Abstract
Background. Inflammation plays a key role in cancer. In the current study, we proposed a novel inflammation-based stage, named I stage, for patients with resectable esophageal squamous cell carcinoma (ESCC). Methods. Three hundred and twenty-three patients with resectable ESCC were enrolled in the current study. The I stage was calculated as follows: patients with high levels of C-reactive protein (CRP) (>10 mg/L), neutrophil-to-lymphocyte ratio (NLR) (>3.5), and platelet-count-to-lymphocyte ratio (PLR) (>150) were defined as I3. Patients with two, one, or no abnormal value were defined as I2, I1, or I0, respectively. The prognostic factors were evaluated by univariate and multivariate analyses. Results. There were 112 patients for I0, 97 patients for I1, 66 patients for I2, and 48 patients for I3, respectively. The 5-year cancer-specific survival (CSS) in patients with I0, I1, I2, and I3 was 50.0%, 30.9%, 18.2%, and 8.3%, respectively (I0 versus I1, P = 0.002; I1 versus I2, P = 0.012; I2 versus I3, P = 0.020). Multivariate analyses revealed that I stage was an independent prognostic factor in patients with resectable ESCC (P < 0.001). Conclusion. The inflammation-based stage (I stage) is a novel and useful predictive factor for CSS in patients with resectable ESCC.
1. Introduction
The cancer incidence and mortality have been increasing worldwide. Esophageal cancer (EC) is one of the most common cancers and remains the 4th leading cause of cancer death [1]. There are two major histologic types of EC: esophageal squamous cell carcinoma (ESCC) and esophageal adenocarcinoma (EAC). ESCC is the most common pathological type in China [2, 3]. However, the prognosis for patients with ESCC is still poor [3]. Therefore, assessing the prognostic factors in ESCC patients will become more and more important.
Recent reports revealed that inflammation plays an important role in cancer [4, 5]. Therefore, a series of inflammation-based biomarkers, such as C-reactive protein (CRP), neutrophil-to-lymphocyte ratio (NLR), and platelet-count-to-lymphocyte ratio (PLR), have been analysed in various cancers [6–11]. However, the prognostic values of these biomarkers in patients with ESCC remain uncertain [12–17]. In addition, most of these studies only evaluated one or two biomarkers without considering others. In the current study, therefore, we proposed a novel inflammation-based stage, named I stage (combination of CRP, NLR, and PLR), for predicting the prognosis for patients with resectable ESCC.
2. Patients and Methods
A retrospective analysis was conducted for patients with ESCC in our hospital from January 2005 to December 2008. Patients with ESCC were confirmed by histopathology. All patients underwent surgery with curative esophagectomy and standard lymphadenectomy. Patients who had received preoperative therapy were excluded. Patients with any form of acute infection or chronic inflammatory disease were also excluded. At last, 323 patients were enrolled in our study. In the current study, a cancer-specific survival (CSS) analysis was ascertained. The last follow-up was on 30 June 2013. This study was approved by the Ethical Committees of Zhejiang Cancer Hospital (Hangzhou, China). All patients were staged according to the 7th edition of the American Joint Committee on Cancer (AJCC) Cancer Staging [18].
Routine laboratory results (including CRP, neutrophil, lymphocyte, and platelet count) were extracted in retrospective medical records. The definitions of NLR and PLR were described as follows: NLR is neutrophil-to-lymphocyte ratio and PLR is platelet-count-to-lymphocyte ratio. The cut-off values for CRP, NLR, and PLR were 10 mg/L, 3.5, and 150 according to the previous studies [12, 13, 16, 17]. Therefore, the I stage was calculated as follows: patients with high levels of CRP (>10 mg/L), NLR (>3.5), and PLR (>150) were defined as I3. Patients with two, one, or no abnormal value were defined as I2, I1, or I0, respectively.
2.1. Statistical Analysis
The 5-year CSS was analysed by the Kaplan-Meier method. Univariate and multivariate Cox analyses were performed to analyse the prognostic factors. Pearson correlation analyses were performed to analyse the correlation. Receiver operating characteristic (ROC) curves were plotted to determine the accuracy of CRP, NLR, and PLR. A P < 0.05 was considered to be statistically significant. Statistical analyses were conducted with SPSS 17.0 (SPSS Inc., Chicago, IL, USA).
