Abstract
Identifying statistical patterns between environmental stimuli enables organisms to respond adaptively when cues are later observed. However, stimuli are often obscured from detection, necessitating behavior under conditions of ambiguity. Considerable evidence indicates decisions under ambiguity rely on inference processes that draw on past experiences to generate predictions under novel conditions. Despite the high demand for this process and the observation that it deteriorates disproportionately with age, the underlying mechanisms remain unknown. We developed a rodent model of decision-making during ambiguity to examine features of experience that contribute to inference. Rats learned either a simple (positive patterning) or complex (negative patterning) instrumental discrimination between the illumination of one or two lights. During test, only one light was lit while the other relevant light was blocked from physical detection (covered by an opaque shield, rendering its status ambiguous). We found experience with the complex negative patterning discrimination was necessary for rats to behave sensitively to the ambiguous test situation. These rats behaved as if they inferred the presence of the hidden light, responding differently than when the light was explicitly absent (uncovered and unlit). Differential expression profiles of the immediate early gene cFos indicated hippocampal involvement in the inference process while localized microinfusions of the muscarinic antagonist, scopolamine, into the dorsal hippocampus caused rats to behave as if only one light was present. That is, blocking cholinergic modulation prevented the rat from inferring the presence of the hidden light. Collectively, these results suggest cholinergic modulation mediates recruitment of hippocampal processes related to past experiences and transfer of these processes to make decisions during ambiguous situations. Our results correspond with correlations observed between human brain function and inference abilities, suggesting our experiments may inform interventions to alleviate or prevent cognitive dysfunction.
Keywords: Representation, Negative Patterning, Positive Patterning, Stimulus Ambiguity, Acetylcholine, Inference
We rarely have direct access to all environmental information, necessitating decision-making under conditions of cue ambiguity. Discerning critical information from ambiguous situations requires prior knowledge or contextual information. For example, to select between a “financial institution” and the “river border” as the appropriate meeting location when your presence is requested at the “bank” requires additional contextual clues (Taler et al., 2009). These clues guide predictions about the ambiguous circumstance, allowing an inference to be drawn. Despite the pervasiveness of decisions under ambiguity, little is known about the underlying mechanisms. Recent evidence suggests that, in addition to its well-known involvement in contextual processing (Fanselow, 2009), the hippocampus plays a role during inferences (Kumaran, 2012; Reber et al., 2012; Zeithamova et al., 2012a; Barron et al., 2013), but it remains unclear if the hippocampus influences decision-making under ambiguity.
Rats, like humans (Hagmayer and Waldmann, 2007), distinguish between the ambiguous and explicit absence of events (Blaisdell et al., 2009; Fast and Blaisdell, 2011; Waldmann et al., 2012; Fast et al., in press). We investigated if and when rats use this distinction in making behavioral decisions and to what extent the hippocampus mediates this ability. Rats were trained to lever-press during one of two lights (A or B) or when both lights co-occurred (AB). Some rats learned a simple positive-patterning discrimination with lever presses rewarded during both lights (AB+) but not when either light appeared independently (A−, B−). Other rats learned a negative-patterning discrimination that rewarded lever presses to either light, but not both (A+, B+, AB−). Rats were then tested with only A illuminated, while B either was explicitly absent (unlit) or ambiguous (covered by an opaque shield). When B is unlit, rats should respond identically to A-alone training trials (little to no responding if A served in positive-patterning and high rates of responding if A served in negative-patterning); however, if rats infer the presence of the hidden light, they should respond similarly to AB trials when B is covered (increased responding for positive-patterning and decreased responding for negative-patterning). Interestingly, only rats exposed to negative-patterning treated the test situations differently, responding less when B was covered compared to unlit. This behavior is consistent with the rat inferring the perceptually-absent light B because lever presses were never reinforced during AB negative-patterning training trials.
