SUMMARY
Fanconi Anemia (FA) is an inherited DNA repair disorder characterized by progressive bone marrow failure (BMF) from hematopoietic stem and progenitor cell (HSPC) attrition. A greater understanding of the pathogenesis of BMF could improve the therapeutic options for FA patients. Using a genome-wide shRNA screen in human FA fibroblasts, we identify transforming growth factor-β (TGF-β) pathway-mediated growth suppression as a cause of BMF in FA. Blocking the TGF-β pathway improves the survival of FA cells and rescues the proliferative and functional defects of HSPCs derived from FA mice and FA patients. Inhibition of TGF-β signaling in FA HSPCs results in elevated homologous recombination (HR) repair with a concomitant decrease in non-homologous end-joining (NHEJ), accounting for the improvement in cellular growth. Together, our results suggest that elevated TGF-β signaling contributes to the BMF in FA by impairing HSPC function, and may be a potential therapeutic target for the treatment of FA.
INTRODUCTION
Hematopoietic stem cells (HSCs) replenish the blood system throughout the life span of an organism and maintain homeostasis. Consequently, depletion or impairment of the HSC reservoir leads to hematopoietic failure or hematologic malignancies. Accumulation of DNA damage and genomic instability, due to physiological stress or exposure to genotoxic agents, is one of the mechanisms of loss of HSC reserve (Garaycoechea et al., 2012; Mandal et al., 2011; Walter et al., 2015). Protective cellular DNA repair mechanisms exist in HSCs to combat the accumulation of DNA damage. Accordingly, the genetic ablation of proteins (e.g. Atm, Lig4, ATR) involved in DNA repair pathways in mice results in severe HSC defects (Ito et al., 2006; Niedernhofer, 2008; Rossi et al., 2007; Ruzankina et al., 2007), and the NHEJ pathway is the most commonly used DNA repair mechanism in quiescent HSCs (Milyavsky et al., 2010; Mohrin et al., 2010).
Fanconi anemia (FA) is the most common inherited bone marrow failure (BMF) syndrome. FA patients develop progressive bone marrow failure during childhood. In addition, FA patients also develop other hematologic manifestations, including myelodisplastic syndrome (MDS) and acute myeloid leukemia (AML) due to clonal evolution (Shimamura and Alter, 2010). Current therapies for hematologic manifestations in FA include hematopoietic stem cell transplant and treatment with growth factors and androgens. While an allogenic bone marrow transplant from a histocompatible sibling donor remains the optimal long-term therapy, many FA patients do not have a suitable donor. Moreover, transplant may contribute to increased incidence of later, non-hematologic consequence such as secondary solid tumors and endocrine failure (Shimamura and Alter, 2010). Therefore, novel alternative treatment options to treat marrow failure in FA patients are needed.
FA is caused by biallelic mutation in one of eighteen FANC genes (FANC-A, B, C, D1, D2, E, F, G, I, J, L, M, N, O, P, Q, S, T), the products of which cooperate in the FA/BRCA DNA repair pathway and regulate cellular resistance to DNA cross-linking agents (Kottemann and Smogorzewska, 2013; Sawyer et al., 2015). Due to the underlying DNA repair defect, FA cells are hypersensitive to genotoxic DNA crosslinking agents. Although the precise biochemical functions of the FA/BRCA pathway in DNA repair remain unclear, the pathway promotes homologous recombination (HR) repair (Deans and West, 2011). The FA/BRCA pathway also regulates cytokinesis, and pathway disruption results in increased binucleate bone marrow cells and apoptosis (Chan et al., 2009; Naim and Rosselli, 2009; Vinciguerra et al., 2010). FA cells are also uniquely hypersensitive to oxidative stress and apoptotic cytokines, such as IFNγ and TNFα (Pang and Andreassen, 2009).
All blood lineages are deficient in FA patients (Butturini et al., 1994), suggesting that the FA pathway regulates the function of hematopoietic stem and progenitor cells (HSPCs). Accordingly, recent studies indicate that the BMF in FA is attributable to an impaired HSPC pool. HSPCs in FA patients and FA mice exhibit reduced cell number and compromised stem cell function (Ceccaldi et al., 2012; Crossan et al., 2011; Parmar et al., 2009; Parmar et al., 2010; Zhang et al., 2010). The primary mechanism of the impairment of HSPCs leading to the BMF in FA, however, remains elusive. Due to the well-defined role of the FA/BRCA pathway in repairing DNA damage (Deans and West, 2011; Kottemann and Smogorzewska, 2013), DNA damage accumulation in HSPCs may be one of the major causes for BMF in FA. Indeed, recent studies suggest that BMF in FA and impaired HSPC function result from the genotoxicity of endogenous crosslinking agents or from physiological stress (Garaycoechea et al., 2012; Langevin et al., 2011; Walter et al., 2015). Bone marrow failure in FA may also result, directly or indirectly, from hyperactivation of cell autonomous or microenvironmental growth suppressive pathways, induced in part, due to genotoxic stress. We have recently shown that one suppressive mechanism results from a hyperactive p53 response in HSPCs from FA patients (Ceccaldi et al., 2012). Here, we performed an unbiased shRNA screen to identify additional growth suppressive pathways which may account for the impairment/attrition of the FA bone marrow HSPCs. We identified hyperactive TGF-β signaling in HSPCs as a mechanism of bone marrow suppression in FA. Disruption of TGF-β signaling significantly protected HSCs in FA from genotoxic stress by increasing HR activity and decreasing NHEJ activity.
RESULTS
TGF-β Pathway Blocks FA Cell Survival in the Presence of Genotoxic Agents
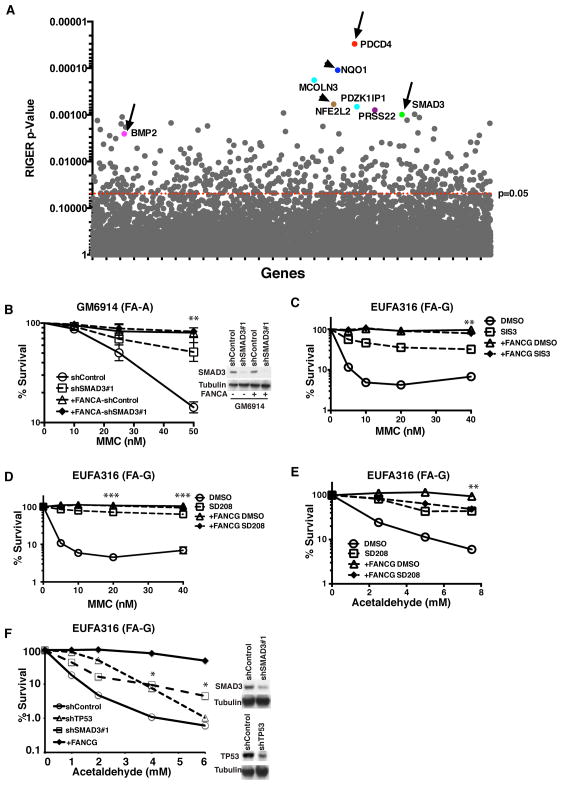
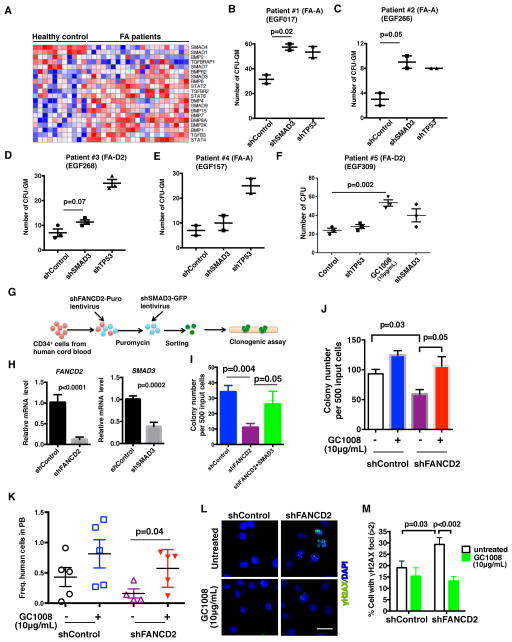
We initially performed a genome-wide shRNA screen in a patient-derived FANCA−/− fibroblast line in order to identify genes whose knockdown can rescue the FA phenotype in the presence of a genotoxic agent, Mitomycin C (MMC) (Figure S1A and S1B). Multiple TGF-β pathway genes (e.g. SMAD3, BMP2, PDCD4) were among the top hits (Figure 1A). shRNAs targeted against NQO1 and NFE2L2 genes involved in MMC metabolism (Siegel et al., 2012) also scored positive (Figure 1A), validating our screen for MMC resistance. The TGF-β pathway is known to contribute to hematopoietic suppression in other human diseases, such as myelodysplastic syndrome (MDS) (Suragani et al., 2014; Zhou et al., 2011; Zingariello et al., 2013), suggesting a more general mechanism in BMF. We next validated our primary screening data in a number of FA cell lines. The respective cell lines, which were corrected by reconstitution with FANCA or FANCG cDNAs, were used as positive controls as they were more resistant to MMC compared to the uncorrected FA cells (Figure 1B–1D). Both knockdown of SMAD3 and pharmacologic inhibition of the TGF-β pathway with small molecules, SD208 and SIS3, significantly enhanced the survival of FA cells in the presence of MMC (Figure 1B–1D, and S1C).
