Abstract
Behaviors associated with sickness (food consumption, weight maintenance, exploratory activity and grooming frequency) were examined on post-surgical days 1, 3, 5, 7 and 9 in male rats treated with progesterone (4 mg/kg) and/or vehicle. Rats with medial frontal cortex contusions showed reduced food consumption on days 1 and 3 (p<0.01), reduced weight maintenance on days 1, 3, 5, 7 and 9 (p<0.01), reduced grooming frequency on day 1 (p < .01), and reduced exploratory activity on day 1 (p<0.01), after injury compared to sham rats. Contusion induced behaviors were not attenuated with five days of progesterone treatment (p>0.05). Progesterone did reduce lesion size at 9 days after injury (p < 0.05). Our results suggest sickness behaviors occur after traumatic brain injury and that they might not respond to some neurosteroidal agents.
Keywords: Sickness behavior, Traumatic brain injury, Progesterone, Weight maintenance, Food consumption, Grooming, Exploratory activity, Lesion size
1. Introduction
Infection-related behaviors (i.e. sickness behaviors) include reduced eating, drinking, grooming and locomotion (as reviewed by [14]). The expression of sickness behaviors might reflect changes in motivational state, which could actually help recovery from illness [19]. For example, decreasing metabolically expensive activities (e.g., food foraging, grooming) would allow more energy to be used for fighting the infection.
Sickness behaviors have been observed after brain injury in humans. For example, it has been reported that traumatic brain injury (TBI) patients show rapid weight loss [6]. This is important since the expression of sickness behaviors (e.g., decreased food consumption) could potentiate the increased nutritional needs after TBI [7]. Caloric consumption approximately doubles in steroid-treated TBI patients compared to normal values [6]. Additionally, motivational deficits in TBI patients are considered to be a major problem following the injury [1]. Motivational deficits could impede successful rehabilitation and could be related to the expression of trauma-induced psychosomatic illnesses. Furthermore, research suggests that the effects of experimental brain injury are more widespread than just in the brain. For example, free radical production increases in the brain as well as in the kidney, lung and intestine in males rats 24 hours following TBI [33].
Sickness-like behaviors have also been observed after experimental brain injury. As compared to shams, male rats given medial frontal cortex contusions had a lower mean percentage of their preinjury weight through 18 days post-surgery [17]. Additionally, analysis of activity measures at 1, 3, 5 and 14 days after injury revealed that rats with contusions spent less time locomoting in an open activity box as compared to shams [34]. Additionally, animals with frontal cortex contusions might groom less compared to controls (unpublished observations).
In summary, research studies suggest that infection-related behaviors (i.e., sickness behaviors), such as decreased locomotion, eating and grooming, are expressed after traumatic brain injury (TBI) and that these behaviors could impede functional recovery. Reducing the expression of these behaviors by administering an immune suppressant could facilitate recovery from TBI. One such pharmacological intervention could be progesterone. Progesterone is considered an immune suppressant and reduces edema and the inflammatory response after TBI [13, 15, 23, 28, 29, 36].
Research on the effects of progesterone on recovery has also shown that the neurosteroid reduces behavioral deficits and neuronal degeneration. In addition to its regulatory effects on edema, progesterone attenuates free radical damage [31], reduces neurological abnormalities following ischemia [5, 19, 22] and spinal cord injury [35]. Neuronal cell death in the medial dorsal nucleus (MDN) of the thalamus, which has afferent and efferent connections with the medial frontal cortex (MFC), is reduced following progesterone treatment [29]. Progesterone also reduces neuronal cell death in ventral medial nucleus of the thalamus, striatum and globus pallidus following MFC cortical contusions [32] as well as the caudate nucleus and hippocampus following ischemic injury [4]. Additionally, progesterone promotes behavioral recovery in a spatial navigation task (e.g., Morris water maze) [29] an avoidance learning task [2], and sensory neglect [32] after bilateral MFC injuries.
Since infection-related behaviors are expressed after TBI we predicted that frontal cortical contusions would induce these sickness behaviors. We also predicted that progesterone, a neuroprotective and immunosuppressant steroid, would decrease the frequency of sickness behaviors (i.e., increase food consumption, weight maintenance, exploratory locomotor activity and grooming frequency).
Materials and Methods
Subjects
Twenty-four male Sprague-Dawley rats, approximately 90 days of age, were used in this experiment. Half the animals received sham surgeries and the remaining half received bilateral frontal contusions. Rats from both groups were randomly assigned to progesterone or control (vehicle) treatment groups. They were housed individually in hanging wire cages and maintained on a 12-hr dark/light reversed light cycle. Food and water were provided ad libitum. Animals were weighed daily throughout the entire study. Rats were tamed by daily handling one week before surgery. Procedures using animals were approved by the Emory University Institutional Animal Care and Use Committee (Protocol # 098-2001).
Surgical Procedures
Rats were anesthetized with sodium pentobarbital (Nembutal, 50 mg/kg, intraperitoneal). Aseptic stereotaxic and surgical procedures were used. A mid-line incision was made, the scalp retracted, and a bilateral 6-mm craniotomy was drilled and centered at 3.0 mm anterior to bregma. The impactor tip was placed over the medial frontal cortex at 3.0 mm A/P and 0.0 mm M/L. These coordinates correspond to the medial frontal cortex described by Paxinos and Watson [24]. A pneumatically driven contusion device was used to produce the cortical contusion injury [17]. The brain was impacted for approximately 0.5 seconds with a 5 mm stainless steel tip with a force of 20 psi, a velocity of 2.25 m/s, and to a depth of 2 mm. After impact, bleeding was controlled. The fascia was sutured closed and the scalp was then closed using surgical staples. A homeothermic blanket system (Harvard Apparatus, Holliston, MA) was used to maintain body temperature during surgery at approximately 37°C. Following surgery, the animals were allowed to recover from the anesthetic in a warm, dry cage until they were fully awake. Sham surgeries were a control for anesthesia and stress. The sham surgeries followed the same procedure as contusion surgeries, except that the sham rats did not receive a craniotomy and contusion. Sham surgeries did not include a craniotomy since previous studies have not found an affect of craniotomy on traumatic injury or progesterone treatment in our injury model [11, 16].
