Abstract
Improving lower-limb prostheses is important to enhance the mobility of amputees. The purpose of this paper is to introduce an impedance-based control strategy (consisting of four novel algorithms) for an active knee and ankle prosthesis and test its generalizability across multiple walking speeds, walking surfaces, and users. The four algorithms increased ankle stiffness throughout stance, decreased knee stiffness during terminal stance, as well as provided powered ankle plantarflexion and knee swing initiation through modifications of equilibrium positions of the ankle and knee, respectively. Seven amputees (knee disarticulation and transfemoral levels) walked at slow, comfortable, and hurried speeds on level and inclined (10°) surfaces. The prosthesis was tuned at their comfortable level ground walking speed. We further quantified trends in prosthetic knee and ankle kinematics, and kinetics across conditions. Subjects modulated their walking speed by ±25% (average) from their comfortable speeds. As speed increased, increasing ankle angles and velocities as well as stance phase ankle power and plantarflexion torque were observed. At slow and comfortable speeds, plantarflexion torque was increased on the incline. At slow and comfortable speeds, stance phase positive knee power was increased and knee torque more flexor on the incline. As speed increased, knee torque became less flexor on the incline. These algorithms were shown to generalize well across speed, produce gait mechanics that compare favorably with non-amputee data, and display evidence of scalable device function. They have the potential to reduce the challenge of clinically configuring such devices and increase their viability during daily use.
Keywords: Biomechanics, gait, transfemoral amputee, powered knee and ankle prosthesis, sloped surface, walking speed
Improving lower-limb prostheses is important to enhance the mobility of amputees. This paper introduces an impedance-based control strategy (consisting of four novel algorithms) for an active knee and ankle prosthesis and tests of its generalizability across multiple walking speeds, walking surfaces and users. In clinical tests, the developed algorithms for an active knee and ankle prosthesis were shown to generalize well across speeds, walking surfaces and users. They also displayed evidence of scalable device function and have the potential to reduce the challenge of clinically configuring such devices, increasing their viability during daily use.
I. Introduction
Major lower-limb amputations account for roughly 40% of all amputations to human extremities [1], [2]. Individuals with these amputations commonly experience abnormal gait characteristics including asymmetrical mechanics [3], [4] and elevated metabolic energy expenditures [5]. These characteristics are often coupled with increased incidences of joint disorders [for review, see 6] as well as chronic leg and back pain [7]–[10]. Furthermore, the prevalence and/or intensity of these behaviors have been shown to generally increase as the level of leg amputation increases [5], [6]. Thus, improving the design and control of prostheses is important, especially for individuals with a high level of amputation such as transfemoral (above-knee) amputees. However, nearly all commercially-available prosthetic knees and ankles are mechanically-passive devices, which cannot perform net positive work about their degrees-of-freedom. This constraint is in contrast to the observed behavior of muscle in able-bodied non-amputees.
Biological musculotendon units have been shown to satisfy whole-body energetic requirements of locomotion as they produce, dissipate or transfer power about the hip, knee and ankle [11]–[14]. In particular, the late stance phase of walking is characterized by high angular velocities of the knee and ankle, while muscles spanning these joints contribute to important walking subtasks [12], [14], [15]. During the second half of stance, the soleus contributes to vertical support and forward propulsion of the body while delivering mechanical energy to the trunk [12], [15]. The gastrocnemius contributes to vertical support and forward propulsion of the body while delivering energy to the leg for swing initiation [12], [15]. In addition, the rectus femoris redistributes energy from the leg to the trunk for forward propulsion [14]. Collectively, the ankle plantarflexors (gastrocnemius and soleus) are the primary contributors to trunk and leg power during this period [16]. Given these roles of muscles spanning the knee and ankle, appropriately functioning lower-limb prostheses are especially important during this phase of the gait cycle.
Recent development of mechanically-active (i.e., motorized) prostheses [17]–[24] that can deliver physiological levels of joint power offer an opportunity to more closely approximate muscular functions during human ambulation. However, the simultaneous control of an active knee and ankle is challenging, especially when attempting to restore the functions of multiple uniarticular and biarticular muscles spanning the knee and ankle joints (e.g., [25]). Previous case studies have relied on empirical tuning of the virtual joint impedance (i.e., the stiffness, damping and equilibrium position of each joint during a finite set of states) to fit an active knee and ankle prosthesis to a user [21], [22], [24]. This tuning method consisted of manually adjusting all impedance parameters within each state as user feedback and joint kinematics and kinetics were assessed. Using this method, these studies have shown the active prosthetic knee and ankle kinematics of an above-knee amputee can better replicate those of non-amputees, relative to passive prostheses [24], [26]. Walking at moderate and altered speeds [24] as well as additional modes of ambulation (e.g., stair ascent/descent) were demonstrated [25]–[27]. In addition, the impedance parameters selected for the amputee user varied across states within each ambulation mode and across speeds during walking [21], [22], [24]. Control algorithms that can be generalized across various users as well as user-initiated modulations of ambulation speed are needed to demonstrate how active knee and ankle prostheses can benefit a greater percentage of the amputee population.
The terrains and walking speeds we experience during daily movement are variable. Previous studies have found that while using passive prostheses, above-knee amputees typically have difficulty with or avoid ramped surfaces, and walk at a slower self-selected speed (e.g., [3]). This may in part be due to inherent constraints on the actuation of their prostheses, which limit the ability of amputees to accommodate to increased task demands of traversing an incline or increasing walking speed. In order to improve these outcomes by using active prostheses, non-time-based control algorithms are beneficial. One reason is that the duration of stance decreases as walking speed increases. Thus, any action of a prosthesis tuned with respect to time for a particular patient and walking speed would likely be limited or exaggerated if the patient were to walk faster or slower, respectively. Furthermore, the action would likely be perceived differently for another patient who walks with different temporal characteristics (e.g., stance duration). Demonstrating effective control algorithms of an active knee and ankle prosthesis that are generalizable across level and inclined surfaces, variable walking speeds and multiple users would be an important step toward improving lower-limb prostheses and the mobility of amputees.
The purpose of this study was to introduce an impedance-based control strategy (consisting of four novel algorithms to control stance) for an active knee and ankle prosthesis and test its generalizability across multiple walking speeds, walking surfaces and amputee users. We evaluated the control strategy by assessing the ability of the users to comfortably and seamlessly modulate their walking speed. We further evaluated our approach by quantifying trends in prosthetic knee and ankle kinematics and kinetics throughout the gait cycle.
