Abstract
Background:
The objective of this study was to study and compare the effects of intravenous dexmedetomidine and fentanyl on intraoperative hemodynamics, opioid consumption, and recovery characteristics in hypertensive patients.
Methods:
Fifty-seven hypertensive patients undergoing major surgery were randomized into two groups, Group D (dexmedetomidine, n = 29) and Group F (fentanyl, n = 28). The patients received 1 μg/kg of either dexmedetomidine or fentanyl, followed by 0.5 μg/kg/h infusion of the same drug, followed by a standard induction protocol. Heart rate (HR), mean arterial pressures (MAPs), end-tidal isoflurane concentration, and use of additional fentanyl and vasopressors were recorded throughout.
Results:
Both dexmedetomidine and fentanyl caused significant fall in HR and MAP after induction and dexmedetomidine significantly reduced the induction dose of thiopentone (P = 0.026). After laryngoscopy and intubation, patients in Group D experienced a fall in HR and a small rise in MAP (P = 0.094) while those in Group F showed significant rise in HR (P = 0.01) and MAP (P = 0.004). The requirement of isoflurane and fentanyl boluses was significantly less in Group D. The duration of postoperative analgesia was longer in Group D (P = 0.015) with significantly lower postoperative nausea and vomiting (PONV) (P < 0.001).
Conclusion:
Infusion of dexmedetomidine in hypertensive patients controlled the sympathetic stress response better than fentanyl and provided stable intraoperative hemodynamics. It reduced the dose of thiopentone, requirement of isoflurane and fentanyl boluses. The postoperative analgesia was prolonged, and incidence of PONV was less in patients who received dexmedetomidine.
Keywords: Dexmedetomidine, hemodynamic stability, hypertension, postoperative nausea and vomiting, stress response
INTRODUCTION
The perioperative management of hypertensive patients is always a challenge to anesthesiologists. The progressive ventricular hypertrophy, which is known to occur in hypertensive patients, leads to diastolic dysfunction. This diastolic dysfunction worsens whenever there is tachycardia in the perioperative period and may lead to myocardial infarction resulting in severe morbidity and even mortality.[1] In poorly controlled hypertensive patients, a more severe hemodynamic response to laryngoscopy and intubation can be observed when compared with well-controlled hypertensive patients.[2]
A variety of pharmacological agents namely opioids, beta blockers, calcium channel blockers, combined alpha and beta blockers, lignocaine, and alpha-2 receptor agonists have been used to maintain stable hemodynamics in the perioperative period. Fentanyl is a popular choice for control of hemodynamics in the perioperative period. However, it is associated with side effects such as respiratory depression and increased incidence of postoperative nausea and vomiting (PONV).[3,4] Dexmedetomidine, a highly selective alpha-2 agonist, has sympatholytic, antinociceptive, sedative, and anti-sialagogue properties without causing respiratory depression.[5,6,7,8]
Numerous studies have shown the benefit of dexmedetomidine in blunting the stress response to laryngoscopy and intubation.[9,10,11,12,13,14] Literature is scarce regarding the role of dexmedetomidine in providing stable intraoperative hemodynamics in hypertensive patients. Hence, we designed this double-blinded randomized controlled trial to compare the effect of infusions of dexmedetomidine and fentanyl on intraoperative hemodynamics, recovery characteristics, and PONV in hypertensive patients.
METHODS
Ethical approval for this randomized double-blinded controlled study (SEC/2011/4/50) was given by the Institute Ethics Committee (Human Studies), in a statement dated February 2, 2012. The sample size was calculated based on a previous study in hypertensive patients by Uysal et al., at a significance level of 5%.[10] Fifty-seven hypertensive patients were enrolled for the study between March 2012 and January 2014 conducted in the operation theaters of a Tertiary Care Teaching Hospital. Hypertensive patients aged 18–70 years scheduled for major surgery were included in the study. The patients with uncontrolled hypertension (systolic blood pressure [BP] >160 mm Hg or diastolic BP >100 mm Hg), history of ischemic heart disease, cerebrovascular disease, impaired liver or renal functions, and suspected full stomach patients including pregnant patients were excluded from the study. Patients with anticipated difficult airway, laparoscopic procedures, and those with expected blood loss >25% of blood volume were also excluded from the study.
