Abstract
Breast and prostate cancers are malignancies in which steroid hormones drive cellular proliferation. Over the past century, this understanding has led to successful treatment strategies aimed to inhibit hormone-mediated tumor growth. Nonetheless, disease relapse and progression still pose significant clinical problems, with recurrent and metastatic tumors often exhibiting resistance to current drug therapies. The central role of androgens and estrogens in prostate and breast cancer etiology explains not only why endocrine therapies are often initially successful but also why many tumors ultimately become resistant. It is hypothesized that reducing the concentration of active hormones in the systemic circulation may be insufficient to block cancer progression, as this action selects for tumor cells that can generate active steroids from circulating precursors. This review aims to highlight the currently known differences of steroid biosynthesis in normal physiology versus hormone-dependent cancers, modern approaches to the assessment and targeting of these pathways, and priorities for future research.
Keywords: Tamoxifen, Androgen Receptor, Androgen Deprivation Therapy, Finasteride, Fulvestrant
Introduction
Steroid hormones and their precursors are synthesized and extensively metabolized primarily in the adrenals and gonads of healthy men and women [1]. These steroid products are secreted into the systemic circulation and exert their physiological effects by (1) binding to their cognate receptors in target tissues and initiating signaling pathways required for cellular growth and sexual maturation and (2) acting as substrates for further metabolism to active hormones, which then act on target tissues. The testes and the ovaries primarily synthesize testosterone or estradiol, respectively, which promote the development of secondary sexual characteristics, enable reproduction, and serve additional functions in the skeleton, brain, and other organs.
Among the most common malignancies in humans are prostate cancer in men and breast cancer in women, neoplasias of epithelial cells in glands whose development is driven by sex-specific gonadal steroids [2]. In many cases, these gonadal steroids fuel the growth and progression of these tumors, and hormone-deprivation therapies are used with or without surgery as first-line treatments. Unfortunately, these cancers often demonstrate either de novo resistance to hormonal therapies or subsequently acquire compensatory mechanisms to proliferate despite castrate concentrations of androgens and estrogens in the circulation. Here, we will review the current state of knowledge on how tumors obtain and synthesize these steroids, approaches to study the acquisition of resistance to treatment, and future areas of investigation.
Normal Physiology
The Hypothalamic–Pituitary–Adrenal Axis
Under the regulation of higher brain centers, neurons in the paraventricular nucleus of the hypothalamus release corticotropin-releasing hormone (CRH) [3] into the portal circulation, which stimulates adrenocorticotropin (ACTH) secretion from the corticotrope cells in the anterior pituitary [4]. ACTH binds to its extracellular receptor on cells of the adrenal cortex to stimulate the synthesis of cortisol and androgen precursors [5], which are not stored but are continuously released in the systemic circulation. Cortisol exerts negative feedback on CRH and ACTH production, achieving homeostasis. Aldosterone production is primarily under the control of a separate axis, renin–angiotensin–aldosterone system.
Adrenal Steroidogenesis
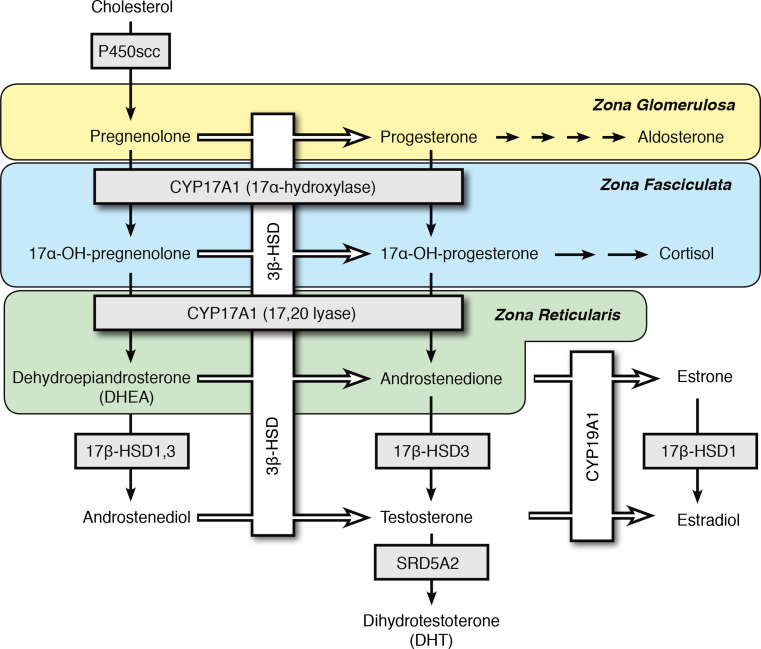
The adrenal glands are responsible for the synthesis of mineralocorticoids, glucocorticoids, and small amounts of androgens but relatively large amounts of androgen precursors. Specifically, within the adrenal gland, the adrenal cortex cells express steroidogenic enzymes and cofactor proteins in a zone-specific manner (Fig. 1). The adrenal cortex is comprised of the three zones, each expressing their own complement of proteins necessary for efficient synthesis of a dominant steroid product. The zona glomerulosa (ZG) expresses the enzymes necessary for aldosterone synthesis, while the zona fasiculata (ZF) primarily synthesizes cortisol. The zona reticularis (ZR) is the adrenal zone responsible for the production of androgens under the stimulation of ACTH, but these cells primarily synthesize androgen precursors. The ZR is characterized by very little 3β-hydroxysteroid dehydrogenase/isomerase (3βHSD) expression in the adult human. Consequently, steroid synthesis mostly follows the Δ5-pathway from pregnenolone to dehydroepiandrosterone (DHEA) [6, 7], which is sulfated and exported as dehydroepiandrosterone sulfate (DHEAS). DHEAS is the predominant circulating 19-carbon androgen precursor steroid, with a plasma concentration of about 10 μmol/L throughout most of adult life but declining progressively after about age 60 [8].
Fig. 1.
Adrenal steroidogenesis. This schematic illustrates the biosynthesis pathways of mineralocorticoids, glucocorticoids, and sex steroids in the adrenal cortex by highlighting the predominant substrates and products within each zone. The three zones, zona glomerulosa (ZG), zona fasiculata (ZF), and zona reticularis (ZR), are labeled and designated with different background colors. Boxes denote steroidogenic enzymes, and arrows represent directionality of the enzymatic reactions. The pathway begins in the upper left hand corner with the conversion of cholesterol to pregnenolone. Multistep conversions are indicated with multiple arrows when the enzymes are not specified
Cholesterol is the sole precursor for all steroid hormone synthesis. Steroid synthesis begins with the steroidogenic acute regulatory (StAR) protein aiding in the translocation of cholesterol from a pool in the outer mitochondrial membrane to the inner mitochondrial membrane. The mitochondrial cytochrome P450 (CYP) cholesterol side chain cleavage enzyme (P450scc, CYP11A1) cleaves the bond between the 20–22 carbons of cholesterol through a series of three oxygenation reactions. The final product of this reaction is the 21-carbon, Δ5-steroid pregnenolone, which is the common initial precursor for downstream synthesis of mineralocorticoids, glucocorticoids, and sex steroids. Pregnenolone is a substrate for both 3βHSD and steroid 17-hydroxylase/17,20-lyase (P450c17, CYP17A1). 3βHSD is the enzyme responsible for converting pregnenolone to its 21-carbon, Δ4-steroid congener, progesterone [9]. CYP17A1 is a bifunctional P450 that catalyzes two major reactions within the endoplasmic reticulum of steroidogenic cells. CYP17A1 can hydroxylate the 17-carbon of progesterone and pregnenolone to form their 17-hydroxy products, 17OH-pregnenolone or 17OH-progesterone [10–12]. These 17-hydroxy products are substrates for other enzymes in their further metabolism to cortisol, or they are further metabolized by CYP17A1’s second function, which is the 17,20-lyase activity. This 17,20-lyase activity cleaves the C–C bond between carbons 17 and 20 of the aforementioned 17-hydroxy substrates to form the 19-carbon androgen precursors DHEA (major Δ5-pathway) or androstenedione (minor Δ4 pathway). CYP17A1’s 17,20-lyase activity is enhanced by the coexpression of cytochrome b 5 (CYB5A), which allosterically stimulates this reaction [13, 14]. While the ZR expresses both CYB5A and CYP17A1, the ZF expresses only CYP17A1 [15, 16]. This zone-specific expression of CYB5A helps to explain why the ZF primarily synthesizes the 21-carbon steroid cortisol, while the ZR synthesizes large amounts of 19-carbon androgens and their precursors.
Using human adrenal vein samples, Nakamura et al. showed that testosterone is synthesized in small amounts in the human adrenal [17]. Type 5 17β-hydroxysteroid dehydrogenase (17βHSD5 or AKR1C3) has been implicated as the steroidogenic enzyme responsible for catalyzing the limited conversion of androstenedione to testosterone in the ZR. Microarray analysis and qPCR studies confirmed that the ZR expresses AKR1C3 mRNA and protein. Knockdown of AKR1C3 via siRNA in the human adrenal H295R cell line reduced testosterone production by 40 % compared to scrambled control siRNA [17]. These data highlight the potential for direct adrenal testosterone synthesis beyond the well-known production of 19-carbon androgen precursors, which are metabolized to active androgens in peripheral organs and target tissues.
The Hypothalamic–Pituitary–Gonadal Axis
With the onset of puberty, loss of repression from higher brain centers allows neurons in the arcuate nucleus of the hypothalamus to resume the pulsatile secretion of gonadotropin-releasing hormone (GnRH) every 90–120 min. This GnRH enters the portal circulation and stimulates pulsatile release of the gonadotropins, luteinizing hormone (LH), and follicle-stimulating hormone (FSH) from the gonadotropes in the anterior pituitary. Pulsatile secretion is critical for reproductive function, because constant exposure to GnRH downregulates its receptor on gonadotropes and thwarts axis function [18]. In males, LH acts on the testicular Leydig cells to stimulate testosterone synthesis, and in peripheral tissues, steroid 5α-reductases convert testosterone to the more potent androgen, 5α-dihydrotestosterone (DHT). In women, LH acts on the ovarian theca cells and, to a lesser extent, the granulosa cells, to drive androgen synthesis, but the ovary lacks 17βHSD type 3, the enzyme that most efficiently converts androstenedione into testosterone. In the ovary, FSH induces the expression of the aromatase (P450aro, CYP19A1) enzyme, which converts androstenedione and testosterone from the theca cells to the estrogens estrone (E1) and estradiol (E2) [19], as well as 17βHSD type 1, the specific 17βHSD isoform that efficiently converts E1 to E2 [20]. In men, FSH acts on the Sertoli cells to facilitate spermatogenesis. In both males and females, androgens and estrogens exert negative feedback on GnRH and LH production [21]. FSH production is primarily under the tonic negative feedback of inhibin B, a protein produced in the Sertoli and granulosa cells [22].
Gonadal Steroidogenesis
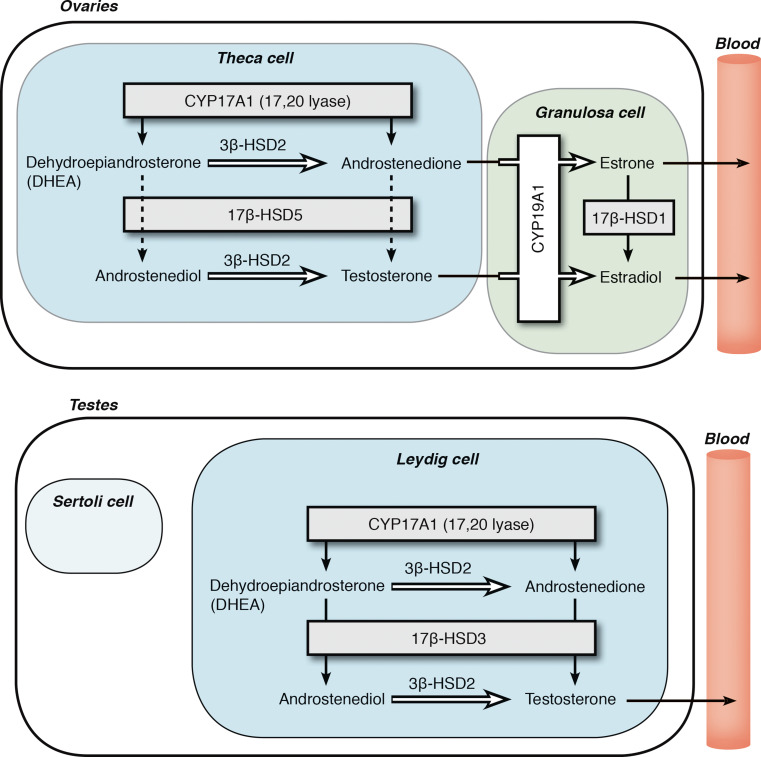
Although the ZR in the adrenal cortex produces less sex steroids than the gonads and large amounts of their precursors, the primary site of sex steroid synthesis is the gonads, using the same enzymes and pathways to get as far as DHEA. Similar to the adrenal cortex, the theca and granulosa cells of the ovaries express their own host of different steroid-metabolizing enzymes that orchestrate the synthesis of specific steroids (Fig. 2a). Immunohistochemical (IHC) studies in human ovaries by Sasano and colleagues revealed high CYP11A1 and CYP17A1 in the theca interna cells adjacent to the developing follicles but found CYP19A1 expression confined to the granulosa cells [23]. Therefore, the granulosa cells are responsible for estrogen synthesis and secretion by way of aromatizing the androgens produced in the ovarian theca cells. In the testes, only the Leydig cells express CYP17A1 (Fig. 2b), and Leydig cells are the only human cells that normally express the androgenic 17βHSD3, which efficiently converts 19-carbon, 17-ketosteroids to active androgens, such as androstenedione to testosterone.