II. Methods
A. Active Knee and Ankle Prosthesis
Mechanical and electronic characteristics of the active knee and ankle prosthesis used during this study have been previously reported [22], [24], [26]. Briefly, the device consisted of two DC electric motors and belt-driven transmissions to power the ankle and knee degrees-of-freedom. Onboard sensors measured the current sent to each motor. In addition, a uniaxial load cell in series between the knee and ankle that measured the axial shank force (500 Hz). Lastly, potentiometers and motor encoders at the knee and ankle measured joint angles and velocities. The device was powered by a 30 V battery.
B. Knee and Ankle Finite State Impedance Control
An onboard microprocessor and custom software used sensor data of the knee and ankle angles and velocities sampled at 500 Hz to compute torque signals sent to each motor. The torque signals of each joint ( were computed according to impedance-based models of the knee and ankle, consisting of a virtual angular stiffness (
were computed according to impedance-based models of the knee and ankle, consisting of a virtual angular stiffness ( , damping (
, damping ( and equilibrium position (
and equilibrium position ( .
.
 |
The impedance parameters were specified within a finite number of states and updated at 50 Hz, while a standard set of triggers (based on mechanical sensor values) were used to switch between states (Fig. 1). This overall control framework of a finite state, impedance-based state machine is similar to approaches described elsewhere [22], [24]. Impedance control allows the user to interact dynamically with their environment (i.e., the ground) within each state, rather than attempting to enforce strict kinematic trajectories. Previously, impedance control has been implemented to enforce passive dynamics by maintaining constant joint impedance parameters within each state. Joint power is typically generated by step changes of impedance parameters at each joint between states.
FIGURE 1.
State machine of walking consisting of 4 finite states (early-mid stance, late stance, swing flexion and swing extension). Onboard mechanical sensor thresholds were used to switch between states and were constant across conditions. Specifically, to detect heel strike and toe off, thresholds of the axial shank force were used. To switch between early-mid stance and late stance, ankle dorsiflexion thresholds were used. To switch between swing flexion and swing extension states, knee flexion velocity thresholds were used.
The state machine used in this study consisted of two states within stance (early through mid stance and late stance) and two states within swing (swing flexion and swing extension), which were consistent across level and incline walking modes of ambulation. Control loops used thresholds on mechanical sensors to switch states (e.g., [22], [24]) (Fig. 1). Mechanical sensor thresholds were also consistent across level and incline walking modes and variable speeds. Within this framework, we integrated biologically-based empirical evidence during the stance phase of walking regarding ankle stiffness [28], [29] and knee quasi stiffness [11], [22], as well as a straightforward approach to perform powered ankle plantarflexion and knee swing-initiation. Furthermore, an important difference in the control framework used in this study was that joint impedances were not constrained to be constant within a given state. Thus, this impedance style of control did not enforce passive dynamics during every state, as in previous studies. Rather, the four new algorithms modulated respective impedance parameters as functions of joint angle or axial shank force, which attempt to mimic biological joint impedance or enabled the subjects to smoothly alter their rates of power generation within and across states.
C. Rate-Based Prosthesis Control Algorithms
Four algorithms were implemented during the stance phase of walking, which altered the rate of change of a given impedance parameter as a function of either the rate of ankle dorsiflexion or the rate of shank unloading. Based on previous experiments of non-amputee walking, [28], [29] we facilitated increasing ankle stiffness ( , Nm/deg) during controlled dorsiflexion (spanning both finite states of stance) as a linear function of ankle angle (
, Nm/deg) during controlled dorsiflexion (spanning both finite states of stance) as a linear function of ankle angle ( , deg), and scaled to a given user’s weight (
, deg), and scaled to a given user’s weight ( , kg).
, kg).
 |
Previous studies determined this ankle stiffness relationship of non-amputees during unshod walking between approximately 10% to 65% of stance by applying angular perturbations about the ankle using a robotic platform [28], [29]. In the current study, ankle stiffness was constrained to never decrease throughout stance. An initial minimum stiffness was set equal to ankle stiffness in the swing extension state. Also, maximum stiffness was capped in late stance (7 Nm/deg) to satisfy an electrical constraint on the ankle motor current.
Based on data from non-amputees [11] and further analyzed by [22], decreasing knee stiffness ( , Nm/deg) (to a final value of 0 Nm/deg) during late stance was facilitated. This modulation was specified as a linear function of decreasing axial shank force (Eq. 3). Similarly, knee swing initiation and powered ankle plantarflexion were controlled by changes of their equilibrium positions (
, Nm/deg) (to a final value of 0 Nm/deg) during late stance was facilitated. This modulation was specified as a linear function of decreasing axial shank force (Eq. 3). Similarly, knee swing initiation and powered ankle plantarflexion were controlled by changes of their equilibrium positions ( as linear functions of decreasing shank force during late stance (Fig. 2, Eq. 3). These three algorithms to modify given impedance parameters (
as linear functions of decreasing shank force during late stance (Fig. 2, Eq. 3). These three algorithms to modify given impedance parameters ( as a function of decreasing shank force,
as a function of decreasing shank force,  , (i.e., a load cell measurement) were governed by a rate-based equation containing straightforward tuning constants (in bold).
, (i.e., a load cell measurement) were governed by a rate-based equation containing straightforward tuning constants (in bold).
 |
Two tuning constants included proportionality constants,  , which scaled each rate-of-change, and final “desired” impedance values,
, which scaled each rate-of-change, and final “desired” impedance values,  . Other constants in the equation were either detected at state changes or constrained by the state machine (i.e., were mechanical sensor values used to switch between states). Finally, this equation was constrained to ensure always increasing or decreasing impedance parameters within the state and impedance parameters between
. Other constants in the equation were either detected at state changes or constrained by the state machine (i.e., were mechanical sensor values used to switch between states). Finally, this equation was constrained to ensure always increasing or decreasing impedance parameters within the state and impedance parameters between  and
and  .
.
FIGURE 2.
Conceptual example depicting the use of the decreasing axial shank force during late stance to control powered ankle plantarflexion and knee swing initiation by modifying ankle and knee equilibrium angles, respectively. Left-hand column shows how at a fixed walking speed, tuning of a proportionality constant, C, can increase or decrease the rate of the function output. In this example, final equilibrium angle of the ankle is set to −12° (plantarflexion) and final equilibrium angle of the knee is set to −45° (flexion). For proportionality constants >1, the function is constrained to stop at each final value. Similarly, the right-hand column shows how the function output changes for an altered walking speed, with fixed tuning parameters (final equilibrium angles and proportionality constants). Note, changes of the equilibrium angles occur sooner and at a faster rate for increasing walking speed. Conversely, these changes occur later and at a slower rate for decreasing walking speed. Axial shank force data across a range of walking speeds are averaged from a group of non-amputees [46].
