Abstract
Background and Aims Many gymnosperms produce an ovular secretion, the pollination drop, during reproduction. The drops serve as a landing site for pollen, but also contain a suite of ions and organic compounds, including proteins, that suggests diverse roles for the drop during pollination. Proteins in the drops of species of Chamaecyparis, Juniperus, Taxus, Pseudotsuga, Ephedra and Welwitschia are thought to function in the conversion of sugars, defence against pathogens, and pollen growth and development. To better understand gymnosperm pollination biology, the pollination drop proteomes of pollination drops from two species of Cephalotaxus have been characterized and an ovular transcriptome for C. sinensis has been assembled.
Methods Mass spectrometry was used to identify proteins in the pollination drops of Cephalotaxus sinensis and C. koreana. RNA-sequencing (RNA-Seq) was employed to assemble a transcriptome and identify transcripts present in the ovules of C. sinensis at the time of pollination drop production.
Key Results About 30 proteins were detected in the pollination drops of both species. Many of these have been detected in the drops of other gymnosperms and probably function in defence, polysaccharide metabolism and pollen tube growth. Other proteins appear to be unique to Cephalotaxus, and their putative functions include starch and callose degradation, among others. Together, the proteins appear either to have been secreted into the drop or to occur there due to breakdown of ovular cells during drop production. Ovular transcripts represent a wide range of gene ontology categories, and some may be involved in drop formation, ovule development and pollen–ovule interactions.
Conclusions The proteome of Cephalotaxus pollination drops shares a number of components with those of other conifers and gnetophytes, including proteins for defence such as chitinases and for carbohydrate modification such as β-galactosidase. Proteins likely to be of intracellular origin, however, form a larger component of drops from Cephalotaxus than expected from studies of other conifers. This is consistent with the observation of nucellar breakdown during drop formation in Cephalotaxus. The transcriptome data provide a framework for understanding multiple metabolic processes that occur within the ovule and the pollination drop just before fertilization. They reveal the deep conservation of WUSCHEL expression in ovules and raise questions about whether any of the S-locus transcripts in Cephalotaxus ovules might be involved in pollen–ovule recognition.
Keywords: Cephalotaxus, proteome, transcriptome, development, gymnosperm, pollen selection
INTRODUCTION
In most extant gymnosperms, pollination relies on the wind-mediated transfer of pollen to ovulate cones. Many gymnosperm species produce an ovular secretion, the pollination drop, which extends beyond the micropyle and forms a liquid surface on which the pollen lands. Subsequently, the drop withdraws and transports pollen grains into the ovule where they germinate, form pollen tubes and ultimately release sperm that fertilize eggs (Doyle and O’Leary, 1935; Singh, 1978). The sole function of the pollination drop was once thought to be transport of pollen into the ovule. These ovular secretions, however, contain a suite of organic and inorganic compounds including sugars, amino acids, organic acids, proteins and calcium (Ziegler, 1959; Seridi-Benkaddour and Chesnoy, 1988; Carafa et al., 1992). The nature of the proteins in the drops suggests that in addition to nourishing the developing pollen, they play diverse roles in pollination (Gelbart and von Aderkas, 2002; Nepi et al., 2009; Coulter et al., 2012).
Studies of pollination drops using immunological methods detected arabinogalactans in Taxus × media (O’Leary et al., 2004), while chemical methods detected acid phosphatase in Welwitschia mirabilis (Carafa et al., 1992). Studies using proteomic methods have focused on the conifers Pseudotsuga menziesii (Poulis et al., 2005), Taxus × media (O’Leary et al., 2007), Juniperus communis, Juniperus oxycedrus and Chamaecyparis lawsoniana; and on the gnetophytes, Welwitschia mirabilis (Carafa et al., 1992; Wagner et al., 2007) and several species of Ephedra (von Aderkas et al., 2015). These studies detected thaumatin-like proteins, chitinases, invertase, galactosidase, peroxidase and subtilisin-like protease, among others (Table 1). Putative functions of the proteins include the conversion of sugars, cleavage of polysaccharides, defence against pathogens, and the expansion and growth of pollen tubes (Table 1) (Gelbart and von Aderkas, 2002; Poulis et al., 2005; Wagner et al., 2007). Chitinases in Pseudotsuga menziesii have antifungal activity (Coulter et al., 2012). Some of the proteins in Table 1 are conserved among taxa, such as chitinase and glucosidase, while others, such as galactosidase, have so far been observed in fewer species.
Table 1.
Proteins previously identified in the pollination drops of gymnosperms
| Chae. laws. | Jun. com. | Jun. oxy. | Pseu. men. | Tax. × med. | Wel. mir. | Eph. comm. | Eph. foe. | Eph. min. | Eph. tri. | Eph. lik | Eph. mon. | Eph. comp. | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| β-d-Glucan exohydrolase | x | ||||||||||||
| Glucan 1,3-β-glucosidase (or precursor) | x | x | x | x | |||||||||
| Subtilisin-like proteinase | x | ||||||||||||
| Glycosyl hydrolase | x | ||||||||||||
| Glucanase-like protein | x | ||||||||||||
| Chitinase | x | x | x | x | |||||||||
| Thaumatin-like protein | x | x | x | x | |||||||||
| Xylosidase | x | x | x | x | x | x | |||||||
| Galactosidase | x | x | x | x | x | ||||||||
| Peroxidase | x | x | x | x | x | ||||||||
| Invertase | x | ||||||||||||
| Aspartyl protease | x | x | x | x | x | ||||||||
| Serine carboxypeptidase (-like) protein | x | x | x | x | |||||||||
| Arabinogalactan protein | x | ||||||||||||
| Malate dehydrogenase | x | ||||||||||||
| Peptidase | x | ||||||||||||
| Superoxide dismutase | x |
Plant names are abbreviated as follows: Chae. laws., Chamaecyparis lawsonia; Jun. com., Juniperus communis; Jun. oxy., Juniperus oxycedrus; Pseud. menz., Pseudotsuga menziesii; Tax. × med., Taxus × media; Wel. mir., Welwitschia mirabilis; Eph. comm., Ephedra communis; Eph. foe., Ephedra foeminea; Eph. min., Ephedra minuta; Eph. tri., Ephedra trifurca; Eph. lik., Ephedra likiangensis; Eph. mon., Ephedra monosperma; Eph. comp., Ephedra compacta.
Adapted from Coulter et al. (2012) and von Aderkas et al. (2015).
Developmental studies suggest that pollen selection conceivably could occur within pollination drops. In the ovules of several species of Pinaceae, differences in pollen germination and pollen tube growth and development were observed, depending on whether the pollen was conspecific, heterospecific (McWilliam, 1959; Fernando et al., 2005) or heterogeneric (von Aderkas et al., 2012) with respect to the ovule. The underlying mechanisms controlling these differential responses of pollen and pollen tubes are unknown, however. It is possible that nutritional requirements of male gametophytes are species specific, leading some to thrive in certain pollination drops while others languish or die (Gelbart and von Aderkas, 2002). Alternatively, protein–protein interactions may occur between the pollen and the ovule, beginning with pollen recognition and culminating in the destruction or inhibition of the growth of certain pollen types. Protein–protein interactions between pollen and/or pollen tubes and ovules have been well documented in angiosperms, including those of the incompatibility reactions of S-locus proteins. Self-incompatibility (SI) systems are found in at least 100 angiosperm families (Igic et al., 2008), and comprise diverse molecular mechanisms, the most well characterized of which are the sporophytic self-incompatibilty (SSI) system found in members of the Brassicaceae, and the gametopythic self-incompatibilty (GSI) systems found in the Papaveraceae, Solanaceae, Plantaginaceae and Rosaceae (for reviews, see Takayama and Isogai, 2005; Franklin-Tong, 2008; Iwano and Takayama, 2012; Gibbs, 2014).
Proteomic studies are needed in additional gymnosperms to better understand pollination drop composition and function in conifers, cycads, Ginkgo and gnetophytes. Here, we documented the proteins present in the pollination drops of Cephalotaxus, for which no data were previously available. Cephalotaxus, or ‘plum yew’ (Fig. 1), is native to southern and eastern Asia, and includes 8–11 species (Bassett et al., 2005). It is the sole genus in the Cephalotaxaceae, although some taxonomic treatments do not recognize Cephalotaxaceae, but consider Cephalotaxus to be a divergent genus within the Taxaceae (Rai et al., 2008). Regardless, the divergence of Cephalotaxus from Taxaceae sensu stricto is ancient, having occurred about 150 Ma (Leslie et al., 2012). We focused on Cephalotaxus sinensis and Cephalotaxus koreana. We also generated RNA-sequencing (RNA-Seq) data to document the transcripts present in ovules of C. sinensis at the time of pollination drop production and to provide additional insight on ovule metabolic processes and pollination drop functions. This is the first transcriptome assembled from a Cephalotaxus ovule at the time of pollination drop production, and the first transcriptome of any gymnosperm ovule.
Fig. 1.
Cephalotaxus koreana. (A) Branch with male cones before pollen shedding. (B) Female cones with pollination drops.
MATERIALS AND METHODS
Pollination drop and ovule collection
Pollination drops and ovules were collected from species of Cephalotaxus at the Arnold Arboretum of Harvard University (Boston, MA, USA). Pollination drops were collected in April 2011 from each of C. sinensis Rehder and Wilson and C. koreana Nakai using a flame-drawn capillary tube. Drops were pooled to obtain a minimum of 100 μL per species and were stored at –20 °C until analysis.
