Abstract
Aim:
Hyperoside is a flavonol glycoside mainly found in plants of the genera Hypericum and Crataegus, which has shown anti-oxidant, anti-cancer and anti-inflammatory activities. In this study, we investigated the effects of hyperoside on human rheumatoid fibroblast-like synoviocytes (FLSs) in vitro and on mouse collagen-induced arthritis (CIA) in vivo.
Methods:
FLSs were isolated from primary synovial tissues obtained from rheumatoid arthritis (RA) patients and exposed to LPS (1 μg/mL). Cell viability and proliferation were measured with MTT and BrdU assay. Cell migration was assessed using wound-healing assay and Transwell assay. DNA binding of NF-κB was measured using a TransAM-NFkappaB kit. The localization of p65 subunit was detected with immunocytochemistry. CIA was induced in mice by primary immunization with Bovine Type II collagen (CII) emulsified in CFA, followed by a booster injection 3 weeks later. The arthritic mice were treated with hyperoside (25, 50 mg·kg−1·d−1, ip) for 3 weeks, and the joint tissues were harvested for histological analysis.
Results:
Hyperoside (10, 50, 100 μmol/L) dose-dependently inhibited LPS-induced proliferation and migration of human RA FLSs in vitro. Furthermore, hyperoside decreased LPS-stimulated production of TNF-α, IL-6, IL-1 and MMP-9 in the cells. Moreover, hyperoside inhibited LPS-induced phosphorylation of p65 and IκBα, and suppressed LPS-induced nuclear translocation of p65 and DNA biding of NF-κB in the cells. Three-week administration of hyperoside significantly decreased the clinical scores, and alleviated synovial hyperplasia, inflammatory cell infiltration and cartilage damage in mice with CIA.
Conclusion:
Hyperoside inhibits LPS-induced proliferation, migration and inflammatory responses in human RA FLSs in vitro by suppressing activation of the NF-κB signaling pathway, which contributes to the therapeutic effects observed in mice with CIA.
Keywords: hyperoside, rheumatoid arthritis, collagen-induced arthritis, fibroblast-like synoviocytes, LPS, NF-κB, pro-inflammatory cytokines
Introduction
Rheumatoid arthritis (RA) is a chronic autoimmune disease of unknown origin characterized by inflammation of joint tissue and synovial hyperplasia with proliferation of fibroblast-like synoviocytes (FLSs), eventually leading to cartilage and bone destruction1. The pathogenic mechanisms of RA have not been fully elucidated, but activated FLSs are considered to play a pivotal role in the initiation and perpetuation of destructive joint inflammation2,3. It has been reported that stable activated RA FLSs exhibit characteristics of tumor cells such as mutant p53 transcripts4 and play a prominent role in the development of pannus by migration and invasiveness towards cartilage and bone5. One of the major signaling pathways implicated in joint inflammation is the nuclear factor-kappa B (NF-κB) pathway. NF-κB regulates the expression of various proinflammatory mediators, including cytokines, such as tumor necrosis factor (TNF)-α, interleukin (IL)-1β and IL-6, chemokines, and cellular adhesion molecules6,7,8. NF-κB activation in FLSs also contributes to the pathogenesis of RA by activating the transcription of matrix metalloproteinases (MMPs), which are responsible for the invasion properties of FLSs9,10,11.
NF-κB is a family of five proteins and is typically a heterodimer composed of p50 and p65 (RelA) subunits. Its regulation depends on its translocation and subsequent DNA binding. In quiescent cells, NF-κB is retained in the cytoplasm through an interaction with inhibitory IκB proteins. In response to a variety of stimuli, including proinflammatory cytokines such as TNF-α, IL-6 and IL-1β as well as LPS, inhibitory IκB is degraded by the ubiquitin-proteasome pathway, leading to nuclear translocation of NF-κB and transcription of many genes encoding proinflammatory proteins12,13. NF-κB is highly activated in the synovial tissue of RA patients14,15 and in mice with collagen-induced arthritis (CIA)16. Electromobility shift assays demonstrate that NF-κB binding is significantly higher in RA synovium compared with osteoarthritis. Inhibiting phosphorylation of the kinase or blocking translocation of NF-κB has been shown to suppress inflammatory arthritis17. It has been reported that p50-deficient mice do not show joint destruction18. A number of anti-RA drugs currently in clinical use have been shown to suppress NF-κB activities19,20,21. In addition, direct intraarticular transfection of NF-κB decoy oligonucleotides in rats with CIA ameliorated the severity of joint destruction of CIA22. Furthermore, in vitro overexpression of IκBα super-repressor in FLSs from RA patients inhibits IL-1β-induced production of chemokines, activation of MMP-1 and MMP-3, and FLS proliferation23. Taken together, these studies confirm that NF-κB is one of the key regulators promoting joint inflammation and FLS proliferation in arthritis. Development of drugs targeting the NF-κB signaling pathway may be considered a potential therapy for arthritis.
It is well-known that several biological drugs targeting proinflammatory cytokines, such as TNF-α blockers and IL-6 receptor antagonists, have proven effective in patients with RA24,25,26. However, some populations of RA patients remain refractory to various biological agents. In addition, the risk of severe infections is increased by biological drugs27,28. Therefore, the development of novel therapeutic agents is still needed. In recent years, an increasing number of plant-derived herbal products have been considered for the treatment of RA. Hyperoside is a flavonol glycoside mainly found in plants of the genera Hypericum and Crataegus29. Pharmacological investigations have demonstrated that hyperoside has anti-oxidant30, anti-cancer31 and anti-inflammatory activities32,33,34. This compound exerts anti-inflammatory effects by suppressing the production of proinflammatory cytokines through inhibiting the activation of the NF-κB signaling pathway in human endothelial cells33. These observations suggest that hyperoside could be used to treat NF-κB-related inflammatory diseases, including RA. Here, we investigated the effects of hyperoside on cultured human RA FLSs and on mice with CIA. We found that hyperoside was effective at suppressing the production of proinflammatory mediators in vitro. Furthermore, therapeutic administration of hyperoside significantly reduced the severity of CIA in mice.
Materials and methods
Reagents
Hyperoside (MW: 464.38, HPLC ≥98%) was purchased from Nanjing Spring & Autumn Biological Engineering Co, Ltd (Nanjing, China). It was dissolved in aqueous DMSO and delivered to cells in media containing the solvent at a final concentration of 0.1% (v/v). The 5-bromo-2-deoxyuridine (BrdU) Cell Proliferation Assay Kit was purchased Merck Millipore (Darmstadt, Germany). Collagenase and LPS were purchased from Sigma (St Louis, MO, USA).
Isolation and culture of RA FLS from patients
Human synovial tissue was obtained with informed consent from all RA patients, and the study protocol was approved by the Ethics Committee of Liaoning Medical University Hospital. RA FLSs were isolated from primary synovial tissue obtained from RA patients who met the 1987 American College of Rheumatology (ACR) criteria35 for the diagnosis of RA and had undergone synovectomy, as previously described36. Synovial tissues were cut into small pieces and digested with collagenase in Dulbecco's modified Eagle's medium/nutrient mixture F-12 (DMEM/F12) for 2 h at 37 °C. Cells were grown at 37 °C in a humidified atmosphere containing 5% CO2/95% O2 in high glucose-containing DMEM/F12 (GIBCO; Grand Island, NY, USA) supplemented with 10% fetal bovine serum (FBS), 2 mmol/L glutamine, 100 units/mL penicillin, and 100 μg/mL streptomycin. Cells were used at passages four to nine, at which time they consisted of a homogeneous population. All treatments were performed in a serum-free medium.
Cell viability assay and LPS-induced FLS proliferation
Cell viability was measured by the 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) method. Briefly, FLSs were seeded in a 96-well plate at a density of 1×104 cells/well and allowed to adhere for 24 h. Cells were deprived of serum for 12 h, treated with hyperoside (1, 10, and 100 μmol/L) for an additional 48 h, and then incubated in 0.5 mg/mL MTT solution. After 4 h of incubation, the dark blue formazan crystals that had formed in intact cells were dissolved in DMSO, and the absorbance at 570 nm was read using a microplate reader (Thermo; Waltham, MA, USA).
