Abstract
Vascular cognitive impairment (VCI) is a heterogeneous group of diseases linked together by cerebrovascular disease. Treatment of VCI has been hindered by the lack of a coherent pathophysiological process that could provide molecular targets. Of the several forms of VCI, the small vessel disease form is both the most prevalent and generally has a progressive course. Binswanger’s disease (BD) is the small vessel form of VCI that involves extensive injury to the deep white matter. Growing evidence suggests that there is disruption of the blood–brain barrier (BBB) secondary to an inflammatory state. Matrix metalloproteinases (MMPs) are increased in the brain and CSF of patients with BD, and have been shown to disrupt the BBB in animal studies, suggesting that they may be biomarkers and therapeutic targets. Multimodal biomarkers derived from clinical, neuropsychological, imaging, and biochemical data can be used to narrow the VCI population to the progressive inflammatory form that will be optimal for treatment trials. This review describes the role of the MMPs in pathophysiology and their use as biomarkers.
Keywords: Vascular cognitive impairment, Blood–brain barrier, White matter, Neuroinflammation, MRI
Introduction
Vascular cognitive impairment (VCI) includes all forms of vascular disease leading to dementia (Gorelick et al. 2011; O’Brien et al. 2003). There is general agreement that small vessel disease (SVD), which is the most common form of VCI, has a progressive course, making it optimal for treatment trials (Erkinjuntti et al. 2004; Roman et al. 2010). Binswanger’s disease (BD) is a form of SVD with extensive injury to the deep white matter, but diagnosis is challenging because it is a spectrum disorder, lacking unified criteria (Babikian and Ropper 1987; Bennett et al. 1990; Caplan 1995; Fisher 1989; Roman et al. 2002). Therefore, it is necessary to develop criteria for the selection of these patients based on characteristic disease markers, including clinical, imaging, and biochemical data, which in combination can improve diagnostic accuracy, particularly for early detection when treatment trials would be most beneficial (Gorelick et al. 2011; Rosenberg et al. 2015).
Neuroinflammation appears to play a role in BD based on the finding of disruption of the blood–brain barrier (BBB), which is shown by an increase in albumin in the cerebrospinal fluid (CSF) compared to serum (Blennow et al. 1990; Wallin et al. 1990), and the more recent MRI studies with dynamic contrast-enhanced MRI (DCEMRI) (Taheri et al. 2011). Evidence from human and animal suggests that matrix metalloproteinases (MMPs) contribute to BBB opening, demyelination, and oligodendrocyte death (Jalal et al. 2012; Nakaji et al. 2006; Sood et al. 2009).
While there remains controversy about which patients to diagnose with BD, there is a need to develop criteria for diagnosis of patients, and for the optimal set of surrogate markers. The progressive form of BD is the optimal form for that purpose, and disruption of the BBB, growth of deep white matter lesions, and neuropsychological test scores provide potential markers. MMPs contribute to both the opening of the BBB and growth of the white matter lesions. Understanding the mechanism of action of MMPs in VCI could provide clues to the pathogenesis of the illness and indicate potential molecular targets to reduce damage to the white matter. This review will focus the role of MMPs as biomarkers of neuroinflammation in BD.
Chronic Vascular Disease
Large vessel disease (LVD) is a sporadic illness with step-wise progression leading to multi-infarct dementia (MID) that will be difficult to study (Hachinski 1991). On the other hand, SVD of the BD type is progressive, making it possible to discern the natural history. Currently, there is little epidemiological data on the relative prevalence of LVD versus SVD. Autopsy studies of vascular dementia patients suggest that SVD is the most prevalent form of VCI (Schneider et al. 2007). In one such study from Lund University in Sweden, involving all vascular dementia patients within a specific catchment area from 1979 to 1995, 175 of these came to autopsy. They found that SVD was the most prevalent form although many patients had multiple pathologies with LVD, SVD, and Alzheimer’s disease (AD); pure SVD was found in 36, while pure LVD was only found in 7, and in the multiple disease group, SVD was found in 110 of 126 patients (Andin et al. 2005). A more recent pathological report from the AD consortium found a high incidence of small infarcts in the AD patients by the time they reached autopsy (Toledo et al. 2013). A large autopsy series of relatively normal persons showed that by the time of death, it was common to have pathological protein accumulations of β-amyloid, α-synuclein, along with microinflarcts (Sonnen et al. 2011).