3. Results
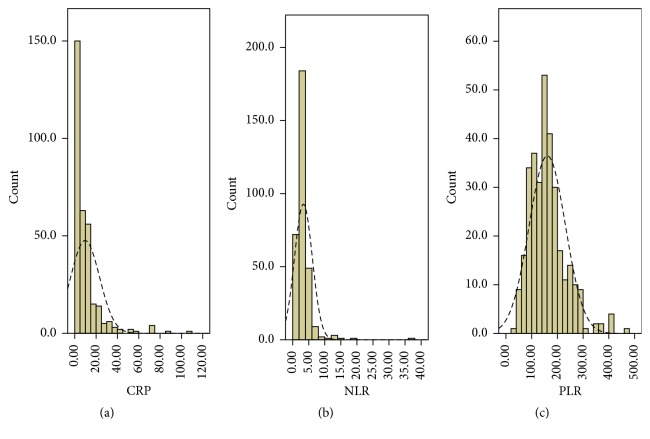
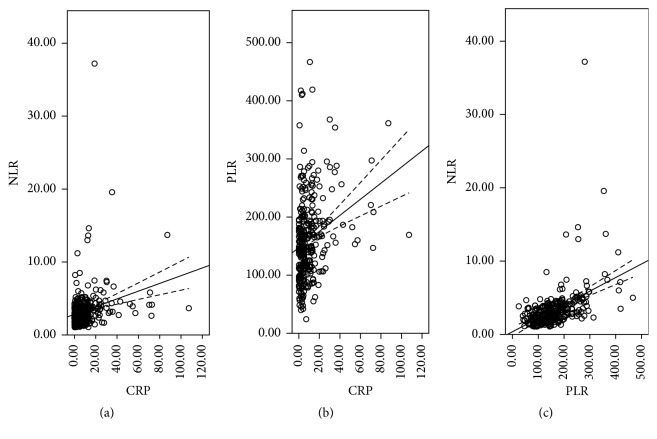
Clinicopathologic characters were shown in Table 1. The mean CRP, NLR, and PLR were 9.7 ± 13.5 (mg/L), 3.3 ± 2.8, and 160.9 ± 70.6, respectively. The histograms of CRP, NLR, and PLR were shown in Figure 1. There were significant positive correlations in CRP and NLR (r = 0.258, P < 0.001), CRP and PLR (r = 0.265, P < 0.001), and NLR and PLR (r = 0.470, P < 0.001) (Figure 2). ROC curves for CSS prediction were shown in Figure 3. The area under the curve (AUC) was 0.713 (95% CI: 0.653–0.772, P < 0.001) for CRP, 0.650 (95% CI: 0.589–0.711, P < 0.001) for NLR, and 0.685 (95% CI: 0.626–0.744, P < 0.001) for PLR.
Table 1.
Clinicopathological characteristics for patients with ESCC.
| Cases (n, %) | |
|---|---|
| Age (years, mean ± SD) | 59.1 ± 7.9 |
| Gender | |
| Female | 42 (13.0) |
| Male | 281 (87.0) |
| Tumor length (cm, mean ± SD) | 4.3 ± 1.9 |
| Tumor location | |
| Upper | 17 (5.3) |
| Middle | 151 (46.7) |
| Lower | 155 (48.0) |
| Differentiation | |
| Good | 44 (13.6) |
| Moderate | 216 (66.9) |
| Poor | 63 (19.5) |
| T grade | |
| T1 | 55 (17.0) |
| T2 | 55 (17.0) |
| T3 | 179 (55.4) |
| T4 | 34 (10.6) |
| N stage | |
| N0 | 174 (53.9) |
| N1 | 87 (26.9) |
| N2 | 37 (11.5) |
| N3 | 25 (7.7) |
| TNM stage | |
| I | 81 (25.1) |
| II | 104 (32.2) |
| III | 138 (42.7) |
| I stage | |
| I0 | 112 (34.7) |
| I1 | 97 (30.0) |
| I2 | 66 (20.4) |
| I3 | 48 (14.9) |
| CRP (mg/L, mean ± SD) | 9.7 ± 13.5 |
| NLR (mean ± SD) | 3.3 ± 2.8 |
| PLR (mean ± SD) | 160.9 ± 70.6 |
Figure 1.
The histograms of the CRP (a), NLR (b), and PLR (c).