The BrdU assay was used to examine the effect of hyperoside on the proliferation of FLSs. A cell proliferation ELISA was used to measure the incorporation of BrdU during DNA synthesis, following the manufacturer's protocols. Briefly, the cells were pretreated with hyperoside (1, 10, 50, and 100 μmol/L) for 1 h and then incubated with or without LPS (1 μg/mL) in the medium for another 48 h. Eighteen hours before termination of the experiments, BrdU (10 μmol/L) was added to the culture medium for 18 h. Then, the BrdU-labeled cells were fixed, and the DNA was denatured in fixative solution for 30 min at room temperature. The cells were then incubated with peroxidase-conjugated anti-BrdU antibody for 1 h at room temperature, followed by washing three times with washing solution. The immune complex was detected using a 3,3′,5,5′-tetramethylbenzidine substrate reaction, and absorbance was measured at 450 nm.
Enzyme-linked immunosorbent assay (ELISA)
RA FLSs were cultured in 96-well plates at a density of 2×105 cells/mL for 24 h and then pretreated with different concentrations of hyperoside (10, 50, and 100 μmol/L) for 4 h; LPS (1 μg/mL) was then added before incubation for another 48 h. The culture media were collected at the end of the culture period and centrifuged at 10 000×g for 5 min to remove the particulate matter. The levels of TNF-α, IL-6, IL-1β, and MMP-9 in the culture supernatants were assessed using ELISA kits (R&D Systems; Minneapolis, MN, USA) according to the manufacturer's instructions. Optical density was determined by a microplate reader (Model ELx800, BioTek). The standard curves of TNF-α, IL-6, IL-1β, and MMP-9 were established using known concentrations of cytokines by plotting optical density vs log of the concentration.
Migration assay using wound-healing assay chambers
Wound-healing assays were performed using specific wound assay chambers (ibidi; Munich, Germany). Seventy microliters of cell suspension (5×105 cells/mL) was seeded into each well of the culture insert. After appropriate cell attachment for 24 h, the culture inserts were removed and the cells were washed twice with PBS. Then, the cells were pretreated with hyperoside (50 μmol/L) for 1 h, followed by incubation with LPS (1 μg/mL) in FBS-free DMEM/F12 for 24 h. The cell migration into the defined cell free gap (500 μm) was observed for 12 h and 24 h under a phase-contrast inverted microscope (Olympus). Images were captured with a TUCSEN camera. A cell-free area was measured using Wimasis Image Analysis (ibidi) at 0, 12, and 24 h, and the ratio was then calculated (cell-free area at 12 or 24 h per cell-free at 0 h).
Migration assay using Transwell chambers
For migration assays, Transwell chambers (6.5 mm, 8 μm pore size; Corning, NY, USA) combined with 24-well culture companion plates were used. Before migration, the cells were serum-starved overnight in DMEM/F12 containing 1% FBS. Hyperoside pretreated cells were trypsinized for 2 h and seeded onto filter membrane of the insert (5×105 cells/mL) with a serum-free medium containing hyperoside. The bottom compartment of the chambers was filled either with 0.6 mL DMEM/F12 containing 0.5% FBS (unstimulated control) or 0.6 mL DMEM/F12 containing 10% FBS. Cells were allowed to migrate through the filter membrane for 24 h at 37 °C in a CO2 incubator. Following incubation, the non-migrating cells were removed from the upper surface with cotton swabs, and the migrated cells, now at the bottom surface of the filter membrane, were stained with 0.1% crystal violet for 30 min at 37 °C. Chemotaxis was quantified by counting the stained cells using an optical microscope (magnification 100). The stained cells were counted as the mean number of cells per nine random fields for each assay. All treatments were performed in triplicate.
Measurement of NF-κB DNA binding activity
DNA binding activity of NF-κB was measured with a sensitive multiwell colorimetric assay37 using a TransAM-NFkappaB kit (Active Motif; Carlsbad, CA, USA). Briefly, nuclear extracts (10 μg) from each sample were incubated in 96-well plates coated with NF-κB consensus double-stranded oligonucleotide sequence (5′-AGTTGAGGGGACTTTCCCAGGC-3′) for 1 h and then with primary NF-κB antibody (1:1000) for 1 h, and subsequently with peroxidase-conjugated secondary antibody (1:1000) for 1 h at room temperature. After colorimetric reaction, optical density was read at 450 nm with a microplate reader (ELx800, BioTek). The results are expressed after subtraction of the blank values. For competition assays, the nuclear extracts were incubated with 22-bp double-stranded DNA, either wild-type or mutated (5′-AGTTGAGCTCACTTTCCC AGGC-3′ [underline denotes the substitution]).
Western blot analysis
Human RA FLSs were seeded in 6-well plates at a density of 5×106 cells/well and pretreated with hyperoside (10, 50, and 100 μmol/L) for 8 h before being exposed to LPS (1 μg/mL) for 30 min or 48 h. Whole-cell lysates or cytoplasmic and nuclear protein extracts were prepared from RA FLSs using a radioimmune precipitation assay lysis buffer or cytoplasmic and nuclear protein extraction reagents. Equivalent amounts of protein (40 μg) from each sample were mixed with gel loading buffer in a ratio of 1:1, heated at 95 °C for 5 min, electrophoresed in a 10%–12% SDS-PAGE and transferred onto a nitrocellulose membrane (Millipore; Bedford, MA, USA). The nitrocellulose membranes were blocked with 5% BSA. The blots were probed with antibodies against the NF-κB p65, phospho-NF-κB p65 (Ser536), IκBα, phospho-IκBα (Ser32), and β-actin (all from Cell Signaling; Beverly, MA, USA). HRP-conjugated anti-IgG (Cell Signaling; Beverly, MA, USA) was used as a secondary antibody. Membranes were visualized with a chemiluminescence system (Pierce; Rockford, IL, USA). Staining intensity was quantified from 3 blots derived from 3 independent experimental trials. The density of each band was quantified using Image J software and normalized β-actin expression. The protein levels were expressed as the ratio of the band intensity for the protein of interest to that for β-actin and were used as loading controls.
Immunocytochemistry for NF-κB p65 localization
Human RA FLSs cultured on coverslips were pretreated with hyperoside (50 μmol/L) for 4 h before incubation with LPS (1 μg/mL) for 30 min. The cells were then washed and fixed with a 4% paraformaldehyde for 30 min at 37 °C, permeabilized with 0.3% Triton X-100 for 10 min, and blocked with 5% normal goat serum for 1 h at room temperature. Next, cells were incubated with rabbit monoclonal anti-NF-κB p65 antibody (1:100, Cell Signaling Technology; Beverly, MA, USA) overnight at 4 °C. After extensive washing with PBS, the slides were incubated with anti-rabbit secondary antibody conjugated with rhodamine (TRITC, 1:1000; The Jackson Labs, West Grove, PA, USA) at room temperature for 2 h and counterstained for nuclei with Hoechst 33258 (50 ng/mL) for 5 min.
Animals
Pathogen-free male DBA/1 mice were purchased from Beijing HFK Bioscience Co, Ltd (Beijing, China). Mice were housed three to four per cage, under specific pathogen-free conditions, with a 12-h light/dark cycle and fed standard rodent chow and water ad libitum. The experimental protocol was approved by the Experimental Animal Care and Use Committee of Liaoning Medical University.
Induction and assessment of CIA
CIA was induced as previously described16. Bovine Type II collagen (CII; Sigma; St Louis, MO, USA) was dissolved to a concentration of 2 mg/mL in 0.01 mol/L acetic acid at 4 °C, with constant overnight mixing. CII was emulsified with an equal volume of Complete Freund′s adjuvant (CFA, Sigma; St Louis, MO, USA). Mice were immunized intradermally at the base of the tail with 150 μL emulsion. The day of the first immunization was defined as day 0. The mice were then given a booster injection of an equal amount of CII emulsified in CFA on day 21. CII solution and emulsion with CFA was always freshly prepared. The control mice were injected in the same way, whether with saline or CFA. Beginning 4 weeks after the primary immunization, the mice were examined 2–3 times per week for onset and severity of arthritis in their four paws using a blind procedure. To examine the therapeutic effect of hyperoside, arthritic mice (with an arthritis score of approximately 2 on day 31) were randomly divided into three groups (n=8 mice per group), including the vehicle group and hyperoside-treated group (25 mg/kg and 50 mg/kg, respectively). Hyperoside or vehicle solution was intraperitoneally administered to each group every day until day 52, after primary immunization. Clinical arthritis scores were evaluated using a scale of 0–4 for each limb: 0, normal; 1, erythema and mild swelling confined to the ankle joint and toes; 2, erythema and mild swelling extending from the ankle to the midfoot; 3, erythema and severe swelling extending from the ankle to the metatarsal joints; 4, severe swelling, erythema, and joint rigidity of the ankle, foot and digits. The clinical scores for each mouse are the sum of the scores for four limbs, and the maximal score for each mouse is sixteen. On day 52, the mice were killed with an overdose of anesthetic, and joint tissues were harvested from each animal for histologic analysis.