Fig. 2.
Gonadal steroidogenesis. This figure depicts the enzymes expressed in the cells that comprise the gonads of females and males. a Ovarian theca cells express CYP17A1 to produce androstenedione and a small amount of testosterone, and these androgens are further aromatized into estrogens in ovarian granulosa cells before entering the circulation. b The testicular Leydig cells are the major steroidogenic cells in the male gonads, and these cells express CYP17A1 to convert androgen precursors into testosterone. Note the different 17βHSD isoenzymes present in the ovary and testis, which afford the major products E2 and testosterone, respectively
Peripheral Steroid Metabolism
Despite interventions that prevent gonadal hormone secretion, sufficient amounts of androgens and estrogens may remain in the circulation to activate their respective receptors [24, 25]. As discussed above, the adrenal glands produce very small amounts of testosterone and estradiol directly, yet the adrenal is a source of abundant 19-carbon androgen precursors such as DHEAS. Even a small portion of orally administered DHEA is converted to testosterone, indicating that tissues other than the adrenals and gonads possess the enzymatic machinery to complete the pathways to androgens and estrogens. One major reason for this capacity for extragonadal hormone generation is the redundancy of key enzyme activities. While the gonads and adrenals primarily express 3βHSD type 2 [9], the liver, skin, and other tissues contain a second, highly homologous isoenzyme, 3βHSD type 1 [26]. The human 17βHSD family includes at least 14 isoenzymes, each with its characteristic spectrum of activities and tissue-specific expression patterns [27]. Even steroid 5α-reductase activity derives from the type 1 and type 2 isoenzymes, with two genes bearing different ontogenies [28]. Among the AKR1C enzymes, all isoforms possess both 17βHSD and 3αHSD activities, which interconvert active and inactive hormones in peripheral and target tissues. The complexity of peripheral steroid metabolism provides a conduit for active hormones, and these hormones can drive breast and prostate cancer progression despite strategies to suppress gonadal steroid synthesis.
An early demonstration of the importance of peripheral hormone synthesis is in the estrogen dependence of most breast cancers in postmenopausal women, who lack ovarian-derived estrogens [29]. In an attempt to pinpoint the source of postmenopausal estrogen production, Grodin et al. analyzed plasma samples from six postmenopausal women and measured the conversion of androstenedione to E1, the predominant estrogen in postmenopausal women [30]. Patients were administered [14C]-androstenedione and subsequent conversion to E1 was measured in urine samples. Because the investigators were able to attribute nearly all of the measured E1 to the administered [14C]-androstenedione, they concluded that peripheral aromatization of androstenedione is the primary source of postmenopausal E1, as opposed to being of ovarian or gonadal origin.
Nuclear Hormone Receptor Signaling
Androgen receptor (AR) and estrogen receptor (ER) are steroid receptors and members of the protein superfamily known as nuclear hormone receptors [31]. These steroid receptors exist most commonly as unbound monomers in a dynamic equilibrium between the nucleus and cytoplasm under the regulation of heat shock and other chaperone proteins [32]. The receptors possess a unique ligand-binding domain (LBD) [33], and upon ligand binding, these receptors dissociate from the chaperone complex and undergo characteristic conformational changes that promote receptor homodimerization [34]. These ligand-activated receptor dimers translocate to the nucleus, where they bind to cognate response elements on DNA and initiate the transcription or repression of genes involved in growth and development. The recruitment of accessory proteins known as coactivators and corepressors to the transcription start site aid in the determination of which genes are expressed or repressed [35]. These coregulator proteins represent a potential strategy for further modulation of nuclear hormone receptor signaling.
Estrogens and Breast Cancer
Estrogen Receptor and Breast Cancer
In the later part of the 19th century, surgeons began performing bilateral oophorectomies to treat women with breast cancer, and Dr. George Beatson was a prominent early investigator [36]. Although there were differing opinions on the rationale for why this treatment was successful, the general consensus was that the ovaries secreted factors that promoted tumor growth. Over the next 100 years, our knowledge of these factors—primarily E2, its cognate receptor (ER), and ER signaling—has led to considerable advancements in treating women with ER-positive breast cancers [37].
Over 231,000 new cases of invasive breast cancer will be diagnosed this year in the USA [2]. Over two thirds of these cases will express ER (“ER-positive tumors”), and for these patients with ER-positive cancers, hormonal manipulation reduces the risk of recurrence or death, particularly in postmenopausal women [38, 39]. Drugs that antagonize estrogen action are effective treatments for patients with metastatic disease and clearly reduce breast cancer mortality when given in the adjuvant setting [37, 40–42]. These data are consistent with Beatson’s success performing oophorectomies in premenopausal women, which led to research over the following 40 years exploring the sources of estrogens in postmenopausal women and subsequent strategies to block estrogen synthesis and ER signaling in breast cancer.
Lippmann and colleagues first reported the importance of ER in breast cancer in vitro in the early 1970s. Using breast cancer cell culture models, specifically the ER-positive, E2-dependent MCF-7 cells, they demonstrated increased cellular proliferation by measuring DNA, RNA, and protein synthesis after E2 treatment. In addition, they showed that competitive inhibition of E2 binding to ER using the antiestrogen tamoxifen blocked the E2-induced effects [43, 44].
Pathways of Estrogen Synthesis in Breast Cancer
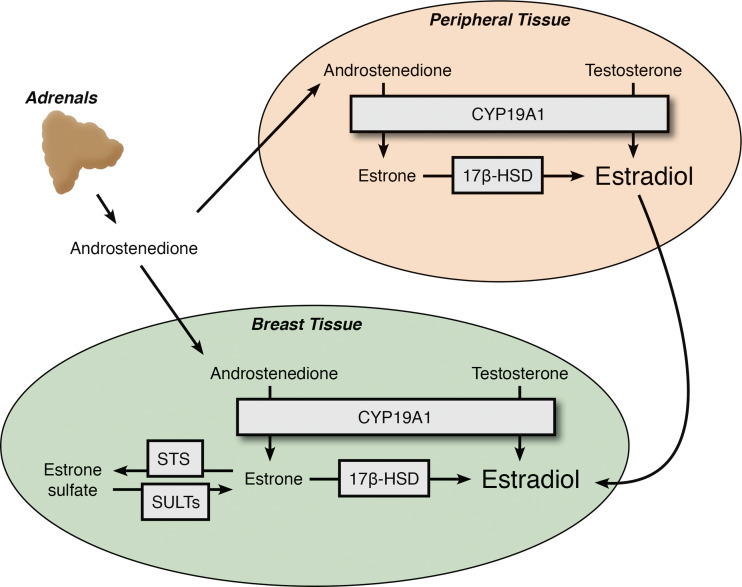
In the absence of functional ovaries, the adrenals were suspected as the source of estrogens in postmenopausal women. Adrenalectomy or hypophysectomy were modestly successful in these patients with remission rates between 25–50 %, and “medical adrenalectomy” with aminoglutethimide showed similar efficacy [45]. Nevertheless, the adrenal gland is known to produce abundant DHEAS, but not E2. It is now recognized that peripheral adipose tissue expresses CYP19A1 and contributes to circulating estrogens in the postmenopausal setting [46]. A study using reverse transcription polymerase chain reaction (RT-PCR) showed that CYP19A1 mRNA expression levels in fat from the buttocks, thighs, and abdomens of postmenopausal women were 2–4 times higher than those observed in young women [47]. Indeed, CYP19A1 mRNA is highly expressed in breast adipose and breast epithelial tissues, and tissue concentrations of E2 are approximately twice as high in breast tumor tissue compared to normal tissue [48], consistent with the local aromatization of adrenal-derived precursors.
In addition to the CYP19A1-mediated aromatization of androgens in peripheral tissues, a sulfatase enzyme has also been implicated in contributing to the delivery of E2 precursors to tumors [49] (Fig. 3). The steroid sulfatase (STS) enzyme removes the sulfate group of estrone sulfate (E1S) to yield E1. E1 can then be converted to E2 via 17βHSD1, which is also expressed in many of the same peripheral tissues as CYP19A1 [50]. An analysis of STS expression and function in breast cancer revealed that STS activity is higher in breast tumor tissue compared to healthy controls and that E1S and E2 were also elevated in breast tumor tissue [48]. The hydrolysis of the sulfate group is reversible, as local expression of sulfotransferases (known as SULTs) can repeat the sulfonation reaction. Over 44 SULT isoforms have been discovered, but only a handful of these sulfonate steroids. Notable SULTs include SULT1E1 (estrogens) and SULT2A1 (non-aromatic steroids.) Given that the risk of developing breast cancer is highly associated with endogenous sex hormone levels, particularly E2, E1, and E1S [51], this pathway represents a source of estrogens contributing to breast cancer progression.
Fig. 3.
Sources of estrogen in postmenopausal women. Extragonadal estrogen secretion via intracrine and paracrine pathways significantly contributes to breast cancer progression. Breast tissue, in addition to other peripheral tissues, expresses CYP19A1 that mediates the conversion of circulating androgen precursors originating primarily in the adrenal, into E2. Following menopause, E2 remains a potent growth stimulus to the roughly 70 % of breast cancer cells expressing ER. AIs work by blocking the local conversion of androgens into estrogens in these extragonadal tissues and significantly reducing circulating E2 levels to prevent E2-induced tumor growth. E1S is an additional source of E2, as the local expression of estrone sulfatase (STS) is able to convert E1S back to the E2 precursor, E1
Targeting of Estrogen Synthesis and Action in Breast Cancer
Two major pharmacological approaches have been developed to block the action of estrogen: (1) direct competition with estrogen for ER binding (e.g., tamoxifen and fulvestrant) and (2) blocking the production of estrogen in postmenopausal women (e.g., letrozole, anastrozole, and exemestane). Both of these approaches have been shown to reduce disease recurrence and prolong survival in postmenopausal breast cancer patients with ER-positive disease [37, 42]. Although the use of ER expression in breast cancers is essential to determine if a patient should receive any form of endocrine therapy, there is no other biomarker to further personalize the type of endocrine therapy that should be administered.
The first successful approach to targeting estrogen’s action in breast cancer was the development of antiestrogens [52]. Tamoxifen is an ER antagonist, or more precisely, a selective estrogen-receptor modulator (SERM), because it has tissue-specific estrogenic and antiestrogenic effects. SERMs, including tamoxifen, can be ER agonists or ER antagonists depending on tissue expression of the nuclear regulatory proteins (coactivators and corepressors) that regulate the expression of estrogen receptor-regulated genes [35]. Therefore, it is an effect of the recruited coregulatory proteins that mediates a SERM’s pharmacologic activity. The composition of coregulatory proteins in complex with ligand-bound ER appears to be ligand specific and determined by receptor conformation [53]. Endogenous ligands such as E2 induce a different conformational change [54] than a SERM like tamoxifen [55] and therefore recruit different coregulatory proteins to the site of DNA binding within the nucleus. Tamoxifen has been shown to reduce disease recurrence and to prolong survival in both premenopausal and postmenopausal women with ER-positive breast cancer as well as prevent breast cancer in high-risk women [56]. Several of its metabolites, including 4-hydroxytamoxifen [57] and endoxifen [57, 58], are also ER antagonists, which are even more potent than tamoxifen itself. However, one of the major drawbacks of SERMs is that their tissue-specific properties can lead to off-target effects by acting as ER agonists in other tissues. For example, tamoxifen acts as an agonist in bone [59] as well as in the uterus and endometrium [60], where ER agonism by SERMs can lead to endometrial hyperplasia and cancer [60]. In addition, tamoxifen therapy carries a similar risk of venous thrombosis as other estrogen therapies [56].