The patients were randomized into dexmedetomidine (Group D) and fentanyl (Group F) groups using a computer-generated randomization table. The patients, the attending anesthesiologist, and the primary investigator were blinded to the study drug allocated to patients. Sequentially numbered opaque sealed envelope technique was used for blinding the anesthesiologists attending to the patients [Figure 1].
Figure 1.
CONSORT flow diagram for the present study
Anesthetic management
All the patients were premedicated with tablet famotidine 20 mg and tablet diazepam 5 mg on the night before and on the morning of the surgery. All anti-hypertensives except angiotensin-converting enzyme inhibitors and angiotensin receptor antagonists were continued in the preoperative period.
Induction
Standard monitors were attached, and baseline parameters such as heart rate (HR), BP, and oxygen (O2) saturation were noted. Dexmedetomidine or fentanyl 100 µg was loaded in a 50 ml syringe and diluted with normal saline (concentration 2 µg/ml). The attending anesthesiologist was blinded to the study drug. Intravenous infusion of the study drug was started to deliver 1 µg/kg bolus over 10 min. Midway through the bolus infusion (after 5 min), fentanyl 1 µg/kg was given intravenously in both groups. After 7 min of starting the bolus infusion, patients were induced with intravenous thiopentone 2.5% titrated to loss of eyelash reflex. Vecuronium bromide 0.1 mg/kg was used for muscle relaxation. The patients were manually ventilated with nitrous oxide (N2O) and O2 mixture (66% +33%) with isoflurane 1–1.3 minimum alveolar concentration (MAC). At the 10th min, laryngoscopy and intubation were done using appropriate size endotracheal tube and the infusion rate of the study drug was changed to 0.5 µg/kg/h.
Maintenance
Anesthesia was maintained on controlled ventilation with N2O in O2 mixture (66%:33%) with isoflurane between 0.7 and 2 MAC. If mean arterial pressure (MAP) dropped >20% from the baseline, isoflurane concentration was reduced up to 0.7 MAC for 3 min. If MAP failed to normalize, boluses of intravenous mephentermine 1.5 mg were used until MAP normalized to within 20% from the baseline. If HR or MAP increased by >20% of the baseline, isoflurane concentration was increased up to 2 MAC and intravenous fentanyl 10 µg boluses were given if MAP failed to normalize. A fall in the HR to <50/min was treated with atropine 0.3 mg intravenously. HR, MAP, and end-tidal isoflurane concentrations were recorded every 1 min for the first 5 min and every 10 min thereafter. The requirement of additional boluses of fentanyl and mephentermine were noted.
Recovery
Study drug infusion and isoflurane were stopped at the end of the surgery. Neuromuscular blockade was reversed with neostigmine 50 µg/kg and glycopyrrolate 10 µg/kg. The time for eye opening on call or spontaneously was noted as the recovery time. On adequate reversal, patients were extubated and monitored in the PACU for 2 h. The time of the first request for analgesia and the occurrence of PONV were noted.
Statistical analysis
The analysis was carried out using SPSS 16 statistical software (SPSS 16.0 by IBM). Results on continuous measurements are presented on mean ± standard deviation (minimum-maximum) and results on categorical measurements are presented in number (%). Significance is assessed at 5% level of significance. The dependent variables were tested for normality. Student's t-test (two-tailed, dependent) was used to find the significance of study parameters on continuous scale with in each group and Leveneuo test has been performed to assess the homogeneity of variance. Repeated measures ANOVA have been used to find the significance of variation in HR and MAP within groups and between the groups. The Chi-square/Fisher exact test has been used to find the significance of study parameters on categorical scale between two or more groups.
RESULTS
Patient demographics such as age, sex, and body weight were comparable in both groups. The duration of surgery was also comparable in the two groups [Table 1].
Table 1.