A number of reports have described the clinical and radiological findings in BD, leading to improved diagnosis during life (Babikian and Ropper 1987; Caplan and Schoene 1978; Fisher 1989; Rosenberg et al. 1979). Separation of BD patients from other forms of neurodegenerative diseases and white matter changes of aging, which is challenging, can be facilitated by the use of biomarkers derived from clinical factors multimodal imaging, and biochemical analysis of the blood and CSF (Rosenberg et al. 2015).
Pathophysiology of White Matter Injury in BD
White matter structural damage is the hallmark of BD pathology and the basis for the clinical features, including executive dysfunction on neuropsychological testing, hyperreflexia, gait, and continence problems. Because WMHs on FLAIR MRI are common in normal elderly, a number of other MRI methods have been employed to define regions of ischemic injury. Proton magnetic resonance spectroscopy (1H-MRS) shows a reduced signal from N-acetylaspartate (NAA) and creatine (Cr) in injured white matter in BD patients compared to controls (Brooks et al. 1997; Sappey Marinier et al. 1992). Similarly, diffusion tensor imaging (DTI) shows structural white matter damage in patients with VCI (Kantarci et al. 2011; Maillard et al. 2013; Nitkunan et al. 2008; O’Sullivan et al. 2004). More importantly, DTI is abnormal both within WMHs and in normal appearing white matter (Maniega et al. 2015; O’Sullivan et al. 2004). Several studies have found increased BBB permeability in lacunar stroke and BD (Hanyu et al. 2002; Taheri et al. 2011; Topakian et al. 2010; Wardlaw et al. 2013).
One theory of the mechanism of white matter damage in SVD postulates hypoxic hypoperfusion of the deep white matter, which is a watershed area that is poorly supplied with blood, making it a vulnerable region (De Reuck et al. 1980). Initially, there is damage to the white matter most likely from long-standing hypertension beginning in mid-life (Maillard et al. 2012). The underlying mechanism of these early changes in the white matter is poorly understood. Long-standing hypertension narrows the vessel lumen and thickens the vessel wall from the fibrosis (Rigsby et al. 2007), compromising cerebral blood flow in the deep white matter (Yonas et al. 1985). Hypertension damages the small vessels leading to thrombosis, which could damage the white matter through a series of small strokes (Smith et al. 2012).
Proteases in Inflammation
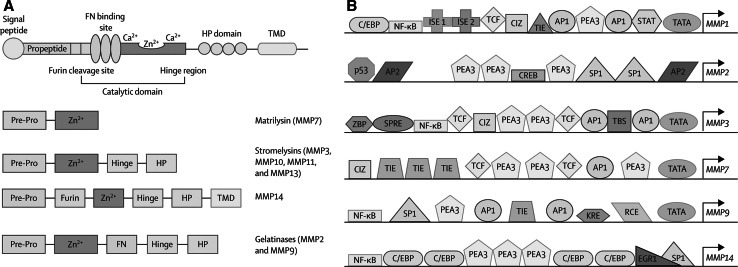
MMPs are a family of 26 extracellular and intracellular matrix-degrading enzymes that are important in development, tumor growth, brain development and injury (Rivera et al. 2010). They are generally classified into major groups based on the molecular components linked together to form the enzyme (Fig. 1) (Rosenberg 2009). Extracellular matrix is the primary site of action, but they are also involved in releasing molecules from the cell surface, cell signaling, and cell death (Yong 2005). The two major constitutive enzymes, gelatinase A (MMP-2) and membrane type-1 metalloproteinase (MMP-14), are present in latent forms normally, becoming active in hypoxic/ischemic injury (Table 1). Activation of proMMP-2 requires the action of the trimolecular complex formed by proMMP-2, tissue inhibitor to metalloproteinases-2 (TIMP-2), and MMP-14, which act close to the membrane due to the membrane bound state of MMP-14 (Strongin et al. 1995). This tethering limits the action of MMP-2 to the region close to the site of activation near the blood vessel. MMP-2 initiates the disruption of the basal lamina proteins and the opening of the tight junction proteins (Fig. 2) (Yang et al. 2007). In the acute injury, this initial phase is reversible and most likely short-lived. However, as hypoxia progresses, the inducible MMPs, stromelysin-1 (MMP-3) and gelatinase B (MMP-9), are released and activated. This starts the more disruptive phase, which generally causes irreversible damage.