In April 2012, cones were collected from C. sinensis at the time of pollination drop production and were frozen immediately in liquid nitrogen for RNA-Seq analyses.
Protein preparation, electrophoresis, mass spectrometry and data analysis
Sample preparation, electrophoresis and protein sequencing followed previous methods (Prior et al., 2013). In brief, 1-D SDS–PAGE was performed, and gels were stained with Coomassie blue. Bands were excised from gels, digested with trypsin, and analysed at the University of Victoria Genome BC Proteomics Center via high-performance liquid chrtomatography–tandem mass spectrometry (HPLC-MS/MS). HPLC was performed on a RP nano-analytical column Magic C-18AQ (Michrom BioResources Inc., Auburn, CA, USA). The chromatography system was coupled online with an LTQ Orbitrap Velos mass spectrometer equipped with a Nanospray II source (Thermo Fisher Scientific).
The raw data files were searched using Thermo Scientific Proteome Discoverer software version 1.2 (Thermo Fisher Scientific) with Mascot version 2.2.1 (Matrix Science, Boston, MA, USA) and PEAKS Studio v. 6 (Bioinformatics Solutions Inc., Ontario, Canada) against the UniProt-SwissProt and Uniprot-Trembl databases. Several post-translational modifications were tested for during searches. These included the fixed carbamidomethylation of cysteines when iodoacetamide was used in sample processing, oxidation of methionine and deamidation (N, Q). Due to the paucity of gymnosperm data available in these databases, de novo discovery of peptides was also performed using PEAKS Studio. SPIDER homology searches implemented in PEAKS Studio were also performed to compensate for sequencing errors and mutations which may otherwise inhibit the correct identification of peptides (Han et al., 2005). PEAKS ‘In Chorus’ was used to summarize the various search results. In both MASCOT and PEAKS, peptides were accepted as correctly identified if their scores had values of at least P < 0.01. The false discovery rate, as determined by a decoy database search, was kept below 1 %. Proteins were considered as correctly identified if they contained at least one unique peptide that fulfilled the above criteria. We did not include ‘uncharacterized protein’ hits in this report.
RNA-Seq
Ovules, including the nucellus and integuments, were dissected from cones on ice and pooled into a single sample. RNA was extracted using a CTAB–PVP–Tris–HCl extraction buffer as described previously (Chang et al., 1993). However, we excluded spermidine from our extraction buffer, used only 100 mm Tris–HCl as opposed to 1 m, and adjusted our centrifugation speed from 14 000 rpm to 10 000 rpm. Library preparation and sequencing were performed by the FAS Center for Systems Biology (Harvard University). In brief, RNA was amplified using the PrepX SPIA RNA-Seq Library Kit (Nugen), sheared, and made into 200 bp insert libraries using the PrepX ILM DNA Library Preparation kit (IntengenX, Pleasanton, CA, USA). Samples were sequenced on an Illumina HiSeq 2000.
Quality of sequences was assessed using FastQC (Andrews). Adaptors were removed using CutAdapt (Martin, 2011), and low quality sequences were trimmed using Sickle (Joshi and Fass, 2011) and Trimmomatic (Lohse et al., 2012). Trimmed sequences were assembled using Trinity (Grabherr et al., 2011) with default parameters. Similarity searches were performed using BLASTx against the NCBI non-redundant (nr) database and Uniprot [SwissProt (SP) and TReMBL (TR)] databases. Transcripts were annotated by using blast2go (Conesa et al., 2005) against the Gene Ontology (GO) database.
RESULTS
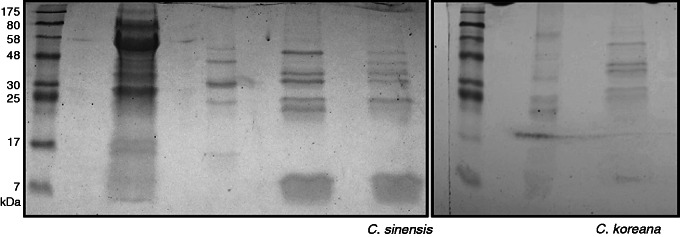
Proteome
Proteins ranged in molecular weight from approx. 7 to 50 kDa. Six SDS–PAGE gel bands from C. koreana extracts and eight bands from C. sinensis extracts were sequenced (Fig. 2). A total of 30 proteins from C. sinensis and 32 from C. koreana were identified (Table 2). Peptides can be found in Supplementary Data Table S1. Twelve proteins were detected in both samples: chitinase IV, chitinase I, peroxidase, thaumatin-like protein (TLP), pollen allergen CJP-38, α-galactosidase, β-galactosidase, α-amylase, cup a 3 protein, pathogenesis-related protein, PR5 allergen jun r/cup s, and luminal binding protein (BiP) (Table 2).
Fig. 2.
SDS–PAGE of pollination drops from C. koreana and C. sinensis and others. Lanes 1 and 6, protein marker; lanes 2–4 and 7, samples not discussed in this paper; lane 5, C. sinensis; lane 8, C. koreana.
Table 2.
Proteins identified in the pollination drops of C. koreana and, C. sinensis
| C. sinensis (outdoor) | C. koreana (outdoor) |
|---|---|
| Alpha-galactosidase | Alpha-galactosidase |
| Alpha-amylase type B isozyme | Alpha-amylase type B isozyme |
| Beta-galactosidase | Beta-galactosidase |
| Class I chitinase | Class I chitinase |
| Class IV chitinase | Class IV chitinase |
| Cup a 3 protein | Cup a 3 protien |
| Luminal-binding protein (BiP) | Luminal binding protein (BiP) |
| Pathogenesis-related protein | Pathogenesis-related protein |
| Peroxidase | Peroxidase |
| Pollen allergen CJP38 | Pollen allergen CJP38 |
| PR5 allergen Jun r | PR5 allergen Jun r |
| Thaumatin-like protein | Thaumatin-like protein |
| Alpha-1_4-glucan-protein synthase | Acidic endochitinase |
| Alpha-amylase isozyme | Acidic thaumatin-like protein |
| Ascorbate peroxidase | Allergen Ara h 1 |
| Aspartate aminotransferase | Allergen Arah3/Arah4 |
| Calmodulin | Alpha-amylase |
| Cytosolic glyceraldehyde-3-phosphate dehydrogenase | Ara h 1 allergen |
| Elongation factor 1-alpha | Arachin |
| Elongation factor | Conarachin |
| Enolase | Endo-beta-mannanase |
| Eukaryotic translation elongation factor | Glucan 1_3-beta-glucosidase |
| Glutathione-S-transferase | Glucan endo-1_3-beta-glucosidase |
| Glyceraldehyde 3-phosphate dehydrogenase | Glutathione reductase |
| Heat shock protein 70 | Gly1 |
| Histone H2A variant 1 | Glycinin |
| Isocitrate dehydrogenase [NADP] | Iso-Ara h3 |
| Monodehydroascorbate reductase | Malate dehydrogenase |
| Probable histone H2A variant 1 | Neutral ceramidase |
| Triosephosphate isomerase | Polygalacturonase |
| UDP-glucose:protein transglucosylase-like protein SlUPTG | Storage protein |
| Zinc finger protein_ putative |
Bold indicates proteins that were detected in previous studies.
The first 12 proteins are found in both samples, followed by proteins unique to each sample. Proteins in each are listed in alphabetical order.
Transcriptome
Paired-end sequencing yielded 314 781 368 paired-end reads from a 200 bp insert library, encompassing 21.8 Gb of data. After stringent quality filtering, only reads of QV > 20 were accepted (mean QV = 37). Sequences have been deposited in the NCBI SRA database under the accession number SRP058054.
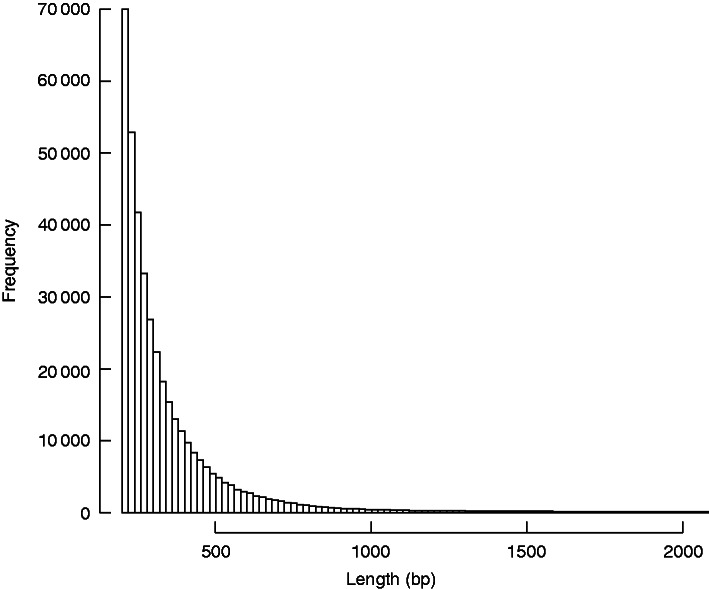
Using the Trinity de novo assembly program, 402 215 transcripts were assembled with N50 = 390, with 56 370 transcripts above 500 bp and 17 201 transcripts above 1000 bp (Fig. 3). Searches against the NCBI nr database and the combined SP and TR Uniprot databases, using BLASTx and an e-value cut-off of 10–5, yielded 49 277 transcripts with significant matches against the nr database, 27 837 against SP and 47,558 against TR (Supplementary Data Table S2). In total, 49 769 transcripts were matched to putative homologues using this approach.
