Abstract
The position of the mitotic spindle determines the cleavage plane in animal cells, but what controls spindle positioning? Kern et al. (2016. J. Cell Biol. http://dx.doi.org/10.1083/jcb.201510117) demonstrate that the microtubule plus end–associated SKAP/Astrin complex participates in this process, possibly by affecting dynein-dependent pulling forces exerted on the tips of astral microtubules.
The mitotic spindle is a complex molecular machine that rapidly remodels a mother cell to create two daughter cells during cell division. The best known function of the spindle microtubules and associated proteins is to provide the tracks and trains that segregate the chromosomes to opposite poles in mitosis. As a second major task, the spindle determines the plane of cell cleavage through its interactions with the cell cortex (Kiyomitsu, 2015). The importance of these processes is evident: proper chromosome segregation is critical for the genetic stability and survival of all cells, whereas the regulation of the cell-cleavage plane is essential for normal development and maintenance of tissue homeostasis.
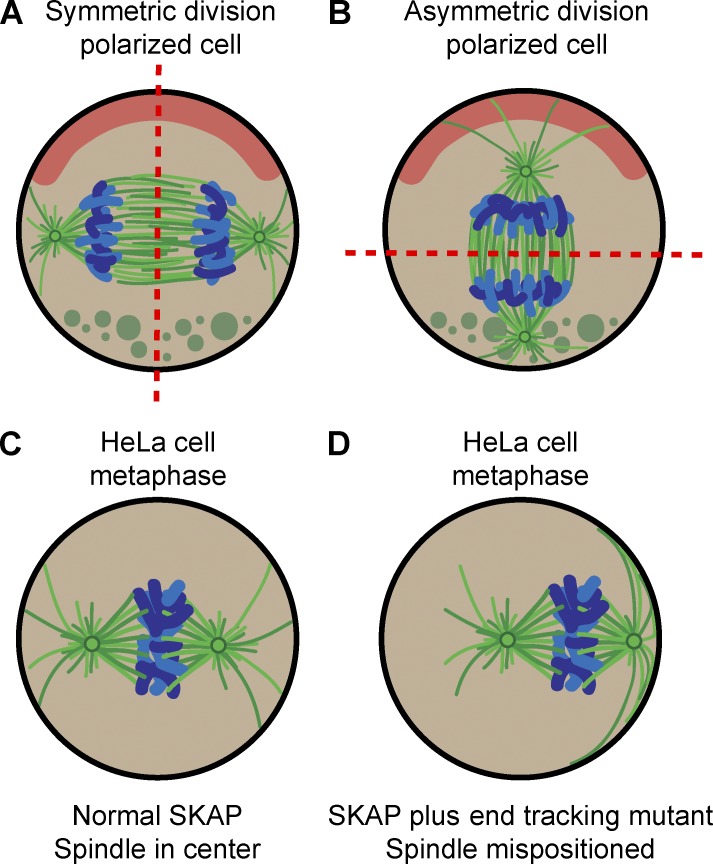
By dictating the plane of cell cleavage, the position of the spindle controls the relative size and location of daughter cells and determines whether cell division is symmetric or asymmetric. Symmetric divisions create identical daughter cells, a process needed to exponentially expand cell populations. Asymmetric cell divisions, in contrast, generate daughter cells that differ in inherited components and developmental fate (Fig. 1). This provides a mechanism by which cell diversity can arise during development. Thus, correct spindle positioning is needed for maintaining the integrity of epithelia, creating cell diversity, and coordinating proliferation and differentiation of stem cell–like progenitors.
Figure 1.
The position of the spindle determines the cell cleavage plane. Schematic representations of symmetric (A) or asymmetric (B) division of polarized cells in late anaphase. (C and D) HeLa cells with normal SKAP and metaphase spindle in the center (C) or plus end tracking-deficient SKAP mutant with a laterally displaced spindle (D). Dashed lines, cleavage plane position; green lines, spindle microtubules; blue, chromosomes; red, polarized cortex; green circles, cytoplasmic determinants.
