Abstract
Scanning electron microscopy with energy dispersive x-ray spectroscopy (SEM-EDS) supplies information that is complementary to those data traditionally obtained using inductively coupled plasma-mass spectrometry for analysis of inorganic tobacco and tobacco smoke constituents. The SEM-EDS approach was used to identify select inorganic constituents of mainstream cigarette smoke “tar.” The nature of SEM-EDS instrumentation makes it an ideal choice for microstructural analyses as it provides information relevant to inorganic constituents that could result from exposure to combusted tobacco products. Our analyses show that aluminum silicates, silica, and calcium compounds were common constituents of cigarette mainstream smoke “tar.”
Identifying inorganic tobacco smoke constituents is important because inhalation of fine inorganic particles could lead to inflammatory responses in the lung and systemic inflammatory responses. As cigarette smoking causes chronic inflammation in the respiratory tract, information on inorganic particulate in mainstream smoke informs efforts to determine causative agents associated with increased morbidity and mortality from tobacco use.
Keywords: Smoking, Cigarette, Silica, Aluminum silicate, Particulate, Smoke, Tar, Cancer, Inflammation, Chronic Obstructive Pulmonary Disease
Introduction
Cigarette smoking and disease caused by exposure to secondhand smoke are estimated to cause more than 480,000 deaths annually in the U.S. (1). Though the prevalence of cigarette smoking has declined in the U.S. adult population over the last decade, 17.8% of U.S. adults reported being current cigarette smokers (smoking every day or some days) in 2013 (2). Notable among tobacco related diseases is the presence and mechanistic role of chronic inflammation (3, 4). In particular, inflammation is thought to play a role in cancer and pulmonary diseases caused by inhaling tobacco smoke (3–6).
Cigarette smoke is a complex chemical mixture containing multiple chemical classes of more than 7,000 constituents (3,7). Mainstream cigarette smoke consists of vapor phase constituents and a particulate phase dispersed in an aerosol. Depending on the total lung particulate burden, some fraction of the inhaled particles can be absorbed into the systemic circulation or lymphatic ducts, cleared by the mucociliary system (8) unless it is impaired as a consequence of smoking, or eliminated by alveolar macrophages if insoluble (9).
Less attention has been paid to insoluble inorganic particles such as silicates in cigarette smoke. It is probable that these were not taken into account in previous risk assessments (7,10) because available data regarding the presence of aluminum and silicon in cigarette smoke is sparse. The lack of data is likely related to analytical measurement difficulties, analytical interferences, incompatibilities of essential reagents such as hydrofluoric acid with glass or quartz components of instrument introduction systems, and to background from plasma torches of inductively coupled plasma instrumentation.
We previously characterized the major inorganic substances present on the surfaces and interiors of tobacco leaves using Scanning Electron Microscopy with Energy Dispersive X-Ray Spectroscopy (SEM-EDS) (12). Aluminum and silicon-containing particles were found in abundance on the leaf surfaces in various forms of silica and aluminum silicate from soil (11). Silica was also found in the interiors of the leaves (11), in the form of phytolithic silica, a biogenic mineral that is commonly produced in plants (12, 13).
Here, we continue the study of major inorganic substances from tobacco leaves to begin characterizing the transfer of silica and aluminum silicate along with trace metals and other substances to tobacco smoke. This study provides a perspective on the transfer of inorganic substances present inside tobacco leaf or on tobacco leaf surfaces to mainstream smoke where they avoid removal by the cigarette filter and are available for inhalation by the smoker. The data that we present here demonstrates that SEM-EDS enables exploration of insoluble particles in tar from mainstream cigarette smoke in a manner that is complimentary to studies on trace metals when Inductively Coupled Plasma-Mass Spectrometry (ICP-MS) is used for total particulate matter (TPM) analyses.