A second class of estrogen antagonists is the selective estrogen-receptor downregulators (SERDs). SERDs differ in their mechanism of action from SERMs in that they promote the degradation of ER protein [61], whereas SERMs like tamoxifen still allow for ligand-bound receptor to bind to DNA within the nucleus. Fulvestrant is the only FDA-approved SERD that is used clinically; however, its clinical use is limited because it must be administered via intramuscular injection as opposed to an orally administered antiestrogen like tamoxifen. This drawback has led to the development of newer orally bioavailable SERDs, some of which are currently being tested in early phase I and II clinical trials for the management of ER-positive breast cancer [62].
A third approach to treating ER-positive breast cancer is to block the production of E2 by inhibiting CYP19A1 [63]. Pharmacological inhibition of CYP19A1 was first achieved with aminoglutethimide (AG) [25]. Trials comparing AG to tamoxifen demonstrated similar efficacy in each treatment arm, but AG therapy was associated with worse side effects [64]. Despite its ability to inhibit estrogen synthesis, AG lacks selectivity for CYP19A1 and requires hydrocortisone replacement. These properties limited the use of AG for the treatment of ER-positive breast cancer and illustrated the need for more selective aromatase inhibitors (AIs). The first rationally designed AIs were mechanism-based substrate analogs, including 4-hydroxyandrostenedione, testolactone, 10-propargylestr-4-ene-3,17-dione, and exemestane [65–67]. Second- and third-generation inhibitors are azole-based non-steroidal compounds with high affinity and irreversible binding to the heme iron of the enzyme, including fadrozole, anastrozole, and letrozole. Of these, exemestane, anastrozole, and letrozole are used clinically in the adjuvant setting to treat ER-positive breast cancer.
Two large phase III clinical trials compared the efficacy of tamoxifen to an AI, alone or in combination. The Arimidex, Tamoxifen Alone, or in Combination (ATAC) trial showed that in postmenopausal women with localized breast cancer, AI therapy was superior to tamoxifen over the course of 5 years of treatment. Anastrozole (trade name Arimidex) significantly prolonged disease-free survival and significantly reduced distant metastases compared to tamoxifen [68]. The Breast International Group (BIG) 1–98 trial compared the efficacy of the AI letrozole to tamoxifen and again showed that AI therapy is superior to tamoxifen in postmenopausal women with ER-positive breast cancer [69]. The letrozole-treatment arm showed significantly increased progression-free survival and also a reduced incidence of distant metastases compared to tamoxifen [69]. The ATAC and BIG 1–98 trial data resulted in the adoption of AIs as the standard of care for postmenopausal women with ER-positive tumors.
Androgens and Prostate Cancer
Androgen Receptor and Prostate Cancer
The observation that prostate gland development is absent in 46,XY individuals with complete androgen insensitivity and steroid 5α-reductase type 2 deficiency firmly established the dependence of prostate growth on androgens [70]. Nearly all prostate cancers express AR, and prostate hyperplasia is androgen-dependent. Androgen deprivation therapy (ADT) was first described as a viable treatment option for prostate cancer in the early 1940s. Huggins and Hodges reported that removal of the testes (orchiectomy) promoted prostate tumor regression [71, 72]. ADT causes tumor regression or stabilization in the majority of patients; however, a substantial number of patients experience disease relapse months to years later. Originally, prostate-cancer recurrences during ADT were assumed to be “androgen independent,” but several groups have shown that androgen-dependent genes are expressed in relapsing tumors and their metastases [73]. Hence, this clinical condition has been renamed “castration-resistant prostate cancer” (CRPC), and most prostate cancer deaths are due to CRPC [74]. Among the possible mechanisms of resistance include amplification or overexpression of AR, which makes the receptor more sensitive to lower levels of circulating androgens [75]; gain-of-function mutations in AR, which render the receptor “promiscuous” and activated by host of other steroids including AR antagonists [76]; and the acquisition of mechanisms to produce androgens either de novo or by limited metabolism of circulating precursor steroids [77].
Pathways of Androgen Synthesis in Prostate Cancer
Belanger et al. observed that castration in adult males reduced circulating testosterone and DHT levels to approximately 5–10 % of their precastration values. Of note, however, is that castration had no effect on adrenal 19-carbon androgen precursors such as DHEA, DHEAS, and androstenedione [78]. A more recent study by Titus et al. examined patient samples to better understand androgen signaling in recurrent prostate cancers upon progression during ADT. By comparing recurrent prostate tumor tissue to androgen-stimulated benign prostate tissue, they noted similar concentrations of testosterone but with 91 % lower amounts of DHT in the recurrent tumor tissue compared to control [24]. It is believed that these remaining concentrations of DHT are still sufficient to activate AR and induce cancer growth.
Given the abundance of DHEAS in the circulation and the limited number of steps to testosterone (3) or DHT (4) via redundant pathways, adrenal-derived 19-carbon steroids and their metabolism have received considerable study as a mechanism driving CRPC. In some prostate cancer cell lines and tumor xenografts, DHEA stimulates growth similar to that of testosterone, but only if converted to Δ4-metabolites. The limiting enzyme in this conversion to active androgens is 3βHSD [79], but in prostate cancers, the major species is generally the type 1 isoenzyme rather than the type 2 found in the adrenal and testis. Inhibitors of 3βHSD shift the dose–response curve for DHEA in proportion to the enzymatic blockade [80]. In 2013, a common allelic variant of the HSD3B1 gene was reported to increase enzyme stability and to prevent proteasomal degradation. The prolonged half-life of the 3βHSD1-N367T variant results in greater amounts of DHT synthesis from DHEA compared to wild-type enzyme [77]. In human CRPC metastases, the selection pressure leads to overrepresentation of this allele, and the presence of this variant portends poor prognosis. The 3βHSD1-N367T variant has major implications for prostate cancer, as its increased expression can promote increased androgen synthesis from adrenal-derived precursors.
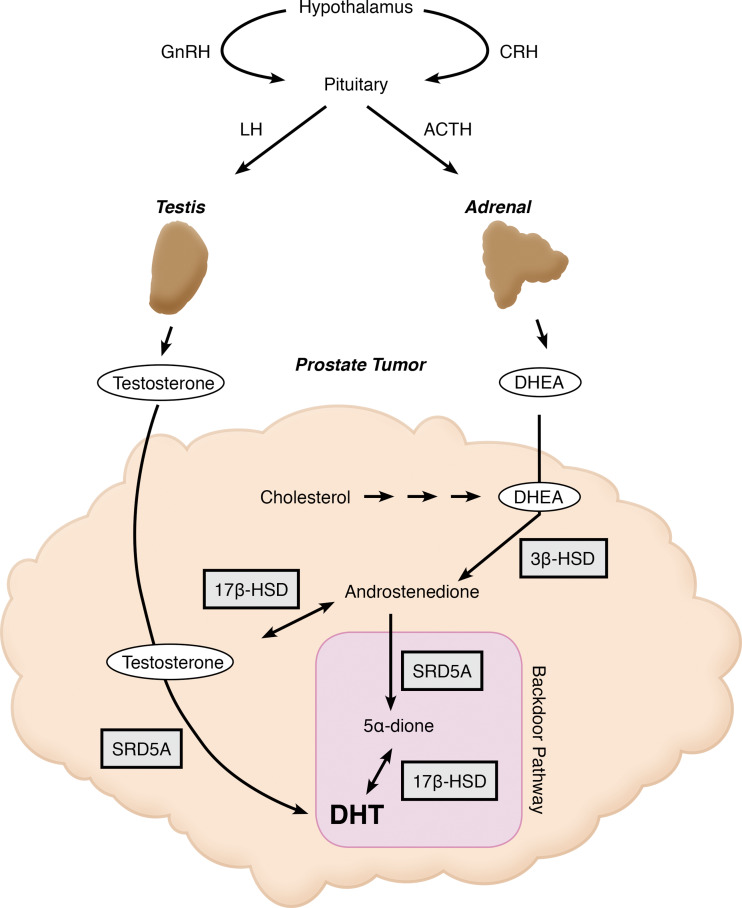
While the conversion of testosterone to the more potent androgen DHT is required for normal prostate development and prostate hyperplasia, the importance of DHT in prostate cancer is not as clear. Of the two 5α-reductase isoenzymes, the type 2 (SRD5A2) is the principal enzyme expressed in the normal or hyperplastic prostate tissue as well as genital skin, where it catalyzes the synthesis of DHT in the fetus during male sexual development. The type 1 isoenzyme (SRD5A1) is normally expressed in the liver and all other skin; however, SRD5A1 is also the predominant isoenzyme in prostate cancers [28]. While both isoenzymes have broad substrate specificity for most 21-carbon and 19-carbon Δ4-steroids, their relative efficiencies for various substrates varies somewhat, particularly under castrate conditions when circulating testosterone concentrations are low. In prostate cancer cell lines and tumor xenografts, Chang et al. demonstrated that SRD5A1 converts androstenedione—derived from DHEA via 3βHSD—to 5α-androstanedione, which is then converted to DHT via 17βHSD-mediated catalysis. This alternative pathway to DHT, which bypasses testosterone as an intermediate, appears to be the dominant route to DHT from circulating adrenal-derived 19-carbon steroids in CRPC [81]. More recent studies using metastatic tumor samples from patients have confirmed this pathway to DHT and characterized its impact in men with prostate cancer, who had stopped responding to traditional AR antagonists. In addition to this pathway, another alternative or “backdoor pathway” to DHT involves the SRD5A1-catalyzed 5α- and subsequent 3α-reduction of 21-carbon steroids, which then undergo cleavage via the 17,20-lyase activity of CYP17A1 to androsterone [82, 83]. Androsterone undergoes 17βHSD-catalyzed reduction to 5α-androstane-3α,17β-diol and then 3αHSD-catalyzed oxidation to DHT. Evidence for contributions from these alternate pathways to DHT, neither of which use testosterone as an intermediate, in the progression of CRPC derive from several laboratories and independent studies.
In addition to further metabolism of gonadal and adrenal precursors, other studies show that androgens can derive de novo from the CRPC tumor itself (Fig. 4). Dillard et al. showed that, in cell culture models of prostate cancer that have been passaged to mimic an androgen-deprived state, the expression of steroidogenic enzymes necessary for intracine testosterone synthesis are increased [7]. Thin-layer chromatography (TLC) analysis suggested that these cells could convert radiolabeled cholesterol into testosterone, presumably due to higher expression of steroidogenic enzymes not present in the parental prostate cancer cells. Montgomery and colleagues confirmed these findings by extensively characterizing which androgen signaling mechanisms are still present in human tissues of those with CRPC. They also identified several steroidogenic enzymes that are upregulated in CRPC tumor metastases compared to the primary tumor tissue, including CYP17A1, 3βHSD1, 17βHSD3, and CYP19A1 [84].
Fig. 4.
Pathways of androgen synthesis in CRPC. Shown in this figure are the pathways contributing to androgen synthesis that occur in the testes, adrenals, and prostate tumor itself in men with CRPC. Expression of CYP17A1 in all three tissues produces androgens, and further metabolism of these androgenic substrates results in the synthesis of the potent androgen receptor agonists, testosterone and DHT. Highlighted in purple is the backdoor pathway to DHT synthesis that bypasses testosterone as an intermediate. Independent studies have shown evidence to support this particular pathway being intact in CRPC
Targeting of Androgen Synthesis and Action in Prostate Cancer
Long-acting GnRH agonists and antagonists achieve medical castration by suppressing LH release and thus ablating testicular androgen synthesis. Long-acting GnRH agonists such as leuprolide acetate produce an initial surge in LH and testosterone, and then disrupt the pulsatile stimulation of pituitary gonadotropin receptors, resulting in receptor desensitization. GnRH antagonists such as degarelix competitively inhibit GnRH binding and do not produce an initial hormone surge; chronically, both treatments decrease LH and testosterone concentrations to castrate levels. GnRH analogs are the cornerstone of ADT in prostate cancer, and these drugs have been used also for ovarian estrogen suppression in premenopausal women with breast cancer [85–87]. Although GnRH agonists and antagonists effectively ablate most androgen and estrogen production in the tissues primarily responsible for sex steroid production, these drugs do not block adrenal steroid synthesis or intracrine steroid production.