Swing flexion and swing extension states did not contain any of these algorithms to modulate joint impedance. In particular, in swing flexion, the knee equilibrium angle was held constant and set equal to its final “flexed” value from late stance. In swing extension, knee equilibrium angle was constant at 0°.
D. Amputee Subjects and Experimental Protocol
We tested 7 amputees (Table 1) using the active knee and ankle (e.g., Fig. 3). Amputation level varied from knee disarticulation to the proximal third of the femur. Subjects provided informed consent to an Institutional Review Board approved protocol prior to participation in the study. All had a minimum of 6 hours experience using the device.
TABLE 1.
Above-knee amputee subject characteristics, including if subjects wore a microprocessor (MP) or non-microprocessor (NMP) controlled knee as their prescribed “home” prosthesis.
FIGURE 3.
Three of the various above-knee amputee users of the active knee and ankle prostheses, using the device to walk inside the laboratory up an incline as part of this experiment (left and middle), as well as outside the laboratory over level ground (right).
Following donning, subjects first accommodated to the device while walking in parallel bars, and then walked outside the bars to an overground walkway of  meters in length. Next, the subjects practiced walking on the inclined (10°) ramped surface. During this process, tuning adjustments were made as needed until the subjects, physical therapist, prosthetists and engineers were comfortable with the operation of the device. The accommodation periods were quite short (i.e., all were less than 15 minutes total) given their previous experience using the prosthesis. For each user, the prosthesis was tuned at their comfortable level ground speed. No tuning adjustments were made across speeds. Furthermore, only one within-state impedance parameter adjustment was made between level and incline trials. Knee flexion angle (equilibrium position during swing flexion) was decreased by 10°. After accommodating to the device, level and incline blocks of trials were collected. Within each block, subjects walked at their self-selected comfortable, hurried and slow speeds. An average of 20 strides were collected per condition.
meters in length. Next, the subjects practiced walking on the inclined (10°) ramped surface. During this process, tuning adjustments were made as needed until the subjects, physical therapist, prosthetists and engineers were comfortable with the operation of the device. The accommodation periods were quite short (i.e., all were less than 15 minutes total) given their previous experience using the prosthesis. For each user, the prosthesis was tuned at their comfortable level ground speed. No tuning adjustments were made across speeds. Furthermore, only one within-state impedance parameter adjustment was made between level and incline trials. Knee flexion angle (equilibrium position during swing flexion) was decreased by 10°. After accommodating to the device, level and incline blocks of trials were collected. Within each block, subjects walked at their self-selected comfortable, hurried and slow speeds. An average of 20 strides were collected per condition.
E. Data Collection Methods and Statistical Analyses
To evaluate the utility of our approach, average walking speeds were first assessed. Then, to provide insight into how the prosthesis assisted the users modulating their speed, prosthesis knee and ankle kinematics and kinetics were compared, with an emphasis on the terminal stance and early swing phases of gait. Speeds and joint kinematic and kinetic quantities were compared across conditions using two-factor (speed, walking surface) repeated-measures analyses of variance (ANOVAs). The speed factor consisted of three levels (comfortable, hurried and slow). The surface factor had two levels (level walking and incline walking). When significant main or interaction effects were detected ( ), pairwise tests were made using a Bonferroni adjustment for multiple comparisons.
), pairwise tests were made using a Bonferroni adjustment for multiple comparisons.
III. Results
A. Walking Speed
Each subject modulated their walking speed on both level and incline walking surfaces (Table 2). Decreases and increases from their comfortable speeds were between 23 and 29% on average across subjects. All pairwise increases or decreases in walking speed were statistically significant. In addition, at all speed conditions the subjects walked significantly slower on the incline (Table 2).
TABLE 2.
Individual subject (TF) and group averaged walking speed over level ground and an incline. Speed and surface main effects were significant as well as all pairwise comparisons.
B. Prosthetic Knee and Ankle Kinematics
As speed increased, trends toward increasing peak ankle dorsiflexion and plantarflexion angles were observed (Figs. 4 and 5). Significant speed main effects and pairwise differences were found in both of these quantities across speeds during both level and incline walking (Figs. 4 and 5). In addition, there was significantly less dorsiflexion during slow walking and more plantarflexion at all speeds on level ground relative to the incline.
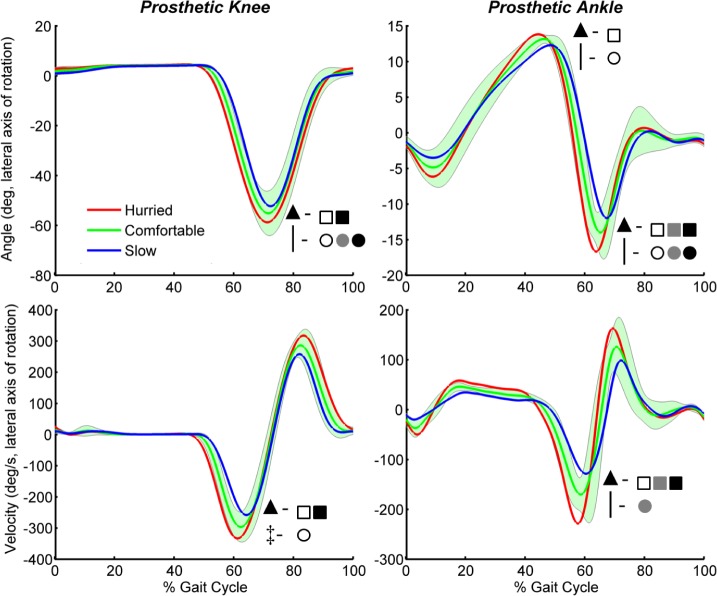
FIGURE 4.
Group-averaged prosthetic knee and ankle angles and velocities during level ground walking. One standard deviation of the comfortable speed condition is shaded. Peak knee flexion, ankle dorsiflexion and ankle plantarflexion angles were compared as well as peak knee flexion and ankle plantarflexion velocities. Significant ( ) speed (
) speed ( ), surface (
), surface ( ) and interaction (
) and interaction ( ) effects are shown. Significant slow to hurried (□), slow to comfortable (
) effects are shown. Significant slow to hurried (□), slow to comfortable ( ) and comfortable to hurried (■) pairwise comparisons using a Bonferroni adjustment for significance are indicated. Significant differences between level and incline walking at slow (○), comfortable (
) and comfortable to hurried (■) pairwise comparisons using a Bonferroni adjustment for significance are indicated. Significant differences between level and incline walking at slow (○), comfortable ( ) and hurried (■) conditions are also indicated.