Unlike positive-patterning, which can be solved through linear summation, negative-patterning requires a rule-based or configural approach to treat the combination of cues (AB) differently than the sum of its elements (A and B). Similar to contextual processing, this approach relies on hippocampal (Phillips and LeDoux, 1992; Alvarado and Rudy, 1995; Rudy and Sutherland, 1995; Sakimoto and Sakata, 2013) and cholinergic function (Moran, 1992; Richmond et al., 1997; Gale et al., 2001; Butt et al., 2002; Hata et al., 2007). Furthermore, configural processes transfer to situations where they are normally unnecessary (Mehta and Williams, 2002; Williams and Braker, 2002; Sakimoto et al., 2013), including positive-patterning. Interestingly, rats tested with positive-patterning cues behave as if they infer the presence of a hidden light if they are also exposed to negative-patterning with auditory cues (Fast and Blaisdell, 2011), suggesting the configural process of negative-patterning may also transfer to ambiguous positive-patterning cues. Given that inferential reasoning involves the transfer of prior knowledge to a novel situation and this process has been correlated with hippocampal activity in humans, we hypothesized similar hippocampal involvement with negative-patterning mediates the rat’s ability to transfer this experience to the ambiguous test situation. We examined if hippocampal-dependent (negative-patterning) training is necessary for rats to infer the presence of a hidden event by controlling for training cue modality, ratio of reinforcement, and difficulty and duration. We then directly assessed hippocampal involvement by examining cFos expression within the dorsal hippocampus (DH). Finally, we conducted a series of tests following microinfusion of the muscarinic antagonist, scopolamine, or vehicle into the DH. If hippocampal cholinergic integrity is needed for decision-making under ambiguity, scopolamine should cause rats to respond as if only one light is lit, regardless of whether the second light is covered or not.
MATERIALS AND METHODS
Subjects
One-hundred-sixty-eight experimentally-naïve female Long-Evans food-restricted (to 85% ad lib weight) rats (Rattus norvegicus) acquired from Harlan (Indianapolis, IN) were randomly assigned to one of eight experimental conditions (Table 1) and pair-housed in transparent plastic tubs with wood shaving substrate in a vivarium maintained on a reverse 12-hr light/dark cycle. Experiments occurred during the dark cycle, six days a week. All experiments adhered to the policies established and approved by the IACUC of UCLA.
Table 1.
Summary of Experimental Designs
| Experiment | Primary IV/DV | Condition | Training | n | B during Test |
|---|---|---|---|---|---|
| Cue Modality | Learning history/Lever presses | P | 12A−, 12B−, 12AB+ | 5 5 |
Unlit Covered |
| PN | 6A−, 6B−, 6AB+, 6C+, 6D+, 6CD− | 5 6 |
Unlit Covered |
||
| PP | 6A−, 6B−, 6AB+, 6C−, 6D−, 6CD+ | 5 6 |
Unlit Covered |
||
| Reinforcement | Learning history/Lever presses | LOW | 12A−, 12B−, 12AB+ | 8 8 |
Unlit Covered |
| HIGH | 6A−, 6B−, 24AB+ | 8 8 |
Unlit Covered |
||
| Uncertainty/Duration | Learning history/Lever presses | PN | 6A−, 6B−, 6AB+, 6C+, 6D+, 6CD− | 8 8 |
Unlit Covered |
| PS | 6A−, 6B−, 6AB+, 3C+, 3C−, 3D+, 3D−, 3CD+, 3CD− | 8 8 |
Unlit Covered |
||
| DH Involvement | Test strategy/cFos | P | 12A−, 12B−, 12AB+ | 8 8 |
Unlit Covered |
| N | 12A+, 12B+, 12AB− | 8 8 |
Unlit Covered |
||
| Homecage | 12A+, 12B+, 12AB− | 8 | NA | ||
| Cholinergic Modulation | Pharmacology/Lever presses | PN | 6A−, 6B−, 6AB+, 6C+, 6D+, 6CD− | 8 8 |
Unlit: PBS/Scop Covered: PBS/Scop |
| PS | 6A−, 6B−, 6AB+, 3C+, 3C−, 3D+, 3D−, 3CD+, 3CD− | 8 8 |
Unlit: PBS/Scop Covered: PBS/Scop |
A and B: visual cues (counter-balanced). C and D: auditory cues (counter-balanced). The ‘− ‘ and ‘+’ sign refer to nonreinforced and reinforced trials, respectively. Subscripts indicate the number of trials presented (quasi-randomly) during each training session. Tests with A- trials were conducted between-subjects with B either explicitly absent (Unlit) or ambiguous (Covered by an opaque shield). Pharmacological manipulations were conducted within-subject via microinfusions of phosphate buffered saline (PBS; vehicle) or the muscarinic antagonist scopolamine (Scop) prior to test sessions.
