PubMed] [Google Scholar]
- 20.Zhou Q, Caudle RM, Price DD, et al. Visceral and somatic hypersensitivity in a subset of rats following TNBS-Induced colitis. Pain 2008;134:9–15. 10.1016/j.pain.2007.03.029 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Malykhina AP, Qin C, Lei Q, et al. Differential effects of intravesical resiniferatoxin on excitability of bladder spinal neuorns upon colon-bladder cross-sensitization. Brain Rsearch 2013;1491:213–24. 10.1016/j.brainres.2012.11.003 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Van Den Wijingaard RM, Welting O, Bulmer DC, et al. Possible role for TRPV1 in neomycin-induced inhibition of visceral hypersensitivity in rat. Neurogastroenterol Motil 2009;21:863–e60. 10.1111/j.1365-2982.2009.01287.x [DOI] [PubMed] [Google Scholar]
- 23.Bejerano G, Pheasant M, Makunin I, et al. Ultraconserved elements in the human genome. Science 2004;304:1321–5. 10.1126/science.1098119 [DOI] [PubMed] [Google Scholar]
- 24.Guttman M, Amit I, Garber M, et al. Chromatin signature reveals over a thousand highly conserved large non-coding RNAs in mammals. Nature 2009;458:223–7. 10.1038/nature07672 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Carninci P, Kasukawa T, Katayama S, et al. The transcriptional landscape of the mammalian genome. Science 2005;309:1559–63. 10.1126/science.1112014 [DOI] [PubMed] [Google Scholar]
- 26.Moazad D. Small RNAs in transcriptional gene silencing and genome defense. Nature 2009;457:413–20. 10.1038/nature07756 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Farh KK, Grimson A, Jan C, et al. The widespread impact of mammalian microRNA on mRNA repression and evolution. Science 2005;310:1817–21. 10.1126/science.1121158 [DOI] [PubMed] [Google Scholar]
- 28.Kim J, Krichevsky A, Grad Y, et al. Identification of many microRNAs that copurify with polyribomes in mammalian neurons. PNAS 2004;101:360–5. 10.1073/pnas.2333854100 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Kapeller J, Houghton LA, Monnikes H, et al. First evidence for an association of a functional variant in the microRNA-510 target site of the serotonin receptor-type 3E gene with diarrea predominant irritable bowel syndrome. Hum Mol Genet 2008;17:2967–77. 10.1093/hmg/ddn195 [DOI] [PubMed] [Google Scholar]
- 30.Haberle J, Gorg B, Rutsch F, et al. Congenital glutamine deficiency with glutamine synthetase mutations. NEJM 2005;353:1926–33. 10.1056/NEJMoa050456 [DOI] [PubMed] [Google Scholar]
- 31.Caterina MJ, Schumacher MA, Tominaga M, et al. The capsaicin receptor: a heat-activated ion channel in the pain pathway. Nature 1997;389: 816–24. 10.1038/39807 [DOI] [PubMed] [Google Scholar]
- 32.Spiller RC, Jenkins D, Thornley JP, et al. Increased rectal mucosal enteroendocrine cells, T lymphocytes, and increased gut permeability following acute Campylobacter enteritis and in post-dysenteric irritable bowel syndrome. Gut 2000;47:804–11. 10.1136/gut.47.6.804 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Zhou Q, Verne GN. Role of microRNA in chronic visceral nociception. Pain 2013;154:9–10. 10.1016/j.pain.2012.10.015 [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.