Androgen-receptor antagonists directly inhibit ligand binding to AR [88, 89]. Because testosterone and DHT have such high affinity (∼1 nM) for AR, the early generations of antiandrogens were not sufficiently potent to block all androgen action and showed limited efficacy in men with CRPC. Flutamide and bicalutamide bind to AR with affinities approximately 30-fold less than DHT. These drugs bind AR in the cytoplasm and inhibit ligand binding but still permit nuclear translocation. A next-generation, more potent AR antagonist is enzalutamide, which has a much higher affinity for AR compared to older drugs and also prevents nuclear translocation [90]. Enzalutamide treatment after chemotherapy in men with CRPC resulted in a 4.8-month increase in overall survival and 37 % reduction in risk of death compared to placebo [91]. In chemotherapy-naïve men with metastatic prostate cancer, enzalutamide decreased the risk of death by 29 % and delayed chemotherapy initiation by a median of 17 months compared to placebo [92].
Beyond suppressing LH secretion and blocking AR, a third strategy to treat CRPC is to inhibit the synthesis of testosterone. Ketoconazole is an azole drug commonly used to treat fungal infections by inhibiting lanosterol demethylase (CYP51A1) [93] and thus ergosterol production, which is essential for fungal cell membrane integrity. Ketoconazole gained traction as a viable treatment option for CRPC, because ketoconazole demonstrates clinically relevant off-target inhibition of several human cytochrome P450s, including CYP11A1, CYP11B1, and CYP17A1 [94, 95]. Unfortunately, ketoconazole is a weak CYP17A1 inhibitor (Ki ∼130 nM) [96], and it strongly inhibits the important drug-metabolizing enzyme CYP3A4, thus limiting its clinical use. Consequently, considerable effort has been expended to develop selective CYP17A1 inhibitors to treat CRPC, and the “holy grail” of these efforts is the development of a drug that specifically inhibits only the 17,20-lyase activity.
Abiraterone is a potent (∼3 nM) [96], functionally irreversible inhibitor of both the 17α-hydroxylase and 17,20-lyase activities of CYP17A1. Inhibition of 17,20-lyase activity with abiraterone significantly reduces circulating concentrations of all 19-carbon steroids, including DHEA, androstenedione, and testosterone. Simultaneous inhibition of 17α-hydroxylase activity prevents the conversion of pregnenolone into cortisol, relieves cortisol negative feedback, allows ACTH to rise, and drives the accumulation of cortisol precursors with mineralocorticoid activity, primarily 11-deoxycorticosterone (DOC) and corticosterone [97]. DOC accumulation causes hypertension and hypokalemia similar to genetic 17-hydroxylase deficiency [98], and administration of mineralocorticoid antagonist or glucocorticoid normalizes these side effects [97]. Consequently, abiraterone treatment requires concomitant administration of a glucocorticoid (such as prednisolone 5 mg BID) to avoid these side effects. An improved CYP17A1 inhibitor that only blocks the 17,20-lyase activity could have a profound impact on the clinical care of CRPC patients, allowing early stage treatment without chronic glucocorticoid coadministration.
Early clinical trials demonstrated abiraterone’s ability to completely suppress testosterone, DHEA, and androstenedione synthesis in men with CRPC to below the limits of detection within 20 days of starting treatment [99]. In the first randomized phase III trial, de Bono and colleagues showed that abiraterone prolongs overall survival in men with CRPC, who had been previously treated with docetaxel, a commonly used chemotherapeutic agent. Overall survival increased by 3.9 months in the abiraterone-treatment group compared to placebo [100]. A subsequent study in docetaxel-naïve patients with CRPC demonstrated that abiraterone plus prednisone prolonged radiographic-free survival by 8.2 months over placebo plus prednisone and showed a trend toward improved survival [101].
Some of the CYP17A1 inhibitors under current development also bind directly to AR and antagonize its activity. In vitro binding studies have shown that abiraterone binds to AR with rather weak affinity in the high micromolar range compared to 1 nM for T and DHT [102]. In contrast, the Δ4-metabolite of abiraterone is a more potent AR antagonist than enzalutamide, and this compound also inhibits 3βHSD and 5α-reductase [103]. Galeterone represents another drug that has exhibited preclinical success with respect to androgen synthesis and androgen signaling blockade. Galeterone has the same chemical Δ5-background structure as DHEA and abiraterone with the Δ16-modification of abiraterone but a benzimidazole moiety to bind the heme iron rather than the 3′-pyridyl group of abiraterone. Galeterone shows some preferential inhibition of 17,20-lyase activity and also antagonizes AR in the 1–10 μM range [104]. Galeterone not only antagonizes AR activity but it also promotes AR protein degradation, representing a novel antiandrogen mechanism of action [105]. In phase I and II trials of men with CRPC, galeterone was well tolerated at 2550 mg/day administered orally. Galeterone treatment decreased serum testosterone without an increase in DOC or hypertension and hypokalemia characteristic of abiraterone treatment, suggesting preferential inhibition of 17,20-lyase activity [106].
VT-464 is a CYP17A1 inhibitor that has been shown to preferentially inhibit 17,20-lyase activity in preclinical models. VT-464 was rationally designed to both inhibit CYP17A1 and antagonize AR [107]. Orteronel (TAK-700) is another purported 17,20-lyase-specific CYP17A1 inhibitor that underwent clinical testing. Preclinical studies of orteronel demonstrated a 5.4-times greater potency for 17,20-lyase activity compared to the 17α-hydroxylase activity in cell-free assays; however, circulating progesterone concentrations rose in monkeys treated with orteronel, consistent with significant 17α-hydroxylase inhibitory activity [108]. In phase III clinical testing, orteronel plus prednisone failed to prolong overall survival compared to placebo plus prednisone in men with CRPC who failed docetaxel chemotherapy [109].
The 5α-reductase inhibitors finasteride and dutasteride reduce the conversion of testosterone to the DHT, which is five times more potent than testosterone as an AR agonist. Finasteride is selective for SRD5A2, but dutasteride inhibits both SRD5A1 and SRD5A2. The Prostate Cancer Prevention Trial (PCPT) aimed to determine the effectiveness of prophylactic SRD5A2 inhibition at preventing or delaying the onset of prostate cancer [110]. The study results showed that finasteride blocked DHT synthesis and demonstrated a 24.8 % reduction in prostate cancer prevalence over the 7 years of treatment; however, the risk for high-grade tumors increased to 6.4 % in finasteride-treated men compared to 5.1 % in the placebo group [110]. These risks have outweighed any potential benefit of using 5α-reductase inhibitors for prostate cancer prevention or treatment.
Alternative Agonists, Estrogen Receptor Mutations, and Androgens in Breast Cancer
Despite the high initial response rate to tamoxifen and AI therapies, breast cancer recurrence still poses a major treatment hurdle for women already treated with hormonal therapy. One possible mechanism of tumor recurrence and drug resistance is alternative pathways to steroid synthesis and non-canonical endogenous ER ligands. One example of such a ligand is the androgen metabolite 5α-androstane-3β,17β-diol (3βAdiol). Sikora et al. showed that 3βAdiol binds to and activates ER, and this binding can be blocked with the pure antiestrogen fulvestrant [111].
Another example of an endogenous ligand with estrogenic properties is 27-hydroxycholesterol (27HC). The oxysterol 27HC is synthesized from cholesterol by the cytochrome P450 27A1 (CYP27A1) enzyme [112]. 27HC was first shown to exhibit SERM properties in the cardiovascular system where it antagonized the cardioprotective effects of estrogen in smooth muscle and endothelial cells using mouse and rat models [113]. Dusell et al. later characterized 27HC’s agonist activity in the ER-positive breast cancer cell line MCF-7 and showed that 1 μM 27HC induced expression of ER-regulated genes, while 100-nM fulvestrant blocked this induction. Additionally, 27HC treatment in MCF-7 cells resulted in a dose-dependent increase in cell number [114]. Such findings illustrate the potential impact alternative endogenous steroid-receptor ligands can have on disease progression and therapy response.
Mutations in the ligand-binding domain of ER have also been recently identified [115, 116]. Of interest, these mutations seem to be significantly more frequent in women that have been treated with AIs, suggesting that estrogen deprivation selected for cells bearing these mutations. Consistent with this model, preclinical data suggest that these patients might still respond to direct ER antagonists [116]; however, this strategy has not been validated in appropriate clinical trials.
During AI treatment, local conversion of androgens to estrogens is impaired, therefore leading to accumulation of androgens. Consequently, another plausible mechanism of resistance to AI therapy is the acquired expression of AR and an active signaling pathway. Indeed, AR expression in breast cancers has been recognized for some time, and recent evidence suggests that AR expression is increased during AI treatment [117], with increases in circulating androgens also detected [118]. Because abiraterone acts upstream of aromatase and blocks the production of androgen precursors, CYP17A1 inhibition has been tested for the management of ER-positive breast cancer. The first clinical trial testing abiraterone in breast cancer patients compared the efficacy of abiraterone plus prednisone to the AI exemestane, alone or in combination [119]. The patient population for this study was women with metastatic, ER-positive breast cancer, who had failed previous endocrine therapies. The trial’s pharmacodynamic endpoints showed that abiraterone use successfully suppressed both circulating androgen and estrogen concentrations; however, this reduction in circulating sex steroids did not translate into significant clinical benefit. Progression-free survival in the three treatment arms was similar, 3.7, 3.7, and 4.5 months in exemestate, abiraterone, and abiraterone-plus-exemestane arms, respectively [119]. A limitation of this study is that only heavily pretreated patients with advanced tumors were randomized, raising the possibility that they were unlikely to respond to any form of treatment. Indeed, these data are consistent with studies on alternative growth signaling pathways beyond AR and ER, which are not targeted with a CYP17A1 inhibitor like abiraterone and might be active in some breast cancers [120].
A subset of triple-negative breast cancers (TNBCs) expresses AR, and these tumors are believed to be androgen-dependent [121]. TNBCs, which account for approximately 10 % of all breast cancers, are characterized by lacking expression of ER, progesterone receptor, and the receptor tyrosine-protein kinase erbB-2, also known as HER2 [122, 123]. These tumors have historically been harder to treat, due to limited options for targeted treatment [124]. Antiandrogen therapies commonly used to treat CRPC have been investigated recently in AR-positive TNBC, based on the hypothesis that these tumors are dependent upon AR for cellular growth. One study using a mouse xenograft model of AR-positive TNBC cells demonstrated that these tumors were sensitive to bicalutamide treatment [121]. A clinical case study highlighted the potential success of bicalutamide as an option for TNBC with intact AR signaling pathways. A 55-year-old woman with metastatic AR-positive TNBC exhibited a complete response to bicalutamide despite disease progression on all previous forms of chemotherapy [125]. A phase II trial testing bicalutamide in women with AR-positive, ER-negative metastatic breast cancer showed promising but modest activity. Of the 26 patients evaluated for the primary endpoint, 5 exhibited evidence of stable disease translating to a clinical benefit rate of 19 % [126]. Similar preclinical data have been reported using the more potent antiandrogen, enzalutamide [127]. Preliminary analysis from a phase II trial assessing enzalutamide therapy in advanced AR-positive breast cancer suggests that patients with tumors characterized by androgen-driven gene signatures display a robust response to enzalutamide, as evidenced by a significant increase in progression-free survival compared to patients with tumors lacking this gene signature [128]. These studies demonstrate that AR is a viable target in AR-positive breast cancers that rely on AR-mediated signaling for growth.
Limitations to Current Knowledge and Future Directions
Because metastatic breast and prostate cancers are treated medically and not surgically, few studies have been able to obtain adequate amounts of metastatic tumor specimens for detailed biological studies. Among the critical questions that need to be answered include: What are the intracellular concentrations of androgens and estrogens in these tumors? To what extent do CYP17A1 inhibitors and AIs inhibit the targeted enzymatic steps in these cells, and how much precursors accumulate? Do the tumors use precursor steroids and metabolites as agonists for driving growth pathways? What is the level of AR and ER activation in the presence of antagonists? These studies require informed consent for invasive procedures that are not standard of care, and informative studies with limited samples require sophisticated and sensitive analytical techniques. The use of tandem mass spectrometry (LC-MS/MS) allows for sensitive and specific measurement of multiple steroids in a single sample [129], but current assays struggle to meet performance characteristics necessary for some analytes, particularly E2 and DHT. An alternative source of metastatic tumor cells is the harvesting of circulating tumor cells (CTCs), but current methods yield insufficient cells for most experiments using even the most sensitive analytical techniques, except for DNA and RNA analyses.