Patient demographics

Heart rate
The baseline HR was comparable in both the groups (P = 0.386). At the end of the bolus infusion, HR decreased significantly in both Groups D and F (P < 0.001). After intubation, there was significant increase in HR in Group F (from 87.54 ± 16.70/min at baseline to 95.43 ± 15.87/min, P = 0.01) whereas HR was lower than the baseline in Group D (from 91.62 ± 18.52/min at baseline to 89.38 ± 15.46/min, P = 0.456). Overall, both dexmedetomidine and fentanyl reduced the HR significantly throughout the duration of surgery as compared to the baseline (intra-group analysis). However, the mean HR was comparable in Group D and Group F (inter-group analysis P > 0.05). At extubation, the mean HRs in both the groups were not raised significantly from the baseline (Group D 84.38 ± 20.34/min, P = 0.162; Group F 84.54 ± 15.64/min, P = 0.473) [Figure 2].
Figure 2.
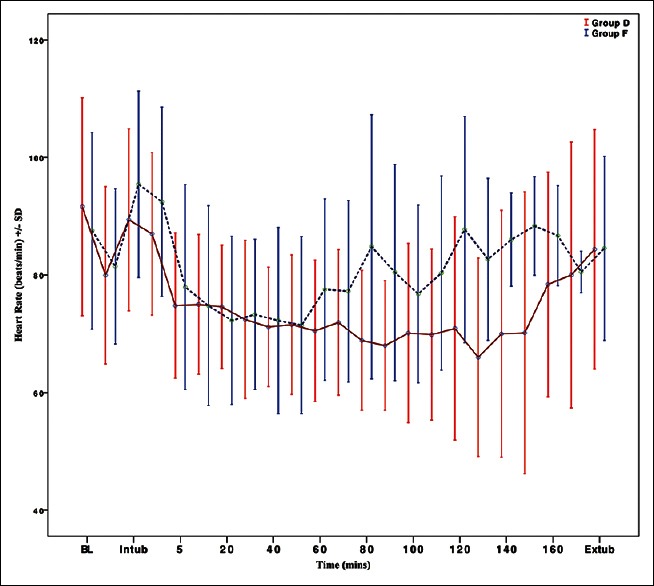
Intraoperative variation in the mean heart rate in the two groups
Mean arterial pressure
The baseline MAP were comparable in both the groups (P = 0.841). There was significant reduction in MAP at the end of the bolus infusion in both the groups (P < 0.01, intra-group analysis). Significant increase in MAP was noted in Group F at laryngoscopy and intubation (from 101.82 ± 13.46 to 113.86 ± 18.69 mm Hg, P = 0.004 in Group F vs. 102.45 ± 9.74 to 108.07 ± 19.2 mm Hg P = 0.094 in Group D). The patients receiving dexmedetomidine experienced significant reductions in MAP after 5 min of intubation and the trend continued throughout the surgery. Fluctuating levels of MAP were observed in Group F. At extubation, the rise in MAP was significant from the baseline values in Group F (112.83 ± 15.85 mm Hg, P = 0.008 within-group analysis) compared to Group D (109.18 ± 20.75 mm Hg, P = 0.097 within-group analysis) [Figure 3].
Figure 3.
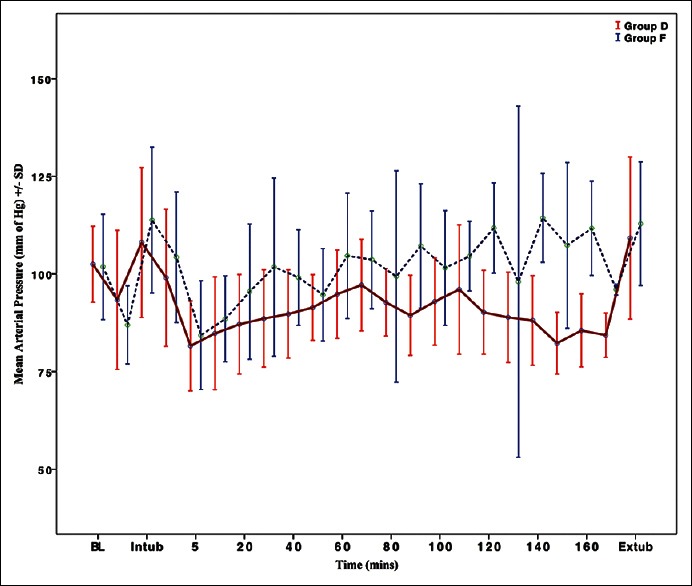
Intraoperative variation in the mean arterial pressure in the two groups
Induction dose of thiopentone
The induction dose of thiopentone was titrated to loss of eyelash reflex. The patients in Group D required significantly less amount of thiopentone as compared to Group F (206.03 ± 48.94 mg vs. 229.46 ± 23.62 mg; P = 0.026).