Histopathological assessment
The hind paws and knee joints were fixed in 4% buffered formaldehyde, decalcified for 3 weeks in 12% disodium EDTA, dehydrated, and embedded in paraffin. Sections (5 μm) were stained with hematoxylin and eosin or Safranin O for light macroscopic examination. Histopathological assessment of the joints of the CIA mice was carried out as previously described38.
Statistical analysis
The data are expressed as the mean±SEM and were analyzed by one-way ANOVA followed by an LSD post hoc multiple-comparison test or by Student's t-test for two-group comparisons. A P-value of less than 0.05 was considered significant.
Results
Effect of hyperoside on LPS-induced cell proliferation in human RA FLSs
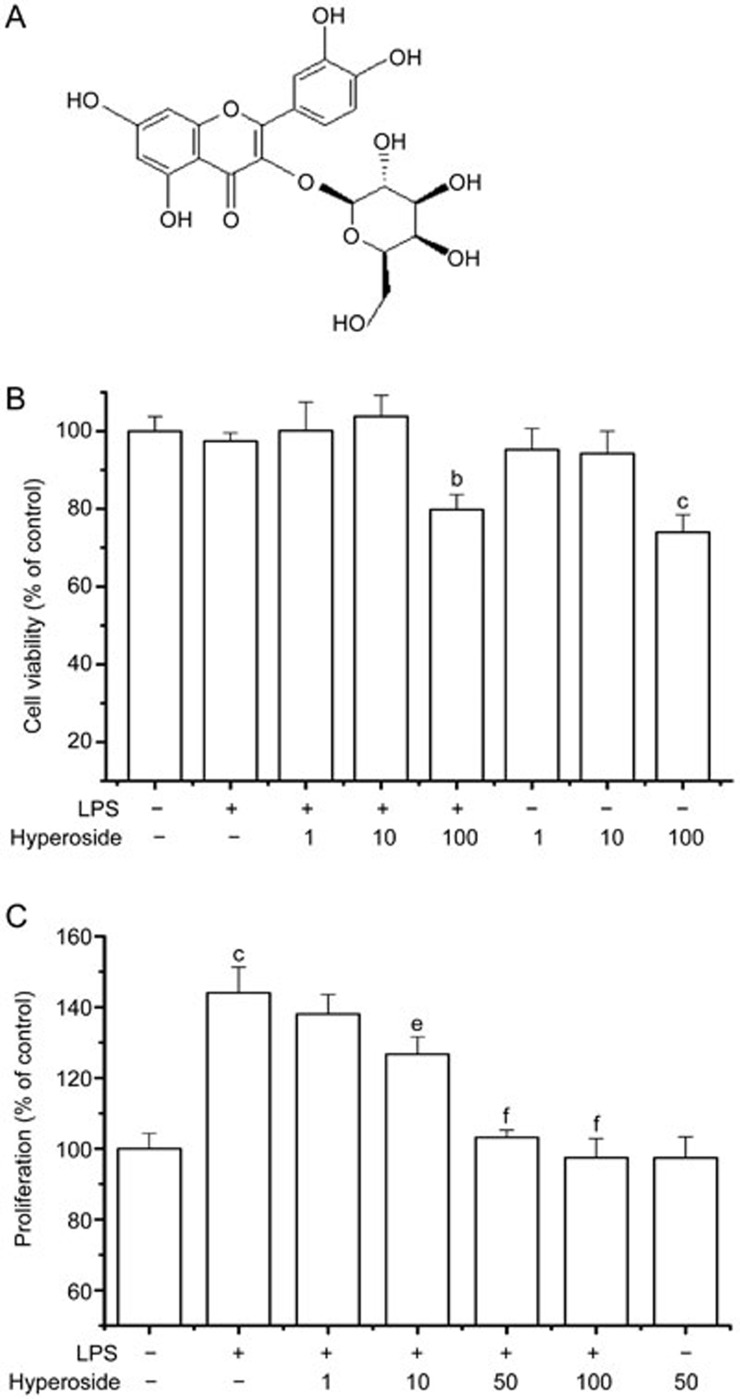
The chemical structure of hyperoside is presented in Figure 1A. The cytotoxic effect of hyperoside on human RA FLSs was examined (Figure 1B). RA FLSs were treated with hyperoside (1, 10, and 100 μmol/L) in an FCS-free medium for 48 h, and cell viability was determined using the MTT assay. Treatment with 1 and 10 μmol/L hyperoside had no significant effect on cell viability for 48 h. However, 100 μmol/L hyperoside decreased cell viability by 27%. Based on data from preliminary studies, three different concentrations of hyperoside (10, 50, and 100 μmol/L) were chosen for the following experiments.
Figure 1.
Effects of hyperoside on LPS-induced RA FLSs proliferation. (A) Chemical structure of hyperoside. (B) FLSs were incubated with the indicated concentrations of hyperoside in a serum-free medium for 48 h, and cell viability was measured by the MTT assay. (C) RA FLSs were pretreated with hyperoside (1, 10, 50, and 100 μmol/L) for 1 h and exposed to LPS (1 μg/mL) for 48 h. Cellular proliferation was measured using the BrdU incorporation assay. Each value represents the mean±SEM of the three independent experiments. *P<0.05, **P<0.01 compared with the control group. #P<0.05, ##P<0.01 compared with the LPS-treated group.
Synovial hyperplasia, also called pannus, in RA is primarily associated with uncontrolled FLS proliferation39. RA FLSs produce a variety of proinflammatory cytokines, chemokines, and matrix-degrading enzymes and are responsible for the destruction of articular cartilage and bone1. LPS is well-known as a potent growth-promoting agent in various cell types40. The effect of hyperoside on proliferation of RA FLSs was evaluated using the BrdU incorporation assay in the presence or absence of LPS (Figure 1C). Treatment with LPS (1 μg/mL) alone for 48 h significantly increased the RA FLS proliferative potential. Pretreatment of hyperoside (1, 10, 50, and 100 μmol/L) significantly inhibited this increased proliferation of LPS-treated RA FLSs in a concentration-dependent manner. Incubation with hyperoside (50 μmol/L) in the absence of LPS did not affect RA FLS proliferation.
Effect of hyperoside on human RA FLS migration abilities
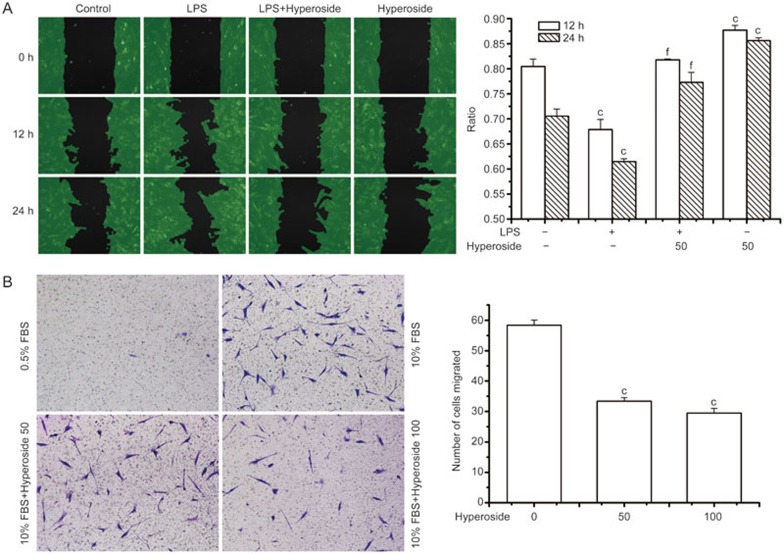
In the RA synovium, the migration of RA FLSs into the cartilage and bone is considered important for pannus development3. Therefore, by using an in vitro wound-healing model, we investigated whether hyperoside could alter RA FLS motility. As shown in Figure 2A, incubation with LPS significantly decreased the cell-free area in 12 or 24 h, indicating that LPS induced RA FLS migration. Moreover, pretreatment with hyperoside significantly suppressed LPS-induced motility in RA FLSs. To further confirm the role of hyperoside on FLS migration, we also used the Transwell chamber assay to assess the cell migration. As expected, RA FLSs treated with either the vehicle or hyperoside did not migrate in the presence of 0.5% FBS. The presence of 10% FBS triggered a chemotactic response demonstrated by the marked (∼50-fold) increase in RA FLSs migration following treatment with the vehicle; by contrast, hyperoside treatment significantly attenuated RA FLSs migration (Figure 2B). It is important to note that these effects were independent of cell proliferation, as RA FLSs require ∼36 h for one replication.