Acknowledgments
This work was supported by grants from the Breast Cancer Research Foundation (BCRF) (N003173 to JMR) and the National Institutes of Health (R01GM099143 to JMR, R01GM086596 to RJA, and R01CA168899 [RJA subcontract from Dr. Nima Sharifi, PI]). We wish to thank Kayla Capper for helping to design figure illustrations.
Compliance with Ethical Standards
Disclosure Statement
R.J.A. reports consulting fees from Janssen Pharmaceuticals, Tokai Pharmaceuticals, and Innocrin Pharmaceuticals.
References
- 1.Miller WL, Auchus RJ. The molecular biology, biochemistry, and physiology of human steroidogenesis and its disorders. Endocr Rev. 2011;32:81–151. doi: 10.1210/er.2010-0013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.American Cancer Society. Cancer facts & figures, 2015. Atlanta
- 3.Vale W, Spiess J, Rivier C, Rivier J. Characterization of a 41-residue ovine hypothalamic peptide that stimulates secretion of corticotropin and beta-endorphin. Science. 1981;213:1394–1397. doi: 10.1126/science.6267699. [DOI] [PubMed] [Google Scholar]
- 4.Rivier C, Vale W. Modulation of stress-induced ACTH release by corticotropin-releasing factor, catecholamines and vasopressin. Nature. 1983;305:325–327. doi: 10.1038/305325a0. [DOI] [PubMed] [Google Scholar]
- 5.Catalano RD, Stuve L, Ramachandran J. Characterization of corticotropin receptors in human adrenocortical cells. J Clin Endocrinol Metab. 1986;62:300–304. doi: 10.1210/jcem-62-2-300. [DOI] [PubMed] [Google Scholar]
- 6.Endoh A, Kristiansen SB, Casson PR, Buster JE, Hornsby PJ. The zona reticularis is the site of biosynthesis of dehydroepiandrosterone and dehydroepiandrosterone sulfate in the adult human adrenal cortex resulting from its low expression of 3 beta-hydroxysteroid dehydrogenase. J Clin Endocrinol Metab. 1996;81:3558–3565. doi: 10.1210/jcem.81.10.8855801. [DOI] [PubMed] [Google Scholar]
- 7.Dillard PR, Lin MF, Khan SA. Androgen-independent prostate cancer cells acquire the complete steroidogenic potential of synthesizing testosterone from cholesterol. Mol Cell Endocrinol. 2008;295:115–120. doi: 10.1016/j.mce.2008.08.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Orentreich N, Brind JL, Rizer RL, Vogelman JH. Age changes and sex differences in serum dehydroepiandrosterone sulfate concentrations throughout adulthood. J Clin Endocrinol Metab. 1984;59:551–555. doi: 10.1210/jcem-59-3-551. [DOI] [PubMed] [Google Scholar]
- 9.Rheaume E, Lachance Y, Zhao HF, Breton N, Dumont M, de Launoit Y, Trudel C, Luu-The V, Simard J, Labrie F. Structure and expression of a new complementary DNA encoding the almost exclusive 3 beta-hydroxysteroid dehydrogenase/delta 5-delta 4-isomerase in human adrenals and gonads. Mol Endocrinol. 1991;5:1147–1157. doi: 10.1210/mend-5-8-1147. [DOI] [PubMed] [Google Scholar]
- 10.Picado-Leonard J, Miller WL. Cloning and sequence of the human gene for P450c17 (steroid 17 alpha-hydroxylase/17,20 lyase): similarity with the gene for P450c21. DNA. 1987;6:439–448. doi: 10.1089/dna.1987.6.439. [DOI] [PubMed] [Google Scholar]
- 11.Chung BC, Picado-Leonard J, Haniu M, Bienkowski M, Hall PF, Shively JE, Miller WL. Cytochrome P450c17 (steroid 17 alpha-hydroxylase/17,20 lyase): cloning of human adrenal and testis cDNAs indicates the same gene is expressed in both tissues. Proc Natl Acad Sci U S A. 1987;84:407–411. doi: 10.1073/pnas.84.2.407. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Matteson KJ, Picado-Leonard J, Chung BC, Mohandas TK, Miller WL. Assignment of the gene for adrenal P450c17 (steroid 17 alpha-hydroxylase/17,20 lyase) to human chromosome 10. J Clin Endocrinol Metab. 1986;63:789–791. doi: 10.1210/jcem-63-3-789. [DOI] [PubMed] [Google Scholar]
- 13.Auchus RJ, Lee TC, Miller WL. Cytochrome b5 augments the 17,20-lyase activity of human P450c17 without direct electron transfer. J Biol Chem. 1998;273:3158–3165. doi: 10.1074/jbc.273.6.3158. [DOI] [PubMed] [Google Scholar]
- 14.Geller DH, Auchus RJ, Miller WL. P450c17 mutations R347H and R358Q selectively disrupt 17,20-lyase activity by disrupting interactions with P450 oxidoreductase and cytochrome b5. Mol Endocrinol. 1999;13:167–175. doi: 10.1210/mend.13.1.0219. [DOI] [PubMed] [Google Scholar]
- 15.Kominami S, Ogawa N, Morimune R, De-Ying H, Takemori S. The role of cytochrome b5 in adrenal microsomal steroidogenesis. J Steroid Biochem Mol Biol. 1992;42:57–64. doi: 10.1016/0960-0760(92)90011-7. [DOI] [PubMed] [Google Scholar]
- 16.Katagiri M, Kagawa N, Waterman MR. The role of cytochrome b5 in the biosynthesis of androgens by human P450c17. Arch Biochem Biophys. 1995;317:343–347. doi: 10.1006/abbi.1995.1173. [DOI] [PubMed] [Google Scholar]
- 17.Nakamura Y, Hornsby PJ, Casson P, Morimoto R, Satoh F, Xing Y, Kennedy MR, Sasano H, Rainey WE. Type 5 17beta-hydroxysteroid dehydrogenase (AKR1C3) contributes to testosterone production in the adrenal reticularis. J Clin Endocrinol Metab. 2009;94:2192–2198. doi: 10.1210/jc.2008-2374. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Belchetz PE, Plant TM, Nakai Y, Keogh EJ, Knobil E. Hypophysial responses to continuous and intermittent delivery of hypothalamic gonadotropin-releasing hormone. Science. 1978;202:631–633. doi: 10.1126/science.100883. [DOI] [PubMed] [Google Scholar]
- 19.Simpson ER, Mahendroo MS, Means GD, Kilgore MW, Hinshelwood MM, Graham-Lorence S, Amarneh B, Ito Y, Fisher CR, Michael MD, et al. Aromatase cytochrome P450, the enzyme responsible for estrogen biosynthesis. Endocr Rev. 1994;15:342–355. doi: 10.1210/edrv-15-3-342. [DOI] [PubMed] [Google Scholar]
- 20.Luu The V, Labrie C, Zhao HF, Couet J, Lachance Y, Simard J, Leblanc G, Cote J, Berube D, Gagne R, et al. Characterization of cDNAs for human estradiol 17 beta-dehydrogenase and assignment of the gene to chromosome 17: evidence of two mRNA species with distinct 5′-termini in human placenta. Mol Endocrinol. 1989;3:1301–1309. doi: 10.1210/mend-3-8-1301. [DOI] [PubMed] [Google Scholar]
- 21.Santen RJ. Feedback control of luteinizing hormone and follicle-stimulating hormone secretion by testosterone and estradiol in men: physiological and clinical implications. Clin Biochem. 1981;14:243–251. doi: 10.1016/S0009-9120(81)90964-4. [DOI] [PubMed] [Google Scholar]
- 22.Meachem SJ, Nieschlag E, Simoni M. Inhibin B in male reproduction: pathophysiology and clinical relevance. Eur J Endocrinol. 2001;145:561–571. doi: 10.1530/eje.0.1450561. [DOI] [PubMed] [Google Scholar]
- 23.Sasano H, Okamoto M, Mason JI, Simpson ER, Mendelson CR, Sasano N, Silverberg SG. Immunolocalization of aromatase, 17 alpha-hydroxylase and side-chain-cleavage cytochromes P-450 in the human ovary. J Reprod Fertil. 1989;85:163–169. doi: 10.1530/jrf.0.0850163. [DOI] [PubMed] [Google Scholar]
- 24.Titus MA, Schell MJ, Lih FB, Tomer KB, Mohler JL. Testosterone and dihydrotestosterone tissue levels in recurrent prostate cancer. Clin Cancer Res. 2005;11:4653–4657. doi: 10.1158/1078-0432.CCR-05-0525. [DOI] [PubMed] [Google Scholar]
- 25.Santen RJ, Santner S, Davis B, Veldhuis J, Samojlik E, Ruby E. Aminoglutethimide inhibits extraglandular estrogen production in postmenopausal women with breast carcinoma. J Clin Endocrinol Metab. 1978;47:1257–1265. doi: 10.1210/jcem-47-6-1257. [DOI] [PubMed] [Google Scholar]
- 26.Dumont M, Luu-The V, Dupont E, Pelletier G, Labrie F. Characterization, expression, and immunohistochemical localization of 3 beta-hydroxysteroid dehydrogenase/delta 5-delta 4 isomerase in human skin. J Invest Dermatol. 1992;99:415–421. doi: 10.1111/1523-1747.ep12616131. [DOI] [PubMed] [Google Scholar]
- 27.Mindnich R, Moller G, Adamski J. The role of 17 beta-hydroxysteroid dehydrogenases. Mol Cell Endocrinol. 2004;218:7–20. doi: 10.1016/j.mce.2003.12.006. [DOI] [PubMed] [Google Scholar]
- 28.Russell DW, Wilson JD. Steroid 5 alpha-reductase: two genes/two enzymes. Annu Rev Biochem. 1994;63:25–61. doi: 10.1146/annurev.bi.63.070194.000325. [DOI] [PubMed] [Google Scholar]
- 29.Judd HL, Judd GE, Lucas WE, Yen SS. Endocrine function of the postmenopausal ovary: concentration of androgens and estrogens in ovarian and peripheral vein blood. J Clin Endocrinol Metab. 1974;39:1020–1024. doi: 10.1210/jcem-39-6-1020. [DOI] [PubMed] [Google Scholar]
- 30.Grodin JM, Siiteri PK, MacDonald PC. Source of estrogen production in postmenopausal women. J Clin Endocrinol Metab. 1973;36:207–214. doi: 10.1210/jcem-36-2-207. [DOI] [PubMed] [Google Scholar]
- 31.Mangelsdorf DJ, Thummel C, Beato M, Herrlich P, Schutz G, Umesono K, Blumberg B, Kastner P, Mark M, Chambon P, Evans RM. The nuclear receptor superfamily: the second decade. Cell. 1995;83:835–839. doi: 10.1016/0092-8674(95)90199-X. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Smith DF, Toft DO. Steroid receptors and their associated proteins. Mol Endocrinol. 1993;7:4–11. doi: 10.1210/mend.7.1.8446107. [DOI] [PubMed] [Google Scholar]
- 33.Wurtz JM, Bourguet W, Renaud JP, Vivat V, Chambon P, Moras D, Gronemeyer H. A canonical structure for the ligand-binding domain of nuclear receptors. Nat Struct Biol. 1996;3:87–94. doi: 10.1038/nsb0196-87. [DOI] [PubMed] [Google Scholar]
- 34.Gronemeyer H, Gustafsson JA, Laudet V. Principles for modulation of the nuclear receptor superfamily. Nat Rev Drug Discov. 2004;3:950–964. doi: 10.1038/nrd1551. [DOI] [PubMed] [Google Scholar]
- 35.O’Malley BW, Kumar R. Nuclear receptor coregulators in cancer biology. Cancer Res. 2009;69:8217–8222. doi: 10.1158/0008-5472.CAN-09-2223. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Beatson G. On the treatment of inoperable cases of carcinoma of the mamma: suggestions for a new method of treatment, with illustrative cases. Lancet. 1896;148:162–165. doi: 10.1016/S0140-6736(01)72384-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Early Breast Cancer Trialists’ Collaborative Group. Dowsett M, Forbes JF, Bradley R, Ingle J, Aihara T, Bliss J, Boccardo F, Coates A, Coombes RC, Cuzick J, Dubsky P, Gnant M, Kaufmann M, Kilburn L, Perrone F, Rea D, Thurlimann B, van de Velde C, Pan H, Peto R, Davies C, Gray R. Aromatase inhibitors versus tamoxifen in early breast cancer: patient-level meta-analysis of the randomised trials. Lancet. 2015;386:1341–1352. doi: 10.1016/S0140-6736(15)61074-1. [DOI] [PubMed] [Google Scholar]