Materials and Methods
Sample preparation
A random selection of commercially available filtered cigarettes (American Spirit Blue Pack®, Camel Blue®, Marlboro Medium®, Newport Non-Menthol 100®) were obtained from retail outlets in the greater Atlanta area between 2010 and 2014. Cooperation Centre for Scientific Research Relative to Tobacco (CORESTA, Paris, France) CM7 test cigarettes were obtained from Cerulean (Richmond, VA, USA). Cigarettes were conditioned according to ISO 3402 (14) prior to smoking with an RM20H rotary smoking machine (Borgwaldt KC, North Chesterfield, VA, USA) with electrostatic precipitation. Electrostatic precipitation of Total Particulate Matter (TPM) was performed at 24 kV. Samples were smoked using the Canadian Intense smoking regimen (15). Following smoking, the mouth end of the machine smoked cigarette filter used for the SEM-EDS analysis was carefully sliced from the cigarette with a razor blade (Figure 1).
Figure 1.

An agglomerate of particles held together by carbonaceous TPM tar on the filter fibers of a CORESTA CM7 cigarette filter (mouth end) smoked according to the Canadian Intense smoking regimen (11). This image was obtained in Low Vacuum mode with an accelerating voltage of 20 kV, spot size 3.0, 110 Pa vapor pressure in the sample chamber, with a 500μm pressure limiting aperture on a Concentric Backscatter Detector (CBS).
Two different procedures were utilized to prepare TPM samples for analysis. TPM was wiped from the electrostatic precipitator tube with a plastic spatula and transferred directly to 0.1 μm pore size polytetrafluoroethylene (PTFE) membrane filters (EMDMillipore, Billerica, MA, USA) followed by desiccation for 48 hours (Figure 2, Figure 7). Alternatively, the TPM was suspended in semiconductor VLSI grade methanol, (Sigma, St. Louis, Mo, USA), filtered thru a 0.05 μm (Tisch Scientific, North Bend, OH, USA) or 0.1 μm pore size PTFE filter, and allowed to dry prior to analysis (Figures 3–6, 8). Prior to analysis, a section of the PTFE filter was mounted on double coated conductive carbon tab on an aluminum stub and placed in a desiccator overnight before analysis. Other variations in sample preparation are described below.
Figure 2.
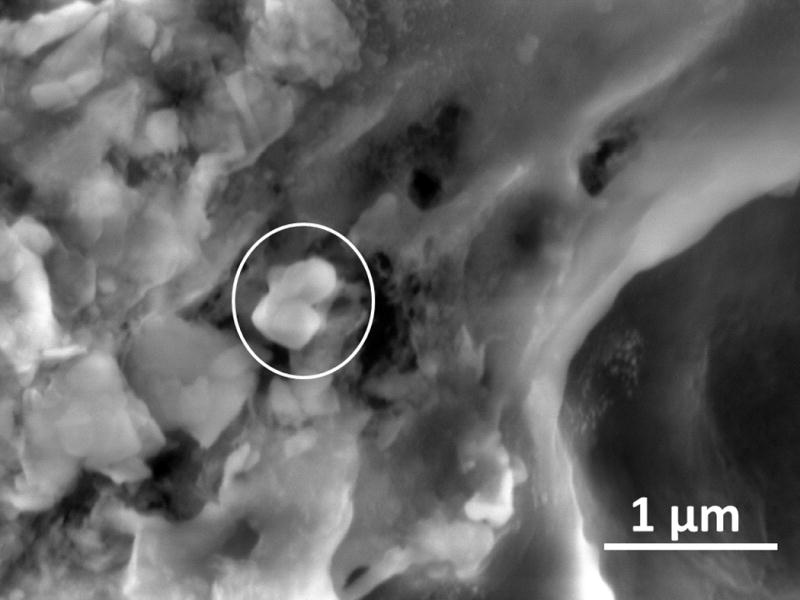
This image of two fine aluminum silicate particles in TPM from a Marlboro Medium® cigarette was obtained in Environmental (ESEM) mode with an accelerating voltage of 5kV, spot size 3.0, 200 Pa vapor pressure in the sample chamber, with a 500μm pressure limiting aperture on a Gaseous Secondary Electron Detector (GSED).
Figure 7.

Silica particles rinsed partially free from TPM. The image of TPM from an American Spirit Natural ® cigarette was obtained in Low Vacuum mode with an accelerating voltage of 5.0 kV, spot size 3.0, 60 Pa vapor pressure in the sample chamber, with a 500 μm pressure limiting aperture and LFD detector.
Figure 3.

