Abstract
Objective
Systemic lupus erythematosus (SLE) is associated with increased risk of adverse pregnancy outcomes, including pre-eclampsia, particularly in association with antiphospholipid antibody syndrome (APS). While significant placental abnormalities are expected in pre-eclampsia, less is known about how lupus activity and APS in pregnancy affect the placenta. We describe placental pathology from a population of lupus pregnancies, several of which were complicated by APS-related thromboses, in which pre-eclampsia and other complications developed. We performed standard histopathological placental review and quantified neutrophils and neutrophil extracellular traps (NETs) in the intervillous space, given the recognised association of NETs with lupus, APS and pre-eclampsia.
Methods
Pre-eclampsia, SLE and control placentas were scored for histological features, and neutrophils were quantified on H&E and immunohistochemical staining for the granular protein myeloperoxidase. NETs were identified by extracellular myeloperoxidase staining in the setting of decondensed nuclei. Non-parametric analysis was used to evaluate differences in netting and intact neutrophils between groups, with Kruskal–Wallis testing for associations between histological findings and neutrophils.
Results
Placentas were evaluated from 35 pregnancies: 10 controls, 11 pre-eclampsia, 4 SLE+pre-eclampsia and 10 SLE, including one complicated by catastrophic APS and one complicated by hepatic and splenic vein thromboses during pregnancy. Intrauterine growth restriction and oligohydramnios were observed in lupus cases but not controls. Significantly more NETs were found infiltrating placental intervillous spaces in pre-eclampsia, SLE+pre-eclampsia and all 10 SLE non-pre-eclampsia cases. The ratio of NETs to total neutrophils was significantly increased in all case groups compared with controls. When present, NETs were associated with maternal vasculitis, laminar decidual necrosis, maternal–fetal interface haemorrhage and non-occlusive fetal thrombotic vasculopathy.
Conclusions
In this pilot study of placental tissue from lupus pregnancies, outcomes were more complicated, particularly if associated with APS. Placental tissue revealed marked inflammatory and vascular changes that were essentially indistinguishable from placental tissue of pre-eclampsia pregnancies.
Keywords: Systemic Lupus Erythematosus, Autoimmune Diseases, pregnancy
Introduction
Pregnancy management in systemic lupus erythematosus (SLE) has improved over years, with more emphasis on multidisciplinary management and continuation of antimalarial therapy in pregnancy leading to increasingly better outcomes.1 While recent evidence suggests that lupus pregnancies, particular those with low disease activity, have better outcomes than previously thought, SLE pregnancies remain at significantly increased risk for complications, including pre-eclampsia, intrauterine growth restriction (IUGR), prematurity and maternal and fetal mortality, particularly if disease activity is high during pregnancy or if antiphospholipid antibodies are present.2–4 Because it can present similarly to proliferative lupus nephritis, pre-eclampsia, a hypertensive disease of pregnancy, can be one of the most difficult complications to diagnose and treat in pregnant patients with lupus. Pre-eclampsia is characterised by new hypertension and proteinuria after the 20th week of gestation, occurring in about 2–8% of pregnancies in general,4 and a full 22.5% of lupus pregnancies. Both lupus and pre-eclampsia are characterised by similar histopathological placental pathology indicative of underlying derangements in implantation, vascular remodelling, and immune regulation.4 5 These similarities raise the possibility that a common pathogenesis may exist along a spectrum in both lupus and pre-eclampsia pregnancies, sharing joint mechanisms and outcomes.
A recently proposed mechanism for the increased endothelial dysfunction, thrombosis and perpetuation of the inflammatory milieu observed in SLE involves the finding of increased neutrophil extracellular traps (NETs) in the circulation, skin and renal tissues of patients with lupus.6 7 Neutrophils form NETs by releasing a web of nuclear contents including chromatin, DNA and antimicrobial proteins such as neutrophil elastase, myeloperoxidase (MPO), cathepsin G and proteinase 3 into the extracellular space in response to microbial triggers including bacteria, viruses and fungi.8 As such, NET release (or NETosis) represents an important arm of antimicrobial innate immunity, and one distinctly different from the process of phagocytosis, which typically leads to neutrophil apoptosis.9 10 In addition to the response to microbes, NET release is triggered by various stimuli, including inflammatory cytokines,11 12 immune complexes13 and placental syncytiotrophoblast microparticles found in the maternal circulation in pre-eclampsia.12 Upon release, NETs mediate toxic effects on other bystander cells, including vascular endothelial cells,14 and exert a powerful prothrombotic effect.15 16
Interestingly, a propensity of neutrophils to form NETs has been observed in patients with SLE, in whom both an impaired ability to degrade NETs and a predilection of neutrophils to undergo NETosis has been observed.14 17–19 NETs may therefore have a prominent role in inducing and perpetuating the vasculopathy observed in SLE, as well as other autoimmune diseases such as antineutrophil cytoplasmic antibody-associated vasculitis, antiphospholipid antibody syndrome (APS) and possibly pre-eclampsia, where previous research has identified NETs infiltrating the placental intervillous space in pregnancies complicated by pre-eclampsia.16 20 21
To test the hypothesis that the presence of neutrophils and NETs in the placental intervillous space are common features of pregnancies in both lupus and pre-eclampsia, and that their presence corresponds to ongoing inflammatory and vasculopathic changes throughout the placenta, we examined histological features of placental tissue from women who had lupus or pre-eclampsia (or both) complicating their pregnancies, and compared them with placental tissue from normal pregnancies. In addition to scoring the extent of tissue inflammation, maternal–fetal interface haemorrhage and infarction in these samples, we quantified intact and netting neutrophils detected by immunohistochemical staining, focusing on the placental intervillous space, the site of oxygen and nutrient exchange. We hypothesised that both lupus pregnancies and pre-eclampsia would be associated with inflammatory histological features, as well as the infiltration of neutrophils/NETs, compared with normal pregnancies. We expected to see an overall more benign histology and fewer total neutrophils and NETs in the lupus pregnancies than in the pre-eclampsia group, in whom abnormal placental pathology would be expected.
Materials and methods
Approval for this research was obtained through the University of Michigan Institutional Review Board.