Figure 2.
Effect of hyperoside on RA FLSs migration. (A) Migration analysis using wound assay chambers. RA FLSs were seeded into wound assay culture inserts (ibidi) and incubated for 24 h. Removal of the insert yielded a uniform “wound” to the monolayer. After preincubation with hyperoside (50 μmol/L) for 1 h, cells were stimulated with LPS (1 μg/mL) for 24 h. The cell-free areas were measured using Wimasis Image Analysis (ibidi) and the ratio was calculated (cell-free area in 12 h or 24 h to cell-free area in 0 h). (B) Migration was performed in a Transwell chamber and chemotaxis was quantified by counting the migrated cells. The RA FLSs were seeded in a Transwell chamber and allowed to migrate for a further 24 h. FBS (10%) was used as a chemoattractant. Data represent the mean±SEM of five independent experiments. **P<0.01 compared with the control group. ##P<0.01 compared with the LPS-treated group.
Effect of hyperoside on LPS-induced production of pro-inflammatory mediators in human RA FLSs
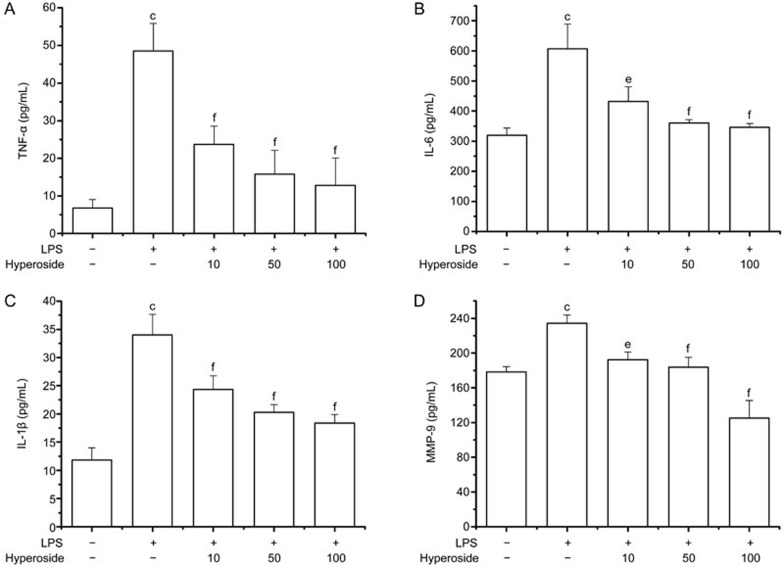
It is well-known that pro-inflammatory cytokines play key roles in the pathogenesis of RA. To examine the anti-inflammatory effect of hyperoside, RA FLSs were incubated with different concentrations of hyperoside (10, 50, and 100 μmol/L) in culture media, followed by stimulation with 1 μg/mL LPS for 48 h. The addition of LPS significantly increased the production of TNFα, IL-6, and IL-1β compared with that of the control. However, pretreatment with hyperoside for 4 h greatly inhibited the production of TNFα (Figure 3A), IL-6 (Figure 3B), and IL-1β (Figure 3C) in a concentration-dependent manner.
Figure 3.
Inhibitory effect of hyperoside on LPS-induced cytokines and MMP-9 production in RA FLSs. FLSs from RA patients were pretreated with different concentrations of hyperoside for 4 h and exposed to LPS (1 μg/mL) for 48 h. The presence of TNF-α (A), IL-6 (B), IL-1β (C), and MMP-9 (D) in the cell-free culture supernatants was then evaluated by ELISA. Values are the mean±SEM of five independent experiments. **P<0.01 compared with the untreated control; #P<0.05, ##P<0.01 compared with the LPS-treated group.
RA FLSs have the ability to secrete zinc-dependent proteases such as MMPs, which promote degradation of the extracellular matrix proteins in joints and bones41. MMP-9, also called collagenases, are induced in RA FLSs by pro-inflammatory cytokines and LPS3,42. A recent report suggests that MMP-9 plays an important role in RA FLS migration and invasion induced by LPS42. Because hyperoside inhibited RA FLS migration, we examined the effect of hyperoside on MMP-9 secretion in cultured media. As shown in Figure 3D, the secretion of MMP-9 in RA FLSs was induced when cultured in a serum-free medium with LPS (1 μg/mL) for 48 h. The treatment of RA FLSs with hyperoside suppressed LPS-induced MMP-9 secretion in a dose-dependent manner.
Effects of hyperoside on LPS-induced NF-κB signal activation in human RA FLSs
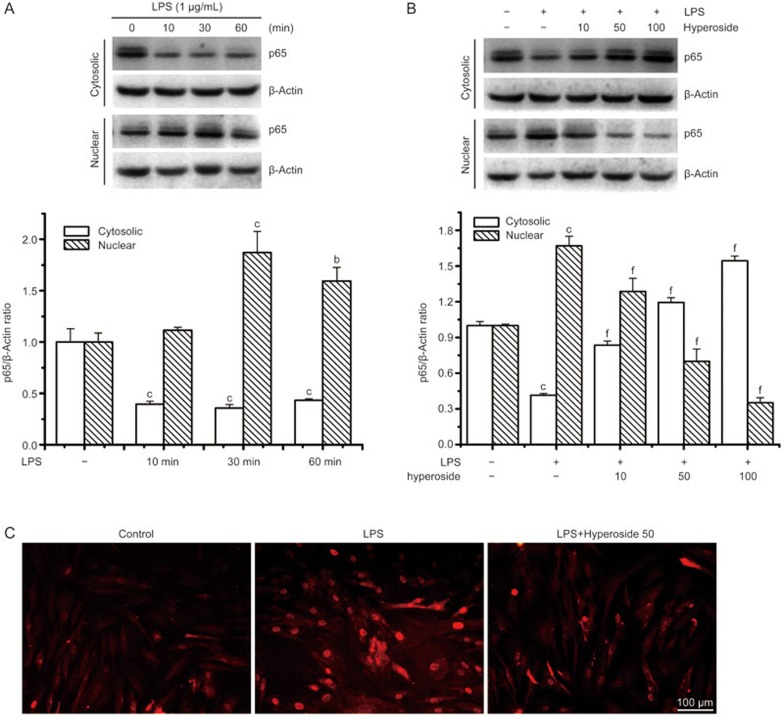
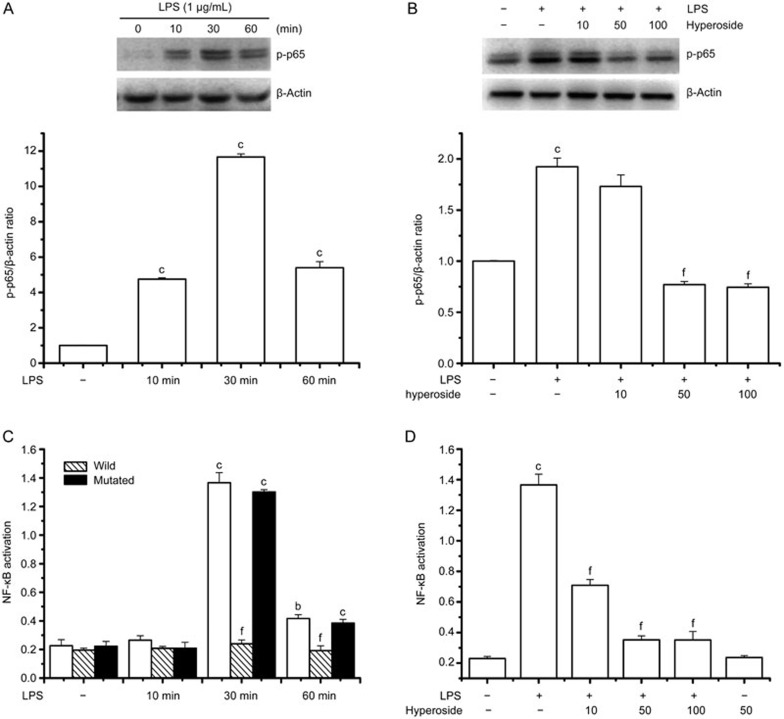
Next, we studied the role of hyperoside in the activation of NF-κB, a critical regulator of inflammatory cytokine gene transcription. LPS has been shown to induce the translocation of the cytosolic p65 subunit into the nucleus after phosphorylation. Therefore, we first tested the effect of hyperoside on LPS-induced nuclear translocation of p65 by an immunofluorescene assay and Western blot analyses. Western blot analyses using cytosolic and nuclear fractions showed that the protein level of p65 in the nucleus was markedly increased after 30 min of exposure to LPS, concomitant with a decrease of p65 in the cytoplasm (Figure 4A), compared with a similar fraction from unstimulated cells. Nuclear translocation of p65 was markedly suppressed by hyperoside in a concentration-dependent manner (Figure 4B). Furthermore, the shift of NF-κB to the nucleus in RA FLSs was analyzed using immunofluorescence staining. The immunofluorescence images revealed that basal p65 was distributed in the cytoplasm and that nuclear translocation of p65 was increased in LPS-stimulated RA FLSs. sBy contrast, the LPS-induced translocation of p65 was significantly inhibited in hyperoside-treated RA FLS (Figure 4C).