Fig. 1.
MMP protein structure and binding sites on the promoters of the MMP genes. a A signal peptide and the propeptide region form part of the cysteine switch, which folds over the zinc in the catalytic site and maintains a latent state. A cleavage site enables the proconvertase furin to activate MMP by cleaving the propeptide. An FN binding site is present in MMP2 and MMP9, connecting them with the basal lamina. The catalytic zinc site is present in all MMPs. A haemopexin domain is joined to the catalytic site by the hinge region. MMP14 (an MT-MMP) has a TMD. b MMP2 has AP2 and SP1 binding sites, which are found in promoter regions of constitutively expressed enzymes. MMP1, MMP3, MMP7, and MMP9 have AP1 binding sites, which are responsive to oncogenes and cytokines, consistent with their expression during inflammation. NF-κB is another inflammatory binding site. AP activator protein, C/EBP CCAAT/enhancer-binding protein, CIZ CAS-interacting zinc-finger protein, CREB cyclic AMP response-element binding protein, EGR early growth response site, FN fibronectin, HP haemopexin, ISE immortalisation-sensitive element, KRE keratinocyte differentiation-factor responsive element, MMP matrix metalloproteinase, MT membrane type, NF-κB nuclear factor-κB, PEA polyomavirus enhancer-A binding-protein, Pre-Pro prepropeptide, RCE retinoblastoma control element, SP specificity protein, SPRE stromelysin-1 platelet-derived growth factor β responsive element, STAT signal transducer and activator of transcription, TBS translocation-ETS-leukaemia binding site, TCF T cell factor site, TIE transforming growth factor β inhibitory element, TMD transmembrane domain, ZBP zinc-binding protein (Rosenberg 2009)
Table 1.
Matrix metalloproteinases in CSF and brain
| MMPs | Molecular weight (kD) | Normal CSF concentration | Brain cells | |
|---|---|---|---|---|
| Latent | Active | |||
| Gelatinases | ||||
| A (MMP-2) | 72 | 62 | High | Astrocytes |
| B (MMP-9) | 92 | 82 | Low | Microglia and endothelial cells |
| Stromelysin | ||||
| MMP-3 | 54 | 43 | Low | Macrophages and pericytes |
| MMP-14 | 78–85 (variable) | ND | Not measured | Astrocytes |
Fig. 2.
a Confocal immunohistochemistry shows glial fibrillary acidic protein (GFAP)-positive astrocytes around a vessel (V) that expresses MMP-2 (arrows) in intact rat brain tissue. The arrowheads indicate the astrocyte end feet around the vessel. b Expression of furin and MT-MMP immunohistochemistry in brain cells. c Immunostaining for MMP-2 (red) and MT1-MMP (green). Merging the two images shows the co-localization (yellow). d Schematic drawing to show that the activation of MMP-2 occurs through the action of the trimolecular complex during the early opening of the BBB in 3-h reperfusion after 90-min middle cerebral artery occlusion (MCAO). In the astrocytic foot processes (AFP), the MT-MMP joins with TIMP-2 to activate proMMP-2 in a spatially constrained manner close to the basal lamina (BL). In the PL are the pericytes (PC). The endothelial cells (EC) have tight junctions (TJ). The activated MMP-2 has direct access to the portion of the BL beneath the AFP and components of the BL are degraded. When the hypoxia is prolonged and severe, there is exprssion of MMP-9 and MMP-3, leading to further tissue destruction (Candelario-Jalil et al. 2009) (Color figure online)
The pathology of BD involves extensive injury to the white matter with gliosis and oligodendrocyte loss, fibrosis of the arterioles, inflammation around the arterioles, and MMPs in reactive astrocytes and microglia/macrophages (Fig. 3). The MMPs have two actions that make them particularly damaging in the chronic hypoxia-driven injuries: they attack the components of the neurovascular unit, including the basal lamina proteins and tight junction proteins, and in addition they break down myelin, leading to demyelination and oligodendrocyte damage (Chandler et al. 1995). This nonimmunological disruption of myelin by proteases was first shown to occur with plasmin and was termed “by-stander” demyelination (Cammer et al. 1978).
Fig. 3.