Figure 1. TGF-β Pathway Disruption Enhances Human FA Cellular Growth under Genotoxic Stress.
(A) RIGER P value analysis to identify top candidate genes in genome-wide screen. Top candidates are highlighted with different colors. Genes of the TGF-β pathway (BMP2, SMAD3, PDCD4) are pointed out by arrow. Genes involved in MMC metabolism (NQO1 and NFE2L2) are pointed out by arrowhead.
(B) Clonogenic survival of GM6914 (FA-A fibroblasts) and FANCA-corrected GM6914 cells after shRNA mediated knockdown of SMAD3. Cells were exposed MMC and cell survival was determined after 10–12 days in culture. The average of two independent experiments is presented. Immunoblot in the right panel shows SMAD3 knockdown efficiency in GM6914 cells.
(C,D) MMC sensitivity of EUFA316 (FA-G lymphoblasts) and FANCG-corrected EUFA316 cells incubated with or without 10μM SMAD3 inhibitor SIS3 or 10μM TGF-β inhibitor SD208. Cells were cultured in triplicates in presence of MMC and DMS0 or inhibitor for 5–6 days and survival was determined. Data are representative of 3 independent experiments.
(E) Acetaldehyde sensitivity of EUFA316 and FANCG-corrected EUFA316 cells incubated with or without 10μM SD208. Cells were exposed to acetaldehyde for 3 hours and survival was determined after culturing them for 5–6 days in presence of DMSO or SD208 in triplicates. The data are representative of 3 independent experiments.
(F) Acetaldehyde sensitivity of EUFA316 cells after shRNA mediated knockdown SMAD3 and TP53. Right panel shows the immunoblots of the lysates from EUFA316 cells showing shRNA-mediated knockdown of SMAD3 or p53.
Error bars represent mean ± s.e.m. *, p<0.05; **, p<0.01; ***, P<0.001. See also Figure S1.
DNA damage caused by an endogenous genotoxic agent, acetaldehyde, requires the FA pathway for repair (Garaycoechea et al., 2012). Accordingly, we identified an FA patient-derived FANCG-deficient lymphoblast line, EUFA316, which is hypersensitive to acetaldehyde (Figure S1D). Both pharmacologic inhibition by SD208 and genetic depletion by shRNA of the TGF-β pathway promoted cellular survival in the presence of acetaldehyde (Figure 1E and 1F). Additionally, disruption of the TGF-β pathway by shRNA or by SD208 resulted in reduced p53 and p21 protein levels (Figure S1E). Together, these data demonstrate that targeting the TGF-β pathway can reduce the genotoxic stress mediated growth inhibition of human FA cells.
TGF-β Pathway Inhibition Rescues the Functional Defects of HSCs from FA Mice
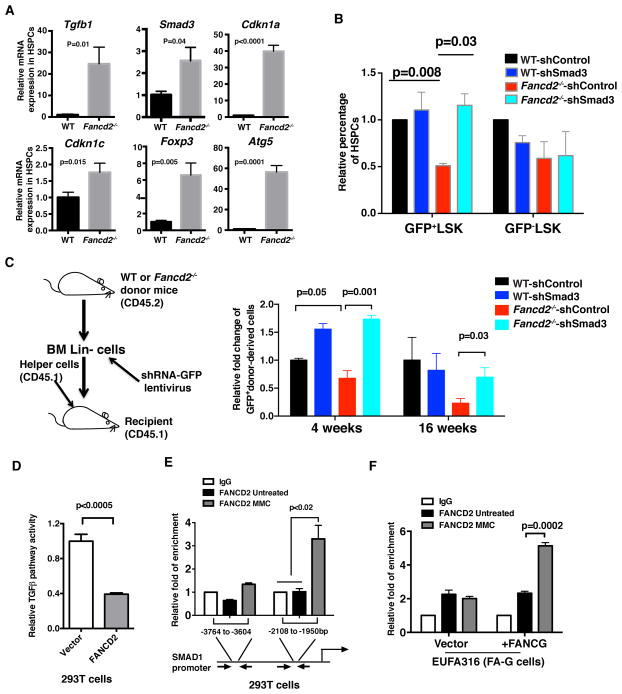
As FA mice exhibit HSPC defects, the possible suppressive function of the TGF-β pathway in bone marrow HSPCs of FA mice was next examined. Elevated expression levels of both Tgfb1 and Smad3 were observed in Fancd2−/− HSPCs [Lin−Sca-1+c-Kit+ (LSK) population] (Figure 2A). Elevated pSmad2/3 protein levels were also observed in Fancd2−/− HSPCs (Figure S2A and S2B). Moreover, enhanced mRNA expression of TGF-β pathway target genes, including Cdkn1a, Cdkn1c, Foxp3, and Atg5, was also observed in Fancd2−/− HSPCs (Figure 2A), implying that the TGF-β pathway is upregulated in FA HSPCs. Fancd2−/− bone marrow HSPCs were more sensitive to TGF-β1 than wild-type HSPCs (Figure S2C). Therefore, we hypothesized that inhibition of this pathway might rescue the defect of FA HSPCs. Colony forming assays showed that the clonogenic ability of Fancd2−/− HSPCs was inhibited by TGF-β1; blockade of the TGF-β signaling by 1D11 (a neutralizing antibody against mouse TGF-β) (Brenet et al., 2013) or SD208 markedly enhanced the clonogenic ability (Figure S2D–S2F). Further, genetic knockdown of Smad3 promoted the in vitro expansion of HSPCs and in vivo engraftment of Lin− cells from bone marrow of Fancd2−/− mice (Figure 2B, 2C, and S2G–S2J). Taken together, inhibition of the TGF-β pathway can rescue the functional defects of Fancd2−/− HSPCs.
Figure 2. Inhibition of the TGF-β Pathway Rescues the Functional Defects of HSPCs from FA Mice.
(A) qRT-PCR analysis showing the mRNA expression levels of Tgfb1, Smad3, Cdkn1a, Cdkn1c, Foxp3 and Atg5 in LSK populations from bone marrow of WT and Fancd2−/− mice.
(B) Quantification of the percentage of transduced (GFP+) and non-transduced (GFP−) LSK cells after 5 days in vitro culture. Bone marrow Lin− cells from WT and Fancd2−/− mice were transduced with lentiviruses encoding shSmad3-GFP or shControl-GFP and cultured in triplicates for 5 days. GFP+ and GFP- LSK cells were analyzed by flow cytometry. Data shown are the average of three independent experiments.
(C) Depletion of Smad3 promotes the engraftment ability of Fancd2−/− cells. Equal numbers of transduced Lin−GFP+ cells (20,000, CD45.2 cells) were transplanted into lethally irradiated recipients (CD45.1) along with 1×105 helper cells (CD45.1). Percentages of donor-derived cells (GFP+CD45.2+) in peripheral blood of recipients were analyzed at 4 and 16 weeks post bone marrow transplantation (n=5 recipient mice per group).
(D) Overexpression of FANCD2 in 293T cells suppresses TGF-β pathway activity as measured by TGF-β luciferase reporter activity.
(E) Binding of FANCD2 to SMAD1 promoter upon DNA damage in 293T cells as detected by ChIP assay. FANCD2 biding was observed in the predicted binding region −2108 to −1950 bp of SMAD1 promoter. 293T cells were treated with MMC (1μM) for 8 hrs, and used in ChIP assays with an anti-FANCD2 antibody or IgG control antibody, followed by real-time PCR with indicated primer sets. ChIP data are presented as enrichment fold of FANCD2 binding to the specific regions after normalization with IgG.
(F) Binding of FANCD2 to SMAD1 promoter upon DNA damage in corrected EUFA316 (+FAG) but not EUFA316 (+Vector) cells. ChIP assays with an anti-FANCD2 antibody or IgG control antibody were performed as described in (E) using a primer set for SMAD1 promoter region −2108 to −1950 bp.
Error bars represent mean ± s.e.m. See also Figure S2.