Injections
Depending on their survival time, rats received injections (intraperitoneal (i.p.) for the first and subcutaneously (s.c.) for the remaining six) of either progesterone and peanut oil (4 mg/kg) or oil at 1, 6, 24 hours, 2, 3, 4 and 5 days after the surgery. This vehicle, dose, and treatment schedule were chosen because they have been shown to reduce behavioral deficits and anterograde/retrograde degeneration caused by medial frontal contusion injuries in male rats [29]. A different vehicle [15], progesterone dose [11] and shorter treatment regimen [32], reduce or eliminate progesterone’s neuroprotective effects in this cortical contusion injury model. Control (vehicle) and progesterone treatments were prepared by a member of the laboratory and labeled “A” or “B.” The experimenters (the author and a laboratory technician) were blinded to which treatment (progesterone or control) corresponded to which label (A or B). Assignment of rats to A or B treatment groups was determined after surgery by a coin toss.
Histology
The animals were euthanized at the completion of behavioral testing by a lethal dose of Nembutal and perfused intracardially with 100 ml 0.9% 0.1M Phosphate Buffered Saline, pH 7.4 followed by 500 ml of 4% Paraformaldehyde solution (200ml 0.2M Phosphate Buffer, 100 ml 20% paraformaldehyde, 200 ml dH2O, pH 7.4). The brains were extracted and placed in a series of sucrose solutions (10%, 20% and 30%). The forebrains were sliced frozen on a sliding microtome into 40 μm coronal sections. One series was collected and mounted on slides, stained for thionin and then used for lesion analysis.
Lesion Analysis
As described elsewhere [12], the mean percent area of original tissue in frontal cortex within the impact area was estimated to verify that the contusion injury resulted in more tissue loss in the frontal cortex compared to shams. Three evenly spaced sections (+4.20, +3.20, +2.20 A/P relative to bregma) through the epicenter of the contusion were used in this analysis. Digital images (1x) of the cortical tissue were imported into, and area measured with, Image Pro Plus v4.0 Program (developed at the University of Health Science Center at San Antonio). The area of remaining tissue and the area of estimated original tissue were measured and averaged for the brains of rats given contusions. The area of tissue was measured and averaged in shams. The effect of contusion injury on the amount of remaining tissue was measured as the mean percent area of original tissue. The experimenter was blind to the treatment conditions of the animals.
Behavioral Testing
Since rats are nocturnal, behavioral testing was conducted under red light in the dark phase of the (12:12) light cycle. White noise was used to reduce the effects of sudden background noise from outside of the testing room on the rats’ behavior and equipment was cleaned with 70% EtOH after each use. Testing proceeded in the following order: exploratory locomotor activity and grooming. The tests were conducted 30 minutes apart, during which interval animals were returned to their home cages. Food consumption and weight maintenance were assessed following the grooming test. An exception to this routine was that during the first 5 days, weight maintenance was assessed earlier when the rats were injected with progesterone or vehicle. The testing order of the rats was alternated on each testing day. Behavioral measures taken before surgery were averaged to indicate rats’ pre-surgical performance on those measures. Observer bias was controlled by maintaining awareness of it and by limiting information given to any observers regarding lesion and treatment conditions of the animals. The observer was blind to the treatment conditions of the animals and did not attempt to recall the lesion conditions (contusion or sham) of the animals. Another observer assessed grooming frequency and was blind to the lesion and treatment conditions of the animals.
Weight Maintenance
Body weight was assessed before surgery (the day before surgery) and then daily for 9 days after surgery. Weight maintenance was measured as the daily mean percent of pre-surgical weight.
Food Consumption
Food consumption was assessed before surgery (the three days before surgery) and then daily for 9 days after surgery. The amount of food remaining in the food container was measured. Additionally, the amount of food that had fallen through the wire cage into a plexiglass tray placed underneath the cage was air-dried for one day and then measured. Food consumption was measured as the total grams of food consumed for each day (i.e., the difference in the weight of the remaining food from the food container and food from the tray compared to the weight of the food in the food container from the previous day).
Locomotor Activity
Exploratory locomotor activity was assessed two days before surgery and following surgery on days 1, 3, 5, 7 and 9. Activity was monitored while the animals were in an Omnitech activity box (40 cm × 40 cm) for 10 minutes on each day and was measured as the total amount of time spent moving.
Grooming Behavior
Grooming behavior (frequency) was assessed two days before and following surgery on days 1, 3, 5, 7 and 9. Rats were placed in a plexiglass box (40 cm × 40 cm) for 65 minutes. The number of grooming instances was observed from previously recorded videotapes when the rats were in a plexiglass box (40 cm × 40 cm) for the last 50 minutes. The first 15 minutes were not included in this analysis since any instances of grooming behavior during this time period could be in response to the novel environment [10]. Grooming frequency was reported as the number of grooming instances.
Data Analysis
All statistical tests were conducted using SPSS (version 10.0 for PC) and a p value of .05, unless otherwise noted. Most of the behavioral data were analyzed with a 2 × 2 × 6 (Lesion × Treatment × Days) mixed analysis of variance (ANOVA), where days served as a repeated measure. Alpha (α) was reduced to .025 when the homogeneity of variance assumption was not met and the Huyn-Feldt corrected df value was used when the sphericity assumption was not met ([20]; SPSS v.10.0). Significant effects were followed up with simple effects analyses. Power and effect sizes (Cohen’s f ) were calculated to determine the probability of having a type II error and to determine the magnitude of the treatment effect, respectively.
