Abstract
Sprague-Dawley rats selectively-bred for susceptibility to stress in our laboratory (Susceptible, or SUS rats) voluntarily consume large amounts of alcohol, and amounts that have, as shown here, pharmacological effects, which normal rats will not do. In this paper, we explore neural events in the brain that underlie this propensity to readily consume alcohol. Activity of locus coeruleus neurons (LC), the major noradrenergic cell body concentration in the brain, influences firing of ventral tegmentum dopaminergic cell bodies of the mesocorticolimbic system (VTA-DA neurons), which mediate rewarding aspects of alcohol. We tested the hypothesis that in SUS rats, alcohol potently suppresses LC activity to markedly diminish LC-mediated inhibition of VTA-DA neurons, which permits alcohol to greatly increase VTA-DA activity and rewarding aspects of alcohol. Electrophysiological single-unit recording of LC and VTA-DA activity showed that in SUS rats, alcohol decreased LC burst firing much more than in normal rats and as a result markedly increased VTA-DA activity in SUS rats while having no such effect in normal rats. Consistent with this, in a behavioral test for reward using conditioned place preference (CPP), SUS rats showed alcohol, given by intraperitoneal (i.p.) injection, to be rewarding. Next, manipulation of LC activity by microinfusion of drugs into the LC region of SUS rats showed that (a) decreasing LC activity increased alcohol intake and increasing LC activity decreased alcohol intake in accord with the formulation described above, and (b) increasing LC activity blocked both the rewarding effect of alcohol in the CPP test and the usual alcohol-induced increase in VTA-DA single-unit activity seen in SUS rats. An important ancillary finding in the CPP test was that an increase in LC activity was rewarding by itself, while a decrease in LC activity was aversive; consequently, effects of LC manipulations on alcohol-related reward in the CPP test were perhaps even larger than evident in the test. Finally, when increased LC activity was associated with (i.e., conditioned to) i.p. alcohol, subsequent alcohol consumption by SUS rats was markedly reduced, indicating that SUS rats consume large amounts of alcohol because of rewarding physiological consequences requiring increased VTA-DA activity. The findings reported here are consistent with the view that the influence of alcohol on LC activity leading to changes in VTA-DA activity strongly affects alcohol-mediated reward, and may well be the basis of the proclivity of SUS rats to avidly consume alcohol.
Keywords: selectively-bred rats, alcohol, locus coeruleus, dopamine, ventral tegmentum, conditioned place preference
Introduction
In our laboratory, we have selectively bred albino Sprague-Dawley rats for being differentially susceptible to stress. We selectively bred rats for showing susceptibility to having their motor activity in a swim test reduced after they had been exposed to a stressful event (which we called “Swim-test Susceptible” or “SUS” rats), and also, in parallel, selectively bred rats for showing resistance to having their activity in a swim test reduced by stress (called “Swim-test Resistant” or “RES” rats) (Scott, Cierpial, Kilts, & Weiss, 1996; West & Weiss, 2005). The SUS and RES lines of rats, now maintained for over 50 generations since their inception, show, respectively, less swim-test activity after exposure to a stressor than do randomly-bred (normal) rats, and more swim-test activity after exposure to a stressor than do randomly-bred (normal) rats. After the lines had been in existence for several years, we discovered (19th generation) that SUS rats voluntarily consume large amounts of alcohol. In over 30+ subsequent generations, we have observed SUS rats to consume large amounts of alcohol, in fact consuming similar amounts of alcohol to rat lines that have been specifically bred for the proclivity to consume alcohol (see discussion in West & Weiss, 2006). SUS rats do this even though consumption of alcohol had not played any part in the selective breeding development of the SUS rat. In comparison, normal Sprague-Dawley rats (and also RES rats) show notable resistance to consuming any alcohol voluntarily.
This paper describes studies that investigated possible neural events underlying the proclivity of SUS rats to avidly consume alcohol. The mesocorticolimbic dopaminergic system, originating from dopaminergic neurons in the ventral tegmentum (VTA-DA neurons), which project to prefrontal cortex and nucleus accumbens (NAC), has long been associated with the reinforcing actions of drugs of abuse. We first considered this system. Alcohol stimulates activity of VTA-DA neurons (Brodie, Pesold, & Appel, 1999; Brodie, Shefner, & Dunwiddie, 1990; Doyon et al., 2003; Imperato & Di Chiara, 1986; Melis, Diana, Enrico, Marinelli, & Brodie, 2009; Xiao et al., 2009), and activation of VTA-DA neurons has been associated with ethanol-induced reward (Gessa, Muntoni, Collu, Vargiu, & Mereu, 1985; McBride et al., 1991; Nowak, McBride, Lumeng, Li, & Murphy, 2000; Rodd et al., 2005). VTA-DA neuronal activity is higher in rats that drink significant amounts of alcohol compared to rats that do not drink alcohol; such studies have assessed VTA-DA activity in high responders to novelty vs. low responders (Marinelli & White, 2000), Lewis vs. Fischer 344 rats (Minabe, Emori, & Ashby, 1995), P vs. NP rats (Morzorati, 1998; Morzorati & Marunde, 2006), and Sardinian alcohol preferring vs. non-preferring rats (Melis et al., 2009). High alcohol-seeking behavior is also associated with lower content of dopamine (DA) in the NAC (McBride, Bodart, Lumeng, & Li, 1995; McBride et al., 1991; Murphy, McBride, Lumeng, & Li, 1987; Murphy et al., 2002; reviewed in McBride & Li, 1998), which will induce higher activity in VTA-DA neurons of alcohol-preferring animals insofar as low DA levels in NAC will not exert normal “end-product negative feedback” and thus will lead to elevated VTA-DA activity. Interestingly, we have found that SUS rats, like some other alcohol-preferring lines, also have notably lower DA content in NAC than do normal and RES rats (Weiss et al., 2008). Therefore, evidence points to VTA-DA activity as mediating the reward produced by alcohol.
A brain region that plays an important role in regulating activity of VTA-DA neurons is the locus coeruleus (LC), site of the major concentration of noradrenergic cell bodies in the brain. Via axonal projections to the ventral tegmentum, LC neurons can both stimulate and inhibit VTA-DA neurons. Low, steady levels of LC activity stimulate VTA-DA neurons by norepinephrine release (NE) onto alpha-1 receptors (Andén & Grabowska, 1976; Donaldson, Dolphin, Jenner, Marsden, & Pycock, 1976; Grenhoff, Nisell, Ferré, Aston-Jones, & Svensson, 1993; Grenhoff & Svensson, 1993), while high rates of LC activity, particularly burst firing of LC, dramatically inhibit VTA-DA firing by causing release of the hyperpolarizing peptide galanin (GAL) from LC terminals in the VTA (Grenhoff et al., 1993). The latter condition may play a significant role in causing depression: we have proposed that elevated LC activity, which is seen in stressed animals and in human depression, brings about many of the behavioral changes in depression through inhibition of VTA-DA neuronal activity as a result of GAL being released from LC terminals in VTA due to excessive LC burst firing (Weiss, Demetrikopoulos, West, & Bonsall, 1996; Weiss, Bonsall, Demetrikopoulos, Emery, & West, 1998; Weiss et al., 2005). This LC-to-VTA-DA neuronal circuit also could be highly relevant to alcohol consumption. Studies have found that ethanol inhibits LC activity (Pohorecky & Brick, 1977; Strahlendorf & Strahlendorf, 1983), and Aston-Jones, Foote, and Bloom (1982) showed that ethanol predominantly inhibits sensory-evoked burst firing of LC neurons. Consistent with this, c-Fos activation of LC neurons was less in alcohol-preferring lines of rats (P, AA) than in non-preferring lines (NP, ANA) in response to high doses of ethanol (Thiele, van Dijk, & Bernstein, 1997), this difference in alcohol-induced LC activation being the one consistent effect these investigators found that distinguished both alcohol-preferring lines they studied from both non-preferring lines. Thus, the propensity of alcohol to reduce LC activity could be important in determining alcohol-mediated reward, through reduction of LC-mediated inhibition of VTA-DA activity.
Based on the foregoing, we tested the following: insofar as high rates of LC activity will inhibit VTA-DA neuronal activity and low rates of LC activity stimulate VTA-DA neurons, reducing LC activity should facilitate alcohol reward and consumption by disinhibiting and/or stimulating VTA-DA neuronal activity to enhance alcohol-derived reward, and increasing LC activity, particularly burst firing, should have the opposite effect on alcohol reward. First, we measured the effect of alcohol, given by i.p. injection, on single-unit electrophysiological activity of LC and then on VTA-DA neurons. After finding that alcohol markedly decreased LC activity and also increased VTA-DA activity in SUS rats, we manipulated LC activity by microinfusion of drugs into LC to determine whether this would affect voluntary alcohol consumption by SUS rats. We then tested whether one could demonstrate that alcohol was rewarding for SUS rats in the conditioned place preference (CPP) test. We note that it has been quite difficult to demonstrate that any rat, even lines of alcohol-preferring rats, experience a rewarding effect of alcohol in the CPP test (Busse, Lawrence, & Riley, 2005; Ciccocioppo, Panocka, Froldi, Quitadamo, & Massi, 1999; Matsuzawa, Suzuki, & Misawa, 1998; Quertemont & De Witte, 2001; Stewart, Murphy, McBride, Lumeng, & Li, 1996); thus, a positive finding in SUS rats would be noteworthy. Lastly, we again manipulated LC activity by microinfusion of drugs into LC to determine if this would alter reward that SUS rats experienced in the CPP test, and also to determine what effects the conditioning experience that rats underwent during the CPP procedure would have on their subsequent voluntary alcohol consumption.
Materials and methods
Animals
Rats were male, albino Sprague-Dawley rats, 3–6 months of age at the time of procedures and testing, and weighing 350–600 grams. Rats were bred and housed in our rat colony on the Briarcliff Campus of Emory University. Rats were from the Swim-test Susceptible (SUS) rat line developed in our laboratory and also from randomly-bred (non-selected, normally-bred) rats that have been maintained in our colony under the same conditions. Details of the selective breeding procedure for the SUS line and also for the Swim-test Resistant (RES) line have been given previously (Scott et al., 1996; West & Weiss, 2005). Briefly, rats were selected for breeding based on the amount of activity (specifically, duration of “struggling” behavior) observed in a swim test immediately after the animals were exposed to an acute stressor. Struggling behavior is vigorous escape-directed behavior in which all four limbs are in motion with the front limbs breaking the surface of the water. SUS rats were chosen for breeding based on their showing a large decrease in struggling after exposure to the stressor compared to nonstressed littermates. The parallel line of RES rats were chosen for breeding based on their showing little or no decrease in struggling after exposure to the same stressor compared to their nonstressed litter-mates. The stressor used initially was a 3.0-h session of electric tail shocks, but beginning in the eighth generation of selective breeding a much milder stressor condition consisting of exposure for 30 min to a novel environment with the presence of 95 dB of white noise was found to be effective in generating behavioral differences. Male and female rats of each generation of these rat lines were tested in this manner and selected for breeding to continue the rat lines.
Rats used in the studies reported here were from generations 43–50 of SUS rats and generations 48–55 of non-selected rats maintained in our colony. From the time of weaning to the beginning of the experiment, animals were housed with two rats of the same line in a standard “shoebox” cage (44 × 23 × 20 cm). Cages were kept in laminar flow racks that have enclosed, sound-attenuating shelves in the colony, with temperature maintained at 21 ± 1 °C and with a 12:12-h light/dark cycle, lights on at 7:00 AM. Body weights of the animals were determined weekly throughout all experiments; body weights did not differ significantly between lines of rats. All procedures used in breeding and testing of animals were in accord with U.S. Department of Agriculture regulations (Laboratory Animal Welfare Acts, P.L. 89-544, as amended P.L. 94-279 and 99-198) and with the “Principles of laboratory animal care” (NIH publication No. 85-23, revised 1985) and have been approved by the Institutional Animal Care and Use Committee of Emory University.
Exposure to alcohol and measurement of alcohol consumption
When exposure to alcohol and measurement of alcohol consumption was begun, animals were moved from pair housing to single housing in standard “shoebox” cages 1 week prior to initiation of any alcohol presentation. At all times, the animals were allowed free access to food and water 24 h/day. When alcohol was presented, a second bottle containing a 10% alcohol solution was placed on the cage; thus, a two-bottle, free-choice procedure was used. The two test fluids were contained in two 500-mL glass bottles fitted with stainless steel sipper tubes with small drinking holes to minimize spillage. Bottles were placed symmetrically on each side of the cage top with the sipper tube protruding through the wire lid, and the positions of the alcohol solution and water were alternated on each successive presentation of alcohol. For the first week, alcohol was presented daily and remained on the cage 24 h per day. Beginning with the second week, the alcohol solution was presented for 5.0 h/day every other day, which was done five times. By the end of this exposure schedule, all animals, and particularly SUS rats, would begin to consume the alcohol solution as soon as it was placed onto the cage. The amount of alcohol solution and water consumed by the animal was determined by weighing the bottles. It is important to note that all animals used in all experiments were exposed to alcohol in this way prior to any of the procedures described below.