MRI scan and histological samples from patients with small-vessel form of vascular cognitive impairment or Binswanger’s disease. a Fluid attenuated inversion recovery MRI scan showing extensive white matter lesions in the periventricular region. b Histological section from the white matter in a patient with Binswanger’s disease shows extensive gliosis. The arrow shows an astrocyte immunostained with glial fibrillary acidic protein. c Fibrotic blood vessel surrounded by inflammatory macrophages. d Blood vessel exposed to long-term hypertension with damage to the blood vessel wall and eosinophilic deposits. e Immunostaining with antibodies to MMP-2 detects an immunoreactive astrocyte (arrow). f Immunostaining with antibodies to MMP-3 detects an MMP-3-positive macrophage around a fibrotic blood vessel in a demyelinated area (arrow). MMP matrix metalloproteinase (Rosenberg 2009)
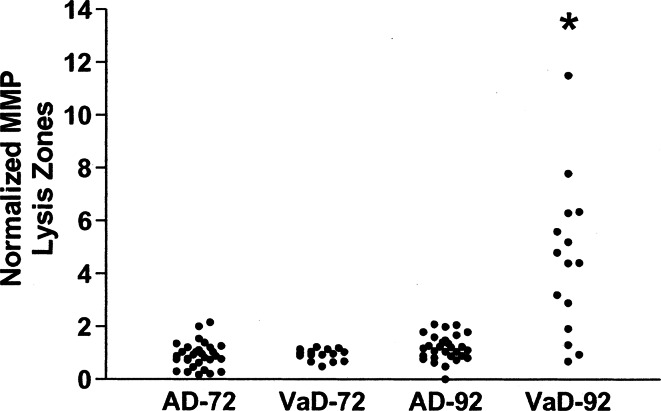
Another line of evidence implicating the MMPs in BD comes from studies of the MMPs in CSF. Several highly sensitive methods are available to study MMP proteins in the CSF, including gelatin zymography and ELISA. Levels of MMP-9 are elevated in VCI patients and not in those with Alzheimer’s disease (Fig. 4) (Adair et al. 2004). Since some of the protein may cross the BBB from the blood, a more accurate measurement of endogenous brain production can be obtained by normalizing the MMPs in CSF and serum with albumin in each of those compartments. Since albumin is a large liver-derived protein that has limited access to the CSF unless the BBB is compromised, it can be used to form the MMP Index, which is similar to the method used to calculate the IgG Index in the multiple sclerosis panel (Liuzzi et al. 2002). Indexing the MMP-2 and MMP-9 shows a reduction in the MMP-2 index. In addition, using fluorescent assay the active form of MMP-3 can be shown to be elevated in VCI patients (Fig. 5) (Candelario-Jalil et al. 2011; Hawkins et al. 2013). The reduction on MMP-2 index was seen only in the VCI patients with the BD form of the illness. One reason for the reduction in MMP-2 index is that gelatin zymograms measure latent MMP-2, suggesting that the latent form was being converted to the active form, resulting in a reduction of the MMP-2 index. In the original study, using the indexing method, we failed to find an increase in MMP-9 index. More recently, in a larger series of patients, the MMP-9 index was elevated in BD (unpublished data). High levels of MMP-2 are normally present in the CSF, while MMP-9 is not found. When inflammation occurs, an increase in MMP-9 can be detected even if the change is small. On the other hand, MMP-2 is already found at high concentration in CSF, and changes are more difficult to detect. Another group, using the ELISA method to measure MMP-9, has confirmed elevated levels of MMP-9 in CSF in VCI (Bjerke et al. 2014).
Fig. 4.
Comparison of normalized values of MMP-9 between patients with AD and VaD. A significant elevation of the levels of MMP-9 is seen in the VaD group compared with those with AD (P < 0.0001). No difference in MMP-2 was observed (Adair et al. 2004)
Fig. 5.
Cerebrospinal fluid values for the matrix metalloproteinase-2 (MMP-2) index and MMP-3 activity. a MMP-2 index for VCI and control subjects. The MMP-2 index for the VCI group was significantly lower than controls. b MMP-3 activity is increased compared with control subjects (P < 0.01). Significance levels are shown in parentheses. c MMP-2 index values for all patients with VCI (open circles) and control subjects (black dots) are plotted against albumin index (Qalb). There was a significant negative correlation (P < 0.0001). d MMP-9 index plotted similarly failed to show a correlation. VCI vascular cognitive impairment (Candelario-Jalil et al. 2011)
Stromelysin-1 (MMP-3) is found mainly in macrophage-like cells, and has an important role in tissue breakdown during the injury phase and in angiogenesis in the recovery phase; the pericytes contain MMP-3, which is important in angiogenesis (Yang et al. 2013). Patients with BD have decreased MMP-2 index and increased MMP-3 activity, which improves separation of BD and controls (Candelario-Jalil et al. 2011).