The estimated percent of remaining tissue was analyzed with a 2 × 2 (Lesion × Treatment) ANOVA. Significant effects were followed up with simple effects analyses.
Pearson’s correlation was used to determine if there was a linear relationship between sickness and mean percent area of tissue in rats with contusions. Spearman’s correlation was used if visual inspection of scatterplots indicated heteroscedasticity. Type I error associated with multiple comparisons was reduced by correcting alpha. Twenty comparisons were made between lesion and behavioral measures. Lesion size on day 9 was compared to weight maintenance, food consumption, exploratory locomotor activity and grooming. Using the Bonferroni correction, alpha would be reduced to .0025. This correction may be considered too stringent, increasing type II error, so alpha was reduced to .01.
Results
Weight Maintenance
The effects of lesion and treatment on weight maintenance were analyzed with a 2 × 2 × 5 (Lesion × Treatment × Days) mixed ANOVA, where days served as a repeated measure. The results showed that post-surgical weight maintenance across days was independent of progesterone treatment because there was no significant Lesion × Treatment × Days interaction, F(4,80) = 0.99, p > .05. The statistical power (1-β) was < 0.40, indicating that there was less than a 40% chance of detecting a significant effect if one actually existed. The small effect size (f = 0.05) indicates that there was only a very small difference in weight maintenance between rats given contusion or sham surgeries and those treated with progesterone or vehicle.
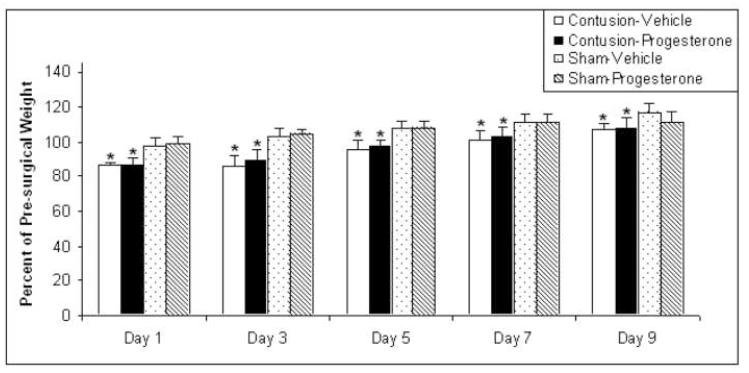
There was a significant Lesion × Days interaction, F(4,80) = 14.72, p < .001. Simple effects analysis of the effect of lesion on weight maintenance across days was performed to follow up this significant two-way interaction. As displayed in Figure 1, the analysis showed that rats with lesions had lower weight maintenance on days 1, 3, 5, 7 and 9 compared to shams (F(1,22) = 55.74, p < .001; F(1,22) = 58.39, p < .001; F(1,22) = 40.83, p < .001; F(1,22) = 21.33, p < .001; F(1,22) = 15.38, p < .01, respectively).
Figure 1. Effect of lesion on weight maintenance.
Percent pre-surgical weight was analyzed using a 2 × 2 × 5 (lesion × treatment × days) mixed ANOVA, with days as a repeated measure. A significant lesion × days interaction showed that rats with contusions had lower weight maintenance compared to shams after injury, F(4,80) = 14.72; p < 0.001. Simple effects analysis showed that rats with lesions had lower weight maintenance on days 1, 3, 5, 7 and 9 compared to sham injury, p < 0.01. (*indicates p < .01; Standard error of the mean (S.E.M.) indicated by vertical error bars.)
Food Consumption
The effects of lesion and treatment on food consumption were analyzed with a 2 × 2 × 6 (Lesion × Treatment × Days) mixed ANOVA, where days served as a repeated measure. The results showed that post-surgical food consumption across days was independent of progesterone treatment as there was no significant Lesion × Treatment × Days interaction, F(2.3,46) = 0.11, p > .05. The statistical power (1-β) was < 0.40 indicating that there was less than a 40% chance of detecting a significant effect if one actually existed. The small effect size (f = 0.07) indicates that there was only a very small difference in food consumption between rats given contusion or sham surgeries and treated with progesterone or vehicle.
There was a significant Lesion × Days interaction, F(2.3,46) = 6.73, p < .01. Simple effects analysis of the effect of lesion on food consumption across days was performed to follow up this significant two-way interaction. As displayed in Figure 2, the analysis showed that rats with lesions ate less food on days 1 and 3 compared to shams (F(1,22) = 66.47, p < .001; F(1,22) = 13.25, p < .01, respectively), but not before surgery or on days 5, 7 and 9 (F(1,22) = 3.16, p > .025; F(1,22) = 0.79, p > .05; F(1,22) = 0.66, p > .05; F(1,22) = 0.04, p > .05, respectively).
Figure 2. Effect of lesion on food consumption.
Food consumption was analyzed using a 2 × 2 × 6 (lesion × treatment × days) mixed ANOVA, with days as a repeated measure. There were no pre-surgery differences in food consumption. A significant lesion × days interaction showed that rats with contusions had lower food consumption compared to shams after injury, F(2.3, 46) = 6.73; p < 0.01. Simple effects analysis showed that rats with lesions consumed less food on days 1 and 3 compared to sham injury, p < 0.01. (*indicates p < .01; S.E.M. indicated by vertical error bars.)