Hyperactive TGF-β pathway signaling in Fancd2−/− HSPCs prompted us to assess whether the FA pathway components directly transcriptionally regulate the expression of TGF-β pathway genes. We first confirmed that mutation in FA genes results in hyperactive TGF-β activity. We transfected a TGF-β luciferase reporter vector containing SMAD binding element, into FA cells or corrected FA cells. Interestingly, increased TGF-β luciferase activity was observed in FA-A cells compared to FANCA-corrected cells (Figure S2K). Conversely, overexpression of FANCD2 in 293T cells significantly reduced the TGF-β luciferase reporter activity (Figure 2D). Using chromatin immunoprecipitation (ChIP) assays, we previously showed that activated FANCD2 increases the transcriptional activity of the TAp63 gene (Park et al., 2013). A genome-wide ChIP sequencing (ChIP-seq) analysis, using FANCD2 antibody, demonstrated that FANCD2 also binds to SMAD gene promoters (Park et al., 2013). ChIP confirmed that FANCD2 can directly bind to the promoter region of a SMAD gene, suggesting that FANCD2 decreases the transcriptional activity of SMAD genes. Interestingly, FANCD2 bound to the promoter of SMAD1 in 293T cells (Figure 2E). The binding of FANCD2 to the promoter of SMAD1 was observed in corrected FA cells, but not in the parental FA cells (EUFA316 and GM6914), suggesting that a functional FA pathway is required for the FANCD2-mediated transcriptional decrease of the TGFβ pathway gene (Figure 2F and S2L). Collectively, these results demonstrate that disruption of the FA pathway causes transcriptional changes, resulting in hyperactive TGF-β signaling.
TGF-β Pathway Inhibition Rescues Acetaldehyde-induced Genotoxicity in HSPCs from FA Mice
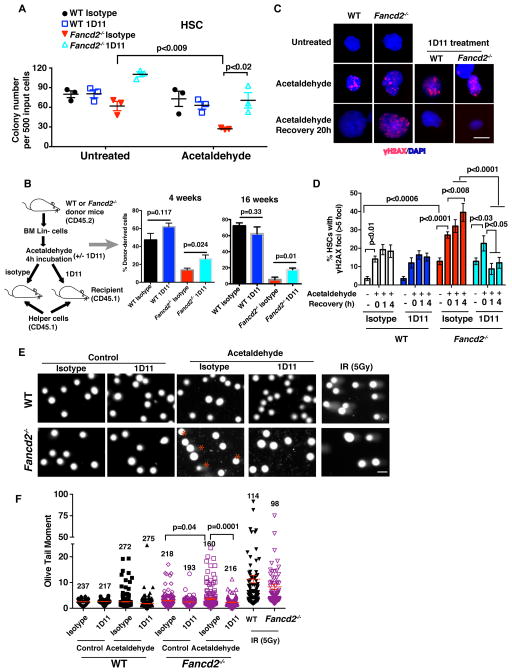
DNA damage induced by the endogenous genotoxic agents, such as acetaldehyde, causes the attrition of HSCs, leading to spontaneous bone marrow failure in murine FA models (Garaycoechea et al., 2012; Langevin et al., 2011). We reasoned that blockade of the TGF-β pathway may contribute to the repair of acetaldehyde-induced DNA damage in FA HSCs. As expected, bone marrow cells from Fancd2−/− mice were sensitive to acetaldehyde exposure. The neutralizing anti-TGF-β antibody 1D11 treatment counteracted the genotoxic effects of acetaldehyde, and rescued the growth and clonogenic capacity of Fancd2−/− HSCs or Lin− cells from bone marrow (Figure 3A and S3A). To directly measure the protective effect of 1D11 on the survival of HSPCs after acetaldehyde exposure, a stem cell transplant was performed. The percentage of donor-derived cells at 4 and 16 weeks post transplantation was higher in recipients of 1D11 treated Fancd2−/− group than those in the isotype control group (Figure 3B and S3B), and 1D11 did not significantly affect the lineage differentiation of HSPCs (Figure S3C). Furthermore, γH2AX foci, a marker for DNA damage, resolved more rapidly in 1D11 treated Fancd2−/− cells, suggesting an improvement of DNA repair (Figure 3C, 3D, and S3D). Repair of DNA damage was further confirmed by comet assay (Figure 3E, 3F, and S3E). 1D11 treated Fancd2−/− cells also showed improvement in DNA repair when DNA damage was directly assessed by alkaline comet assay and confirmed that acetaldehyde-induced γH2AX foci indeed correspond to unrepaired double strand breaks (Figure 3E, 3F, and S3E), Taken together, pharmacologic inhibition of the TGF-β pathway by 1D11 promotes resistance of FA cells to endogenous genotoxic agents, and partially corrects the impaired stem cell function by promoting DNA repair.
Figure 3. TGF-β Pathway Inhibition Rescues Acetaldehyde-induced Genotoxicity in HSPCs from FA Mice.
(A) Colony forming assay showing acetaldehyde resistance of Fancd2−/− HSCs (CD48−CD150+LSK cells) with 1D11 treatment. HSCs from WT and Fancd2−/− mice were exposed 2 mM acetaldehyde for 4h and then cultured in methycellulose medium containing 10 μg/mL 1D11 or its isotype control antibodies for 10 days. Survival of the hematopoietic progenitors was determined by colony quantification.
(B) Inhibition of TGF-β pathway enhances acetaldehyde resistance of HSCs. Lin− cells from bone marrow (BM) of WT and Fancd2−/− mice were pretreated with 1D11 or isotype control antibody for 30min, followed by exposure to 2mM acetaldehyde for 4h. After 24h in culture with 1D11 or isotype control antibody, equal numbers of cells were transplanted into lethally irradiated recipients (CD45.1) along with 1×105 helper cells (CD45.1). Donor-derived cells (CD45.2) in peripheral blood were analyzed by flow cytometric analysis at 4 and 16 weeks post transplantation (n=4~5 recipient mice per group).
(C, D, E, F) 1D11 rescues acetaldehyde-induced DNA damage in HSCs and Lin− cells. Representative images (C) and quantification (D) of γH2AX foci in HSCs from WT and Fancd2−/− mice are shown. HSCs were pretreated with 1D11 or isotype control antibody followed by exposure to acetaldehyde for 4h, and harvested for immunofluorescence at the indicated time points. Hundred to 200 cells with more than 5 foci were counted for each sample. (Scale bar: 20μm). (E) Representative images of alkaline comets of bone marrow Lineage-negative cells from WT and Fancd2−/− mice. Comet tails of Fancd2−/− cells in the isotype plus acetaldehyde treatment group are highlighted by a star. (Scale bar: 50μm). (F) Olive tail moment demonstrating that 1D11 significantly prevents acetaldehyde-induced DNA damage in Fancd2−/− bone marrow in vitro. Ninety-eight to 275 cells from each group were scored.
Error bars represent s.e.m. See also Figure S3.
Inhibition of TGF-β Pathway Rescues Physiological Stress-induced Bone Marrow Failure in FA Mice
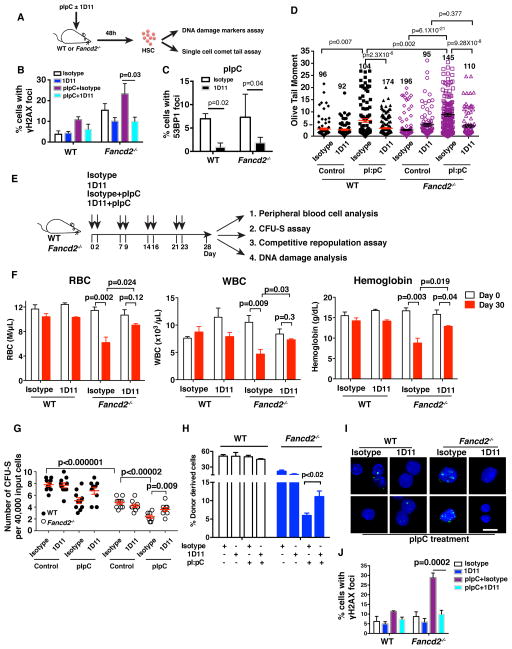
Physiological stress-induced DNA damage is a source of DNA damage in HSCs in FA. The accumulation of physiological stress-induced DNA damage results in a collapse of the hematopoietic system in a murine FA model (Walter et al., 2015). Interestingly, polyinosinic:polycytidylic acid (pI:pC) exposure caused activation of TGF-β pathway in HSPCs (Figure S4A). To determine whether inhibition of TGF-β pathway also counteracts the genomic instability of HSCs caused by physiological stress, we simultaneously treated WT and Fancd2−/− mice for 48h with pI:pC (to induce stress) and 1D11 (to block the TGF-β pathway). As previously described (Walter et al., 2015), administration of pI:pC induced DNA damage in HSCs and caused increased levels of γH2AX and 53BP1 foci (Figure 4A–4D and S4B); inhibition of the TGF-β pathway by 1D11 markedly reduced the pI:pC-induced DNA damage (Figure 4A–4D, S4B, and S4C). Importantly, inhibition of the TGF-β pathway rescued the viability of Fancd2−/− total bone marrow cells following pI:pC treatment (Figure S4D). Consistent with the previous study (Walter et al., 2015), pI:pC exposure induced oxidative stress in HSPCs from Fancd2−/− mice, however, 1D11 did not affect the level of oxidative stress (Figure S4E). As expected, pI:pC exposure induced proliferation of HSPCs, resulting in a concomitant reduction in the frequencies of quiescent cells. Interestingly, 1D11 treatment reduced pI:pC-induced cell cycling and increased the percentage of quiescent HSPCs, perhaps accounting for the improved DNA repair (Figure S4F). Thus, TGF-β pathway inhibition counteracts physiological stress-induced DNA damage in HSCs.
