Abstract
Background
Corpus callosal abnormalities (CCA) in dogs have been only sporadically reported and are poorly characterized.
Hypothesis/Objectives
To describe the clinical presentation and magnetic resonance imaging (MRI) characteristics of dogs with CCA.
Animals
Fifteen client‐owned dogs.
Methods
Retrospective study. Records of the contributing institutions were reviewed to identify dogs diagnosed with malformations affecting the corpus callosum (CC); cases in which the CCA was thought to be secondary were excluded.
Results
The most represented breeds were Staffordshire Bull Terriers (5/15) and Miniature Schnauzers (3/15; n = 3, 20%) and the mean age at time of presentation of 19 months (range 3–81 months). The clinical signs most commonly reported were adipsia/hypodipsia with associated hypernatremia (12/15), tremors (6/15), and seizures (6/15). Review of the MR images revealed that 10 dogs had absence of the rostral CC and hypoplasia of the caudal portion, 4 dogs had a diffusely hypoplastic and dysplastic CC, and 1 dog had a diffusely hypoplastic CC. In 14 cases, there was abnormal cortical development with fusion of the ventral frontal lobes and part of the diencephalon, indicating lobar holoprosencephaly.
Conclusions and Clinical Importance
Previous literature has mainly associated CCA with adipsia and only 12 of 15 dogs in the current series demonstrated this abnormality. There are different degrees of the malformation but in 10 dogs the rostral portion of the CC is most severely affected. Fourteen dogs have simultaneous fusion of the midline structures rostral to the CC; this region has several structures involved in thirst regulation and might explain this derangement.
Keywords: Adipsia, Corpus callosum, Dog, Holoprosencephaly
Abbreviations
- AV3V
anteroventral third ventricle
- CCA
corpus callosal abnormalities
- CC
corpus callosum
- HPE
holoprosencephaly
- MRI
magnetic resonance imaging
The cerebral commissures are cortico‐cortical bundles of white matter connecting 1 hemisphere with the other, usually symmetrically, and the corpus callosum (CC) is the principal supratentorial commissure.1 There are 2 other telencephalic commissures, the rostral and the hippocampal commissures while the latter are common to all vertebrates, the corpus callosum is only found in placental mammals.2
The CC is typically divided into 4 segments: the rostrum, genu, body, and splenium (Fig 1). The fibers of the body form the roof of the rostral horn and central part of the lateral ventricle, and are attached ventrally to the septum pellucidum. Caudally, the CC is thickened to form the splenium. The rostral end of the CC is hook‐shaped where it bends ventrally at the genu and then tapers as the rostrum becoming continuous with the lamina terminalis. The fibers of the CC fan out laterally into each hemisphere from its body and also rostrally and caudally from its genu and splenium, respectively.1 The complete CC can be consistently seen on magnetic resonance imaging (MRI) by 8 weeks of age in dogs and has an adult appearance by 16 weeks. As it is a densely packed white matter structure, the mature CC has high signal on T1‐weighted and low signal on T2‐weighted magnetic resonance imaging (MRI); sagittal images are the most useful for evaluation of the CC.3
Figure 1.

Midsagittal T2‐weighted MR image of a normal corpus callosum.
Corpus callosal abnormalities (CCA) are a common cerebral malformation in people, with an incidence within the disabled population of 2–3 per 1004 and an estimated prevalence of 0.3–0.7% in patients undergoing brain imaging.5 Classically, CCA in humans are divided into partial (hypogenesis) or complete agenesis.1 The first type is more common and the most frequently seen anomalies tend to affect the caudal portion of the CC.1, 6 Recently, a more detailed classification system has been proposed6 dividing CCA into hypoplasia (in which all the major anatomic components are present but are reduced in size), hypoplasia with dysplasia (small CC with distorted overall shape), and agenesis (absence of CC). CCA are found commonly in association with other brain malformations.7
Sporadic cases of CCA in dogs have been reported in recent years because of the more widespread use of advanced imaging techniques in veterinary medicine8, 9, 10, 11; nonetheless these malformations are still poorly understood in this species. The aims of this case series were therefore to describe the clinical presentation, MRI findings, and eventual outcome as well of dogs with CCA.