Exploratory Locomotor Activity
The effects of lesion and treatment on exploratory locomotor activity were analyzed with a 2 × 2 × 6 (Lesion × Treatment × Days) mixed ANOVA, where days served as a repeated measure. The results showed that post-surgical exploratory activity across days was independent of progesterone treatment as there was no significant Lesion × Treatment × Days interaction, F(5,100) = 0.75, p > .05. The statistical power (1-β) was < 0.43, indicating that there was less than a 43% chance of detecting a significant effect if one actually existed. The small to medium effect size (f = 0.20) indicates that there was a moderate difference in exploratory behavior between rats given contusion or sham surgeries and treated with progesterone or vehicle.
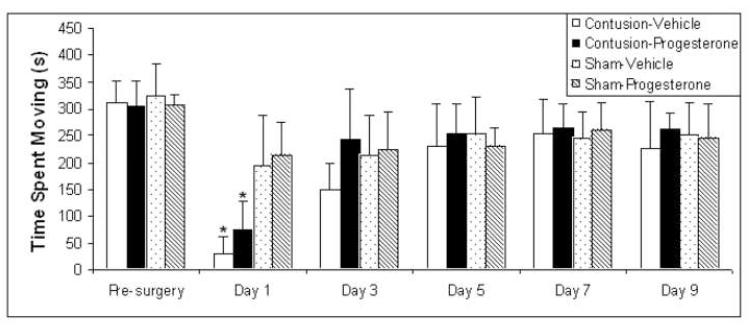
There was a significant Lesion × Days interaction, F(5,100) = 11.41, p <.01. Simple effects analysis of the effect of lesion on exploratory locomotor activity across days was performed to follow up this significant two-way interaction. As displayed in Figure 3, the analysis showed that rats with lesions spent less time moving on day 1 compared to shams (F(1,22) = 35.42, p < .001), but not before surgery or on days 3, 5, 7 and 9 (F(1,22) = 0.11, p > .05; F(1,22) =0.48, p > .05; F(1,22) = 0.00, p > .05; F(1,22) = 0.13, p > .05; F(1,22) = 0.02, p > .05, respectively).
Figure 3. Effect of lesion on exploratory locomotor activity.
Exploratory locomotor activity was analyzed using a 2 × 2 × 6 (lesion × treatment × days) mixed ANOVA, with days as a repeated measure. There were no pre-surgery differences in exploratory locomotor activity. A significant lesion × days interaction showed that rats with contusions had lower locomotor activity compared to shams after injury, F(5, 100) = 11.408; p < 0.001. Simple effects analysis showed that rats with lesions had lower locomotor activity on day 1 compared to sham injury, p < 0.001. (*indicates p < .001; S.E.M. indicated by vertical error bars.)
Grooming Behavior
The effects of lesion and treatment on grooming were analyzed with a 2 × 2 × 6 (Lesion × Treatment × Days) mixed ANOVA, where days served as a repeated measure. The results showed that post-surgical grooming across days was independent of progesterone treatment as there was no significant Lesion × Treatment × Days interaction, F(5,100) = 2.02, p > .05. The statistical power (1-β) was < 0.43, indicating that there was less than a 43% chance of detecting a significant effect if one actually existed. The medium effect size (f = 0.25) indicates that there was a moderate difference in grooming behavior between rats given contusion or sham surgeries and treated with progesterone or vehicle.
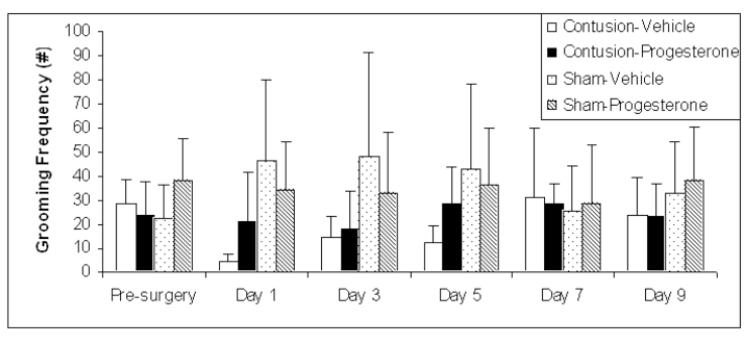
There was a significant Lesion × Days interaction, F(5,100) = 2.66, p < .05. Simple effects analysis of the effect of lesion on grooming across days was performed to follow up this significant two-way interaction. As displayed in Figure 4, the analysis showed that rats with lesions groomed less on day 1 compared to shams (F(1,22) = 8.97, p < .01), but not before surgery or on days 3, 5, 7 and 9 (F(1,22) = 0.54, p > .05; F(1,22) =5.12, p > .025; F(1,22) = 3.45, p > .05; F(1,22) = 0.12, p > .05; F(1,22) = 2.82, p > .05, respectively).
Figure 4. Effect of lesion on grooming frequency.
Grooming frequency was analyzed using a 2 × 2 × 6 (lesion × treatment × days) mixed ANOVA, with days as a repeated measure. There were no pre-surgery differences in grooming frequency. A significant lesion × days interaction showed that rats with contusions had lower grooming frequency compared to shams after injury, F(5,100) = 2.66; p < 0.05. Simple effects analysis showed that rats with lesions groomed less on day 1 compared to sham injury, p < 0.01. (* indicates p < .01; S.E.M. indicated by vertical error bars.)
Lesion Analysis
Figure 5 displays representative coronal sections of the damaged frontal cortex through the epicenter of the impact. Alpha was equal to .01. The effects of treatment on mean percent area of original tissue was analyzed with a 2 × 2 (Lesion × Treatment) ANOVA. The significant lesion effect indicated that rats with contusions had a lower mean percent area of original tissue compared to shams after injury, F(1,20) = 298.52; p < 0.001. The significant treatment effect and the significant Lesion × Treatment interaction indicated that progesterone treatment affected the mean percent area of original tissue, F(1,20) = 6.459; p < 0.025 and F(1,20) = 6.459; p < 0.025, respectively. Simple effects analysis of the effect of treatment on mean percent area of original tissue in rats with contusions was performed to follow up this significant two-way interaction. As displayed in Figure 6, the analysis showed that lesion size was reduced in rats treated with progesterone compared to rats treated with oil, F(1,11) = 6.459; p < 0.05. Pearson’s and Spearman’s Correlations showed that lesion size at day 9 after injury was not related to sickness behavior (see Table 1).