) and hurried (■) conditions are also indicated.
FIGURE 5.
Group-averaged prosthetic knee and ankle angles and velocities during incline walking. One standard deviation of the comfortable speed condition is shaded. Peak knee flexion, ankle dorsiflexion and ankle plantarflexion angles were compared as well as peak knee flexion and ankle plantarflexion velocities. Significant ( ) speed (▲), surface (
) speed (▲), surface ( ) and interaction (
) and interaction ( ) effects are shown. Significant slow to hurried (□), slow to comfortable (
) effects are shown. Significant slow to hurried (□), slow to comfortable ( ) and comfortable to hurried (■) pairwise comparisons using a Bonferroni adjustment for significance are indicated. Significant differences between level and incline walking at slow (○), comfortable (
) and comfortable to hurried (■) pairwise comparisons using a Bonferroni adjustment for significance are indicated. Significant differences between level and incline walking at slow (○), comfortable ( ) and hurried (●) conditions are also indicated.
) and hurried (●) conditions are also indicated.
Peak plantarflexion velocity significantly increased with speed on both surfaces (Figs. 4 and 5). At comfortable speeds, peak plantarflexion velocity was significantly less on the incline relative to level ground.
During incline walking, peak knee flexion angle increased with speed. Similarly, peak knee flexion velocity significantly increased across speed conditions on the incline (Figs. 4 and 5). At all speeds, there was less peak knee flexion (Figs. 4 and 5) in incline walking, reflecting the one within-state impedance parameter difference between the two surfaces (described above), which was a reduced knee flexion equilibrium angle in incline walking.
C. Prosthetic Knee and Ankle Kinetics
Average positive and negative ankle power in stance significantly increased as speed increased for both walking surfaces (Figs. 6 and 7). Average negative ankle power was significantly larger during level slow and hurried walking when compared to incline (Figs. 6 and 7). This difference also approached significance at the comfortable speed condition ( ).
).
FIGURE 6.
Group-averaged prosthetic knee and ankle torques and powers during level ground walking. One standard deviation of the comfortable speed condition is shaded. Knee torque at terminal stance and peak ankle plantarflexion torque were compared as well as average stance phase positive and negative knee and ankle power. Significant ( ) speed (▲), surface (
) speed (▲), surface ( ) and interaction (
) and interaction ( ) effects are shown. Significant slow to hurried (□), slow to comfortable (
) effects are shown. Significant slow to hurried (□), slow to comfortable ( ) and comfortable to hurried (■) pairwise comparisons using a Bonferroni adjustment for significance are indicated. Significant differences between level and incline walking at slow (○), comfortable (
) and comfortable to hurried (■) pairwise comparisons using a Bonferroni adjustment for significance are indicated. Significant differences between level and incline walking at slow (○), comfortable ( ) and hurried (●) conditions are also indicated.
) and hurried (●) conditions are also indicated.
FIGURE 7.
Group-averaged prosthetic knee and ankle torques and powers during incline walking. One standard deviation of the comfortable speed condition is shaded. Knee torque at terminal stance and peak ankle plantarflexion torque were compared as well as average stance phase positive and negative knee and ankle power. Significant ( ) speed (▲), surface (
) speed (▲), surface ( ) and interaction (
) and interaction ( ) effects are shown. Significant slow to hurried (□), slow to comfortable (
) effects are shown. Significant slow to hurried (□), slow to comfortable ( ) and comfortable to hurried (◀) pairwise comparisons using a Bonferroni adjustment for significance are indicated. Significant differences between level and incline walking at slow (○), comfortable (
) and comfortable to hurried (◀) pairwise comparisons using a Bonferroni adjustment for significance are indicated. Significant differences between level and incline walking at slow (○), comfortable (  ) and hurried ( ●) conditions are also indicated.
) and hurried ( ●) conditions are also indicated.
In level and incline conditions, peak plantarflexion torque increased as speed increased (Figs. 6 and 7). All speed comparisons were significant on level ground, and the slow to hurried comparison was significant on the incline. In addition, at slow and comfortable speeds, peak plantarflexion torque was significantly larger on the incline compared to level ground.
No significant differences were found when comparing average negative knee power during stance. However, average positive knee power at the slow and comfortable speeds was significantly higher on the incline. This difference also approached significance in the hurried conditions ( ).
).
Terminal stance knee torque displayed significant speed and surface effects (Figs. 6 and 7). On the incline, knee torque became significantly less flexor when comparing slow to hurried speeds. Similar changes of the knee torque becoming less flexor (and in some cases an extensor torque) in terminal stance approached significance during level walking (slow to hurried,  ). Finally, at the slow and comfortable speeds, terminal stance knee torque was more flexor in the incline compared to level walking trials (Figs. 6 and 7). This difference approached significance at hurried speeds (
). Finally, at the slow and comfortable speeds, terminal stance knee torque was more flexor in the incline compared to level walking trials (Figs. 6 and 7). This difference approached significance at hurried speeds ( ).
).
IV. Discussion
The purpose of this study was to introduce a novel impedance-based control strategy for an active knee and ankle prosthesis and test its generalizability across multiple walking speeds, walking surfaces and amputee users. We implemented four novel control algorithms to modulate joint impedance as functions of joint angle or axial shank force. These algorithms attempt to mimic biological joint impedance (i.e., increasing ankle stiffness during stance [29] or decreasing knee stiffness in terminal stance [11], [22]) or enable users to smoothly generate power during the stance to swing transition (Eq. 3, Fig. 2).