Tissue inhibitors of the metalloproteinases (TIMPs) are the endogenous MMP inhibitors (Brew and Nagase 2010). The TIMPs form complexes with the MMPs that complicate the measurements of the TIMPs in the CSF. Few studies have examined the role of the TIMPs in the MMPs. In one study, there was a increase in TIMP-1 in CSF in VCI (Bjerke et al. 2011).
Since BD is a spectrum disorder with the diagnosis made when a number of the factors are present, a BD score can be constructed from the relevant factors; several factors are related to neuroinflammation, including elevated albumin ratio (a biomarker of BBB opening), increased permeability using dynamic contrast-enhanced MRI, and the MMP indexes (Rosenberg et al. 2015). Three MMPs are altered in the CSF during inflammation: elevated MMP-9 index, reduced MMP-2 index, and activated MMP-3. Other MMPs may be found to be important as improvements in the sensitivity of assay systems in the CSF are developed. Neurofilament light and sulfatide are biomarkers of myelin breakdown (Bjerke et al. 2014).
Conclusion
BD is a progressive form of VCI that is most compatible with clinical trials since the natural history can be determined and compared to drug treatments. However, the challenge remains to separate those with BD from VCI patients. Biomarkers provide the best method and many investigators are attempting to identify the optimal set of biomarkers. MMPs contribute to the inflammation by disrupting the basal lamina and tight junction proteins. Since neuroinflammation is an important part of the pathophysiology, the measurement of MMPs in the CSF, which can be done with sensitive zymography and ELISA methods, provides an indication of the pathological processes occurring in the brain. Biomarkers based on the pathophysiology provide a guide to future treatments.
Acknowledgments
The study was supported by Funding from NIH (RO1 NS052305-07), the US-Israeli Binational Foundation, and Bayer Pharmaceutical Corp (GR), and the University of New Mexico NIH National Center for Advancing Translational Sciences (UL1 TR000041).
References
- Adair JC, Charlie J, Dencoff JE, Kaye JA, Quinn JF, Camicioli RM, Stetler-Stevenson WG, Rosenberg GA (2004) Measurement of gelatinase B (MMP-9) in the cerebrospinal fluid of patients with vascular dementia and Alzheimer disease. Stroke 35:e159–e162 [DOI] [PubMed] [Google Scholar]
- Andin U, Gustafson L, Passant U, Brun A (2005) A clinico-pathological study of heart and brain lesions in vascular dementia. Dement Geriatr Cogn Disord 19:222–228 [DOI] [PubMed] [Google Scholar]
- Babikian V, Ropper AH (1987) Binswanger’s disease: a review. Stroke 18:2–12 [DOI] [PubMed] [Google Scholar]
- Bennett DA, Wilson RS, Gilley DW, Fox JH (1990) Clinical diagnosis of Binswanger’s disease [see comments]. J Neurol Neurosurg Psychiatry 53:961–965 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bjerke M, Zetterberg H, Edman A, Blennow K, Wallin A, Andreasson U (2011) Cerebrospinal fluid matrix metalloproteinases and tissue inhibitor of metalloproteinases in combination with subcortical and cortical biomarkers in vascular dementia and Alzheimer’s disease. J Alzheimers Dis 27:665–676 [DOI] [PubMed] [Google Scholar]
- Bjerke M, Jonsson M, Nordlund A, Eckerstrom C, Blennow K, Zetterberg H, Pantoni L, Inzitari D, Schmidt R, Wallin A (2014) Cerebrovascular biomarker profile is related to white matter disease and ventricular dilation in a LADIS substudy. Dement Geriatr Cogn Disord Extra 4:385–394 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Blennow K, Wallin A, Fredman P, Karlsson I, Gottfries CG, Svennerholm L (1990) Blood-brain barrier disturbance in patients with Alzheimer’s disease is related to vascular factors. Acta Neurol Scand 81:323–326 [DOI] [PubMed] [Google Scholar]