Determination of blood alcohol levels
To determine whether the amount of alcohol that SUS rats consumed voluntarily was sufficient to have a pharmacological effect, blood alcohol concentration (BAC) was measured in SUS rats as well as in randomly-bred, normal SD rats for comparison. Before measurement, animals in this study had been individually housed and given the opportunity to consume a 10% alcohol solution for 2 weeks before measurement with the procedure described directly above. On the test day, the 10% alcohol solution was again presented, the amount drunk in 45 min recorded, and a blood sample then taken by tail nick. Blood alcohol was measured using an enzymatic rate method with a Beckman Coulter DXC chemistry analyzer (Gadsden, Taylor, & Steindel, 1986). BAC also was determined by this procedure 45 min after SUS rats had received an i.p. injection of 1.0 g/kg alcohol.
Procedure to increase or decrease LC activity
The procedure to alter specifically LC activity was to infuse into the LC region, through cannulae implanted bilaterally into LC, a small amount of artificial cerebrospinal fluid (aCSF) containing appropriate drug(s) to affect LC activity. For surgery to implant the cannulae, animals maintained under isoflurane anesthesia were mounted in a Kopf stereotaxic instrument. The skull was opened by incision, overlying skin retracted, and two small holes drilled in the skull 1.2 mm on either side of the midline at 3.0 mm posterior to lambda. Two 26-gauge stainless steel guide cannulae held 2.4 mm apart in a plastic mount (Plastics One, Inc., Roanoke, Va.) were lowered through these holes to a depth of 5.5 mm below the top of the brain and the assembly was then affixed to the skull with dental cement. All surgical procedures were conducted under appropriate aseptic conditions and in accord with NIH Guidelines and U.S. Department of Agriculture regulations (Laboratory Animal Welfare Acts, P.L. 89-544, as amended P.L. 94-279 and 99-198). Animals were allowed 2 weeks to recover following surgery before any infusions into LC and subsequent procedures were undertaken. Infusions were administered to awake, freely-moving animals. While the animal was gently held, the cap to which was attached internal “dummy” cannulae was removed and replaced by a cap having two infusion needles attached which were lowered through the 26-gauge guide cannulae; these infusion needles protruded 1.0 mm below the end of the guide cannulae into the brain. Above the cap, the two infusion needles were each attached to 2.5 ft. of PE-10 polyethylene tubing that, at the distal end, were each attached to a separate microsyringe, and both microsyringes were mounted on a hand-held motor-driven microdoser (Delsaga Instruments, Heidelberg, Germany) that was activated to deliver the infusion solution through the cannulae. With the infusion needles and attached tubing in place, the rat was placed into an open-top Plexiglas® box with a 38 × 38-cm floor covered with standard bedding and walls also 38 cm high, and the animal was allowed to move freely about this enclosure. Over a period of 10 min, 3.0 μL of solution was infused slowly through each cannula. To inhibit LC activity, animals were infused with artificial cerebrospinal fluid (aCSF) containing clonidine (Clon) (100 ng/μL, or 300 ng/side of brain), which stimulates inhibitory somatodendritic alpha-2 receptors on LC neurons, and, by so doing, decreases LC activity and burst firing in particular (Simson & Weiss, 1987, 1989). To stimulate LC activity, aCSF containing Substance P (Sub P) and idazoxan (Ida) (100 ng/μL of each drug, or 300 ng of each drug/side of brain) was infused. Sub P is a profound activator of LC activity, and Ida, opposite to the effect of Clon, blocks alpha–2 receptors, thereby facilitating LC burst firing. Based on previous studies, infusion of the drugs and doses described above has been shown, by measurement of noradrenergic metabolites in LC target regions, to selectively decrease and increase, respectively, LC activity (Weiss et al., 1994). For control animals in which LC activity was not decreased or increased, aCSF containing no drug was infused. Following the 10-min infusion procedure, animals were removed from the Plexiglas® box, infusion needles removed from guide cannulae, and the cap replaced so that the internal “dummy” cannulae were placed back into the guides. The animals then began the next stage of the procedure, as described below.
Measurement of alcohol consumption after LC activity has been increased or decreased
To determine the effect that an increase or decrease in LC activity would have on alcohol consumption of SUS rats, immediately following an infusion into LC as described above animals were returned to their home cage in the colony room, and at that time a bottle containing a 10% alcohol solution as well as a second bottle containing normal tap water was present on the home cage. The amount of alcohol solution and water consumed during the following hour was quantified. Each animal included in this study had alcohol consumption measured after infusions to both increase and decrease LC activity as well as vehicle infusions for comparison; details regarding the sequence and schedule of infusions are given in the legend for Fig. 4.
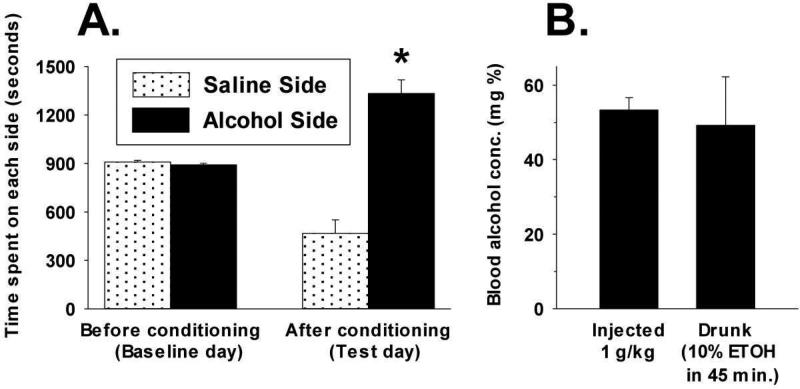
Fig. 4.
A (left). Conditioned Place Preference (CPP) of SUS rats produced by injections of alcohol (1.0 g/kg) versus saline shown by time (seconds) spent during a 30-min test session on either the alcohol-paired side or the saline-paired side of the CPP test chamber. Means and standard errors are shown. n = 6. * = significant difference (p < 0.002) in time spent on alcohol-associated side of CPP apparatus before vs. after conditioning. B (right). Blood Alcohol Concentration (BAC) found in SUS rats after voluntary consumption of a 10% alcohol solution and after injection of 1.0 g/kg alcohol as used in CPP conditioning. Blood samples were taken 45 min after onset of ethanol presentation for drinking and after i.p. injection. Means and standard errors are shown. n = 6 (same rats as in A).
Conditioned place preference (CPP)
The initial procedure used to determine if rewarding consequences of alcohol could be demonstrated for SUS rats by using the CPP test was as follows: rats were habituated to the CPP apparatus, which was done by allowing the animals to explore the apparatus freely for 30 min on three consecutive days. The CPP apparatus was made up of two distinctly different chambers separated by an opening in the center wall, each chamber 48 × 48 cm in size. Each of the chambers differed in (a) black wall vs. white walls, (b) floor covered with wood shaving bedding vs. corncob bedding, and (c) bedding impregnated with distinctly different odors, d-limonene vs. clove oil. On the third day of habitation, the amount of time that each rat chose to spend in each of two chambers was measured; this time served as a rat's pre-conditioning baseline. On each of three days thereafter, rats received an injection of physiological saline (0.85% saline) and then were confined on one side of the chamber for 30 min, and 4 h later on the same day received an injection of alcohol (1.0 g/kg) and then were confined on the other side of the chamber for 30 min (for half the animals, substances injected in the reverse order; i.e., counterbalanced). After the third day of such conditioning, no treatment was given on the next day, and on the following day, the CPP test was conducted. On the test day, the rats were given no injections but simply returned to the apparatus (initially placed near the opening between the sides in the saline-paired side) and allowed to move freely between the two chambers of the apparatus for 30 min. Time spent in each chamber of the apparatus on this test day was quantified.
Conditioned place preference (CPP) when LC activity is either increased or decreased
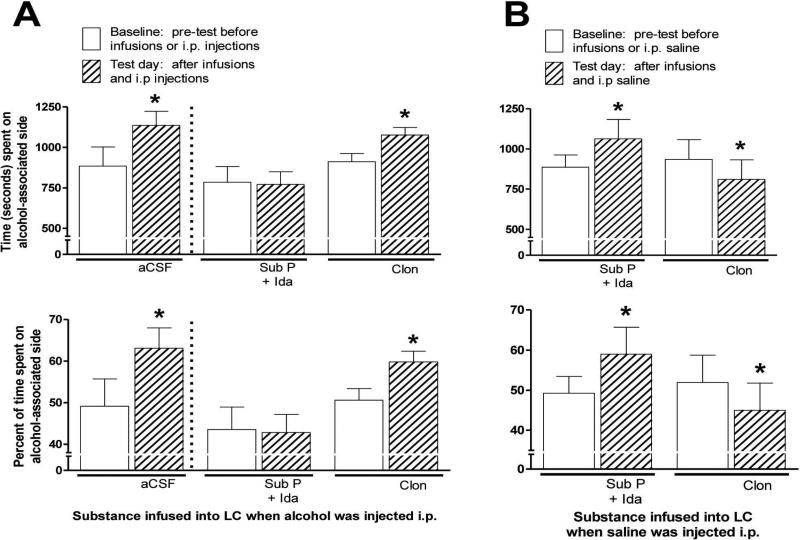
In this study, the CPP test was conducted in a similar manner as described above except that conditioning (i.e., i.p. injection followed by confinement on one side of the apparatus) was done only once per day and was done after animals had been infused with drug to either increase or decrease LC activity or, for comparison, with vehicle (aCSF) to produce no change in LC activity. As described above, SUS rats in this study underwent surgery to implant bilateral cannulae aimed at LC. Following recovery 2 weeks later, the rats were familiarized with the CPP apparatus and then baseline time spent on the two sides of the apparatus was determined. Following this, animals were exposed to the CPP apparatus six times at intervals of 4–5 days between each exposure. As described above, on three of these six exposures, animals were injected i.p. with alcohol and then were confined for 30 min on one distinctive side of the CPP apparatus, and on the other three occasions were injected i.p. with physiological saline and then confined for 30 min on the other distinctive side. On the three occasions when alcohol was injected i.p., Sub P + Ida was infused into some animals to increase LC activity before alcohol was injected. Clon was infused into other animals to decrease LC activity, and vehicle was infused into other animals to not alter LC activity. On the three occasions when animals had saline injected i.p. and the animals were then confined on the distinctive saline-associated side of the CPP apparatus, all animals were infused with artificial CSF vehicle. Thus, when alcohol was injected and animals were subsequently confined on the alcohol-associated side of the CPP apparatus, LC activity was either increased, decreased, or not changed, and effects of these manipulations were to be compared with saline injection and confinement on the saline-associated side of the CPP apparatus with no manipulation of LC activity (i.e., aCSF infused). After the six infusion/i.p. injection sessions were completed, 5 days later animals were given the test session in the CPP as described above; i.e., animals were allowed to freely explore the CPP apparatus for 30 min, during which the time spent on both sides of the CPP apparatus was recorded.
To interpret the findings of the study described directly above, it was important to determine if manipulation of LC activity by itself had any rewarding or aversive effects that could influence responding in the CPP test. To assess this, an ancillary study was conducted in which additional SUS rats underwent exactly the same procedure as described directly above for the infusion/i.p. injection process, except that all i.p. injections were saline (i.e., no alcohol after any infusion). In this study, on three occasions Sub P + Ida was infused into some animals to increase LC activity, while Clon was infused into other animals to decrease LC activity, after which all animals were given an i.p. injection of saline and confined on one distinctive side of the apparatus. Thus, in contrast to the procedure described directly above, when LC activity was altered alcohol played no role in conditioning. On the three other occasions when these animals were infused with aCSF into LC to have no effect on LC activity, they were then also injected i.p. with saline and confined on the other distinctive side of the CPP apparatus. Consequently, performance of these animals on the test day in the CPP of this ancillary study was exclusively the result of their LC activity having been either increased or decreased.
Electrophysiological recording
Anesthesia, electrodes, recording equipment, and stereotaxic procedure
Animals were anesthetized with chloral hydrate (400 mg/kg i.p.) and given supplemental doses of anesthetic as needed. After induction of anesthesia, animals were mounted in a Kopf stereotaxic instrument. Body temperature was monitored and maintained at near 37 °C by a water-circulating heating pad. The skull was opened over the appropriate brain area for unit recording and the dura incised and retracted. The brain surface was kept moist throughout the recording session with warm saline solution. Micropipette glass electrodes used were pulled from 1.5-mm capillary tubing, filled with 3.6% NaCl, and were used for recording. Micropipette tips were broken back to 2–3 μm (2–7 MΩ impedance measured at 135 Hz). The electrical signal from the electrode was fed into a preamplifier (Fintronics model WDR-420), the filtered output from the preamplifier fed into an oscilloscope and audio monitor and into a computer for analysis, display, and storage using a commercial program (Experimenter's Workbench, DataWave Technologies).