Figure 2.
Pearson correlation analysis. Positive correlations in CRP and NLR (r = 0.258, P < 0.001; (a)), CRP and PLR (r = 0.265, P < 0.001; (b)), and NLR and PLR (r = 0.470, P < 0.001; (c)).
Figure 3.
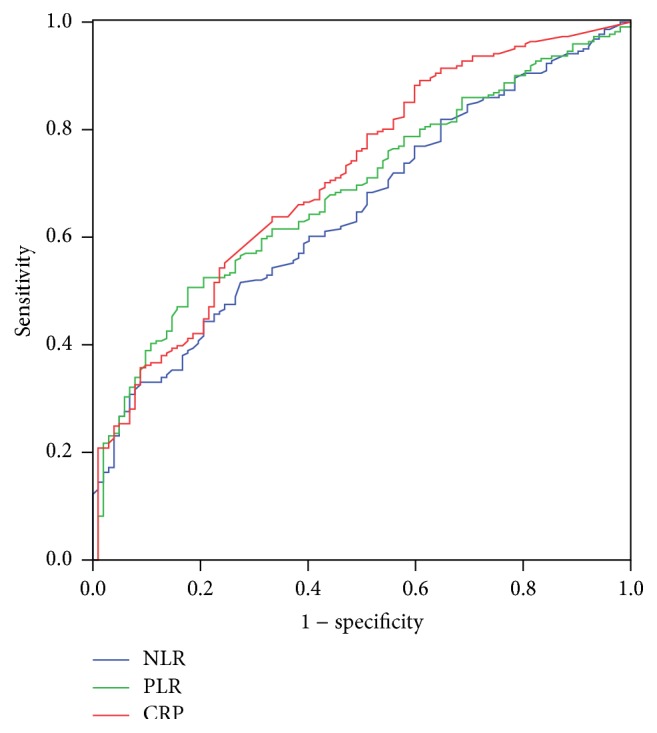
ROC curves for CSS prediction. The area under the curve (AUC) was 0.713 (95% CI: 0.653–0.772, P < 0.001) for CRP, 0.650 (95% CI: 0.589–0.711, P < 0.001) for NLR, and 0.685 (95% CI: 0.626–0.744, P < 0.001) for PLR.
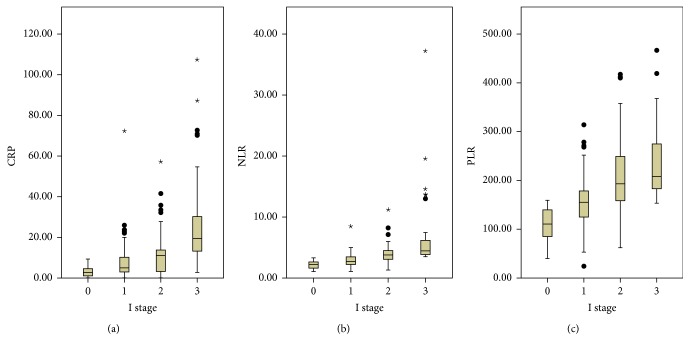
Of the 323 patients, 112 (34.7%) were allocated an I stage 0, 97 (30.0%) were allocated an I stage 1, 66 (20.4%) were allocated an I stage 2, and 48 (14.9%) were allocated an I stage 3, respectively. The relationships between the I stage and clinicopathological characteristics were shown in Table 2. Our study demonstrated that I stage was associated with tumor length (P < 0.001), perineural invasion (P = 0.043), T stage (P < 0.001), N stage (P < 0.001), and TNM stage (P < 0.001). In addition, our study revealed that CRP, NLR, and PLR were significantly higher in patients with high I stage (P < 0.001, Figure 4).
Table 2.