Case ascertainment
SLE and pre-eclampsia cases were identified using corresponding ICD-9 codes at admission and discharge (see online supplementary appendix 1 for list of codes). A cross-reference of SLE and pre-eclampsia cases was then performed to capture lupus pregnancies complicated by pre-eclampsia. From each group, social history was reviewed, and cases with positive tobacco use during pregnancy were excluded. Cases with pre-existing or gestational hypertension and diabetes were excluded from the study. Multifetal gestations and fetuses with structural/chromosomal anomalies were excluded.
lupus-2015-000134supp.pdf (87.5KB, pdf)
Verification of SLE cases
Charts review was performed for subjects identified by ICD-9 search in order to confirm SLE diagnosis by ≥4/11 American College of Rheumatology classification criteria.22 23 Data were obtained from inpatient and outpatient records, including scanned documents within subject files from outside rheumatologists where, in some cases, the lupus diagnosis was made prior to receiving care at the University of Michigan. Other data reviewed included laboratory results with particular attention to autoantibody profile, haematological and renal testing, as well as pathology review of renal biopsies. Assessment of SLE disease activity during pregnancy was based on the treating rheumatologists' assessment and on whether an increase in the patient's daily prednisone dose of ≥10 mg/day was made at any time during pregnancy. All but one patient with SLE were evaluated and treated by University of Michigan rheumatologists during their pregnancies. The one patient treated outside the University of Michigan system had inactive lupus during pregnancy based on obstetric notes, concurrent medications and laboratory measures.
Forty-three per cent of patients with SLE in the study had antiphospholipid antibodies, categorised as follows: IgG and IgM isotypes of anticardiolipin (aCL) and β-2-glycoprotein 1 (β-2GP1) were considered positive if greater than the 99th percentile at our institution, on two or more occasions, at least 12 weeks apart, 6 months prior to or during the study case pregnancy. This corresponded to the following values: aCL IgG (≥22 GPL); aCL IgM (≥25 MPL); β2GP1 IgG (≥24 U/mL); β2GP1 IgM (≥26 U/mL). The lupus anticoagulant was identified either by prolongation of activated partial thromboplastin time or dilute Russell's viper-venom time with a positive confirmatory test. 28.5% of patients with SLE fulfilled diagnostic criteria for APS according to expert consensus criteria,24 25 which is independently associated with pregnancy complications.26 For purposes of analysis, patients with secondary APS were included within the ‘SLE’ group.
Verification of pre-eclampsia cases
Chart review was performed for subjects identified by ICD-9 search in order to confirm pre-eclampsia diagnosis occurring in both the antepartum and postpartum periods. Pre-eclampsia is defined by the International Society for the Study of Hypertension in pregnancy as hypertension of at least 140/90 mm Hg on two separate occasions ≥4 h apart, in addition to proteinuria of at least 0.3 g/24 h collection (or >30 mg/mmol protein/creatinine ratio), occurring after the 20th week of gestation in previously normotensive women, with resolution by the 6th week of the postpartum period.4 27 This diagnosis was met in 9/11 of the subjects; the diagnosis in the remainder of cases was based on the clinical judgement of the treating obstetrician in the context of hypertensive emergency and proteinuria as they were delivered emergently.
Control cases
A prospective control group of sequentially enrolled healthy women undergoing planned caesarean-sections between 38 and 39 weeks gestation was identified, and placental tissue obtained at the time of delivery. One patient delivered spontaneously at the time of her planned section.
Tissue procurement and processing
Formalin-fixed, paraffin-embedded tissue was obtained from the University of Michigan Pathology Slide/Block Library Repository. Placental tissue from women who deliver at the University of Michigan Hospital is saved and fixed in 10% formalin solution when a pathologist's review is requested by the obstetrician or perinatologist. In the majority of SLE pregnancies (approximately ≥90%) at our hospital, a formal placental pathology review is performed due to recognition of higher risk and adverse outcomes.
Review of pathology
All slides were reviewed with an experienced placental pathologist (RWL), who was blinded to the clinical outcome.
Histological review
All specimens were first characterised by H&E staining. We used standard diagnostic criteria and performed a systematic pathologic review adapted from work by Salafia and Popek, developed with the intention of identifying underlying pathophysiological processes with the most relevant clinicopathological correlations.28 29 These factors included but were not limited to acute and chronic inflammation, vasculopathy, maternal–fetal interface haemorrhage and ischaemic changes detailed below and described in online supplementary table (appendix 2). The separate histological categories were scored as follows: ‘absent=0; mild=1; moderate=2; severe=3’ for categorical variables, or ‘present/not present’ for binary variables. The relationship between these variables and the presence of neutrophils and NETs in the intervillous space was then assessed.
Acute and chronic inflammation
An acute maternal inflammatory response was considered mild if neutrophils were present, including amnionitis, focal deciduitis or intervillositis; moderate if more global inflammatory changes and obvious neutrophils were apparent on low power; or severe if microabscess formation and coalescing neutrophils were seen. An acute fetal inflammatory response was mild if focal vasculitis was seen; moderate with acute fetal inflammation (funisitis, which is defined by the finding of polymorphic mononuclear cells in the umbilical cord vasculature and stroma), of at least one vessel; or severe if funisitis was present. Chronic decidual inflammation was considered mild if a non-occlusive perivascular infiltrate was noted with intramuscular histiocytes and vascular degeneration; moderate with multifocal, partially occlusive thrombosis; or severe with thrombosed vessels and palisading inflammatory in the vessel wall, with or without fibrinoid necrosis.
Vasculopathy and infarction
Histological changes of vasculopathy of the maternal and fetal circulations were qualitatively assessed. Fetal thrombotic vasculopathy was assigned to distal vessel and large vessel involvement and scored based upon degree of involvement. Villous hypervascularity as documented by the presence or absence of chorangiosis, which is defined by at least 10 villous capillaries in 10 villi on a 10× objective, seen in multiple locations, was also determined. Maternal decidual vasculopathy was scored as mild to moderate depending on the presence and extent of atherosis and/or perivascular alteration of spiral arterioles, and severe if multiple vascular lumens were replaced by organised thrombosis and fibrinoid necrosis of vessel walls.
Maternal–fetal interface haemorrhage
The presence of acute and chronic maternal–fetal interface haemorrhage was also assessed as it implies an implantation site defect, which has been traditionally associated with pre-eclamptic pregnancies. Due to the small sample size, acute and chronic haemorrhage were considered as one group.