Intraoperative anesthetic requirement
Dexmedetomidine showed significantly better anesthetic sparing effects, with end-tidal concentrations of isoflurane were significantly less in Group D than in Group F [Figure 4].
Figure 4.
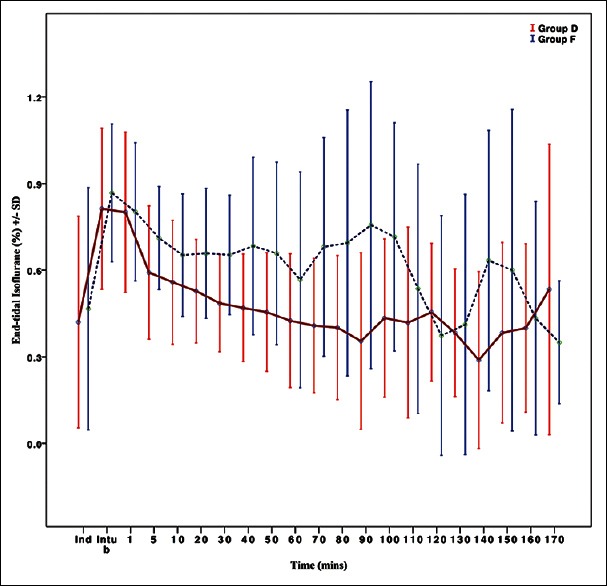
End-tidal isoflurane concentration in the two groups
Intraoperative interventions
The patients in Group F received higher amount of fentanyl boluses compared to patients in Group D (P = 0.003). The patients in Group D required higher amount of mephentermine boluses for maintaining MAP (P = 0.039).
Recovery from anesthesia
The time for eye-opening and time for extubation were comparable in both groups. Postoperative pain relief was better in Group D compared to Group F, and time to request for first analgesic was longer in patients receiving dexmedetomidine [Table 2].
Table 2.
Recovery characteristics and postoperative nausea and vomiting

Postoperative nausea and vomiting
Dexmedetomidine group had significantly reduced incidence of PONV (P < 0.001). Fourteen patients in Group F had postoperative nausea or vomiting compared to only one patient in Group D [Table 2].
DISCUSSION
Hemodynamic stability is of paramount importance in the perioperative period, more so in hypertensive patients.[2,15,16] In the present study, we compared the dexmedetomidine-based anesthesia technique with the standard opioid (fentanyl)-based technique for hemodynamic stability, intraoperative anesthetic requirement, and recovery characteristics.
With administration of the loading dose of dexmedetomidine, we found a reduced requirement of the induction dose of thiopentone, when compared to fentanyl administration. This is similar to studies performed earlier by various researchers.[9,10,17] The reduction in the dose of thiopentone (by about 30%) has been attributed to reduction in its volume of distribution and clearance in a study by Bührer et al.[18]
In our study, we found a significant fall in HR and MAP after dexmedetomidine and fentanyl infusion. In a study by Uysal et al. that compared dexmedetomidine with esmolol and sufentanil in hypertensive patients, administration of dexmedetomidine before induction of anesthesia reduced the HR, systolic and diastolic BP.[10]
In the present study, dexmedetomidine provided better hemodynamic stability during laryngoscopy and intubation. It successfully prevented the increases in HR and MAP as compared to fentanyl. Uysal et al. similarly found a reduced hemodynamic response to intubation with dexmedetomidine compared with esmolol and sufentanil. Previous studies by various investigators also showed reduced stress response to this stimulus by administration dexmedetomidine in healthy normotensive patients.[9,11,12]
In the present study, the bolus dose of the study drug was followed by a maintenance dose until the end of the surgery. HR was maintained on the lower side in the dexmedetomidine and fentanyl groups. MAP then fell significantly in the both the groups 5 min after intubation. MAP was significantly lower in the dexmedetomidine group, and those patients received more mephentermine boluses than those who received fentanyl infusion.