The relationship between I stage and clinicopathological characteristics.
| I stage 0 | I stage 1 | I stage 2 | I stage 3 | P value | |
|---|---|---|---|---|---|
| (n = 112) | (n = 97) | (n = 66) | (n = 48) | ||
| Age (years) | 0.817 | ||||
| ≤60 | 66 | 58 | 37 | 25 | |
| >60 | 46 | 39 | 29 | 23 | |
| Gender | 0.375 | ||||
| Female | 18 | 14 | 5 | 5 | |
| Male | 94 | 83 | 61 | 43 | |
| Tumor length (cm) | <0.001 | ||||
| ≤3 | 45 | 31 | 9 | 4 | |
| >3 | 67 | 66 | 57 | 44 | |
| Tumor location | 0.488 | ||||
| Upper | 8 | 4 | 1 | 4 | |
| Middle | 51 | 49 | 28 | 23 | |
| Lower | 53 | 44 | 37 | 21 | |
| Vessel involvement | 0.385 | ||||
| Negative | 99 | 79 | 54 | 38 | |
| Positive | 13 | 18 | 12 | 10 | |
| Perineural invasion | 0.043 | ||||
| Negative | 98 | 70 | 52 | 40 | |
| Positive | 14 | 27 | 14 | 8 | |
| Differentiation | 0.310 | ||||
| Good | 17 | 10 | 12 | 5 | |
| Moderate | 80 | 65 | 41 | 30 | |
| Poor | 15 | 22 | 13 | 13 | |
| T stage | <0.001 | ||||
| T1 | 33 | 18 | 3 | 1 | |
| T2 | 23 | 14 | 11 | 7 | |
| T3 | 50 | 58 | 42 | 29 | |
| T4 | 6 | 7 | 10 | 11 | |
| N stage | <0.001 | ||||
| N0 | 71 | 55 | 32 | 16 | |
| N1 | 30 | 30 | 12 | 15 | |
| N2 | 6 | 9 | 12 | 10 | |
| N3 | 5 | 3 | 10 | 7 | |
| TNM stage | <0.001 | ||||
| I | 46 | 21 | 9 | 5 | |
| II | 31 | 41 | 22 | 10 | |
| III | 35 | 35 | 35 | 33 |
Figure 4.
The CRP (a), NLR (b), and PLR (c) were significantly higher in patients with high I stage (P < 0.001). The “∗” and “•” were created by SPSS statistical software.
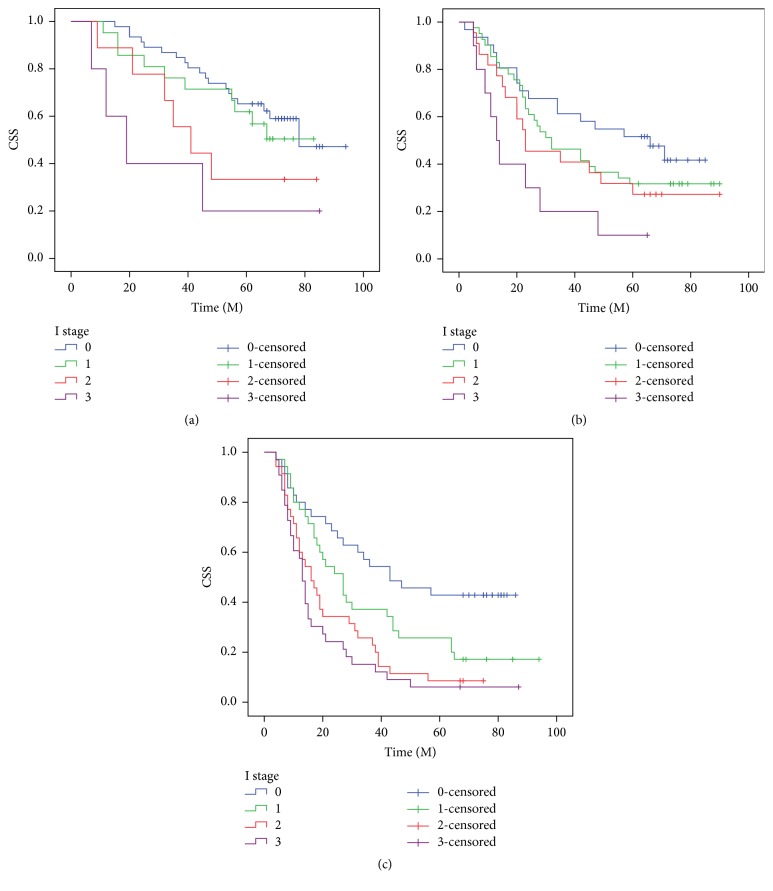
The 5-year CSS in patients with I0, I1, I2, and I3 was 50.0%, 30.9%, 18.2%, and 8.3%, respectively (P < 0.001, Figure 5) (I0 versus I1, P = 0.002; I1 versus I2, P = 0.012; I2 versus I3, P = 0.020). In addition, our study revealed that patients with CRP (>10.0 mg/L), NLR (>3.5), or PLR (>150) were significantly associated with decreased CSS, respectively (P < 0.001). Then, we further stratified patients into different groups based on TNM stage. Our results demonstrated that I stage was also significantly correlated with CSS based on TNM stage (Figure 6).