Fig. 3.
Histogram of Trinity-assembled transcript sizes.
BLAST searches will not find matches for all sequences because short reads are rarely matched to known genes. For example, only 7·8 % of all transcripts <500 bp in length had blast hits against the nr database, but 36·0 % of transcripts sized ≥500 bp had hits. The paucity of gymnosperm data available in the databases further limits the total number of BLAST hits.
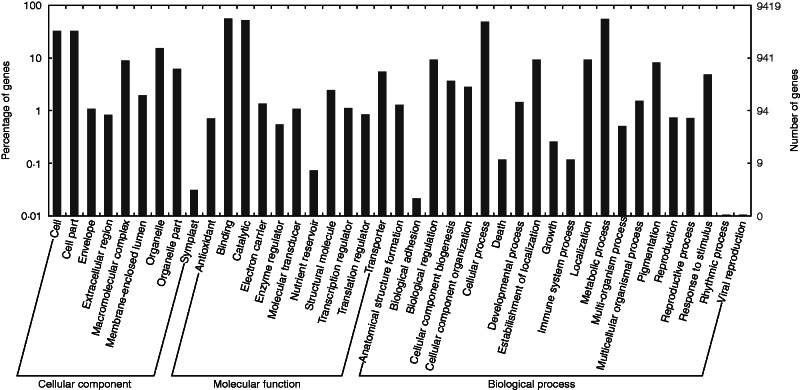
Transcripts with BLAST matches against the nr database were annotated using Gene Ontology (GO) terms to predict their possible functions. A total of 9644 transcripts were annotated (Supplemetnary Data Table S3). GO annotations classify the function of transcripts into three categories: biological processes, cellular components and molecular functions. Within these categories, the greatest numbers of transcripts were assigned to the sub-categories ‘binding’ and ‘catalytic’ (molecular processes), ‘cellular process’ and ‘metabolic process’ (biological processes), and ‘cell’ and ‘cell part’ (cellular component) (Fig. 4).
Fig. 4.
Histogram of gene ontology classifications, sub-divided into biological processes, cellular components and molecular functions. The right y-axis indicates the number of genes in a category. The left y-axis indicates the percentage of genes in a category.
DISCUSSION
Proteome of the pollination drops of C. koreana and C. sinensis
The pollination drops of C. koreana and C. sinensis harbored at least 30 proteins (Table 2). Their potential roles include defence of nutrient-rich pollination drops from microbial pathogens, promotion and support of pollen tube growth, metabolism during drop production, and response to stress. It is likely that some of these proteins are actively secreted into the pollination drop (O’Leary et al., 2007), while others may enter the drop as a result of nucellar breakdown (von Aderkas et al., 2015). As pollination drops form in Cephalotaxus (C. Pirone-Davies, unpubl. res.) and in some other gymnosperms, the most micropylar cells of the nucellus break down (O’Leary et al., 2004).
Several of the proteins we detected in Cephalotaxus are found in the pollination drops of other gymnosperms, while others are reported here for the first time (Table 2). Defensive, or pathogenesis-related proteins (PR proteins), are conserved across all species examined to date. PR proteins are classified into diverse families, including chitinases (PR-3), TLPs (PR-5) and peroxidases (PR-9) (van Loon et al., 2006). Chitinases have been detected previously in two species of Juniperus, Ephedra foeminea, E. trifurca and Welwitschia mirabilis (Wagner et al., 2007), TLPs have been detected in species of Juniperus, E. minuta and Taxus × media (O’Leary et al., 2007), peroxidase in Pseudotsuga menziesii, E. compacta, E. likiangensis and E. trifurca, and galactosidase in E. minuta, E. compacta, E. trifurca and P. menziesii (Poulis et al., 2005). Three of the six proteins shared by both species of Cephalotaxus are PR proteins, i.e. they are induced in the presence of a pathogen. The mechanisms by which the defence-related proteins function in defence are diverse. Chitin is the primary component of fungal cell walls, and thus chitinases, which break down various polymers including chitin (Grover, 2012), often function as antifungals, as has been shown in the pollination drops of Pseudotsuga menziesii (Coulter et al., 2012). TLPs may affect fungal growth via β-glucanase activity, or the hydrolysis of β-1,3-glucan in fungal cell walls (Grenier et al., 1999), and they may also exhibit α-amylase activity that hinders the digestion of plant starches and proteins in the insect gut, thus deterring insect predation (Franco et al., 2002). All of these proteins, however, have other known roles in plants. Chitinases are involved in various growth and developmental processes (Grover, 2012). TLPs accumulate in tissue in response to some environmental stresses and developmental cues (Liu et al., 2010), and some TLPs can inhibit the formation of ice crystals (Hon et al., 1995). Peroxidases are involved in auxin metabolism, lignin and suberin formation, and the linking of cell wall components (Passardi et al., 2005). Nonetheless, in Cephalotaxus, other conifers and gnetophytes, it is likely that the function of one or more of these proteins is to defend the nutrient-rich drop from pathogens (Gelbart and von Aderkas, 2002).
Cup s and Jun r are members of the PR-5 protein family and thus may play a role in defence. They have been identified as pollen allergens in Cupressus sempervirens and Juniperus rigida, respectively (Cortegano et al., 2004; Breiteneder and Mills, 2005). Other pollen allergens, such as CJP-38, share homology with β-1,3-glucanase, which degrades β-1,3-glucan, or callose. Enzymes with glucanase or glucosidase activity are also found in Chamaecyparis lawsoniana and Juniperus oxycedrus (Table 1). Functions of β-1,3-glucanase are diverse, and include the regulation of plasmodesmata (Levy et al., 2007), hydrolysis of β-1,3-glucan in fungal cell walls and pollen tube growth in angiosperms (Kotake et al., 2000; Sela-Buurlage et al., 1993). In the pollination drops of Cephalotaxus, CJP-38 may have antifungal activity, or, if it originates from nucellar cells, it may regulate plasmodesmata, and thus cell–cell communication within the ovule. It is unclear whether CJP-38 is involved in pollen tube growth in Cephalotaxus as is the case in angiosperms. Callose is present in the pollen tubes of some gymnosperms (Yatomi et al., 2002), but it is not ubiquitous, and its distribution within the tube varies depending on species and developmental stage (for a review, see Fernando et al., 2010). In contrast, the angiosperm tube wall is composed predominantly of callose, and callose septae, or plugs, are formed throughout the tube as it grows (Abercrombie et al., 2011).
α-Amylase hydrolyses starch, thus mobilizing energy for growth and development (Huang et al., 1992). Starch has been observed to accumulate in the nucellar cells of some gymnosperms just before drop formation (Carafa et al., 1992; Takaso and Owens, 1995) and, in some species, it decreases at the time of pollination drop production (Owens and Simmons, 1987).
Galactosidase enzymes are less widespread in pollination drop proteomes than defence-related proteins, but they occur in drops of Pseudotsuga mensziesii and some species of Ephedra. They are also present in various plant tissues where they metabolize a variety of polysaccharides and are involved in fruit ripening, growth and the hydrolysis of lactose. In flowering plants, galactosidases are observed in both the stigma exudate and the pollen, and are hypothesized to loosen the cell wall components of the intine and assist in pollen germination and elongation (Hruba et al., 2005; Rejon et al., 2013). It has been suggested that galactosidase and xylosidase present in the pollination drops of Pseudotsuga menziesii may also help loosen the cell walls of pollen via degradation of xyloglucan support chains of the pollen intine, thus promoting pollen tube growth (Poulis et al., 2005).
The luminal binding protein (BiP) occurs in the endoplasmic reticulum (ER) lumen, where it assists in the proper folding of proteins (Boston et al., 1996; Galili et al., 1998). Based on its typical sub-cellular location, BiP is probably present in the drop as a result of nucellar breakdown. It is conserved across both samples, suggesting that it may have an important metabolic role at the time of pollination drop production. BiP activity increases during biotic and abiotic stress responses to pathogens, nutrient deficiency, temperature changes and water stress (Alvim et al., 2001). It has been proposed that increased BiP production is needed to support an increase in the synthesis of PR proteins (Jelitto-Van Dooren et al., 1999). Numerous PR proteins are present in the pollination drop; thus, it is possible that BiP could support their synthesis.
Additional proteins were detected in either C. koreana or C. sinensis, but not in both (Table 2). Technical replicates are needed to verify that their presence is species specific (Elias et al., 2005; Ham et al., 2008). Many of these other proteins are involved in cellular metabolic processes, and may have been released from nucellar cells, as appears to be the case for similar proteins recently detected in pollination drops of seven species of Ephedra (von Aderkas et al., 2015).