Neutrophil quantification
Using an ocular micrometer, the diameter of a 60× high-power field was measured as 0.36 mm (360 µm). Neutrophils were identified by MPO immunohistochemistry (as described below), as well as by nuclear morphology. Placental sections were evaluated at 60×, with neutrophil counts obtained in 10 consecutive fields in which the maternal intervillous space was identified. A neutrophil was defined as ‘intact if MPO staining was confined within the cytoplasm of the cell, and if an intact, multilobulated nucleus was present. A neutrophil was defined as ‘netting’ when extracellular MPO was detected in the presence of a disrupted, decondensed nucleus. Finally, neutrophils identified as ‘indeterminate’ when extracellular MPO was present, but with unclear nuclear-cytoplasmic integrity. These cells were counted as part of the total number of neutrophils in the intervillous space but were not considered to be forming NETs.
Immunohistochemistry
Deparaffinisation and rehydration were performed with standard xylene-to-ethanol washes. Heat-induced epitope retrieval was achieved by boiling samples for 30 min in sodium citrate buffer (10 mM sodium citrate, 0.05% Tween 20, pH 6.0). Samples were blocked in phosphate buffered saline plus 0.025% Tween 20 with 10% foetal bovine serum. The primary antibody was to MPO (Dako, Carpinteria, California, USA), while the secondary antibody was HRP-conjugated anti-rabbit IgG (Amersham Biosciences, Pittsburgh, Pennsylvania, USA). In between the primary and secondary antibodies, tissue was incubated for 15 min in 0.3% hydrogen peroxide to block endogenous peroxidase activity. Colour change was detected with the DAB-Plus Substrate Kit (Invitrogen, Grand Island, New York, USA). Samples were counterstained with haematoxylin. For a subset of the samples, the identity of netting neutrophils was confirmed (as MPO-DNA overlap) by immunofluorescence staining with anti-MPO (Dako) and DAPI as previously described.30
Statistical analysis
Summary statistics were computed to describe the distributions of variables of interest, and boxplots stratified by patient group were used for graphical display. Logistic regression was used to model the associations between number of netting neutrophils and binary histopathological outcome measures. Due to the sample size and data restriction, the non-parametric Kruskal–Wallis test was used to evaluate whether the ratio of netting neutrophils to intact neutrophils differed between groups.
Results
Demographics
Placentas from 10 healthy controls, all of whom were scheduled for planned delivery by caesarean-section were compared with placentas from 10 SLE, 11 pre-eclampsia and 4 SLE+pre-eclampsia cases. SLE cases from deliveries occurring during years 2004–2011 were included. Detailed demographic and clinical data are presented in table 1.
Table 1.
Characteristics of study population
| Characteristics | Control (n=10) |
Pre-eclampsia (N=11) |
SLE (N=10) |
SLE+pre-eclampsia (N=4) |
|---|---|---|---|---|
| Maternal | ||||
| Age at delivery (mean; SD) | 35.5 (4.12) | 29 (4.98) | 31.7 (5.93) | 31.5 (7.77) |
| Race | ||||
| White (not Hispanic) | 5 (50%) | 6 (55%) | 7 (70%) | 4 (100%) |
| African/African–American | 2 (20%) | 1 (9%) | 2 (20%) | 0 |
| Asian | 2 (20%) | 3 (27%) | 0 | 0 |
| Hispanic | 0 | 1 (9%) | 1 (10%) | 0 |
| Multiracial | 1 (10%) | 0 | 0 | 0 |
| Delivery by caesarean- section | 9 (90%) | 9 (82%) | 7 (70%) | 1 (25%) |
| SLE active during pregnancy* | n/a | n/a | 1 (10%) | 1 (25%) |
| Medications during pregnancy† | ||||
| Azathioprine | n/a | n/a | 4 (40%) | 0 |
| Hydroxychloroquine | n/a | n/a | 5 (50%) | 0 |
| SQ heparin and/or 81 mg aspirin | n/a | n/a | 6 (60%) | 4 (100%) |
| Daily prednisone | 4 (40%) | 2 (60%) | ||
| APS | n/a | n/a‡ | 3 (30%) | 1 (25%) |
| ANA+ | ‡ | 1 (9%) | 10 (100%) | 4 (100%) |
| APLAb+ | ‡ | ‡ | 5 (50%) | 1 (25%) |
| ENA+ | ‡ | ‡ | 4 (40%) | 1 (25%) |
| Child | ||||
| Weight, g (mean; SD) | 3505 (264.82) | 2020.36 (1055.97) | 2920.1 (699.33) | 1495 (1190.4)§ |
| Gestational age, weeks (median; IQR) | 39.15 (0.35) | 35 (8.4) | 36.39 (2.55) | 30 (5.88)¶ |
| Duration ROM (median; IQR) | n/a | 0 (0) | 1 (511) | 94 (94) |
*SLE activity: increase in prednisone dose >10 mg/day or new requirement for intravenous steroids.
†Medications included if they were given for duration of pregnancy; steroids included for indication SLE treatment; dexamethasone given for indication of congenital heart block was not included, although two patients with SLE received it for this indication.
‡Not routinely performed in patients with non-rheumatic disease.
§Normal versus pre-eclampsia; normal versus SLE+pre-eclampsia (p=0.0015and 0.0137), respectively.
¶Normal versus pre-eclampsia; normal versus SLE; normal versus SLE+pre-eclampsia (p=0.034, 0.0019, and 0.0105), respectively.
ANA, antinuclear antibody; APLAb, antiphospholipid antibodies; APS, antiphospholipid antibody syndrome; NA, not applicable; ROM, rupture of membranes; SLE, systemic lupus erythematosus; SQ, subcutaneous.