Figure 4. Inhibition of TGF-β Pathway Rescues Physiological Stress-induced Bone Marrow Failure in FA Mice.
(A) TGF-β pathway inhibition by 1D11 prevents pI:pC induced DNA damage in HSCs in vivo. WT and Fancd2−/− mice were injected intraperitoneally with pI:pC (5mg/kg) and 1D11 or isotype control antibody (10mg/kg). Forty-eight hours after the treatments, HSCs were sorted for DNA damage analysis by immunofluorescence staining or by the single cell comet tail assay.
(B) Percentages of HSCs with γH2AX foci) and (C) 53BP1 foci are shown. Hundred to 150 cells were counted for each sample.
(D) Olive tail moment in a comet assay demonstrating that 1D11 significantly reduces pI:pC-induced DNA damage in Fancd2−/− HSCs. Ninety-two to 196 HSCs from each group were scored.
(E) Schematic of pI:pC-induced bone marrow failure mouse model.
(F) Peripheral blood analysis of pI:pC plus isotype or 1D11-treated WT and Fancd2−/− mice as shown in (E). Red blood cell (RBC) counts, white blood cell counts (WBCs) and hemoglobin levels are shown (n=4–5 mice per group).
(G) CFU-S content in the bone marrow of WT or Fancd2−/− mice after four weeks of pI:pC plus isotype or 1D11 treatment as shown in (E)
(H) Inhibition of TGF-β pathway rescues pI;pC-mediated functional defects of HSPCs. WT and Fancd2−/− mice were exposed to pI:pC plus isotype or 1D11 for four weeks as shown in (E) and bone marrow cells were transplanted into lethally irradiated recipients (CD45.1) along with 1×105 helper cells (CD45.1). Donor-derived cells (CD45.2) in peripheral blood were analyzed at 4 weeks post transplantation (n=4~5 recipient mice per group).
(I, J) 1D11 rescues pI:pC-induced DNA damage in HSPCs. (I) Representative images and (J) quantification of γH2AX foci in HSPCs from WT and Fancd2−/− mice after four weeks treatment with pI:pC plus isotype or 1D11 as shown in (E). (Scale bar: 20μm).
Error bars represent s.e.m. See also Figure S4.
To determine whether TGF-β pathway inhibition rescues pI:pC induced bone marrow failure, we treated WT and Fancd2−/− mice with pI:pC and 1D11 (Figure 4E). Interestingly, four weeks of pI:pC treatment caused peripheral blood pancytopenia in isotype control antibody-treated Fancd2−/− mice with reduction in red blood cells (RBC), white blood cells (WBC) and hemoglobin levels (Figure 4F). However, administration of 1D11 significantly prevented these peripheral blood abnormalities (Figure 4F). To examine the bone marrow HSC function, we next transplanted bone marrow cells from these pI:pC (plus or minus 1D11) treated donor mice into lethally irradiated recipients and evaluated spleen colony-forming units (CFU-S), thus allowing the quantification of the surviving multipotent short-term HSCs. As expected, pI:pC treatment caused a marked reduction in the CFU-S content of Fancd2−/− bone marrow, and 1D11 improved this defect (Figure 4G). Consistent with these results, in a competitive repopulation experiment, recipient mice transplanted with bone marrow from 1D11 treated Fancd2-/- mice showed improved short-term donor cell engraftment compared to the recipients transplanted with isotype control (Figure 4H). Bone marrow HSPCs from Fancd2−/− mice treated with 1D11 also exhibited less DNA damage upon pI:pC exposure (Figure 4I, 4J, and S4G). Together, these data suggest that TGF-β pathway inhibition rescues pI:pC-induced bone marrow failure in FA mice and may be effective in preventing the bone marrow collapse of FA patients observed after viral infections.
TGF-β Pathway Inhibition Restores the Function of HSPCs Derived from Humans with FA
We next extended our studies in HSPCs derived from patients with FA. Primary human FA bone marrow cells demonstrated increased mRNA expression of multiple TGF-β pathway components (Figure 5A and S5A). TGF-β pathway inhibition, by knockdown of SMAD3 or by exposure to anti-human TGF-β neutralizing antibody GC1008, rescued the clonogenic defects of primary HSPCs from bone marrow of FA patients, although to a lesser extent than the rescue observed following p53 knockdown (Figure 5B–5F and S5B). Variable response to TGF-β pathway inhibition in primary human FA patient HSPCs may result from differences in the age, gender, intrinsic genetic differences, the levels of SMAD3/p53 in HSPCs, or the severity of the pre-existing bone marrow failure. Depletion of SMAD3 in FA-like primary HSPCs established by FANCD2 knockdown in human cord blood CD34+ cells, also rescued their clonogenicity, and partially restored MMC resistance (Figure 5G–5I and S5C). Inhibition of TGF-β signaling by GC1008 also recapitulated this phenotype by markedly rescuing the clonogenic capacity of FA-like HSPCs (Figure 5J and S5D). Further, since the very low CD34+ cell numbers in FA patients did not allow efficient xenograft assays for analysis of clonogenicity in vivo, we performed a surrogate in vivo xenograft assay using FA-like CD34+ cells. Strikingly, GC1008 treatment in vivo enhanced the engraftment of primary FA-like CD34+ cells (Figure 5K and S5E). In addition, we observed a higher frequency of FA-like HSPCs with γH2AX and 53BP1 foci upon MMC exposure, compared to the control HSPCs (Figure 5L, 5M, S5F and S5G). Exposure to GC1008 significantly reduced the number of FA-like HSPCs with γH2AX and 53BP1 foci (Figure 5L, 5M, S5F and S5G), suggesting that TGF-β pathway inhibition improves DNA repair in these primary human FA-like HSPCs. Together, our data indicate that the TGF-β signaling is increased in human FA HSPCs and that blockade of this pathway partially restores their function.
Figure 5. TGF-β Pathway Blockade Rescues the Function of Primary HSPCs from Patients with FA.
(A) Hierarchial clustering and heat-map of the expression of TGF-β pathway genes in human FA and non-FA bone marrow samples using gene set enrichment analysis.
(B–F) Colony forming assays using primary CD34+ bone marrow cells from five FA patients. Cells were transduced with lentiviruses encoding shControl, shTP53 or shSMAD3 followed by selection for puromycin resistance and then plated in methycellulose cultures or the cells were directly plated in methycellulose cultures containing GC1008 for colony growth. The hematopoietic colonies were counted after 10 days in culture.
(G–I) Colony forming assays using FA-like CD34+ cells from human cord blood. As shown in the schematic (G), FA-like CD34+ cells were generated by transducing human cord blood CD34+ cells with lentivirus encoding shFANCD2. After selection for puromycin resistance, cells were transduced with lentivirus encoding shSMAD3-GFP and GFP+ cells were sorted by FACS and subjected to clonogenic assay in triplicates. qRT-PCR analysis (H) shows significant reduction of FANCD2 and SMAD3 expression in cells. Hematopoietic colonies were counted after 10–14 days in culture (I).
(J) Colony forming assay of FA-like CD34+ cells with GC1008 treatment. Hematopoietic colonies were counted after 10–14 days in culture.
(K) In vivo xenograftment assay. Transduced human cord blood CD34+ cells with shFANCD2 or shControl were selected with puromycin and transplanted into sub-lethally irradiated NSG mice. Recipient mice were treated with GC1008 at 3 doses per week for 2 weeks. Human cells were analyzed in the peripheral blood at 2 weeks post transplantation. Data shown are combined from two independent experiments (n= 4–5 recipient mice).