Materials and Methods
The medical records of the contributing institutions were retrospectively reviewed to identify dogs that had been diagnosed with intracranial malformations affecting the CC (2 dogs have been reported previously by Jeffery et al, 2003 and 1 dog in MacKillop 2011). Cases in which the CCA was thought to be associated with hydrocephalus, porencephaly, or hydranencephaly were excluded. Data collected for each case included the signalment, clinical history, findings of the physical and neurologic examinations, serum biochemistry results, and MR images of the brain. Outcome data were subsequently collected through telephone consultation with the referring veterinarian.
Magnetic resonance imaging examinations included the following sequences: T2‐weighted images, fluid attenuated inversion recovery (FLAIR) images, and pre‐ and postcontrast (intravenous injection of 0.1 mmol/kg of gadopentetate dimeglumine) T1‐weighted images; all cases had transverse, sagittal, and dorsal plane images available for review. The MR images were reviewed to determine the degree of CC abnormality (agenesis, hypoplasia, dysplasia, or all three) and the presence and type of associated malformations.
Results
Fifteen cases were identified in the medical records of the different institutions. The mean age at presentation was 19 months (with a range of 3–81 months and a median of 11 months) with 9 dogs presenting within the first year of life. There were 5 Staffordshire Bull Terriers (SBT), 3 Miniature Schnauzers and 1 Miniature Schnauzer cross, and 1 each of the following breeds: Affenpinsher, Border Collie, Cairn Terrier, Pug, Rottweiler, and Tibetan Spaniel. Nine dogs were female and 6 were male (3 were neutered).
The most common clinical sign identified was adipsia/hypodipsia associated with hypernatremia (median 186 mmol/L, range 169–205 mmol/L), which was reported in 12 cases. Seizures and tremors were reported in 6 cases each. Interestingly, 5 dogs (4 SBTs and 1 Rottweiler) were reported to have hyperkeratosis and ichthyosis affecting different areas, but most frequently the feet and the nasal planum. Abnormal behavior was described in 4 cases and circling in 3 cases.
The neurologic examination was normal in 6 dogs (5 of which had hypernatremia recorded at the time of examination); some of these dogs had abnormalities described by the owners such as episodes of tremoring or circling and training difficulties that were not observed during the examination. In the other 9 dogs, neurologic deficits were identified (6 of which had hypernatremia recorded at the time of examination); these included tremors (5), proprioceptive deficits (4), ataxia (4), reduced menace response bilaterally (3), obtundation (4), circling to both sides (2), and head tilt and nystagmus (1).
Magnetic resonance imaging revealed that 10 dogs had an absence of the rostral CC and hypoplasia of the caudal portion, 4 dogs had a diffusely hypoplastic and dysplastic CC whereas 1 dog had a diffusely hypoplastic CC (Fig 2). The septum pellucidum and fornix could only be identified in 2 dogs. There was abnormal cortical development with fusion of the ventral frontal lobes, cyngulate gyri and part of the diencephalon in 14 cases; this was associated with loss of normal structures in the region rostral to the third ventricle (Fig 3). A focal circular T2 hyperintensity dorsal to the midbrain was detected in 7 dogs; this was thought to be consistent with either dilatation of the pineal recess or an arachnoid diverticulum. Chiari‐like malformation was seen in 3 dogs (Fig 4).
Figure 2.