Partial dissolution of tar from the TPM of a Camel Blue® cigarette revealed inorganic particles embedded in the tar. This false color SEM micrograph was obtained in Low Vacuum mode with an accelerating voltage of 10 kV, 100 Pa vapor pressure in the sample chamber, spot size 4.0, with a 500μm pressure limiting aperture and mixed Large Field Detector (LFD) and CBS. EDS analysis revealed that the composition of the particle on the left was silica, whereas the particle on the right was an agglomerate of calcium phosphate and calcium carbonate or oxalate. The fluorine peak is from the fluoropolymer filter beneath the sample.
Figure 6.

Silica particles embedded in TPM. The image of TPM from a Newport Non-Menthol 100’s ® cigarette was obtained in Environmental (ESEM) mode with an accelerating voltage of 10 kV, spot size 3.5, 266 Pa vapor pressure in the sample chamber, with a 500μm pressure limiting aperture with GSED.
Figure 8.

Calcium-containing crystalline particles composed of calcium, carbon, and oxygen. The crystals, which resemble calcium oxalate partially rinsed free of carbonaceous TPM tar, were obtained from the TPM from a Marlboro® Medium cigarette with an accelerating voltage of 5 kV, spot size 3.0, 130 Pa vapor pressure in the sample chamber, with a 500μm pressure limiting aperture and LFD detector.
SEM-EDS Instrumentation
Images were acquired with a Quanta 250 Field Emission Gun Scanning Electron Microscope (SEM) with “environmental mode (ESEM)” capabilities (FEI Co., Hillsboro, OR, USA). The SEM was equipped with an Energy Dispersive X-Ray (EDS) attachment with an 80 mm2 X-MaxN Silicon Drift Detector (Oxford Instruments, Concord, MA, USA). EDS analysis data was obtained with the Oxford EDS system with 80 mm2 X-MaxN Silicon Drift Detector (SDD) and Aztec software. Individual instrument settings are described in the respective figure captions.
Results
The utilization of low water vapor pressures in the sample chamber in Low Vacuum and Environmental (ESEM) modes of the Quanta 250 made it possible to acquire images of particles in non-conductive TPM samples on a fluoropolymer filter without the need for carbon coating or heavy metal complexation steps during sample preparation to mitigate sample or filter charging.
During preliminary work, few individual particles were seen in desiccated cigarette smoke TPM, because they were embedded in the tar. High Vacuum mode conditions dehydrated the samples further than was possible with desiccation but resulted in apparent precipitation of artefactual potassium sulfate crystals from the TPM. Calcium and silicate-containing particles larger than the mean diameters typically reported for tobacco smoke (16–18) and large agglomerates of smaller particles were visible when TPM was wiped from electrostatic precipitation tubes.
Figure 1 shows a particle agglomerate in carbonaceous TPM tar at the mouth end of the filter tip from a smoked CORESTA CM7 cigarette. EDS analysis of three particle agglomerates at the mouth end of a CM7 cigarette filter showed they consisted predominantly of carbon, oxygen, silicon, aluminum, iron, sulfur, potassium, calcium, magnesium, chlorine, sulfur, phosphorus, and small amount of titanium. The high carbon and oxygen TPM matrix surrounding the particles prevented a more definitive EDS analysis of these agglomerates. The individual particles in the agglomerate ranged in size from below 1 μm to approximately 4 μm. The largest individual particles were within two geometric standard deviations (σg = 2 μm) of the geometric mean for particle sizes reported for mainstream cigarette smoke particles (19) and were consistent with the size range for silicates observed in smokers’ lungs (20).
Two aluminum silicate particles in TPM from a Marlboro Medium® cigarette are shown adhering to a TPM particle agglomerate in Figure 2. These particles (approximately 160 × 170 nm) are consistent with the particle size range for silica and silicate particles in smokers lungs reported by Broday and Craighead (20) and are close to the mean particle size ranges reported by others (16–18). The high carbon and oxygen TPM matrix surrounding the particles again prevented a more definitive EDS analysis of the particles beyond the determination that the major elemental constituents were oxygen, silicon, and aluminum adhering to a predominantly carbonaceous matrix.