(L,M) GC1008 rescues MMC-induced DNA damage in primary FA-like HSPCs. Representative images (L) and quantification (M) of γH2AX foci in cord blood CD34+ cells transduced with lentivirus encoding shFANCD2 or shControl. Puromycin resistant cord blood CD34+ cells transduced with lentivirus were exposed to MMC (100 ng/ml) for 2 hrs and allowed to recover for 24 hrs in presence of GC1008. Cells were then analyzed for γH2AX foci by immunofluorescence. Thirty to hundred cells with more than 5 foci were counted for each sample. (Scale bar: 20μm).
Error bars represent mean ± s.e.m. See also Figure S5.
TGF-β Pathway Inhibition Upregulates HR and Downregulates NHEJ in HSCs of FA Mice
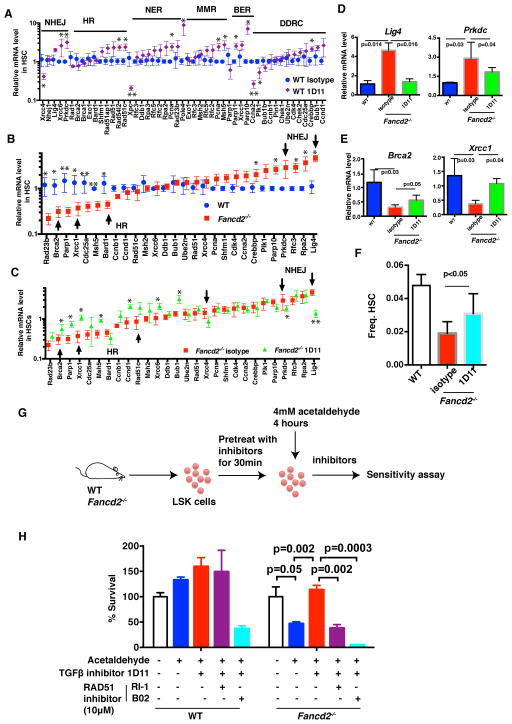
To determine the molecular mechanism by which TGF-β pathway regulates DNA repair and rescues genotoxicity in HSCs, we next analyzed the expression of DNA repair genes in HSCs. Genes associated with NHEJ, HR, nucleotide excision repair (NER), mismatch repair (MMR), and the DNA damage response (DDR) were evaluated. Interestingly, over 70% of DNA repair genes examined were significantly upregulated in HSCs from WT mice exposed to 1D11, the neutralizing anti-murine TGF-β monoclonal antibody (Figure 6A and S6A). In contrast, many genes involved in NHEJ were upregulated and many genes involved in HR were downregulated in HSCs from Fancd2−/− mice, when compared to WT HSCs (Figure 6B). This skewed expression pattern in Fancd2−/− HSCs was reversed by TGF-β pathway inhibition, as 1D11 antibody treatment induced expression of HR genes such as Brca2 and Xrcc1 in Fancd2−/− HSCs, and caused a concomitant reduction of NHEJ gene expression, such as Lig4 and Prkdc (Figure 6C–6E). Previous studies have shown that HR is upregulated when HSCs are driven into the cell cycle, thereby resulting in repair of double strand breaks (Beerman et al., 2014). Accordingly, the increase in HR activity in FA HSCs following TGF-β pathway inhibition may result, at least in part, from the release from cell cycle arrest (Figure S6B), subsequently leading to a higher frequency of HSCs in FA mice (Figure 6F). To confirm that inhibition of TGF-β pathway in HSCs creates an HR-competent state and thereby functionally protects them from genotoxicity, we inhibited both HR and TGF-β pathway and examined the survival of cells after acetaldehyde exposure (Figure 6G). We used pharmacologic inhibitors which blocked HR, resulting in the reduction of RAD51 foci and the inhibition of DR-GFP plasmid recombination in a reporter assay (Figure S6D–S6F). As expected, 1D11 exposure inhibited TGF-β pathway signaling in bone marrow from FA mice (Figure S6C). Interestingly, 1D11 did not protect the HSPCs from acetaldehyde-induced genotoxicity when HR was inhibited by pharmacologic inhibitors of RAD51 (Budke et al., 2012; Huang et al., 2012) (Figure 6H, S6G and S6H). Collectively, these data indicate that TGF-β pathway inhibition upregulates HR and downregulates NHEJ in HSPCs of Fancd2−/− mice and functionally promotes their survival.
Figure 6. TGF-β Pathway Inhibition Upregulates HR and Downregulates NHEJ in HSCs from Fancd2−/− mice. FA.
(A) TGF-β pathway inhibition induces expression of the majority genes involved in DNA damage repair in HSCs from WT mice. HSCs were sorted from WT mice after 48h treatment with 1D11 or isotype control antibody, and used for qRT-PCR analysis.
(B) The expression levels of DNA damage repair genes in Fancd2−/− and WT HSCs. Some genes involved in HR and NHEJ pathways were pointed out by arrow.
(C) Blockade of TGF-β pathway induces HR gene expression and downregulates NHEJ gene expression in HSCs from Fancd2−/− mice. HSCs were sorted from Fancd2−/− mice after 48h treatment with 1D11 or isotype control antibody, and used for qRT-PCR analysis. Some genes involved in HR and NHEJ pathways were pointed out by arrow.
(D, E) Gene expression of representative NHEJ (D) and HR (E) genes Lig4, Prkdc, Brca2, and Xrcc1 in HSCs.
(F) The frequency of HSCs in Fancd2−/− mice after 48h treatment with 1D11 or isotype control antibody (n=4 mice per group).
(G) Schematic of acetaldehyde sensitivity assay in bone marrow HSPCs from WT or Fancd2−/− mice.
(H) 1D11 does not protect the Fancd2−/− HSPCs from genotoxic stress when HR is inhibited. HSPCs from WT or Fancd2−/− mice were exposed to 1D11 and RAD51 inhibitors (10 μM) for 30 min followed by exposure to acetaldehyde for 4 hrs. The cells were then washed and cultured in presence of 1D11 and RAD51 inhibitors for five days and survival was determined.
Error bars represent mean ± s.e.m. *p < 0.05; **p < 0.01. See also Figure S6.
TGF-β Pathway Inhibition Increases HR and Decreases NHEJ in FA Cells
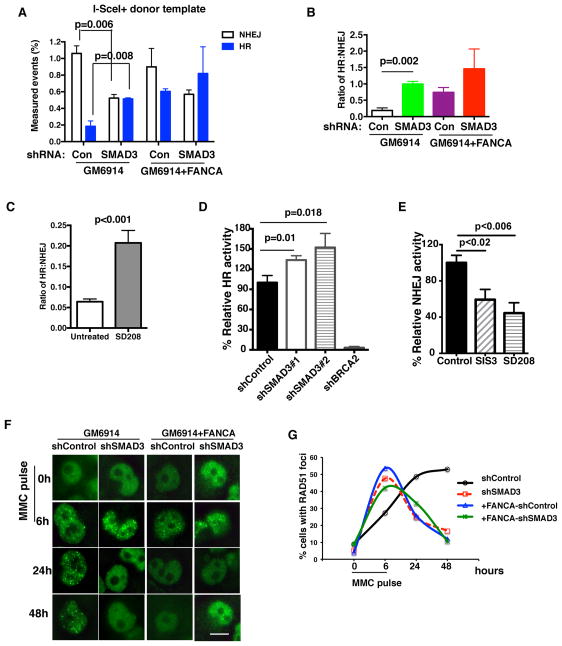
To directly confirm that inhibition of TGF-β pathway modulates HR or NHEJ activity, we engineered an individual DNA breakpoint in FA (GM6914) cells and employed the traffic light reporter (TLR) system which quantifies both HR and NHEJ activities (Certo et al., 2011). In the TLR assay, double strand break (DSB) generated by I-Sce1 endonuclease can be repaired by either HR or NHEJ pathway when the donor repair template is provided, however, the DSB can be repaired by NHEJ only when the repair template is missing (Figure S7A). NHEJ and not HR activity was observed in FA cells when the donor repair template was missing (Figure S7B), validating the assay. As predicted, FA (GM6914) cells exhibited fewer HR events compared to the FA-A-corrected (GM6914+FANCA) cells (Figure 7A). Interestingly, TGF-β pathway inhibition, by knockdown of SMAD3, in FA cells resulted in increased HR events with a concomitant decrease in NHEJ events (Figure 7A and 7B). Inhibition of TGF-β pathway by the small molecule inhibitor SD208 also resulted in increased HR activity in FA cells (Figure 7C). Similar findings were revealed when different reporter systems were used to quantify the HR and NHEJ activities individually (Figure 7D and 7E). Consistently, knockdown of SMAD3 in FA cells resulted in decreased phosphorylation of DNA-PK, a marker of NHEJ (Figure S7C). Additionally, damage-induced RAD51 foci resolved more quickly in SMAD3-depleted FA cells (Figure 7F and 7G), implying that HR-mediated DNA repair is more efficient in these cells. Analysis of the genome-wide siRNA screening data (Adamson et al., 2012) revealed that siRNA-mediated knockdown of several TGF-β pathway genes enhanced HR activity (Figure S7D). Taken together, these data indicate that TGF-β pathway inhibition promotes DNA repair by directly increasing HR activity in FA cells.
