Abstract
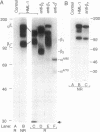
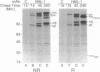
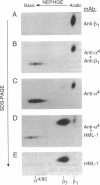
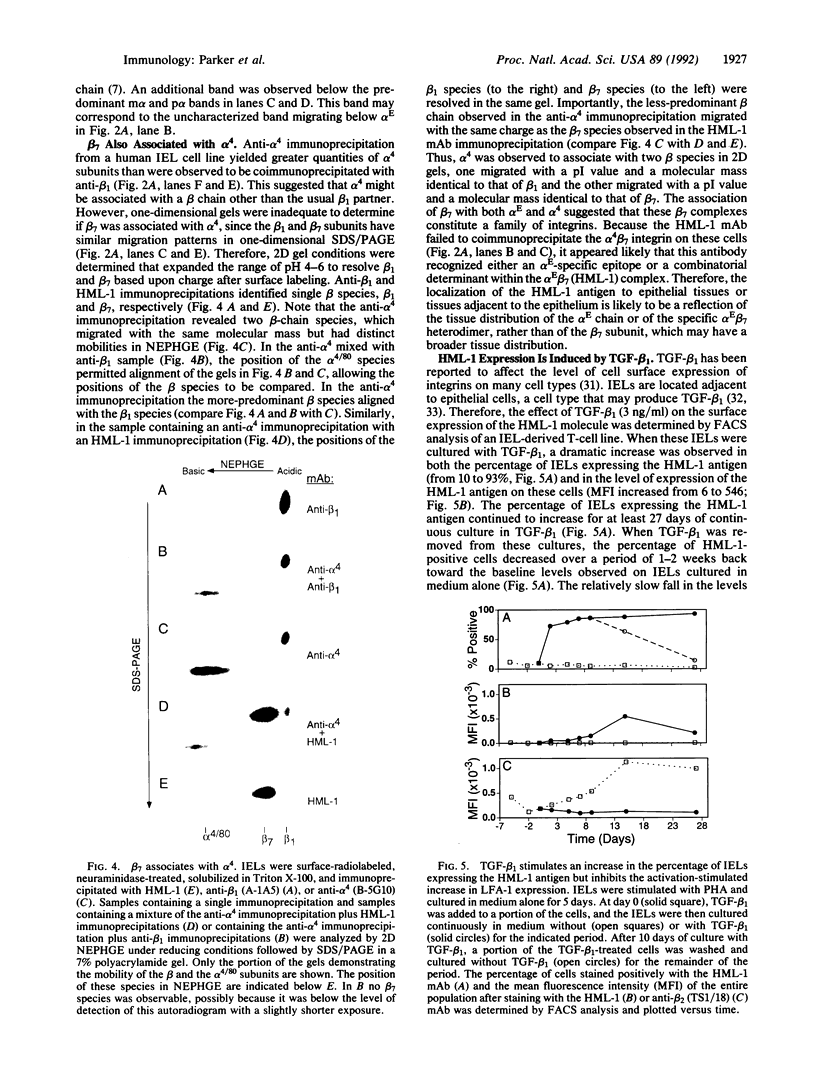
The heterodimeric protein complex recognized by the human mucosal lymphocyte 1 (HML-1) monoclonal antibody is expressed on 95% of intraepithelial lymphocytes but on only 1-2% of peripheral blood lymphocytes [Cerf-Bensusson, N., Jarry, A., Brousse, N., Lisowska-Grospierre, B., Guy-Grand, D. & Griscelli, C. (1987) Eur. J. Immunol. 17, 1279-1285]. We purified the smaller HML-1 subunit (105 kDa under nonreducing conditions) from hairy-cell leukemia cells and determined the N-terminal amino acid sequence of this chain. The 17 residues determined were identical to the deduced amino acid sequence encoded by an integrin beta 7 cDNA clone [Yuan, Q., Jiang, W.-M., Krissansen, G.W. & Watson, J.D. (1990) Int. Immunol. 2, 1097-1108]. Biochemical analysis of the larger HML-1 subunit (175 kDa under nonreducing conditions) suggested that it was a distinct member of the cleaved group of integrin alpha chains, which we designated alpha E. The beta 7 chain also was associated with the integrin alpha 4 subunit, suggesting that the HML-1 antigen (alpha E beta 7) and alpha 4 beta 7 constitute a beta 7 integrin family on mucosal lymphocytes. Interestingly, regulation of the expression of the HML-1 antigen was reciprocal to that of lymphocyte function-associated molecule 1 in the presence of transforming growth factor beta 1. We suggest that these beta 7 integrins may play a specific role in mucosal localization or adhesion and that the expression of the HML-1 antigen might be regulated by transforming growth factor beta 1 produced at or near epithelial tissues.