Apparatus
Eight experimental MedAssociates (Georgia, VT) chambers (30L × 25W × 20H cm) equipped with plexiglass front and back walls and ceiling, aluminum side walls, and stainless-steel rod floors were housed in sound and light insulating chests. A food niche equipped with a water-dipper to deliver sucrose solution (20%) was positioned centrally on one side wall. A 3.5-cm wide lever was positioned 1 cm left of the niche, a 28-V incandescent bulb was located 9 cm left of the niche (Cue A or B, counterbalanced), and an incandescent light flashed at 2 Hz was located 9 cm right of the niche (Cue B or A; Fig. 1A). Ventilation fans and a white noise generator outside of the enclosures provided constant background noise (62 dB). One speaker located on the left wall delivered a 3000 Hz tone, 8 dB(A) above background and another speaker on the ceiling delivered a 4 Hz click-train 8 dB(A) above background (stimuli C and D, counterbalanced). A 4-cm square, stainless steel block affixed to a 7.8-cm × 4.1-cm aluminum plate served as the test cover.
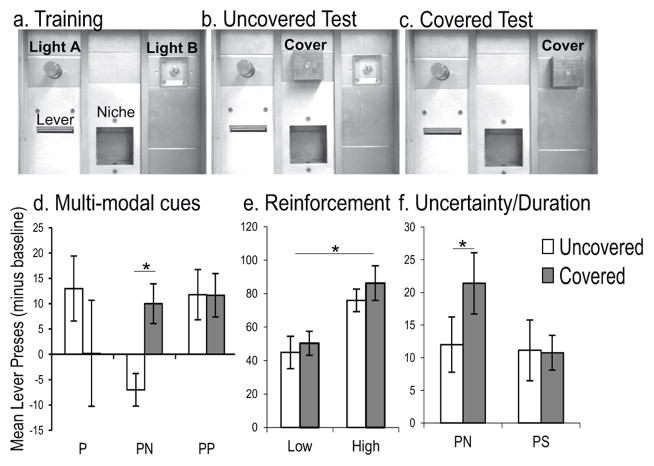
Fig 1. Negative-patterning training is necessary for rats to infer the presence of a hidden cue.
Rats learned to discriminate between illumination of one or two lights (A; lights A and B, counterbalanced), before being tested with only A lit while B either was uncovered and unlit (B) or covered by an opaque shield (C). Lever-presses during test trials minus responses that occurred during the 30 s prior to cue onset (baseline) are illustrated, error bars represent the standard error of the mean and ‘*’ denotes significant differences between conditions. Average responses to A-alone trials did not differ between groups at the conclusion of training (not shown). (D) PN rats (n=11) learned positive- and negative-patterning with visual and auditory cues, respectively. These rats responded more when B was covered compared to uncovered and unlit at test (p = .01). Mere exposure to a secondary auditory discrimination (positive-patterning, PP, n=11) did not cause rats to treat the test conditions differently. PP rats showed similar insensitivity as rats that had only learned a single positive-patterning discrimination (P, n=10) with the visual cues. (E) Although increasing the ratio of reinforced to nonreinforced trials (2:3; HIGH; n=16) during positive-patterning training caused rats to commit more lever presses overall than rats that had experienced a LOW ratio (1:3; n=16), neither group responded differently when B was covered compared to uncovered. (F) Rats trained for the equivalent duration as others learning positive- and negative-patterning (PN, n=16) but with an unsolvable, auditory pseudo-discrimination (PS, n=16) failed to treat the tests differently. Collectively, these result indicate cognitive processes more complex than those impacted by cross-modal cue exposure, amount of reinforcement, and experience with cue uncertainty or training duration must account for the disparity in sensitivity to ambiguity between rats trained with positive- and negative-patterning.
Behavioral Procedures
Sessions terminated after 60 min (unless otherwise noted) with a mean inter trial interval of 70 s (range: 40 to 100 s). Discriminative cues were presented for 30 s. Lever-presses were measured 30 s prior to cue onset and during the 30-s stimuli.
Rats received two days of training to approach and drink sucrose by delivering it every 20 ± 15 s. They were then shaped to press the lever by delivering sucrose for 10-s every 120 s and immediately following every lever press (CRF) for two sessions. Noncontingent reinforcement was removed in three following sessions and an average of every four lever presses was reinforced (VR-4; range: 1 to 7). Discrimination training began on the following session. Trials were quasi-randomly selected during each session with restrictions regarding consecutive presentations of the same trial type. A CRF schedule was gradually reduced to VR-4 across sessions. Multiple 5-s reinforcements were delivered during each trial if the subject repeatedly met the schedule of reinforcement while the cue was present.