- Brew K, Nagase H (2010) The tissue inhibitors of metalloproteinases (TIMPs): an ancient family with structural and functional diversity. Biochim Biophys Acta 1803:55–71 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brooks WM, Wesley MH, Kodituwakku PW, Garry PJ, Rosenberg GA (1997) 1H-MRS differentiates white matter hyperintensities in subcortical arteriosclerotic encephalopathy from those in normal elderly. Stroke 28:1940–1943 [DOI] [PubMed] [Google Scholar]
- Cammer W, Bloom BR, Norton WT, Gordon S (1978) Degradation of basic protein in myelin by neutral proteases secreted by stimulated macrophages: a possible mechanism of inflammatory demyelination. Proc Natl Acad Sci USA 75:1554–1558 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Candelario-Jalil E, Yang Y, Rosenberg GA (2009) Diverse roles of matrix metalloproteinases and tissue inhibitors of metalloproteinases in neuroinflammation and cerebral ischemia. Neuroscience 158:983–994 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Candelario-Jalil E, Thompson J, Taheri S, Grossetete M, Adair JC, Edmonds E, Prestopnik J, Wills J, Rosenberg GA (2011) Matrix metalloproteinases are associated with increased blood-brain barrier opening in vascular cognitive impairment. Stroke 42:1345–1350 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Caplan LR (1995) Binswanger’s disease–revisited. [Review]. Neurology 45:626–633 [DOI] [PubMed] [Google Scholar]
- Caplan LR, Schoene WC (1978) Clinical features of subcortical arteriosclerotic encephalopathy (Binswanger disease). Neurology 28:1206–1215 [DOI] [PubMed] [Google Scholar]
- Chandler S, Coates R, Gearing A, Lury J, Wells G, Bone E (1995) Matrix metalloproteinases degrade myelin basic protein. Neurosci Lett 201:223–226 [DOI] [PubMed] [Google Scholar]
- De Reuck J, Crevits L, De Coster W, Sieben G, vander Eecken H (1980) Pathogenesis of Binswanger chronic progressive subcortical encephalopathy. Neurology 30:920–928 [DOI] [PubMed] [Google Scholar]
- Erkinjuntti T, Roman G, Gauthier S, Feldman H, Rockwood K (2004) Emerging therapies for vascular dementia and vascular cognitive impairment. Stroke 35:1010–1017 [DOI] [PubMed] [Google Scholar]
- Fisher CM (1989) Binswanger’s encephalopathy: a review. J Neurol 236:65–79 [DOI] [PubMed] [Google Scholar]
- Gorelick PB, Scuteri A, Black SE, Decarli C, Greenberg SM, Iadecola C, Launer LJ, Laurent S, Lopez OL, Nyenhuis D et al (2011) Vascular contributions to cognitive impairment and dementia: a statement for healthcare professionals from the American Heart Association/American Stroke Association. Stroke 42:2672–2713 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hachinski VC (1991) Multi-infarct dementia: a reappraisal. [Review]. Alzheimer Dis Assoc Disord 5:64–68 [DOI] [PubMed] [Google Scholar]
- Hanyu H, Asano T, Tanaka Y, Iwamoto T, Takasaki M, Abe K (2002) Increased blood-brain barrier permeability in white matter lesions of Binswanger’s disease evaluated by contrast-enhanced MRI. Dement Geriatr Cogn Disord 14:1–6 [DOI] [PubMed] [Google Scholar]
- Hawkins KE, DeMars KM, Yang C, Rosenberg GA, Candelario-Jalil E (2013) Fluorometric immunocapture assay for the specific measurement of matrix metalloproteinase-9 activity in biological samples: application to brain and plasma from rats with ischemic stroke. Mol Brain 6:14 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jalal FY, Yang Y, Thompson J, Lopez AC, Rosenberg GA (2012) Myelin loss associated with neuroinflammation in hypertensive rats. Stroke 43:1115–1122 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kantarci K, Senjem ML, Avula R, Zhang B, Samikoglu AR, Weigand SD, Przybelski SA, Edmonson HA, Vemuri P, Knopman DS et al (2011) Diffusion tensor imaging and cognitive function in older adults with no dementia. Neurology 77:26–34 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liuzzi GM, Trojano M, Fanelli M, Avolio C, Fasano A, Livrea P, Riccio P (2002) Intrathecal synthesis of matrix metalloproteinase-9 in patients with multiple sclerosis: implication for pathogenesis. Mult Scler 8:222–228 [DOI] [PubMed] [Google Scholar]