The maternal ages at the time of delivery were comparable between controls and patients with SLE (35.5 (SD±4.12) and 31.7 (SD±5.93), respectively), whereas patients with pre-eclampsia were younger at age of delivery (29 (SD±4.98)). Not surprisingly, all three case groups had significantly shorter mean gestational periods than controls (control vs pre-eclampsia, p=0.034; control vs SLE, p=0.0019; control vs SLE+pre-eclampsia, p=0.0105). The indications for delivery among non-pre-eclampsia lupus subjects were as follows: oligohydramnios (three); premature rupture of membranes (two); IUGR (two), one in a mother with hepatic and splenic thromboses due to APS; breech presentation (one); and increasing MCA Doppler (concern for fetal cerebral oedema) (one). All SLE cases had positive antinuclear antibody (by HEp-2 immunofluorescence). A remote history of biopsy-proven lupus nephritis was present in two in the SLE group and one of the SLE+pre-eclampsia group, but none had baseline renal impairment. Among SLE cases, two developed severe third trimester lupus/APS complications: one developed lupus nephritis and subsequent catastrophic APS in the third trimester, and one developed hepatic and splenic infarction due to APS. One woman experienced a third trimester lupus flare based on malar rash and inflammatory arthritis, with hypertension and proteinuria. She was treated with moderate-dose steroids for lupus and delivered urgently out of concern for pre-eclampsia, which was confirmed by subsequent prompt resolution of hypertension and proteinuria. Among the remainder of SLE cases, disease control was achieved during pregnancy, and no adjustments of prednisone >10 mg/day were made with the exception of dexamethasone administered for the indication of congenital heart block (in two anti-Ro/SSA, anti-La/SSB antibody-positive SLE cases). No cases of chorioamnionitis were identified. One case was excluded from analysis due to prolonged rupture of membranes (eg, lasting longer than 18–24 h between time of rupture and time of delivery),31 and concern for NET induction in response to ascending bacterial infection.
Numbers of neutrophils and neutrophil NETS, and the ratio of NETs/total neutrophils are significantly higher in intervillous space of SLE and pre-eclampsia than controls
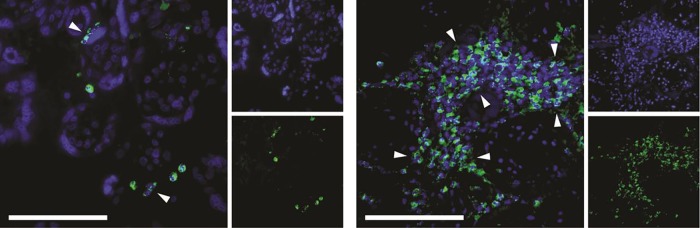
Representative histological examples of intact and netting MPO-stained neutrophils in placental intervillous space are shown in figure 1. Neutrophils with clearly granular MPO staining are representative of intact neutrophils (figure 1A). Neutrophils in which there is a disrupted, decondensed nucleus, with MPO staining, extracellularly represent netting neutrophils (figure 1B). As further evidence of the netting process, a representative image of immunofluorescence staining with anti-MPO and DAPI (for DNA) for two SLE cases is demonstrated in figure 2.
Figure 1.
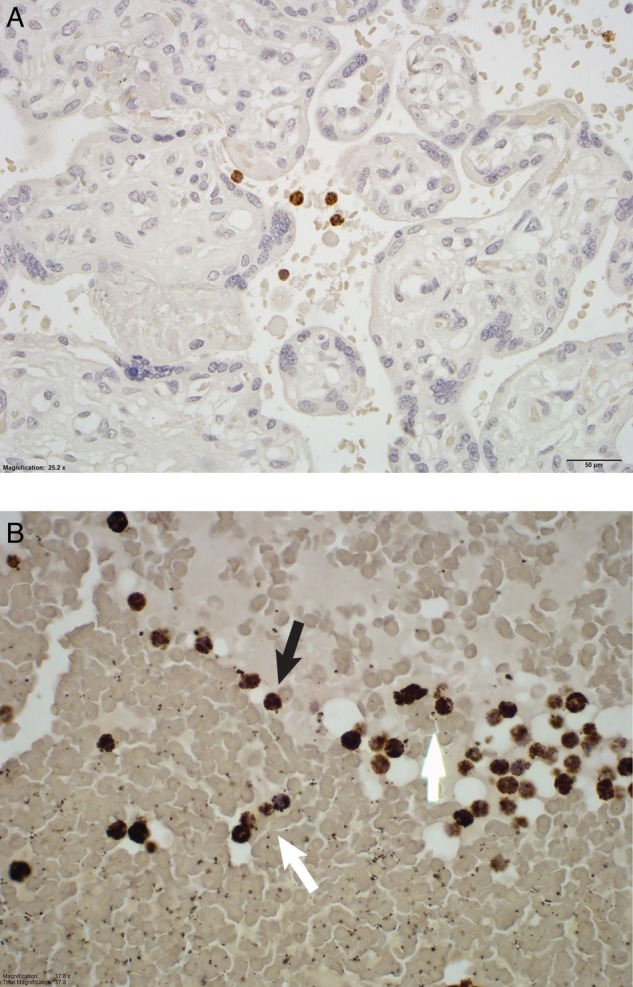
Myeloperoxidase staining of the intervillous space from a healthy control pregnancy (A), a lupus pregnancy (B). Intact neutrophils (black arrows) and netting or non-intact neutrophils (white arrows) are identified.
Figure 2.
Netting neutrophils were confirmed (as myeloperoxidase (MPO)-DNA overlap) by immunofluorescence staining with anti-MPO (green) and DAPI (DNA=blue). These are representative images from two separate patients with systemic lupus erythematosus. The left image shows two discrete neutrophil extracellular traps, while the right image highlights an area of widespread netting. Scale bar=100 μm.
The data for neutrophil and NET quantification in 10 consecutive fields at 60× high-power field are presented in table 2.
Table 2.
Neutrophil and neutrophil extracellular trap (NET) quantification (median (IQR)) in the placental intervillous space
| Control (N=10) | Pre-eclampsia (N=11) | SLE (N=10) | SLE+pre-eclampsia (N=4) | |
|---|---|---|---|---|
| Total neutrophils* | 20.50 (14.75) | 95 (36)† | 99‡ (176.75) | 42.5 (57) |
| Total NETs | 0 (0.75) | 13 (18)§ | 20.5 (43)¶ | 12.50 (23.75)** |
Total neutrophils included intact, netting and indeterminate cells.
*Per 10 high-power field.
†Control versus pre-eclampsia (p=0.0003).
‡Control versus SLE (p=0.0044).
§Control versus pre-eclampsia (p=0.0003).
¶Control versus SLE (p=0.0010).
**Control versus SLE+pre-eclampsia (p=0.0717).
SLE, systemic lupus erythematosus.
Of note, scarce, if any, NETs were seen in the controls. The numbers of both total neutrophils and neutrophil NETs in placental intervillous space were significantly higher in the pre-eclampsia and SLE groups compared with controls. These values were also higher in the SLE+pre-eclampsia group compared with controls, though not reaching statistical significance, likely due to the small sample size in this group (n=4). Among the case groups (pre-eclampsia, SLE and SLE+pre-eclampsia), the counts of total neutrophils and NETs did not differ statistically.