Figure 5.
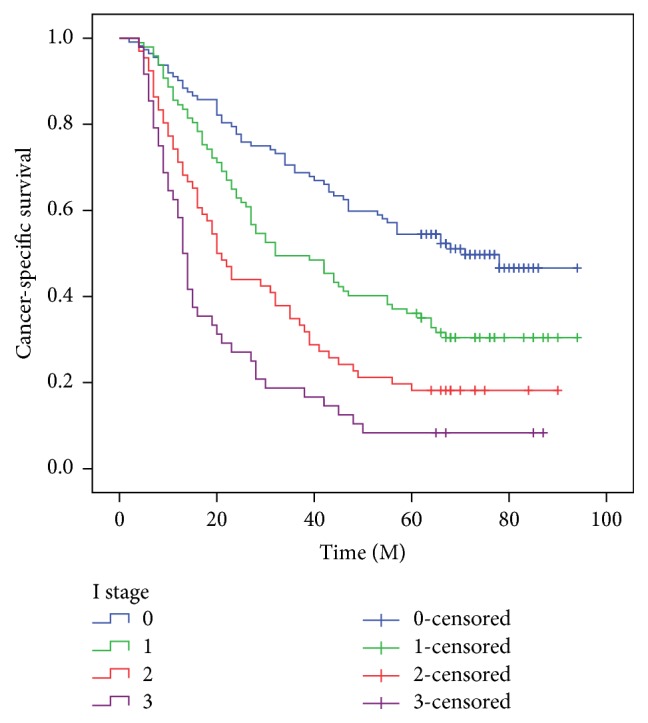
The 5-year CSS in patients with I0, I1, I2, and I3 was 50.0%, 30.9%, 18.2%, and 8.3%, respectively (P < 0.001) (I0 versus I1, P = 0.002; I1 versus I2, P = 0.012; I2 versus I3, P = 0.020).
Figure 6.
The predictive values of I stage were significant in patients based on TNM stage. TNM I stage (P = 0.035, (a)), TNM II stage (P = 0.028, (b)), and TNM III stage (P < 0.001, (c)).
Among the above variables, univariate analyses revealed that tumor length (P = 0.004), vessel involvement (P = 0.008), perineural invasion (P = 0.006), TNM stage (P < 0.001), and I stage (P < 0.001) were predictive of CSS (Table 3). In multivariate analyses, we demonstrated that I stage was an independent prognostic factor in patients with resectable ESCC (P < 0.001) (Table 4).
Table 3.
Univariate analyses for patients with ESCC.
| 5-year CSS (%) | P value | HR (95% CI) | P value | |
|---|---|---|---|---|
| Age (years) | 0.978 | 0.978 | ||
| ≤60 | 30.1 | 1.000 | ||
| >60 | 33.6 | 0.996 (0.762–1.302) | ||
| Gender | 0.322 | 0.327 | ||
| Female | 38.1 | 1.000 | ||
| Male | 30.6 | 1.227 (0.815–1.848) | ||
| Tumor length (cm) | 0.003 | 0.004 | ||
| ≤3 | 41.6 | 1.000 | ||
| >3 | 27.8 | 1.580 (1.157–2.157) | ||
| Tumor location | 0.556 | 0.564 | ||
| Upper | 41.2 | 1.000 | ||
| Middle | 33.1 | 0.735 (0.385–1.404) | 0.351 | |
| Lower | 29.0 | 0.908 (0.693–1.190) | 0.483 | |
| Differentiation | 0.198 | 0.207 | ||
| Good | 38.6 | 1.000 | ||
| Moderate | 31.0 | 1.185 (0.786–1.786) | 0.417 | |
| Poor | 28.6 | 1.504 (0.933–2.424) | 0.098 | |
| Vessel involvement | 0.007 | 0.008 | ||
| Negative | 34.1 | 1.000 | ||
| Positive | 18.9 | 1.577 (1.129–2.202) | ||
| Perineural invasion | 0.005 | 0.006 | ||
| Negative | 35.0 | 1.000 | ||
| Positive | 17.5 | 1.551 (1.135–2.119) | ||
| TNM stage | <0.001 | <0.001 | ||
| I | 51.9 | 1.000 | ||
| II | 32.7 | 1.878 (1.269–2.780) | 0.002 | |
| III | 18.8 | 2.943 (2.039–4.248) | <0.001 | |
| I stage | <0.001 | <0.001 | ||
| I0 | 50.0 | 1.000 | ||
| I1 | 30.9 | 1.696 (1.189–2.420) | 0.004 | |
| I2 | 18.2 | 2.676 (1.837–3.900) | <0.001 | |
| I3 | 8.3 | 4.372 (2.924–6.536) | <0.001 | |
| Adjuvant therapy | 0.398 | 0.402 | ||
| No | 32.0 | 1.000 | ||
| Yes | 30.6 | 1.130 (0.849–1.504) | ||
| CRP (mg/L) | <0.001 | <0.001 | ||
| ≤10.0 | 39.2 | 1.000 | ||
| >10.0 | 17.1 | 2.217 (1.692–2.906) | ||
| NLR | <0.001 | <0.001 | ||
| ≤3.5 | 39.0 | 1.000 | ||
| >3.5 | 17.7 | 1.925 (1.471–2.519) | ||
| PLR | <0.001 | <0.001 | ||
| ≤150 | 43.9 | 1.000 | ||
| >150 | 17.3 | 2.260 (1.729–2.955) |