- 38.Ariazi EA, Ariazi JL, Cordera F, Jordan VC. Estrogen receptors as therapeutic targets in breast cancer. Curr Top Med Chem. 2006;6:181–202. doi: 10.2174/156802606776173483. [DOI] [PubMed] [Google Scholar]
- 39.Winer EP, Hudis C, Burstein HJ, Wolff AC, Pritchard KI, Ingle JN, Chlebowski RT, Gelber R, Edge SB, Gralow J, Cobleigh MA, Mamounas EP, Goldstein LJ, Whelan TJ, Powles TJ, Bryant J, Perkins C, Perotti J, Braun S, Langer AS, Browman GP, Somerfield MR. American Society of Clinical Oncology technology assessment on the use of aromatase inhibitors as adjuvant therapy for postmenopausal women with hormone receptor-positive breast cancer: status report 2004. J Clin Oncol. 2005;23:619–629. doi: 10.1200/JCO.2005.09.121. [DOI] [PubMed] [Google Scholar]
- 40.Osborne CK. Tamoxifen in the treatment of breast cancer. N Engl J Med. 1998;339:1609–1618. doi: 10.1056/NEJM199811263392207. [DOI] [PubMed] [Google Scholar]
- 41.Dowsett M, Cuzick J, Ingle J, Coates A, Forbes J, Bliss J, Buyse M, Baum M, Buzdar A, Colleoni M, Coombes C, Snowdon C, Gnant M, Jakesz R, Kaufmann M, Boccardo F, Godwin J, Davies C, Peto R. Meta-analysis of breast cancer outcomes in adjuvant trials of aromatase inhibitors versus tamoxifen. J Clin Oncol. 2010;28:509–518. doi: 10.1200/JCO.2009.23.1274. [DOI] [PubMed] [Google Scholar]
- 42.Coates AS, Winer EP, Goldhirsch A, Gelber RD, Gnant M, Piccart-Gebhart M, Thurlimann B, Senn HJ, Panel Members Tailoring therapies-improving the management of early breast cancer: St Gallen International Expert Consensus on the Primary Therapy of Early Breast Cancer 2015. Ann Oncol. 2015;26:1533–1546. doi: 10.1093/annonc/mdv221. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Lippman M, Bolan G, Huff K. The effects of estrogens and antiestrogens on hormone-responsive human breast cancer in long-term tissue culture. Cancer Res. 1976;36:4595–4601. [PubMed] [Google Scholar]
- 44.Lippman ME, Bolan G. Oestrogen-responsive human breast cancer in long term tissue culture. Nature. 1975;256:592–593. doi: 10.1038/256592a0. [DOI] [PubMed] [Google Scholar]
- 45.Wells SA, Jr, Santen RJ, Lipton A, Haagensen DE, Jr, Ruby EJ, Harvey H, Dilley WG. Medical adrenalectomy with aminoglutethimide: clinical studies in postmenopausal patients with metastatic breast carcinoma. Ann Surg. 1978;187:475–484. doi: 10.1097/00000658-197805000-00004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Santen RJ, Brodie H, Simpson ER, Siiteri PK, Brodie A. History of aromatase: saga of an important biological mediator and therapeutic target. Endocr Rev. 2009;30:343–375. doi: 10.1210/er.2008-0016. [DOI] [PubMed] [Google Scholar]
- 47.Bulun SE, Simpson ER. Competitive reverse transcription-polymerase chain reaction analysis indicates that levels of aromatase cytochrome P450 transcripts in adipose tissue of buttocks, thighs, and abdomen of women increase with advancing age. J Clin Endocrinol Metab. 1994;78:428–432. doi: 10.1210/jcem.78.2.8106632. [DOI] [PubMed] [Google Scholar]
- 48.Chetrite GS, Cortes-Prieto J, Philippe JC, Wright F, Pasqualini JR. Comparison of estrogen concentrations, estrone sulfatase and aromatase activities in normal, and in cancerous, human breast tissues. J Steroid Biochem Mol Biol. 2000;72:23–27. doi: 10.1016/S0960-0760(00)00040-6. [DOI] [PubMed] [Google Scholar]
- 49.Santner SJ, Feil PD, Santen RJ. In situ estrogen production via the estrone sulfatase pathway in breast tumors: relative importance versus the aromatase pathway. J Clin Endocrinol Metab. 1984;59:29–33. doi: 10.1210/jcem-59-1-29. [DOI] [PubMed] [Google Scholar]
- 50.Labrie F, Luu-The V, Lin SX, Simard J, Labrie C, El-Alfy M, Pelletier G, Belanger A. Intracrinology: role of the family of 17 beta-hydroxysteroid dehydrogenases in human physiology and disease. J Mol Endocrinol. 2000;25:1–16. doi: 10.1677/jme.0.0250001. [DOI] [PubMed] [Google Scholar]
- 51.Key T, Appleby P, Barnes I, Reeves G, Endogenous H, Breast Cancer Collaborative Group Endogenous sex hormones and breast cancer in postmenopausal women: reanalysis of nine prospective studies. J Natl Cancer Inst. 2002;94:606–616. doi: 10.1093/jnci/94.8.606. [DOI] [PubMed] [Google Scholar]
- 52.Jordan VC, Murphy CS. Endocrine pharmacology of antiestrogens as antitumor agents. Endocr Rev. 1990;11:578–610. doi: 10.1210/edrv-11-4-578. [DOI] [PubMed] [Google Scholar]
- 53.Hall JM, McDonnell DP. Coregulators in nuclear estrogen receptor action: from concept to therapeutic targeting. Mol Interv. 2005;5:343–357. doi: 10.1124/mi.5.6.7. [DOI] [PubMed] [Google Scholar]
- 54.Beekman JM, Allan GF, Tsai SY, Tsai MJ, O’Malley BW. Transcriptional activation by the estrogen receptor requires a conformational change in the ligand binding domain. Mol Endocrinol. 1993;7:1266–1274. doi: 10.1210/mend.7.10.8264659. [DOI] [PubMed] [Google Scholar]
- 55.Paige LA, Christensen DJ, Gron H, Norris JD, Gottlin EB, Padilla KM, Chang CY, Ballas LM, Hamilton PT, McDonnell DP, Fowlkes DM. Estrogen receptor (ER) modulators each induce distinct conformational changes in ER alpha and ER beta. Proc Natl Acad Sci U S A. 1999;96:3999–4004. doi: 10.1073/pnas.96.7.3999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Fisher B, Costantino JP, Wickerham DL, Redmond CK, Kavanah M, Cronin WM, Vogel V, Robidoux A, Dimitrov N, Atkins J, Daly M, Wieand S, Tan-Chiu E, Ford L, Wolmark N. Tamoxifen for prevention of breast cancer: report of the National Surgical Adjuvant Breast and Bowel Project P-1 Study. J Natl Cancer Inst. 1998;90:1371–1388. doi: 10.1093/jnci/90.18.1371. [DOI] [PubMed] [Google Scholar]
- 57.Stearns V, Johnson MD, Rae JM, Morocho A, Novielli A, Bhargava P, Hayes DF, Desta Z, Flockhart DA. Active tamoxifen metabolite plasma concentrations after coadministration of tamoxifen and the selective serotonin reuptake inhibitor paroxetine. J Natl Cancer Inst. 2003;95:1758–1764. doi: 10.1093/jnci/djg108. [DOI] [PubMed] [Google Scholar]
- 58.Johnson MD, Zuo H, Lee KH, Trebley JP, Rae JM, Weatherman RV, Desta Z, Flockhart DA, Skaar TC. Pharmacological characterization of 4-hydroxy-N-desmethyl tamoxifen, a novel active metabolite of tamoxifen. Breast Cancer Res Treat. 2004;85:151–159. doi: 10.1023/B:BREA.0000025406.31193.e8. [DOI] [PubMed] [Google Scholar]
- 59.Love RR, Mazess RB, Barden HS, Epstein S, Newcomb PA, Jordan VC, Carbone PP, DeMets DL. Effects of tamoxifen on bone mineral density in postmenopausal women with breast cancer. N Engl J Med. 1992;326:852–856. doi: 10.1056/NEJM199203263261302. [DOI] [PubMed] [Google Scholar]
- 60.Gail MH, Costantino JP, Bryant J, Croyle R, Freedman L, Helzlsouer K, Vogel V. Weighing the risks and benefits of tamoxifen treatment for preventing breast cancer. J Natl Cancer Inst. 1999;91:1829–1846. doi: 10.1093/jnci/91.21.1829. [DOI] [PubMed] [Google Scholar]
- 61.Nicholson RI, Gee JM, Manning DL, Wakeling AE, Montano MM, Katzenellenbogen BS. Responses to pure antiestrogens (ICI 164384, ICI 182780) in estrogen-sensitive and -resistant experimental and clinical breast cancer. Ann N Y Acad Sci. 1995;761:148–163. doi: 10.1111/j.1749-6632.1995.tb31376.x. [DOI] [PubMed] [Google Scholar]
- 62.Lai A, Kahraman M, Govek S, Nagasawa J, Bonnefous C, Julien J, Douglas K, Sensintaffar J, Lu N, Lee KJ, Aparicio A, Kaufman J, Qian J, Shao G, Prudente R, Moon MJ, Joseph JD, Darimont B, Brigham D, Grillot K, Heyman R, Rix PJ, Hager JH, Smith ND. Identification of GDC-0810 (ARN-810), an orally bioavailable selective estrogen receptor degrader (SERD) that demonstrates robust activity in tamoxifen-resistant breast cancer xenografts. J Med Chem. 2015;58:4888–4904. doi: 10.1021/acs.jmedchem.5b00054. [DOI] [PubMed] [Google Scholar]
- 63.Smith IE, Dowsett M. Aromatase inhibitors in breast cancer. N Engl J Med. 2003;348:2431–2442. doi: 10.1056/NEJMra023246. [DOI] [PubMed] [Google Scholar]
- 64.Smith IE, Harris AL, Morgan M, Ford HT, Gazet JC, Harmer CL, White H, Parsons CA, Villardo A, Walsh G, McKinna JA. Tamoxifen versus aminoglutethimide in advanced breast carcinoma: a randomized cross-over trial. Br Med J (Clin Res Ed) 1981;283:1432–1434. doi: 10.1136/bmj.283.6304.1432. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Covey DF, Hood WF, Parikh VD. 10 beta-propynyl-substituted steroids. Mechanism-based enzyme-activated irreversible inhibitors of estrogen biosynthesis. J Biol Chem. 1981;256:1076–1079. [PubMed] [Google Scholar]
- 66.Siiteri PK, Thompson EA. Studies of human placental aromatase. J Steroid Biochem. 1975;6:317–322. doi: 10.1016/0022-4731(75)90149-1. [DOI] [PubMed] [Google Scholar]
- 67.Brodie AM, Schwarzel WC, Shaikh AA, Brodie HJ. The effect of an aromatase inhibitor, 4-hydroxy-4-androstene-3,17-dione, on estrogen-dependent processes in reproduction and breast cancer. Endocrinology. 1977;100:1684–1695. doi: 10.1210/endo-100-6-1684. [DOI] [PubMed] [Google Scholar]
- 68.Howell A, Cuzick J, Baum M, Buzdar A, Dowsett M, Forbes JF, Hoctin-Boes G, Houghton J, Locker GY, Tobias JS, ATAC Trialists’ Group Results of the ATAC (Arimidex, Tamoxifen, Alone or in Combination) trial after completion of 5 years’ adjuvant treatment for breast cancer. Lancet. 2005;365:60–62. doi: 10.1016/S0140-6736(05)74803-0. [DOI] [PubMed] [Google Scholar]
- 69.Breast International Group 1–98 Collaborative Group. Thurlimann B, Keshaviah A, Coates AS, Mouridsen H, Mauriac L, Forbes JF, Paridaens R, Castiglione-Gertsch M, Gelber RD, Rabaglio M, Smith I, Wardley A, Price KN, Goldhirsch A. A comparison of letrozole and tamoxifen in postmenopausal women with early breast cancer. N Engl J Med. 2005;353:2747–2757. doi: 10.1056/NEJMoa052258. [DOI] [PubMed] [Google Scholar]
- 70.Griffin JE. Androgen resistance—the clinical and molecular spectrum. N Engl J Med. 1992;326:611–618. doi: 10.1056/NEJM199202273260906. [DOI] [PubMed] [Google Scholar]