Partial dissolution of the organic portion of the TPM permitted discovery of particles that were embedded in the organic material. Figure 3 shows two particles within the TPM of a Camel Blue® cigarette that are still beneath the organic portion of TPM tar after partial dissolution of the TPM with methanol. These particles are approximately 2.0 μm × 2.5 μm. The organic layers were vaporized by focusing the electron beam on these particles for several minutes, enabling EDS analyses of the exposed particles. The particle on the left consisted of silicon and oxygen (silica) in a carbonaceous matrix (spectrum shown in Fig. 3). The particle on the right consisted of calcium, phosphorus, carbon, and oxygen.
Partial dissolution of the organic portion of the TPM from a Marlboro Medium® cigarette revealed a crystalline silica particle emerging from the remaining TPM. Figure 4 shows a fine particle sized (1 μm × 2 μm) silica particle partially embedded in TPM beside a PTFE filter that was rinsed with 15 mL methanol to control for any artifacts originating from the PTFE filter or from the semiconductor VLSI grade solvent. EDS analysis revealed a composition of silicon and oxygen in a matrix of predominantly carbon and oxygen.
Figure 4.
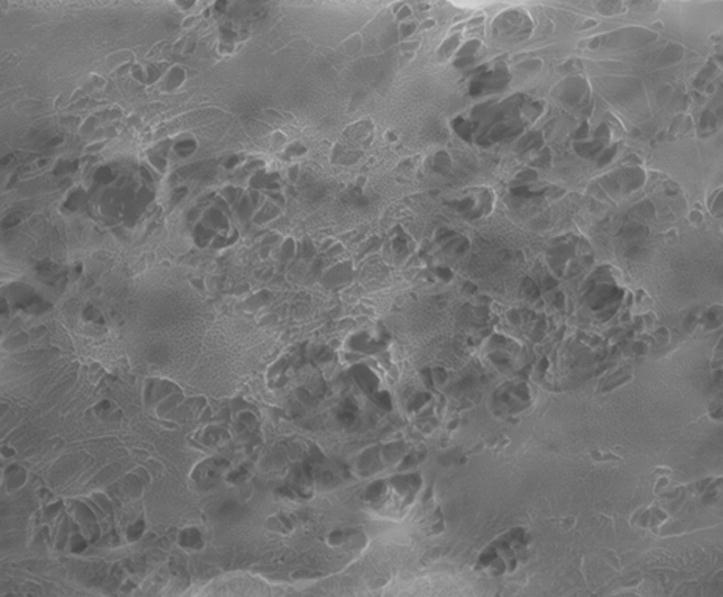

a. Control PTFE filter image (left) was obtained in Low Vacuum mode with an accelerating voltage of 10 kV, spot size 3.0, 100 Pa vapor pressure in the sample chamber, with LFD.
b. Fine silica particle partially rinsed free of organic tar. This image was obtained in Low Vacuum mode with an accelerating voltage of 20 kV, spot size 3.0, 110 Pa vapor pressure in the sample chamber, with a 500μm pressure limiting aperture and LFD detector.
In Figure 5, TPM from a Marlboro Medium® cigarette more extensively rinsed free of TPM was found to consist predominantly of aluminum silicate with additional elemental constituents including calcium, sodium, magnesium, potassium, and chloride. The platelike layers together with the composition suggest kaolinite particulate. The smaller particles appear to be less than 1 μm, whereas the larger particles could be up to 5 μm.
Figure 5.

Agglomerate of aluminum silicate particles partially rinsed free of carbonaceous TPM tar. The image of TPM from a Marlboro Medium ® cigarette was obtained in Low Vacuum mode with an accelerating voltage of 10 kV, spot size 4.0, 100 Pa vapor pressure in the sample chamber, with CBS.
Figure 6 shows an image of multiple particles from the TPM of a Newport Non-Menthol 100® cigarette. The particles ranged in size from approximately 50 nm to approximately 250 nm. The composition of the largest particle was determined by EDS to consist of only silicon and oxygen in a carbonaceous matrix.