Furthermore, we also assessed the relative proportions of neutrophils forming NETs within each group by calculating the ratio of NETs to total neutrophils (intact, indeterminate and netting). As displayed in figure 3, the NET:total neutrophil ratios were significantly higher in each of the three case groups compared with controls, demonstrating a higher level of NETosis accounting for the absolute neutrophil numbers. The magnitude of the NET:neutrophil ratio was highest in the combined SLE+pre-eclampsia group. There was no correlation between neutrophils or NETs and the duration of membrane rupture, making ascending infection as a cause of neutrophil infiltration less likely. In summary, these data suggest that the large neutrophilic infiltrate in the intervillous space of SLE and pre-eclampsia pregnancies is a prominent and common feature of both processes that is not present in normal pregnancies.
Figure 3.
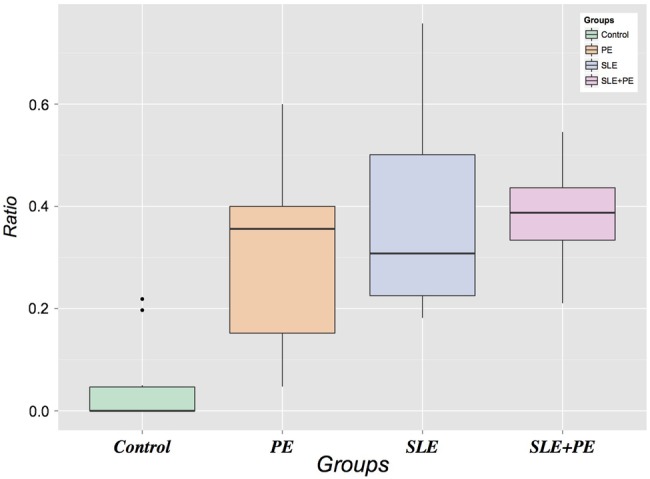
Ratio of neutrophil extracellular traps to total neutrophils (intact, indeterminate and netting). SLE, systemic lupus erythematosus.
NETs predict the presence of histopathological findings
We examined histopathological features on H&E-stained placental tissue with particular attention to vascular and inflammatory phenomena for correlation with the presence of NETs in the intervillous space (table 3).
Table 3.
The presence of intervillous neutrophil extracellular traps (NETs) versus no NETs in all cases corresponds to vascular pathology and inflammation*
| Variables | No NETs (median; IQR) |
NETs (median; IQR) |
p Value |
|---|---|---|---|
| Inflammation | |||
| Maternal | |||
| Chronic | 8 (26) | 16.5 (6.5) | NS |
| Acute | 4 (14) | 19 (14) | 0.06559 |
| Fetal | |||
| Chronic | 9.5 (23) | 10 (16) | NS |
| Acute | 9 (20.75) | 32 (0) | NS |
| Circulatory | |||
| Decidual vasculopathy | |||
| Perivasculitis; mild | 4 (26) | 14 (10.25) | NS |
| Vasculitis severe; fibrinoid necrosis | 4.5 (14.75) | 23 (39) | 0.02286 |
| Maternal thrombosis: occlusive | 10.5 (24) | 4 (13) | NS |
| Maternal; non-occlusive | 9 (25) | 11 (0) | NS |
| Laminar decidual necrosis (>10%) | 4.5 (14.75) | 23 (31) | 0.03458 |
| Infarct | |||
| Fetal: focal villous infarction | 8 (26) | 11.5 (10.5) | NS |
| Fetal: multifocal villous infarction | 6.5 (14.75) | 23 (12) | NS |
| Maternal–fetal interface haemorrhage† | 4.5 (13.75) | 29 (40) | 0.02116 |
| Fetal vasculopathy | |||
| Distal villous lesions | 19 (19.75) | 13.5 (31.75) | NS |
| Large vessel lesions | 15 (19) | 13 (30.5) | NS |
| Choriangiosis | 24.5 (17.25) | 13.5 (32) | NS |
| Fetal thrombotic vasculopathy | |||
| Occlusive | 9 (16.25) | 45 (26.5) | NS |
| Non-occlusive | 4 (13.25) | 29 (23) | 0.003374 |
*Comparison between groups tested by Kruskal–Wallis.
†Acute and chronic haemorrhage are not presented, but were NS separately likely due to small numbers.
Of the histopathological features, the presence of NETs in the intervillous space corresponded primarily to pathologic vascular and inflammatory changes of maternal decidua. In particular, NETs were associated with laminar necrosis of placental membranes, and maternal–fetal interface haemorrhage. NETs also correlated to the presence of fetal non-occlusive thrombotic vasculopathy. However, we did not observe a correlation with NETs and more extensive fetal vascular pathology: for example, there was no association with the presence of atherosis, distal villous or large vessel lesions, or villous infarct, although that could be due in part to limited sample size. Acute maternal inflammation as a group (intervillositis, amnionitis) neared a significant association with the presence of NETs in the intervillous space; however, chronic inflammatory changes did not.
Discussion
In this study, we performed a detailed pathological review of placentas from lupus pregnancies and from pre-eclampsia pregnancies. The lupus pregnancies occurred between years 2004 and 2011, during which time improvements in management of lupus pregnancy were occurring, due to multidisciplinary approaches in care including preconception counselling, a general acknowledgement that 6 months of good lupus disease control prior to conception improved outcomes and the continuation of antimalarial therapy throughout pregnancy.32 The placental pathology from lupus pregnancies exhibited features of inflammatory vasculopathy comparable to pre-eclampsia, arguably the most severe and potentially fatal disease of pregnancy. In particular, a more robust non-infectious infiltrate of both intact neutrophils and NETs in the placental intervillous space was present in both lupus and pre-eclampsia compared with control pregnancies. Further, these infiltrates corresponded to histological changes of the placenta consistent with inflammation and vasculopathy (decidual vasculitis, laminar necrosis, maternal–fetal interface haemorrhage, fetal thrombotic vasculopathy). These findings are intriguing in the context of the well-recognised toxic effect of NETs on vascular endothelium and raise the possibility that NETs are actually driving the observed histopathological changes.