- 71.Huggins C. Effect of orchiectomy and irradiation on cancer of the prostate. Ann Surg. 1942;115:1192–1200. doi: 10.1097/00000658-194206000-00030. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Huggins C, Hodges CV. Studies on prostatic cancer. I. The effect of castration, of estrogen and androgen injection on serum phosphatases in metastatic carcinoma of the prostate. Cancer Res. 1941;22:232–240. doi: 10.3322/canjclin.22.4.232. [DOI] [PubMed] [Google Scholar]
- 73.Mohler JL, Gregory CW, Ford OH, 3rd, Kim D, Weaver CM, Petrusz P, Wilson EM, French FS. The androgen axis in recurrent prostate cancer. Clin Cancer Res. 2004;10:440–448. doi: 10.1158/1078-0432.CCR-1146-03. [DOI] [PubMed] [Google Scholar]
- 74.Scher HI, Sawyers CL. Biology of progressive, castration-resistant prostate cancer: directed therapies targeting the androgen-receptor signaling axis. J Clin Oncol. 2005;23:8253–8261. doi: 10.1200/JCO.2005.03.4777. [DOI] [PubMed] [Google Scholar]
- 75.Visakorpi T, Hyytinen E, Koivisto P, Tanner M, Keinanen R, Palmberg C, Palotie A, Tammela T, Isola J, Kallioniemi OP. In vivo amplification of the androgen receptor gene and progression of human prostate cancer. Nat Genet. 1995;9:401–406. doi: 10.1038/ng0495-401. [DOI] [PubMed] [Google Scholar]
- 76.Veldscholte J, Ris-Stalpers C, Kuiper GG, Jenster G, Berrevoets C, Claassen E, van Rooij HC, Trapman J, Brinkmann AO, Mulder E. A mutation in the ligand binding domain of the androgen receptor of human LNCaP cells affects steroid binding characteristics and response to anti-androgens. Biochem Biophys Res Commun. 1990;173:534–540. doi: 10.1016/S0006-291X(05)80067-1. [DOI] [PubMed] [Google Scholar]
- 77.Chang KH, Li R, Kuri B, Lotan Y, Roehrborn CG, Liu J, Vessella R, Nelson PS, Kapur P, Guo X, Mirzaei H, Auchus RJ, Sharifi N. A gain-of-function mutation in DHT synthesis in castration-resistant prostate cancer. Cell. 2013;154:1074–1084. doi: 10.1016/j.cell.2013.07.029. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Belanger B, Belanger A, Labrie F, Dupont A, Cusan L, Monfette G. Comparison of residual C-19 steroids in plasma and prostatic tissue of human, rat and guinea pig after castration: unique importance of extratesticular androgens in men. J Steroid Biochem. 1989;32:695–698. doi: 10.1016/0022-4731(89)90514-1. [DOI] [PubMed] [Google Scholar]
- 79.Evaul K, Li R, Papari-Zareei M, Auchus RJ, Sharifi N. 3beta-hydroxysteroid dehydrogenase is a possible pharmacological target in the treatment of castration-resistant prostate cancer. Endocrinology. 2010;151:3514–3520. doi: 10.1210/en.2010-0138. [DOI] [PubMed] [Google Scholar]
- 80.Li R, Evaul K, Sharma KK, Chang KH, Yoshimoto J, Liu J, Auchus RJ, Sharifi N. Abiraterone inhibits 3beta-hydroxysteroid dehydrogenase: a rationale for increasing drug exposure in castration-resistant prostate cancer. Clin Cancer Res. 2012;18:3571–3579. doi: 10.1158/1078-0432.CCR-12-0908. [DOI] [PubMed] [Google Scholar]
- 81.Chang KH, Li R, Papari-Zareei M, Watumull L, Zhao YD, Auchus RJ, Sharifi N. Dihydrotestosterone synthesis bypasses testosterone to drive castration-resistant prostate cancer. Proc Natl Acad Sci U S A. 2011;108:13728–13733. doi: 10.1073/pnas.1107898108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Wilson JD, Auchus RJ, Leihy MW, Guryev OL, Estabrook RW, Osborn SM, Shaw G, Renfree MB. 5alpha-androstane-3alpha,17beta-diol is formed in tammar wallaby pouch young testes by a pathway involving 5alpha-pregnane-3alpha,17alpha-diol-20-one as a key intermediate. Endocrinology. 2003;144:575–580. doi: 10.1210/en.2002-220721. [DOI] [PubMed] [Google Scholar]
- 83.Auchus RJ. The backdoor pathway to dihydrotestosterone. Trends Endocrinol Metab. 2004;15:432–438. doi: 10.1016/S1043-2760(04)00214-0. [DOI] [PubMed] [Google Scholar]
- 84.Montgomery RB, Mostaghel EA, Vessella R, Hess DL, Kalhorn TF, Higano CS, True LD, Nelson PS. Maintenance of intratumoral androgens in metastatic prostate cancer: a mechanism for castration-resistant tumor growth. Cancer Res. 2008;68:4447–4454. doi: 10.1158/0008-5472.CAN-08-0249. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Eisenberger MA, O’Dwyer PJ, Friedman MA. Gonadotropin hormone-releasing hormone analogues: a new therapeutic approach for prostatic carcinoma. J Clin Oncol. 1986;4:414–424. doi: 10.1200/JCO.1986.4.3.414. [DOI] [PubMed] [Google Scholar]
- 86.Pagani O, Regan MM, Walley BA, Fleming GF, Colleoni M, Lang I, Gomez HL, Tondini C, Burstein HJ, Perez EA, Ciruelos E, Stearns V, Bonnefoi HR, Martino S, Geyer CE, Jr, Pinotti G, Puglisi F, Crivellari D, Ruhstaller T, Winer EP, Rabaglio-Poretti M, Maibach R, Ruepp B, Giobbie-Hurder A, Price KN, Bernhard J, Luo W, Ribi K, Viale G, Coates AS, Gelber RD, Goldhirsch A, Francis PA, TEXT and SOFT Investigators. International Breast Cancer Study Group Adjuvant exemestane with ovarian suppression in premenopausal breast cancer. N Engl J Med. 2014;371:107–118. doi: 10.1056/NEJMoa1404037. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Crawford ED, Eisenberger MA, McLeod DG, Spaulding JT, Benson R, Dorr FA, Blumenstein BA, Davis MA, Goodman PJ. A controlled trial of leuprolide with and without flutamide in prostatic carcinoma. N Engl J Med. 1989;321:419–424. doi: 10.1056/NEJM198908173210702. [DOI] [PubMed] [Google Scholar]
- 88.Masiello D, Cheng S, Bubley GJ, Lu ML, Balk SP. Bicalutamide functions as an androgen receptor antagonist by assembly of a transcriptionally inactive receptor. J Biol Chem. 2002;277:26321–26326. doi: 10.1074/jbc.M203310200. [DOI] [PubMed] [Google Scholar]
- 89.Singh SM, Gauthier S, Labrie F. Androgen receptor antagonists (antiandrogens): structure-activity relationships. Curr Med Chem. 2000;7:211–247. doi: 10.2174/0929867003375371. [DOI] [PubMed] [Google Scholar]
- 90.Tran C, Ouk S, Clegg NJ, Chen Y, Watson PA, Arora V, Wongvipat J, Smith-Jones PM, Yoo D, Kwon A, Wasielewska T, Welsbie D, Chen CD, Higano CS, Beer TM, Hung DT, Scher HI, Jung ME, Sawyers CL. Development of a second-generation antiandrogen for treatment of advanced prostate cancer. Science. 2009;324:787–790. doi: 10.1126/science.1168175. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Scher HI, Fizazi K, Saad F, Taplin ME, Sternberg CN, Miller K, de Wit R, Mulders P, Chi KN, Shore ND, Armstrong AJ, Flaig TW, Flechon A, Mainwaring P, Fleming M, Hainsworth JD, Hirmand M, Selby B, Seely L, de Bono JS, Investigators A. Increased survival with enzalutamide in prostate cancer after chemotherapy. N Engl J Med. 2012;367:1187–1197. doi: 10.1056/NEJMoa1207506. [DOI] [PubMed] [Google Scholar]
- 92.Beer TM, Armstrong AJ, Rathkopf DE, Loriot Y, Sternberg CN, Higano CS, Iversen P, Bhattacharya S, Carles J, Chowdhury S, Davis ID, de Bono JS, Evans CP, Fizazi K, Joshua AM, Kim CS, Kimura G, Mainwaring P, Mansbach H, Miller K, Noonberg SB, Perabo F, Phung D, Saad F, Scher HI, Taplin ME, Venner PM, Tombal B, Investigators P. Enzalutamide in metastatic prostate cancer before chemotherapy. N Engl J Med. 2014;371:424–433. doi: 10.1056/NEJMoa1405095. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Strushkevich N, Usanov SA, Park HW. Structural basis of human CYP51 inhibition by antifungal azoles. J Mol Biol. 2010;397:1067–1078. doi: 10.1016/j.jmb.2010.01.075. [DOI] [PubMed] [Google Scholar]
- 94.Santen RJ, Van den Bossche H, Symoens J, Brugmans J, DeCoster R. Site of action of low dose ketoconazole on androgen biosynthesis in men. J Clin Endocrinol Metab. 1983;57:732–736. doi: 10.1210/jcem-57-4-732. [DOI] [PubMed] [Google Scholar]
- 95.Pont A, Williams PL, Azhar S, Reitz RE, Bochra C, Smith ER, Stevens DA. Ketoconazole blocks testosterone synthesis. Arch Intern Med. 1982;142:2137–2140. doi: 10.1001/archinte.1982.00340250097015. [DOI] [PubMed] [Google Scholar]
- 96.Garrido M, Peng HM, Yoshimoto FK, Upadhyay SK, Bratoeff E, Auchus RJ. A-ring modified steroidal azoles retaining similar potent and slowly reversible CYP17A1 inhibition as abiraterone. J Steroid Biochem Mol Biol. 2014;143:1–10. doi: 10.1016/j.jsbmb.2014.01.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Attard G, Reid AH, Auchus RJ, Hughes BA, Cassidy AM, Thompson E, Oommen NB, Folkerd E, Dowsett M, Arlt W, de Bono JS. Clinical and biochemical consequences of CYP17A1 inhibition with abiraterone given with and without exogenous glucocorticoids in castrate men with advanced prostate cancer. J Clin Endocrinol Metab. 2012;97:507–516. doi: 10.1210/jc.2011-2189. [DOI] [PubMed] [Google Scholar]
- 98.Costa-Santos M, Kater CE, Auchus RJ, Brazilian Congenital Adrenal Hyperplasia Multicenter Study Group Two prevalent CYP17 mutations and genotype-phenotype correlations in 24 Brazilian patients with 17-hydroxylase deficiency. J Clin Endocrinol Metab. 2004;89:49–60. doi: 10.1210/jc.2003-031021. [DOI] [PubMed] [Google Scholar]
- 99.Attard G, Reid AH, Yap TA, Raynaud F, Dowsett M, Settatree S, Barrett M, Parker C, Martins V, Folkerd E, Clark J, Cooper CS, Kaye SB, Dearnaley D, Lee G, de Bono JS. Phase I clinical trial of a selective inhibitor of CYP17, abiraterone acetate, confirms that castration-resistant prostate cancer commonly remains hormone driven. J Clin Oncol. 2008;26:4563–4571. doi: 10.1200/JCO.2007.15.9749. [DOI] [PubMed] [Google Scholar]