In Figure 7, this agglomerate of silica particles from the TPM of an American Spirit Natural® cigarette appears to consist of a large number of sub-100 nm particles glued together after being partially rinsed relatively free of TPM. EDS analysis revealed that the composition was greater than 99% silicon and oxygen, though carbon was the major component of the adhesive matrix. Additional smaller agglomerates also appear trapped by the filter in the upper right of the figure and lower right, deeper in the PTFE filter matrix.
In Figure 8, an agglomerate of calcium-containing crystals and several individual crystals is shown after partial dissolution of tar from Marlboro Medium®. The individual particles were approximately 50 to 250 nm. The overall size of the agglomerate was approximately 5 μm diameter. The composition was determined with EDS to be calcium, carbon, and oxygen. The remaining carbonaceous TPM prevented exact formula determination, but it is possible that these are calcium oxalate crystals freed from the tobacco matrix (11) during combustion, since they do not appear as plate like as calcium carbonate crystals typically do.
Discussion
Silica, aluminum silicates, and calcium compounds were previously reported on the surfaces of tobacco leaves (11). Phytolithic silica was observed in the interiors of tobacco leaves (11). This study of inorganic particulate in the machine generated mainstream smoke of filtered cigarettes demonstrated that silica, aluminum silicates, and calcium compounds are the major inorganic particulate phase constituents in the smoke available for inhalation by the smoker. Silicon-containing TPM agglomerates at the mouth end of a cigarette filter and aluminum silicates, silica, and calcium-containing particles embedded in the carbonaceous TPM tar were observed for all brands examined. The brands included in the study were a random selection of commercially available cigarettes from different manufacturers and are not intended to make comparisons between brands. Particle sizes ranged from significantly less than 100 nm to several μm. Though we presented data on particles that were less than 100 nm, it is possible that even smaller particles passed unrecovered through the 0.05 or 0.1 μm pore size filters when the TPM organic matrix in which they were embedded was partially dissolved and filtered. In addition, we observed potassium sulfate, magnesium, chloride, and other constituents in the TPM. This agrees well with Langer et al., who reported that the mineral content in cigarette smoke consists principally of potassium chloride, potassium sulfate, potassium carbonate, calcium carbonate, and “quartz” (silica, 21).
Our results agree with Brody and Craighead’s suggestion that silica and aluminum silicates contribute substantially to the particulate matter in mainstream cigarette smoke, based on the widespread observation of these substances in pulmonary macrophages from the lungs of smokers (20). They and others reported large numbers of bronchiolar, alveolar, and interstitial pigmented macrophages with “smokers’ inclusions” consisting of aluminum silicate (20,22,23), for which they could find no source other than cigarette smoke. Heckman and Lehman also reported aluminum and silicon-containing inclusions in lung cells of rats after chronic exposure to tobacco smoke (24).The silica and aluminum silicate particles that we observed were found in the size ranges that Brody and Craighead described in smokers’ lungs.
Associations between the presence of aluminum and silicon compounds in the lungs as a consequence of smoking and pulmonary disease with carcinogenic outcome have been reported. Indeed, silicon compounds in various forms including silica, kaolinite (an aluminum silicate), and others have been found associated with adenocarcinomas and bronchoalveolar carcinomas (25). Terzakis reported silicon as the prominent element in particulate associated with peripherally located lung carcinomas with scar (scar carcinoma, a consequence of tissue inflammation or tumor necrosis followed by collagen formation) (25).