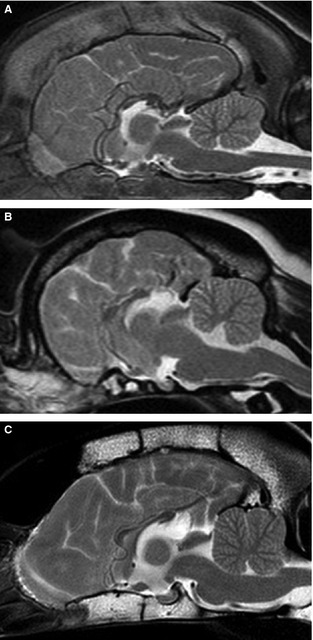
Midsagittal T2‐weighted MR images of (A) 3‐month Rottweiler with absence of the rostral CC and hypoplasia of the caudal portion, (B) 18‐month Pug with a diffusely hypoplastic and dysplastic CC, and (C) 3‐year 6‐month Border Collie with a diffusely hypoplastic CC.
Figure 3.
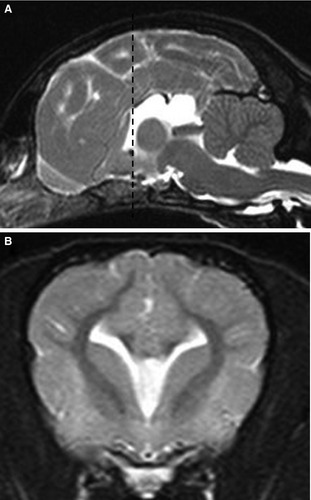
Midsagittal (A) and transverse (B) T2‐weighted MR images of a 7‐month Schnauzer crossbreed dog demonstrating fusion of the cyngulate gyri, lateral ventricles, and ventral frontal lobes with associated loss of some normal midline structures. On transverse MR images, the lateral ventricles have upturned, pointed corners reminiscent of bat wings as is typically seen with complete or focal aplasia of the CC.
Figure 4.
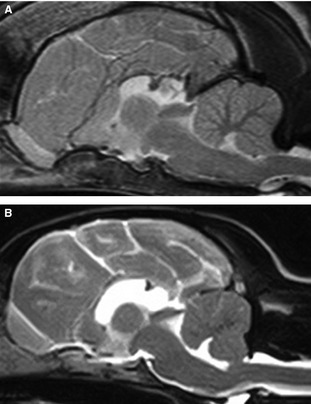
Midsagittal T2‐weighted MR images of a (A) 5‐month Staffordshire Bull Terrier and a (B) 3‐month Miniature Schnauzer with dilatation of the pineal recess and Chiari‐like malformation.
Treatments used included phenobarbital in 1 dog and fluvoxamine in another dog. All dogs that were reported as hypo‐ or adipsic were managed medically long term by adding water to their meals. In 1 dog, a feeding tube was placed to directly control water consumption. Four dogs were lost to follow‐up within 1–6 years after diagnosis; they had been alive for a median of 4 years before becoming lost to follow‐up. Four dogs had been euthanized at the time of review. In 3 cases, this was directly associated with the neurologic problems: one during a period of hypernatremia 6 months after diagnosis; one during an episode of cluster seizures 1 year after diagnosis and one during a period of hypernatremia that had resulted in nonambulatory tetraparesis and stupor 7 years after diagnosis (this dog underwent a postmortem examination that confirmed the MRI findings). One case was euthanized because of the severity of the skin abnormalities (severe recurrent pododermatitis secondary to ichthyosis and hyperkeratosis), which were considered by the owners to significantly affect quality of life; this occurred 15 months after diagnosis. The remaining 7 dogs were alive at the time of review with a median survival time of 20 months (10–77 months after diagnosis).
Discussion
This case series describes the clinical presentation of dogs with CCA. Previous literature has mainly associated this malformation with adipsia but not all dogs affected by the malformation present in this manner. The severity of the malformation varies but in most dogs the rostral portion of the CC is affected, which differs from human patients with the condition. Also, most dogs have simultaneous fusion of the midline structures rostral to the CC; this region has several structures involved in thirst regulation and may explain this derangement. Overall outcome is fair to good as most affected dogs are still alive with just dietary adjustments.