The transcriptome of C. sinensis at the time of pollination drop production
The sequences we identified in the pollination drop proteome are a small sub-set of the sequences in our RNA-Seq data. This is not surprising given that one sample is from an aqueous secretion and the other from a multicellular tissue. Also, the synthesis and degradation of mRNA and protein are differently affected by various factors including numerous post-transcription regulatory processes that affect mRNA stability, the timing of protein synthesis and rates of protein turnover (for a review, see Vogel and Marcotte, 2012). The degree of overlap in the identities of transcripts and the proteins is small, with just 174 matching hits (Supplementary Data Table S4).
The transcripts detected in the ovule of C. sinensis represent 41 GO categories broadly distributed across biological processes, cellular components and molecular functions (Fig. 4). Among the more surprising results, we detected homologues of S-locus proteins, which could function in pollen–ovule interactions, and B-class MADS-box genes, which are expected in male cones based on previous studies (Sundstrom and Engstrom, 2002; Thiessen and Becker, 2004). We focus our discussion here on transcripts for proteins that potentially are involved in pollen recognition, pollination drop formation and reproductive development.
Pollen recognition
We detected several transcripts that may be involved in pollen recognition, based on BLAST results and GO annotations (Supplementary Data Table S4). Among the five proteins with ‘recognition of pollen’ in the GO annotation were one transcript that matched an S-locus lectin protein kinase and four transcripts that matched a g-type lectin S-receptor-like serine threonine-protein kinase in either the NCBI nr or the SP database. Additional transcripts had top blast hits to a g-type lectin S-receptor-like serine/threonine kinase, but did not include pollen recognition in the GO annotation (Supplementary Data Table S3). S-locus proteins determine the specificity of pollen rejection in angiosperm SI systems, but SI systems differ with respect to the proteins encoded by the S-loci (Takayama and Isogai, 2005). It is the SSI system of Brassicaceae that uses S-receptor kinase (SRK) as the female determinant of pollen rejection (Stein et al., 1991; Sherman-Broyles et al., 2007). It belongs to the diverse receptor-like kinase (RLK) family and is the best characterized member of the S-domain RLKs (SRLKs) (Shiu and Bleecker, 2003; Xing et al., 2013). SRLKs are differentiated from other RLKs by the presence of an extracellular S-domain composed of three sub-domains, B_Lectin, SLG and PAN_APPLE, one of which, SLG, is responsible for binding the male determinant during SI reactions (Kemp and Doughty, 2007). Apart from SRK in Brassica, the functions of SRLK members are largely unknown. They are present in non-reproductive tissues and are predicted to be involved in roles other than pollen recognition, including development and defence (Dwyer et al., 1994; Bassett et al., 2005). A recent transcriptomic profile for soybean under salt stress detected one SRLK that was upregulated when stressed (Ge et al., 2010). A subsequent study sought to validate the role of this SRLK but relied heavily on its overexpression in arabidopsis, leaving open the question of its function in soybean (Sun et al., 2013).
Self-incompatibility is widespread in angiosperms, and the fact that unrelated proteins are encoded by S-loci in different SI systems is consistent with independent evolution of multiple SI systems, both of the SSI and the GSI type (Gibbs, 2014). In GSI and SSI, pollen either fails to germinate on the stigma or growth of pollen tubes arrests in the style. There is genetic evidence for a third type of SI, which may be more widespread than GSI and SSI. Late-acting self-incompatibility (LSI), in the strict sense, describes self-sterility resulting from the failure of pollen tubes to penetrate the ovule or failure of egg and sperm to fuse (reviewed in Gibbs, 2014). LSI may be widespread in angiosperms, and it may be the ancestral type of SI in angiosperms (Gibbs, 2014). A similar phenomenon has been observed in Picea glauca, where both abortion of pollen tubes and failure of the tubes to release sperm were observed after self-pollination but not after cross-pollination (Runions and Owens, 1998), and in Abies alba, where archegonia broke down in self- but not in cross-pollinated ovules (Kormatuk, 1999). These pre-zygotic events are distinguished from cases in which expression of lethal genes in the zygote or embryo causes breakdown of the ovule and low seed set. Post-zygotic breakdown may in fact explain other cases in conifers in which self-pollination results in low seed set.
There are intriguing suggestions from pollination studies in conifers of signalling between pollen and ovules, which, if present, could function in LSI or other SI systems. These include the dependence of ovule development on the germination and growth of the pollen tube in species of Pinus and Tsuga (Pinaceae) (McWilliam, 1959; Dogra, 1967; Owens and Blake, 1983; Owens et al., 2005) and Thuja (Cupressaceae) (Owens et al., 1990), aberrant pollen tube growth and/or ovular breakdown in heterospecific crosses in Pinus (McWilliam, 1959) and Abies alba (Kormatuk, 1999), and the retraction of the pollination drop in the presence of conspecific, but not heterospecific pollen in Juniperus communis (Mugnaini et al., 2007). Signalling between pollen and ovule also could explain pollen selection in the pollination drop. In several cross-pollination studies, conspecific pollen germinated and grew better in the pollination drop than heterospecific or heterogeneric pollen (McWilliam, 1959; Fernando et al., 2005; von Aderkas et al., 2012). These studies, however, did not distinguish whether this resulted from nutritional needs being better met by conspecific pollination drops, or by pollen–ovule signalling, or by both. Outcrossing in Cephalotaxus is promoted by dioecy, although dioecy does not necessarily prevent crossing among individuals with nearly identical genotypes. Outcrossing would be further promoted, however, if pollen selection was also occurring in the pollination drop.
Self-incompatibility systems help promote outcrossing and are often cited as a unique feature of angiosperms that contributes to reproductive isolation and speciation (Stebbins, 1957; Jain, 1976). The presence of SI systems in gymnosperms would require a revision of this hypothesis. The observations pointing to pollen–ovule signalling are perhaps the most compelling, whereas the SRLK transcripts in the ovule might be for proteins that function in ovule development or defence. In the latter case, one might expect SRLK members in the proteome of the pollination drop. We did not detect SRLK proteins in this study. However, it is possible that SRLK was present in our samples, but in a quantity too low to be detected by the methods used here. Also still needed from studies of pollination in conifers and other gymnosperms are data from the diallel crosses that can reveal the presence of SI (Gibbs, 2014), and a sufficient number of histological observations of self- and cross-pollination events.
Pollination drop formation
The pollination drop, like nectar, is a liquid secretion from reproductive tissue, and it is possible that the mechanisms controlling secretion in these systems are similar. Ovule transcripts in our RNA-Seq data may have a bearing on the question of whether similar mechanisms are used. The pollination drop originates in the nucellus, as shown by immunolocalization studies of pollination drop proteins (Poulis et al., 2005), and then passes through the micropyle to form a droplet. Ultimately, it retracts, bringing pollen into the ovule. In Cephalotaxus, drop production and retraction follow a diurnal cycle, with drops produced in the early morning and slowly retracting throughout the day, a phenomenon that has been observed in podocarpaceous conifers (Tomlinson et al., 1991). Drop retraction is also induced when pollen enters the drops of Cephalotaxus (C. Pirone-Davies, pers. comm.); this also has been noted in other conifers, including the Pinaceae, Podocarpaceae and Cupressaceae (Doyle and O’ Leary, 1935; Tomlinson et al., 1997; Mugnaini et al., 2007). The regulation of water movement and its role in drop production is not well understood. Conifer ovules are not vascularized, and thus water associated with pollination drop formation and retraction apparently is not associated with the osmotic potential of the xylem. Pollination drop formation must therefore be controlled by water dynamics within the ovule or cone (O’Leary and von Aderkas, 2005). One explanation is that changes in the osmotic potential of the drop facilitate the movement of water (Ziegler, 1959; Coulter et al., 2012). Sugars are proposed to regulate water movement during nectar secretion in angiosperms (Lin et al., 2014), and a similar process could be occurring during drop formation in gymnosperms, with the various sugars present in the drops serving as osmotic regulators (Seridi-Benkaddour and Chesnoy, 1988; Nepi et al., 2009). Consistent with this idea, we detected several transcripts for proteins involved in the transport of sugars (Supplementary Data Table S5), including several members of the SWEETS protein family, a superfamily of sugar transporters (Xuan et al., 2013), some of which are involved in nectar secretion (Lin et al., 2014). Transcripts of β-glucosidase and P-loop-containing nucleoside triphosphate hydrolases, additional proteins involved in nectar production (Bender et al., 2012), also are present in the Cephalotaxus drops.
Reproductive development
Ovules are a signature feature of the seed plants, and the presence of the ovule and placental tissue connecting it to a megasporophyll determines female identity. Ovules and pollen typically are borne in separate structures in gymnosperms. This leads to the expectation that genes known to control ovule development and female identity in angiosperms will be expressed in the ovulate cones of gymnosperms, while those controlling male identity will be expressed in pollen cones. In particular, one might expect similar sets of genes to be expressed in ovules across seed plants, since ovules are conserved, unlike the structures on which they are borne (Mathews and Kramer, 2012). Transcripts we detected in the ovules of Cephalotaxus that are homologues of genes involved in reproductive development in angiosperms bear on these expectations. These include ULTRAPETALA (ULT1), MADS-box transcription factors, AP2-related, CLAVATA (CLV), WUSCHEL (WUS), JOINTLESS and LEAFY (LFY) (Supplementary Data Table S3). The roles of several of these loci are understood in angiosperms, but fewer data are available for gymnosperms (for a review, see Mathews and Kramer, 2012).