Figure 4.
Effect of hyperoside on LPS-induced nuclear translocation of NF-κB in human RA FLSs. RA FLSs were incubated with LPS (1 μg/mL) for the indicated time with or without the indicated concentration of hyperoside. In coincubation experiments, hyperoside was added to cells 4 h prior to LPS and then treated with LPS for 30 min. (A and B) Representative immunoblot comparing NF-κB subunit p65 to β-actin in RA FLSs after the indicated treatment. Group data showing the normalization of p65 to β-actin as determined in each group from the 3 experiments. (C) Representative immunofluorescence photographs show the location of p65 in RA FLSs. Values are the mean±SEM of three independent experiments. **P<0.01 compared with the untreated control. ##P<0.01 compared with the LPS.
LPS has been shown to induce the phosphorylation of p65, which is required for translocation of p65 to the nucleus. Therefore, we also tested the effect of hyperoside on LPS-induced phosphorylation of p65. As shown in Figure 5A and B, LPS induced the phosphorylation of p65 in a time-dependent manner (Figure 5A), and hyperoside pretreatment significantly suppressed LPS-induced phosphorylation of p65 in a concentration-dependent manner (Figure 5B).
Figure 5.
Effect of hyperoside on LPS-induced phosphorylation of p65 and DNA binding activity of NF-κB. RA FLSs were incubated with LPS (1 μg/mL) for the indicated time with or without the indicated concentration of hyperoside. In coincubation experiments, hyperoside was added to cells 4 h prior to LPS and then treated with LPS for 30 min. The nuclear extracts of these cells were prepared for Western blotting and measurement of NF-κB DNA binding activity. (A and B) Representative immunoblot comparing phosphorylated p65 to β-actin in RA FLSs after the indicated treatment. Group data showing the normalization of phosphorylated p65 to β-actin as determined in each group from the 3 independent experiments. (C and D) Nuclear extracts prepared from cells exposed to LPS for 30 min exhibited increased NF-κB DNA binding activity, which was significantly prevented by hyperoside. The NF-κB DNA binding capacity was effectively completed by the wild-type consensus oligonucleotide, but not mutated oligonucleotide. Values are the mean±SEM of the three independent experiments. *P<0.05, **P<0.01 compared with the untreated control. ##P<0.01 compared with the LPS-treated group.
To further investigate the role of hyperoside on the NF-κB signaling pathway, NF-κB DNA binding activity was also measured with a sensitive colorimetric assay using a specific oligonucleotide probe for NF-κB. Binding activity of the p65 subunit to an NF-κB consensus sequence was increased in LPS-stimulated RA FLS and this was time dependent, with DNA binding activity peaking at 30 min (Figure 5C). By contrast, nuclear extracts prepared from hyperoside-treated RA FLSs revealed markedly suppressed DNA binding of NF-κB in a concentration-dependent manner (Figure 5D).
Effects of hyperoside on LPS-induced phosphorylation and degradation of IκBα
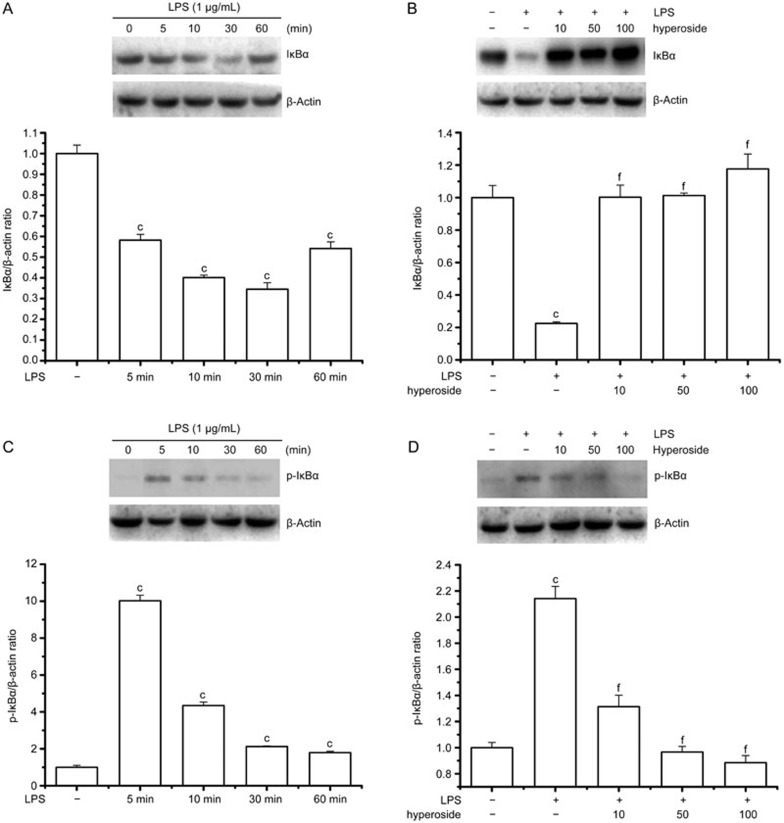
The translocation of NF-κB to the nucleus is preceded by the phosphorylation, ubiquitination, and proteolytic degradation of IκBα43. To determine whether the inhibitory action of hyperoside on LPS-induced NF-κB activation was due to an effect on IκBα degradation, the cytoplasmic IκBα protein levels were examined by Western blot analysis. Time course analysis revealed that IκBα was maximally degraded 30 min after LPS treatment of RA FLS and resynthesis began thereafter (Figure 6A). The pretreatment of cells with hyperoside completely abolished the LPS-induced degradation of IκBα (Figure 6B).
Figure 6.
Effect of hyperoside on LPS-induced phosphorylation and degradation of IκBα. RA FLSs (2×106/mL) were incubated with LPS (1 g/mL) for the indicated time with or without the indicated concentration of hyperoside. In coincubation experiments, hyperoside was added to cells 4 h prior to LPS and then treated with LPS for 30 min (IκBα) or 15 min (phospho-IκBα). The cytosolic extracts of these cells were prepared for Western blotting. (A and B) Representative immunoblot comparing IκBα to β-actin in RA FLSs after the indicated treatment. Group data showing the normalization of IκBα to β-actin as determined in each group from the 3 independent experiments. (C and D) Representative immunoblot comparing phosphorylated IκBα to β-actin in RA FLSs after the indicated treatment. Group data showing the normalization of phosphorylated IκBα to β-actin as determined in each group from the 3 independent experiments. **P<0.01 compared with the untreated control. ##P<0.01 compared with the LPS-treated group.
To determine whether inhibition of LPS-induced IκBα degradation by hyperoside was due to suppression of IκBα phosphorylation, we examined the phosphorylated form of IκBα using an antibody that recognizes only the serine-phosphorylated form of IκBα. As shown in Figure 6C, LPS induced IκBα phosphorylation, and the activation could be seen as early as 5 min after LPS treatment. Pretreatment of hyperoside markedly suppressed the LPS-induced IκBα phosphorylation in a concentration-dependent manner (Figure 6D).