Corpus callosal abnormalities are rare malformations affecting dogs with only small numbers of cases seen across a number of institutions. Defects of the CC occur as a result of the failure of fibers to develop or to cross the midline.12 In the former case, there is a failure of the parent cell bodies or their axons to form in the cerebral cortex and in the latter, the axons form but are unable to cross the midline. This likely occurs because of absence of the massa commissuralis, leaving large aberrant longitudinal fiber bundles known as Probst bundles along the medial hemispheric walls.13 Probst bundles were not identified in any of these cases, so the first explanation seems most likely in this group of patients.
Recently, an association between early onset lysosomal storage diseases (namely the gangliosidoses) and a small CC size has been reported.14 It was postulated that the CC hypoplasia was most likely because of underdevelopment, similarly to what happens to the subcortical white matter in these conditions. An association between CCAs and inborn errors of metabolism has long been established in human patients.15 It is thought that at a molecular level, the development of the CC is complex and involves the synchronization of many cellular events; disruption or desynchronization of these mechanisms may lead to complete (early insult) or partial (late insult) callosal agenesis. In the dogs presented in this case series, lysosomal storage diseases were not considered likely because of their presentation and lack of other characteristic MRI abnormalities; therefore urine metabolic screening and genetic testing were not performed. Nonetheless, in view of the recent description of CC hypoplasia in certain canine and feline lysosomal storage diseases, testing for these conditions should be considered in clinical cases with CCA.
The most common clinical sign identified in association with CCA was hypodipsia/adipsia. Adipsic hypernatremia is characterized by chronic and recurrent episodes of hypernatremia associated with lack of thirst; in humans, this is thought to be associated with an impaired function of the hypothalamic osmoreceptors that regulate thirst and antidiuretic hormone (arginine vasopressin) secretion.16, 17 Vertebrates maintain plasma osmolality and extracellular volume primarily by regulating the ingestion and urinary excretion of water and electrolytes. An increase in the plasma osmolality, and consequent cellular dehydration, is the most potent stimulus of thirst.18 In the genesis of thirst, there is an important role for osmoreceptor‐Na+ receptor cells located in the circumventricular organs of the anterior aspect of the third ventricle. These structures contain sensory cells that respond to variations in the plasma osmotic pressure or the sodium concentration of plasma and cerebral spinal fluid.19 Lesions of the anteroventral third ventricle (AV3V), which includes 2 of the circumventricular organs, the median preoptic nucleus and the organum vasculosum lamina terminalis, induce permanent or temporary adipsia.18 Besides the AV3V, other midline structures such as the subfornical organ, medial septal area, anterior lateral hypothalamus, paraventricular nucleus, and the stria medullaris are also involved in the regulation of water/sodium intake and excretion18 and may be affected in these cohort of patients.
Most neurologic deficits were associated with forebrain dysfunction. A significant proportion of cases also presented tremors (which were persistent or temporary in some cases). This is difficult to explain with the malformations identified, as there are no anatomic structures in the affected region that could readily explain this clinical finding. White matter abnormalities, including reduced white matter volume and myelination delays, are reported to affect almost all cases (94%) of human patients with CCA.7 A possible explanation for the tremors is therefore that the affected animals had additional white matter abnormalities that we failed to detect. All dogs that presented tremors were hypodipsic and were either hypernatremic at examination or had episodes of hypernatremia recorded in their clinical history. It is therefore possible that the electrolyte disturbance could be associated with the tremors but these were reported as permanent in some dogs, making this relation less likely. In 1 dog, unexplained lateralized vestibular deficits (horizontal nystagmus and right‐sided head tilt) were found and described by the owners as present since birth. Review of the MRI in this case did not reveal any lateralized abnormalities; this patient had a diffusely hypoplastic and dysplastic CC.
There appeared to be a breed predisposition for CCA in both SBTs and Schnauzers. Also, we found a previously unreported association with dermatologic conditions (hyperkeratosis and ichthyosis), mainly in the SBTs. Similar associations have been reported in human patients and a genetic cause is often suspected.20, 21
The outcome of dogs with CCA appears fair to good although some dogs were euthanized because of repeated episodes of hypernatremia. Nonetheless, most dogs remained stable with either symptomatic medication or simple dietary management designed to improve fluid intake through the addition of water to the meals.