These abnormal placental findings may represent another manifestation of subclinical vascular disease that is well described in SLE, both in studies of carotid intimal media thickness33–35 and abnormal myocardial perfusion.36 Indeed, increased risk of cardiovascular disease in both lupus and in pre-eclampsia, in the absence of traditional Framingham risk factors, is recognised to contribute to the excess morbidity and mortality observed in long-term follow-up of these populations.37–41 Our findings suggest a similar phenomenon occurring within the placenta, a highly vascularised target organ, corresponding to the observed high rates of IUGR and oligohydramnios in these pregnancies. IUGR in turn has been observed at a rate of 5.6% in lupus pregnancies from the Nationwide Inpatient Sample, as opposed to 0.09% in the general population,2 and in 20% of the small lupus population from our study.
In SLE vasculopathy, the imbalance between vascular damage and repair is characterised in part by increased endothelial cell apoptosis and decreased number and function of endothelial progenitor cells.35 A similar phenomenon has been observed in the placentas of patients with pre-eclampsia, in which increased trophoblast apoptosis and impaired spiral artery structure and function are observed.42 In SLE, increased endothelial cell apoptosis is associated with a novel proinflammatory subset of lupus neutrophils, the ‘low-density granulocyte’ (LDG), which expresses significantly higher levels of type I interferon mRNA compared with control or autologous lupus neutrophils, and is especially predisposed to NET formation.14 43 44 The same NET contents that disarm and kill bacteria extracellularly are toxic to endothelial cells in vitro14 and may have similar toxic or ‘pro-apoptotic’ effects on trophoblasts, resulting in impaired placentation.
NET formation may also result in externalisation of matrix metalloproteinases (MMPs), which are also present within neutrophil granules.45 MMPs, a family of proteases that degrade extracellular matrix proteins, can damage vascular cell endothelial integrity and function.46 Higher levels of MMP-9 in particular are externalised by NETs formed from lupus LDGs, which, after activation with MMP-2, have been shown to impair aortic endothelium-dependent vasorelaxation and induce endothelial cell apoptosis in lupus-prone mice.45 The imbalance between MMPs and their inhibitors may also impact uterine spiral artery remodelling,47 thereby adversely affecting placental implantation, growth and development in both SLE and pre-eclampsia.
A causal role for inflammation in the genesis of pre-eclampsia has not been established. However, a 2005 paper by Gupta et al12 described NETs infiltrating the intervillous space of placentas from patients with pre-eclampsia, with proposed neutrophil activation by syncytiotrophoblast microparticles as the triggering event for NET formation. Here, using different methods to identify neutrophil NETs, we observed NETs in the intervillous space of patients with pre-eclampsia and for the first time in the intervillous space of patients with SLE. In this setting, NETs may provide a source of immunostimulatory, pro-inflammatory and antiangiogenic mediators that are passed on to the fetus with unknown consequences.
Study limitations include the lack of prospectively obtained lupus disease assessment using a validated activity instrument (eg, the SLE Disease Activity Index):48 rather, assessment of lupus activity was based on chart review by a study rheumatologist with experience in lupus (WM) with attention to medication changes, including prednisone adjustments. Additionally, the number of cases, particularly among the SLE+pre-eclampsia group, was small: a larger sample size could adequately assess whether the combination of both processes results in a more aggressive immunological process. Finally, it is difficult to ascertain whether the pathological findings observed in this placental tissue represent cause or effect of observed birth outcomes such as oligohydramnios or IUGR, and to what extent underlying SLE and APS contribute.
The strengths of this study include a rigorous histopathological review and neutrophil quantification of all placental tissue by an experienced placental pathologist, as well as novel immunohistochemical staining for neutrophil NETs in an experienced lab with a focus on NET biology. The findings of this novel work add to a growing body of research related to lupus pregnancy outcomes. Over the past several decades, increasingly more women with SLE are achieving successful pregnancies thanks to improvements in disease diagnosis and management.49–51 Indeed, recent findings from the PROMISSE study reveal better outcomes of lupus pregnancies than had been previously observed.52 The findings from the current study, revealing a higher rate of adverse pregnancy outcomes and abnormal placental structure and function among this group of generally well-controlled lupus pregnancies, raise the issue of long-term implications for the developing fetus. Our findings support observations that lupus pregnancies, particularly in association with antiphospholipid antibodies, are more at risk for complications, even with good disease control. Future directions for this work should focus on characterising the role of neutrophils and NETs on placental development and function in lupus pregnancies as well as focusing on associations between placental pathology observed in this study with long-term developmental outcomes of lupus offspring.
Footnotes
Contributors: All authors involved in this paper contributed substantially to the conception, design or conduction of the research or the acquisition, analysis or interpretation of data for the work. Additionally, all authors were involved in drafting the work or revising it critically for important intellectual content. Furthermore, all authors gave final approval of the version to be published. All authors agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Funding: WM was supported by K12HD001438 from NIH. ECS was supported by NIH 5K01ES019909. JSK was supported by NIH K08AR066569 and a career development award from the Burroughs Wellcome Fund. This work was performed while MJK was employed at the University of Michigan.
Disclaimer: The opinions expressed in this article are the author's own and do not reflect the views of the National Institutes of Health, the Department of Health and Human Services, or the US government.
Competing interests: None declared.
Ethics approval: University of Michigan Medical School Internal Review Board.
Provenance and peer review: Not commissioned; externally peer reviewed.
Data sharing statement: No additional data are available.