Many previous studies, including that by Patel et al., found a significant decrease and less variability in the HRs of patients who received dexmedetomidine, and a low and stable systolic and diastolic BPs as well. However, Patel et al. had administered the maintenance dose of dexmedetomidine while the others administered a 1 µg/kg loading dose alone.[13] Lawrence and De Lange had also demonstrated lower perioperative serum catecholamine concentrations in the dexmedetomidine group.
The requirement of isoflurane was found to be significantly less in the patients who received dexmedetomidine than in those who received fentanyl. Thornton et al. studied evoked responses in healthy volunteers and found a reduced requirement of isoflurane concentrations in patients receiving dexmedetomidine.[19] This is similar to previous studies that also noted significantly lower requirements of isoflurane concentrations with the administration of dexmedetomidine.[9,20,21]
In the present study, the mean opioid consumption was significantly higher in the fentanyl group than in the dexmedetomidine group. This finding of the opioid-sparing effect of dexmedetomidine corroborates with that in previous studies comparing the two drugs.[12,22,23] This property confers a distinct advantage to dexmedetomidine-based anesthesia as a host of side effects of opioids, such as nausea and vomiting, respiratory depression, pruritus, postoperative ileus, and urinary retention, can be minimized.
Extubation is challenging in hypertensive patients. Any sympathetic response during emergence from anesthesia can increase morbidity. In our study, by continuing a maintenance infusion of the drug throughout surgery, we achieved lower but comparable HRs at extubation in both the groups. However, the hypertensive response was significantly lower in the dexmedetomidine group compared to the baseline unlike that seen with fentanyl, indicating better control of BP compared to fentanyl.
The time to recovery from anesthesia and extubation after stopping the infusion, in our study, was found to be comparable. In the study by Patel et al., the test group received dexmedetomidine maintenance infusion of 0.2–0.8 µg/kg/h after the bolus dose, while the control group did not receive an infusion. The test group experienced greater sedation postoperatively compared to the control group. In our study, the fentanyl group continued to receive maintenance infusion of the test drug throughout the surgery, similar to the dexmedetomidine group.[13]
Our study found that the duration of postoperative analgesia, as seen by the delayed request for first analgesic dose by the patients, was significantly prolonged in the dexmedetomidine group, indicating more satisfactory postoperative pain relief and comfort. Ohtani et al. used dexmedetomidine as 1 µg/kg/h infusion and found lower requirement of analgesic postoperatively, similar to the observation in our study.[24]
Increased use of opioids and inhalational agents has been associated with higher incidence of PONV.[3,4] By its opioid-sparing properties, dexmedetomidine can reduce the incidence of PONV. In our study, only one patient who received dexmedetomidine infusion had PONV as compared to 14 patients who received fentanyl infusion. Previous studies and a systematic review have also suggested a reduced incidence of PONV when dexmedetomidine was used as an adjunct to anesthesia.[22,25]
A limitation of our study was that only hemodynamic parameters and end-tidal concentration of isoflurane were used to monitor the depth of anesthesia. No electroencephalogram-based monitors were used.
CONCLUSION
In our study, dexmedetomidine, when compared to fentanyl, reduced the sympathetic response to laryngoscopy and intubation in hypertensive patients, maintained stable hemodynamics in the intraoperative period as well as during extubation. It reduced the requirement for isoflurane and intraoperative opioid consumption. The patients who received dexmedetomidine had lower incidence of PONV and comparatively prolonged postoperative analgesia. Thus, overall dexmedetomidine infusion was found to be better than fentanyl as an adjuvant to general anesthesia in hypertensive patients.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest
Acknowledgments
We would like to thank professor and head of the department Dr. Pankaj Kundra, Prof. Ashok S. Badhe, and Prof. Hemavathy for helping with the study design.