Table 4.
Multivariate analyses for patients with ESCC.
| HR (95% CI) | P value | |
|---|---|---|
| Tumor length (cm) | 0.603 | |
| ≤3 | 1.000 | |
| >3 | 1.075 (0.818–1.412) | |
| Vessel involvement | 0.742 | |
| Negative | 1.000 | |
| Positive | 1.060 (0.747–1.505) | |
| Perineural invasion | 0.077 | |
| Negative | 1.000 | |
| Positive | 1.341 (0.968–1.857) | |
| TNM stage | <0.001 | |
| I | 1.000 | |
| II | 1.586 (1.048–2.400) | 0.029 |
| III | 2.220 (1.456–3.384) | <0.001 |
| I stage | <0.001 | |
| I0 | 1.000 | |
| I1 | 1.543 (1.076–2.214) | 0.018 |
| I2 | 2.356 (1.602–3.466) | <0.001 |
| I3 | 3.594 (2.363–5.467) | <0.001 |
| CRP (mg/L) | 0.493 | |
| ≤10.0 | 1.000 | |
| >10.0 | 1.151 (0.770–1.719) | |
| NLR | 0.786 | |
| ≤3.5 | 1.000 | |
| >3.5 | 1.050 (0.740–1.488) | |
| PLR | 0.065 | |
| ≤150 | 1.000 | |
| >150 | 1.440 (0.978–2.121) |
4. Discussion
In the current study, we initially proposed a novel inflammation-based prognostic system, named I stage (combination of CRP, NLR, and PLR), in patients with resectable ESCC. Our study revealed that I stage was associated with tumor length, perineural invasion, and TNM stage. In multivariate analyses, we revealed that I stage is a useful predictor of postoperative CSS in patients with resectable ESCC (P < 0.001).
Several hematological biomarkers have shown prognostic values in cancers. In particular, the CRP has been well validated. CRP is a representative acute-phase reactant for inflammation [19]. Recently, several previous studies have shown that CRP is associated with prognosis in several cancers, including ECs [6, 8–12]. In our study, patients with CRP ≤ 10.0 mg/L had a significantly better 5-year CSS than patients with CRP > 10.0 mg/L (39.2% versus 17.1%, P < 0.001). However, CRP was not an independent prognostic factor in multivariate analyses (P = 0.493).
The prognostic values of NLR and PLR in patients with EC remain uncertain. Several reports demonstrated that NLR is an independent prognostic factor in patients with EC [14, 15]. However, Rashid et al. [13] and Dutta et al. [16] revealed that NLR does not correlate with prognosis for patients with EC. Moreover, there have been few studies regarding PLR in EC patients. Dutta et al. [16] demonstrated that PLR does not correlate with prognosis in patients with EC. A retrospective study by Liu et al. [20] on 326 ESCC patients revealed PLR to be a potential prognostic factor. In our study, NLR and PLR were correlated with survival; however, NLR and PLN were not independent prognostic factors in multivariate analyses.