In all but one of the cases reported here, the CCA was associated with midline fusion in the region rostral to the CC and the third ventricle. By definition, these dogs therefore had a mild subtype of lobar holoprosencephaly.22 Recently, a very mild form of holoprosencephaly (HPE) has been reported in people by Hahn;23 in this subtype, named septopreoptic HPE, the nonseparation is restricted to the septal (subcallosal), preoptic regions, or both. In all cases, the CC was abnormally formed, with aplasia or hypoplasia of the rostrum and less severe hypoplasia of the genu. Although the canine cases reported here do not completely mirror the findings in septopreoptic HPE, a mild subtype of lobar HPE is suspected in these cases. Concurrent brain anomalies are commonly seen in humans with agenesis/hypogenesis of the CC or HPE.7 In these patients, a small diverticulum dorsal to the midbrain was found in 7 dogs. Based on routine imaging, it could not be determined if these cerebrospinal fluid accumulations represented dilatation of the caudodorsal third ventricle (pineal recess) or subarachnoid space (arachnoid diverticulum). A so‐called dorsal cyst is frequently seen in humans with holoprosencephaly and the diverticulum seen in these patients may represent a similar entity.24
The major limitation of this study is the lack of histopathologic confirmation in most cases (this is only available in 1 dog). This was not possible in most cases as patients were not euthanized in the short term and most are still alive. Also, the lack of hormonal testing, namely serum vasopressin measurement, is a significant limitation. This was unfortunately not considered at the time of evaluation of these patients but will be undertaken in future cases to evaluate osmoreceptor function.
Acknowledgments
The authors thank Erika Bersan for helping in the data collection.
Conflict of Interest Declaration: This study has not been supported by any grants or otherwise and the authors have no conflict of interests to declare.
Preliminary results were presented as an oral presentation at the 26th Annual Symposium of the ESVN‐ECVN, Paris, September 26–28, 2013.
References
- 1. Raybaud C. The corpus callosum, the other great forebrain commissures, and the septum pellucidum: Anatomy, development and malformation. Neuroradiology 2010;52:447–477. [DOI] [PubMed] [Google Scholar]
- 2. Aboitiz F, Montiel J. One hundred million years of interhemispheric communication: The history of the corpus callosum. Braz J Med Biol Res 2003;36:409–420. [DOI] [PubMed] [Google Scholar]
- 3. Gross B, Garcia‐Tapia D, Riedesel E, et al. Normal canine brain maturation at magnetic resonance imaging. Vet Radiol Ultrasound 2010;51:361–373. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Jeret JS, Serur D, Wisniewski K, Fisch C. Frequency of agenesis of the corpus callosum in the developmentally disabled population as determined by computerized tomography. Pediatr Neurosci 1985‐1986;12:101–103. [DOI] [PubMed] [Google Scholar]
- 5. Bedeschi MF, Bonaglia MC, Grasso R, et al. Agenesis of the corpus callosum: Clinical and genetic study in 63 young patients. Pediatr Neurol 2006;34:186–193. [DOI] [PubMed] [Google Scholar]
- 6. Hanna RM, Marsh SE, Swistun D, et al. Distinguishing 3 classes of corpus callosal abnormalities in consanguineous families. Neurology 2011;76:373–382. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Hetts SW, Sherr EH, Chao S, et al. Anomalies of the corpus callosum: An MR analysis of the phenotypic spectrum of associated malformations. AJR Am J Roentgenol 2006;187:1343–1348. [DOI] [PubMed] [Google Scholar]