REFERENCES
- 1.Pirracchio R, Cholley B, De Hert S, Solal AC, Mebazaa A. Diastolic heart failure in anaesthesia and critical care. Br J Anaesth. 2007;98:707–21. doi: 10.1093/bja/aem098. [DOI] [PubMed] [Google Scholar]
- 2.Prys-Roberts C, Greene LT, Meloche R, Foëx P. Studies of anaesthesia in relation to hypertension. II. Haemodynamic consequences of induction and endotracheal intubation. Br J Anaesth. 1971;43:531–47. doi: 10.1093/bja/43.6.531. [DOI] [PubMed] [Google Scholar]
- 3.Apfel CC, Heidrich FM, Jukar-Rao S, Jalota L, Hornuss C, Whelan RP, et al. Evidence-based analysis of risk factors for postoperative nausea and vomiting. Br J Anaesth. 2012;109:742–53. doi: 10.1093/bja/aes276. [DOI] [PubMed] [Google Scholar]
- 4.Gan TJ. Mechanisms underlying postoperative nausea and vomiting and neurotransmitter receptor antagonist-based pharmacotherapy. CNS Drugs. 2007;21:813–33. doi: 10.2165/00023210-200721100-00003. [DOI] [PubMed] [Google Scholar]
- 5.Nelson LE, Lu J, Guo T, Saper CB, Franks NP, Maze M. The alpha2-adrenoceptor agonist dexmedetomidine converges on an endogenous sleep-promoting pathway to exert its sedative effects. Anesthesiology. 2003;98:428–36. doi: 10.1097/00000542-200302000-00024. [DOI] [PubMed] [Google Scholar]
- 6.Bhana N, Goa KL, McClellan KJ. Dexmedetomidine. Drugs. 2000;59:263–8. doi: 10.2165/00003495-200059020-00012. [DOI] [PubMed] [Google Scholar]
- 7.Hsu YW, Cortinez LI, Robertson KM, Keifer JC, Sum-Ping ST, Moretti EW, et al. Dexmedetomidine pharmacodynamics: Part I: Crossover comparison of the respiratory effects of dexmedetomidine and remifentanil in healthy volunteers. Anesthesiology. 2004;101:1066–76. doi: 10.1097/00000542-200411000-00005. [DOI] [PubMed] [Google Scholar]
- 8.Ebert TJ, Hall JE, Barney JA, Uhrich TD, Colinco MD. The Effects of increasing plasma concentrations of dexmedetomidine in humans. Anesthesiology. 2000;93:382–94. doi: 10.1097/00000542-200008000-00016. [DOI] [PubMed] [Google Scholar]
- 9.Lawrence CJ, De Lange S. Effects of a single pre-operative dexmedetomidine dose on isoflurane requirements and peri-operative haemodynamic stability. Anaesthesia. 1997;52:736–44. doi: 10.1111/j.1365-2044.1997.169-az0303.x. [DOI] [PubMed] [Google Scholar]
- 10.Uysal HY, Tezer E, T zer36- M, Aslanargun P, Balan H. The effects of dexmedetomidine on hemodynamic responses to tracheal intubation in hypertensive patients: A comparison with esmolol and sufentanil. J Res Med Sci. 2012;17:22–31. [PMC free article] [PubMed] [Google Scholar]
- 11.Lee JH, Kim H, Kim HT, Kim MH, Cho K, Lim SH, et al. Comparison of dexmedetomidine and remifentanil for attenuation of hemodynamic responses to laryngoscopy and tracheal intubation. Korean J Anesthesiol. 2012;63:124–9. doi: 10.4097/kjae.2012.63.2.124. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Bajwa SJ, Kaur J, Singh A, Parmar S, Singh G, Kulshrestha A, et al. Attenuation of pressor response and dose sparing of opioids and anaesthetics with pre-operative dexmedetomidine. Indian J Anaesth. 2012;56:123–8. doi: 10.4103/0019-5049.96303. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Patel CR, Engineer SR, Shah BJ, Madhu S. Effect of intravenous infusion of dexmedetomidine on peri-operative hemodynamic changes and postoperative recovery: A study with entropy analysis. Indian J Anaesth. 2012;56:542–6. doi: 10.4103/0019-5049.104571. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Gogus N, Akan B, Serger N, Baydar M. The comparison of the effects of dexmedetomidine, fentanyl and esmolol on prevention of hemodynamic response to intubation. Rev Bras Anestesiol. 2014;64:314–9. doi: 10.1016/j.bjan.2013.10.010. [DOI] [PubMed] [Google Scholar]