Ovule identity in angiosperms is determined by loci in the AGAMOUS (AG) protein lineage (Pinyopich et al., 2003), and we detected several AG and AG-like transcripts in the C. sinensis ovule. Angiosperm ovules also express WUS, a member of the WUSCHEL-related homeobox domain (WOX) family, but they do not express CLAVATA3 (CLV3). In angiosperm shoot apical meristems (Laux et al., 1996) WUS and CLV signalling pathways interact to maintain the stem cell niche of the central zone. In arabidopsis ovules, WUS is critical for initiation of the integuments, consistent with the origin of the nucellus of the ovule from a shoot apical meristem (Gross-Hardt et al., 2002; Mathews and Kramer, 2012), and the presence of WUS in the ovules of Gnetum (Nardmann et al., 2009) and C. sinensis suggests that the role of WUS during integument formation may be conserved across the seed plants. Consistent with their determinate nature, CLV3 is not expressed in arabidopsis ovules. It is thus interesting that we detected CLV transcripts in the dissected ovules of C. sinensis. This could point to the presence of a stem cell niche, maintained by the signalling of WUS and CLV. Alternatively, the presence of WUS and CLV in the ovule could result from meristematic activities in the integuments during early stages of their development. Perhaps less probably, they could represent a novel set of functions in ovules.
Within both shoot and floral meristems of arabidopsis, control of cell accumulation involves ULT1 (Fletcher, 2001). ULT1 also positively regulates floral meristem determinacy, possibly through the AG pathway (Prunet et al., 2008). ULT1 encodes a trithorax group protein, a class of proteins involved in chromatin remodelling. In arabidopsis, ULT1 interacts with KANADI to organize the gynoecium along two polarity axes (Pires et al., 2014) In Cephalotaxus, the ULT1 homologue probably is also involved in chromatin remodelling and may be controlling some aspect of ovule development. It would be interesting to determine whether it is involved in polarity determination in the ovule.
MADS-box genes are transcription factors that contain a MADS DNA-binding domain, which is conserved across eukaryotes and metazoans (Gramzow et al., 2014). MADS-domain proteins are sub-divided into Type 1, or serum response factor (SRF)-like proteins, and Type 2, or myocyte enhancer factor (MEF)-like proteins. Few Type 1 genes have been functionally characterized, but Type 1 genes in arabidopsis are involved in female gametophyte, embryo sac and seed development (for a review, see Gramzow and Theissen, 2010). Type 1 genes were detected in several members of the Pinaceae as well as in Sciadopitys verticillata. However, detection of transcripts is infrequent and is limited to shoot, bud, male cone and embryo tissues (Gramzow et al., 2014). The functions of these transcripts are unknown. Our data set contains a single Type 1 MADS-box transcript.
Type 2 MADS-box genes, in contrast, have been extensively studied in angiosperms, where they are best known for their roles in floral organ identity. They also are involved in diverse developmental processes in fruits, seeds, embryos, roots and leaves (for reviews, see Becker, 2003; Theissen, 2001). Determination of organ identity in flowers is described by the ABCDE model, with A + E class genes specifying sepals, A + B + E class petals, B + C + E class stamens, C + E class carpels, and D + E class ovules (for reviews, see Causier et al., 2010; O’Maoileidigh et al., 2014). Most genes in these classes are Type 2 MADS-box genes. In gymnosperms, B and C class genes appear to be involved in the specification of reproductive structures and the differentiation of male and female cones (Melzer et al., 2010). Using BLAST searches, we detected several Cephalotaxus transcripts that showed greatest similarity to Type 2 MADS-box genes, including B class and B-sister (Bs) transcripts, AG and AG-like (C class), TM8 and DEFICIENS AGAMOUS-LIKE 10 (DAL10). We verified via phylogenetic analyses (S. Mathews, E. Kramer, C. Pirone-Davies, unpubl. res.) that among the BLAST hits are single homologues of B class, Bs and AG transcripts.
The MADS-box genes from the A class have not previously been detected in gymnosperms (Melzer et al., 2010). However, the A class gene AP2 from the AP2/ERF family of transcription factors has been detected in several conifers (e.g. Nilsson et al., 2007). In arabidopsis, AP2 specifies the identity of sepals and petals, regulates C class genes, is involved in seed development and may play a role in development of non-floral organs (Jofuku et al., 1994). Homologues of AP2 from Picea probably control diverse developmental events, but also share features of their angiosperm counterparts, as PiAP2 has the capacity to substitute for an A class gene in arabidopsis (Nilsson et al., 2007). We detected one AP2-related transcript in C. sinensis.
The MADS-box genes from the B class have been found in Ginkgo, Gnetum gnemon and some conifers (Mouradov et al., 1999; Sundstrom et al., 1999; Gramzow et al., 2014), where their expression is largely restricted to male cones. It appears that the role of these genes in the development of pollen-bearing structures is conserved across seed plants (Sundstrom and Engstrom, 2002; Theissen and Becker, 2004). B class transcripts were, however, found by Gramzow and colleagues (Gramzow et al., 2014) in transcriptomes assembled from the female cones of Picea abies. Our detection of B class and Bs transcripts in the ovules of C. sinensis, suggests that B class genes are indeed involved in a role beyond male reproductive development. Since we collected ovules from outdoor sites where pollen was likely to be present, we cannot completely rule out the possibility that we detected transcripts from the pollen. It is unclear, however, whether B class transcripts would occur in mature pollen. In situ data indicate their presence in various pollen cone tissues, but no transcripts were detected in the pollen mother cells, and mature pollen was not analysed (Sundstrom and Engstrom, 2002). The expression of one or more B gene homologues in ovules is not uncommon in angiosperms (for a review, see Kramer and Irish, 2000), so it may be that this pattern is also common among gymnosperms.
Homologues of the C class gene AG have been identified in all major gymnosperm lineages (Rutledge et al., 1998; Tandre et al., 1998; Winter et al., 1999; Kramer et al., 2003), and AG and AG-like are expressed in the female cones of four cycads and several conifers, the male cones of Cryptomeria, the shoots of Gnetum gnemon, non-reproductive tissues of Picea abies and in the nucellus of Taxus globosa (Englund et al., 2011; Gramzow et al., 2014). In flowering plants, AG is involved in stamen and carpel identity and in establishing determinancy of the floral meristem (Bowman et al., 1989). In gymnosperms, AG genes are generally involved in the development of both male and female cones (Melzer et al., 2010), and may be involved in the development of ovuliferous scales (Tandre et al., 1998) and in the transition from vegetative to reproductive identity (Carlsbecker et al., 2013).
Additional Type 2 MADS-box genes in the Cephalotaxus ovular transcriptome include TM8, DAL10 and JOINTLESS. TM8 and DAL10 belong to a large clade that is sister to the C class clade (Melzer et al., 2010). TM8 expression occurs in reproductive and non-reproductive tissues of diverse gymnosperms (Gramzow et al., 2014), including the ovules of Taxus baccata and G. biloba (Lovisetto et al., 2012). Although the function of TM8 remains poorly understood, it may be involved in controlling A class expression in tomato (Daminato et al., 2014). Given the diversity of TM8 expression patterns across seed plants, further research into its function is needed. DAL10 has also been found in numerous gymnosperm species and tissues, but is absent from angiosperm lineages (Carlsbecker et al., 2003). Similar to TM8, little is known about the function of this gene, although it appears to be involved in the shift from vegetative to reproductive buds, and its presence in developing seed and pollen cones is similar to that of B and C class genes (Carlsbecker et al., 2003, 2013). JOINTLESS is involved in the formation of the abscission zone in tomato (Mao et al., 2000). It works at least in part via the regulation of transcription factors involved in meristem identity, genes involved in cell wall formation and lipid metabolism, and phytohormones (Nakano et al., 2012).
The presence in Cephalotaxus ovules of transcripts similar to the transcription factor gene LFY is consistent with its activity in early female and male cone development (Vasquez-Lobo et al., 2007). It may regulate some ABC class MADS-box proteins as it does in angiosperms (Moyroud et al., 2010). In angiosperms, LFY enables the transition from vegetative to floral meristems (Weigel et al., 1992), and also activates floral homeotic genes (Weigel and Meyerowitz, 1994). Members of the LFY lineage have been detected in non-reproductive and reproductive tissues of diverse non-flowering plants (Moyroud et al., 2010), including the nucellus of Picea (Carlsbecker et al., 2013).
In summary, our proteome data strongly support the hypothesis that pollination drop proteins are important in defence, polysaccharide metabolism and pollen tube growth. We detected additional, novel proteins that may be involved in defence (Cup a3, Cup s and Jun r), starch degradation (α-amylase) and callose degradation (pollen allergen CJP-38), while others are likely to be by-products of nucellar degradation (luminal binding protein, histone and others). The implications of the latter in pollination drop function, if any, are unclear. Examination of the transcriptome of Cephalotaxus ovules revealed several transcripts that may be important in pollen–ovule recognition, pollination drop formation and retraction, and reproductive and developmental processes within the ovule. Functional validation of the pollination drop proteins and ovular transcripts characterized here will deepen our knowledge of pollination biology in gymnosperms. Transcriptome data, moreover, from additional stages of ovule development and from a broad phylogenetic sample across gymnosperms will greatly advance our understanding of gymnosperm reproductive development. Finally, we note the need for histological and genetic data from rigorous self- and cross-pollination experiments to explore the intriguing observations that suggest that pollen–ovule signalling occurs in at least some gymnosperm reproductive systems.
