Abstract
Background
The benefits of pioglitazone in patients with type 2 diabetes mellitus (T2DM) after percutaneous coronary intervention (PCI) is unclear.
Objectives
To evaluate the effect of pioglitazone on prevention of in-stent restenosis (ISR) in patients with T2DM after PCI.
Methods
All full-text published relevant studies compared the effect of pioglitazone with control group (placebo or no pioglitazone treatment) on ISR in patients with T2DM after PCI were identified by searching the databases including PubMed, EMBASE, Cochrane Library and ISI Web of Science through October 2015. The endpoints were defined as the rate of ISR, late lumen loss, in-stent neointimal volume, target lesion revascularization (TLR) and major adverse cardiac events (MACE).
Results
Six studies (5 RCTs and 1 retrospective study), comprising 503 patients, were included into this meta-analysis. In the pioglitazone group, as compared with the control group, the risk ratio for ISR was 0.48 (I2 = 14.5%, P = 0.322; 95%CI 0.35 to 0.68, P<0.001), the risk ratio for TLR was 0.58 (I2 = 6.0%, P = 0.363; 95%CI 0.38 to 0.87, P = 0.009). The result showed there was no association between the use of pioglitazone and the events of MACE (I2 = 36.7%, P = 0.209; RR 0.56, 95%CI 0.30 to 1.05, P = 0.071). For the considerable heterogeneity, further analysis was not suitable for the endpoints of late lumen loss (I2 = 81.9%, P<0.001) and neointimal volume (I2 = 75.9%, P = 0.016).
Conclusions
The treatment of pioglitazone was associated with a reduction in ISR and TLR in T2DM patients suffering from PCI, except the incidence of MACE.
Introduction
In-stent restenosis (ISR) is considered as the leading problem after percutaneous coronary intervention (PCI), especially in patients with type 2 diabetes mellitus (T2DM)[1,2]. Thiazolidinediones (TZDs), agonists of the peroxisome proliferation-activated receptor-γ (PPAR-γ), usually used to improve insulin sensitivity and reduce blood glucose levels in diabetic patients, could also inhibit proliferation and migration of vascular smooth muscle cells (VSMCs) and ameliorate inflammation after vascular injury[3–5]. Among the three TZDs, pioglitazone may be the only one which shows beneficial effects on cardiovascular outcomes and appears to be promising in clinical application[6–8]. Previous meta-analysis has suggested that pioglitazone is effective in decreasing ISR and the need for revascularization after bare-metal stents (BMS) implantation in diabetic patients[9]. However, drug-eluting stents (DES) are more widely used in recent years. Diabetes mellitus was also regarded as one of the most powerful clinical predictors of ISR after DES implantation as same as in BMS era[10–12]. A hypothesized independent anti-restenotic effect of pioglitazone in patients with T2DM has not been clearly demonstrated[13]. Thus, we performed this meta-analysis to investigate the effect of pioglitazone on prevention of ISR in patients with T2DM after PCI, with update information both on BMS and DES implantation.
Methods
This meta-analysis was conducted according to the PRISMA statement [14], and the PRISMA checklist is provided as S1 Table.
Search Strategy
We searched the databases including PubMed (http://www.ncbi.nlm.nih.gov/pubmed), EMBASE (http://www.embase.com), Cochrane Library (www.cochranelibrary.com) and ISI Web of Science (http://www.webofknowledge.com) for all full-text published relevant studies compared the effect of pioglitazone with control group (placebo or no pioglitazone treatment) in T2DM patients after PCI through October 2015. Search terms included any possible combination of the keywords of ‘‘restenosis”, ‘‘percutaneous coronary intervention or PCI”, ‘‘pioglitazone” and ‘‘diabetes”.
Study selection
Two independent reviewers (S.-J.Z. and L.C.) were involved in the search for potentially eligible studies and in the inclusion process. In case of disagreement, a decision was made under the supervision of the senior author (W.T.). Studies will be included if they met the following criteria: (1) RCT or high quality retrospective study, (2) pioglitazone was administered and compared with control group (placebo or no pioglitazone treatment) for patients who suffered from diabetes and performed PCI, and (3) the studies included at least one of the following interesting outcomes: ISR (defined as stenosis more than 50% at the site of stent), late loss (change in minimum lumen diameter at the stent site from baseline to follow-up), in-stent neointimal volume (neointimal volume/length of stent) measured by intravenous ultrasound (IVUS), target lesion revascularization (TLR) and major adverse cardiac events (MACE, including target vessel revascularization, acute myocardial infarction, cardiac death, etc.).