Given this central role, it is important to understand what molecular mechanisms determine the position of the spindle in mitosis. Does the spindle align with the cell axis, migrate off-center, or even rotate to alter the plane of cell cleavage by 90°? These examples of spindle behavior are reproducibly seen in genetic model systems. In particular, studies of the one-cell Caenorhabditis elegans embryo, as well as neuroblasts and sensory organ precursor cells in Drosophila melanogaster, have revealed much insight in spindle positioning (Knoblich, 2010). The components identified in these systems form part of a spindle-positioning complex that appears conserved in all animals. This complex consists of the α subunit of a heterotrimeric G protein (Gα), which is lipid anchored to the membrane. Gα binds a TPR-GoLoco domain protein (e.g., LGN), which in turn associates with a NuMA-related coiled-coil protein. This trimeric complex recruits the dynein motor and captures astral microtubules at the cortex. The interplay between cortically attached dynein and depolymerizing astral microtubules generates cortical pulling forces that promote aster movement and spindle positioning.
In mammals, spindle positioning has been best studied in neuroepithelial precursors during brain formation and in basal progenitors during the development of the stratified epidermis (Mora-Bermúdez et al., 2014; Williams et al., 2014). Remarkably, even tumor-derived cells in 2D cell cultures show extensive control over spindle positioning. While normally positioning the spindle in the cell center, HeLa cells grown on printed fibronectin micropatterns orient their spindle in response to adhesive contacts established in interphase and maintained through retraction fibers in mitosis (Théry et al., 2007). Membrane recruitment of dynein by the human Gαi–LGN–NuMA complex was shown to also be critical for orienting the spindle in HeLa cells (Kotak et al., 2012).
Although the molecular players of the cortical dynein complexes exerting force on astral microtubules may be identified, how these complexes interact with microtubule tips is still unclear. The plus ends of astral microtubules are highly dynamic: they grow from the spindle pole toward the cell cortex, where they persist for a few seconds and then depolymerize (Samora et al., 2011). During these short encounters, dynein with its cortical partners must form a force-generating complex that pulls on the microtubule plus end. In vitro experiments have shown that the dynein motor alone can capture a microtubule plus end, trigger its depolymerization, and, by holding onto a shrinking microtubule, generate force in an end-on configuration (Laan et al., 2012). Dynein can also generate force by microtubule sliding, i.e., walking on microtubules laterally interacting with the cortex (Samora et al., 2011).
In cells, the interactions of dynein with microtubule plus ends are affected by the presence of different microtubule-associated proteins (MAPs), which can bind along the entire microtubule or accumulate at microtubule ends. Among these, microtubule plus end–tracking proteins (+TIPs) are particularly relevant (Akhmanova and Steinmetz, 2015). This group of very diverse factors specifically recognizes growing microtubule ends and can either stimulate or inhibit dynein interaction with microtubule tips. The core of the +TIP complexes is formed by end-binding (EB) proteins, which autonomously recognize growing microtubule ends and recruit a plethora of other +TIPs (Akhmanova and Steinmetz, 2015). Dynein can be targeted to EB-bound microtubule plus ends by its cofactor dynactin, which associates with EBs either directly or indirectly (Akhmanova and Steinmetz, 2015). These +TIP interactions contribute to dynein loading onto the plus ends for minus end–directed cargo transport; however, it is currently unclear whether they also participate in the formation of cortical dynein complexes and generation of pulling forces.
Proteins that form a large group of EB-dependent +TIPs possess a so-called SxIP motif embedded in an intrinsically unstructured basic polypeptide region (Honnappa et al., 2009). In this issue, Kern et al. study a SxIP-containing +TIP, small kinetochore associated protein (SKAP). Previous work has shown that SKAP, together with its binding partner Astrin, is required for multiple aspects of the formation and function of the mitotic spindle (Dunsch et al., 2011; Wang et al., 2012; Tamura et al., 2015). Here, Kern et al. (2016) demonstrate that the ability of SKAP to track growing microtubule plus ends is important for dynein-dependent symmetric positioning of the metaphase spindle in HeLa cells.