Training sessions included 36 trials. Rats assigned to groups P, LOW, HIGH, N, and Homecage were trained with a single visual discrimination with trials restricted to no more than four of the same type consecutively. Groups P and LOW received positive-patterning training with12 A−, 12 B−, and 12 AB+ trials. Group HIGH also received positive-patterning training but with only 6 A−, 6 B−, and 24 AB+ trials. Groups N and Homecage received 12 A+, 12 B+, and 12 AB− trials serving in a negative-patterning discrimination. Rats assigned to groups PN, PP, and PS were trained with concurrent visual and auditory discriminations involving 6 trials of each type with the restriction that no more than two of the same type could occur consecutively. Visual trials included positive-patterning A−, B−, and AB+ trials. Group PN also received negative-patterning (C+, D+, CD−) trials with the auditory cues while Group PP received positive-patterning (C−, D−, CD+) and Group PS received 3 C+, 3 C−, 3 D+, 3 D−, 3 CD+, and 3 CD− auditory trials.
A discrimination ratio (DR) was calculated for each stimulus modality by subtracting the mean elevation score (trial – pretrial responses) on nonreinforced trials from the mean elevation score on reinforced trials and dividing by the mean elevation scores across all trial types. For example, the DRvisual = [mean AB − (mean A + mean B)/2] / [mean AB + (mean A + mean B)/2]. DRs ranged between −1 and +1 with 0 representing no discrimination and +1 indicating perfect performance. Rats were required to meet DR criterion based on the training discrimination for three out of five consecutive training days, including the two most recent. Rats trained with only a visual discrimination (P, LOW, HIGH, N, and Homecage) were required to obtain DRs greater than or equal to 0.65. One Homecage subject failed to meet this criterion and was excluded from all analyses. Groups PN and PP were required to meet DRs greater than or equal to 0.50 for the visual task and 0.20 for the auditory task. For testing purposes, each PS subject was yoked to a PN subject such that whenever the PN rat met test criterion, a PS rat with a DR greater than or equal to 0.5 on the visual task advanced to test. Because sessions were conducted six days a week, subjects never tested following a break, but instead received one additional training session and advanced to test on the following session if they again met criterion. Two subjects in Groups PN and PS failed to reach test criterion after 100 days of training and were omitted from analyses. One additional PN subject was removed due to health complications.
Test
After demonstrating mastery of the training discrimination, according to the DR criterion described above, rats were tested with only one visual cue presented while the second cue remained unlit (explicitly absent), or covered by an opaque shield (ambiguous). Rats trained with only a visual discrimination (P, LOW, HIGH, and N) experienced a single 20-min test session with 12 A- trials. The light bulb that had served as B was replaced by the stainless steel cover for rats assigned to Covered conditions. We controlled for generalization decrement (Ghirlanda and Enquist, 2003) by placing the cover between lights A and B during Uncovered tests while B remained unlit. Homecage rats were not tested, but were transported to the room housing the chambers and then returned to the vivarium where they remained for the following 90 mins.
Rats trained with a secondary auditory discrimination (PN, PS, PP) received 10-min test sessions involving 6 A- trials while B was either covered or uncovered and unlit. Rats tested following microinfusion into DH experienced three separate test sessions after microinfusions of PBS, scopolamine, and PBS, in that order. A minimum of one training session separated each test to ensure testing was not disruptive to discrimination. Subjects were required to meet test criterion (described above) before re-testing. Rats were given a 7 day break following scopolamine administration.