Figure 5.
Representative coronal sections of the damaged frontal cortex through the epicenter of the impact at 9 days after injury.
Figure 6. Effects of lesion and treatment on the mean percent area of original tissue.
*A 2 × 2 (lesion × treatment) ANOVA showed that rats with contusions had a smaller estimated mean % area of original tissue between sections +4.20 A/P and +2.20 A/P than shams, F(1, 20) = 6.459, p < 0.025. †Simple effects analysis showed that progesterone treatment resulted in a larger % of original tissue relative to vehicle treatment, F(1, 11) =6.459, p < 0.05. (S.E.M. indicated by vertical bars.)
Table 1.
Correlations of Mean Percent Area of Original Tissue and Behavioral Measures
| Mean Percent Area of Tissue | |||||
|---|---|---|---|---|---|
| Behavioral Measures | Day1 | Day3 | Day5 | Day7 | Day9 |
| Body Weight | 0.19 | 0.20b | 0.04 | 0.15b | −0.02 |
| Food Consumption | −0.18 | −0.13b | 0.07 | 0.07 | −0.16 |
| Exploratory Locomotor Activity | 0.36 | 0.42 | 0.08b | 0.45b | 0.03b |
| Grooming | 0.23 | 0.22 | 0.27 | 0.15b | −0.15b |
Note. Data represent Pearson’s Correlation Coefficients, unless indicated otherwise.
Spearman’s Correlation Coefficient.
Discussion
The objective of this experiment was to determine whether sickness behaviors occurred after TBI and to examine the effects of progesterone on these behaviors. Our findings suggest that: (1) medial frontal cortex contusions produce acute changes in behaviors consistent with sickness behaviors; (2) sustained post-injury weight loss is not due to reduced food intake; (3) sickness behaviors might not respond to exogenous progesterone (4 mg/kg) in male rats; (4) progesterone reduces lesion size; (5) lesion size reduction is not related to sickness behaviors.
MFC contusions result in short-term reductions in food consumption, exploratory locomotor activity and grooming and in a relatively more persistent reduction in weight maintenance. These behavioral changes are consistent with sickness behaviors observed during infection and following the injection of cytokines (reviewed by [14]).
Grooming and exploratory locomotor activity are reduced on day 1, food consumption is reduced on days 1 and 3, and weight maintenance is reduced on days 1, 3, 5 and 9 after injury. The change in weight maintenance following MFC contusions is consistent with previous research [17]. The change in exploratory locomotor activity differs from a previously discussed study where rats with contusions decreased their exploratory locomotor activity at 1, 3 and 5 days after injury as compared to shams [34]. It is possible that the lesion size in the Smith et al. [34] study was larger compared to the lesion size in the present study, creating a longer lasting change in exploratory locomotor activity.
The decrease in weight maintenance persisted following resumption of food consumption in rats with contusions compared to sham operates, suggesting that the sustained post-injury weight loss is not due to lowered food intake. This pattern, where body weight decreases despite increased food intake, is similar to what is observed in mice deficient in orexin, a neuropeptide involved in feeding behavior, and has been attributed to hypermetabolism (personal communication reported in [18]).
The sustained weight-loss observed in the present study could be due to an increased metabolic rate. A hypermetabolic response to brain injury has been observed in both humans (as reviewed by [25]) and rats [27]. Resting oxygen consumption, an indicator of metabolic rate, was increased in rats with brain injuries compared to shams during the 7-hour post-injury period of investigation. It is not known when this hypermetabolic response to brain injury in the rat dissipates. If a hypermetabolic response to brain injury in the rat persists for a longer period of time, this could explain why rats continued to show poor weight maintenance despite the return to sham levels of food consumption.
Sickness behaviors might not respond to progesterone in the dose applied here. Progesterone-treated rats with contusions did not recover significantly faster than vehicle-treated rats with contusions compared to shams on measures of sickness behaviors. Three possible explanations are that: (1) differences between the contusion groups simply did not exist because progesterone did not facilitate recovery; or (2) for some measures of sickness behaviors, the experiment was not sensitive enough to detect differences between the groups; or (3) the dose was not sufficient to elicit an effect on these measures.
Assuming that the experiment was sensitive enough to detect differences between the experimental groups, reasons progesterone did not affect recovery to sham levels include the timing of progesterone’s effect on the inflammatory response and on behavioral recovery.
Research on how progesterone exerts its beneficial effects suggests that it might be targeting different aspects of the inflammatory response very early after the injury and then days later. Progesterone (8 mg/kg) reduces IL-1β protein levels and the expression of IL-1β and TNFα mRNA at 3 hours after injury, but not at 8 and 12 hours or at 6 days after injury [15]. Additionally, progesterone reduces edema at 3 days after injury when administration was delayed to 24, but not 48, hours after the injury [30]. This suggests that progesterone’s actions that reduce edema at 3 days occur between 24 to 48 hours after the injury. However, it is not known how delayed progesterone treatment affects edema when measured at a later time. Additionally, progesterone is more effective at reducing edema at 6 days after injury when administered for 5 days compared to 3 days [9]. These studies suggest that five days of progesterone administration more effectively ameliorates components of the pathophysiological response.