Data abstraction and quality assessment
For all included studies, we recorded the baseline information of the study, including the number of subjects, patients’ characteristics, interventions in each group, methodology quality of the study, outcomes with ascertained method and length of follow-up. The data were extracted independently by two authors(S.-J.Z. and L.C.) using a pre-designed data extraction form. As before, disagreements will be resolved by discussion and referral to the senior author(W.T.) if necessary.
Statistical analysis
The treatment effect for continuous outcomes are expressed as weighted mean difference (WMD) with 95% confidence intervals (CI) for all studies used the same scale[15]. For dichotomous data, the pooled risk ratios (RRs) would be used. Statistical heterogeneity across the studies was measured by I2 test, and a fixed-effects (FE) model (Mantel-Haenszel method) would be used if I2≤50%. In case of important heterogeneity (50%<I2≤75%), further exploration was made if appropriate, including sensitivity analysis and subgroup analysis. If heterogeneity remained significant, a random-effects (RE) model would be used. If there was considerable heterogeneity (I2>75%), the data would be regarded as unsuitable for pooling[15]. The publication bias was evaluated by using funnel plots with Begg’s test[16]. A value of P<0.05 indicated statistically significant. All analysis was completed in Stata v12.0 (Stata Corp, College Station, TX,USA) using the metan function.
Results
Baseline characteristics of included studies
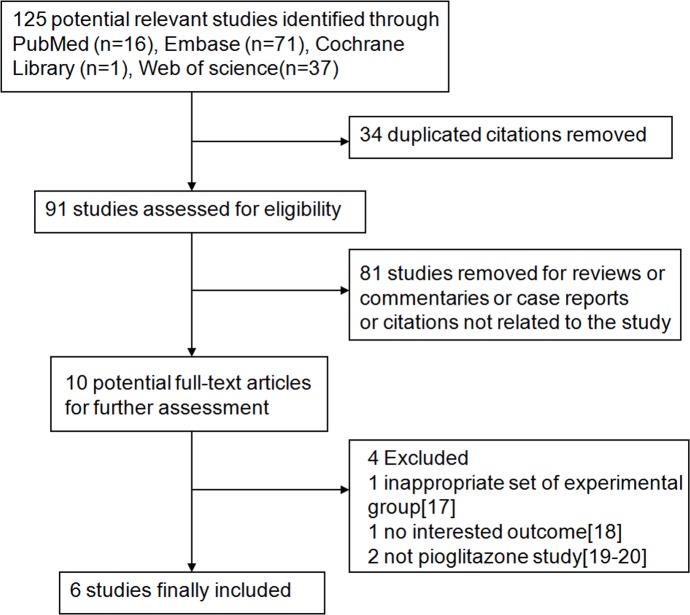
Our search strategy resulted in the initial identification of 125 records. After duplicated removal, 91 studies were left, in which 81 records were further excluded as non-clinical studies, reviews, case reports or irrelevant studies after the title and abstract screening. As a result, a total of 10 studies were left for full-text screening to determine eligibility for analysis.
After full-text screening, 4 studies were removed due to either inappropriate set of experimental group, lack of interested outcome or other reasons[17–20]. Finally, 6 studies (5 RCTs and 1 retrospective study) involved a total of 503 cases, were selected for inclusion in the meta-analysis. A flow chart summarizes the selection process of the studies (Fig 1), and the detail information of the included studies is provided in Table 1[21–26].
Fig 1. Flow chart of study selection.
Table 1. Baseline characteristics of selected studies.