- Maillard P, Seshadri S, Beiser A, Himali JJ, Au R, Fletcher E, Carmichael O, Wolf PA, DeCarli C (2012) Effects of systolic blood pressure on white-matter integrity in young adults in the Framingham Heart Study: a cross-sectional study. Lancet Neurol 11:1039–1047 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Maillard P, Carmichael O, Harvey D, Fletcher E, Reed B, Mungas D, DeCarli C (2013) FLAIR and diffusion MRI signals are independent predictors of white matter hyperintensities. AJNR 34:54–61 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Maniega SM, Valdes Hernandez MC, Clayden JD, Royle NA, Murray C, Morris Z, Aribisala BS, Gow AJ, Starr JM, Bastin ME et al (2015) White matter hyperintensities and normal-appearing white matter integrity in the aging brain. Neurobiol Aging 36:909–918 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nakaji K, Ihara M, Takahashi C, Itohara S, Noda M, Takahashi R, Tomimoto H (2006) Matrix metalloproteinase-2 plays a critical role in the pathogenesis of white matter lesions after chronic cerebral hypoperfusion in rodents. Stroke 37:2816–2823 [DOI] [PubMed] [Google Scholar]
- Nitkunan A, Charlton RA, McIntyre DJ, Barrick TR, Howe FA, Markus HS (2008) Diffusion tensor imaging and MR spectroscopy in hypertension and presumed cerebral small vessel disease. Magn Reson Med 59:528–534 [DOI] [PubMed] [Google Scholar]
- O’Brien JT, Erkinjuntti T, Reisberg B, Roman G, Sawada T, Pantoni L, Bowler JV, Ballard C, DeCarli C, Gorelick PB et al (2003) Vascular cognitive impairment. Lancet Neurol 2:89–98 [DOI] [PubMed] [Google Scholar]
- O’Sullivan M, Morris RG, Huckstep B, Jones DK, Williams SC, Markus HS (2004) Diffusion tensor MRI correlates with executive dysfunction in patients with ischaemic leukoaraiosis. J Neurol Neurosurg Psychiatry 75:441–447 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rigsby CS, Pollock DM, Dorrance AM (2007) Spironolactone improves structure and increases tone in the cerebral vasculature of male spontaneously hypertensive stroke-prone rats. Microvasc Res 73:198–205 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rivera S, Khrestchatisky M, Kaczmarek L, Rosenberg GA, Jaworski DM (2010) Metzincin proteases and their inhibitors: foes or friends in nervous system physiology? J Neurosci 30:15337–15357 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Roman GC, Erkinjuntti T, Wallin A, Pantoni L, Chui HC (2002) Subcortical ischaemic vascular dementia. Lancet Neurol 1:426–436 [DOI] [PubMed] [Google Scholar]
- Roman GC, Salloway S, Black SE, Royall DR, Decarli C, Weiner MW, Moline M, Kumar D, Schindler R, Posner H (2010) Randomized, placebo-controlled, clinical trial of donepezil in vascular dementia: differential effects by hippocampal size. Stroke 41:1213–1221 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rosenberg GA (2009) Matrix metalloproteinases and their multiple roles in neurodegenerative diseases. Lancet Neurol 8:205–216 [DOI] [PubMed] [Google Scholar]
- Rosenberg GA, Kornfeld M, Stovring J, Bicknell JM (1979) Subcortical arteriosclerotic encephalopathy (Binswanger): computerized tomography. Neurology 29:1102–1106 [DOI] [PubMed] [Google Scholar]
- Rosenberg GA, Prestopnik J, Adair JC, Huisa BN, Knoefel J, Caprihan A, Gasparovic C, Thompson J, Erhardt EB, Schrader R (2015) Validation of biomarkers in subcortical ischaemic vascular disease of the Binswanger type: approach to targeted treatment trials. J Neurol Neurosurg Psychiatry 12:1324–1330 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sappey Marinier D, Calabrese G, Hetherington HP, Fisher SN, Deicken R, Van Dyke C, Fein G, Weiner MW (1992) Proton magnetic resonance spectroscopy of human brain: applications to normal white matter, chronic infarction, and MRI white matter signal hyperintensities. Magn Reson Med 26:313–327 [DOI] [PubMed] [Google Scholar]