Five days of progesterone administration might also be more effective at facilitating behavioral recovery. Progesterone has been shown to facilitate recovery from sensory neglect and to improve Morris water maze performance after 5 days of treatment, but not after only 3 days [32]. Sickness behaviors examined in the present study were transiently expressed, returning to sham levels within five days after brain injury. Thus, most of the behaviors were no longer expressed during the time when progesterone treatment has been shown to affect behavior, indicating that progesterone could influence behavioral recovery at later time points by continuing to reduce components of the pathophysiological response around five days after injury.
Perhaps the only report showing an effect of progesterone on behavioral recovery before five days after injury is Chen et al. [5]. Their experiment examined the effects of progesterone treatment on neurological functional tests (rotarod, somatosensory neglect and neurologic tests) on days 1 through 7 after ischemic injury. The 4mg/kg dose of progesterone reduced sensory neglect only on the second day relative to injured rats given the vehicle. However, in their model of ischemia, the progesterone was administered intravenously (i.v.) at three different times during the first 24 hours after injury rather than i.p and s.c. as done in this study. This suggests that i.v. administration facilitates progesterone’s effectiveness at promoting behavioral recovery at an earlier time point compared to other routes of administration. However, the effectiveness of i.v. progesterone administration on behavioral recovery at earlier time points could be limited considering that i.v. progesterone treatment only influenced recovery on one behavioral measure at one time point.
Reduced weight maintenance persists beyond the five days of progesterone treatment. It is not known why progesterone did not improve weight maintenance in rats given contusions during the nine-day testing period. Part of the explanation could be related to the dose of progesterone used, the vehicle used, or the length of the post-surgical testing period.
In the present study, a progesterone dose of 4 mg/kg did not improve post-surgical weight maintenance by nine days after injury. Djebaili, Hoffman & Stein [8] also showed that progesterone (16 mg/kg in cyclodextrin) did not reduce weight loss in rats through 5 days after injury, but did reduce weight loss at 21 days after injury. In contrast, the same dose of progesterone did improve weight maintenance across five days after the injury in progesterone-treated rats relative to shams, whereas two other doses (8mg/kg and 32 mg/kg in cyclodextrin) did not [11]. The reason for the discrepancy in the effectiveness of the 16 mg/kg progesterone dose (in cyclodextrin) on weight loss between the two studies is not known, but could be related to the type of vehicle used. These studies suggest that the 4 mg/kg dose of progesterone takes longer than a more potent dose to exert its effects on post-surgical weight maintenance, although a higher dose of progesterone might be necessary to affect recovery when cyclodextrin is used as a vehicle.
Goss, Simkins, Hoffman & Stein [11] showed that a higher dose of progesterone is necessary to promote behavioral recovery on the Morris water maze when cyclodextrin is used as the treatment vehicle. Additionally, He et al. [16] did not find a beneficial effect of progesterone (4 mg/kg in cyclodextrin) on the Morris water maze and have attributed this to the vehicle used. Progesterone forms a complex with cyclodextrin, reducing its availability [3]. Therefore, a higher dose of progesterone might be required to affect behavioral recovery when administered using cyclodextrin as the vehicle. In the present study, peanut oil was used as the treatment vehicle. The potency of the 4 mg/kg progesterone dose in oil on behavioral recovery compared to the progesterone doses in cyclodextrin is not known.
The length of the post-surgical testing period could also explain why progesterone did not alter weight maintenance in the present study. Considering that no spontaneous recovery of weight maintenance occurred during the nine-day testing period, perhaps any effects of progesterone on post-surgical weight maintenance would have been observed in the present study if the testing period had been extended.
Our findings support previous research showing that progesterone treatment reduces lesion size after TBI [32] and after ischemia [5, 22]. In the present study, progesterone treatment increased the estimated mean percent of original tissue in rats with contusions relative to rats with contusions and treated only with the vehicle. An increase in the percent of original tissue could indicate a reduction of lesion size and an increase in neuronal survival.
Although progesterone was effective at reducing lesion size and perhaps increasing neuronal survival, this did not influence the expression of sickness behaviors. Two possible reasons why the increase in remaining tissue did not affect sickness behaviors are the timing of the effects of progesterone treatment and the relationship between behavioral changes and lesion size at nine days after injury.
Progesterone might not affect lesion size during the three days after injury when most of the significant behavioral changes in the present study occurred. This idea is supported by Shear et al. [32], who showed that progesterone did not significantly affect lesion size in rats treated with progesterone for only three days. Kumon et al. [22] found similar results in a cerebral ischemia model. Seven days of progesterone treatment (8 mg/kg) reduced lesion volume at seven days post-ischemia, relative to controls. The two-day progesterone treatment schedule did not reduce lesion volume at two days post-ischemia, relative to controls. However, Chen et al. [5] showed that a shorter progesterone treatment schedule (8.0 mg/kg at 2 h, 6 h 24 h) administered intravenously (i.v.) reduced lesion volume at seven days post-ischemia, suggesting that i.v. administration facilitates progesterone’s effectiveness of reducing lesion size. These studies suggest that the progesterone treatment did not affect lesion volume early after the injury or that the effects are not apparent until a longer period of time after the injury.
The extent of the lesion at nine days after the injury could be unrelated to behavioral changes that occur within three days after the injury. In the present study, behavioral measures did not correlate with the mean percent of remaining tissue at nine days after injury. Shear et al. [32] also did not find a significant correlation between lesion size at 21 days after injury and sensory neglect. However, they did find a significant correlation between lesion size and performance on the Morris water maze within the second week after surgery. This suggests that the mean percent of original tissue at a later time point might not be related to the magnitude of behavioral recovery at earlier time points.