| Study | Year | Study design | Age(PG/CG) | Sample size(PG/CG) | Stent type | Interventions | Outcomes | Follow-up | ||
|---|---|---|---|---|---|---|---|---|---|---|
| PG | CG | Routine Follow-up | Clinically Driven | |||||||
| Lee, H et al.[21] | 2013 | RCT | 60±10/62±9 | 60/61 | DES | pioglitazone, 15 mg/d | placebo | ISR; Late loss; Neointimal volume; MACE | TLR | 12 months |
| Soon Jun Hong et al.[22] | 2011 | RCT | 64±7/62±8 | 47/47 | DES | pioglitazone, 30 mg/d | placebo | ISR; Late loss; Neointimal volume; MACE | - | 8 months |
| Takagi, T et al.[23] | 2009 | RCT | 64±9/62±10 | 48/49 | BES | pioglitazone, 30 mg/d | placebo | ISR; Late loss; MACE | TLR | 6 months |
| Yokoyama, J et al.[24] | 2007 | Retrospective | 63±10/64±9 | 56/37 | BES | pioglitazone, 15 mg/d | without pioglitazone | ISR; Late loss | TLR | 6 months |
| Nishio, K et al.[25] | 2006 | RCT | 66±9/68±10 | 26/28 | BES | pioglitazone, 30 mg/d | without pioglitazone | ISR; Late loss | - | 6 months |
| Takagi, T et al.[26] | 2003 | RCT | 64±10/65±9 | 23/21 | BES | pioglitazone, 30 mg/d | without pioglitazone | ISR; Neointimal volume | TLR | 6 months |
PG = pioglitazone group; CG = control group; DES = drug-eluting stent; BES = bare-metal stent; ISR = in-stent restenosis; TLR = target lesion revascularization; MACE = major adverse cardiac events.
The quality assessment and publication bias
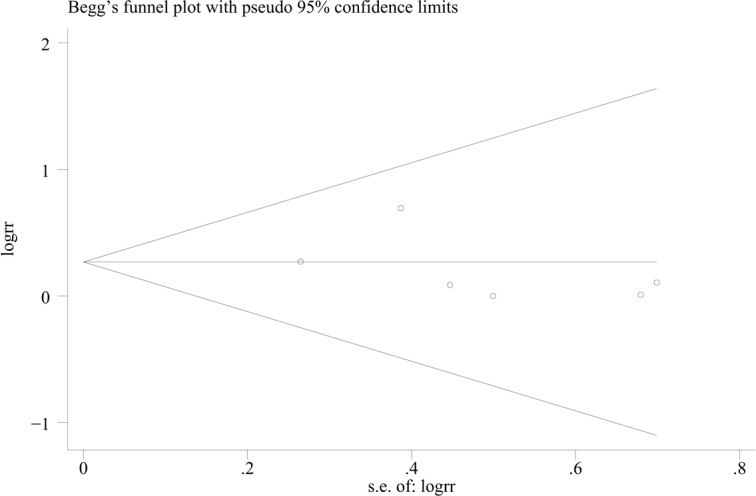
The assessment of study quality was performed by application of Modified Jadad scale, which is composed of randomization method, double-blinding, withdrawals and dropouts and allocation concealment[27]. The Jadad scores of the studies are summarized in Table 2. The publication bias was assessed by a funnel plot, which was based on the outcome of ISR (Fig 2), and no publication bias was found (Begg’s test, P = 0.707).
Table 2. Methodologic quality assessment of selected studies.
| Author | Randomization | Randomization method | Blinding | Withdrawals/dropouts | Allocation concealment | Scores |
|---|---|---|---|---|---|---|
| Lee, H et al.[21] | Yes | Unclear | Unclear | No | Unclear | 3 |
| Soon Jun Hong et al.[22] | Yes | Unclear | single blind | Yes | Unclear | 3 |
| Takagi, T et al.[23] | Yes | Unclear | open label | No | Unclear | 2 |
| Yokoyama, J et al.[24] | No | No | No | No | No | 0 |
| Nishio, K et al.[25] | Yes | Unclear | Unclear | No | Unclear | 3 |
| Takagi, T et al.[26] | Yes | Unclear | double blind | No | Unclear | 4 |
Fig 2. Funnel plot for the events of ISR.
Statistical Analysis Results
In Stent Restenosis
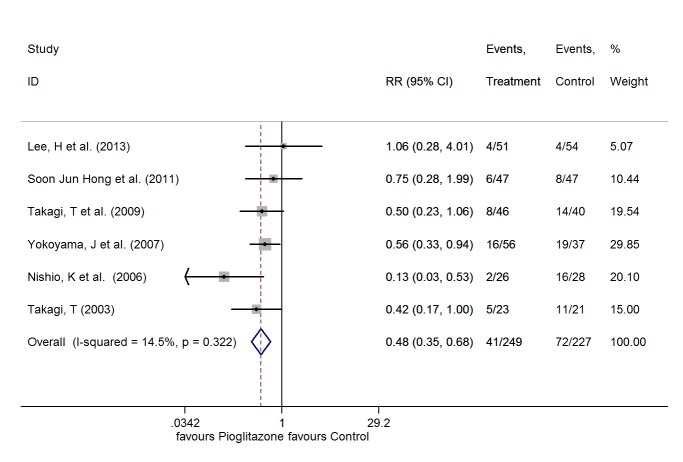
There was no significant heterogeneity among the six studies[21–26] (I2 = 14.5%, P = 0.322), so the FE model was used. In the six studies, the events of ISR occurred in 41 of 249 patients (16.5%) treated with pioglitazone and in 72 of 227 patients (31.7%) without pioglitazone treatment. The use of pioglitazone was associated with a significant reduction in the events of ISR (RR 0.48, 95% CI 0.35 to 0.68, P<0.001) (Fig 3).