SKAP is expressed as two splice forms, which encode proteins of substantially different sizes. Previous studies of cell division mostly focused on the long isoform. The experiments by Kern et al. (2016), as well as the recently published work by Grey et al. (2016), demonstrated that this isoform is only present during spermatogenesis, whereas dividing cells express the short isoform. In contrast to the long isoform, the short SKAP isoform effectively rescued the mitotic phenotypes induced by SKAP depletion in HeLa cells, and thus could be effectively used for functional analysis of SKAP domains. Kern et al. (2016) identified a positively charged sequence responsible for the interaction of SKAP with microtubules and showed that this sequence is essential for spindle organization and chromosome alignment.
SKAP also contains a single SxIP motif, which can recruit SKAP to the growing microtubule ends by binding to EB proteins both in cells and in vitro (Wang et al., 2012; Tamura et al., 2015). Kern et al. (2016) found that mutation of this SxIP motif prevents the plus end association of SKAP but does not interfere with the majority of its microtubule-associated functions. However, this mutation caused a strong displacement of the spindle to one side of the cell in the horizontal plane (Fig. 1 D). This defect correlated with enhanced lateral interactions of the astral microtubules on the side where the spindle was closest to the cell cortex. Kern et al. (2016) showed that depletion of LGN, one of the components of the cortical trimeric complex that recruits dynein, restored spindle positioning, suggesting that SKAP might somehow regulate dynein-based pulling of astral microtubules.
Metaphase spindle positioning defects are corrected by spindle pole–derived Polo-like kinase 1 signaling, which attenuates the binding between LGN-NuMA and dynein-dynactin in a distance-dependent manner (Kiyomitsu, 2015). When a pole moves too closely to one side of the cell, this signaling mechanism suppresses dynein pulling on that side and corrects spindle mispositioning. Kern et al. (2016) show that this spindle-centering pathway is not affected by the SKAP mutant deficient in plus end tracking and its misregulation cannot explain spindle mispositioning.
The mechanism underlying the involvement of SKAP in spindle positioning likely involves its effect on the composition of +TIP complexes. One of the abundant +TIPs that binds to the SKAP/Astrin complex is CLASP1 (Maffini et al., 2009). Kern et al. (2016) confirmed this interaction and showed that when SKAP cannot bind to microtubule plus ends, the recruitment of CLASPs to microtubule tips is specifically suppressed in mitotic cells. CLASP1 was previously proposed to regulate cortical capture of astral microtubule tips (Samora et al., 2011). In interphase cells, CLASPs associate with the cell cortex by interacting with the phosphatidylinositol-3,4,5-triphosphate–binding protein LL5β (Lansbergen et al., 2006). It should be noted, however, that it is currently unknown whether a functional CLASP-LL5β interaction is retained in mitosis. Furthermore, CLASP1 binds to LL5β and the SKAP/Astrin complex through the same C-terminal domain (Lansbergen et al., 2006; Kern et al., 2016). Therefore, it is questionable that CLASP can bind to both factors simultaneously and in this way provide a link to the cortex when recruited to microtubule tips by SKAP.
Immunoprecipitation-based analysis of the SKAP interactome identified multiple associated proteins, including dynein and NuMA (Kern et al., 2016). Therefore, it is possible that SKAP affects dynein engagement with microtubule tips directly. For example, previous work has shown that MAP4, a “classical” MAP similar to neuronal tau and MAP2, binds to dynactin and participates in metaphase spindle positioning by suppressing excessive dynein-based engagement of the astral microtubule tips with the cell cortex (Samora et al., 2011). It is conceivable that SKAP might act in a similar way.