Full text
PDF



Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Asarnow D. M., Goodman T., LeFrancois L., Allison J. P. Distinct antigen receptor repertoires of two classes of murine epithelium-associated T cells. Nature. 1989 Sep 7;341(6237):60–62. doi: 10.1038/341060a0. [DOI] [PubMed] [Google Scholar]
- Barnard J. A., Beauchamp R. D., Coffey R. J., Moses H. L. Regulation of intestinal epithelial cell growth by transforming growth factor type beta. Proc Natl Acad Sci U S A. 1989 Mar;86(5):1578–1582. doi: 10.1073/pnas.86.5.1578. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brandtzaeg P., Sollid L. M., Thrane P. S., Kvale D., Bjerke K., Scott H., Kett K., Rognum T. O. Lymphoepithelial interactions in the mucosal immune system. Gut. 1988 Aug;29(8):1116–1130. doi: 10.1136/gut.29.8.1116. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brenner M. B., McLean J., Scheft H., Warnke R. A., Jones N., Strominger J. L. Characterization and expression of the human alpha beta T cell receptor by using a framework monoclonal antibody. J Immunol. 1987 Mar 1;138(5):1502–1509. [PubMed] [Google Scholar]
- Cerf-Bensussan N., Jarry A., Brousse N., Lisowska-Grospierre B., Guy-Grand D., Griscelli C. A monoclonal antibody (HML-1) defining a novel membrane molecule present on human intestinal lymphocytes. Eur J Immunol. 1987 Sep;17(9):1279–1285. doi: 10.1002/eji.1830170910. [DOI] [PubMed] [Google Scholar]
- Davies M. D., Parrott D. M. Preparation and purification of lymphocytes from the epithelium and lamina propria of murine small intestine. Gut. 1981 Jun;22(6):481–488. doi: 10.1136/gut.22.6.481. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Erle D. J., Rüegg C., Sheppard D., Pytela R. Complete amino acid sequence of an integrin beta subunit (beta 7) identified in leukocytes. J Biol Chem. 1991 Jun 15;266(17):11009–11016. [PubMed] [Google Scholar]
- Hemler M. E., Elices M. J., Parker C., Takada Y. Structure of the integrin VLA-4 and its cell-cell and cell-matrix adhesion functions. Immunol Rev. 1990 Apr;114:45–65. doi: 10.1111/j.1600-065x.1990.tb00561.x. [DOI] [PubMed] [Google Scholar]
- Hemler M. E., Huang C., Takada Y., Schwarz L., Strominger J. L., Clabby M. L. Characterization of the cell surface heterodimer VLA-4 and related peptides. J Biol Chem. 1987 Aug 25;262(24):11478–11485. [PubMed] [Google Scholar]
- Hemler M. E. VLA proteins in the integrin family: structures, functions, and their role on leukocytes. Annu Rev Immunol. 1990;8:365–400. doi: 10.1146/annurev.iy.08.040190.002053. [DOI] [PubMed] [Google Scholar]
- Hemler M. E., Ware C. F., Strominger J. L. Characterization of a novel differentiation antigen complex recognize by a monoclonal antibody (A-1A5): unique activation-specific molecular forms on stimulated T cells. J Immunol. 1983 Jul;131(1):334–340. [PubMed] [Google Scholar]