Recording of LC neuronal activity
The glass micropipette was lowered into the brain in the region of the LC until single-unit activity was detected. Stereotaxic coordinates for the LC region were given previously under Procedure to increase or decrease LC activity. Recording of LC neurons was verified by criteria described in various references (Aston-Jones & Bloom, 1981; Borsody & Weiss, 1996; Foote, Aston-Jones, & Bloom, 1980; Graham & Aghajanian, 1971; Korf, Bunney, & Aghajanian, 1974; Simson & Weiss, 1987); these criteria were: (a) a long-duration action potential with a positive-negative waveform and a notch on the ascending limb, (b) a spontaneous firing pattern of 0.5 to 3.5 Hz, and (c) elicitation of a rapidly-occurring succession of spikes (“burst” firing) followed by a period of quiescence (post-stimulus inhibition) in response to application of a salient sensory stimulus (in this case, compression of the contralateral hind paw). When a single-unit of stable amplitude demonstrating these characteristics was isolated, spontaneous rate of firing was recorded for 4 min, with activity during the last 3 min used to determine the spontaneous firing rate for that unit. The magnitude of the sensory-evoked “burst” response of the neuron was then measured. For this procedure, the paw was compressed (PC) for 1.0 sec between the ends of a pair of 13.0-cm surgical forceps (Malony, curved end, Jarit Surgical Instrument Co.) by applying pressure midway along the forceps such that the opposite sides of the forceps, at this midpoint, came into contact. The magnitude of the “burst” response (i.e., the number of spikes in a “burst”) has been shown to be little affected by the intensity of the PC; rather, a “burst” will occur when the intensity of the PC exceeds the threshold needed to elicit a “burst” response, with the number of spikes in a burst then determined by factors unrelated to PC intensity such as activation of receptors on the LC neuron, resting potential of the cell, etc. (see Simson & Weiss, 1989). In addition, application of successive PCs has not been observed to produce habituation or sensitization. To determine the amount of sensory-evoked “burst” firing by a unit, 10 PCs were applied, with each PC spaced at least 10 sec apart. Spikes occurring in the first 0.5 sec of the PC were counted; this was done because almost all sensory-evoked “burst” firing for both drug-treated and vehicle-treated animals took place within the first 0.5 sec of the PC. The amount of sensory-evoked “burst” firing of a unit was established by using the average of the 10 elicited “bursts”. Following the last of the 10 PC applications, spontaneous activity was recorded for 1 min (to verify that the cell returned to its normal baseline firing rate), after which this entire process (recording of spontaneous activity and “burst firing” as described above) was repeated with doses of alcohol being given as described below.
Determining response of LC neurons to alcohol
Alcohol administration was done as follows: after the initial determination of spontaneous firing rate and burst firing for a unit, animals were injected i.p. with alcohol. The pre-alcohol activity recording value is shown in the figures at the “0.0” alcohol dose. All subsequent determinations were done while recording from that unit. Alcohol (20% solution in physiological saline) was given i.p. to achieve an initial alcohol dose of 0.5 g/kg, after which, beginning 2 min later, recording from that unit was done as described above. Five minutes after conclusion of this recording procedure, an equal amount of alcohol as injected initially was again given, making the cumulative dose 1.0 g/kg, and recording was again conducted. This sequence was repeated twice more, thus obtaining final recordings at cumulative doses of 1.5 and 2.0 g/kg.
Recording of VTA-DA neuronal activity
Procedures for single-unit recording of dopaminergic neurons in the ventral tegmentum given here follow those used by several other major laboratories in this field, as noted below. The electrode was lowered into the brain at the following stereotaxic coordinates for VTA: posterior to bregma 5.0–6.0 mm, lateral 0.6–1.2 mm, ventral from cortical surface 7.5–9.5 mm. Dopaminergic neurons in the VTA were distinguished from non dopaminergic neurons in this brain region using criteria employed by laboratories with recognized expertise in identification of DA neuronal extracellular recording (Bunney, Walters, Roth, & Aghajanian, 1973; Chiodo, 1988; Grace & Bunney, 1980, 1983; Guyenet & Aghajanian, 1978; Wang, 1981; White & Wang, 1983). These criteria for identifying putative VTA-DA neurons are: (1) bi- or tri-phasic waveform (positive first), (2) long-duration action potential of 2.5–5.0 msec, (3) an IS-SD break notch on the ascending phase of the action potential, and (4) spontaneous firing rates of 0.1– 10.0 spikes/sec. We used a lower limit to the firing rate criterion (i.e., 0.1 spikes/sec) than is typically reported (i.e., 0.5–1.0 spikes/sec) because we encountered some very slow firing neurons with quite distinct dopaminergic neuronal characteristics except for a low spontaneous rate (although such slow-firing units were rare). Prior to recording of any units included in the data reported here, recording was done from several animals, with VTA-DA neurons identified as explained above, and the recording site was then dye-marked. The brains of these animals were sectioned to establish that recordings were being done exclusively from the VTA region, which was confirmed by this procedure.
VTA-DA neuronal activity was quantified as follows: the recording electrode was lowered into the brain and VTA-DA units were identified using the criteria described above. After isolation and stabilization of recording of a VTA-DA unit, baseline spontaneous activity was recorded for 4 min, the last 2 min of which will be used for collection of spontaneous firing measurements. In addition to spontaneous firing rate, the percentage of spikes occurring as a “burst” was also quantified; most, but not all, VTA-DA neurons show a bursting firing pattern with no other stimulus required. The onset of a burst was defined as an inter-spike interval (ISI) of less than 80 msec, and the termination of a burst is defined as an ISI of longer than 160 msec (Grace & Bunney, 1984). The percentage of burst firing was determined by expressing the number of spikes occurring within bursts during a time period as a percentage of the total number of spikes occurring in that time period.
Determining response of VTA-DA neurons to alcohol
Response to alcohol of VTA-DA neurons was determined in exactly the same way as described above for LC units. When VTA-DA activity in response to alcohol was determined in conjunction with LC activity being increased by infusion of drug into the LC region, the following procedure was added initially: a cannula was lowered into the LC region at the beginning of the recording session, and, after obtaining a stable firing rate for a dopaminergic unit in the VTA region ipsilateral to the LC cannula placement, a microinfusion into LC of Sub P + Ida (10-min infusion, 3.0 μL, 300 ng of each drug dissolved in aCSF as described earlier) was given. Five minutes later, the baseline-firing rate of the VTA-DA unit was again determined (0.0 dose point) and the procedure for administration of alcohol and recording then proceeded as described above. In this study, other animals were also infused similarly into LC with aCSF (3 μL) for the comparison condition in which LC activity was not altered, and the response of VTA-DA neurons to alcohol then determined similarly.
Data analysis
Statistical analysis was conducted with SPSS version 20 as follows: When single-unit electrophysiological activity of LC and VTA units in response to increasing doses of alcohol was analyzed, repeated measures analysis of covariance was used, with the value obtained before alcohol injection (0.0 dose) being the covariate for each animal. A similar repeated-measures analysis of covariance was used on the post-CPP alcohol consumption test data, with consumption of each animal prior to the CPP procedure being the covariate. All other comparisons were done by paired t tests (when the same animal could be compared before and after a treatment) or by independent t tests (when animals in different conditions were compared).
Results
Effects of alcohol on electrophysiological activity (spontaneous and burst firing) of LC and VTA-DA neuron
The hypothesis being examined states that high rates of LC activity, particularly burst firing of LC neurons, inhibit VTA-DA neuronal activity, and, consequently, decreases in LC activity would allow firing of VTA-DA neurons to increase readily. It has been reported that alcohol reduces burst firing of LC neurons (Aston-Jones et al., 1982). The first experiment therefore measured single-unit electrophysiological activity of LC neurons to determine how LC neurons respond to alcohol in SUS rats as compared to randomly-bred normal Sprague-Dawley (SD) rats.
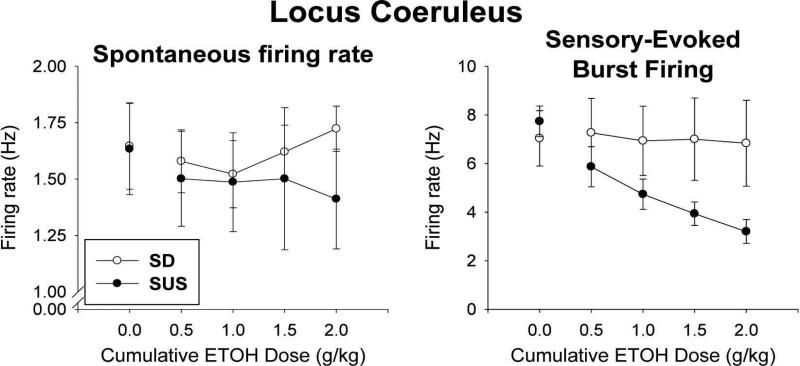
The salient result, shown in Fig. 1, was that burst firing of LC neurons (see right side of Fig. 1) was markedly decreased in response to alcohol in SUS rats but did not change in randomly-bred, normal SD rats. With regard to spontaneous firing rate (shown at left of Fig. 1), increasing doses of alcohol had little effect on this measure in either group of rats. An analysis of covariance (covariate was the baseline [pre-alcohol] rate at “0.0” dose point) revealed no significant effect of rat type (or group) (F[1,9] = .61, p = .45) and alcohol dose (F[3,27] = .26, p = .851), although the rat type × dose interaction approached significance, because a low spontaneous firing rate was seen in SUS rats consistently across the alcohol doses (F[3,27] = 2.55, p = .076). In contrast to spontaneous firing rate, sensory-evoked burst firing of LC neurons was affected by alcohol quite differently in the two rat lines. In randomly-bred, normal SD rats, burst firing was hardly affected by increasing doses of alcohol, but in SUS rats LC burst firing decreased markedly and progressively with increasing doses of alcohol. An analysis of covariance (covariate was baseline at 0.0 dose level) revealed a significant difference between rat type (F[1,9] = 9.03, p <. 02) and a highly significant rat type × alcohol dose interaction (F[3,27] = 8.38, p < .001). Because of the large effect of different doses of alcohol in SUS rats, the dose factor was also significant overall (F[3,27] = 5.47, p < .005). Thus, while burst firing of LC neurons was little affected in normal rats, SUS rats showed a large decrease in LC burst firing in response to alcohol.
Fig. 1.
Firing activity of locus coeruleus (LC) neurons in SUS and randomly-bred normal SD rats in response to alcohol injected intraperitoneally (i.p.). Single-unit electrophysiological activity (spikes per second [Hz]) of LC neurons (means and standard errors) is shown. Following determination of baseline activity (0.0 dose), the effect of increasing (cumulative) doses of i.p.-injected alcohol was assessed on Spontaneous firing rate (Left) and Sensory-evoked burst firing (Right). Number of rats/group = 6, one unit per rat.
The effect of alcohol on single-unit electrophysiological activity of VTA-DA neurons was then measured. The hypothesis being tested here proposes that, based on inhibition of the LC burst firing caused by alcohol in SUS rats, alcohol could then produce a large excitatory response in VTA-DA neurons of SUS rats.
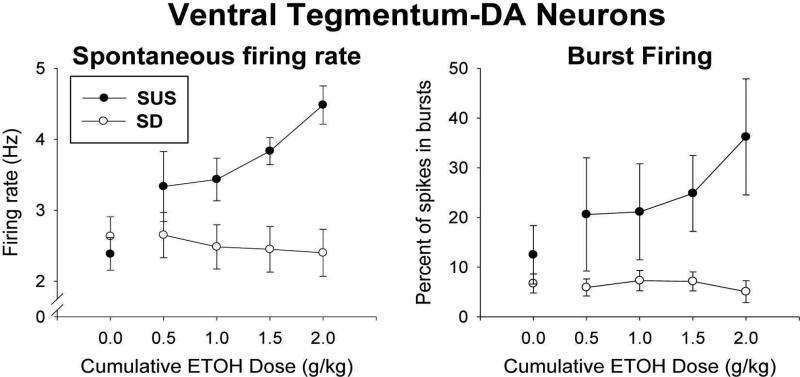
Fig. 2 shows that both spontaneous firing rate and burst firing increased markedly in response to alcohol in SUS rats but did not change in randomly-bred, normal SD rats. For spontaneous firing rate of VTA-DA neurons (shown at left of Fig. 2), an analysis of covariance (covariate was the baseline [pre-alcohol] rate at “0.0” dose point) revealed a highly significant difference between rat type (F[1,9] = 15.45, p = .003), and a significant rat type × alcohol dose interaction (F[3,27] = 6.77, p < .002). The alcohol dose factor was not significant (F[3,27] = .90, p = .455). Similar effects were seen in regard to VTA-DA burst firing (shown at right of Fig. 2), which, for VTA-DA neurons, is expressed as the percent of spikes that occur within a burst. For burst firing, an analysis of covariance (covariate was baseline at 0.0 dose) yielded a significant difference between rat type (F[1,9] = 6.99, p < .03), and a significant rat type × alcohol dose interaction (F[3,27] = 3.25, p < .04). The dose factor also was significant overall (F[3,27] = 3.38, p < .04). Thus, both spontaneous firing rate and burst firing of VTA-DA neurons were markedly elevated by increasing doses of alcohol in SUS rats, while both measures were essentially unaffected by alcohol in randomly-bred, normal SD rats.
Fig. 2.
Firing activity of dopaminergic neurons in the ventral tegmental area (VTA-DA neurons) in SUS and randomly-bred normal SD rats in response to alcohol injected intraperitoneally (i.p.). Single-unit electrophysiological activity (spikes per second [Hz]) of VTA-DA neurons (means and standard errors) is shown. Following determination of baseline activity (0.0 dose), the effect of increasing (cumulative) doses of i.p.-injected alcohol was assessed on Spontaneous firing rate (Left) and percentage of spikes occurring within bursts (Right). Number of rats/group = 6; one unit per rat.