The presence of aluminum and silicon-containing particulate from cigarette smoke in pulmonary tissue has also been associated with fibrotic diseases such as Idiopathic Pulmonary Fibrosis (26). Aluminum silicates accumulated in the brown pigmented “smokers’ macrophages” that are characteristic of Respiratory Bronchiolitis-Interstitial Lung Disease (RB-ILD) (27), a chronic obstructive pulmonary disease (COPD) observed in the majority of smokers after smoking for seven to seventy-five pack years (20,28). Though there is also strong evidence for the involvement of cadmium in the development of obstructive lung disease (29), the widely cited work of Girod and King presented strong evidence that COPD resulting from smoking, and specifically that RB-ILD as a precursor lesion to emphysema, is a “dust-induced” disease where aluminum silicates such as kaolinite are described as the dust (30). Indeed, aluminum silicates have been shown to induce fibrotic obstructive lung disease in rats (31), and are known to cause fibrotic and pneumoconiotic disease in humans (32). Both aluminum salt and silica particles have been shown to independently activate the NALP3 inflammasome and induce synthesis of proinflammatory cytokine interleukin-1β, and NALP3-independent prostaglandin E2 (33). The presence of inflammation-inducing aluminum silicates (4,34) as principal constituents of pulmonary macrophage cytoplasmic inclusions has been described as strong evidence that aluminum silicates and silica may be principal etiologic agents in cigarette smoke for COPD (30,34). Smokers constitute the majority of cases of the otherwise rare pulmonary Langerhans’ cell hystiocytosis interstitial lung disease. The lysosomes of smoker’s macrophages that are associated with Langerhans’ hystiocytosis cells (dendritic cells) are described as containing numerous needle-like inclusions of aluminum silicate (35). Therefore, it is possible that multiple components of cigarette smoke, including cadmium and silica (carcinogens), and aluminum silicates or other silicates may be agents involved in tumorigenesis (25) and in the progression of chronic inflammation from which fibrotic, obstructive, and interstitial pulmonary disease result (25,27,30–35) as a consequence of cigarette smoking.
Conclusions
SEM-EDS was used to characterize common inorganic components of mainstream cigarette smoke particulate. Silica and aluminum silicates were prevalent in the carbonaceous matrix of TPM. Calcium compounds were also observed, though less frequently than silicates. Previous work showed that these inorganic particles were common on both the surfaces and interiors of tobacco leaves, apparently the source of these particles in cigarette smoke.
Silica and aluminum silicates have been understudied due to analytical interferences when the bulk TPM matrix was analyzed using ICP-MS. However, use of electron microscopic analyses to complement ICP-MS analyses of TPM can add information regarding commonly observed substances found in pulmonary macrophage inclusions and in pulmonary tissues of smokers. Our direct investigation into the nature of such silica and silicate particles in TPM from mainstream cigarette smoke may help in further investigations of cigarette smoke toxicity and the etiology of cancer and obstructive diseases that are consequences of tobacco smoking.
Acknowledgments
This study was funded by an interagency agreement by the U.S. Food and Drug Administration Center for Tobacco Products.
Footnotes
Disclaimer
The findings and conclusions in this report are those of the authors and do not necessarily represent the official position of the Centers for Disease Control and Prevention. Use of trade names and commercial sources is for identification only and does not constitute endorsement by the U.S. Department of Health and Human Services, or the U.S. Centers for Disease Control and Prevention.
Contributor Information
R. Steven Pappas, Email: RPappas@cdc.gov.
Mary M. Halstead, Email: MHalstead@cdc.gov.
Clifford H. Watson, Email: CWatson@cdc.gov.
References