At present, the prognosis of cancer is commonly based on the TNM staging system [21, 22]. Inflammation plays an important role in cancer. Therefore, in our study, we proposed a novel inflammation-based prognostic system (I stage) in resectable ESCC patients. A significant association was found between the I stage and clinical characteristics. In multivariate analyses, we revealed that I stage is a useful predictor of postoperative CCS in patients with resectable ESCC (P < 0.001). It may well be that the influence of I stage on the subgroup with TNM stage is important for the understanding of its role in patients with ESCC. Our results demonstrated that I stage was also significantly correlated with CSS based on TNM stage.
Limitations should be acknowledged. Firstly, our study was a retrospective study. Secondly, we excluded patients with neoadjuvant treatment, which may have influenced the results. Neoadjuvant treatment will inevitably have an impact on the systemic inflammation. Thus, evaluation of I stage in neoadjuvant therapy does not reflect the baseline impact of systemic inflammation for ESCC patients. Therefore, larger prospective studies will need to be performed to confirm these preliminary results.
In summary, there was a significant association between the I stage (combination of CRP, NLR, and PLR) and clinical characteristics. Based on the results of the current study, we believe that I stage is a novel and useful predictive factor for CSS in patients with resectable ESCC.
Competing Interests
The authors have no competing interests to disclose.
References
- 1.Siegel R. L., Miller K. D., Jemal A. Cancer statistics, 2015. CA: A Cancer Journal for Clinicians. 2015;65(1):5–29. doi: 10.3322/caac.21254. [DOI] [PubMed] [Google Scholar]
- 2.Lin Y., Totsuka Y., He Y., et al. Epidemiology of esophageal cancer in Japan and China. Journal of Epidemiology. 2013;23(4):233–242. doi: 10.2188/jea.je20120162. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Keditsu K. K., Jiwnani S., Karimundackal G., Pramesh C. S. Multimodality management of esophageal cancer. Indian Journal of Surgical Oncology. 2013;4(2):96–104. doi: 10.1007/s13193-013-0216-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Balkwill F., Mantovani A. Inflammation and cancer: back to Virchow? The Lancet. 2001;357(9255):539–545. doi: 10.1016/s0140-6736(00)04046-0. [DOI] [PubMed] [Google Scholar]
- 5.Mantovani A., Allavena P., Sica A., Balkwill F. Cancer-related inflammation. Nature. 2008;454(7203):436–444. doi: 10.1038/nature07205. [DOI] [PubMed] [Google Scholar]
- 6.Nakamura T., Matsumine A., Asanuma K., Matsubara T., Sudo A. The role of C-reactive protein in predicting post-metastatic survival of patients with metastatic bone and soft tissue sarcoma. Tumor Biology. 2015;36(10):7515–7520. doi: 10.1007/s13277-015-3464-5. [DOI] [PubMed] [Google Scholar]
- 7.Jiang N., Deng J.-Y., Liu Y., Ke B., Liu H.-G., Liang H. The role of preoperative neutrophil-lymphocyte and platelet-lymphocyte ratio in patients after radical resection for gastric cancer. Biomarkers. 2014;19(6):444–451. doi: 10.3109/1354750X.2014.926567. [DOI] [PubMed] [Google Scholar]
- 8.Nozoe T., Iguchi T., Adachi E., Matsukuma A., Ezaki T. Preoperative elevation of serum C-reactive protein as an independent prognostic indicator for gastric cancer. Surgery Today. 2011;41(4):510–513. doi: 10.1007/s00595-009-4297-x. [DOI] [PubMed] [Google Scholar]
- 9.Nakamura T., Matsumine A., Matsubara T., Asanuma K., Uchida A., Sudo A. The combined use of the neutrophil-lymphocyte ratio and C-reactive protein level as prognostic predictors in adult patients with soft tissue sarcoma. Journal of Surgical Oncology. 2013;108(7):481–485. doi: 10.1002/jso.23424. [DOI] [PubMed] [Google Scholar]