A. Control Algorithm Generalization
Despite various etiologies, genders, amputation levels, heights and weights (Table 1), each subject was able to seamlessly increase or decrease their walking speed on both level and inclined surfaces (Table 2). Furthermore, their speeds were comparable with previous reports of amputees using microprocessor and non-microprocessor mechanically-passive prostheses. For example, self-selected walking speed of above-knee amputees on level ground with these devices ( m/s, [30]) was between our “hurried” and “comfortable” walking conditions (Table 2). No tuning adjustments were made across speeds, and only one consistent change was made from level to incline conditions (i.e., a 10° decrease in knee flexion equilibrium angle). Furthermore, across subjects, several tuning parameters were constant. Proportionality constants governing knee swing initiation, decreasing knee stiffness and powered ankle plantarflexion in late stance were 1.0, 1.0 and 1.5 (Eq. 3), respectively. Final plantarflexion equilibrium angle in late stance was 12° for all subjects (Eq. 3). An exception was final knee flexion equilibrium angle, which varied (45-70°) across subjects. However, meaningful changes of this parameter were made to achieve ground clearance for various amputation levels, while the distance between the knee and ankle was fixed at its minimum value (i.e., a current device constraint) [26].
m/s, [30]) was between our “hurried” and “comfortable” walking conditions (Table 2). No tuning adjustments were made across speeds, and only one consistent change was made from level to incline conditions (i.e., a 10° decrease in knee flexion equilibrium angle). Furthermore, across subjects, several tuning parameters were constant. Proportionality constants governing knee swing initiation, decreasing knee stiffness and powered ankle plantarflexion in late stance were 1.0, 1.0 and 1.5 (Eq. 3), respectively. Final plantarflexion equilibrium angle in late stance was 12° for all subjects (Eq. 3). An exception was final knee flexion equilibrium angle, which varied (45-70°) across subjects. However, meaningful changes of this parameter were made to achieve ground clearance for various amputation levels, while the distance between the knee and ankle was fixed at its minimum value (i.e., a current device constraint) [26].
B. Trends of Prosthetic Knee and Ankle Mechanics
Kinematics and kinetics were favorable in their comparison to non-amputee data (relative to passive prostheses) and in support of scalable prosthesis function. Ankle range of motion from mid to late stance was between 25-30° across speeds on both level ground and the incline (Figs. 4 and 5). In addition, negative and positive ankle power in stance showed systematic increases with walking speed on both surfaces (Figs. 6 and 7). In particular, the peak magnitudes of mid-stance negative ankle power and late stance positive ankle power during level ground walking follow consistent trends across speeds and are comparable in magnitude at these walking velocities when compared to non-amputees [31]. Similarly, ankle plantarflexion torque in late stance showed consistent increases with increasing walking speed on both surfaces. The roles of muscles have been shown to primarily scale with respect to walking speed [32]. In mid to late stance, the plantarflexors provide vertical support and forward propulsion of the body, and increase their contributions as speed increases [32]. These data from mid to late stance suggest the prosthetic ankle with our control strategy behaved similarly.
During terminal stance, we observed a trend toward a more negative knee power and a more extensor knee torque as walking speed increased (Figs. 6 and 7). A significant speed main effect was observed for the terminal stance knee torque, with significant pairwise difference on the incline and pairwise difference approaching significance on level ground (slow to hurried  ). In fact, some subjects generated a knee extensor torque across all level ground speed conditions despite the control system modulating knee equilibrium angle from a relatively straight position to a flexed position (i.e., actively flexing the equilibrium position) (e.g., Fig. 2). These behaviors demonstrate the influence of dynamic coupling (e.g., [33]). In this phase of gait, a knee flexion acceleration was likely induced by the residual limb hip flexors, contributing to increased knee flexion velocities and damping contributions to the overall knee torque (Eq. 1) as speed increased. When these individuals walked slower, a more positive knee power and knee flexor torque were observed, perhaps as a result of less induced knee flexion.
). In fact, some subjects generated a knee extensor torque across all level ground speed conditions despite the control system modulating knee equilibrium angle from a relatively straight position to a flexed position (i.e., actively flexing the equilibrium position) (e.g., Fig. 2). These behaviors demonstrate the influence of dynamic coupling (e.g., [33]). In this phase of gait, a knee flexion acceleration was likely induced by the residual limb hip flexors, contributing to increased knee flexion velocities and damping contributions to the overall knee torque (Eq. 1) as speed increased. When these individuals walked slower, a more positive knee power and knee flexor torque were observed, perhaps as a result of less induced knee flexion.
When an individual walks up an incline at a steady walking speed, every stride results in an increase in the potential energy of the body. Thus, in addition to fulfilling typical roles associated with level walking, the demands on a prosthetic knee and/or ankle to facilitate this increase in energy are likely increased relative to level walking. In this study, we found the peak ankle plantarflexion torque to be greater on the incline at the two slowest speeds relative to level ground walking (Figs. 6 and 7), which may provide more support and forward propulsion to these users [34]. In addition, we found the slow and comfortable incline conditions to have a more positive knee power and flexor torque in terminal stance relative to over level ground (Figs. 6 and 7). In the hurried condition, a more knee flexor torque approached significance ( ). These data from the incline conditions suggest the prosthetic knee may be delivering more energy to the leg of these subjects (i.e., providing leg swing initiation) needed to increase their potential energy from step to step.
). These data from the incline conditions suggest the prosthetic knee may be delivering more energy to the leg of these subjects (i.e., providing leg swing initiation) needed to increase their potential energy from step to step.
C. Stance to Swing Transition
These data suggest that the timing of the stance to swing transition was appropriate. As speed increased, knee flexion angles were initiated earlier on both surfaces (Figs. 4 and 5). In addition, knee flexion velocities on the incline and ankle plantarflexion velocities on both surfaces increased with speed. Collectively, these results suggest the device was keeping up with each user as they increased speed. In our experience, the stance to swing transition can be a “sticking” point in terms of amputees feeling non-smooth behaviors of the device. In addition, a poorly-timed stance to swing transition can negatively affect their perception of whether or not the device will be ready for a subsequent heel strike. Thus, one intention of these strategies was to smooth the prosthesis response between late stance and swing flexion states (Fig. 1), while another was to initiate swing sufficiently early such that each user was confident the device would be prepared for a subsequent heel strike. Anecdotally, the feedback received from these individuals regarding this transition was consistently positive.
D. Study Limitations
Some knee and ankle kinematics and kinetics were inconsistent with previously published reports of non-amputees. For example, some increases of ankle dorsiflexion with increasing speed were shown (Figs. 4 and 5 compared to small decreases or no change of non-amputees [35], [36]). An explanation of this difference is that the offset of stance phase ankle stiffness Eq. 2 was not modulated in response to speed. However, studies have shown that electromyography of muscles spanning the ankle increases with speed [35]–[38]. In addition, other studies have found that increasing ankle muscle activity results in increased ankle stiffness (e.g., [39]). Thus, an improvement may be to apply an offset to Eq. 2 based on electromyography or a predicted stride velocity. Another difference was a substantially smaller peak ankle plantarflexion torque observed during stance (compare Fig. 6 to  -1.7 Nm/kg moments of non-amputees at comparable speeds [40], [41]), which is a known limitation of the current version of the device. A hard cap on the maximum ankle stiffness was applied due to a known electromechanical constraint on the peak current delivered to the motors. Newer generations of the device are anticipated to include a parallel ankle spring to offset the demand on the motor and increase the joint torque output (e.g., [18]).