References
- 1.Gordon C. Pregnancy and autoimmune diseases. Best Pract Res Clin Rheumatol 2004;18:359–79. doi:10.1016/j.berh.2004.02.012 [DOI] [PubMed] [Google Scholar]
- 2.Clowse ME, Jamison M, Myers E et al. . A national study of the complications of lupus in pregnancy. Am J Obstet Gynecol 2008;199:1–12. doi:10.1016/j.ajog.2008.03.012 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Clowse MEB, Magder LS, Witter F et al. . The impact of increased lupus activity on obstetric outcomes. Arthritis Rheum 2005;52:514–21. doi:10.1002/art.20864 [DOI] [PubMed] [Google Scholar]
- 4.Steegers EA, von Dadelszen P, Duvekot JJ et al. . Pre-eclampsia. Lancet 2010;376:631–44. doi:10.1016/S0140-6736(10)60279-6 [DOI] [PubMed] [Google Scholar]
- 5.Magid MS, Kaplan C, Sammaritano LR et al. . Placental pathology in systemic lupus erythematosus: a prospective study. Am J Obstet Gynecol 1998;179:226–34. doi:10.1016/S0002-9378(98)70277-7 [DOI] [PubMed] [Google Scholar]
- 6.Knight JS, Kaplan MJ. Lupus neutrophils: “NET” gain in understanding lupus pathogenesis. Curr Opin Rheumatol 2012;24:441–50. doi:10.1097/BOR.0b013e3283546703 [DOI] [PubMed] [Google Scholar]
- 7.Elkon KB, Wiedeman A. Type I IFN system in the development and manifestations of SLE. Curr Opin Rheumatol 2012;24: 499–505. doi:10.1097/BOR.0b013e3283562c3e [DOI] [PubMed] [Google Scholar]
- 8.Rao AN, Kazzaz NM, Knight JS. Do neutrophil extracellular traps contribute to the heightened risk of thrombosis in inflammatory diseases? World J Cardiol 2015;7:829–42. doi:10.4330/wjc.v7.i12.829 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Segal AW. The function of the NADPH oxidase of phagocytes and its relationship to other NOXs in plants, invertebrates, and mammals. Int J Biochem Cell Biol 2008;40:604–18. doi:10.1016/j.biocel.2007.10.003 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.HIRSCH JG, COHN ZA. Degranulation of polymorphonuclear leucocytes following phagocytosis of microorganisms. J Exp Med 1960;112:1005–14. doi:10.1084/jem.112.6.1005 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Keshari RS, Jyoti A, Dubey M et al. . Cytokines induced neutrophil extracellular traps formation: implication for the inflammatory disease condition. PLoS ONE 2012;7:e48111 doi:10.1371/journal.pone.0048111 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Gupta AK, Hasler P, Holzgreve W et al. . Induction of neutrophil extracellular DNA lattices by placental microparticles and IL-8 and their presence in preeclampsia. Hum Immunol 2005;66:1146–54. doi:10.1016/j.humimm.2005.11.003 [DOI] [PubMed] [Google Scholar]
- 13.Behnen M, Leschczyk C, Möller S et al. . Immobilized immune complexes induce neutrophil extracellular trap release by human neutrophil Granulocytes via Fc RIIIB and Mac-1. J Immunol 2014;193:1954–65. doi:10.4049/jimmunol.1400478 [DOI] [PubMed] [Google Scholar]
- 14.Villanueva E, Yalavarthi S, Berthier CC et al. . Netting neutrophils induce endothelial damage, infiltrate tissues, and expose immunostimulatory molecules in systemic lupus erythematosus. J Immunol 2011;187:538–52. doi:10.4049/jimmunol.1100450 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Andrews RK, Arthur JF, Gardiner EE. Neutrophil extracellular traps (NETs) and the role of platelets in infection. Thromb Haemost 2014;112:659–65. doi:10.1160/TH14-05-0455 [DOI] [PubMed] [Google Scholar]
- 16.Yalavarthi S, Gould TJ, Rao AN et al. . Release of neutrophil extracellular traps by neutrophils stimulated with antiphospholipid antibodies: a newly identified mechanism of thrombosis in the antiphospholipid syndrome. Arthritis Rheumatol 2015;67:2990–3003. doi:10.1002/art.39247 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Zhang S, Lu X, Shu X et al. . Elevated plasma cfDNA may be associated with active lupus nephritis and partially attributed to abnormal regulation of neutrophil extracellular traps (NETs) in patients with systemic lupus erythematosus. Intern Med 2014;53:2763–71. doi:10.2169/internalmedicine.53.2570 [DOI] [PubMed] [Google Scholar]
- 18.Garcia-Romo GS, Caielli S, Vega B et al. . Netting neutrophils are major inducers of type I IFN production in pediatric systemic lupus erythematosus. Sci Transl Med 2011;3:73ra20 doi:10.1126/scitranslmed.3001201 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Lande R, Ganguly D, Facchinetti V, et al Neutrophils activate plasmacytoid dendritic cells by releasing self-DNA-peptide complexes in systemic lupus erythematosus. Sci Transl Med 2011;3:73ra19 doi:10.1126/scitranslmed.3001180 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Kessenbrock K, Krumbholz M, Schönermarck U et al. . Netting neutrophils in autoimmune small-vessel vasculitis. Nat Med 2009;15:623–5. doi:10.1038/nm.1959 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Gupta AK, Hasler P, Holzgreve W et al. . Neutrophil NETs: a novel contributor to preeclampsia-associated placental hypoxia? Semin Immunopathol 2007;29:163–7. doi:10.1007/s00281-007-0073-4 [DOI] [PubMed] [Google Scholar]
- 22.Tan EM, Cohen AS, Fries JF et al. . The 1982 revised criteria for the classification of systemic lupus erythematosus. Arthritis Rheum 1982;25:1271–7. doi:10.1002/art.1780251101 [DOI] [PubMed] [Google Scholar]
- 23.Hochberg MC. Updating the American College of Rheumatology revised criteria for the classification of systemic lupus erythematosus. Arthritis Rheum 1997;40:1725 doi:10.1002/1529-0131(199709)40:9<1725::AID-ART29>3.0.CO;2-Y [DOI] [PubMed] [Google Scholar]
- 24.Wilson WA, Gharavi AE, Koike T et al. . International consensus statement on preliminary classification criteria for definite antiphospholipid syndrome: report of an international workshop. Arthritis Rheum 1999;42:1309–11. doi:10.1002/1529-0131(199907)42:7<1309::AID-ANR1>3.0.CO;2-F [DOI] [PubMed] [Google Scholar]
- 25.Miyakis S, Lockshin MD, Atsumi T et al. . International consensus statement on an update of the classification criteria for definite antiphospholipid syndrome (APS). J Thromb Haemost 2006;4:295–306. doi:10.1111/j.1538-7836.2006.01753.x [DOI] [PubMed] [Google Scholar]