Fig 3. RR of the events of ISR.
Late Loss
The late loss was reported after pioglitazone treatment in the diabetic patients suffered from PCI in 5 studies[21–25]. There was considerable heterogeneity among these studies (I2 = 81.9%, P<0.001). As a result, the data about late loss in the studies were not suitable for further analysis.
Neointimal Volume
The neointimal volume of the target lesion was investigated by IVUS at 1 year follow-up in 3 studies[21,22,26]. After pooling the data of the studies, we were not able to perform further analysis for the results of neointimal volume because of the considerable heterogeneity among the studies (I2 = 75.9%, P = 0.016).
Target Lesion Revascularization
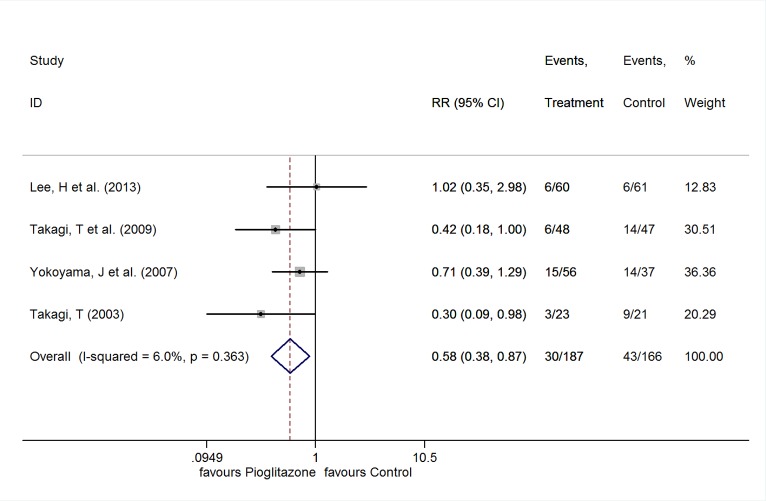
The meta-analysis was performed with FE model for there was no significant heterogeneity among the 4 studies [21,23,24,26] (I2 = 6.0%, P = 0.363). Pioglitazone treatment was associated with a significant reduction in the events of TLR (RR 0.58, 95%CI 0.38 to 0.87, P = 0.009) (Fig 4).
Fig 4. RR of the events of TLR.
Major Adverse Cardiac Events
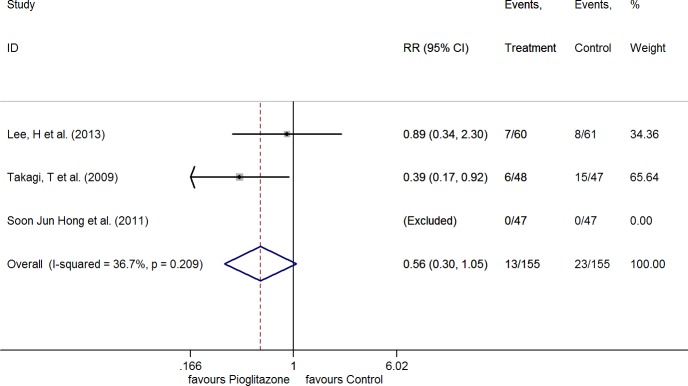
The incidence of MACE was reported in three studies[21–23], and there was no significant heterogeneity among them (I2 = 36.7%, P = 0.209). After the application of FE model for the meta-analysis, the result showed there was no association between the treatment of pioglitazone and MACE (RR 0.56, 95%CI 0.30 to 1.05, P = 0.071) (Fig 5).
Fig 5. RR of the events of MACE.
Discussion
Inflammation response evoked by stent implantation, which initiates several vascular remodeling processes including neointimal hyperplasia, is crucial contributor to the development of restenosis after PCI[28], especially in patients with T2DM[29]. As anti-diabetic agents involved in insulin sensitivity and lipid metabolism, TZDs can activate PPAR-γ, which is highly expressed in activated macrophages after PCI[30], to exert its anti-inflammatory and anti-proliferative effect in the process of ISR[31–34]. However, among the three TZDs, troglitazone is not approved due to its liver toxicity[8], and rosiglitazone is associated with increased risk of myocardial infarction[6]. So, as the only one which shows beneficial effects on cardiovascular outcomes in TZDs, pioglitazone is considered to be a more promising drug on the prevention of ISR[23,35].