Suppression of synovial inflammation and bone destruction in mice with CIA by treatment with hyperoside
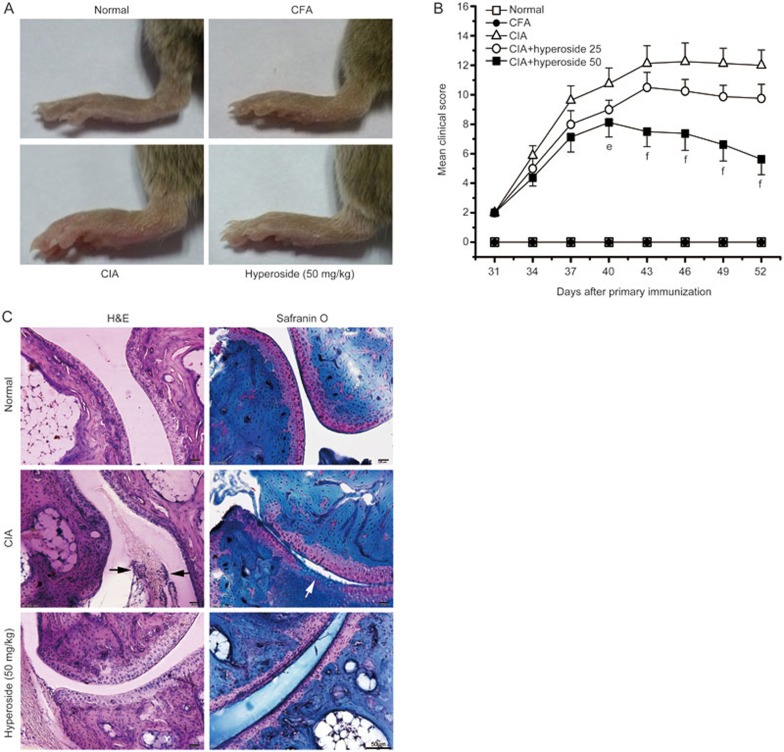
Next, we assessed the anti-arthritic role of hyperoside in mice with CIA. To test the possible therapeutic effect of hyperoside on the CIA animal model, we included only those mice that had apparently begun to develop arthritis, and injected hyperoside (25 mg/kg and 50 mg/kg) every day until day 52, after primary immunization. The clinical score was used to evaluate the progression of arthritis development. Vehicle-treated mice developed severe swelling, erythema, and joint rigidity of the hind paws. By contrast, in mice treated with hyperoside, the severity of CIA was significantly attenuated according to mean arthritis score (Figure 7A and 7B). In addition, CFA-treated (as a negative control) mice did not develop any clinical signs of arthritis (Figure 7A and 7B).
Figure 7.
Therapeutic effect of hyperoside treatment on established arthritis in mice with collagen-induced arthritis (CIA). Following the development of clinical arthritis (average clinical score of 2) on day 31, mice with CIA were randomized to receive injection with either the vehicle or hyperoside (25 mg/kg and 50 mg/kg, respectively). (A) Clinical evidence of arthritis with redness and swelling of the paws involving the tarsus, ankle and toes in the three treated groups. Pictures were taken at day 52. (B) Clinical scores were measured on the indicated days after the primary immunization. (C) Histopathological changes in hyperoside-treated mice with CIA. Hind paws from mice with CIA were obtained on day 52 and were then sectioned and stained with hematoxylin and eosin (H&E) or Safranio O. Photomicrographs are representative of the 3 independent experiments (Original magnification 200 for H&E- and Safranin O–stained sections). Black arrows indicate synovium inflammation. White arrows indicate cartilage damage. Data represent the mean± SEM(n=8 mice per group). #P<0.05, ##P<0.01 compared with the vehicle-treated CIA mice.
Histopathological assessment showed synovial proliferation, cartilage damage, and inflammatory cell infiltration in the ankle joints of vehicle-treated mice. By contrast, the destruction of cartilage and inflammation was remarkably alleviated by hyperoside treatment compared with vehicle-treated mice (Figure 7C).
Discussion
In the present study, we showed that hyperoside effectively suppressed the inflammatory response and cartilage damage in both in vitro and in vivo RA models. We found that hyperoside inhibited LPS-induced cell proliferation and migration, cytokine production, and MMP-9 secretion in cultured human RA FLSs. Subsequently, hyperoside alleviated the clinical scores, synovial hyperplasia, and inflammatory cell infiltration in CIA mice, which is the prototype of an animal model of RA. These beneficial effects of hyperoside correlated with decreased activity of the NF-κB signal.
In RA, FLSs and various inflammatory cells can produce pro-inflammatory cytokines, such as TNFα, IL-1β, and IL-644. Increasing levels of cytokines in the synovium regulate growth factor expression and lead to activation of FLSs, which undergo hyperplasia, a hallmark event in RA. Activated FLSs can undergo migration and induce and/or enhance the production of MMPs that in turn mediate tissue destruction45,46. As a first step towards application of hyperoside, we tested the effects of hyperoside on proliferation and migration of RA FLSs. The results demonstrated that hyperoside significantly suppressed LPS-induced FLS proliferation and migration. In this study, we also confirmed that hyperoside reduced the LPS-induced increase in production of TNFα, IL-1β, and IL-6 in cultured human RA FLSs.
Over 20 members of the MMP family have been identified in humans to date. Elevated gene expression of MMPs is critical for the progression of RA47,48. LPS can stimulate FLS to secrete MMPs, and this induction is regulated at the transcriptional and translational levels49. MMP-9 is one of two major gelatinases in the MMP family, which, in addition to efficiently and rapidly cleaving unfolded collagen (gelatin), has been reported to cleave other matrix and non-matrix components50. MMP-9 expression is activated by proinflammatory cytokines51 and has been shown to decrease in response to anti-TNF therapy52. In the present study, LPS treatment of human RA FLSs caused an increase in MMP-9 levels. The increase in MMP-9 secretion was inhibited by hyperoside in cultured RA FLSs. Therefore, it is likely that hyperoside blocks cartilage damage mediated by MMPs in the inflamed joints of RA.
It has been reported that the translocation and subsequent DNA binding of NF-κB in inflamed synovium transactivates the gene expression of inflammatory cytokines and MMPs, including TNFα, IL-1β and IL-6, and MMP-942,53. Therefore, NF-κB has been extensively studied to determine its role in the pathogenesis of RA, and NF-κB inhibition has been explored as a therapeutic approach to the disease. A recent report showed that hyperoside inhibited the NF-κB pathway in lipopolysaccharide-stimulated mouse peritoneal macrophages32. Thus, we postulated that hyperoside might suppress NF-κB activation in RA FLSs. Our results indeed demonstrate that hyperoside can suppress NF-κB activation induced by LPS in cultured human RA FLSs. In response to LPS stimulation, NF-κB is translocated into a nucleus through IκB degradation by activating the TLR4-mediated MyD88-dependent signaling pathway54. In the present study, we showed that hyperoside significantly attenuated LPS-induced IκB degradation and nuclear translocation of p65 in LPS-stimulated RA FLSs, as indicated by the results of western blotting and immunocytochemistry for NF-κB p65. Furthermore, hyperoside significantly decreased the NF-κB DNA binding activities in LPS-stimulating RA FLSs. These findings suggest that hyperoside may indicate an anti-inflammatory activity by inhibiting NF-κB activation.
CIA is a common model of RA and has been used in numerous studies to examine the pathogenesis of arthritis and identify potential therapeutic targets. To confirm our results from a local to a systemic level, we treated CIA DBA/1 mice with hyperoside (25 mg/kg and 50 mg/kg). The treatment of arthritic mice with hyperoside significantly reduced the clinical scores and alleviated synovial hyperplasia and inflammatory cell infiltration, as demonstrated by clinical evaluation and histomorphometry.