Effects of manipulating LC activity on intake of alcohol by SUS rats
Having found that alcohol affected LC and VTA-DA neuronal firing in SUS rats as predicted, it was then determined if experimentally manipulating LC activity, both to increase or decrease LC activity, would affect voluntary alcohol intake of SUS rats. Through implanted bilateral cannulae aimed at the LC, drugs were infused with a 10-min procedure into the LC region to either increase or decrease LC activity of SUS rats, and then alcohol consumption was measured during the next 60 min.
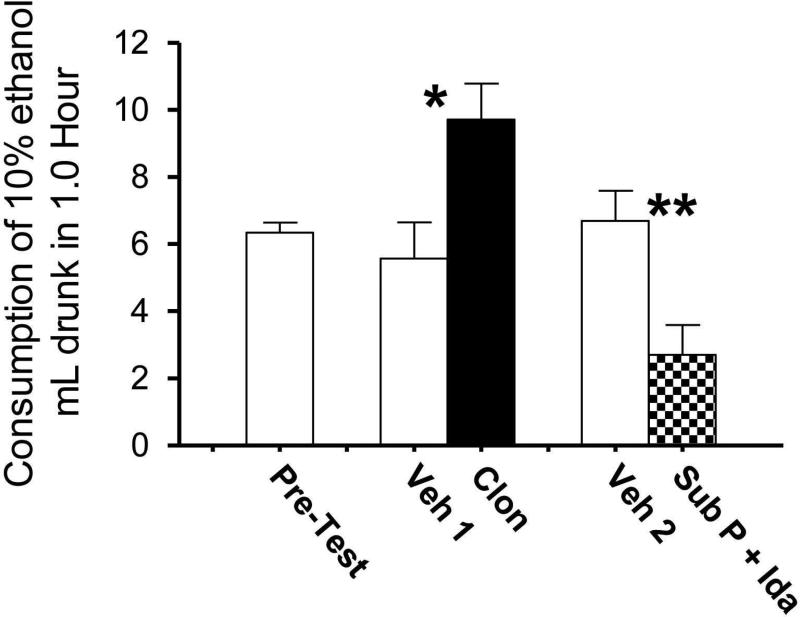
Fig. 3 shows consumption of 10% ethanol solution by SUS rats at 1.0 h after infusion. All animals in the study (n = 7) received vehicle (aCSF) infusions as well as infusions to both decrease or increase LC activity. The bar at the far left shows the consumption of all animals when no infusion procedure had been given (Pre-test). The bars at the middle and right of the figure show alcohol consumption in each of 2 weeks following the Pre-test; in each week, animals received an alcohol consumption test following infusion of vehicle (aCSF) and following infusion of drug (see figure legend for details). After animals received Clon to decrease LC activity, their alcohol intake increased significantly relative to what they consumed after vehicle (paired t = 3.0, df = 6, p < .004), and after infusion of Sub P + Ida to increase LC activity, their alcohol intake decreased significantly relative to vehicle (paired t = 3.6, df = 6, p < .002). Plain tap water was available in a second bottle on cages throughout all testing sessions, but consumption of water in the 1.0-h test period was negligible and did not differ in any infusion condition. The results therefore showed that experimentally decreasing LC activity led to an increase in voluntary alcohol intake by SUS rats, and that experimentally increasing LC activity led to a decrease in their alcohol intake.
Fig. 3.
Consumption of 10% ethanol solution (g/kg) in 1.0 h by SUS rats after no procedure (Pre-test) or after infusion of artificial CSF vehicle (Veh) and drugs to either decrease LC activity (clonidine [Clon]) or increase LC activity (substance P + Idazoxan [Sub P + Ida]). Means and standard errors are shown. The same rats (n = 7) underwent all phases of this study. In the week following the pre-test measure, all animals received two infusions, one of vehicle (Veh 1) and the other of Clon to decrease LC activity, with the infusions given 4 days apart (with consumption test after each infusion); three rats received vehicle first and then Clon and four rats received infusions in the reverse order. In the following week, all animals again received two infusions, one of vehicle (Veh 2) and the other of Sub P + Ida to increase LC activity, with infusions again 4 days apart; again, three rats received vehicle first and then drug while four rats received infusions in reverse order. * = Clon differs from Veh 1, p < .005; ** = Sub P + Ida differs from Veh 2, p < .003.
Assessment of rewarding effects of alcohol in SUS rats
A. Examining blood alcohol levels after voluntary alcohol intake
Addressing the possibility that SUS rats found the consequences of alcohol intake to be rewarding, it was first determined if SUS rats consumed a sufficient amount of alcohol to have a pharmacological effect. For this, blood alcohol concentration (BAC) was measured after SUS rats had voluntarily consumed alcohol. Using SUS rats and randomly-bred, normal SD rats that had equal exposure to alcohol prior to the test occasion (see Procedure), animals were given access to 10% alcohol solution for 45 min and then a blood sample was taken by tail nick. SUS rats (n = 12) consumed an average of 6.8 ± 0.7 mL of alcohol solution during the 45-min test period, significantly more than SD rats (n = 4), which consumed an average of 2.0 ± 0.3 mL of the alcohol solution (independent groups t = 6.8, df = 14, p < .001). This represented an average consumption by SUS rats of 2.0 ± 0.2 g/kg of alcohol solution in the 45-min period vs. 0.6 ± 0.1 g/kg of alcohol solution by the SD rats. Analysis of BAC revealed that SUS rats had a mean (± S.E.) blood alcohol level at the end of the 45-min test period of 54.2 ± 7.4 mg%, while this value for the normal SD rats was 6.8 ± 1.2 mg% (independent groups t = 7.3, df = 14, p < .001). These BAC levels in SUS rats are comparable to what is found in rat lines specifically bred for alcohol preference when these rats voluntarily consume alcohol (see review by Rodd, Bell, Sable, Murphy, & McBride, 2004) and are sufficient to have pharmacological effects (e.g., Gill, France, & Amit, 1986; Waller, Murphy, McBride, Lumeng, & Li, 1986).
B. Conditioned place preference (CPP) based on alcohol
It was then determined whether SUS rats experienced reward from alcohol (given by i.p. injection) as reflected behaviorally in the CPP test. The findings are shown in Part A of Fig. 4 (left side). Time that rats spent on the “saline” side and on the “alcohol” side before and after conditioning is shown. While the animals showed no preference for either side of the apparatus prior to any injections (i.e., Before conditioning, Baseline day), on the test day (i.e., After conditioning, Test day) all SUS rats (n = 6) spent considerably more time on the side that had been associated with injection of alcohol than they did on the side that had been associated with injection of saline. The difference in time spent on the alcohol side before conditioning vs. after conditioning on the test day was statistically significant (paired t = 5.7, df = 5, p < .002). Thus, SUS rats clearly showed a “place preference” for the location where they had received alcohol.
In Part B of Fig. 4 (right side) are shown, in the same rats that underwent the CPP procedure, results from determining blood alcohol concentration (BAC) after i.p. injection of the amount of alcohol given in that procedure (i.e., 1.0 mg/kg) and after voluntary alcohol consumption by the same rats. BACs found in the six rats used in this study 45 min after they were injected with 1.0 g/kg alcohol was a mean of 53.3 ± 3.3 mg%. This BAC was not significantly different from, and in fact quite similar to, the blood alcohol level measured in the same six rats at the end of a 45-min period of voluntary consumption of the 10% alcohol solution (49.2 ± 13.0 mg%; paired t = 0.13, df = 5, p = .74 two tailed), which had been measured 2 weeks before measurement after i.p. injection. Thus, the rewarding effect of alcohol shown in Fig. 4 (Part A) occurred when animals were injected with an amount of alcohol that resulted in a BAC level similar to what these same SUS rats achieve when they voluntarily consumed alcohol.
C. Conditioned place preference (CPP) based on alcohol when LC activity is increased or decreased
It was then determined whether reward from alcohol as manifested in the CPP test would be altered if LC activity was either experimentally increased or decreased. Findings are shown in Part A (left side) of Fig. 5. When animals were infused with Sub P + Ida into LC to increase LC activity prior to i.p. alcohol injections, this completely blocked the rewarding effect of alcohol, evidenced by no increase in time spent on the side associated with alcohol. This result should be compared to the increase in time spent on the alcohol-related side in animals whose LC activity was not increased prior to i.p. alcohol (i.e., in aCSF-infused control animals, shown at the far left of Part A). Animals infused with Clon to decrease LC activity prior to alcohol injection showed a distinct preference for the side where alcohol was injected, but the magnitude of this side preference was similar to that seen in aCSF-infused animals; thus, Clon infusion did not increase the preference for, or reward of, alcohol shown in the CPP test. For results of statistical analysis, see Fig. 5 legend.
Fig. 5.
A (Left). Conditioned Place Preference (CPP) of SUS rats produced by i.p. injection of alcohol (1.0 g/kg) versus saline shown by animals that had been infused into locus coeruleus (LC) with vehicle (aCSF) (n = 6) or Substance P + Idazoxan (Sub P + Ida) to increase LC activity (n = 6) or clonidine (Clon) to decrease LC activity (n = 7) immediately before the injections of alcohol in this procedure. Shown is time spent during the 30-min test session on the alcohol-paired side of two-compartment CPP apparatus; at top is shown time (in seconds) spent on the alcohol-paired side in the test session, and at bottom is shown the percentage of total time in CPP apparatus during the test session spent on the alcohol-paired side. Means and standard errors are shown. “Baseline” shows time spent on what became the alcohol-paired side of the CPP apparatus before any injection of alcohol or any other procedure; “Test day” shows time spent on that side after i.p. injections of alcohol followed by confinement on that distinctive side of the CPP apparatus and after an equal number of injections of i.p. physiological saline (0.85% saline) and confinement on the other distinctive side of the CPP apparatus (i.e., post conditioning). Statistics: * = significantly higher than the “Baseline” for these same animals (atleast p < .02; by paired t tests), and also significantly higher than Sub P + Ida-infused rats on “Test day” (at least p < .005; by independent groups t tests). B (Right). Conditioned Place Preference (CPP) of SUS rats with details exactly as above except that all i.p. injections were physiological saline (i.e., no alcohol given). Thus, animals were infused into locus coeruleus (LC) with Sub P + Ida to increase LC activity (n = 6) or Clon to decrease LC activity (n = 7) followed by i.p. saline, and consequently this CPP procedure determined preference for, or aversion of, the side of the CPP apparatus paired with LC activity having been either increased (vs. no change in LC activity by aCSF infusion) or decreased (vs. no change in LC activity by aCSF infusion). Statistics: * = significantly different (higher or lower) than the “Baseline” for these same animals (at least p < .05 by paired t tests).
However, an important aspect of this CPP study was to assess the effect of the drug infusions into LC by themselves on side preference in the CPP; i.e., to assess how side preference in the CPP would be affected simply by altering LC activity when no alcohol is injected. The importance of doing this can be seen by the following: the salient finding described in the preceding paragraph was that increasing LC activity (by infusing Sub P + Ida) was able to block the rewarding effect of alcohol; i.e., when Sub P + Ida was given into LC, this clearly blocked the preference that rats usually show for the side of the CPP where alcohol is injected i.p. However, if the effect produced by infusing Sub P + Ida into LC was itself aversive, the conclusion that this manipulation blocked the rewarding effect of alcohol could well be invalid, because decreased preference for the alcohol-associated side could have been simply due to aversion from infusion of Sub P + Ida being associated with that side of the CPP. Part B (right side) of Fig. 5 shows the effect of infusing either Sub P + Ida or Clon into LC with no alcohol injection afterward (saline given i.p. instead). The findings of this ancillary study were directly opposite to any finding that would compromise the conclusion made in the first paragraph above. When infused alone into LC, Sub P + Ida caused animals to choose that side of the CPP apparatus, while Clon caused animals to avoid that side, i.e., the animals’ behavior indicates that having the LC activated is rewarding, and, conversely, that having the LC inhibited has aversive consequences. Consequently, blocking of alcohol reward by increasing LC activity was seen in the CPP test despite the fact that increasing LC activity is, by itself, rewarding to rats, and, conversely, sustained alcohol reward was seen in the CPP test despite the fact that decreased LC activity is somewhat aversive to rats. Results of statistical analysis are shown in Fig. 5 legend.