Although the incidence of ISR was markedly reduced since DES have been widely used [36,37], a fairly high rate of ISR (10.1% to 17.6%) after DES implantation still exists in patients with T2DM[10,38], which probably due to the amplified inflammatory response and increased neointimal hyperplasia. Therefore, diabetes is one of the most powerful clinical predictors of ISR after PCI regardless of the kind of stents used[1,10,11,39]. Besides, little data is available about the dosage of pioglitazone that can appropriately prevent ISR after PCI with DES, especially in T2DM patients. Therefore, we made the meta-analysis without selection of the kind of stent and the dosage of pioglitazone, which might mainly account for the considerable heterogeneity of late loss and neointimal volume. Nevertheless, as the results have shown, no statistical heterogeneity was found among the included studies for most endpoints, indicating that the results of the study are convincing in some degree.
This study, comprising 503 T2DM patients, demonstrates that the use of pioglitazone was associated with a reduction in ISR and TLR. Although late loss and neointimal volume were not further analyzed due to the considerable heterogeneity among the studies, the beneficial effects of pioglitazone on these endpoints could not be ignored [22,23,25,26]. The benefits of pioglitazone for T2DM patients could be attributed to its favorable effect on lipid profile[40], attenuation of atherosclerotic plaque inflammation[4] and efflux of cholesterol from macrophages[41]. In this regards, Pioglitazone could be an useful agent on the prevention of ISR [42–44]. However, it is worth noting that no studies, including this meta-analysis, have found that pioglitazone reduces cardiovascular events, even the best available data for pioglitazone from the PROactive trial[45]. For this reason, the results of the study should be interpreted with care and more high quality clinical trials are needed.
Several limitations should be considered in this study. First, for only six studies met our inclusion criteria, the publication bias of the study could not be totally excluded for the low power of funnel plot asymmetry test. Second, the considerable heterogeneity mentioned above, together with the small sample size, lower quality of the RCT trials (Modified Jadad score ranged from 0–4) and the enrollment of one retrospective study, could make the conclusion less convincing. Finally, the lack of subgroup analysis, such as condition of the patients, selection of the stents and the dosage of pioglitazone might also lead to significant bias in the study. Therefore, the results of the study should be interpreted with caution and additional clinical trials are required.
Conclusions
The limited evidence indicates that the treatment of pioglitazone is associated with a reduction in ISR and TLR in T2DM patients suffering from PCI, except the incidence of MACE. However, due to the limitations of the present study, additional high quality RCTs are needed and the results of the study should be interpreted with care.
Supporting Information
(DOC)
Acknowledgments
We thank Yan-zhao Lu, Hua Li, Yu-jiao Sun and Li-na Ren for helpful suggestions on the study design and data collection.
Data Availability
All relevant data are within the paper and its Supporting Information file.
Funding Statement
The authors have no support or funding to report.