The most important goal in RA therapy is the prevention of bone destruction to maintain normal joint function. Some studies have indicated that bone-resorbing osteoclasts in the synovium play an important role in bone and cartilage destruction in RA55,56. IL-6 is a pleiotropic cytokine with multiple roles in the regulation of inflammation and hematopoiesis. IL-6 derived from activated synovial macrophages and FLS stimulates local osteoclasts to resorb the bone matrices in the affected joints56. Consistent with these findings, this study demonstrated that hyperoside inhibited an LPS-induced increase in production of IL-6 RA FLSs. Given the essential effect of NF-κB for the expression of genes required for osteoclastogenesis, it is possible that hyperoside minimizes joint destruction by inhibiting the NF-κB pathway and decreasing the number and activity of osteoclasts. Although we argue that NF-κB inhibition is the principal therapeutic effect of hyperoside on RA, we do not exclude the possibility of unidentified effects of hyperoside in other cell types.
In summary, the present study provides evidence to support the notion that hyperoside, by acting as an anti-inflammatory and suppressing joint destruction, has a potent therapeutic effect on the development and progression of CIA in mice. This therapeutic effect of hyperoside may be related to the decreased production of pro-inflammatory cytokines and suppression of the NF-κB signaling pathway. Hyperoside has the potential to be developed into a novel therapeutic agent for the treatment of human RA.
Author contribution
Ying JIN and Wei GAO designed the research; Xiang-nan JIN, En-zhi YAN, Han-ming WANG, Hai-juan SUI, and Zhou LIU performed the research; Xiang-nan JIN analyzed the data, and Ying JIN wrote the paper.
Acknowledgments
We thank Prof Dan-ping LIU (Liaoning Medical University, Jinzhou, China) for providing FLSs of RA patients. This study was supported by grants from the National Natural Science Foundation (No 81400875) and the Natural Science Foundation of Liaoning Province (No 2014022017).
References
- Huber LC, Distler O, Tarner I, Gay RE, Gay S, Pap T. Synovial fibroblasts: key players in rheumatoid arthritis. Rheumatology (Oxford) 2006; 45: 669–75. [DOI] [PubMed] [Google Scholar]
- Bottini N, Firestein GS. Duality of fibroblast-like synoviocytes in RA: passive responders and imprinted aggressors. Nat Rev Rheumatol 2013; 9: 24–33. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bartok B, Firestein GS. Fibroblast-like synoviocytes: key effector cells in rheumatoid arthritis. Immunol Rev 2010; 233: 233–55. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Firestein GS, Echeverri F, Yeo M, Zvaifler NJ, Green DR. Somatic mutations in the p53 tumor suppressor gene in rheumatoid arthritis synovium. Proc Natl Acad Sci U S A 1997; 94: 10895–900. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Muller-Ladner U, Pap T, Gay RE, Neidhart M, Gay S. Mechanisms of disease: the molecular and cellular basis of joint destruction in rheumatoid arthritis. Nat Clin Pract Rheumatol 2005; 1: 102–10. [DOI] [PubMed] [Google Scholar]
- McInnes IB, Schett G. Cytokines in the pathogenesis of rheumatoid arthritis. Nat Rev Immunol 2007; 7: 429–42. [DOI] [PubMed] [Google Scholar]
- Koch AE. Chemokines and their receptors in rheumatoid arthritis: future targets? Arthritis Rheum 2005; 52: 710–21. [DOI] [PubMed] [Google Scholar]
- Gerlag DM, Ransone L, Tak PP, Han Z, Palanki M, Barbosa MS, et al. The effect of a T cell-specific NF-kappa B inhibitor on in vitro cytokine production and collagen-induced arthritis. J Immunol 2000; 165: 1652–8. [DOI] [PubMed] [Google Scholar]
- Murphy G, Nagase H. Progress in matrix metalloproteinase research. Mol Aspects Med 2008; 29: 290–308. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vincenti MP, Coon CI, Brinckerhoff CE. Nuclear factor kappaB/p50 activates an element in the distal matrix metalloproteinase 1 promoter in interleukin-1beta-stimulated synovial fibroblasts. Arthritis Rheum 1998; 41: 1987–94. [DOI] [PubMed] [Google Scholar]
- Barchowsky A, Frleta D, Vincenti MP. Integration of the NF-kappaB and mitogen-activated protein kinase/AP-1 pathways at the collagenase-1 promoter: divergence of IL-1 and TNF-dependent signal transduction in rabbit primary synovial fibroblasts. Cytokine 2000; 12: 1469–79. [DOI] [PubMed] [Google Scholar]
- Palombella VJ, Conner EM, Fuseler JW, Destree A, Davis JM, Laroux FS, et al. Role of the proteasome and NF-kappaB in streptococcal cell wall-induced polyarthritis. Proc Natl Acad Sci U S A 1998; 95: 15671–6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- di Meglio P, Ianaro A, Ghosh S. Amelioration of acute inflammation by systemic administration of a cell-permeable peptide inhibitor of NF-kappaB activation. Arthritis Rheum 2005; 52: 951–8. [DOI] [PubMed] [Google Scholar]
- Handel ML, McMorrow LB, Gravallese EM. Nuclear factor-kappa B in rheumatoid synovium. Localization of p50 and p65. Arthritis Rheum 1995; 38: 1762–70. [DOI] [PubMed] [Google Scholar]
- Marok R, Winyard PG, Coumbe A, Kus ML, Gaffney K, Blades S, et al. Activation of the transcription factor nuclear factor-kappaB in human inflamed synovial tissue. Arthritis Rheum 1996; 39: 583–91. [DOI] [PubMed] [Google Scholar]
- Hah YS, Lee YR, Jun JS, Lim HS, Kim HO, Jeong YG, et al. A20 suppresses inflammatory responses and bone destruction in human fibroblast-like synoviocytes and in mice with collagen-induced arthritis. Arthritis Rheum 2010; 62: 2313–21. [DOI] [PubMed] [Google Scholar]
- Okazaki Y, Sawada T, Nagatani K, Komagata Y, Inoue T, Muto S, et al. Effect of nuclear factor-kappaB inhibition on rheumatoid fibroblast-like synoviocytes and collagen induced arthritis. J Rheumatol 2005; 32: 1440–7. [PubMed] [Google Scholar]
- Campbell IK, Gerondakis S, O'Donnell K, Wicks IP. Distinct roles for the NF-kappaB1 (p50) and c-Rel transcription factors in inflammatory arthritis. J Clin Invest 2000; 105: 1799–806. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Auphan N, DiDonato JA, Rosette C, Helmberg A, Karin M. Immunosuppression by glucocorticoids: inhibition of NF-kappa B activity through induction of I kappa B synthesis. Science 1995; 270: 286–90. [DOI] [PubMed] [Google Scholar]
- Yin MJ, Yamamoto Y, Gaynor RB. The anti-inflammatory agents aspirin and salicylate inhibit the activity of I(kappa)B kinase-beta. Nature 1998; 396: 77–80. [DOI] [PubMed] [Google Scholar]
- Majumdar S, Aggarwal BB. Methotrexate suppresses NF-kappaB activation through inhibition of IkappaBalpha phosphorylation and degradation. J Immunol 2001; 167: 2911–20. [DOI] [PubMed] [Google Scholar]
- Tomita T, Takeuchi E, Tomita N, Morishita R, Kaneko M, Yamamoto K, et al. Suppressed severity of collagen-induced arthritis by in vivo transfection of nuclear factor kappaB decoy oligodeoxynucleotides as a gene therapy. Arthritis Rheum 1999; 42: 2532–42. [DOI] [PubMed] [Google Scholar]
- Lee YR, Kweon SH, Kwon KB, Park JW, Yoon TR, Park BH. Inhibition of IL-1beta-mediated inflammatory responses by the IkappaB alpha super-repressor in human fibroblast-like synoviocytes. Biochem Biophys Res Commun 2009; 378: 90–4. [DOI] [PubMed] [Google Scholar]
- Lipsky PE, van der Heijde DM, St Clair EW, Furst DE, Breedveld FC, Kalden JR, et al. Infliximab and methotrexate in the treatment of rheumatoid arthritis. Anti-tumor necrosis factor trial in rheumatoid arthritis with concomitant therapy study group. N Engl J Med 2000; 343: 1594–602. [DOI] [PubMed] [Google Scholar]