- 1.U.S. Department of Health and Human Services. The Health Consequences of Smoking—50 Years of Progress: A Report of the Surgeon General. Atlanta, GA: U.S. Department of Health and Human Services, Centers for Disease Control and Prevention, National Center for Chronic Disease Prevention and Health Promotion, Office on Smoking and Health; 2014. p. 2,17. http://www.surgeongeneral.gov/library/reports/50-years-of-progress/exec-summary.pdf, last accessed 3 February 2015. [Google Scholar]
- 2.Jamal A, Agaku IT, O’Connor E, King BA, Kenemer JB, Linda Neff L. Current Cigarette Smoking Among Adults — United States, 2005–2013. Morb Mortal Wkly Rep. 2014;63(47):1108–1112. [PMC free article] [PubMed] [Google Scholar]
- 3.U.S. Department of Health and Human Services. 2010 Surgeon General’s Report—How Tobacco Smoke Causes Disease: The Biology and Behavioral Basis for Smoking-Attributable Disease. Atlanta, GA: U.S. Department of Health and Human Services, Centers for Disease Control and Prevention, National Center for Chronic Disease Prevention and Health Promotion, Office on Smoking and Health; 2010. p. 9,11,17,33,408,442. http://www.cdc.gov/tobacco/data_statistics/sgr/2010/index.htm, last accessed 3 February 2015. [Google Scholar]
- 4.Pappas RS. Toxic elements in tobacco and in cigarette smoke: inflammation and sensitization. Metallomics. 2011;3:1181–1198. doi: 10.1039/c1mt00066g. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Aggarwal BB, Shishodia S, Sandur SK, Pandey MK, Sethi G. Inflammation and cancer: How hot is the link? Biochem Pharmacol. 2006;72:1605–1621. doi: 10.1016/j.bcp.2006.06.029. [DOI] [PubMed] [Google Scholar]
- 6.Balkwill F, Mantovani A. Inflammation and cancer: back to Virchow? Lancet. 2001;357:539–545. doi: 10.1016/S0140-6736(00)04046-0. [DOI] [PubMed] [Google Scholar]
- 7.Fowles J, Dybing E. Application of toxicological risk assessment principles to the chemical constituents of cigarette smoke. Tob Control. 2003;12:424–430. doi: 10.1136/tc.12.4.424. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Lippmann M, Yeates DB, Albert RE. Deposition, retention and clearance of inhaled particles. Brit J Ind Med. 1980;37:337–362. doi: 10.1136/oem.37.4.337. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Lehnert BE. Pulmonary and thoracic macrophage subpopulations and clearance of particles from the lung. Environ Health Perspect. 1992;97:17–46. doi: 10.1289/ehp.929717. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Burns DM, Dybing E, Gray N, Hecht S, Anderson C, Sanner T, O’Connor R, Djordjevic M, Dresler C, Hainaut P, Jarvis M, Opperhuizen A, Straif K. Mandated lowering of toxicants in cigarette smoke: a description of the World Health Organization TobReg proposal. Tob Control. 2008;17:132–141. doi: 10.1136/tc.2007.024158. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Halstead MM, Watson CH, Pappas RS. Scanning electron microscopic analysis of inorganic substances on smokeless and cigarette tobacco. J Anal Toxicol. 2015;39:698–701. doi: 10.1093/jat/bkv097. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Huitu O, Forbes KM, Helander M, Julkunen-Tiitto R, Lambin X, Saikkonen K, Stuart P, Sulkama S, Hartley S. Silicon, endophytes and secondary metabolite as grass defenses against mammalian herbivores. Front Plant Sci. 2014;5 doi: 10.3389/fpls.2014.00478. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Sivanesan I, Park SW. The role of silicon in plant tissue culture. Front Plant Sci. 2014;5 doi: 10.3389/fpls.2014.00571. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.International Organization for Standardization. Tobacco and tobacco products – atmosphere for conditioning and testing. ISO. 1999;3402:1–4. [Google Scholar]
- 15.Hammond D, Wiebel F, Kozlowski LT, Borland R, Cummings KM, O’Connor RJ, McNeill A, Connolly GN, Arnott D, Fong GT. Revising the machine smoking regime for cigarette emissions: implications for tobacco control policy. Tob Control. 2007;16:8–14. doi: 10.1136/tc.2005.015297. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Broday DM, Robinson R. Application of cloud dynamics to dosimetry of cigarette smoke particles in the lungs. Aerosol Sci Tech. 2003;37:510–527. [Google Scholar]