D. Assessment of electrophysiological activity of VTA-DA neuronal response to alcohol when activity of LC is increased
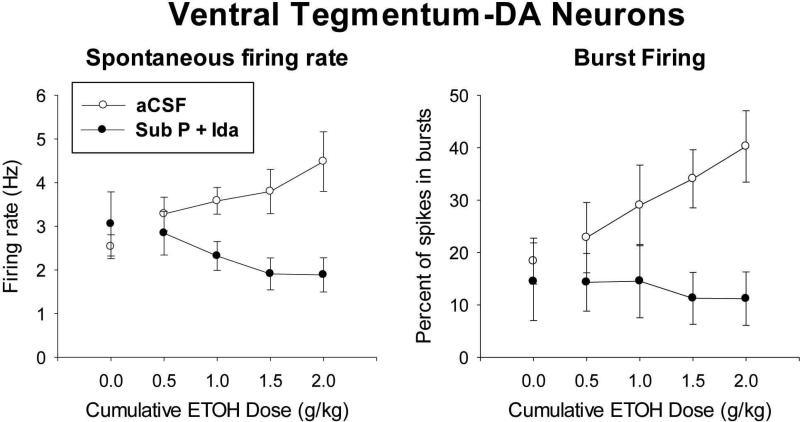
Insofar as infusion of Sub P + Ida into LC to increase LC activity blocked the rewarding effect of i.p. alcohol in the CPP test, it was of interest to determine if this manipulation might have done so by blocking the increase in VTA-DA activity that alcohol usually produces in SUS rats. Therefore, single-unit electrophysiological activity was recorded from VTA-DA neurons of the VTA ipsilateral to where these animals received a unilateral infusion of Sub P + Ida into LC. The results, shown in Fig. 6, make it clear that infusion of Sub P + Ida to increase LC activity completely blocked the increase in VTA-DA neuronal firing that normally results from alcohol in SUS rats. These results show that the change in LC activity was responsible for the change in the response of VTA-DA neurons to alcohol.
Fig. 6.
Firing activity of dopaminergic neurons in the ventral tegmental area (VTA-DA neurons) in response to alcohol injected intraperitoneally (i.p.) in SUS rats infused into LC with Substance P + Idazoxan (Sub P + Ida) to increase LC activity (n = 6) or aCSF to have no effect on LC activity (n = 9). Single-unit electrophysiological activity (spikes per second [Hz]) of VTA-DA neurons (means and standard errors) is shown. Recording was done in the VTA on the side of brain ipsilateral to where unilateral infusion into LC was done. Following infusion of aCSF or drug, baseline activity was determined (0.0 dose), and then the effect of increasing (cumulative) doses of i.p.-injected alcohol was assessed on Spontaneous firing rate (Left) and percentage of spikes occurring within bursts (Right). Statistics: Analysis of both spontaneous firing rate and burst firing was done by 2-way (Group × alcohol dose) repeated-measures analysis of covariance (repeated measure for each subject across alcohol doses; covariate was the first measure after infusion shown at 0.0 alcohol dose). For spontaneous firing rate, Group factor (i.e., aCSF vs. Sub P + Ida) was significantly different (F[1,12] = 5.92, p < .032) as was the interaction (Group × alcohol dose) (F[3,36] = 2.93, p < .047). For burst firing, the Group factor was also significantly different (F[1,12] = 10.59, p < .007) as was the interaction (Group × alcohol dose) (F[3,36] = 3.84, p < .017). The alcohol dose factor was also significant (F[3,36] = 1.77, p < .043) in analysis of burst firing.
E. Voluntary alcohol consumption after animals had received alcohol together with their LC activity being increased or decreased
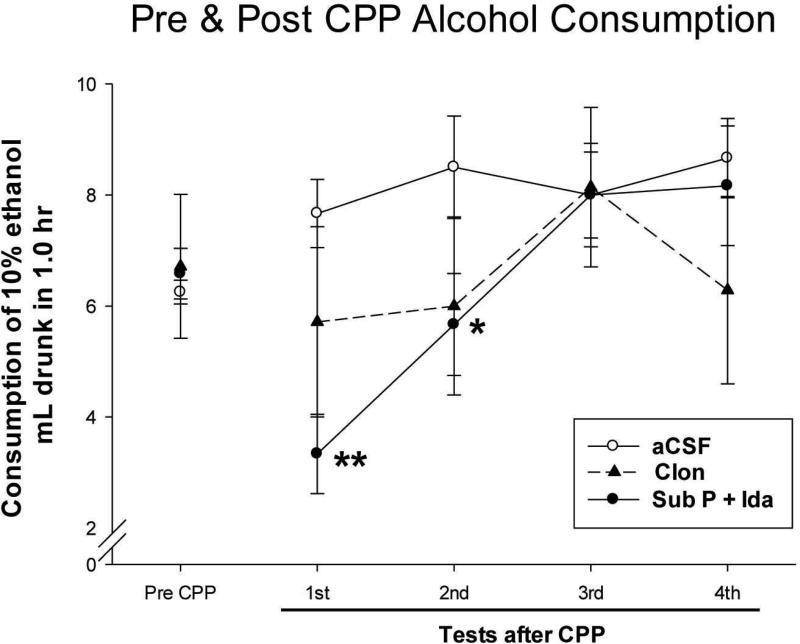
After completion of CPP testing in which alcohol had been given i.p. in conjunction with LC activity being either increased or decreased (results described above), voluntary consumption of alcohol in the home cage by animals that had undergone this procedure was determined. Alcohol was presented for 1 h in the home cage (two-bottle testing; 10% alcohol in one bottle and tap water in the other) four times, first done 2 weeks after the CPP test had concluded and then repeated three more times with a 2-week interval between each presentation. The amount of alcohol consumed is shown in Fig. 7. As would have been expected, animals whose LC activity had not been altered because they received “control” aCSF infusions into the LC region showed similar elevated alcohol ingestion in the home cage, typical of SUS rats, as they had shown prior to any infusion, and did so on all four assessments. In contrast to this, animals whose LC activity had been increased (i.e., Sub P + Ida infused into LC) when alcohol was injected i.p. for the CPP procedure showed a marked and significant reduction in alcohol consumption on the first subsequent presentation of alcohol in the home cage, and this reduction persisted, though somewhat diminished, on the second presentation 2 weeks later (see Figure legend for statistical results). By the third presentation, their voluntary alcohol consumption returned to its normal “pre-conditioning” level. For animals whose LC activity had been decreased in conjunction with i.p. alcohol (i.e., Clon infused into LC), their consumption was somewhat reduced, but not significantly so, on three of the four alcohol presentations. These observations are confirmed by a repeated-measures analysis of covariance (repeated measures over tests after CPP; consumption amount prior to the CPP procedure as the covariate), which showed a significant Infusion Group × Test interaction (F[6,45] = 3.60, p < .005), while neither the Infusion Group effect (F[2,15] = 2.51, p = .112) nor the Test effect (F[3,45] = 1.39, p = .258) was significant. Following finding of a significant interaction, contrasts of the Infusion Groups on each test day were carried out and indicated individual group differences described above. These results thus reveal that animals that were given alcohol i.p. while at the same time having their LC activity increased show classically-conditioned effects of the LC manipulation related to alcohol, which then influences the amount of alcohol that the animals will subsequently consume.
Fig. 7.
Consumption of 10% alcohol solution (g/kg) in 1.0 h by SUS rats prior to their undergoing the Conditioned Place Preference (CPP) procedure (Pre CPP) and on four consumption tests at 2-week intervals after these animals had undergone the CPP conditioning procedure (Tests after CPP). Means and standard errors are shown. During the CPP procedure, all animals had received bilateral infusions into LC immediately before alcohol was injected i.p. (1.0 g/kg), some animals receiving artificial CSF to have no effect on LC activity (aCSF), other animals receiving Substance P + Idazoxan to increase LC activity (Sub P + Ida), and other animals receiving Clonidine to decrease LC activity (Clon). Statistics: ** = significantly lower (p < .02) and * = significantly lower (p < .05) than aCSF-infused animals on that test by contrasts on each test day.
Discussion
1. Hypothesis underlying the experiments
Experiments described in this paper tested the notion that activity of LC neurons influences reward produced by alcohol and thereby LC activity affects the propensity to consume alcohol. We suggested that high levels of LC activity will decrease reward and inhibit consumption of alcohol and, conversely, low levels of LC activity will enable or facilitate reward and promote consumption of alcohol. The mechanism by which this occurs was said to parallel what we have proposed regarding the influence of LC activity on depression, which is that LC activity influences firing of VTA-DA neurons (Weiss, Demetrikopoulos, West, & Bonsall, 1996; Weiss et al., 1998, 2005). Insofar as reward from alcohol appears to be dependent on firing of VTA-DA neurons, the activity of LC neurons therefore could be an important factor in determining reward produced by alcohol. Specifically, the hypothesis states that high levels of LC activity, particularly burst firing of LC neurons, will release galanin from LC terminals in the VTA-DA region that markedly hyperpolarizes, and therefore inhibits, activity of VTA-DA neurons, whereas, conversely, low levels of LC activity do not stimulate galanin release and thereby will produce little inhibition of VTA-DA neuronal activity. Through this mechanism, LC activity was proposed to influence reward from, and thus consumption of, alcohol.
2. Effects of alcohol on LC and VTA-DA neuronal activity in SUS and normal rats
What does this imply for SUS rats that readily consume alcohol, as opposed to normal rats that will not avidly consume alcohol? It has been reported that alcohol can inhibit the activity of LC neurons (Aston-Jones et al., 1982; Pohorecky & Brick, 1977; Strahlendorf & Strahlendorf, 1983) – consequently, one might predict that alcohol inhibits activity of LC neurons in SUS rats more so than in normal rats. Electrophysiological recording of LC single-unit activity in SUS and normal rats revealed that, as expected by this prediction, LC activity of SUS rats differed from normal rats in response to i.p. -injected alcohol. Specifically, spontaneous firing rate of LC units did not differ, but burst firing of LC neurons, which is what releases galanin to inhibit VTA-DA firing (Bartfai, Iverfeldt, Fisone, & Serfözö, 1988; Consolo et al., 1994), was dramatically and progressively inhibited by increasing doses of alcohol in SUS rats while showing little or no inhibition to alcohol in normal rats (see Fig. 1). It should be noted that a strong sensory stimulus such as paw compression (PC) is needed to elicit burst firing of LC in anesthetized animals, but such burst firing occurs often in response to common environmental stimuli in awake animals (Aston-Jones & Bloom, 1981), so alcohol will inhibit a response of LC neurons that is normally prevalent in awake rats. This effect on burst firing was predicted to enable alcohol to produce larger increases in VTA-DA activity in SUS rats than occur in normal rats. Electrophysiological recording of LC single-unit activity of VTA-DA neurons in SUS and normal rats confirmed this prediction as well. Both spontaneous firing rate and burst firing of VTA-DA neurons increased considerably more in SUS rats in response to increasing amounts of alcohol than was seen in normal rats (see Fig. 2). Thus, effects on both LC and VTA-DA activity were consistent with the proposed mechanism underlying enhanced alcohol consumption of SUS rats.
3. Effects of manipulation of LC activity on alcohol consumption of SUS rats
The next experiment showed that experimental manipulation of LC activity influenced consumption of alcohol in SUS rats in a manner consistent with the formulation presented above. As shown in Fig. 3, when LC activity was increased by infusion of Sub P + Ida into LC, voluntary alcohol consumption of SUS rats decreased. In fact, when this was done alcohol intake of SUS rats was reduced to the level of randomly-bred (normal) Sprague-Dawley rats that have no propensity to consume alcohol (i.e., compare four SD rats that consumed 2.0 ± 0.3 mL of 10% alcohol in 45 min with consumption of 2.6 ± 0.7 mL in 1 h by the seven SUS rats tested after Sub P + Ida [virtually all alcohol consumption occurs immediately after alcohol is available; t = 0.6, df = 9, p = .55 two tailed]). In contrast, when LC activity of SUS rats was decreased by infusion of clonidine into the LC, the voluntary alcohol consumption of these rats increased markedly, becoming significantly higher than their normally-elevated alcohol intake (See Fig. 3).
4. Is alcohol rewarding to SUS rats as determined by conditioned place preference?
We then determined that SUS rats found alcohol, given by i.p. injection, to be rewarding. This was assessed in the conditioned place preference (CPP) test, an often used method to assess reward. In the test session following 3 days of classical conditioning, SUS rats chose to remain on the side of the CPP apparatus associated with alcohol given by i.p. injection for significantly more time than they spent on the non-alcohol side of the apparatus, revealing effects of alcohol to be rewarding (see Fig. 4, Part A). This finding somewhat distinguishes the SUS rat as a model of alcohol consumption when compared to several other rat types that consume alcohol, including several alcohol-preferring rat lines. While one would think that “reward from alcohol” would be seen consistently in animals that readily consume alcohol, it has proved difficult to demonstrate this even in alcohol-preferring lines of rats (Busse et al., 2005; Ciccocioppo et al., 1999; Matsuzawa et al., 1998; Quertemont & De Witte, 2001; Stewart et al., 1996). In contrast, reward from alcohol was shown here in the SUS rat. Determination of blood alcohol levels (BAC) showed that the amount of alcohol injected i.p. into SUS rats during the CPP conditioning procedure was similar to the BAC that these rats produce when they voluntarily consume alcohol (see Fig. 4, Part B).