- Feldmann M. Development of anti-TNF therapy for rheumatoid arthritis. Nat Rev Immunol 2002; 2: 364–71. [DOI] [PubMed] [Google Scholar]
- Nishimoto N, Yoshizaki K, Miyasaka N, Yamamoto K, Kawai S, Takeuchi T, et al. Treatment of rheumatoid arthritis with humanized anti-interleukin-6 receptor antibody: a multicenter, double-blind, placebo-controlled trial. Arthritis Rheum 2004; 50: 1761–9. [DOI] [PubMed] [Google Scholar]
- Komano Y, Tanaka M, Nanki T, Koike R, Sakai R, Kameda H, et al. Incidence and risk factors for serious infection in patients with rheumatoid arthritis treated with tumor necrosis factor inhibitors: a report from the Registry of Japanese Rheumatoid Arthritis Patients for Longterm Safety. J Rheumatol 2011; 38: 1258–64. [DOI] [PubMed] [Google Scholar]
- Nishimoto N, Ito K, Takagi N. Safety and efficacy profiles of tocilizumab monotherapy in Japanese patients with rheumatoid arthritis: meta-analysis of six initial trials and five long-term extensions. Mod Rheumatol 2010; 20: 222–32. [DOI] [PubMed] [Google Scholar]
- Zou Y, Lu Y, Wei D. Antioxidant activity of a flavonoid-rich extract of Hypericum perforatum L in vitro. J Agric Food Chem 2004; 52: 5032–9. [DOI] [PubMed] [Google Scholar]
- Li HB, Yi X, Gao JM, Ying XX, Guan HQ, Li JC. The mechanism of hyperoside protection of ECV-304 cells against tert-butyl hydroperoxide-induced injury. Pharmacology 2008; 82: 105–13. [DOI] [PubMed] [Google Scholar]
- Li FR, Yu FX, Yao ST, Si YH, Zhang W, Gao LL. Hyperin extracted from Manchurian rhododendron leaf induces apoptosis in human endometrial cancer cells through a mitochondrial pathway. Asian Pac J Cancer Prev 2012; 13: 3653–6. [DOI] [PubMed] [Google Scholar]
- Kim SJ, Um JY, Lee JY. Anti-inflammatory activity of hyperoside through the suppression of nuclear factor-kappaB activation in mouse peritoneal macrophages. Am J Chin Med 2011; 39: 171–81. [DOI] [PubMed] [Google Scholar]
- Ku SK, Zhou W, Lee W, Han MS, Na M, Bae JS. Anti-inflammatory effects of hyperoside in human endothelial cells and in mice. Inflammation 2015; 38: 784–99. [DOI] [PubMed] [Google Scholar]
- Ku SK, Kwak S, Kwon OJ, Bae JS. Hyperoside inhibits high-glucose-induced vascular inflammation in vitro and in vivo. Inflammation 2014; 37: 1389–400. [DOI] [PubMed] [Google Scholar]
- Arnett FC, Edworthy SM, Bloch DA, McShane DJ, Fries JF, Cooper NS, et al. The American Rheumatism Association 1987 revised criteria for the classification of rheumatoid arthritis. Arthritis Rheum 1988; 31: 315–24. [DOI] [PubMed] [Google Scholar]
- Lee YR, Lee JH, Noh EM, Kim EK, Song MY, Jung WS, et al. Guggulsterone blocks IL-1beta-mediated inflammatory responses by suppressing NF-kappaB activation in fibroblast-like synoviocytes. Life Sci 2008; 82: 1203–9. [DOI] [PubMed] [Google Scholar]
- Renard P, Ernest I, Houbion A, Art M, Le Calvez H, Raes M, et al. Development of a sensitive multi-well colorimetric assay for active NFkappaB. Nucleic Acids Res 2001; 29: E21. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lee YR, Hwang JK, Lee HS, Cheon YJ, Ryu JH, Lee SI, et al. SPA0355, a thiourea analogue, inhibits inflammatory responses and joint destruction in fibroblast-like synoviocytes and mice with collagen-induced arthritis. Br J Pharmacol 2011; 164: 794–806. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Qu Z, Garcia CH, O'Rourke LM, Planck SR, Kohli M, Rosenbaum JT. Local proliferation of fibroblast-like synoviocytes contributes to synovial hyperplasia. Results of proliferating cell nuclear antigen/cyclin, c-myc, and nucleolar organizer region staining. Arthritis Rheum 1994; 37: 212–20. [DOI] [PubMed] [Google Scholar]
- Wang LY, Wang HY, Ouyang J, Yu L, Chen B, Qin JQ, et al. Low concentration of lipopolysaccharide acts on MC3T3-E1 osteoblasts and induces proliferation via the COX-2-independent NFkappaB pathway. Cell Biochem Funct 2009; 27: 238–42. [DOI] [PubMed] [Google Scholar]
- Murphy G, Nagase H. Reappraising metalloproteinases in rheumatoid arthritis and osteoarthritis: destruction or repair? Nat Clin Pract Rheumatol 2008; 4: 128–35. [DOI] [PubMed] [Google Scholar]
- Li G, Liu D, Zhang Y, Qian Y, Zhang H, Guo S, et al. Celastrol inhibits lipopolysaccharide-stimulated rheumatoid fibroblast-like synoviocyte invasion through suppression of TLR4/NF-kappaB-mediated matrix metalloproteinase-9 expression. PLoS One 2013; 8: e68905. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pahl HL. Activators and target genes of Rel/NF-kappaB transcription factors. Oncogene 1999; 18: 6853–66. [DOI] [PubMed] [Google Scholar]
- Boissier MC. Cell and cytokine imbalances in rheumatoid synovitis. Joint Bone Spine 2011; 78: 230–4. [DOI] [PubMed] [Google Scholar]
- Joosten LA, Helsen MM, Saxne T, van De Loo FA, Heinegard D, van Den Berg WB. IL-1 alpha beta blockade prevents cartilage and bone destruction in murine type II collagen-induced arthritis, whereas TNF-alpha blockade only ameliorates joint inflammation. J Immunol 1999; 163: 5049–55. [PubMed] [Google Scholar]
- Kokkonen H, Soderstrom I, Rocklov J, Hallmans G, Lejon K, Rantapaa Dahlqvist S. Up-regulation of cytokines and chemokines predates the onset of rheumatoid arthritis. Arthritis Rheum 2010; 62: 383–91. [DOI] [PubMed] [Google Scholar]
- Tolboom TC, Pieterman E, van der Laan WH, Toes RE, Huidekoper AL, Nelissen RG, et al. Invasive properties of fibroblast-like synoviocytes: correlation with growth characteristics and expression of MMP-1, MMP-3, and MMP-10. Ann Rheum Dis 2002; 61: 975–80. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ou Y, Li W, Li X, Lin Z, Li M. Sinomenine reduces invasion and migration ability in fibroblast-like synoviocytes cells co-cultured with activated human monocytic THP-1 cells by inhibiting the expression of MMP-2, MMP-9, CD147. Rheumatol Int 2011; 31: 1479–85. [DOI] [PubMed] [Google Scholar]
- Kessenbrock K, Plaks V, Werb Z. Matrix metalloproteinases: regulators of the tumor microenvironment. Cell 2010; 141: 52–67. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bjorklund M, Koivunen E. Gelatinase-mediated migration and invasion of cancer cells. Biochim Biophys Acta 2005; 1755: 37–69. [DOI] [PubMed] [Google Scholar]
- Richardson VJ. Divergent and synergistic regulation of matrix metalloprotease production by cytokines in combination with C-C chemokines. Int J Immunopathol Pharmacol 2010; 23: 715–26. [DOI] [PubMed] [Google Scholar]
- Vandooren B, Kruithof E, Yu DT, Rihl M, Gu J, De Rycke L, et al. Involvement of matrix metalloproteinases and their inhibitors in peripheral synovitis and down-regulation by tumor necrosis factor alpha blockade in spondylarthropathy. Arthritis Rheum 2004; 50: 2942–53. [DOI] [PubMed] [Google Scholar]
- Okamoto T. NF-kappaB and rheumatic diseases. Endocr Metab Immune Disord Drug Targets 2006; 6: 359–72. [DOI] [PubMed] [Google Scholar]
- Kawai T, Akira S. TLR signaling. Cell Death Differ 2006; 13: 816–25. [DOI] [PubMed] [Google Scholar]
- Boyle WJ, Simonet WS, Lacey DL. Osteoclast differentiation and activation. Nature 2003; 423: 337–42. [DOI] [PubMed] [Google Scholar]
- Cronstein BN. Interleukin-6 — a key mediator of systemic and local symptoms in rheumatoid arthritis. Bull NYU Hosp Jt Dis 2007; 65: S11–5. [PubMed] [Google Scholar]