- 15.Howell SJ, Sear JW, Foëx P. Hypertension, hypertensive heart disease and perioperative cardiac risk. Br J Anaesth. 2004;92:570–83. doi: 10.1093/bja/aeh091. [DOI] [PubMed] [Google Scholar]
- 16.James MF, Dyer RA, Rayner BL. A modern look at hypertension and anaesthesia. S Afr J Anaesth Analg. 2011;17:168–73. [Google Scholar]
- 17.Aantaa R, Kanto J, Mika S, Kallio A, Scheinin H. Dexmedetomidine, an [alpha] 2-adrenoceptor agonist, reduces anesthetic requirements for patients undergoing minor gynecologic surgery. Anesthesiology. 1990;73:230–5. doi: 10.1097/00000542-199008000-00007. [DOI] [PubMed] [Google Scholar]
- 18.Bührer M, Mappes A, Lauber R, Stanski DR, Maitre PO. Dexmedetomidine decreases thiopental dose requirement and alters distribution pharmacokinetics. Anesthesiology. 1994;80:1216–27. doi: 10.1097/00000542-199406000-00008. [DOI] [PubMed] [Google Scholar]
- 19.Thornton C, Lucas MA, Newton DE, Doré CJ, Jones RM. Effects of dexmedetomidine on isoflurane requirements in healthy volunteers 2: Auditory and somatosensory evoked responses. Br J Anaesth. 1999;83:381–6. doi: 10.1093/bja/83.3.381. [DOI] [PubMed] [Google Scholar]
- 20.Aantaa R, Jaakola ML, Kallio A, Kanto J. Reduction of the minimum alveolar concentration of isoflurane by dexmedetomidine. Anesthesiology. 1997;86:1055–60. doi: 10.1097/00000542-199705000-00008. [DOI] [PubMed] [Google Scholar]
- 21.Aho M, Lehtinen AM, Erkola O, Kallio A, Korttila K. The effect of intravenously administered dexmedetomidine on peri-operative hemodynamics and isoflurane requirements in patients undergoing abdominal hysterectomy. Anesthesiology. 1991;74:997–1002. doi: 10.1097/00000542-199106000-00005. [DOI] [PubMed] [Google Scholar]
- 22.Blaudszun G, Lysakowski C, Elia N, Tramèr MR. Effect of perioperative systemic α2 agonists on postoperative morphine consumption and pain intensity: Systematic review and meta-analysis of randomized controlled trials. Anesthesiology. 2012;116:1312–22. doi: 10.1097/ALN.0b013e31825681cb. [DOI] [PubMed] [Google Scholar]
- 23.Khalil MA, Azeem MS. The impact of dexmedetomidine infusion in sparing morphine consumption in off-pump coronary artery bypass grafting. Semin Cardiothorac Vasc Anesth. 2013;17:166–71. doi: 10.1177/1089253212463969. [DOI] [PubMed] [Google Scholar]
- 24.Ohtani N, Yasui Y, Watanabe D, Kitamura M, Shoji K, Masaki E. Perioperative infusion of dexmedetomidine at a high dose reduces postoperative analgesic requirements: A randomized control trial. J Anesth. 2011;25:872–8. doi: 10.1007/s00540-011-1239-8. [DOI] [PubMed] [Google Scholar]
- 25.Turgut N, Turkmen A, Gökkaya S, Altan A, Hatiboglu MA. Dexmedetomidine-based versus fentanyl-based total intravenous anesthesia for lumbar laminectomy. Minerva Anestesiol. 2008;74:469–74. [PubMed] [Google Scholar]