5. Can alcohol reward in CPP for SUS rats be altered by manipulating LC activity?
We next assessed whether reward from alcohol in the CPP could be blocked or enhanced by manipulating LC activity. In these experiments, some animals had their LC activity increased (by infusion of Sub P + Ida), others had it decreased (by infusion of Clon), and others had their LC activity not manipulated (by infusion of aCSF) before alcohol was injected in the CPP conditioning procedure. When the CPP test was conducted, as expected, a rewarding effect of alcohol in the CPP test was clearly manifested by SUS rats whose LC activity was not altered (i.e., infused with aCSF) prior to the i.p. alcohol injections during the conditioning procedure. In marked contrast, when LC activity was increased prior to an animal receiving i.p. alcohol, the rewarding effect of alcohol in the CPP test was completely blocked (see Fig. 5, Part A). What occurred when LC activity was decreased prior to the animal receiving i.p. alcohol was more ambiguous; specifically, these animals showed a rewarding effect of alcohol but it was not larger in magnitude than the effect seen in animals that had no LC manipulation, so decreasing LC activity did not enhance the rewarding effect of alcohol. However, an ancillary experiment sheds light on this particular ambiguity, and the results of this ancillary study are extremely interesting in and of themselves as is also discussed below.
6. Effects of manipulation of LC activity alone on reward in the CPP test
To appropriately interpret the results just described, an ancillary study was conducted with additional animals to determine whether simply increasing or decreasing activity of LC neurons would have effects on reward in the CPP test. If, for example, an increase in LC activity (by infusion of Sub P + Ida) produced, in and of itself, aversive consequences, and therefore caused animals to avoid the side of the CPP apparatus associated with this infusion, then it would be erroneous to conclude that the results in the CPP test revealed that increasing LC activity blocked the rewarding aspects of alcohol. In this case, avoidance of that side of the CPP apparatus could have been caused simply by the increase in LC activity resulting from infusion of Sub P + Ida. The ancillary study revealed clear effects on “reward” in the CPP test from having LC activity increased or decreased (see Fig. 5, Part B). The findings of this study did not at all compromise the conclusion that increasing LC activity blocked the rewarding effect on alcohol in SUS rats. To the contrary, when LC activity was increased (i.e., Sub P + Ida infused), animals chose to remain on that side of the CPP apparatus for more time, indicating that they found effects of increased LC activity to be rewarding. Consequently, when animals were injected with alcohol after being similarly infused, they clearly did not fail to spend time on the alcohol side of the apparatus because the LC manipulation had produced aversion; rather, the effects of the LC manipulation itself were, in fact, just the opposite. These animals showed no preference for the alcohol side of the CPP when their LC activity was increased despite the LC manipulation tending to be rewarding in and of itself. Regarding the effects of having LC activity decreased (Clon infused), the CPP results in the ancillary study showed that, conversely, this had somewhat aversive consequences. This result may well explain why animals that received alcohol while also having LC activity decreased did not show evidence of an enhanced rewarding effect of alcohol in the CPP – the consequences of the LC decrease itself could have prevented an augmented rewarding effect of alcohol from being manifested in the CPP test.
In regard to rewarding effects of increased LC activity, a reasonable question is how this can be reconciled with the fact that increased LC activity, especially increased burst firing of LC, inhibits VTA-DA neuronal activity. If reward from alcohol requires increased VTA-DA neuronal activity, how can an increase in LC activity by itself be rewarding? The following quotation from the abstract of the 1989 article by R. A. Wise and P. P. Rompre titled “Brain dopamine and reward” speaks directly to this issue: “One thing is clear: Dopamine is not the only reward transmitter, and dopaminergic neurons are not the final common path for all rewards.” The formulation that underlies the research described here is that increased VTA-DA dopaminergic activity is necessary for reward from alcohol, similar to reward from other substances of abuse, but it is not proposed that this is needed for all rewards. Apparently, rewarding consequences of increased LC activity do not require enhanced VTA-DA activity but is an instance of reward that does not depend on this mediation.
7. Additional significance of the findings from LC manipulation alone in the CPP test
The significance of the ancillary study that assessed whether increasing or decreasing LC activity might be rewarding in and of itself should not be overlooked; these results address an important issue that has been debated for decades. In the 1970s, Redmond and colleagues presented data indicating that activation of LC neurons promoted anxiety (Redmond & Huang, 1979; Redmond, Huang, Snyder, & Maas, 1976; summarized in Redmond, 1979, 1987), and in the years that followed their publication it was widely assumed that high levels of LC activity produced this emotional response. However, this formulation was controversial from the outset, and prominent investigators of LC function expressed doubts about it (e.g., Koob, Thatcher-Britton, Britton, Roberts, & Bloom, 1984). We stated in 1994 (Weiss et al., 1994) that the manipulations used in the early studies which suggested LC activity promoted anxiety (electrolytic lesions and electrical stimulation of LC, systemic administration of drugs) were not specific to LC neurons, and what was needed to test the validity of the proposed association was more specific manipulation of LC neuronal activity. When Julie Stout subsequently used a similar procedure as employed in the present paper – infusion of microquantities of drug directly into LC of rats to block or stimulate receptors on LC neurons to either excite or inhibit LC activity – the results were remarkable in that they showed exactly opposite of the proposal that LC activation promoted anxiety (reported in Weiss et al., 1994). That is, assessing anxiety by measuring the time it took before thirsty rats would drink from a water spout located in the center of an open field (a validated method of assessing anxiety [Stout & Weiss, 1994]), it was found that decreasing LC activity by drug infused into LC increased anxiety, and increasing LC activity by drug infused into LC decreased anxiety. Turning to the ancillary study in the present paper, effects on anxiety were not assessed here but, instead, this study determined whether animals would show a preference for having their LC activity decreased or increased. The finding was that decreased LC activity produced aversion to the side of the CPP apparatus associated with this event, and that increased LC activity produced preference for the side of the CPP apparatus associated with this event. Assuming that heightened anxiety would be an aversive condition and reduced anxiety would be a preferred condition, these results are clearly consistent with the earlier findings of Stout and colleagues. In this regard, another significant finding can be noted: Libet and Gleason (1994) reported effects of stimulating the LC in two communicative human subjects, and found that not only did LC stimulation not cause anxiety but, to the contrary, both patients reported feelings of relaxation and comfort from LC stimulation. In summary, the results of the ancillary study conducted here cast further doubt on the idea that LC activation promotes anxiety. In Weiss et al. (1994), we argued that LC activation, while clearly brought about by many stressful conditions, might actually serve to counteract anxiety and excess emotionality engendered by such circumstances. For further discussion, see Weiss et al. (1994) as the details will not be repeated here.
8. LC activation blocks alcohol stimulation of VTA-DA – electrophysiological evidence
Electrophysiological recording of VTA-DA neurons then demonstrated that what is proposed to be the neurological basis of reward from alcohol is blocked when LC activity is increased. The usual progressive increase in VTA-DA neuronal activity that occurs in SUS rats in response to increasing amounts of alcohol was completely negated when LC activity was increased by infusion of Sub P + Ida into the LC prior to the alcohol administration (see Fig. 6). In contrast, a marked increase in VTA-DA activity in response to alcohol was clearly evident in SUS rats that had no change produced in LC activity (i.e., aCSF infused into LC). The presumption here is that the blockade of the rise in VTA-DA activity normally produced by alcohol is what blocked the expression of reward from alcohol in the CPP test.
9. Association of alcohol with LC manipulations affects later alcohol consumption
As the final aspect of this study, an interesting and potentially significant phenomenon relating to conditioned responses was discovered when alcohol intake following the CPP procedure was measured. During the CPP procedure, SUS rats had their LC activity either increased, decreased, or not changed by drug infusion, and immediately thereafter were injected with 1.0 g/kg of alcohol. This was done three times as part of the CPP procedure. For these animals, the consequences of 1.0 g/kg i.p. alcohol were therefore associated with whatever are the effects of having LC activity increased or decreased, while having LC activity not changed (by infusion of aCSF into LC) would be expected to cause no effect to be associated with alcohol. This last expectation was confirmed – infusion of aCSF into the LC in conjunction with i.p. alcohol during the CPP procedure had no effect on animals’ subsequent voluntary intake of alcohol (see Fig. 7). The most salient and important effects were seen when LC activity had been increased by infusion of Sub P + Ida. When these SUS rats were later given the opportunity to consume alcohol, on the first presentation of alcohol they showed a large decrease in their normal alcohol consumption, and on the second presentation a significant decrease persisted. It was not until the third presentation that they again showed the usual elevated alcohol intake of SUS rats. These results were remarkable, and are informative. Thus, we observed that when an “alcohol reward-blocking neural event” (i.e., increase in LC activity leading to blockade of VTA-DA firing increase from alcohol) was associated with an i.p. injection of alcohol, this association led to a subsequent decrease in voluntary alcohol consumption.
How did this occur? It is noteworthy that alcohol was given by i.p. injection during conditioning in the CPP procedure, and this led to a subsequent decrease in alcohol intake by mouth. The SUS rats, however, were well experienced with alcohol intake by mouth before assessment of intake after the CPP procedure, which is significant. Because of this experience, these animals had repeated opportunities to associate the taste of the 10% alcohol solution with the subsequent physiological/pharmacological effects of the significant amount of alcohol these rats ingest. We presented data showing that (a) the amount of alcohol they normally ingest has a pharmacological effect, and (b) the amount of alcohol injected for the CPP procedure generated a similar blood level of alcohol as did the amount of alcohol that these rats voluntarily ingest, so these animals had repeatedly experienced the association of the taste of alcohol and similar physiological/pharmacological effects as were produced by the injection of alcohol in the CPP procedure. As a result of undergoing the CPP procedure, these animals experienced association of these physiological/pharmacological effects with their rewarding aspects ostensibly blocked because VTA-DA activity did not increase in response to alcohol. When subsequently given the opportunity to ingest alcohol, they initially do not drink any more alcohol than normal rats (see Fig. 7). This result conveys significant information: it indicates that if a SUS rat has experienced the same physiological effects that result from ingestion of the large quantity of alcohol that these rats normally ingest but the rewarding aspects of these effects are blocked, the taste cue previously associated with ingesting alcohol will no longer lead to appreciable alcohol intake. This indicates that the motivation of SUS rats for drinking alcohol, the cue for which is the taste of the alcohol solution, are the rewarding physiological/pharmacological consequences produced by the alcohol ingestion. If reward from the physiological consequences is blocked or does not occur, SUS rats no longer avidly consume alcohol. When Jackie Gleason, the famous hard-drinking comedian, was asked why he drank, he replied, “I drink to get bagged,” which we can assume was rewarding. Apparently, so do SUS rats.
10. Comment on VTA-DA activity, reward, and recent theorizing
Some comment should be made about the relationship between VTA-DA neuronal activity and reward in view of developing findings and theorizing regarding dopamine and reward. Whereas earlier theorizing sought to equate mesolimbic dopaminergic activity with mediation of “pleasure” or “hedonic valuation” (Wise, 1982, 1985), an increasing body of findings and theory indicates that this is an oversimplification. Dopaminergic activity may be critically involved in aspects of rewarding events, including prediction of reward and “wanting/seeking” reward, but may not actually mediate pleasure per se (Berridge & Kringelbach, 2011; Castro & Berridge, 2014; Panksepp, 2005; Salamone & Correa, 2012; Schultz, 1998, 2000). In regard to the findings reported here, these do fit with the notion that (a) alcohol is rewarding for SUS rats, and (b) VTA-DA activity is critical for mediating this reward. In particular, (a) SUS rats expressed a clear preference for an environment associated with a pharmacologically active level of blood alcohol (i.e., in the CPP test), and (b) when the usual rise in VTA-DA neuronal activity produced by this level of alcohol was blocked by increasing LC activity, this preference was no longer expressed. Additionally, when the physiological effects of alcohol that normally support this preference in SUS rats were associated with (i.e., paired with) failure of VTA-DA activity to rise as normally occurs, thereafter SUS rats lost the proclivity to voluntarily consume a large amount of alcohol. All of the findings reported here, including the remarkable demonstration that failure of VTA-DA neuronal activity to rise in response to elevated blood alcohol degrades the ability of the alcohol taste cue to stimulate alcohol consumption, seem readily explained by postulating that VTA-DA neuronal activity mediates reward. We leave it to later investigators to reinterpret these findings in light of more rigorous formulations and explain them by perhaps more sophisticated conceptions.
11. Limitations to the experiments described in this paper
In conclusion, certain limitations of the experiments presented here should be noted. First, perhaps the most salient of these is that the experiments described in this paper have not conclusively established that the changes in LC activity produced by either alcohol injection or by manipulations of the LC firing rate (by microinfusion of drug into the LC region) alter alcohol consumption and reward because of effects on VTA-DA neuronal activity. Insofar as LC neurons project widely throughout the brain, it remains possible that changes in LC activity affected alcohol consumption and alcohol-related reward because LC influenced activity in some brain region(s) or neurons other than VTA-DA neurons. The required experiment to conclusively establish that it was changes in VTA-DA neuronal activity that were responsible for the effects on alcohol consumption and reward seen after changes in LC activity would be to alter LC activity but then prevent this change from affecting only VTA-DA neurons and show that alcohol consumption and reward is not altered in this case. The technical difficulty of carrying out this particular experiment is considerable because of the lack of reliable drugs to block galanin type1 receptors on VTA-DA neurons; nevertheless, in the absence of such an experiment, the LC induced changes in VTA-DA results described above have not been shown definitively to be the cause of changes in alcohol consumption and reward. Of course, considerable experimental evidence has linked alcohol consumption and reward to VTA-DA activity (reviewed in the Introduction). Also, it is evident that changes in LC activity were indeed directly responsible for altering alcohol-induced changes in VTA-DA activity – this was shown by the study where electrophysiological recordings of VTA-DA activity were made in conjunction with microinfusion on Sub P + Ida into LC, and the experimentally-induced change in LC activity was shown to abolish the increase in VTA-DA activity that normally occurs in response to injected alcohol.