-1.7 Nm/kg moments of non-amputees at comparable speeds [40], [41]), which is a known limitation of the current version of the device. A hard cap on the maximum ankle stiffness was applied due to a known electromechanical constraint on the peak current delivered to the motors. Newer generations of the device are anticipated to include a parallel ankle spring to offset the demand on the motor and increase the joint torque output (e.g., [18]).
E. Alternative Control Approaches
Alternative approaches (e.g., [42], [43]) to a finite state joint impedance style of control have been proposed in the literature for controlling robotic assistive devices during human gait, and have yielded positive results in transtibial (below-knee) amputees (e.g., [44]). In particular, when controlling an active prosthetic ankle, these have included an ankle positive force feedback strategy during terminal stance [42]. These approaches have been termed “artificial reflexes” and were inspired by previous literature of non-amputees (e.g., [45]). However, in addition to ankle control, the simultaneous control of a knee have not (to our knowledge) been demonstrated with these approaches. In this study, we constrained ourselves to the joint impedance style of control since it has shown good controllability in previous studies using this device. Furthermore, we integrated the simultaneous control of both a prosthetic ankle and knee. Finally, using our approach to provide knee swing initiation and powered ankle plantarflexion (Fig. 2, Eq. 3), straightforward changes of tuning parameters could be made by a prospective clinician, such as increasing final knee equilibrium angle to achieve more ground clearance. In contrast, the representation of how tuning positive force feedback gains may influence amputee gait mechanics is less intuitive.
F. Future Directions
Control algorithms of active prostheses should take into consideration that even these devices may be limited in fully replacing the functions of muscles (e.g., the complex roles of uniarticular and biarticular muscles, (e.g., [14]) These devices are composed of one actuator per each degree of freedom, as opposed to the muscle redundancy (i.e., multiple muscles spanning the same joint) of the musculoskeletal system. Control strategies that account for these differences across a range of locomotor activities may be needed to more positively influence amputee mobility and quality of life. Previous work has shown that joint-torque-actuated degrees-of-freedom can provide support and forward propulsion of the body [34]. However, there are likely differences in how they deliver energy among the body segments (e.g., delivering energy to the leg for swing initiation) when compared to muscles. Through modeling and simulation approaches (e.g., [13]), we hope to further quantify and optimize for these differences.
V. Conclusion
More effective and generalizable control strategies governing the behaviors of active lower-limb prostheses are needed to help realize their potential benefits. In summary, these developed algorithms for an active knee and ankle prosthesis were shown to generalize well across speeds, walking surfaces and users. They also displayed evidence of scalable device function and have the potential to reduce the challenge of clinically configuring such devices, increasing their viability during daily use.
Acknowledgment
The authors would like to thank Dr. Todd Kuiken, Robert Lipschutz, Suzanne Finucane, Kim Ingraham and Elizabeth Halsne for their contributions.
Biographies

Nicholas P. Fey (M’13) received the B.S.E., M.S.E., and Ph.D. degrees in mechanical engineering from the University of Texas at Austin, Austin, TX, USA, in 2006, 2008, and 2011, respectively.
He is currently a Post-Doctoral Research Fellow with the Center for Bionic Medicine, Rehabilitation Institute of Chicago, Chicago, IL, USA. He also has an appointment with the Department of Physical Medicine and Rehabilitation, Northwestern University, Chicago. His overall research interest is to identify how given task demands and lower-limb assistive devices contribute to human motor control and biomechanics. His previous projects have examined how walking speed and the stiffness of elastic prostheses alter the gait of below-knee amputees. He currently aims to enhance the mobility of above-knee amputees through the control of powered prostheses among various modes of ambulation.
Dr. Fey has been a member of the American Society of Biomechanics, since 2008, and the Biomedical Engineering Society. He has served as an Editorial Reviewer of the IEEE TRANSACTIONS ON BIOMEDICAL ENGINEERING journal and the IEEE Engineering in Medicine and Biology Conferences.

Ann M. Simon (M’12) received the B.S. degree in biomedical engineering from Marquette University, Milwaukee, WI, USA, in 2003, and the M.S. degree in mechanical engineering and the Ph.D. degree in biomedical engineering from the University of Michigan, Ann Arbor, MI, USA, in 2007 and 2008, respectively.
She is a Biomedical Engineering Manager with the Center for Bionic Medicine, Rehabilitation Institute of Chicago, Chicago, IL, USA, and a Research Assistant Professor with the Department of Physical Medicine and Rehabilitation, Northwestern University, Chicago. Her research interests include overcoming clinical challenges associated with the application of advanced pattern recognition myoelectric control systems for both upper- and lower-limb amputees.

Aaron J. Young (S’11) received the B.S. degree in biomedical engineering from Purdue University, West Lafayette, IN, USA in 2009, and the M.S. degree in biomedical engineering from Northwestern University, Chicago, IL, USA, in 2011.
He is currently pursuing the Ph.D. degree with the Center for Bionic Medicine, Rehabilitation Institute of Chicago, Northwestern University. His research interests include neural signal processing and pattern recognition using advanced machine learning techniques for the control of myoelectric prosthesis for the upper and lower limb.

Levi J. Hargrove (S’05–M’08) received the B.Sc., M.Sc., and Ph.D. degrees in electrical engineering from the University of New Brunswick, Fredericton, NB, Canada, in 2003, 2005, and 2007, respectively.
He joined the Center for Bionic Medicine (CBM) at the Rehabilitation Institute of Chicago, Chicago, IL, USA, in 2008. He is the Director of the Neural Engineering for Prosthetics and Orthotics Laboratory with CBM. He is also a Research Assistant Professor with the Department of Physical Medicine and Rehabilitation, Northwestern University, Chicago. His research interests include pattern recognition, biological signal processing, and myoelectric control of powered prostheses.
Dr. Hargrove is a member of the Association of Professional Engineers and Geoscientists of New Brunswick.
Funding Statement
This work was supported in part by the U.S. Army’s Telemedicine and Advanced Technology Research Center under Grant 81XWH-09-2-0020 and the U.S. Department of Education under Grant H133F130034.