Figure 7. TGF-β Pathway Inhibition Increases HR and Decreases NHEJ activities in FA Cells.
(A, B) TGF-β pathway inhibition affects the choice of HR versus NHEJ pathways in repairing individual DNA breakpoints in FA cells. GM6914 cells (FA-A cells) or FANCA corrected GM6914 cells with shControl or shSMAD3 were used to generate traffic light reporter system, and then were infected with GFP-donor template and I-SceI lentivirus to generate DNA breakpoints. Quantification analysis of HR and NHEJ repair events is shown. (B) The ratio of HR to NHEJ activity based on the data in (A).
(C) SD208 mediated TGF-β pathway inhibition increases HR events and decreases NHEJ events. Quantification of HR and NHEJ repair events in GM6914 cells exposed to SD-208 for 72 hrs as detected by traffic light reporter assay described in (A).
(D) SMAD3 knockdown significantly increases HR efficiency. HR assay was measured in U2OS cells with DR-GFP reporter after transduction with lentivirus encoding indicated shRNAs. The representative of three independent experiments is presented.
(E) NHEJ reporter assay showing decreased NHEJ activity in U2OS cells after inhibition of the TGF-β pathway by small molecule inhibitors.
(F, G) TGF-β pathway inhibition promotes HR activity in FA cells. Representative images (F) and quantification (G) of RAD51 foci in MMC treated GM6914 (FA-A) cells or FANCA-corrected GM6914 cells with shRNA-mediated knockdown of SMAD3. Cells were exposed to 1μM MMC for 6h, and allowed to recover for 24h and 48h. RAD51 foci were then identified. One hundred cells were scored for RAD51 foci. (Scale bar: 20μm)
Error bars represent mean ± s.e.m. See also Figure S7.
DISCUSSION
Although the pathophysiology of BMF in FA is poorly understood, anemia in FA patients is known to result from attrition of HSPCs. The attrition of HSPCs in FA results from multiple mechanisms including impaired HSPC function, genotoxicity in HSPCs from the endogenous crosslinking agent, acetaldehyde (Garaycoechea et al., 2012; Langevin et al., 2011), physiological stress (Walter et al., 2015), elevated p53 levels in HSPCs (Ceccaldi et al., 2012), and hypersensitivity of the bone marrow to inflammatory cytokines (Haneline et al., 1998; Li et al., 2007). Here, we show that hyperactive TGF-β signaling also contributes to the impairment of the HSPC function in FA bone marrow. Blocking of TGF-β pathway signaling in FA HSPCs improves their growth and survival under genotoxic stress. TGF-β pathway inhibition in FA cells reduces toxic NHEJ and partially rescues HR activity. Hyperactive NHEJ is known to be toxic to FA cells, and NHEJ inhibitors improve FA cell growth (Adamo et al., 2010; Pace et al., 2010). Thus, the improved growth of FA HSPCs following TGF-β pathway inhibition appears to result largely from restored HR repair and reduced NHEJ repair, resulting in part from an increase in cycling HSPCs. TGF-β inhibition also appears to promote a HR competent state, even in cells which have not entered active cycling. Under physiological stress, the rapid reduction in DNA damage by TGF-β inhibition results in a rapid decline in cycling and in the maintenance or, for some cells, the return to an HSC quiescent state. Our results also support the recent finding that DNA damage accumulation occurs in quiescent, but not cycling HSCs (Beerman I et al, 2014).
Our study shows that mutation in FA genes results in hyperactive TGF-β signaling. This result suggests that the FA pathway normally functions to activate FANCD2 and to repress transcription of SMAD genes. We show that FANCD2, a critical downstream component of the FA pathway, binds to the promoter region of SMAD1. Activated FANCD2 has been previously reported to decrease transcription of the TNFα gene and to increase transcription of the TAp63 gene (Matsushita et al., 2011; Park et al., 2013). Our results indicate that hyperactive TGF-β pathway in FA cells contributes to the impairment of DNA repair due to downregulation of HR activity. The TGF-β pathway is known to play a role in DNA damage response (Barcellos-Hoff and Cucinotta, 2014). For instance, BRCA2 and SMAD3 form a complex and synergize in regulation of gene transcription (Preobrazhenska et al., 2002). SMAD3 also counteracts BRCA1-dependent repair of DNA double-strand breaks in human breast epithelial cells (Dubrovska et al., 2005). In addition, TGF-β pathway induces HR defects in breast cancer cells by regulating BRCA1, ATM, and MSH2 DNA repair genes (Liu et al., 2014). Moreover, TGF-β pathway activation promotes genomic instability by downregulating RAD51 protein expression in lung epithelial cells (Kanamoto et al., 2002). These studies further support our finding that inhibition of the hyperactive TGF-β pathway restores HR in FA HSPCs and improves their function. It remains to be determined whether the SMAD factors directly suppress HR gene transcription and promote NHEJ gene transcription.
Our results elucidate a mechanism that contributes to bone marrow failure in FA patients–namely, the increased TGF-β signaling in FA HSPCs. TGF-β is a myelosuppressive cytokine that has been also implicated in the hematopoietic suppression of other hematopoietic diseases, including myelodysplastic syndrome (MDS), suggesting a more general mechanism of BMF (Suragani et al., 2014; Zhou et al., 2011; Zingariello et al., 2013). Plasma levels of TGF-β are mildly upregulated in MDS patients, and increased TGF-β immunohistochemical staining is evident in the bone marrow. Inhibition of TGF-β pathway after chemotherapy accelerates hematopoietic reconstitution by delaying the return of cycling HSCs to quiescence (Brenet et al., 2013). Murine and human FA HSPC function was enhanced after TGF-β pathway inhibition, consistent with the notion that TGF-β regulates quiescence and self-renewal of HSPCs (Bank and Kalrsson, 2014).
Finally, our findings have implications for the clinical care of FA patients. Pharmacological agents targeting the TGF-β pathway have been developed for a variety of disease conditions, and some have shown efficacy in preclinical murine models and in human phase I and II clinical trials (Akhurst and Hata, 2012; Grafe et al., 2014; Suragani et al., 2014). Pharmacological inhibition of the TGF-β pathway appears to be safe and well tolerated, and 1D11 treatment does not cause obvious negative effects on general health, behavior, or growth in mice (Grafe et al., 2014). Inhibition of the TGF-β pathway may also be a clinically useful strategy during gene therapy for ex vivo expansion of rare HSPCs derived from FA patients (Rio et al., 2014). Additionally, the ability of TGF-β pathway inhibition to increase HR and decrease NHEJ may also provide a tool to improve CRISPR-mediated genome editing and may also be a mechanistic basis for the reduction of tumorigenesis.
EXPERIMENTAL PROCEDURES
Detailed procedures can be found in the Supplemental Experimental Procedures.
Genome-wide shRNA Screen
The screen was performed using the Hannon-Elledge whole genome pooled shRNA library as previously described (Luo et al., 2009).
Mice
Fancd2−/− mice in C57BL/6J background are previously described (Parmar et al., 2010). B6.SJL-PtprcaPep3b/BoyJ (Congenic B6, CD45.1+) mice and NOD.Cg-Prkdcscid Il2rgtm1Wjl/SzJ (NSG) mice were purchased from The Jackson Laboratory. All experimental procedures were approved by the Animal Care and Use Committee of the Dana Farber Cancer Institute.
Hematopoietic Stem/progenitor Cell Culture And Flow Cytometry Analysis
The procedures of enrichment and cell staining for isolating HSPCs (Lin−cKit+Sca-1+) and HSCs (Lin−cKit+Sca-1+CD48−CD150+) from mouse bone marrow were performed as described (Zhang et al., 2012).
Murine Bone Marrow Transplantation
Donor cells (CD45.2+) were transplanted into lethally irradiated (10 Gy dose) recipient (congenic B6-CD45.1+ mice) along with 1×105cells competitive bone marrow cells from congenic B6-CD45.1+ mice. Peripheral blood from recipient mice was analyzed for donor cell engraftment as described (Parmar et al., 2010).
Colony-forming Unit-spleen (CFU-S) Assay
Recipient mice (wild-type, 8–12 weeks old) were irradiated with a split dose of 1100 rad (550 rad each, 4 hours apart) before transplantation. Forty thousand bone marrow cells from donor mice were transplanted into each recipient mouse. Ten to 12 days post-transplantation, spleens were harvested and fixed with Bouin fixative solution.