SUPPLEMENTARY DATA
Supplementary data are available online at www.aob.oxfordjournals.org and consist of the following. Table S1: peptides and proteins identified in the pollination drops of C. koreana and C. sinensis. Table S2: BLAST results (nr, SwissProt and trEMBL) for the ovule transcriptome of C. sinensis. Table S3: Blast2go annotations for the ovule transcriptome of C. sinensis. Table S4: list of transcripts and proteins found in both the transcriptome and proteome data sets. Table S5: transcripts identified in the ovules of C. sinensis with the putative functions of sugar transport, pollen–ovule interactions and development.
ACKNOWLEDGEMENTS
We thank Elena Kramer for her insight on ovule development and for sharing with us her data matrix of MADS-box genes. This work was supported by a Putnam Fellowship from the Arnold Arboretum of Harvard University to C.P.D. The Natural Sciences and Engineering Research Council of Canada’s PGS and Discovery Grant Programs provided financial support to N.A.P. and P.vA., respectively.
LITERATURE CITED
- Abercrombie JM, O’Meara BC, Moffatt AR, Williams JH. 2011. Developmental evolution of flowering plant pollen tube cell walls: callose synthase (CalS) gene expression patterns. Evodevo 2: 14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- von Aderkas P, Nepi M, Rise M, et al. 2012. Post-pollination prefertilization drops affect germination rates of heterospecific pollen in larch and Douglas-fir. Sexual Plant Reproduction 25: 215–225. [DOI] [PubMed] [Google Scholar]
- von Aderkas P, Prior NA, Gagnon S, et al. 2015. Degradome and secretome of pollination drops of Ephedra. Botanical Review 81: 1–27. [Google Scholar]
- Alvim FC, Carolino SM, Cascardo JC, et al. 2001. Enhanced accumulation of BiP in transgenic plants confers tolerance to water stress. Plant Physiology 126: 1042–1054. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Andrews S. FastQC a quality control tool for high throughput sequence data. http://www.bioinformatics.babraham.ac.uk/projects/fastqc/.
- Bassett CL, Nickerson ML, Farrell RE, et al. 2005. Characterization of an S-locus protein kinase-like gene from peach. Tree Physiology 25: 403–411. [DOI] [PubMed] [Google Scholar]
- Becker A. 2003. The major clades of MADS-box genes and their role in the development and evolution of flowering plants. Molecular Phylogenetics and Evolution 29: 464–489. [DOI] [PubMed] [Google Scholar]
- Bender R, Klinkenberg P, Jiang Z, et al. 2012. Functional genomics of nectar production in the Brassicaceae. Flora – Morphology, Distribution, Functional Ecology of Plants 207: 491–496. [Google Scholar]
- Boston RS, Viitanen PV, Vierling E. 1996. Molecular chaperones and protein folding in plants. Plant Molecular Biology 32: 191–222. [DOI] [PubMed] [Google Scholar]
- Bowman JL, Smyth DR, Meyerowitz EM. 1989. Genes directing flower development in Arabidopsis. The Plant Cell 1: 37–52. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Breiteneder H, Mills EN. 2005. Molecular properties of food allergens. Journal of Allergy and Clinical Immunology 115: 14–23. [DOI] [PubMed] [Google Scholar]
- Carafa AM, Carratù G, Pizzolongo P. 1992. Anatomical observations on the nucellar apex of Wellwitschia mirabilis and the chemical composition of the micropylar drop. Sexual Plant Reproduction 5: 275–279. [Google Scholar]
- Carlsbecker A, Sundstrom J, Tandre K, et al. 2003. The DAL10 gene from Norway spruce (Picea abies) belongs to a potentially gymnosperm-specific subclass of MADS-box genes and is specifically active in seed cones and pollen cones. Evolution and Development 5: 551–561. [DOI] [PubMed] [Google Scholar]
- Carlsbecker A, Sundstrom JF, Englund M, et al. 2013. Molecular control of normal and acrocona mutant seed cone development in Norway spruce (Picea abies) and the evolution of conifer ovule-bearing organs. New Phytologist 200: 261–275. [DOI] [PubMed] [Google Scholar]
- Causier B, Schwarz-Sommer Z, Davies B. 2010. Floral organ identity: 20 years of ABCs. Seminars in Cell and Developmental Biology 21: 73–79. [DOI] [PubMed] [Google Scholar]
- Chang S, Puryear J, Cairney J. 1993. A simple and efficient method for isolating RNA from pine trees. Plant Molecular Biology Reporter 11: 113–116. [Google Scholar]
- Conesa A, Gotz S, Garcia-Gomez JM, Terol J, Talon M, Robles M. 2005. Blast2GO: a universal tool for annotation, visualization and analysis in functional genomics research. Bioinformatics 21: 3674–3676. [DOI] [PubMed] [Google Scholar]
- Cortegano I, Civantos E, Aceituno E, et al. 2004. Cloning and expression of a major allergen from Cupressus arizonica pollen, Cup a 3, a PR-5 protein expressed under polluted environment. Allergy 59: 485–490. [DOI] [PubMed] [Google Scholar]
- Coulter A, Poulis BAD, von Aderkas P. 2012. Pollination drops as dynamic apoplastic secretions. Flora – Morphology, Distribution, Functional Ecology of Plants 207: 482–490. [Google Scholar]
- Daminato M, Masiero S, Resentini F, Lovisetto A, Casadoro G. 2014. Characterization of TM8, a MADS-box gene expressed in tomato flowers. BMC Plant Biology 14: 319. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dogra PD. 1967. Seed sterility and disturbances in embryogeny in conifers with particular reference to seed testing and tree breeding in Pinaceae. Studia Forestalia Suecica 45: 4–28. [Google Scholar]
- Doyle J, O’ Leary M. 1935. Pollination in Tsuga, Cedrus, Pseudotsuga, and Larix. With 2 plates. Scientific Proceedings of the Royal Dublin Society XXI: 191–204. [Google Scholar]
- Dwyer KG, Kanakachari MK, Mahosky DI, et al. 1994. A superfamily of S locus-related sequences in Arabidopsis: diverse structures and expression patterns. The Plant Cell 6: 1829–1843. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Elias JE, Haas W, Faherty BK, Gygi SP. 2005. Comparative evaluation of mass spectrometry platforms used in large-scale proteomics investigations. Nature Methods 2: 667–675. [DOI] [PubMed] [Google Scholar]
- Englund M, Carlsbecker A, Engstrom P, Vergara-Silva F. 2011. Morphological ‘primary homology’ and expression of AG-subfamily MADS-box genes in pines, podocarps, and yews. Evolution and Development 13: 171–181. [DOI] [PubMed] [Google Scholar]
- Fernando DD, Long SM, Sniezko RA. 2005. Sexual reproduction and crossing barriers in white pines: the case between Pinus lambertiana (sugar pine) and P. monticola (western white pine). Tree Genetics and Genomes 1: 143–150. [Google Scholar]
- Fernando DD, Quinn CR, Brenner ED, Owens JN. 2010. Male gametophyte development and evolution in extant gymnosperms. International Journal of Plant Developmental Biology 4: 47–63. [Google Scholar]
- Fletcher JC. 2001. The ULTRAPETALA gene controls shoot and floral meristem size in Arabidopsis. Development 128: 1323–1333. [DOI] [PubMed] [Google Scholar]
- Franco OL, Rigden DJ, Melo FR, Grossi-de-Sa M. 2002. Plant alpha-amylase inhibitors and their interaction with insect alpha-amylases. European Journal of Biochemistry 269: 397–412. [DOI] [PubMed] [Google Scholar]
- Franklin-Tong VE. 2008. Self-incompatibility in flowering plants. Berlin: Springer-Verlag. [Google Scholar]
- Galili G, Sengupta-Gopalan C, Ceriotti A. 1998. The endoplasmic reticulum of plant cells and its role in protein maturation and biogenesis of oil bodies. Plant Molecular Biology 38: 1–29. [PubMed] [Google Scholar]
- Ge Y, Li Y, Zhu YM, et al. 2010. Global transcriptome profiling of wild soybean (Glycine soja) roots under NaHCO3 treatment. BMC Plant Biology 10: 153. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gelbart G, von Aderkas P. 2002. Ovular secretions as part of pollination mechanisms in conifers. Annals of Forest Science 59: 345–357. [Google Scholar]
- Gibbs PE. 2014. Late-acting self-incompatibility – the pariah breeding system in flowering plants. New Phytologist 203: 717–734. [DOI] [PubMed] [Google Scholar]
- Grabherr MG, Haas BJ, Yassour M, et al. 2011. Full-length transcriptome assembly from RNA-Seq data without a reference genome. Nature Biotechnology 29: 644–652. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gramzow L, Theissen G. 2010. A hitchhiker’s guide to the MADS world of plants. Genome Biology 11: 214–225. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gramzow L, Weilandt L, Theissen G. 2014. MADS goes genomic in conifers: towards determining the ancestral set of MADS-box genes in seed plants. Annals of Botany 114: 1407–11429. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Grenier J, Potvin C, Trudel J, Asselin A. 1999. Some thaumatin-like proteins hydrolyse polymeric β-1,3-glucans. The Plant Journal 19: 473–480. [DOI] [PubMed] [Google Scholar]
- Gross-Hardt R, Lenhard M, Laux T. 2002. WUSCHEL signaling functions in interregional communication during Arabidopsis ovule development. Genes and Development 16: 1129–1138. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Grover A. 2012. Plant chitinases: genetic diversity and physiological roles. Critical Reviews in Plant Sciences 31: 57–73. [Google Scholar]
- Ham BM, Yang F, Jayachandran H, et al. 2008. The influence of sample preparation and replicate analyses on HeLa cell phosphoproteome coverage. Journal of Proteome Research 7: 2215–2221. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Han Y, Ma B, Zhang K. 2005. SPIDER: software for protein identification from sequence tags with de novo sequencing error. Journal of Bioinformatics and Computational Biology 3: 697–716. [DOI] [PubMed] [Google Scholar]
- Hon WC, Griffith M, Mlynarz A, Kwok YC, Yang DS. 1995. Antifreeze proteins in winter rye are similar to pathogenesis-related proteins. Plant Physiology 109: 879–89. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hruba P, Honys D, Twell D, Capkova V, Tupy J. 2005. Expression of beta-galactosidase and beta-xylosidase genes during microspore and pollen development. Planta 220: 931–940. [DOI] [PubMed] [Google Scholar]
- Huang N, Stebbins GL, Rodriguez RL. 1992. Classification and evolution of alpha-amylase genes in plants. Proceedings of the National Academy of Sciences, USA 89: 7526–7530. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Igic B, Lande R, Kohn JR. 2008. Loss of self-incompatibility and its evolutionary consequences. International Journal of Plant Sciences 169: 93–104. [Google Scholar]
- Iwano M, Takayama S. 2012. Self/non-self discrimination in angiosperm self-incompatibility. Current Opinion in Plant Biology 15: 78–83. [DOI] [PubMed] [Google Scholar]
- Jain SK. 1976. The evolution of inbreeding in plants. Annual Review of Ecology and Systematics 7: 469–495. [Google Scholar]
- Jelitto-Van Dooren EP, Vidal S, Denecke J. 1999. Anticipating endoplasmic reticulum stress. A novel early response before pathogenesis-related gene induction. The Plant Cell 11: 1935–1944. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jofuku KD, den Boer BGW, Montagu MV, Okamuro JK. 1994. Control of Arabidopsis flower and seed development by the homeotic gene APETALA2. The Plant Cell 6: 1211–1225. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Joshi NA, Fass JN. 2011. Sickle: a sliding-window, adaptive, quality-based trimming tool for FastQ files http://github.com/najoshi/sickle.