References
- 1.Silva JA, Ramee SR, White CJ, Collins TJ, Jenkins JS, Nunez E, et al. Primary stenting in acute myocardial infarction: influence of diabetes mellitus in angiographic results and clinical outcome. Am Heart J. 1999; 138: 446–455. [DOI] [PubMed] [Google Scholar]
- 2.Hong SJ, Kim MH, Cha KS, Park HS, Chae SC, Hur SH, et al. Comparison of three-year clinical outcomes between sirolimus-versus paclitaxel-eluting stents in diabetic patients: prospective randomized multicenter trial. Catheter Cardiovasc Interv. 2010; 76: 924–933. 10.1002/ccd.22369 [DOI] [PubMed] [Google Scholar]
- 3.Joner M, Farb A, Cheng Q, Finn AV, Acampado E, Burke AP, et al. Pioglitazone inhibits in-stent restenosis in atherosclerotic rabbits by targeting transforming growth factor-beta and MCP-1. Arterioscler Thromb Vasc Biol. 2007; 27: 182–189. [DOI] [PubMed] [Google Scholar]
- 4.Kasai T, Miyauchi K, Yokoyama T, Kajimoto K, Sumiyoshi K, Kubota N, et al. Pioglitazone attenuates neointimal thickening via suppression of the early inflammatory response in a porcine coronary after stenting. Atherosclerosis. 2008; 197: 612–619. [DOI] [PubMed] [Google Scholar]
- 5.Okura H, Takagi T, Yoshida K. Therapies Targeting Inflammation After Stent Implantation.Curr Vasc Pharmacol. 2013; 11: 399–406. [DOI] [PubMed] [Google Scholar]
- 6.Nissen SE, Wolski K. Effect of rosiglitazone on the risk of myocardial infarction and death from cardiovascular causes. N Engl J Med. 2007; 356: 2457–2471. [DOI] [PubMed] [Google Scholar]
- 7.Lincoff AM, Wolski K, Nicholls SJ, Nissen SE. Pioglitazone and risk of cardiovascular events in patients with type 2 diabetes mellitus: a meta-analysis of randomized trials. Jama. 2007; 298: 1180–1188. [DOI] [PubMed] [Google Scholar]
- 8.Henney JE. Withdrawal of Troglitazone and Cisapride. Jama. 2000; 283: 2228.10807369 [Google Scholar]
- 9.Patel D, Walitt B, Lindsay J, Wilensky RL. Role of pioglitazone in the prevention of restenosis and need for revascularization after bare-metal stent implantation: a meta-analysis. JACC Cardiovasc Interv. 2011; 4: 353–360. 10.1016/j.jcin.2010.10.016 [DOI] [PubMed] [Google Scholar]
- 10.Moussa I, Leon MB, Baim DS, O'Neill WW, Popma JJ, Buchbinder M, et al. Impact of sirolimus-eluting stents on outcome in diabetic patients: a SIRIUS (SIRolImUS-coated Bx Velocity balloon-expandable stent in the treatment of patients with de novo coronary artery lesions) substudy. Circulation. 2004; 109: 2273–2278. [DOI] [PubMed] [Google Scholar]
- 11.Kitoga M, Pasquet A, Preumont V, Kefer J, Hermans MP, Vanoverschelde JL, et al. Coronary in-stent restenosis in diabetic patients after implantation of sirolimus or paclitaxel drug-eluting coronary stents. Diabetes Metab. 2008; 34: 62–67. [DOI] [PubMed] [Google Scholar]
- 12.Hong SJ, Kim MH, Ahn TH, Ahn YK, Bae JH, Shim WJ, et al. Multiple predictors of coronary restenosis after drug-eluting stent implantation in patients with diabetes. Heart. 2006; 92: 1119–1124. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Guerra E, Byrne RA, Kastrati. A Pharmacological inhibition of coronary restenosis: systemic and local approaches. Expert Opin Pharmacother. 2014; 15: 2155–2171. 10.1517/14656566.2014.948844 [DOI] [PubMed] [Google Scholar]
- 14.Moher D, Liberati A, Tetzlaff J, Altman DG. Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. Ann Intern Med. 2009; 151: 264–269, W64. [DOI] [PubMed] [Google Scholar]
- 15.Higgins J, Green S (2010) Cochrane handbook for systematic reviews of interventions version 5.0. 2 [updated September 2009]. The Cochrane Collaboration, 2009. Available: www.cochrane-handbook.org.