In Vivo Xenograft Assay
Human cord blood derived CD34+ cells were transduced with lentivirus encoding shFANCD2 or shControl as described (Ceccaldi et al., 2012). While cells were selected with 2μg/mL puromycin, cells were also treated with 10μg/mL GC1008. After 48h treatment, 2×105 cells were transplanted into sub-lethally irradiated (2.5Gy) NSG mice. The recipient mice were treated with 10mg/kg GC1008 at 3 doses per week for two weeks. Two and eight weeks after transplantation, human cells in peripheral blood (PB) were analyzed using anti-human CD45 antibody (eBioscience, 17–0459–42) by flow cytometry.
Statistics
Two-tailed P values for statistical analysis were obtained using Student’s t-test.
Supplementary Material
Acknowledgments
We are grateful to Raphael Ceccaldi and members of our laboratory for helpful discussion. We thank Benjamin Primack for maintaining Fancd2 mouse colony. We thank Dr. H. Lin (Massachusetts General Hospital), who generously provided the TGF-β reporter CAGA-Luciferase plasmid. We thank Genzyme Corp for providing the 1D11 and GC1008 antibodies. We thank the FA patients and their families. H.Z is supported by the Leukemia & Lymphoma Society Fellow Award. This work was also supported by grants from U.S. National Institutes of Health R01DK43889, R37HL052725 (to A.D.), and P01HL048546 (to M.G.).
Footnotes
AUTHOR CONTRIBUTIONS
H.Z. and A.D.D designed experiments. H.Z., D.E.K., K.W.O., S.V., A.R., A.H., L.M., E.F.G., and K.P., performed experiments with help from J.G., G.B., J.S., and M.G.. H.Z., D.E.K., K.P., and A.D.D. analyzed and interpreted the data. H.Z., K.P., and A.D.D. wrote the manuscript.
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
References
- Adamo A, Collis SJ, Adelman CA, Silva N, Horejsi Z, Ward JD, Martinez-Perez E, Boulton SJ, La Volpe A. Preventing nonhomologous end joining suppresses DNA repair defects of Fanconi anemia. Mol Cell. 2010;39:25–35. doi: 10.1016/j.molcel.2010.06.026. [DOI] [PubMed] [Google Scholar]
- Adamson B, Smogorzewska A, Sigoillot FD, King RW, Elledge SJ. A genome-wide homologous recombination screen identifies the RNA-binding protein RBMX as a component of the DNA-damage response. Nat Cell Biol. 2012;14:318–328. doi: 10.1038/ncb2426. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Akhurst RJ, Hata A. Targeting the TGFbeta signalling pathway in disease. Nat Rev Drug Discov. 2012;11:790–811. doi: 10.1038/nrd3810. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Barcellos-Hoff MH, Cucinotta FA. New tricks for an old fox: impact of TGFbeta on the DNA damage response and genomic stability. Sci Signal. 2014;7:re5. doi: 10.1126/scisignal.2005474. [DOI] [PubMed] [Google Scholar]
- Beerman I, Seita J, Inlay MA, Weissman IL, Rossi DJ. Quiescent hematopoietic stem cells accumulate DNA damage during aging that is repaired upon entry into cell cycle. Cell Stem Cell. 2014;15:37–50. doi: 10.1016/j.stem.2014.04.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brenet F, Kermani P, Spektor R, Rafii S, Scandura JM. TGFbeta restores hematopoietic homeostasis after myelosuppressive chemotherapy. J Exp Med. 2013;210:623–639. doi: 10.1084/jem.20121610. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Budke B, Logan HL, Kalin JH, Zelivianskaia AS, Cameron McGuire W, Miller LL, Stark JM, Kozikowski AP, Bishop DK, Connell PP. RI-1: a chemical inhibitor of RAD51 that disrupts homologous recombination in human cells. Nucleic Acids Res. 2012;40:7347–7357. doi: 10.1093/nar/gks353. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Butturini A, Gale RP, Verlander PC, Adler-Brecher B, Gillio AP, Auerbach AD. Hematologic abnormalities in Fanconi anemia: an International Fanconi Anemia Registry study. Blood. 1994;84:1650–1655. [PubMed] [Google Scholar]
- Ceccaldi R, Parmar K, Mouly E, Delord M, Kim JM, Regairaz M, Pla M, Vasquez N, Zhang QS, Pondarre C, et al. Bone marrow failure in Fanconi anemia is triggered by an exacerbated p53/p21 DNA damage response that impairs hematopoietic stem and progenitor cells. Cell Stem Cell. 2012;11:36–49. doi: 10.1016/j.stem.2012.05.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Certo MT, Ryu BY, Annis JE, Garibov M, Jarjour J, Rawlings DJ, Scharenberg AM. Tracking genome engineering outcome at individual DNA breakpoints. Nat Methods. 2011;8:671–676. doi: 10.1038/nmeth.1648. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chan KL, Palmai-Pallag T, Ying S, Hickson ID. Replication stress induces sister-chromatid bridging at fragile site loci in mitosis. Nat Cell Biol. 2009;11:753–760. doi: 10.1038/ncb1882. [DOI] [PubMed] [Google Scholar]
- Crossan GP, van der Weyden L, Rosado IV, Langevin F, Gaillard PH, McIntyre RE, Gallagher F, Kettunen MI, Lewis DY, Brindle K, et al. Disruption of mouse Slx4, a regulator of structure-specific nucleases, phenocopies Fanconi anemia. Nat Genet. 2011;43:147–152. doi: 10.1038/ng.752. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Deans AJ, West SC. DNA interstrand crosslink repair and cancer. Nat Rev Cancer. 2011;11:467–480. doi: 10.1038/nrc3088. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dubrovska A, Kanamoto T, Lomnytska M, Heldin CH, Volodko N, Souchelnytskyi S. TGFbeta1/Smad3 counteracts BRCA1-dependent repair of DNA damage. Oncogene. 2005;24:2289–2297. doi: 10.1038/sj.onc.1208443. [DOI] [PubMed] [Google Scholar]
- Garaycoechea JI, Crossan GP, Langevin F, Daly M, Arends MJ, Patel KJ. Genotoxic consequences of endogenous aldehydes on mouse haematopoietic stem cell function. Nature. 2012;489:571–575. doi: 10.1038/nature11368. [DOI] [PubMed] [Google Scholar]
- Grafe I, Yang T, Alexander S, Homan EP, Lietman C, Jiang MM, Bertin T, Munivez E, Chen Y, Dawson B, et al. Excessive transforming growth factor-beta signaling is a common mechanism in osteogenesis imperfecta. Nat Med. 2014;20:670–675. doi: 10.1038/nm.3544. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Haneline LS, Broxmeyer HE, Cooper S, Hangoc G, Carreau M, Buchwald M, Clapp DW. Multiple inhibitory cytokines induce deregulated progenitor growth and apoptosis in hematopoietic cells from Fac-/- mice. Blood. 1998;91:4092–4098. [PubMed] [Google Scholar]
- Huang F, Mazina OM, Zentner IJ, Cocklin S, Mazin AV. Inhibition of homologous recombination in human cells by targeting RAD51 recombinase. J Med Chem. 2012;55:3011–3020. doi: 10.1021/jm201173g. [DOI] [PubMed] [Google Scholar]
- Ito K, Hirao A, Arai F, Takubo K, Matsuoka S, Miyamoto K, Ohmura M, Naka K, Hosokawa K, Ikeda Y, et al. Reactive oxygen species act through p38 MAPK to limit the lifespan of hematopoietic stem cells. Nat Med. 2006;12:446–451. doi: 10.1038/nm1388. [DOI] [PubMed] [Google Scholar]
- Kanamoto T, Hellman U, Heldin CH, Souchelnytskyi S. Functional proteomics of transforming growth factor-beta1-stimulated Mv1Lu epithelial cells: Rad51 as a target of TGFbeta1-dependent regulation of DNA repair. EMBO J. 2002;21:1219–1230. doi: 10.1093/emboj/21.5.1219. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kottemann MC, Smogorzewska A. Fanconi anaemia and the repair of Watson and Crick DNA crosslinks. Nature. 2013;493:356–363. doi: 10.1038/nature11863. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Langevin F, Crossan GP, Rosado IV, Arends MJ, Patel KJ. Fancd2 counteracts the toxic effects of naturally produced aldehydes in mice. Nature. 2011;475:53–58. doi: 10.1038/nature10192. [DOI] [PubMed] [Google Scholar]