- Kemp BP, Doughty J. 2007. S cysteine-rich (SCR) binding domain analysis of the Brassica self-incompatibility S-locus receptor kinase. New Phytologist 175: 619–629. [DOI] [PubMed] [Google Scholar]
- Kormatuk A. 1999. Self-incompatibility in silver fir (Abies alba). Biologia Bratislava 54: 101–105. [Google Scholar]
- Kotake T, Li YQ, Takahashi M, Sakurai N. 2000. Characterization and function of wall-bound exo-β-glucanases of Lilium longiflorum pollen tubes. Sexual Plant Reproduction 13: 1–9. [Google Scholar]
- Kramer EM, Irish VF. 2000. Evolution of the petal and stamen developmental programs: evidence from comparative studies of the lower eudicots and basal angiosperms. International Journal of Plant Sciences 161: S29–S40. [Google Scholar]
- Kramer EM, Jaramillo MA, Di Stilio VS. 2003. Patterns of gene duplication and functional evolution during the diversification of the AGAMOUS subfamily of MADS Box genes in angiosperms. Genetics 166: 1011–1023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Laux T, Mayer KFX, Berger J, Jurgens G. 1996. The WUSCHEL gene is required for shoot and floral meristem integrity in Arabidopsis. Development 122: 87–96. [DOI] [PubMed] [Google Scholar]
- Leslie AB, Beaulieu JM, Rai HS, Crane PR, Donoghue MJ, Mathews S. 2012. Hemisphere-scale differences in conifer evolutionary dynamics. Proceedings of the National Academy of Sciences, USA 109: 16217–16221. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Levy A, Erlanger M, Rosenthal M, Epel BL. 2007. A plasmodesmata-associated beta-1,3-glucanase in Arabidopsis. The Plant Journal 49: 669–682. [DOI] [PubMed] [Google Scholar]
- Lin IW, Sosso D, Chen LQ, et al. 2014. Nectar secretion requires sucrose phosphate synthases and the sugar transporter SWEET9. Nature 508: 546–549. [DOI] [PubMed] [Google Scholar]
- Liu JJ, Sturrock R, Ekramoddoullah AK. 2010. The superfamily of thaumatin-like proteins: its origin, evolution, and expression towards biological function. Plant Cell Reports 29: 419–436. [DOI] [PubMed] [Google Scholar]
- Lohse M, Bolger AM, Nagel A, et al. 2012. RobiNA: a user-friendly, integrated software solution for RNA-Seq-based transcriptomics. Nucleic Acids Research 40: W622–W627. [DOI] [PMC free article] [PubMed] [Google Scholar]
- van Loon LC, Rep M, Pieterse CM. 2006. Significance of inducible defense-related proteins in infected plants. Annual Review of Phytopathology, 44: 135–162. [DOI] [PubMed] [Google Scholar]
- Lovisetto A, Guzzo F, Tadiello A, Toffali K, Favretto A, Casadoro G. 2012. Molecular analyses of MADS-box genes trace back to Gymnosperms the invention of fleshy fruits. Molecular Biology and Evolution 29: 409–419. [DOI] [PubMed] [Google Scholar]
- Mao L, Begum D, Chuang H, et al. 2000. JOINTLESS is a MADS-box gene controlling tomato flower abscission zone development. Nature 406: 910–913. [DOI] [PubMed] [Google Scholar]
- Martin M. 2011. Cutadapt removes adapter sequences from high-throughput sequencing reads. EMBNet 17: 10–12. [Google Scholar]
- Mathews S, Kramer EM. 2012. The evolution of reproductive structures in seed plants: a re-examination based on insights from developmental genetics. New Phytologist 194: 910–923. [DOI] [PubMed] [Google Scholar]
- McWilliam JR. 1959. Interspecific incompatibility in Pinus. American Journal of Botany 46: 425–433. [Google Scholar]
- Melzer R, Wang YQ, Theissen G. 2010. The naked and the dead: the ABCs of gymnosperm reproduction and the origin of the angiosperm flower. Seminars in Cell and Developmental Biology 21: 118–128. [DOI] [PubMed] [Google Scholar]
- Mouradov A, Hamdorf B, Teasdale RD, Kim JT, Winter K, Theissen G. 1999. A DEF/GLO-like MADS-box gene from a gymnosperm: Pinus radiata contains an ortholog of angiosperm B class floral homeotic genes. Developmental Genetics 25: 245–252. [DOI] [PubMed] [Google Scholar]
- Moyroud E, Kusters E, Monniaux M, Koes R, Parcy F. 2010. LEAFY blossoms. Trends in Plant Science 15: 346–352. [DOI] [PubMed] [Google Scholar]
- Mugnaini S, Nepi M, Guarnieri M, Piotto B, Pacini E. 2007. Pollination drop in Juniperus communis: response to deposited material. Annals of Botany 100: 1475–1481. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nakano T, Kimbara J, Fujisawa M, et al. 2012. MACROCALYX and JOINTLESS interact in the transcriptional regulation of tomato fruit abscission zone development. Plant Physiology 158: 439–450. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nardmann J, Reisewitz P, Werr W. 2009. Discrete shoot and root stem cell-promoting WUS/WOX5 functions are an evolutionary innovation of angiosperms. Molecular Biology and Evolution 26: 1745–1755. [DOI] [PubMed] [Google Scholar]
- Nepi M, von Aderkas P, Wagner R, Mugnaini S, Coulter A, Pacini E. 2009. Nectar and pollination drops: how different are they? Annals of Botany 104: 205–219. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nilsson L, Carlsbecker A, Sundas-Larsson A, Vahala T. 2007. APETALA2 like genes from Picea abies show functional similarities to their Arabidopsis homologues. Planta 225: 589–602. [DOI] [PubMed] [Google Scholar]
- O’Leary SJB, von Aderkas P. 2005. Postpollination drop production in hybrid larch is not related to the diurnal pattern of xylem water potential. Trees 20: 61–66. [Google Scholar]
- O’Leary SJB, Joseph C, von Aderkas P. 2004. Origin of arabinogalactan proteins in the pollination drop of Taxus × media. Austrian Journal of Forest Science 121: 35–46. [Google Scholar]
- O’Leary SJB, Poulis BAD, von Aderkas P. 2007. The identification of two thaumatin-like proteins (TLPs) in the pollination drop of hybrid yew that may play a role in pathogen defence during pollen collection. Tree Physiology 27: 1649–1659. [DOI] [PubMed] [Google Scholar]
- O’Maoileidigh DS, Graciet E, Wellmer F. 2014. Gene networks controlling Arabidopsis thaliana flower development. New Phytologist 201: 16–30. [DOI] [PubMed] [Google Scholar]
- Owens JN, Blake MD. 1983. Pollen morphology and development of the pollination mechanism in Tsuga heterophylla and T. mertensiana. Canadian Journal of Botany,61: 3041–3049. [Google Scholar]
- Owens JN, Simmons SJ. 1987. The pollination mechanism of Engelmann spruce (Picea engelmanii). Canadian Journal of Botany 65: 1439–1450. [Google Scholar]
- Owens JN, Colangeli AM, Morris SJ. 1990. Factors affecting seed set in Douglas-fir (Pseudotsuga menziesii). Canadian Journal of Botany 69: 229–238. [Google Scholar]
- Owens JN, Bennett J, L’Hirondelle S. 2005. Pollination and cone morphology affect cone and seed production in lodgepole pine seed orchards. Canadian Journal of Forest Research 35: 383–400. [Google Scholar]
- Passardi F, Cosio C, Penel C, Dunand C. 2005. Peroxidases have more functions than a Swiss army knife. Plant Cell Reports 24: 255–265. [DOI] [PubMed] [Google Scholar]
- Pinyopich A, Ditta GS, Savidge B, et al. 2003. Assessing the redundancy of MADS-box genes during carpel and ovule development. Nature 424: 85–88. [DOI] [PubMed] [Google Scholar]
- Pires HR, Monfared MM, Shemyakina EA, Fletcher JC. 2014. ULTRAPETALA trxG genes interact with KANADI transcription factor genes to regulate Arabidopsis gynoecium patterning. The Plant Cell 26: 4345–4361. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Poulis BAD, O’Leary SJB, Haddow JD, von Aderkas P. 2005. Identification of proteins present in the Douglas fir ovular secretion: an insight into conifer pollen selection and development. International Journal of Plant Sciences 166: 733–739. [Google Scholar]