Overall, these results indicate that medial frontal cortex contusions produce acute changes in behaviors consistent with sickness behaviors and that progesterone’s influence on functional recovery could vary depending on the dose and time after injury when behavioral or anatomical changes are measured. They also emphasize that progesterone’s effect on recovery might become apparent after 5 days of treatment, when a dose of 4 mg/kg is used.
Research Highlights.
Contusion injury to the medial frontal cortex produces acute changes in behaviors consistent with sickness behaviors (e.g., grooming, food intake, weight maintenance and exploratory locomotor activity)
Sickness behaviors might not respond to exogenous progesterone (4 mg/kg) in male rats.
Progesterone’s influence on functional recovery could vary depending on the dose and time after injury when behavioral or anatomical changes are measured.
Acknowledgements
Aamir Dam for his assistance with behavioral testing preparations. Research supported by General Cologne Reinsurance Corporation and NIH R01 NS-38664.
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
References
- [1].Al-Adawi S, Powell JH, Greenwood RJ. Motivational deficits after brain injury: a neuropsychological approach using new assessment techniques. Neuropsychology. 1998;12(1):115–124. doi: 10.1037//0894-4105.12.1.115. [DOI] [PubMed] [Google Scholar]
- [2].Asbury ET, Fritts ME, Horton JE, Isaac WL. Progesterone facilitates the acquisition of avoidance learning and protects against subcortical neuronal death following prefrontal cortex ablation in the rat. Behav Brain Res. 1998;97(1-2):99–106. doi: 10.1016/s0166-4328(98)00031-x. [DOI] [PubMed] [Google Scholar]
- [3].Cavalli R, Peira E, Caputo O, Gasco MR. Solid lipid nanoparticles as carriers of hydrocortisone and progesterone complexes with beta-cyclodextrins. Int J Pharm. 1999;182:59–69. doi: 10.1016/s0378-5173(99)00066-6. [DOI] [PubMed] [Google Scholar]
- [4].Cervantes M, González-Vidal MD, Ruelas R, Escobar A, Moralí G. Neuroprotective effects of progesterone on damage elicited by acute global cerebral ischemia in neurons of the caudate nucleus. Arch Med Res. 2002;33:6–14. doi: 10.1016/s0188-4409(01)00347-2. [DOI] [PubMed] [Google Scholar]
- [5].Chen J, Chopp M, Li Y. Neuroprotective effects of progesterone after transient middle cerebral artery occlusion in rat. J Neurol Sci. 1999;171(1):24–30. doi: 10.1016/s0022-510x(99)00247-6. [DOI] [PubMed] [Google Scholar]
- [6].Clifton GL, Robertson CS, Grossman RG, Hodge S, Foltz R, Garza C. The metabolic response to severe head injury. J Neurosurg. 1984;60:687–696. doi: 10.3171/jns.1984.60.4.0687. [DOI] [PubMed] [Google Scholar]
- [7].Dickerson JWT. Recovery of function: Nutritional factors. In: Rose FD, Johnson DA, editors. Recovery from Brain Damage. Plenum Press; New York: 1992. pp. 23–33. [DOI] [PubMed] [Google Scholar]
- [8].Djebaili M, Hoffman SW, Stein DG. Allopregnanolone and progesterone decrease cell death and cognitive deficits after a contusion of the rat pre-frontal cortex. Neuroscience. 2004;123(2):349–59. doi: 10.1016/j.neuroscience.2003.09.023. [DOI] [PubMed] [Google Scholar]
- [9].Galani R, Hoffman SW, Stein DG. Effects of the duration of progesterone treatment on the resolution of cerebral edema induced by cortical contusions in rats. Restor Neurol and Neurosci. 2001;18(4):161–166. [PubMed] [Google Scholar]
- [10].Gipsen WH, Wiegant VM, Greven HM, deWied D. The induction of excessive grooming in the rat by intraventricular application of peptides derived from ACTH: Structure-activity studies. Life Sciences. 1975;17(4):645–652. doi: 10.1016/0024-3205(75)90103-4. [DOI] [PubMed] [Google Scholar]
- [11].Goss CW, Hoffman SW, Stein DG. Behavioral effects and anatomic correlates after brain injury: a progesterone dose-response study. Pharmacol Biochem Behav. 2003;76(2):231–242. doi: 10.1016/j.pbb.2003.07.003. [DOI] [PubMed] [Google Scholar]
- [12].Grossman KJ, Goss CW, Stein DG. Effects of progesterone on the inflammatory response to brain injury in the rat. Brain Res. 2004;1008(1):29–39. doi: 10.1016/j.brainres.2004.02.022. [DOI] [PubMed] [Google Scholar]
- [13].Guo Q, Sayeed I, Baronne LM, Hoffman SW, Guennoun R, Stein DG. Progesterone administration modulates AQP4 expression and edema after traumatic brain injury in male rats. Exp Neurol. 2006;198:469–478. doi: 10.1016/j.expneurol.2005.12.013. [DOI] [PubMed] [Google Scholar]
- [14].Hart BL. Biological basis of the behavior of sick animals. Neurosci Biobehav Rev. 1988;12:123–137. doi: 10.1016/s0149-7634(88)80004-6. [DOI] [PubMed] [Google Scholar]
- [15].He J, Evans CO, Hoffman SW, Oyesiku NM, Stein DG. Progesterone and allopregnanolone reduce inflammatory cytokines after traumatic brain injury. Exp Neurol. 2004a;189:404–412. doi: 10.1016/j.expneurol.2004.06.008. [DOI] [PubMed] [Google Scholar]