Being an abundant mitotic +TIP, SKAP in association with Astrin and other proteins might attenuate dynein interactions with the microtubule tips simply by occluding the binding sites for dynein. Mounting evidence indicates that the functions of microtubule plus end–associated proteins are strongly influenced by both synergistic and competitive interactions between different +TIPs, which form extended and flexible networks (Akhmanova and Steinmetz, 2015). Because the EB proteins themselves participate in the regulation of astral microtubule organization and spindle orientation in mammalian cells (Toyoshima and Nishida, 2007), it is likely that the dynamics and movements of astral microtubule plus ends at the cortex ultimately depend on the complex interplay of multiple tip-associated factors.
Defects in metaphase spindle positioning can be compensated by a backup system, which becomes active when cells start to elongate in anaphase (Kiyomitsu, 2015). Metaphase spindle mispositioning can thus be corrected in anaphase and does not necessarily have dramatic consequences for the ensuing cell division. Still, given the elaborate functions of the SKAP/Astrin complex in spindle formation and chromosome separation in cultured human cells such as HeLa, it remains surprising that knockout mice lacking SKAP expression are viable and develop normally, although they do display defects during spermatogenesis and reduced fertility (Grey et al., 2016). Knockout mice that lack the Astrin homologue SPAG5 are also viable and show no major defects (Xue et al., 2002). It is possible that SKAP/Astrin function during cell division can be compensated by the presence of other spindle proteins, which are not available in HeLa cells. As a provocative possibility, these data could reflect a functional difference between humans and mice. It is noteworthy in this respect that the SPAG5/Astrin-encoding gene evolved rapidly in mammals, with three fixed amino acid differences reported between human and Neanderthal genomes (Prüfer et al., 2014). This observation is potentially interesting because spindle positioning is particularly important for the regulation of brain development. Future studies will reveal how the microtubule end–associated complexes responsible for the critical aspects of cell division combine highly conserved properties with functional features that might result in adaptive changes driving evolution.
Acknowledgments
The authors declare no competing financial interests.
References
- Akhmanova A., and Steinmetz M.O.. 2015. Control of microtubule organization and dynamics: two ends in the limelight. Nat. Rev. Mol. Cell Biol. 16:711–726. 10.1038/nrm4084 [DOI] [PubMed] [Google Scholar]
- Dunsch A.K., Linnane E., Barr F.A., and Gruneberg U.. 2011. The astrin–kinastrin/SKAP complex localizes to microtubule plus ends and facilitates chromosome alignment. J. Cell Biol. 192:959–968. 10.1083/jcb.201008023 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Grey C., Espeut J., Ametsitsi R., Kumar R., Luksza M., Brun C., Verlhac M.H., Suja J.A., and de Massy B.. 2016. SKAP, an outer kinetochore protein, is required for mouse germ cell development. Reproduction. 151:239–251. 10.1530/REP-15-0451 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Honnappa S., Gouveia S.M., Weisbrich A., Damberger F.F., Bhavesh N.S., Jawhari H., Grigoriev I., van Rijssel F.J., Buey R.M., Lawera A., et al. 2009. An EB1-binding motif acts as a microtubule tip localization signal. Cell. 138:366–376. 10.1016/j.cell.2009.04.065 [DOI] [PubMed] [Google Scholar]
- Kern D.M., Nicholls P.K., Page D.C., and Cheeseman I.M.. 2016. A mitotic SKAP isoform regulates spindle positioning at astral microtubule plus ends. J. Cell Biol. 10.1083/jcb.201510117 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kiyomitsu T. 2015. Mechanisms of daughter cell-size control during cell division. Trends Cell Biol. 25:286–295. 10.1016/j.tcb.2014.12.003 [DOI] [PubMed] [Google Scholar]