- 8. Jeffery ND, Watson PJ, Abramson C, Notenboom A. Brain malformations associated with primary adipsia identified using magnetic resonance imaging. Vet Rec 2003;152:436–438. [DOI] [PubMed] [Google Scholar]
- 9. Sullivan SA, Harmon BG, Purinton PT, et al. Lobar holoprosencephaly in a Miniature Schnauzer with hypodipsic hypernatremia. J Am Vet Med Assoc 2003;223:1783–1787. [DOI] [PubMed] [Google Scholar]
- 10. Shimokawa Miyama T, Iwamoto E, Umeki S, et al. Magnetic resonance imaging and clinical findings in a Miniature Schnauzer with hypodipsic hypernatremia. J Vet Med Sci 2009;71:1387–1391. [DOI] [PubMed] [Google Scholar]
- 11. MacKillop E. Magnetic resonance imaging of intracranial malformations in dogs and cats. Vet Radiol Ultrasound 2011;52(1 Suppl 1):S42–S51. [DOI] [PubMed] [Google Scholar]
- 12. Sztriha L. Spectrum of corpus callosum agenesis. Pediatr Neurol 2005;32:94–101. [DOI] [PubMed] [Google Scholar]
- 13. Dobyns WB. Absence makes the search grow longer. Am J Hum Genet 1996;58:7–16. [PMC free article] [PubMed] [Google Scholar]
- 14. Hasegawa D, Tamura S, Nakamoto Y, et al. Magnetic resonance findings of the corpus callosum in canine and feline lysosomal storage diseases. PLoS ONE 2013;8:1–6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Prasad AN, Bunzeluk K, Prasad C, et al. Agenesis of the corpus callosum and cerebral anomalies in inborn errors of metabolism. Congenit Anom (Kyoto) 2007;47:125–135. [DOI] [PubMed] [Google Scholar]
- 16. Komatsu H, Miyake H, Kakita S, Ikuta H. Hypoplasia of the corpus callosum associated with adipsic hypernatremia and hypothalamic hypogonadotropinism: A case report and review of the literature. Pediatr Int 2001;43:683–687. [DOI] [PubMed] [Google Scholar]
- 17. Savasta S, Chiapedi S, Borali E, et al. Holoprosencephaly with neurogenic hypernatremia: A new case. Childs Nerv Syst 2008;24:139–142. [DOI] [PubMed] [Google Scholar]
- 18. Antunes‐Rodrigues J, de Castro M, Elias LL, et al. Neuroendocrine control of body fluid metabolism. Physiol Rev 2004;84:169–208. [DOI] [PubMed] [Google Scholar]
- 19. Bourque CW. Central mechanisms of osmosensation and systemic osmoregulation. Nat Rev Neurosci 2008;9:519–531. [DOI] [PubMed] [Google Scholar]
- 20. Reish O, Gorlin RJ, Hordinsky M, et al. Brain anomalies, retardation of mentality and growth, ectodermal dysplasia, skeletal malformations, Hirschsprung disease, ear deformity and deafness, eye hypoplasia, cleft palate, cryptorchidism, and kidney dysplasia/hypoplasia (BRESEK/BRESHECK): New X‐linked syndrome? Am J Med Genet 1997;68:386–390. [DOI] [PubMed] [Google Scholar]
- 21. Bibas‐Bonet H, Fauze R, Boente MC, et al. IFAP syndrome “plus” seizures, mental retardation, and callosal hypoplasia. Pedriatr Neurol 2001;24:228–231. [DOI] [PubMed] [Google Scholar]
- 22. Hahn JS, Barnes PD. Neuroimaging advances in holoprosencephaly: Refining the spectrum of the midline malformation. Am J Med Genet C Semin Med Genet 2010;154C:120–132. [DOI] [PubMed] [Google Scholar]
- 23. Hahn JS, Barnes PD, Clegg NJ, Stashinko EE. Septopreoptic holoprosencephaly: A mild subtype associated with midline craniofacial anomalies. AJNR Am J Neuroradiol 2010;31:1596–1601. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24. Pavone P, Barone R, Baieli S, et al. Callosal anomalies with interhemispheric cyst: Expanding the phenotype. Acta Pediatr 2005;94:1066–1072. [DOI] [PubMed] [Google Scholar]