- 16.Begg CB, Mazumdar M. Operating characteristics of a rank correlation test for publication bias. Biometrics. 1994; 50: 1088–1101. [PubMed] [Google Scholar]
- 17.Yeh H, Hsieh C, Tsai Y, Huang W. Effects of Thiazolidinediones on Cardiovascular Events in Patients With Type 2 Diabetes Mellitus After Drug-Eluting Stent Implantation: A Retrospective Cohort Study Using the National Health Insurance Database in Taiwan. Clinical Therapeutics. 2012; 34: 885–893. 10.1016/j.clinthera.2012.02.024 [DOI] [PubMed] [Google Scholar]
- 18.Ozawa T, Oda H, Oda M, Hosaka Y, Kashimura T, Ozaki K, et al. Improved cardiac function after sirolimus-eluting stent placement in diabetic patients by pioglitazone: Combination therapy with statin. Journal of Cardiology. 2009; 53: 402–409. 10.1016/j.jjcc.2009.01.011 [DOI] [PubMed] [Google Scholar]
- 19.Takagi T, Yamamuro A, Tamita K, Katayama M, Morioka S. Thiazolidinedione treatment attenuates diffuse neointimal hyperplasia in restenotic lesions after coronary stent implantation in type 2 diabetic patients: An intravascular ultrasound study. Journal of Cardiology. 2005; 45: 139–147. [PubMed] [Google Scholar]
- 20.Takagi T, Akasaka T, Yamamuro A, Honda Y, Hozumi T, Morioka S, et al. Impact of insulin resistance on neointimal tissue proliferation after coronary stent implantationIntravascular ultrasound studies. Journal of Diabetes and its Complications. 2002; 16: 50–55. [DOI] [PubMed] [Google Scholar]
- 21.Lee HW, Lee HC, Kim BW, Yang MJ, Park JS, Oh JH, et al. Effects of low dose pioglitazone on restenosis and coronary atherosclerosis in diabetic patients undergoing drug eluting stent implantation. Yonsei Med J. 2013; 54: 1313–1320. 10.3349/ymj.2013.54.6.1313 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Ahn CM, Hong SJ, Kim JH, Lee KM, Kim JS, Park JH, et al. Cellular and molecular changes associated with inhibitory effect of pioglitazone on neointimal growth in patients with type 2 diabetes after zotarolimus-eluting stent implantation. J Am Coll Cardiol. 2011; 57: E1914. [DOI] [PubMed] [Google Scholar]
- 23.Takagi T, Okura H, Kobayashi Y, Kataoka T, Taguchi H, Toda I, et al. A prospective, multicenter, randomized trial to assess efficacy of pioglitazone on in-stent neointimal suppression in type 2 diabetes: POPPS (Prevention of In-Stent Neointimal Proliferation by Pioglitazone Study). JACC Cardiovasc Interv. 2009; 2: 524–531. 10.1016/j.jcin.2009.04.007 [DOI] [PubMed] [Google Scholar]
- 24.Yokoyama J, Sutoh N, Higuma T, Horiuchi D, Katoh C, Yokota T, et al. Efficacy and safety of low-dose pioglitazone after primary coronary angioplasty with the use of bare metal stent in patients with acute myocardial infarction and with type 2 diabetes mellitus or impaired glucose tolerance. Heart Vessels. 2007; 22: 146–151. [DOI] [PubMed] [Google Scholar]
- 25.Nishio K, Sakurai M, Kusuyama T, Shigemitsu M, Fukui T, Kawamura K, et al. A randomized comparison of pioglitazone to inhibit restenosis after coronary stenting in patients with type 2 diabetes. Diabetes Care. 2006; 29: 101–106. [DOI] [PubMed] [Google Scholar]
- 26.Takagi T, Yamamuro A, Tamita K, Yamabe K, Katayama M, Mizoguchi S, et al. Pioglitazone reduces neointimal tissue proliferation after coronary stent implantation in patients with type 2 diabetes mellitus: an intravascular ultrasound scanning study. Am Heart J. 2003; 146: E5 [DOI] [PubMed] [Google Scholar]
- 27.Oremus M, Wolfson C, Perrault A, Demers L, Momoli F, Moride Y. Interrater reliability of the modified Jadad quality scale for systematic reviews of Alzheimer's disease drug trials. Dement Geriatr Cogn Disord. 2001; 12: 232–236. [DOI] [PubMed] [Google Scholar]
- 28.Kornowski R, Hong MK, Tio FO, Bramwell O, Wu H, Leon MB. In-stent restenosis: contributions of inflammatory responses and arterial injury to neointimal hyperplasia. J Am Coll Cardiol. 1998; 31: 224–230. [DOI] [PubMed] [Google Scholar]
- 29.Gilbert RE. Endothelial loss and repair in the vascular complications of diabetes: pathogenetic mechanisms and therapeutic implications. Circ J. 2013; 77: 849–856. [DOI] [PubMed] [Google Scholar]