- Hochstenbach F., Parker C., McLean J., Gieselmann V., Band H., Bank I., Chess L., Spits H., Strominger J. L., Seidman J. G. Characterization of a third form of the human T cell receptor gamma/delta. J Exp Med. 1988 Aug 1;168(2):761–776. doi: 10.1084/jem.168.2.761. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Holzmann B., Weissman I. L. Peyer's patch-specific lymphocyte homing receptors consist of a VLA-4-like alpha chain associated with either of two integrin beta chains, one of which is novel. EMBO J. 1989 Jun;8(6):1735–1741. doi: 10.1002/j.1460-2075.1989.tb03566.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kanof M. E., Strober W., Fiocchi C., Zeitz M., James S. P. CD4 positive Leu-8 negative helper-inducer T cells predominate in the human intestinal lamina propria. J Immunol. 1988 Nov 1;141(9):3029–3036. [PubMed] [Google Scholar]
- Kilshaw P. J., Murant S. J. A new surface antigen on intraepithelial lymphocytes in the intestine. Eur J Immunol. 1990 Oct;20(10):2201–2207. doi: 10.1002/eji.1830201008. [DOI] [PubMed] [Google Scholar]
- Kitagawa T., Aikawa T. Enzyme coupled immunoassay of insulin using a novel coupling reagent. J Biochem. 1976 Jan;79(1):233–236. doi: 10.1093/oxfordjournals.jbchem.a131053. [DOI] [PubMed] [Google Scholar]
- Kruschwitz M., Fritzsche G., Schwarting R., Micklem K., Mason D. Y., Falini B., Stein H. Ber-ACT8: new monoclonal antibody to the mucosa lymphocyte antigen. J Clin Pathol. 1991 Aug;44(8):636–645. doi: 10.1136/jcp.44.8.636. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Köhler G., Milstein C. Continuous cultures of fused cells secreting antibody of predefined specificity. Nature. 1975 Aug 7;256(5517):495–497. doi: 10.1038/256495a0. [DOI] [PubMed] [Google Scholar]
- Marcantonio E. E., Hynes R. O. Antibodies to the conserved cytoplasmic domain of the integrin beta 1 subunit react with proteins in vertebrates, invertebrates, and fungi. J Cell Biol. 1988 May;106(5):1765–1772. doi: 10.1083/jcb.106.5.1765. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Massagué J. The transforming growth factor-beta family. Annu Rev Cell Biol. 1990;6:597–641. doi: 10.1146/annurev.cb.06.110190.003121. [DOI] [PubMed] [Google Scholar]
- Matsudaira P. Sequence from picomole quantities of proteins electroblotted onto polyvinylidene difluoride membranes. J Biol Chem. 1987 Jul 25;262(21):10035–10038. [PubMed] [Google Scholar]
- Mayer L., Shlien R. Evidence for function of Ia molecules on gut epithelial cells in man. J Exp Med. 1987 Nov 1;166(5):1471–1483. doi: 10.1084/jem.166.5.1471. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Morrissey J. H. Silver stain for proteins in polyacrylamide gels: a modified procedure with enhanced uniform sensitivity. Anal Biochem. 1981 Nov 1;117(2):307–310. doi: 10.1016/0003-2697(81)90783-1. [DOI] [PubMed] [Google Scholar]
- Parker C. M., Groh V., Band H., Porcelli S. A., Morita C., Fabbi M., Glass D., Strominger J. L., Brenner M. B. Evidence for extrathymic changes in the T cell receptor gamma/delta repertoire. J Exp Med. 1990 May 1;171(5):1597–1612. doi: 10.1084/jem.171.5.1597. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sanchez-Madrid F., Nagy J. A., Robbins E., Simon P., Springer T. A. A human leukocyte differentiation antigen family with distinct alpha-subunits and a common beta-subunit: the lymphocyte function-associated antigen (LFA-1), the C3bi complement receptor (OKM1/Mac-1), and the p150,95 molecule. J Exp Med. 1983 Dec 1;158(6):1785–1803. doi: 10.1084/jem.158.6.1785. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schieferdecker H. L., Ullrich R., Weiss-Breckwoldt A. N., Schwarting R., Stein H., Riecken E. O., Zeitz M. The HML-1 antigen of intestinal lymphocytes is an activation antigen. J Immunol. 1990 Apr 1;144(7):2541–2549. [PubMed] [Google Scholar]