References
- [1].Dillingham T. R., Pezzin L. E., and MacKenzie E. J., “Limb amputation and limb deficiency: Epidemiology and recent trends in the United States,” South Med. J., vol. 95, no. , pp. 875–883, Aug. 2002. [DOI] [PubMed] [Google Scholar]
- [2].Ziegler-Graham K., MacKenzie E. J., Ephraim P. L., Travison T. G., and Brookmeyer R., “Estimating the prevalence of limb loss in the United States: 2005 to 2050,” Archit. Phys. Med. Rehabil., vol. 89, no. 3, pp. 422–429, Mar. 2008. [DOI] [PubMed] [Google Scholar]
- [3].Robinson J. L., Smidt G. L., and Arora J. S., “Accelerographic, temporal, and distance gait factors in below-knee amputees,” Phys. Therapy, vol. 57, no. 8, pp. 898–904, Aug. 1977. [DOI] [PubMed] [Google Scholar]
- [4].Winter D. A. and Sienko S. E., “Biomechanics of below-knee amputee gait,” J. Biomech., vol. 21, no. 5, pp. 361–367, 1988. [DOI] [PubMed] [Google Scholar]
- [5].Waters R. L., Perry J., Antonelli D., and Hislop H., “Energy cost of walking of amputees: The influence of level of amputation,” J. Bone Joint Surgery Amer., vol. 58, no. 1, pp. 42–46, Jan. 1976. [PubMed] [Google Scholar]
- [6].Gailey R., Allen K., Castles J., Kucharik J., and Roeder M., “Review of secondary physical conditions associated with lower-limb amputation and long-term prosthesis use,” J. Rehabil. Res. Develop., vol. 45, no. 1, pp. 15–29, 2008. [DOI] [PubMed] [Google Scholar]
- [7].Burke M. J., Roman V., and Wright V., “Bone and joint changes in lower limb amputees,” Ann. Rheumatic Diseases, vol. 37, no. 3, pp. 252–254, Jun. 1978. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [8].Ephraim P. L., Wegener S. T., MacKenzie E. J., Dillingham T. R., and Pezzin L. E., “Phantom pain, residual limb pain, and back pain in amputees: Results of a national survey,” Archives Phys. Med. Rehabil., vol. 86, no. 10, pp. 1910–1919, Oct. 2005. [DOI] [PubMed] [Google Scholar]
- [9].Kulkarni J., Gaine W. J., Buckley J. G., Rankine J. J., and Adams J., “Chronic low back pain in traumatic lower limb amputees,” Clin. Rehabil., vol. 19, no. 1, pp. 81–86, Jan. 2005. [DOI] [PubMed] [Google Scholar]
- [10].Smith D. G., Ehde D. M., Legro M. W., Reiber G. E., del Aguila M., and Boone D. A., “Phantom limb, residual limb, and back pain after lower extremity amputations,” Clin. Orthopaedics Rel. Res., vol. 361, pp. 29–38, Apr. 1999. [DOI] [PubMed] [Google Scholar]
- [11].Winter D. A., The Biomechanics and Motor Control of Human Gait: Normal, Elderly and Pathological. Waterloo, ON, Canada: Univ. Waterloo Press, 1991. [Google Scholar]
- [12].Anderson F. C. and Pandy M. G., “Individual muscle contributions to support in normal walking,” Gait Posture, vol. 17, no. 2, pp. 159–169, Apr. 2003. [DOI] [PubMed] [Google Scholar]
- [13].Liu M. Q., Anderson F. C., Pandy M. G., and Delp S. L., “Muscles that support the body also modulate forward progression during walking,” J. Biomech., vol. 39, no. 14, pp. 2623–2630, 2006. [DOI] [PubMed] [Google Scholar]
- [14].Neptune R. R., Zajac F. E., and Kautz S. A., “Muscle force redistributes segmental power for body progression during walking,” Gait Posture, vol. 19, no. 2, pp. 194–205, Apr. 2004. [DOI] [PubMed] [Google Scholar]
- [15].Neptune R. R., Kautz S. A., and Zajac F. E., “Contributions of the individual ankle plantar flexors to support, forward progression and swing initiation during walking,” J. Biomech., vol. 34, no. 11, pp. 1387–1398, Nov. 2001. [DOI] [PubMed] [Google Scholar]
- [16].Allen J. L. and Neptune R. R., “Three-dimensional modular control of human walking,” J. Biomech., vol. 45, no. 12, pp. 2157–2163, Aug. 2012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [17].Au S. K., Herr H., Weber J., and Martinez-Villalpando E. C., “Powered ankle-foot prosthesis for the improvement of amputee ambulation,” in Proc. 29th Annu. Int. Conf. IEEE Eng. Med. Biol. Soc., Aug. 2007, pp. 3020–3026. [DOI] [PubMed] [Google Scholar]
- [18].Au S. K. and Herr H., “Powered ankle-foot prosthesis,” IEEE Robot. Autom. Mag., vol. 15, no. 3, pp. 52–59, Sep. 2008. [Google Scholar]
- [19].Au S. K., Weber J., and Herr H., “Biomechanical design of a powered ankle-foot prosthesis,” in Proc. IEEE 10th Int. Conf. Rehabil. Robot., Jun. 2007, pp. 298–303. [Google Scholar]
- [20].Fite K., Mitchell J., Sup F., and Goldfarb M., “Design and control of an electrically powered knee prosthesis,” in Proc. IEEE 10th Int. Conf. Rehabil. Robot., Jun. 2007, pp. 902–905. [Google Scholar]
- [21].Sup F., Bohara A., and Goldfarb M., “Design and control of a powered knee and ankle prosthesis,” in Proc. IEEE Int. Conf. Robot. Autom., Apr. 2007, pp. 4134–4139. [Google Scholar]
- [22].Sup F., Bohara A., and Goldfarb M., “Design and control of a powered transfemoral prosthesis,” Int. J. Robot. Res., vol. 27, no. 2, pp. 263–273, Feb. 2008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [23].Au S., Berniker M., and Herr H., “Powered ankle-foot prosthesis to assist level-ground and stair-descent gaits,” Neural Netw., vol. 21, no. 4, pp. 654–666, May 2008. [DOI] [PubMed] [Google Scholar]
- [24].Sup F., Varol H. A., Mitchell J., Withrow T. J., and Goldfarb M., “Preliminary evaluations of a self-contained anthropomorphic transfemoral prosthesis,” IEEE/ASME Trans. Mechatronics, vol. 14, no. 6, pp. 667–676, Dec. 2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [25].Hargrove L. J., et al. , “Robotic leg control with EMG decoding in an amputee with nerve transfers,” New England J. Med., vol. 369, no. 13, pp. 1237–1242, Sep. 2013. [DOI] [PubMed] [Google Scholar]