Second, electrophysiological single-unit activity (of LC and VTA-DA neurons) was recorded under anesthesia (chloral hydrate), while alcohol consumption and reward from alcohol in the CPP test was assessed in awake animals. Consequently, it becomes a question of whether the results obtained in electrophysiological assessment therefore can be related to the behavior seen in awake animals. This is a potential limitation to the results described here. The question is whether the anesthesia alters or distorts the normal (awake state) neural activity so that the recorded electrophysiological activity is not what would be expected in the awake state. However, we do have findings that bear on this question. In Simson and Weiss (1988), we found that application of inescapable electric shock to awake animals resulted in expected alterations in LC activity that were then recorded under anesthesia 2.0–3.0 h later. Also, in the SUS rats used in the present study which, as explained in the Introduction, were selectively-bred for greater susceptibility to stressors, we have found that exposure to a relatively mild stressor (placement into a novel environment for 30 min with 95-dB white noise) that reduces their subsequent swim-test activity, and is not sufficiently stressful to have this effect on the swim-test activity of normal rats, significantly elevated LC activity in SUS rats relative to normal rats when electrophysiological recording of LC activity was conducted under anesthesia approximately 2.0 h after the exposure to this relatively mild stressor (unpublished data). Results such as these indicate that electrophysiological activity recorded under anesthesia will show effects that are relevant to awake, non-anesthetized animals.
Finally, there is the possibility that the use of chloral hydrate as an anesthetic for the electrophysiological recording altered the effects of injected alcohol during single-unit recording because the neural targets of chloral hydrate are similar to those of alcohol. Chloral hydrate is effective by being reduced to its active metabolite, trichloroethanol, which is highly similar to ethanol, differing in structure from ethanol only by hydrogen atoms at position 2 replaced by three chlorine atoms. In particular, both trichloroethanol and ethanol both affect 5 HT3 receptors, which have been importantly implicated in mediating dopamine release in response to ethanol , as well as NMDA receptors that are critically involved in excitatory stimulation (burst firing) of LC neurons (Downie et al., 1995; Lovinger, Sung, & Zhou, 2000; Salous et al., 2009). As a result, using chloral hydrate as an anesthetic for the electrophysiological recording could have altered both VTA-DA activity and LC excitability during the electrophysiological recording. This is acknowledged here. However, one would speculate that if trichloroethanol occupied similar targets/binding sites as ethanol that the effects of injected ethanol would be diminished as a consequence, since target sites would already be occupied by the anesthetic metabolite. Insofar as the effects of injected alcohol were quite large during electrophysiological recording, a functional consequence of having used chloral hydrate is not evident.
Highlights.
Rats of the Stress Susceptible line (SUS) consume much alcohol; normal rats do not
We studied neural control of the large, pharmacological alcohol intake of SUS rats
Locus coeruleus (LC) regulates intake: high LC activity → low intake, and vice versa
This occurs as LC alters reward from alcohol via influence on VTA dopamine neurons
SUS rats show place preference (reward) of alcohol, similarly regulated by LC activity
Acknowledgment
This research was supported in part by Public Health Service Grant #AA021411.
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
References
- Andén N, Grabowska M. Pharmacological evidence for a stimulation of dopamine neurons by noradrenaline neurons in the brain. European Journal of Pharmacology. 1976;39:275–282. doi: 10.1016/0014-2999(76)90136-9. [DOI] [PubMed] [Google Scholar]
- Aston Jones G, Bloom FE. Norepinephrine containing locus coeruleus neurons in behaving rats exhibit pronounced responses to non noxious environmental stimuli. The Journal of Neuroscience. 1981;1:887–900. doi: 10.1523/JNEUROSCI.01-08-00887.1981. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Aston Jones G, Foote SL, Bloom FE. Low doses of ethanol disrupt sensory responses of brain noradrenergic neurones. Nature. 1982;296:857–860. doi: 10.1038/296857a0. [DOI] [PubMed] [Google Scholar]
- Bartfai T, Iverfeldt K, Fisone G, Serfözö P. Regulation of the release of coexisting neurotransmitters. Annual Review of Pharmacology and Toxicology. 1988;28:285–310. doi: 10.1146/annurev.pa.28.040188.001441. [DOI] [PubMed] [Google Scholar]
- Berridge KC, Kringelbach ML. Building a neuroscience of pleasure and well being. Psychology of Well-being. 2011;1:1–3. doi: 10.1186/2211-1522-1-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Borsody MK, Weiss JM. Influence of corticotropin releasing hormone on electrophysiological activity of locus coeruleus neurons. Brain Research. 1996;724:149–168. doi: 10.1016/0006-8993(96)00199-0. [DOI] [PubMed] [Google Scholar]
- Brodie MS, Pesold C, Appel SB. Ethanol directly excites dopaminergic ventral tegmental area reward neurons. Alcoholism: Clinical and Experimental Research. 1999;23:1848–1852. [PubMed] [Google Scholar]
- Brodie MS, Shefner SA, Dunwiddie TV. Ethanol increases the firing rate of dopamine neurons of the rat ventral tegmental area in vitro. Brain Research. 1990;508:65–69. doi: 10.1016/0006-8993(90)91118-z. [DOI] [PubMed] [Google Scholar]
- Bunney BS, Walters JR, Roth RH, Aghajanian GK. Dopaminergic neurons: effect of antipsychotic drugs and amphetamine on single cell activity. The Journal of Pharmacology and Experimental Therapeutics. 1973;185:560–571. [PubMed] [Google Scholar]
- Busse GD, Lawrence ET, Riley AL. The effects of alcohol preexposure on cocaine, alcohol and cocaine/alcohol place conditioning. Pharmacology, Biochemistry, and Behavior. 2005;81:459–465. doi: 10.1016/j.pbb.2005.04.003. [DOI] [PubMed] [Google Scholar]
- Castro DC, Berridge KC. Advances in the neurobiological bases for food 'liking' versus 'wanting'. Physiology & Behavior. 2014;136:22–30. doi: 10.1016/j.physbeh.2014.05.022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chiodo LA. Dopamine containing neurons in the mammalian central nervous system: electrophysiology and pharmacology. Neuroscience and Biobehavioral Reviews. 1988;12:49–91. doi: 10.1016/s0149-7634(88)80073-3. [DOI] [PubMed] [Google Scholar]
- Ciccocioppo R, Panocka I, Froldi R, Quitadamo E, Massi M. Ethanol induces conditioned place preference in genetically selected alcohol preferring rats. Psychopharmacology. 1999;141:235–241. doi: 10.1007/s002130050830. [DOI] [PubMed] [Google Scholar]
- Consolo S, Baldi G, Russi G, Civenni G, Bartfai T, Vezzani A. Impulse flow dependency of galanin release in vivo in the rat ventral hippocampus. Proceedings of the National Academy of Sciences of the United States of America. 1994;91:8047–8051. doi: 10.1073/pnas.91.17.8047. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Donaldson I, Dolphin A, Jenner P, Marsden CD, Pycock C. The roles of noradrenaline and dopamine in contraversive circling behaviour seen after unilateral electrolytic lesions of the locus coeruleus. European Journal of Pharmacology. 1976;39:179–191. doi: 10.1016/0014-2999(76)90126-6. [DOI] [PubMed] [Google Scholar]
- Downie DL, Hope AG, Belelli D, Lambert JJ, Peters JA, Bentley KR, et al. The interaction of trichloroethanol with murine recombinant 5 HT3 receptors. British Journal of Pharmacology. 1995;114:1641–1651. doi: 10.1111/j.1476-5381.1995.tb14952.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Doyon WM, York JL, Diaz LM, Samson HH, Czachowski CL, Gonzales RA. Dopamine activity in the nucleus accumbens during consummatory phases of oral ethanol self administration. Alcoholism: Clinical and Experimental Research. 2003;27:1573–1582. doi: 10.1097/01.ALC.0000089959.66222.B8. [DOI] [PubMed] [Google Scholar]
- Foote SL, Aston Jones G, Bloom FE. Impulse activity of locus coeruleus neurons in awake rats and monkeys is a function of sensory stimulation and arousal. Proceedings of the National Academy of Sciences of the United States of America. 1980;77:3033–3037. doi: 10.1073/pnas.77.5.3033. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gadsden RH, Taylor EH, Steindel SJ. Ethanol in biological fluids by enzymatic analysis. In: Frings CS, Faulkner WR, editors. Selective Methods of Emergency Toxicology, Vol. 11, Selected Methods of Clinical Chemistry. AACC Press; Washington, DC: 1986. pp. 63–65. [Google Scholar]
- Gessa GL, Muntoni F, Collu M, Vargiu L, Mereu G. Low doses of ethanol activate dopaminergic neurons in the ventral tegmental area. Brain Research. 1985;348:201–203. doi: 10.1016/0006-8993(85)90381-6. [DOI] [PubMed] [Google Scholar]
- Gill K, France C, Amit Z. Voluntary ethanol consumption in rats: an examination of blood/brain ethanol levels and behavior. Alcoholism: Clinical and Experimental Research. 1986;10:457–462. doi: 10.1111/j.1530-0277.1986.tb05124.x. [DOI] [PubMed] [Google Scholar]
- Grace AA, Bunney BS. Nigral dopamine neurons: intracellular recording and identification with L dopa injection and histofluorescence. Science. 1980;210:654–656. doi: 10.1126/science.7433992. [DOI] [PubMed] [Google Scholar]
- Grace AA, Bunney BS. Intracellular and extracellular electrophysiology of nigral dopaminergic neurons 1. Identification and characterization. Neuroscience. 1983;10:301–315. doi: 10.1016/0306-4522(83)90135-5. [DOI] [PubMed] [Google Scholar]
- Grace AA, Bunney BS. The control of firing pattern in nigral dopamine neurons: burst firing. The Journal of Neuroscience. 1984;4:2877–2890. doi: 10.1523/JNEUROSCI.04-11-02877.1984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Graham AW, Aghajanian GK. Effects of amphetamine on single cell activity in a catecholamine nucleus, the locus coeruleus. Nature. 1971;234:100–102. doi: 10.1038/234100b0. [DOI] [PubMed] [Google Scholar]
- Grenhoff J, Nisell M, Ferré S, Aston Jones G, Svensson TH. Noradrenergic modulation of midbrain dopamine cell firing elicited by stimulation of the locus coeruleus in the rat. Journal of Neural Transmission. General Section. 1993;93:11–25. doi: 10.1007/BF01244934. [DOI] [PubMed] [Google Scholar]
- Grenhoff J, Svensson TH. Prazosin modulates the firing pattern of dopamine neurons in rat ventral tegmental area. European Journal of Pharmacology. 1993;233:79–84. doi: 10.1016/0014-2999(93)90351-h. [DOI] [PubMed] [Google Scholar]
- Guyenet PG, Aghajanian GK. Antidromic identification of dopaminergic and other output neurons of the rat substantia nigra. Brain Research. 1978;150:69–84. doi: 10.1016/0006-8993(78)90654-6. [DOI] [PubMed] [Google Scholar]
- Imperato A, Di Chiara G. Preferential stimulation of dopamine release in the nucleus accumbens of freely moving rats by ethanol. The Journal of Pharmacology and Experimental Therapeutics. 1986;239:219–228. [PubMed] [Google Scholar]
- Koob GF, Thatcher Britton K, Britton DR, Roberts DC, Bloom FE. Destruction of the locus coeruleus or the dorsal NE bundle does not alter the release of punished responding by ethanol and chlordiazepoxide. Physiology & Behavior. 1984;33:479–485. doi: 10.1016/0031-9384(84)90172-0. [DOI] [PubMed] [Google Scholar]
- Korf J, Bunney BS, Aghajanian GK. Noradrenergic neurons: morphine inhibition of spontaneous activity. European Journal of Pharmacology. 1974;25:165–169. doi: 10.1016/0014-2999(74)90045-4. [DOI] [PubMed] [Google Scholar]
- Libet B, Gleason CA. The human locus coeruleus and anxiogenesis. Brain Research. 1994;634:178–180. doi: 10.1016/0006-8993(94)90274-7. [DOI] [PubMed] [Google Scholar]