- Knoblich J.A. 2010. Asymmetric cell division: recent developments and their implications for tumour biology. Nat. Rev. Mol. Cell Biol. 11:849–860. 10.1038/nrm3010 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kotak S., Busso C., and Gönczy P.. 2012. Cortical dynein is critical for proper spindle positioning in human cells. J. Cell Biol. 199:97–110. 10.1083/jcb.201203166 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Laan L., Pavin N., Husson J., Romet-Lemonne G., van Duijn M., López M.P., Vale R.D., Jülicher F., Reck-Peterson S.L., and Dogterom M.. 2012. Cortical dynein controls microtubule dynamics to generate pulling forces that position microtubule asters. Cell. 148:502–514. 10.1016/j.cell.2012.01.007 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lansbergen G., Grigoriev I., Mimori-Kiyosue Y., Ohtsuka T., Higa S., Kitajima I., Demmers J., Galjart N., Houtsmuller A.B., Grosveld F., and Akhmanova A.. 2006. CLASPs attach microtubule plus ends to the cell cortex through a complex with LL5β. Dev. Cell. 11:21–32. 10.1016/j.devcel.2006.05.012 [DOI] [PubMed] [Google Scholar]
- Maffini S., Maia A.R., Manning A.L., Maliga Z., Pereira A.L., Junqueira M., Shevchenko A., Hyman A., Yates J.R. III, Galjart N., et al. 2009. Motor-independent targeting of CLASPs to kinetochores by CENP-E promotes microtubule turnover and poleward flux. Curr. Biol. 19:1566–1572. 10.1016/j.cub.2009.07.059 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mora-Bermúdez F., Matsuzaki F., and Huttner W.B.. 2014. Specific polar subpopulations of astral microtubules control spindle orientation and symmetric neural stem cell division. eLife. 3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Prüfer K., Racimo F., Patterson N., Jay F., Sankararaman S., Sawyer S., Heinze A., Renaud G., Sudmant P.H., de Filippo C., et al. 2014. The complete genome sequence of a Neanderthal from the Altai Mountains. Nature. 505:43–49. 10.1038/nature12886 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Samora C.P., Mogessie B., Conway L., Ross J.L., Straube A., and McAinsh A.D.. 2011. MAP4 and CLASP1 operate as a safety mechanism to maintain a stable spindle position in mitosis. Nat. Cell Biol. 13:1040–1050. 10.1038/ncb2297 [DOI] [PubMed] [Google Scholar]
- Tamura N., Simon J.E., Nayak A., Shenoy R., Hiroi N., Boilot V., Funahashi A., and Draviam V.M.. 2015. A proteomic study of mitotic phase-specific interactors of EB1 reveals a role for SXIP-mediated protein interactions in anaphase onset. Biol. Open. 4:155–169. 10.1242/bio.201410413 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Théry M., Jiménez-Dalmaroni A., Racine V., Bornens M., and Jülicher F.. 2007. Experimental and theoretical study of mitotic spindle orientation. Nature. 447:493–496. 10.1038/nature05786 [DOI] [PubMed] [Google Scholar]
- Toyoshima F., and Nishida E.. 2007. Integrin-mediated adhesion orients the spindle parallel to the substratum in an EB1- and myosin X-dependent manner. EMBO J. 26:1487–1498. 10.1038/sj.emboj.7601599 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang X., Zhuang X., Cao D., Chu Y., Yao P., Liu W., Liu L., Adams G., Fang G., Dou Z., et al. 2012. Mitotic regulator SKAP forms a link between kinetochore core complex KMN and dynamic spindle microtubules. J. Biol. Chem. 287:39380–39390. 10.1074/jbc.M112.406652 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Williams S.E., Ratliff L.A., Postiglione M.P., Knoblich J.A., and Fuchs E.. 2014. Par3-mInsc and Gαi3 cooperate to promote oriented epidermal cell divisions through LGN. Nat. Cell Biol. 16:758–769. 10.1038/ncb3001 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xue J., Tarnasky H.A., Rancourt D.E., and van Der Hoorn F.A.. 2002. Targeted disruption of the testicular SPAG5/deepest protein does not affect spermatogenesis or fertility. Mol. Cell. Biol. 22:1993–1997. 10.1128/MCB.22.7.1993-1997.2002 [DOI] [PMC free article] [PubMed] [Google Scholar]