- 26.Clark EA, Silver RM, Branch DW. Do antiphospholipid antibodies cause preeclampsia and HELLP syndrome? Curr Rheumatol Rep 2007;9:219–25. doi:10.1007/s11926-007-0035-9 [DOI] [PubMed] [Google Scholar]
- 27.Davey DA, MacGillivray I. The classification and definition of the hypertensive disorders of pregnancy. Am J Obstet Gynecol 1988;158:892–8. doi:10.1016/0002-9378(88)90090-7 [DOI] [PubMed] [Google Scholar]
- 28.Benirschke K, Burton GJ, Baergen RN. Pathology of the human placenta. 6th edn Berlin Heidelberg: Springer-Verlag, 2012. [Google Scholar]
- 29.Salafia CM, Popek EJ. Inflammatory and Vascular Placental Pathology. Glob Libr Women's Med 2009. http://www.glowm.com/index.html?p=glowm.cml/section_view&articleid=152 (accessed 15 Sep 2014). [Google Scholar]
- 30.Khandpur R, Carmona-Rivera C, Vivekanandan-Giri A et al. . NETs are a source of citrullinated autoantigens and stimulate inflammatory responses in rheumatoid arthritis. Sci Transl Med 2013;5:178ra40 doi:10.1126/scitranslmed.3005580 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Callahan TL. Blueprints obstetrics and gynecology. CA: Lippincott Williams & Wilkins, 2008. [Google Scholar]
- 32.Gordon P, Beedham T, Khamashta M et al. . Systemic lupus erythematosus in pregnancy. Obstet Gynaecol 2004;6:80–7. doi:10.1576/toag.6.2.80.26982 [Google Scholar]
- 33.Hahn S, Giaglis S, Hoesli I et al. . Neutrophil NETs in reproduction: from infertility to preeclampsia and the possibility of fetal loss. Front Immunol 2012;3:362 doi:10.3389/fimmu.2012.00362 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Manzi S, Selzer F, Sutton-Tyrrell K et al. . Prevalence and risk factors of carotid plaque in women with systemic lupus erythematosus. Arthritis Rheum 1999;42:51–60. doi:10.1002/1529-0131(199901)42:1<51::AID-ANR7>3.0.CO;2-D [DOI] [PubMed] [Google Scholar]
- 35.Rajagopalan S, Somers EC, Brook RD et al. . Endothelial cell apoptosis in systemic lupus erythematosus: a common pathway for abnormal vascular function and thrombosis propensity. Blood 2004;103:3677–83. doi:10.1182/blood-2003-09-3198 [DOI] [PubMed] [Google Scholar]
- 36.Bruce IN, Burns RJ, Gladman DD et al. . Single photon emission computed tomography dual isotope myocardial perfusion imaging in women with systemic lupus erythematosus. I. Prevalence and distribution of abnormalities. J Rheumatol 2000;27:2372–7. [PubMed] [Google Scholar]
- 37.Urowitz MB, Gladman D, Ibañez D et al. . Accumulation of coronary artery disease risk factors over three years: data from an international inception cohort. Arthritis Rheum 2008;59:176–80. doi:10.1002/art.23353 [DOI] [PubMed] [Google Scholar]
- 38.Urowitz MB, Bookman AA, Koehler BE et al. . The bimodal mortality pattern of systemic lupus erythematosus. Am J Med 1976;60:221–5. doi:10.1016/0002-9343(76)90431-9 [DOI] [PubMed] [Google Scholar]
- 39.Manzi S, Meilahn EN, Rairie JE et al. . Age-specific incidence rates of myocardial infarction and angina in women with systemic lupus erythematosus: comparison with the Framingham Study. Am J Epidemiol 1997;145:408–15. doi:10.1093/oxfordjournals.aje.a009122 [DOI] [PubMed] [Google Scholar]
- 40.Kessous R, Shoham-Vardi I, Pariente G et al. . Long-term maternal atherosclerotic morbidity in women with pre-eclampsia. Heart 2015;101:442–6. doi:10.1136/heartjnl-2014-306571 [DOI] [PubMed] [Google Scholar]
- 41.Funai EF, Friedlander Y, Paltiel O et al. . Long-term mortality after preeclampsia. Epidemiology 2005;16:206–15. doi:10.1097/01.ede.0000152912.02042.cd [DOI] [PubMed] [Google Scholar]
- 42.Allaire A. Placental apoptosis in preeclampsia. Obstet Gynecol 2000;96:271–6. [DOI] [PubMed] [Google Scholar]
- 43.Knight JS, Kaplan MJ. Cardiovascular disease in lupus: insights and updates. Curr Opin Rheumatol 2013;25:597–605. doi:10.1097/BOR.0b013e328363eba3 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Denny MF, Yalavarthi S, Zhao W et al. . A distinct subset of proinflammatory neutrophils isolated from patients with systemic lupus erythematosus induces vascular damage and synthesizes type I IFNs. J Immunol 2010;184:3284–97. doi:10.4049/jimmunol.0902199 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Carmona-Rivera C, Zhao W, Yalavarthi S et al. . Neutrophil extracellular traps induce endothelial dysfunction in systemic lupus erythematosus through the activation of matrix metalloproteinase-2. Ann Rheum Dis 2015;74:1417–24. doi:10.1136/annrheumdis-2013-204837 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Fert-Bober J, Leon H, Sawicka J et al. . Inhibiting matrix metalloproteinase-2 reduces protein release into coronary effluent from isolated rat hearts during ischemia-reperfusion. Basic Res Cardiol 2008;103:431–43. doi:10.1007/s00395-008-0727-y [DOI] [PubMed] [Google Scholar]
- 47.Montagnana M, Lippi G, Albiero A et al. . Evaluation of metalloproteinases 2 and 9 and their inhibitors in physiologic and pre-eclamptic pregnancy. J Clin Lab Anal 2009;23:88–92. doi:10.1002/jcla.20295 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Bombardier C, Gladman DD, Urowitz MB et al. . Derivation of the SLEDAI. A disease activity index for lupus patients. The Committee on Prognosis Studies in SLE. Arthritis Rheum 1992;35:630–40. [DOI] [PubMed] [Google Scholar]
- 49.Clark CA, Spitzer KA, Laskin CA. Decrease in pregnancy loss rates in patients with systemic lupus erythematosus over a 40-year period. J Rheumatol 2005;32:1709–12. [PubMed] [Google Scholar]
- 50.Jain V, Gordon C. Managing pregnancy in inflammatory rheumatological diseases. Arthritis Res Ther 2011;13:206 doi:10.1186/ar3227 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Khamashta MA. Systemic lupus erythematosus and pregnancy. Best Pract Res Clin Rheumatol 2006;20:685–94. doi:10.1016/j.berh.2006.04.003 [DOI] [PubMed] [Google Scholar]
- 52.Buyon JP, Kim MY, Guerra MM et al. . Predictors of pregnancy outcomes in patients with lupus: a cohort study. Ann Intern Med 2015;163:153–63. doi:10.7326/M14-2235 [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
lupus-2015-000134supp.pdf (87.5KB, pdf)