- Lovinger DM, Sung KW, Zhou Q. Ethanol and trichloroethanol alter gating of 5 HT3 receptor channels in NCB 20 neuroblastoma cells. [Research Support, U.S. Gov't, P.H.S.]. Neuropharmacology. 2000;39:561–570. doi: 10.1016/s0028-3908(99)00164-1. [DOI] [PubMed] [Google Scholar]
- Marinelli M, White FJ. Enhanced vulnerability to cocaine self administration is associated with elevated impulse activity of midbrain dopamine neurons. The Journal of Neuroscience. 2000;20:8876–8885. doi: 10.1523/JNEUROSCI.20-23-08876.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Matsuzawa S, Suzuki T, Misawa M. Conditioned fear stress induces ethanol associated place preference in rats. European Journal of Pharmacology. 1998;341:127–130. doi: 10.1016/s0014-2999(97)01456-8. [DOI] [PubMed] [Google Scholar]
- McBride WJ, Bodart B, Lumeng L, Li TK. Association between low contents of dopamine and serotonin in the nucleus accumbens and high alcohol preference. Alcoholism: Clinical and Experimental Research. 1995;19:1420–1422. doi: 10.1111/j.1530-0277.1995.tb01001.x. [DOI] [PubMed] [Google Scholar]
- McBride WJ, Li TK. Animal models of alcoholism: neurobiology of high alcohol drinking behavior in rodents. Critical Reviews in Neurobiology. 1998;12:339–369. doi: 10.1615/critrevneurobiol.v12.i4.40. [DOI] [PubMed] [Google Scholar]
- McBride WJ, Murphy JM, Gatto GJ, Levy AD, Lumeng L, Li TK. Serotonin and dopamine systems regulating alcohol intake. Alcohol and Alcoholism. Supplement. 1991;1:411–416. [PubMed] [Google Scholar]
- Melis M, Diana M, Enrico P, Marinelli M, Brodie MS. Ethanol and acetaldehyde action on central dopamine systems: mechanisms, modulation, and relationship to stress. Alcohol. 2009;43:531–539. doi: 10.1016/j.alcohol.2009.05.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Melis M, Pillolla G, Perra S, Colombo G, Muntoni AL, Pistis M. Electrophysiological properties of dopamine neurons in the ventral tegmental area of Sardinian alcohol preferring rats. Psychopharmacology. 2009;201:471–481. doi: 10.1007/s00213-008-1309-2. [DOI] [PubMed] [Google Scholar]
- Minabe Y, Emori K, Ashby CR., Jr. Significant differences in the activity of midbrain dopamine neurons between male Fischer 344 (F344) and Lewis rats: an in vivo electrophysiological study. Life Sciences. 1995;56:PL261–267. doi: 10.1016/0024-3205(95)00075-5. [DOI] [PubMed] [Google Scholar]
- Morzorati SL. VTA dopamine neuron activity distinguishes alcohol preferring (P) rats from Wistar rats. Alcoholism: Clinical and Experimental Research. 1998;22:854–857. [PubMed] [Google Scholar]
- Morzorati SL, Marunde RL. Comparison of VTA dopamine neuron activity in lines of rats selectively bred to prefer or avoid alcohol. Alcoholism: Clinical and Experimental Research. 2006;30:991–997. doi: 10.1111/j.1530-0277.2006.00113.x. [DOI] [PubMed] [Google Scholar]
- Murphy JM, McBride WJ, Lumeng L, Li TK. Contents of monoamines in forebrain regions of alcohol preferring (P) and nonpreferring (NP) lines of rats. Pharmacology, Biochemistry, and Behavior. 1987;26:389–392. doi: 10.1016/0091-3057(87)90134-1. [DOI] [PubMed] [Google Scholar]
- Murphy JM, Stewart RB, Bell RL, Badia Elder NE, Carr LG, McBride WJ, et al. Phenotypic and genotypic characterization of the Indiana University rat lines selectively bred for high and low alcohol preference. Behavior Genetics. 2002;32:363–388. doi: 10.1023/a:1020266306135. [DOI] [PubMed] [Google Scholar]
- Nowak KL, McBride WJ, Lumeng L, Li TK, Murphy JM. Involvement of dopamine D2 autoreceptors in the ventral tegmental area on alcohol and saccharin intake of the alcohol preferring P rat. Alcoholism: Clinical and Experimental Research. 2000;24:476–483. [PubMed] [Google Scholar]
- Panksepp J. Affective consciousness: Core emotional feelings in animals and humans. [Review]. Consciousness and Cognition. 2005;14:30–80. doi: 10.1016/j.concog.2004.10.004. [DOI] [PubMed] [Google Scholar]
- Pohorecky LA, Brick J. Activity of neurons in the locus coeruleus of the rat: inhibition by ethanol. Brain Research. 1977;131:174–179. doi: 10.1016/0006-8993(77)90039-7. [DOI] [PubMed] [Google Scholar]
- Quertemont E, De Witte P. Conditioned stimulus preference after acetaldehyde but not ethanol injections. Pharmacology, Biochemistry, and Behavior. 2001;68:449–454. doi: 10.1016/s0091-3057(00)00486-x. [DOI] [PubMed] [Google Scholar]
- Redmond DE., Jr. New and old evidence for the involvement of a brain norepinephrine system in anxiety. In: Fann WE, Karacan I, Pokorny AD, Williams RL, editors. Phenomenology and Treatment of Anxiety. Spectrum; New York: 1979. pp. 89–111. [Google Scholar]
- Redmond DE., Jr. Studies of the nucleus locus ceruleus in monkeys and hypotheses for neuropsychopharmacology. In: Meltzer HY, editor. Psychopharmacology: The Third Generation of Progress. Raven; New York: 1987. pp. 967–975. [Google Scholar]
- Redmond DE, Jr., Huang YH. Current concepts. II. New evidence for a locus coeruleus norepinephrine connection with anxiety. Life Sciences. 1979;25:2149–2162. doi: 10.1016/0024-3205(79)90087-0. [DOI] [PubMed] [Google Scholar]
- Redmond DE, Jr., Huang YH, Snyder DR, Maas JW. Behavioral effects of stimulation of the nucleus locus coeruleus in the stump tailed monkey Macaca arctoides. Brain Research. 1976;116:502–510. doi: 10.1016/0006-8993(76)90498-4. [DOI] [PubMed] [Google Scholar]
- Rodd ZA, Bell RL, Sable HJ, Murphy JM, McBride WJ. Recent advances in animal models of alcohol craving and relapse. Pharmacology, Biochemistry, and Behavior. 2004;79:439–450. doi: 10.1016/j.pbb.2004.08.018. [DOI] [PubMed] [Google Scholar]
- Rodd ZA, Bell RL, Zhang Y, Murphy JM, Goldstein A, Zaffaroni A, et al. Regional heterogeneity for the intracranial self administration of ethanol and acetaldehyde within the ventral tegmental area of alcohol preferring (P) rats: involvement of dopamine and serotonin. Neuropsychopharmacology. 2005;30:330–338. doi: 10.1038/sj.npp.1300561. [DOI] [PubMed] [Google Scholar]
- Salamone JD, Correa M. The mysterious motivational functions of mesolimbic dopamine. Neuron. 2012;76:470–485. doi: 10.1016/j.neuron.2012.10.021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Salous AK, Ren H, Lamb KA, Hu XQ, Lipsky RH, Peoples RW. Differential actions of ethanol and trichloroethanol at sites in the M3 and M4 domains of the NMDA receptor GluN2A (NR2A) subunit. British Journal of Pharmacology. 2009;158:1395–1404. doi: 10.1111/j.1476-5381.2009.00397.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schultz W. Predictive reward signal of dopamine neurons. Journal of Neurophysiology. 1998;80:1–27. doi: 10.1152/jn.1998.80.1.1. [DOI] [PubMed] [Google Scholar]
- Schultz W. Multiple reward signals in the brain. Nature Reviews. Neuroscience. 2000;1:199–207. doi: 10.1038/35044563. [DOI] [PubMed] [Google Scholar]
- Scott PA, Cierpial MA, Kilts CD, Weiss JM. Susceptibility and resistance of rats to stress induced decreases in swim test activity: a selective breeding study. Brain Research. 1996;725:217–230. doi: 10.1016/0006-8993(96)00093-5. [DOI] [PubMed] [Google Scholar]
- Simson PE, Weiss JM. Alpha 2 receptor blockade increases responsiveness of locus coeruleus neurons to excitatory stimulation. The Journal of Neuroscience. 1987;7:1732–1740. doi: 10.1523/JNEUROSCI.07-06-01732.1987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Simson PE, Weiss JM. Altered activity of the locus coeruleus in an animal model of depression. Neuropsychopharmacology. 1988;1:287–295. [PubMed] [Google Scholar]
- Simson PE, Weiss JM. Blockade of alpha 2 adrenergic receptors, but not blockade of gamma aminobutyric acidA, serotonin, or opiate receptors, augments responsiveness of locus coeruleus neurons to excitatory stimulation. Neuropharmacology. 1989;28:651–660. doi: 10.1016/0028-3908(89)90147-0. [DOI] [PubMed] [Google Scholar]
- Stewart RB, Murphy JM, McBride WJ, Lumeng L, Li TK. Place conditioning with alcohol in alcohol preferring and nonpreferring rats. Pharmacology, Biochemistry, and Behavior. 1996;53:487–491. doi: 10.1016/0091-3057(95)02102-7. [DOI] [PubMed] [Google Scholar]
- Stout JC, Weiss JM. An animal model for measuring behavioral responses to anxiogenic and anxiolytic manipulations. Pharmacology, Biochemistry, and Behavior. 1994;47:459–465. doi: 10.1016/0091-3057(94)90143-0. [DOI] [PubMed] [Google Scholar]
- Strahlendorf HK, Strahlendorf JC. Ethanol suppression of locus coeruleus neurons: relevancy to the fetal alcohol syndrome. Neurobehavioral Toxicology and Teratology. 1983;5:221–224. [PubMed] [Google Scholar]
- Thiele TE, van Dijk G, Bernstein IL. Ethanol induced c Fos expression in rat lines selected for low and high alcohol consumption. Brain Research. 1997;756:278–282. doi: 10.1016/s0006-8993(97)00228-x. [DOI] [PubMed] [Google Scholar]
- Waller MB, Murphy JM, McBride WJ, Lumeng L, Li TK. Effect of low dose ethanol on spontaneous motor activity in alcohol preferring and nonpreferring lines of rats. Pharmacology, Biochemistry, and Behavior. 1986;24:617–623. doi: 10.1016/0091-3057(86)90567-8. [DOI] [PubMed] [Google Scholar]
- Wang RY. Dopaminergic neurons in the rat ventral tegmental area. I. Identification and characterization. Brain Research Reviews. 1981;3:123–140. [Google Scholar]
- Weiss JM, Bonsall RW, Demetrikopoulos MK, Emery MS, West CH. Galanin: a significant role in depression? Annals of the New York Academy of Sciences. 1998;863:364–382. doi: 10.1111/j.1749-6632.1998.tb10707.x. [DOI] [PubMed] [Google Scholar]
- Weiss JM, Boss Williams KA, Moore JP, Demetrikopoulos MK, Ritchie JC, West CH. Testing the hypothesis that locus coeruleus hyperactivity produces depression related changes via galanin. Neuropeptides. 2005;39:281–287. doi: 10.1016/j.npep.2004.12.028. [DOI] [PubMed] [Google Scholar]
- Weiss JM, Demetrikopoulos MK, West CHK, Bonsall RW. Hypothesis linking the noradrenergic and dopaminergic systems in depression. Depression. 1996;3:225–245. [Google Scholar]
- Weiss JM, Stout JC, Aaron MF, Quan N, Owens MJ, Butler PD, et al. Depression and anxiety: role of the locus coeruleus and corticotropin releasing factor. Brain Research Bulletin. 1994;35:561–572. doi: 10.1016/0361-9230(94)90170-8. [DOI] [PubMed] [Google Scholar]
- Weiss JM, West CH, Emery MS, Bonsall RW, Moore JP, Boss Williams KA. Rats selectively bred for behavior related to affective disorders: proclivity for intake of alcohol and drugs of abuse, and measures of brain monoamines. Biochemical Pharmacology. 2008;75:134–159. doi: 10.1016/j.bcp.2007.09.027. [DOI] [PubMed] [Google Scholar]
- West CH, Weiss JM. A selective test for antidepressant treatments using rats bred for stress induced reduction of motor activity in the swim test. Psychopharmacology. 2005;182:9–23. doi: 10.1007/s00213-005-0048-x. [DOI] [PubMed] [Google Scholar]
- West CH, Weiss JM. Intake of ethanol and reinforcing fluids in rats bred for susceptibility to stress. [Research Support, Non U.S. Gov't]. Alcohol. 2006;38:13–27. doi: 10.1016/j.alcohol.2006.03.005. [DOI] [PubMed] [Google Scholar]
- White FJ, Wang RY. Differential effects of classical and atypical antipsychotic drugs on A9 and A10 dopamine neurons. Science. 1983;221:1054–1057. doi: 10.1126/science.6136093. [DOI] [PubMed] [Google Scholar]
- Wise RA. Neuroleptics and operant behavior: The anhedonia hypothesis. Behavioral and Brain Sciences. 1982;5:39–53. [Google Scholar]
- Wise RA. The anhedonia hypothesis: Mark III. Behavioral and Brain Sciences. 1985;8:178–186. [Google Scholar]
- Wise RA, Rompre PP. Brain dopamine and reward. Annual Review of Psychology. 1989;40:191–225. doi: 10.1146/annurev.ps.40.020189.001203. [DOI] [PubMed] [Google Scholar]
- Xiao C, Shao XM, Olive MF, Griffin WC, 3rd, Li KY, Krnjević K, et al. Ethanol facilitates glutamatergic transmission to dopamine neurons in the ventral tegmental area. Neuropsychopharmacology. 2009;34:307–318. doi: 10.1038/npp.2008.99. [DOI] [PMC free article] [PubMed] [Google Scholar]