- [26].Lawson B., Varol H. A., Huff A., Erdemir E., and Goldfarb M., “Control of stair ascent and descent with a powered transfemoral prosthesis,” IEEE Trans. Neural Syst. Rehabil. Eng., vol. 21, no. 3, pp. 466–473, May 2013. [DOI] [PubMed] [Google Scholar]
- [27].Sup F., Varol H. A., and Goldfarb M., “Upslope walking with a powered knee and ankle prosthesis: Initial results with an amputee subject,” IEEE Trans. Neural Syst. Rehabil. Eng., vol. 19, no. 1, pp. 71–78, Feb. 2011. [DOI] [PubMed] [Google Scholar]
- [28].Rouse E. J., Hargrove L. J., Akhtar A., and Kuiken T. A., “Validation of methods for determining ankle stiffness during walking using the perturberator robot,” in Proc. 4th IEEE Ras Embs Int. Conf. Biomed. Robot. Biomechatron. (Biorob), Jun. 2012, pp. 1650–1655. [Google Scholar]
- [29].Rouse E. J., Hargrove L. J., Perreault E. J., and Kuiken T. A., “Estimation of human ankle impedance during walking using the perturberator robot,” in Proc. 4th IEEE Ras Embs Int. Conf. Biomed. Robot. Biomechatron. (Biorob), Jun. 2012, pp. 373–378. [Google Scholar]
- [30].Hafner B. J., Willingham L. L., Buell N. C., Allyn K. J., and Smith D. G., “Evaluation of function, performance, and preference as transfemoral amputees transition from mechanical to microprocessor control of the prosthetic knee,” Archives Phys. Med. Rehabil., vol. 88, no. 2, pp. 207–217, Feb. 2007. [DOI] [PubMed] [Google Scholar]
- [31].Silverman A. K., Fey N. P., Portillo A., Walden J. G., Bosker G., and Neptune R. R., “Compensatory mechanisms in below-knee amputee gait in response to increasing steady-state walking speeds,” Gait Posture, vol. 28, no. 4, pp. 602–609, Nov. 2008. [DOI] [PubMed] [Google Scholar]
- [32].Neptune R. R., Sasaki K., and Kautz S. A., “The effect of walking speed on muscle function and mechanical energetics,” Gait Posture, vol. 28, no. 1, pp. 135–143, Jul. 2008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [33].Zajac F. E., Neptune R. R., and Kautz S. A., “Biomechanics and muscle coordination of human walking: Part I: Introduction to concepts, power transfer, dynamics and simulations,” Gait Posture, vol. 16, no. 3, pp. 215–232, Dec. 2002. [DOI] [PubMed] [Google Scholar]
- [34].Kepple T. M., Siegel K. L., and Stanhope S. J., “Relative contributions of the lower extremity joint moments to forward progression and support during gait,” Gait Posture, vol. 6, no. 1, pp. 1–8, 1997. [Google Scholar]
- [35].Murray M. P., Mollinger L. A., Gardner G. M., and Sepic S. B., “Kinematic and EMG patterns during slow, free, and fast walking,” J. Orthopaedic Res., vol. 2, no. 3, pp. 272–280, 1984. [DOI] [PubMed] [Google Scholar]
- [36].Pepin A., Norman K. E., and Barbeau H., “Treadmill walking in incomplete spinal-cord-injured subjects: 1. Adaptation to changes in speed,” Spinal Cord, vol. 41, no. 5, pp. 257–270, May 2003. [DOI] [PubMed] [Google Scholar]
- [37].Yang J. F. and Winter D. A., “Surface EMG profiles during different walking cadences in humans,” Electroencephalogr. Clin. Neurophysiol., vol. 60, no. 6, pp. 485–491, Jun. 1985. [DOI] [PubMed] [Google Scholar]
- [38].Fey N. P., Silverman A. K., and Neptune R. R., “The influence of increasing steady-state walking speed on muscle activity in below-knee amputees,” J. Electromyogr. Kinesiol., vol. 20, no. 1, pp. 155–161, Feb. 2010. [DOI] [PubMed] [Google Scholar]
- [39].Lee H., Krebs H. I., and Hogan N., “Multivariable dynamic ankle mechanical impedance with active muscles,” IEEE Trans. Neural Syst. Rehabil. Eng., 2014, to be published. [DOI] [PMC free article] [PubMed]
- [40].Sanderson D. J. and Martin P. E., “Lower extremity kinematic and kinetic adaptations in unilateral below-knee amputees during walking,” Gait Posture, vol. 6, no. 2, pp. 126–136, 1997. [Google Scholar]
- [41].Goldberg S. R. and Stanhope S. J., “Sensitivity of joint moments to changes in walking speed and body-weight-support are interdependent and vary across joints,” J. Biomech., vol. 46, no. 6, pp. 1176–1183, Apr. 2013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [42].Eilenberg M. F., Geyer H., and Herr H., “Control of a powered ankle-foot prosthesis based on a neuromuscular model,” IEEE Trans. Neural Syst. Rehabil. Eng., vol. 18, no. 2, pp. 164–173, Apr. 2010. [DOI] [PubMed] [Google Scholar]
- [43].Vallery H., van Asseldonk E. H. F., Buss M., and van der Kooij H., “Reference trajectory generation for rehabilitation robots: Complementary limb motion estimation,” IEEE Trans. Neural Syst. Rehabil. Eng., vol. 17, no. 1, pp. 23–30, Feb. 2009. [DOI] [PubMed] [Google Scholar]
- [44].Herr H. M. and Grabowski A. M., “Bionic ankle-foot prosthesis normalizes walking gait for persons with leg amputation,” Proc. Roy. Soc. B, Biol. Sci., vol. 279, pp. 457–464, Feb. 2012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [45].Grey M. J., Nielsen J. B., Mazzaro N., and Sinkjaer T., “Positive force feedback in human walking,” J. Physiol., vol. 581, pp. 99–105, May 2007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [46].Fey N. P. and Neptune R. R., “3D intersegmental knee loading in below-knee amputees across steady-state walking speeds,” Clin. Biomech., vol. 27, no. 4, pp. 409–414, May 2012. [DOI] [PubMed] [Google Scholar]