- 10.Nakamura T., Grimer R. J., Gaston C. L., Watanuki M., Sudo A., Jeys L. The prognostic value of the serum level of C-reactive protein for the survival of patients with a primary sarcoma of bone. Bone & Joint Journal B. 2013;95(3):411–418. doi: 10.1302/0301-620x.95b3.30344. [DOI] [PubMed] [Google Scholar]
- 11.Nakamura T., Grimer R., Gaston C., et al. The value of C-reactive protein and comorbidity in predicting survival of patients with high grade soft tissue sarcoma. European Journal of Cancer. 2013;49(2):377–385. doi: 10.1016/j.ejca.2012.09.004. [DOI] [PubMed] [Google Scholar]
- 12.Shimada H., Nabeya Y., Okazumi S.-I., et al. Elevation of pre-operative serum C-reactive protein level is related to poor prognosis in esophageal squamous cell carcinoma. Journal of Surgical Oncology. 2003;83(4):248–252. doi: 10.1002/jso.10275. [DOI] [PubMed] [Google Scholar]
- 13.Rashid F., Waraich N., Bhatti I., et al. A pre-operative elevated neutrophil: lymphocyte ratio does not predict survival from oesophageal cancer resection. World Journal of Surgical Oncology. 2010;8, article 1 doi: 10.1186/1477-7819-8-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Sato H., Tsubosa Y., Kawano T. Correlation between the pretherapeutic neutrophil to lymphocyte ratio and the pathologic response to neoadjuvant chemotherapy in patients with advanced esophageal cancer. World Journal of Surgery. 2012;36(3):617–622. doi: 10.1007/s00268-011-1411-1. [DOI] [PubMed] [Google Scholar]
- 15.Sharaiha R. Z., Halazun K. J., Mirza F., et al. Elevated preoperative neutrophil: lymphocyte ratio as a predictor of postoperative disease recurrence in esophageal cancer. Annals of Surgical Oncology. 2011;18(12):3362–3369. doi: 10.1245/s10434-011-1754-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Dutta S., Crumley A. B. C., Fullarton G. M., Horgan P. G., McMillan D. C. Comparison of the prognostic value of tumour- and patient-related factors in patients undergoing potentially curative resection of oesophageal cancer. World Journal of Surgery. 2011;35(8):1861–1866. doi: 10.1007/s00268-011-1130-7. [DOI] [PubMed] [Google Scholar]
- 17.Feng J.-F., Huang Y., Chen Q.-X. Preoperative platelet lymphocyte ratio (PLR) is superior to neutrophil lymphocyte ratio (NLR) as a predictive factor in patients with esophageal squamous cell carcinoma. World Journal of Surgical Oncology. 2014;12, article 58 doi: 10.1186/1477-7819-12-58. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Rice T. W., Rusch V. W., Ishwaran H., Blackstone E. H. Cancer of the esophagus and esophagogastric junction: data-driven staging for the seventh edition of the American Joint Committee on Cancer/International Union Against Cancer Cancer Staging Manuals. Cancer. 2010;116(16):3763–3773. doi: 10.1002/cncr.25146. [DOI] [PubMed] [Google Scholar]
- 19.Diehl E. E., Haines G. K., III, Radosevich J. A., Potempa L. A. Immunohistochemical localization of modified C-reactive protein antigen in normal vascular tissue. The American Journal of the Medical Sciences. 2000;319(2):79–83. doi: 10.1016/s0002-9629(15)40692-5. [DOI] [PubMed] [Google Scholar]
- 20.Liu J. S., Huang Y., Yang X., Feng J. F. A nomogram to predict prognostic values of various inflammatory biomarkers in patients with esophagealsquamous cell carcinoma. American Journal of Cancer Research. 2015;5:2180–2189. [PMC free article] [PubMed] [Google Scholar]
- 21.Wijnhoven B. P. L., Tran K. T. C., Esterman A., Watson D. I., Tilanus H. W. An evaluation of prognostic factors and tumor staging of resected carcinoma of the esophagus. Annals of Surgery. 2007;245(5):717–725. doi: 10.1097/01.sla.0000251703.35919.02. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Peyre C. G., Hagen J. A., DeMeester S. R., et al. The number of lymph nodes removed predicts survival in esophageal cancer: an international study on the impact of extent of surgical resection. Annals of Surgery. 2008;248(4):549–556. doi: 10.1097/sla.0b013e318188c474. [DOI] [PubMed] [Google Scholar]