- Schwarting R., Dienemann D., Kruschwitz M., Fritsche G., Stein H. Specificities of monoclonal antibodies B-ly7 and HML-1 are identical. Blood. 1990 Jan 1;75(1):320–321. [PubMed] [Google Scholar]
- Shimizu Y., Van Seventer G. A., Horgan K. J., Shaw S. Regulated expression and binding of three VLA (beta 1) integrin receptors on T cells. Nature. 1990 May 17;345(6272):250–253. doi: 10.1038/345250a0. [DOI] [PubMed] [Google Scholar]
- Springer T. A. Adhesion receptors of the immune system. Nature. 1990 Aug 2;346(6283):425–434. doi: 10.1038/346425a0. [DOI] [PubMed] [Google Scholar]
- Suzuki S., Naitoh Y. Amino acid sequence of a novel integrin beta 4 subunit and primary expression of the mRNA in epithelial cells. EMBO J. 1990 Mar;9(3):757–763. doi: 10.1002/j.1460-2075.1990.tb08170.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Van Kerckhove C., Russell G. J., Deusch K., Reich K., Bhan A. K., DerSimonian H., Brenner M. B. Oligoclonality of human intestinal intraepithelial T cells. J Exp Med. 1992 Jan 1;175(1):57–63. doi: 10.1084/jem.175.1.57. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yednock T. A., Rosen S. D. Lymphocyte homing. Adv Immunol. 1989;44:313–378. doi: 10.1016/s0065-2776(08)60645-8. [DOI] [PubMed] [Google Scholar]
- Yelton D. E., Desaymard C., Scharff M. D. Use of monoclonal anti-mouse immunoglobulin to detect mouse antibodies. Hybridoma. 1981;1(1):5–11. doi: 10.1089/hyb.1.1981.1.5. [DOI] [PubMed] [Google Scholar]
- Yokoyama W. M., Maxfield S. R., Shevach E. M. Very early (VEA) and very late (VLA) activation antigens have distinct functions in T lymphocyte activation. Immunol Rev. 1989 Jun;109:153–176. doi: 10.1111/j.1600-065x.1989.tb00024.x. [DOI] [PubMed] [Google Scholar]
- Yssel H., De Vries J. E., Koken M., Van Blitterswijk W., Spits H. Serum-free medium for generation and propagation of functional human cytotoxic and helper T cell clones. J Immunol Methods. 1984 Aug 3;72(1):219–227. doi: 10.1016/0022-1759(84)90450-2. [DOI] [PubMed] [Google Scholar]
- Yuan Q. A., Jiang W. M., Krissansen G. W., Watson J. D. Cloning and sequence analysis of a novel beta 2-related integrin transcript from T lymphocytes: homology of integrin cysteine-rich repeats to domain III of laminin B chains. Int Immunol. 1990;2(11):1097–1108. doi: 10.1093/intimm/2.11.1097. [DOI] [PubMed] [Google Scholar]
- Yuan Q., Jiang W. M., Hollander D., Leung E., Watson J. D., Krissansen G. W. Identity between the novel integrin beta 7 subunit and an antigen found highly expressed on intraepithelial lymphocytes in the small intestine. Biochem Biophys Res Commun. 1991 May 15;176(3):1443–1449. doi: 10.1016/0006-291x(91)90448-g. [DOI] [PubMed] [Google Scholar]