- Prior N, Little SA, Pirone C, et al. 2013. Application of proteomics to the study of pollination drops. Applications in Plant Science 1: doi:10.3732/apps.1300008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Prunet N, Morel P, Thierry AM, et al. 2008. REBELOTE, SQUINT, and ULTRAPETALA1 function redundantly in the temporal regulation of floral meristem termination in Arabidopsis thaliana. The Plant Cell 20: 901–919. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rai HS, Reeves PA, Peakall R, Olmstead RG, Graham SW. 2008. Inference of higher-order conifer relationships from a multi-locus plastid data set. Botany 86: 658–669. [Google Scholar]
- Rejon JD, Delalande F, Schaeffer-Reiss C, et al. 2013. Proteomics profiling reveals novel proteins and functions of the plant stigma exudate. Journal of Experimental Botany 64: 5695–5705. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Runions CJ, Owens JN. 1998. Evidence of pre-zygotic self-incompatibility in a conifer In: PJ Rudall, SJ Owens, eds. Reproductive biology. Kew: Royal Botanic Gardens, 255–264. [Google Scholar]
- Rutledge R, Regan S, Nicolas O, et al. 1998. Characterization of an AGAMOUS homologue from the conifer black spruce (Picea mariana) that produces floral homeotic conversions when expressed in Arabidopsis. The Plant Journal 15: 625–634. [DOI] [PubMed] [Google Scholar]
- Sela-Buurlage MB, Ponstein AS, Bres-Vloemans SA, et al. 1993. Only specific tobacco (Nicotiana tabacum) chitinases and [beta]-1,3-glucanases exhibit antifungal activity. Plant Physiology 101: 857–863. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Seridi-Benkaddour R, Chesnoy L. 1988. Secretion and composition of the pollination drop in Cephalotaxus drupaceae (gymnosperm, Cephalotaxeae) In: M Cresti, P Gori, E Pacini, eds. Sexual reproduction in higher plants. Berlin: Springer Verlag, 345–350. [Google Scholar]
- Sherman-Broyles S, Boggs N, Farkas A, et al. 2007. S locus genes and the evolution of self-fertility in Arabidopsis thaliana. The Plant Cell 19: 94–106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shiu SH, Bleecker AB. 2003. Expansion of the receptor-like kinase/Pelle gene family and receptor-like proteins in Arabidopsis. Plant Physiology 132: 530–543. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Singh H. 1978. Embryology of gymnosperms. Berlin: Gerbrüder Borntraeger. [Google Scholar]
- Stebbins GL. 1957. Self fertlization and population variability in the higher plants. American Naturalist 91: 337–354. [Google Scholar]
- Stein JC, Howlett B, Boyes DC, Nasrallah ME, Nasrallah JB. 1991. Molecular cloning of a putative receptor protein kinase gene encoded at the self-incompatibility locus of Brassica oleracea. Proceedings of the National Academy of Sciences, USA 88: 8816–8820. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sun XL, Yu QY, Tang LL, et al. 2013. GsSRK, a G-type lectin S-receptor-like serine/threonine protein kinase, is a positive regulator of plant tolerance to salt stress. Journal of Plant Physiology 170: 505–515. [DOI] [PubMed] [Google Scholar]
- Sundstrom J, Engstrom P. 2002. Conifer reproductive development involves B-type MADS-box genes with distinct and different activities in male organ primordia. The Plant Journal 31: 161–169. [DOI] [PubMed] [Google Scholar]
- Sundstrom J, Carlsbecker A, Svensson ME, et al. 1999. MADS-Box genes active in developing pollen cones of Norway Spruce (Picea abies) are homologous to the B-Class floral homeotic genes in angiosperms. Developmental Genetics 25: 253–266. [DOI] [PubMed] [Google Scholar]
- Takaso T, Owens JN. 1995. Pollination drop and microdrop secretions in Cedrus. International Journal of Plant Sciences 156: 640–649. [Google Scholar]
- Takayama S, Isogai A. 2005. Self-incompatibility in plants. Annual Review of Plant Biology 56: 467–489. [DOI] [PubMed] [Google Scholar]
- Tandre K, Svenson M, Svensson ME, Engstrom P. 1998. Conservation of gene structure and activity in the regulation of reproductive organ development of conifers and angiosperms. The Plant Journal 15: 615–623. [DOI] [PubMed] [Google Scholar]
- Theissen G. 2001. Development of floral organ identity: stories from the MADS house. Current Opinion in Plant Biology 4: 75–85. [DOI] [PubMed] [Google Scholar]
- Theissen G, Becker A. 2004. Gymnosperm orthologues of class B floral homeotic genes and their impact on understanding flower origin. Critical Reviews in Plant Sciences 23: 129–148. [Google Scholar]
- Tomlinson PB, Braggins JE, Rattenbury JA. 1991. Pollination drop in relation to cone morphology in Podocarpaceae: a novel reproductive mechanism. American Journal of Botany 78: 1289–1303. [Google Scholar]
- Tomlinson PB, Braggins JE, Rattenbury JA. 1997. Contrasted pollen capture mechanisms in Phyllocladaceae and certain Podocarpaceae (Coniferales). American Journal of Botany 84: 214–223. [PubMed] [Google Scholar]
- Vasquez-Lobo A, Carlsbecker A, Vergara-Silva F, Alvarez-Buyila ER, Pineroa D, Engstrom P. 2007. Characterization of the expression patterns of LEAFY/FLORICAULA and NEEDLY orthologues in female and male cones of the conifer genera Picea, Podocarpus, and Taxus: implications for current evo-devo hypotheses for gymnosperms. Evolution and Development 9: 446–459. [DOI] [PubMed] [Google Scholar]
- Vogel C, Marcotte EM. 2012. Insights into the regulation of protein abundance from proteomic and transcriptomic analyses. Nature Reviews Genetics 13: 227–232. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wagner RE, Mugnaini S, Sniezko R, et al. 2007. Proteomic evaluation of gymnosperm pollination drop proteins indicates highly conserved and complex biological functions. Sexual Plant Reproduction 20: 181–189. [Google Scholar]
- Weigel D, Meyerowitz EM. 1994. The ABCs of floral homeotic genes. Cell 78: 203–209. [DOI] [PubMed] [Google Scholar]
- Weigel G, Alvarez J, Smyth DR, Yanofsky MF, Meyerowitz EM. 1992. LEAFY controls floral meristem identity in Arabidopsis. Cell 69: 843–859. [DOI] [PubMed] [Google Scholar]
- Winter K, Becker A, Munster T, Kim JT, Saedler H, Theissen G. 1999. MADS-box genes reveal that gnetophytes are more closely related to conifers than to flowering plants. Proceedings of the National Academy of Sciences, USA 96: 7342–7347. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xing S, Li M, Liu P. 2013. Evolution of S-domain receptor-like kinases in land plants and origination of S-locus receptor kinases in Brassicaceae. BMC Evolutionary Biology 13: 1–11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xuan YH, Hu YB, Chen LQ, Sosso D, Ducat DC, Hou BH, Frommer WB. 2013. Functional role of oligomerization for bacterial and plant SWEET sugar transporter family. Proceedings of the National Academy of Sciences, USA 110: E3685–E3694. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yatomi R, Nakamura S, Nakamura N. 2002. Immunochemical and cytochemical detection of wall components of germinated pollen of gymnosperms. Grana 41: 21–28. [Google Scholar]
- Ziegler H. 1959. Über die Zusammensetzung des ‘Bestäubungstropfens’ und den Mechanismus seiner Sekretion. Planta 52: 587–599. [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.