- [16].He J, Hoffman SW, Stein DG. Allopregnanolone, a progesterone metabolite, enhances behavioral recovery and decreases neuronal loss after traumatic brain injury. Restor Neurol Neurosci. 2004b;22(1):19–31. [PubMed] [Google Scholar]
- [17].Hoffman SW, Fulop Z, Stein DG. Bilateral frontal cortical contusion in rats: behavioral and anatomic consequences. J Neurotrauma. 1994;11(4):417–431. doi: 10.1089/neu.1994.11.417. [DOI] [PubMed] [Google Scholar]
- [18].Inui A. Transgenic approach to the study of body weight regulation. Pharmacol Rev. 2000;52(1):35–61. [PubMed] [Google Scholar]
- [19].Jiang N, Chopp M, Stein D, Feit H. Progesterone is neuroprotective after transient middle cerebral artery occlusion in male rats. Brain Res. 1996;735(1):101–107. doi: 10.1016/0006-8993(96)00605-1. [DOI] [PubMed] [Google Scholar]
- [20].Kelley KW, Hutchison K, French R, Bluthe RM, Parnet P, Johnson RW, Dantzer R. Central Interleukin-1 receptors as mediators of sickness. Ann NY Acad Sci. 1997;823:234–246. doi: 10.1111/j.1749-6632.1997.tb48395.x. [DOI] [PubMed] [Google Scholar]
- [21].Keppel G. Design and Analysis. A Researcher’s Handbook. 3rd ed Prentice Hall; Englewood Cliffs, NJ: 1991. [Google Scholar]
- [22].Kumon Y, Kim SC, Tompkins P, Stevens A, Sakaki S, Loftus CM. Neuroprotective effect of postischemic administration of progesterone in spontaneously hypertensive rats with focal cerebral ischemia. J Neurosurg. 2000;92(5):848–52. doi: 10.3171/jns.2000.92.5.0848. [DOI] [PubMed] [Google Scholar]
- [23].O’Connor CA, Cernak I, Johnson F, Vink R. Both estrogen and progesterone attenuate edema formation following diffuse traumatic brain injury in rats. Brain Res. 2005;1062(1-2):171–174. doi: 10.1016/j.brainres.2005.09.011. [DOI] [PubMed] [Google Scholar]
- [24].Paxinos G, Watson C. The Rat Brain. Academic Press; New York: 1982. [Google Scholar]
- [25].Pepe JL, Barba CA. The metabolic response to acute traumatic brain injury and implications for nutritional support. J Head Trauma Rehab. 1999;14(5):462–474. doi: 10.1097/00001199-199910000-00007. [DOI] [PubMed] [Google Scholar]
- [26].Pettus EH, Wright DW, Stein DG, Hoffman SW. Progesterone treatment inhibits the inflammatory agents that accompany traumatic brain injury. Brain Res. 2005;1049:112–119. doi: 10.1016/j.brainres.2005.05.004. [DOI] [PubMed] [Google Scholar]
- [27].Roe SY, Rothwell NJ. Whole body metabolic responses to brain trauma in the rat. J Neurotrauma. 1997;14(6):399–408. doi: 10.1089/neu.1997.14.399. [DOI] [PubMed] [Google Scholar]
- [28].Roof RL, Duvdevani R, Stein DG. Progesterone treatment attenuates brain edema following contusion injury in male and female rats. Restor Neurol Neurosci. 1992;4:425–427. doi: 10.3233/RNN-1992-4608. [DOI] [PubMed] [Google Scholar]
- [29].Roof RL, Duvdevani R, Braswell L, Stein DG. Progesterone facilitates cognitive recovery and reduces secondary neuronal loss caused by cortical contusion injury in male rats. Exp Neurol. 1994;129:64–69. doi: 10.1006/exnr.1994.1147. [DOI] [PubMed] [Google Scholar]
- [30].Roof RL, Duvdevani R, Heyburn JW, Stein DG. Progesterone rapidly decreases brain edema: treatment delayed up to 24 hours is still effective. Exp Neurol. 1996;138(2):246–251. doi: 10.1006/exnr.1996.0063. [DOI] [PubMed] [Google Scholar]
- [31].Roof RL, Hoffman SW, Stein DG. Progesterone protects against lipid peroxidation following traumatic brain injury in rats. Mol Chem Neuropathol. 1997;31(1):1–11. doi: 10.1007/BF02815156. [DOI] [PubMed] [Google Scholar]
- [32].Shear DA, Galani R, Hoffman SW, Stein DG. Progesterone protects against necrotic damage and behavioral abnormalities caused by traumatic brain injury. Exp Neurol. 2002;178(1):59–67. doi: 10.1006/exnr.2002.8020. [DOI] [PubMed] [Google Scholar]
- [33].Shohami E, Gati I, Beit-Yannai E, Trembovler V, Kohen R. Closed head injury in the rat induces whole body oxidative stress: overall reducing antioxidant profile. J Neurotrauma. 1999;16(5):365–376. doi: 10.1089/neu.1999.16.365. [DOI] [PubMed] [Google Scholar]
- [34].Smith JS, Fulop ZL, Levinsohn SA, Darrell RS, Stein DG. Effects of the novel NMDA receptor antagonist gacyclidine on recovery from medial frontal cortex contusion injury in rats. Neural Plast. 2000;7(1-2):73–91. doi: 10.1155/NP.2000.73. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [35].Thomas A, Nockels R, Hiu P, Sharffrey CI, Chopp M. Progesterone is neuroprotective after acute experimental spinal cord trauma in rats. Spine. 1999;24(20):2134–2138. doi: 10.1097/00007632-199910150-00013. [DOI] [PubMed] [Google Scholar]
- [36].Wright DW, Bauer ME, Hoffman SW, Stein DG. Serum progesterone levels correlate with decreased cerebral edema after traumatic brain injury in male rats. Journal of Neurotrauma. 2001;18(9):901–909. doi: 10.1089/089771501750451820. [DOI] [PubMed] [Google Scholar]