- Schneider JA, Arvanitakis Z, Bang W, Bennett DA (2007) Mixed brain pathologies account for most dementia cases in community-dwelling older persons. Neurology 69:2197–2204 [DOI] [PubMed] [Google Scholar]
- Smith EE, Schneider JA, Wardlaw JM, Greenberg SM (2012) Cerebral microinfarcts: the invisible lesions. Lancet Neurol 11:272–282 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sonnen JA, Santa Cruz K, Hemmy LS, Woltjer R, Leverenz JB, Montine KS, Jack CR, Kaye J, Lim K, Larson EB et al (2011) Ecology of the aging human brain. Arch Neurol 68:1049–1056 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sood R, Yang Y, Taheri S, Candelario-Jalil E, Estrada EY, Walker EJ, Thompson J, Rosenberg GA (2009) Increased apparent diffusion coefficients on MRI linked with matrix metalloproteinases and edema in white matter after bilateral carotid artery occlusion in rats. J Cereb Blood Flow Metab 29:308–316 [DOI] [PubMed] [Google Scholar]
- Strongin AY, Collier I, Bannikov G, Marmer BL, Grant GA, Goldberg GI (1995) Mechanism of cell surface activation of 72-kDa type IV collagenase. Isolation of the activated form of the membrane metalloprotease. J Biol Chem 270:5331–5338 [DOI] [PubMed] [Google Scholar]
- Taheri S, Gasparovic C, Huisa BN, Adair JC, Edmonds E, Prestopnik J, Grossetete M, Shah NJ, Wills J, Qualls C et al (2011) Blood-brain barrier permeability abnormalities in vascular cognitive impairment. Stroke 42:2158–2163 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Toledo JB, Arnold SE, Raible K, Brettschneider J, Xie SX, Grossman M, Monsell SE, Kukull WA, Trojanowski JQ (2013) Contribution of cerebrovascular disease in autopsy confirmed neurodegenerative disease cases in the National Alzheimer’s Coordinating Centre. Brain 136:2697–2706 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Topakian R, Barrick TR, Howe FA, Markus HS (2010) Blood-brain barrier permeability is increased in normal-appearing white matter in patients with lacunar stroke and leucoaraiosis. J Neurol Neurosurg Psychiatry 81:192–197 [DOI] [PubMed] [Google Scholar]
- Wallin A, Blennow K, Fredman P, Gottfries CG, Karlsson I, Svennerholm L (1990) Blood brain barrier function in vascular dementia. Acta Neurol Scand 81:318–322 [DOI] [PubMed] [Google Scholar]
- Wardlaw JM, Doubal FN, Valdes-Hernandez M, Wang X, Chappell FM, Shuler K, Armitage PA, Carpenter TC, Dennis MS (2013) Blood-brain barrier permeability and long-term clinical and imaging outcomes in cerebral small vessel disease. Stroke 44:525–527 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yang Y, Estrada EY, Thompson JF, Liu W, Rosenberg GA (2007) Matrix metalloproteinase-mediated disruption of tight junction proteins in cerebral vessels is reversed by synthetic matrix metalloproteinase inhibitor in focal ischemia in rat. J Cereb Blood Flow Metab 27:697–709 [DOI] [PubMed] [Google Scholar]
- Yang Y, Thompson JF, Taheri S, Salayandia VM, McAvoy TA, Hill JW, Yang Y, Estrada EY, Rosenberg GA (2013) Early inhibition of MMP activity in ischemic rat brain promotes expression of tight junction proteins and angiogenesis during recovery. J Cereb Blood Flow Metab 33:1104–1114 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yonas H, Gur D, Good BC, Latchaw RE, Wolfson SK Jr, Good WF, Maitz GS, Colsher JG, Barnes JE, Colliander KG et al (1985) Stable xenon CT blood flow mapping for evaluation of patients with extracranial-intracranial bypass surgery. J Neurosurg 62:324–333 [DOI] [PubMed] [Google Scholar]
- Yong VW (2005) Metalloproteinases: mediators of pathology and regeneration in the CNS. Nat Rev Neurosci 6:931–944 [DOI] [PubMed] [Google Scholar]