- 30.Delhaye C, Maluenda G, Wakabayashi K, Ben-Dor I, Lemesle G, Collins SD, et al. Long-term prognostic value of preprocedural C-reactive protein after drug-eluting stent implantation. Am J Cardiol. 2010; 105: 826–832. 10.1016/j.amjcard.2009.10.064 [DOI] [PubMed] [Google Scholar]
- 31.Satoh N, Ogawa Y, Usui T, Tagami T, Kono S, Uesugi H, et al. Antiatherogenic effect of pioglitazone in type 2 diabetic patients irrespective of the responsiveness to its antidiabetic effect. Diabetes Care. 2003; 26: 2493–2499. [DOI] [PubMed] [Google Scholar]
- 32.Yki-Järvinen H. Thiazolidinediones. N Engl J Med. 2004; 351: 1106–1118. [DOI] [PubMed] [Google Scholar]
- 33.Barlaka E, Galatou E, Mellidis K, Ravingerova T, Lazou A. Role of pleiotropic properties of peroxisome proliferator-activated receptors in the heart: focus on the non-metabolic effects in cardiac protection. Cardiovasc Ther. 2015. [DOI] [PubMed] [Google Scholar]
- 34.Cheang WS, Fang X, Tian XY. Pleiotropic effects of peroxisome proliferator-activated receptor gamma and delta in vascular diseases. Circ J. 2013; 77: 2664–2671. [DOI] [PubMed] [Google Scholar]
- 35.Hong SJ, Choi SC, Cho JY, Joo HJ, Park JH, Yu CW, et al. Pioglitazone increases circulating microRNA-24 with decrease in coronary neointimal hyperplasia in type 2 diabetic patients- optical coherence tomography analysis. Circ J. 2015; 79: 880–888. 10.1253/circj.CJ-14-0964 [DOI] [PubMed] [Google Scholar]
- 36.Roiron C, Sanchez P, Bouzamondo A, Lechat P, Montalescot G. Drug eluting stents: an updated meta-analysis of randomised controlled trials. Heart. 2006; 92: 641–649. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Wood TC, Marks AR. Drug-eluting stents. Annu Rev Med. 2004; 55: 169–178. [DOI] [PubMed] [Google Scholar]
- 38.Yang TH, Park SW, Hong MK, Park DW, Park KM, Kim YH, et al. Impact of diabetes mellitus on angiographic and clinical outcomes in the drug-eluting stents era. Am J Cardiol. 2005; 96: 1389–1392. [DOI] [PubMed] [Google Scholar]
- 39.Abizaid A, Kornowski R, Mintz GS, Hong MK, Abizaid AS, Mehran R, et al. The influence of diabetes mellitus on acute and late clinical outcomes following coronary stent implantation. J Am Coll Cardiol. 1998; 32: 584–589. [DOI] [PubMed] [Google Scholar]
- 40.Goldberg RB, Kendall DM, Deeg MA, Buse JB, Zagar AJ, Pinaire JA, et al. A comparison of lipid and glycemic effects of pioglitazone and rosiglitazone in patients with type 2 diabetes and dyslipidemia. Diabetes Care. 2005; 28: 1547–1554. [DOI] [PubMed] [Google Scholar]
- 41.Nissen SE, Nicholls SJ, Wolski K, Nesto R, Kupfer S, Perez A, et al. Comparison of pioglitazone vs glimepiride on progression of coronary atherosclerosis in patients with type 2 diabetes: the PERISCOPE randomized controlled trial. Jama. 2008; 299: 1561–1573. 10.1001/jama.299.13.1561 [DOI] [PubMed] [Google Scholar]
- 42.Mizoguchi M, Tahara N, Tahara A, Nitta Y, Kodama N, Oba T, et al. Pioglitazone attenuates atherosclerotic plaque inflammation in patients with impaired glucose tolerance or diabetes a prospective, randomized, comparator-controlled study using serial FDG PET/CT imaging study of carotid artery and ascending aorta. JACC Cardiovasc Imaging. 2011; 4: 1110–1118. 10.1016/j.jcmg.2011.08.007 [DOI] [PubMed] [Google Scholar]
- 43.Nitta Y, Tahara N, Tahara A, Honda A, Kodama N, Mizoguchi M, et al. Pioglitazone decreases coronary artery inflammation in impaired glucose tolerance and diabetes mellitus: evaluation by FDG-PET/CT imaging. JACC Cardiovasc Imaging. 2013; 6: 1172–1182. 10.1016/j.jcmg.2013.09.004 [DOI] [PubMed] [Google Scholar]
- 44.Saremi A, Schwenke DC, Buchanan TA, Hodis HN, Mack WJ, Banerji M, et al. Pioglitazone slows progression of atherosclerosis in prediabetes independent of changes in cardiovascular risk factors. Arterioscler Thromb Vasc Biol. 2013; 33: 393–399. 10.1161/ATVBAHA.112.300346 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Erdmann E, Harding S, Lam H, Perez A. Ten-year observational follow-up of PROactive: a randomized cardiovascular outcomes trial evaluating pioglitazone in type 2 diabetes. Diabetes Obes Metab. 2016;18: 266–273. 10.1111/dom.12608 [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
(DOC)
Data Availability Statement
All relevant data are within the paper and its Supporting Information file.