- Li J, Sejas DP, Zhang X, Qiu Y, Nattamai KJ, Rani R, Rathbun KR, Geiger H, Williams DA, Bagby GC, et al. TNF-alpha induces leukemic clonal evolution ex vivo in Fanconi anemia group C murine stem cells. J Clin Invest. 2007;117:3283–3295. doi: 10.1172/JCI31772. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu L, Zhou W, Cheng CT, Ren X, Somlo G, Fong MY, Chin AR, Li H, Yu Y, Xu Y, et al. TGFbeta induces “BRCAness” and sensitivity to PARP inhibition in breast cancer by regulating DNA-repair genes. Mol Cancer Res. 2014;12:1597–1609. doi: 10.1158/1541-7786.MCR-14-0201. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Luo J, Emanuele MJ, Li D, Creighton CJ, Schlabach MR, Westbrook TF, Wong KK, Elledge SJ. A genome-wide RNAi screen identifies multiple synthetic lethal interactions with the Ras oncogene. Cell. 2009;137:835–848. doi: 10.1016/j.cell.2009.05.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mandal PK, Blanpain C, Rossi DJ. DNA damage response in adult stem cells: pathways and consequences. Nat Rev Mol Cell Biol. 2011;12:198–202. doi: 10.1038/nrm3060. [DOI] [PubMed] [Google Scholar]
- Matsushita N, Endo Y, Sato K, Kurumizaka H, Yamashita T, Takata M, Yanagi S. Direct inhibition of TNF-alpha promoter activity by Fanconi anemia protein FANCD2. PloS one. 2011;6:e23324. doi: 10.1371/journal.pone.0023324. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Milyavsky M, Gan OI, Trottier M, Komosa M, Tabach O, Notta F, Lechman E, Hermans KG, Eppert K, Konovalova Z, et al. A distinctive DNA damage response in human hematopoietic stem cells reveals an apoptosis-independent role for p53 in self-renewal. Cell Stem Cell. 2010;7:186–197. doi: 10.1016/j.stem.2010.05.016. [DOI] [PubMed] [Google Scholar]
- Mohrin M, Bourke E, Alexander D, Warr MR, Barry-Holson K, Le Beau MM, Morrison CG, Passegue E. Hematopoietic stem cell quiescence promotes error-prone DNA repair and mutagenesis. Cell Stem Cell. 2010;7:174–185. doi: 10.1016/j.stem.2010.06.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Naim V, Rosselli F. The FANC pathway and BLM collaborate during mitosis to prevent micro-nucleation and chromosome abnormalities. Nat Cell Biol. 2009;11:761–768. doi: 10.1038/ncb1883. [DOI] [PubMed] [Google Scholar]
- Niedernhofer LJ. DNA repair is crucial for maintaining hematopoietic stem cell function. DNA Repair (Amst) 2008;7:523–529. doi: 10.1016/j.dnarep.2007.11.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pace P, Mosedale G, Hodskinson MR, Rosado IV, Sivasubramaniam M, Patel KJ. Ku70 corrupts DNA repair in the absence of the Fanconi anemia pathway. Science. 2010;329:219–223. doi: 10.1126/science.1192277. [DOI] [PubMed] [Google Scholar]
- Pang Q, Andreassen PR. Fanconi anemia proteins and endogenous stresses. Mutat Res. 2009;668:42–53. doi: 10.1016/j.mrfmmm.2009.03.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Park E, Kim H, Kim JM, Primack B, Vidal-Cardenas S, Xu Y, Price BD, Mills AA, D’Andrea AD. FANCD2 activates transcription of TAp63 and suppresses tumorigenesis. Mol Cell. 2013;50:908–918. doi: 10.1016/j.molcel.2013.05.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Parmar K, D’Andrea A, Niedernhofer LJ. Mouse models of Fanconi anemia. Mutat Res. 2009;668:133–140. doi: 10.1016/j.mrfmmm.2009.03.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Parmar K, Kim J, Sykes SM, Shimamura A, Stuckert P, Zhu K, Hamilton A, Deloach MK, Kutok JL, Akashi K, et al. Hematopoietic stem cell defects in mice with deficiency of Fancd2 or Usp1. Stem Cells. 2010;28:1186–1195. doi: 10.1002/stem.437. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Preobrazhenska O, Yakymovych M, Kanamoto T, Yakymovych I, Stoika R, Heldin CH, Souchelnytskyi S. BRCA2 and Smad3 synergize in regulation of gene transcription. Oncogene. 2002;21:5660–5664. doi: 10.1038/sj.onc.1205732. [DOI] [PubMed] [Google Scholar]
- Rio P, Banos R, Lombardo A, Quintana-Bustamante O, Alvarez L, Garate Z, Genovese P, Almarza E, Valeri A, Diez B, et al. Targeted gene therapy and cell reprogramming in Fanconi anemia. EMBO Mol Med. 2014;6:835–848. doi: 10.15252/emmm.201303374. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rossi DJ, Bryder D, Seita J, Nussenzweig A, Hoeijmakers J, Weissman IL. Deficiencies in DNA damage repair limit the function of haematopoietic stem cells with age. Nature. 2007;447:725–729. doi: 10.1038/nature05862. [DOI] [PubMed] [Google Scholar]
- Ruzankina Y, Pinzon-Guzman C, Asare A, Ong T, Pontano L, Cotsarelis G, Zediak VP, Velez M, Bhandoola A, Brown EJ. Deletion of the developmentally essential gene ATR in adult mice leads to age-related phenotypes and stem cell loss. Cell Stem Cell. 2007;1:113–126. doi: 10.1016/j.stem.2007.03.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sawyer SL, Tian L, Kahkonen M, Schwartzentruber J, Kircher M, Majewski J, Dyment DA, Innes AM, Boycott KM, Moreau LA, et al. Biallelic mutations in BRCA1 cause a new Fanconi anemia subtype. Cancer Discov. 2015;5:135–142. doi: 10.1158/2159-8290.CD-14-1156. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shimamura A, Alter BP. Pathophysiology and management of inherited bone marrow failure syndromes. Blood Rev. 2010;24:101–122. doi: 10.1016/j.blre.2010.03.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Siegel D, Yan C, Ross D. NAD(P)H:quinone oxidoreductase 1 (NQO1) in the sensitivity and resistance to antitumor quinones. Biochem Pharmacol. 2012;83:1033–1040. doi: 10.1016/j.bcp.2011.12.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Suragani RN, Cadena SM, Cawley SM, Sako D, Mitchell D, Li R, Davies MV, Alexander MJ, Devine M, Loveday KS, et al. Transforming growth factor-beta superfamily ligand trap ACE-536 corrects anemia by promoting late-stage erythropoiesis. Nat Med. 2014;20:408–414. doi: 10.1038/nm.3512. [DOI] [PubMed] [Google Scholar]
- Vinciguerra P, Godinho SA, Parmar K, Pellman D, D’Andrea AD. Cytokinesis failure occurs in Fanconi anemia pathway-deficient murine and human bone marrow hematopoietic cells. J Clin Invest. 2010;120:3834–3842. doi: 10.1172/JCI43391. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Walter D, Lier A, Geiselhart A, Thalheimer FB, Huntscha S, Sobotta MC, Moehrle B, Brocks D, Bayindir I, Kaschutnig P, et al. Exit from dormancy provokes DNA-damage-induced attrition in haematopoietic stem cells. Nature. 2015;520:549–552. doi: 10.1038/nature14131. [DOI] [PubMed] [Google Scholar]
- Zhang H, Peng C, Hu Y, Li H, Sheng Z, Chen Y, Sullivan C, Cerny J, Hutchinson L, Higgins A, et al. The Blk pathway functions as a tumor suppressor in chronic myeloid leukemia stem cells. Nat Genet. 2012;44:861–871. doi: 10.1038/ng.2350. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang QS, Marquez-Loza L, Eaton L, Duncan AW, Goldman DC, Anur P, Watanabe-Smith K, Rathbun RK, Fleming WH, Bagby GC, et al. Fancd2-/- mice have hematopoietic defects that can be partially corrected by resveratrol. Blood. 2010;116:5140–5148. doi: 10.1182/blood-2010-04-278226. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhou L, McMahon C, Bhagat T, Alencar C, Yu Y, Fazzari M, Sohal D, Heuck C, Gundabolu K, Ng C, et al. Reduced SMAD7 leads to overactivation of TGF-beta signaling in MDS that can be reversed by a specific inhibitor of TGF-beta receptor I kinase. Cancer Res. 2011;71:955–963. doi: 10.1158/0008-5472.CAN-10-2933. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zingariello M, Martelli F, Ciaffoni F, Masiello F, Ghinassi B, D’Amore E, Massa M, Barosi G, Sancillo L, Li X, et al. Characterization of the TGF-beta1 signaling abnormalities in the Gata1low mouse model of myelofibrosis. Blood. 2013;121:3345–3363. doi: 10.1182/blood-2012-06-439661. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.