Immunohistochemistry
Seventy minutes following completion of the test session (or 90 min after Homacage transport), P, N, and Homecage rats were anesthetized and perfused intracardially with PBS, followed by 4% paraformaldehyde (PFA). Brains were extracted and cryoprotected in 30% sucrose solution for a minimum of 72 hours before being frozen (−80° C) and sectioned (40 μm) on a cryostat. Every third sample (120 μm) within the DH (mean −3.00 mm to bregma) was collected and stored in PBS at 4° C. Free-floating tissue samples were washed in PBS (3 successive 10 min rinses), incubated in 50% ETOH for 20 minutes, and 50% ETOH + 3% H2O2 for an additional 20 minutes, before receiving 3 additional 10 min PBS rinses. Sections were then incubated at room temperature for 1 hour in blocking solution (10% normal goat serum + .3% Triton-X 100 in 1x PBS), and at 4° C for 72 hours in rabbit antiserum against cFos (1:2000; Santa Cruz). Sections underwent 3, 10 min PBS rinses followed by two additional 10 minute washes in blocking solution before being incubated overnight in biotinylated goat anti-rabbit IgG (1:250; Invitrogen). After washing (PBS, 3, 10 min washes), sections were incubated in ABC reagent (avidin biotin complex kit, Vector Labs) at room temperature for 1 hour, washed (PBS, 3, 10 min washes), and reacted in diaminobenzidine (DAB; approximately 5 minutes) to reveal neurons labeled for cFos in brown. Sections were then washed (PBS, 3 washes at 10 min each) and nissl bodies were counterstained using Hematoxylin QS (HQS; Vector Labs), mounted on microscope slides, and coverslipped using Vectashield (Vector Laboratories, Burlingame, CA).
CFos Quantification
Tissue images were captured at 20 X optical magnification using brightfield microscopy. One sample per subject per region of interest (ROI) was selected blind to the experimental condition on the basis of tissue quality. A universal filter, applied through ImageJ software (Rasband, 1997; Abramoff et al., 2004) normalized background and non-specific staining. CFos positive cells were counted manually and independently using ImageJ in a quasi-random sample of imaged tissue by three researchers, blind to the experimental conditions. The same samples were submitted to ImmunoRatio software (Tuominen et al., 2010) to identify the percentage of area of the image occupied by the brown color associated with cell bodies stained positive for cFos (DAB) relative to total area of purple color associated with other cell bodies in the sample (counter-stained HQS). This measure produced high inter-rater-reliability with manually counted cell bodies and quasi-random sampling confirmed ImmunoRatio stain classification accuracy. Thus all samples were subjected to analysis with ImmunoRatio, maintaining identical threshold settings for DAB and for HQS across all samples. The percentage of cFos positive cell area determined by ImmunoRatio was used as the dependent variable for statistical analysis.
Surgery
After meeting test criterion (but before testing), rats were given a minimum of 3 days free access to food before undergoing surgery. Rats were anesthetized with isoflurane in oxygen (1.5% in O2) and treated with atropine (subcutaneous, 0.1 mg/kg) before being mounted in a stereotaxic frame (Kopf Instruments, Tujunga, CA). Their heads were shaved and eyes were treated with artificial tears (Akwa tears) before their scalps were cleansed with alternating wipes of Betadine and 70% ETOH and incised with the fascia retracted. Bregma and lambda were positioned in the same horizontal plane before bilateral stainless steel guide cannulae were lowered into place (−3.7 mm from bregma, ±2.5 mm from midline, and −2.5 mm ventral from skull) through small burr holes and held in place with dental acrylic. Dummy cannulae (33-guage, 7 mm; PlasticsOne, Roanoke, VA) were inserted into the guides and replaced daily. Carprofen was administered subcutaneously (5mg/kg) once per day for 48 hours following surgery and the antibiotic trimethoprim sulfate (TMS) was dissolved in the rat’s drinking water (0.5 mg/ml) for one-week following surgery. Rats were maintained on an ad lib diet for a minimum of 5 days post-surgery before being progressively restricted to no less than 85% their free-feeding body weight and resuming behavioral sessions. Rats were again required to meet discrimination criterion before advancing to test.
Microinfusion Procedures
Infusions occurred 20 minutes prior to behavioral sessions and were conducted according to Gale et al. (2001). Rats received bilateral microinfusions of either scopolamine (50μg/μl) or PBS, infused at 0.25 μl/min for 2 minutes with internals left in place for an additional minute to facilitate diffusion. Rats were given two days of habituation to the infusion context and manual restraint with dummy cannulae removed and internal cannulae (33-guage, 8 mm) connected to PE-20 polythylene tubing inserted into the implanted guides. Tubing was connected to 10-μl Hamilton syringes and mounted on an infusion pump (Harvard Apparatus, South Natick, MA).
Histology
Cannulae placements were assessed after all sessions were completed. Rats were anesthetized and perfused intracardially, their brains were extracted, cryoprotected, and frozen, as described above. Coronal slices (40 μm) including the entire width of the cannula tract were collected and mounted on microscope slides. Tissue was stained for nissl bodies (cresyl violet) and imaged with brightfield microscopy (Zeiss, Oberkochen, Germany).