- 17.Becquemin MH, Bertholon JF, Attoui M, Roy F, Roy M, Dautzenberg B. Particle size in the smoke produced by six different types of cigarette tobacco. Rev Mal Respir. 2009;26:e12–e18. doi: 10.1016/s0761-8425(07)91386-8. [DOI] [PubMed] [Google Scholar]
- 18.Dickens C, McGrath C, Warren N, Biggs P, McAughey J. Puffing and inhalation behaviour in cigarette smoking: Implications for particle diameter and dose. J Phys Conference Series. 2009;151 doi: 10.1088/1742-6596/151/1/012019. [DOI] [Google Scholar]
- 19.Anderson PJ, Wilson JD, Hiller FC. Particle Size Distribution of Mainstream Tobacco and Marijuana Smoke. Am Rev Respir Dis. 1989;140:202–205. doi: 10.1164/ajrccm/140.1.202. [DOI] [PubMed] [Google Scholar]
- 20.Brody AR, Craighead JE. Cytoplasmic inclusions in pulmonary macrophages of cigarette smokers. Lab Invest. 1975;32:125–132. [PubMed] [Google Scholar]
- 21.Langer AM, Nolan RP, Bowes DR, Shirey SB. Inorganic particles found in cigarette tobacco, cigarette ash, and cigarette smoke. In: Wehner AF, Felton D-L, editors. Biological Interaction of Inhaled Mineral Fibers and Cigarette Smoke. Columbus, OH, USA: Battelle; 1989. pp. 421–439. [Google Scholar]
- 22.Lynn WS, Kylstra JA, Sahu SC, Tainer J, Shelburne J, Pratt PC, Gutknecht WF, Shaw R, Ingram P. Investigations of black bronchoalveolar human lavage fluid. Chest. 1977;72:483–488. doi: 10.1378/chest.72.4.483. [DOI] [PubMed] [Google Scholar]
- 23.Choux R, Pautrat J, Viallat J, Farisse P, Boutin C. Inorganic cytoplasmic inclusions in alveolar macrophages. Arch Pathol Lab Med. 1978;102:79–83. [PubMed] [Google Scholar]
- 24.Heckman CA, Lehman GL. Ultrastructure and distribution of intracellular spicules in rat lung following chronic tobacco smoke exposure. J Natl Cancer I. 1985;74:647–657. [PubMed] [Google Scholar]
- 25.Terzakis JA. X-Ray microanalysis of peripheral lung carcinomas. Ultrastruct Pathol. 1995;19:167–173. doi: 10.3109/01913129509064218. [DOI] [PubMed] [Google Scholar]
- 26.Monso E, Tura JM, Marsal M, Morell F, Pujadas J, Morera J. Mineralogical microanalysis of idiopathic pulmonary fibrosis. Arch Environ Health. 1990;45:185–188. doi: 10.1080/00039896.1990.9936714. [DOI] [PubMed] [Google Scholar]
- 27.Carpio C, Gómez-Carrera L, Álvarez-Sala R. Smoking and other interstitial lung diseases. ISRN Pulmonol. 2011 doi: 10.5402/2011/876534. [DOI] [Google Scholar]
- 28.Voelkel NF. In: Chronic Obstructive Lung Disease. MacNee W, editor. People’s Medical Publishing House; USA: 2008. pp. 175–176. www.pmph-usa.com. [Google Scholar]
- 29.Lin Y-S, Caffrey JL, Chang M-H, Dowling N, Lin JW. Cigarette smoking, cadmium exposure, and zinc intake on obstructive lung disorder. Respir Res. 2010;11 doi: 10.1186/1465-9921-11-53. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Girod CE, King TE. COPD: A Dust-Induced Disease? Chest. 2005;128:3055–3064. doi: 10.1378/chest.128.4.3055. [DOI] [PubMed] [Google Scholar]
- 31.Lemaire I, Dionne PG, Nadeau D, Dunnigan J. Rat lung reactivity to natural and man-made fibrous silicates following short-term exposure. Environ Res. 1989;48:193–210. doi: 10.1016/s0013-9351(89)80034-9. [DOI] [PubMed] [Google Scholar]
- 32.Craighead JE. Diseases associated with exposure to silica and nonfibrous silicate minerals. Arch Pathol Lab Med. 1988;112:673–720. [PubMed] [Google Scholar]
- 33.Kuroda E, Ishii KJ, Uematsu S, Ohata K, Coban C, Akira S, Aritake K, Urade Y, Morimoto Y. Silica crystals and aluminum salts regulate the production of prostaglandin in macrophages via NALP3 inflammasome-independent mechanisms. Immunity. 2011;34:514–526. doi: 10.1016/j.immuni.2011.03.019. [DOI] [PubMed] [Google Scholar]
- 34.Portillo K, Morera J. Combined pulmonary fibrosis and emphysema syndrome: a new phenotype within the spectrum of smoking-related interstitial lung disease. Pulmonary Med. 2012 doi: 10.1155/2012/867870. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Hammar SP, Allen TC. Histiocytosis and storage diseases. In: Tomashefski JF, Cagle PT, Farver C, Fraire AE, editors. Dail and Hammar’s Pulmonary Pathology. 3rd. Vol. 1. Springer; New York, NY, USA: 2009. pp. 622–623. Nonneoplastic Lung Disease. [Google Scholar]