- 100.de Bono JS, Logothetis CJ, Molina A, Fizazi K, North S, Chu L, Chi KN, Jones RJ, Goodman OB, Jr, Saad F, Staffurth JN, Mainwaring P, Harland S, Flaig TW, Hutson TE, Cheng T, Patterson H, Hainsworth JD, Ryan CJ, Sternberg CN, Ellard SL, Flechon A, Saleh M, Scholz M, Efstathiou E, Zivi A, Bianchini D, Loriot Y, Chieffo N, Kheoh T, Haqq CM, Scher HI, COU-AA-301 Investigators Abiraterone and increased survival in metastatic prostate cancer. N Engl J Med. 2011;364:1995–2005. doi: 10.1056/NEJMoa1014618. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Ryan CJ, Smith MR, de Bono JS, Molina A, Logothetis CJ, de Souza P, Fizazi K, Mainwaring P, Piulats JM, Ng S, Carles J, Mulders PF, Basch E, Small EJ, Saad F, Schrijvers D, Van Poppel H, Mukherjee SD, Suttmann H, Gerritsen WR, Flaig TW, George DJ, Yu EY, Efstathiou E, Pantuck A, Winquist E, Higano CS, Taplin ME, Park Y, Kheoh T, Griffin T, Scher HI, Rathkopf DE, COU-AA-302 Investigators Abiraterone in metastatic prostate cancer without previous chemotherapy. N Engl J Med. 2013;368:138–148. doi: 10.1056/NEJMoa1209096. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Richards J, Lim AC, Hay CW, Taylor AE, Wingate A, Nowakowska K, Pezaro C, Carreira S, Goodall J, Arlt W, McEwan IJ, de Bono JS, Attard G. Interactions of abiraterone, eplerenone, and prednisolone with wild-type and mutant androgen receptor: a rationale for increasing abiraterone exposure or combining with MDV3100. Cancer Res. 2012;72:2176–2182. doi: 10.1158/0008-5472.CAN-11-3980. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Li Z, Bishop AC, Alyamani M, Garcia JA, Dreicer R, Bunch D, Liu J, Upadhyay SK, Auchus RJ, Sharifi N. Conversion of abiraterone to D4A drives anti-tumour activity in prostate cancer. Nature. 2015;523:347–351. doi: 10.1038/nature14406. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Handratta VD, Vasaitis TS, Njar VC, Gediya LK, Kataria R, Chopra P, Newman D, Jr, Farquhar R, Guo Z, Qiu Y, Brodie AM. Novel C-17-heteroaryl steroidal CYP17 inhibitors/antiandrogens: synthesis, in vitro biological activity, pharmacokinetics, and antitumor activity in the LAPC4 human prostate cancer xenograft model. J Med Chem. 2005;48:2972–2984. doi: 10.1021/jm040202w. [DOI] [PubMed] [Google Scholar]
- 105.Kwegyir-Afful AK, Ramalingam S, Purushottamachar P, Ramamurthy VP, Njar VC. Galeterone and VNPT55 induce proteasomal degradation of AR/AR-V7, induce significant apoptosis via cytochrome c release and suppress growth of castration resistant prostate cancer xenografts in vivo. Oncotarget. 2015;6:27440–27460. doi: 10.18632/oncotarget.4578. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Montgomery B, Eisenberger MA, Rettig MB, Chu FM, Pili R, Stephenson J, Vogelzang NJ, Koletsky AJ, Nordquist LT, Edenfield WJ, Mamlouk K, Ferrante KJ, Taplin ME. Androgen receptor modulation optimized for response (ARMOR) phase I and II studies: galeterone for the treatment of castration-resistant prostate cancer. Clin Cancer Res. 2015 doi: 10.1158/1078-0432.CCR-15-1432. [DOI] [PubMed] [Google Scholar]
- 107.Toren PJ, Kim S, Pham S, Mangalji A, Adomat H, Guns EST, Zoubeidi A, Moore W, Gleave ME. Anticancer activity of a novel selective CYP17A1 inhibitor in preclinical models of castrate-resistant prostate cancer. Mol Cancer Ther. 2015;14:59–69. doi: 10.1158/1535-7163.MCT-14-0521. [DOI] [PubMed] [Google Scholar]
- 108.Yamaoka M, Hara T, Hitaka T, Kaku T, Takeuchi T, Takahashi J, Asahi S, Miki H, Tasaka A, Kusaka M. Orteronel (TAK-700), a novel non-steroidal 17,20-lyase inhibitor: effects on steroid synthesis in human and monkey adrenal cells and serum steroid levels in cynomolgus monkeys. J Steroid Biochem Mol Biol. 2012;129:115–128. doi: 10.1016/j.jsbmb.2012.01.001. [DOI] [PubMed] [Google Scholar]
- 109.Fizazi K, Jones R, Oudard S, Efstathiou E, Saad F, de Wit R, De Bono J, Cruz FM, Fountzilas G, Ulys A, Carcano F, Agarwal N, Agus D, Bellmunt J, Petrylak DP, Lee SY, Webb IJ, Tejura B, Borgstein N, Dreicer R. Phase III, randomized, double-blind, multicenter trial comparing orteronel (TAK-700) plus prednisone with placebo plus prednisone in patients with metastatic castration-resistant prostate cancer that has progressed during or after docetaxel-based therapy: ELM-PC 5. J Clin Oncol. 2015;33:723–731. doi: 10.1200/JCO.2014.56.5119. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Thompson IM, Goodman PJ, Tangen CM, Lucia MS, Miller GJ, Ford LG, Lieber MM, Cespedes RD, Atkins JN, Lippman SM, Carlin SM, Ryan A, Szczepanek CM, Crowley JJ, Coltman CA., Jr The influence of finasteride on the development of prostate cancer. N Engl J Med. 2003;349:215–224. doi: 10.1056/NEJMoa030660. [DOI] [PubMed] [Google Scholar]
- 111.Sikora MJ, Cordero KE, Larios JM, Johnson MD, Lippman ME, Rae JM. The androgen metabolite 5alpha-androstane-3beta,17beta-diol (3betaAdiol) induces breast cancer growth via estrogen receptor: implications for aromatase inhibitor resistance. Breast Cancer Res Treat. 2009;115:289–296. doi: 10.1007/s10549-008-0080-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112.Cali JJ, Russell DW. Characterization of human sterol 27-hydroxylase. A mitochondrial cytochrome P-450 that catalyzes multiple oxidation reaction in bile acid biosynthesis. J Biol Chem. 1991;266:7774–7778. [PubMed] [Google Scholar]
- 113.Umetani M, Domoto H, Gormley AK, Yuhanna IS, Cummins CL, Javitt NB, Korach KS, Shaul PW, Mangelsdorf DJ. 27-Hydroxycholesterol is an endogenous SERM that inhibits the cardiovascular effects of estrogen. Nat Med. 2007;13:1185–1192. doi: 10.1038/nm1641. [DOI] [PubMed] [Google Scholar]
- 114.DuSell CD, Umetani M, Shaul PW, Mangelsdorf DJ, McDonnell DP. 27-hydroxycholesterol is an endogenous selective estrogen receptor modulator. Mol Endocrinol. 2008;22:65–77. doi: 10.1210/me.2007-0383. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Toy W, Shen Y, Won H, Green B, Sakr RA, Will M, Li Z, Gala K, Fanning S, King TA, Hudis C, Chen D, Taran T, Hortobagyi G, Greene G, Berger M, Baselga J, Chandarlapaty S. ESR1 ligand-binding domain mutations in hormone-resistant breast cancer. Nat Genet. 2013;45:1439–1445. doi: 10.1038/ng.2822. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116.Robinson DR, Wu YM, Vats P, Su F, Lonigro RJ, Cao X, Kalyana-Sundaram S, Wang R, Ning Y, Hodges L, Gursky A, Siddiqui J, Tomlins SA, Roychowdhury S, Pienta KJ, Kim SY, Roberts JS, Rae JM, Van Poznak CH, Hayes DF, Chugh R, Kunju LP, Talpaz M, Schott AF, Chinnaiyan AM. Activating ESR1 mutations in hormone-resistant metastatic breast cancer. Nat Genet. 2013;45:1446–1451. doi: 10.1038/ng.2823. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117.Fujii R, Hanamura T, Suzuki T, Gohno T, Shibahara Y, Niwa T, Yamaguchi Y, Ohnuki K, Kakugawa Y, Hirakawa H, Ishida T, Sasano H, Ohuchi N, Hayashi S. Increased androgen receptor activity and cell proliferation in aromatase inhibitor-resistant breast carcinoma. J Steroid Biochem Mol Biol. 2014;144 Pt B:513–522. doi: 10.1016/j.jsbmb.2014.08.019. [DOI] [PubMed] [Google Scholar]
- 118.Rossi E, Morabito A, Di Rella F, Esposito G, Gravina A, Labonia V, Landi G, Nuzzo F, Pacilio C, De Maio E, Di Maio M, Piccirillo MC, De Feo G, D’Aiuto G, Botti G, Chiodini P, Gallo C, Perrone F, de Matteis A. Endocrine effects of adjuvant letrozole compared with tamoxifen in hormone-responsive postmenopausal patients with early breast cancer: the HOBOE trial. J Clin Oncol. 2009;27:3192–3197. doi: 10.1200/JCO.2008.18.6213. [DOI] [PubMed] [Google Scholar]
- 119.O’Shaughnessy J, Campone M, Brain E, Neven P, Hayes D, Bondarenko I, Griffin TW, Martin J, De Porre P, Kheoh T, Yu MK, Peng W, Johnston S. Abiraterone acetate, exemestane or the combination in postmenopausal patients with estrogen receptor-positive metastatic breast cancer. Ann Oncol. 2015;27:106–113. doi: 10.1093/annonc/mdv487. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120.Osborne CK, Schiff R. Mechanisms of endocrine resistance in breast cancer. Annu Rev Med. 2011;62:233–247. doi: 10.1146/annurev-med-070909-182917. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121.Lehmann BD, Bauer JA, Chen X, Sanders ME, Chakravarthy AB, Shyr Y, Pietenpol JA. Identification of human triple-negative breast cancer subtypes and preclinical models for selection of targeted therapies. J Clin Invest. 2011;121:2750–2767. doi: 10.1172/JCI45014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122.Perou CM. Molecular stratification of triple-negative breast cancers. Oncologist. 2011;16(Suppl 1):61–70. doi: 10.1634/theoncologist.2011-S1-61. [DOI] [PubMed] [Google Scholar]
- 123.Dent R, Trudeau M, Pritchard KI, Hanna WM, Kahn HK, Sawka CA, Lickley LA, Rawlinson E, Sun P, Narod SA. Triple-negative breast cancer: clinical features and patterns of recurrence. Clin Cancer Res. 2007;13:4429–4434. doi: 10.1158/1078-0432.CCR-06-3045. [DOI] [PubMed] [Google Scholar]
- 124.Hudis CA, Gianni L. Triple-negative breast cancer: an unmet medical need. Oncologist. 2011;16(Suppl 1):1–11. doi: 10.1634/theoncologist.2011-S1-01. [DOI] [PubMed] [Google Scholar]
- 125.Arce-Salinas C, Riesco-Martinez MC, Hanna W, Bedard P, Warner E. Complete response of metastatic androgen receptor-positive breast cancer to bicalutamide: case report and review of the literature. J Clin Oncol. 2014 doi: 10.1200/JCO.2013.49.8899. [DOI] [PubMed] [Google Scholar]
- 126.Gucalp A, Tolaney S, Isakoff SJ, Ingle JN, Liu MC, Carey LA, Blackwell K, Rugo H, Nabell L, Forero A, Stearns V, Doane AS, Danso M, Moynahan ME, Momen LF, Gonzalez JM, Akhtar A, Giri DD, Patil S, Feigin KN, Hudis CA, Traina TA, Translational Breast Cancer Research C. Phase II trial of bicalutamide in patients with androgen receptor-positive, estrogen receptor-negative metastatic breast cancer. Clin Cancer Res. 2013;19:5505–5512. doi: 10.1158/1078-0432.CCR-12-3327. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 127.Cochrane DR, Bernales S, Jacobsen BM, Cittelly DM, Howe EN, D’Amato NC, Spoelstra NS, Edgerton SM, Jean A, Guerrero J, Gomez F, Medicherla S, Alfaro IE, McCullagh E, Jedlicka P, Torkko KC, Thor AD, Elias AD, Protter AA, Richer JK. Role of the androgen receptor in breast cancer and preclinical analysis of enzalutamide. Breast Cancer Res. 2014;16:R7. doi: 10.1186/bcr3599. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 128.Traina TA, Miller K, Yardley DA, O’Shaughnessy J, Cortes J, Awada A, Kelly CM, Trudeau ME, Schmid P, Gianni L, Garcia-Estevez L, Nanda R, Ademuyiwa FO, Chan S, Steinberg JL, Blaney ME, Tudor IC, Uppal H, Peterson AC, Hudis CA. Results from a phase 2 study of enzalutamide (ENZA), an androgen receptor (AR) inhibitor, in advanced AR+ triple-negative breast cancer (TNBC) ASCO Meeting Abstracts. 2015;33:1003. [Google Scholar]
- 129.McDonald JG, Matthew S, Auchus RJ. Steroid profiling by gas chromatography–mass spectrometry and high performance liquid chromatography-mass spectrometry for adrenal diseases. Horm Cancer. 2011;2:324–332. doi: 10.1007/s12672-011-0099-x. [DOI] [PMC free article] [PubMed] [Google Scholar]