RESULTS
Instrumental lever-pressing in rats was assessed during tests with one training cue, A, illuminated while a second training cue, B, was unlit (explicitly absent; Fig. 1B) or covered by an opaque shield (ambiguous; Fig. 1C). Bonferroni corrections were made wherever multiple unplanned comparisons were performed.
Learning history influences behavior during ambiguity
We hypothesized that the configural or rule-based strategy of negative-patterning transfers to how the rat approaches positive-patterning in the ambiguous test. A univariate analysis of variance (ANOVA) computed on lever presses at test between subjects that did (PN) and did not (P, PP) learn negative-patterning and the two test conditions (B Covered or Uncovered) revealed a Training X Test interaction, F(1, 28) = 5.201, p = .03. Independent-samples t-test planned comparisons revealed that PN rats responded more when light B was covered compared to unlit, t(9) = 3.264, p = .010, d = 2.176 (Fig. 1D). This behavior is consistent with the rats inferring the presence of the hidden light because compound AB trials were always reinforced during positive-patterning training. However, rats that did not learn negative patterning failed to treat the two test conditions differently (p > .38). Importantly, learning two separate positive-patterning discriminations with visual and auditory cues (PP) did not cause rats to respond differently when the cue was covered compared to unlit, (p > .33), indicating mere exposure to multiple discriminations across two modalities is insufficient for rats to behaviorally distinguish between the explicit and ambiguous absence of a cue. Although increasing the ratio of reinforced to nonreinforced positive-patterning training trials (A−, B−, AB+; Group HIGH) to match the ratio experienced under negative-patterning contingencies (A+, B+, AB−) caused rats to respond more to both reinforced and nonreinforced trial types, it did not cause these rats to behaviorally distinguish between test trials when B was covered compared to unlit. An ANOVA computed on test responses between the variables of training reinforcement and test condition supported these observations, revealing a main effect of Reinforcement, F(1, 28) = 15.132, p = .001, but no main effect of Test condition nor interaction between these variables, (Fs < 1). Finally, because negative-patterning is empirically more difficult (Harris et al., 2009; Fast and Blaisdell, 2011) we controlled for experience with uncertainty related to cue-reinforcement contingencies and training duration by exposing rats to an auditory pseudo-discrimination in addition to visual positive-patterning (Group PS). The pseudo-discrimination controlled for experience with combinations of auditory cues and reinforcement without providing a patterning solution by reinforcing each cue and combination of cues half of the time they were presented (C±, D±, CD±). Because this discrimination is insoluble, it also allowed us to control for training duration by testing PS subjects when a PN rat, to which it was yoked, demonstrated mastery over positive- and negative-patterning with visual and auditory cues, respectively. Nonetheless, PS rats failed to treat tests with B covered differently than B unlit (Fig. 1F). An ANOVA computed on test responses between training (PN or PS) and test (B Covered or Uncovered) conditions revealed a significant Train X Test interaction, F(1, 23) = 4.804, p = .039, while independent-samples t-test planned comparisons revealed that only PN rats responded differently when B was Covered at test compared to Uncovered and unlit, t(11) = 2.456, p = .032, d = 1.48. Collectively, these results suggest rats can infer the presence of hidden events, but only if they have experience with negative-patterning.
DH cFos corresponds with inference about a hidden cue
Rats learned either positive- or negative-patterning with visual cues (Fig. 2A). A mixed ANOVA computed on lever press responses at the conclusion of training with trial type (elemental or compound) as within-subjects factors and training type (positive- or negative-patterning) as between-subjects factors revealed a significant Trial Type X Training interaction, F(1, 33) = 888.931, p < .001. Planned t-test comparisons revealed that P rats successfully performed positive-patterning by responding significantly more to the reinforced compound trials than the nonreinforced elemental trials, t(13) = 18.033, p < .001, d = 10.003, while N rats successfully performed negative-patterning by responding less to the nonreinforced compound trials than to the reinforced elemental trials, t(20) = 24.884, p < .001, d = 11.128.
Fig 2. DH cFos corresponds with inference about a hidden cue.
Behavioral results from training (A) and test (B) and cFos expression in the dentate gyrus (DG) of the dorsal hippocampus following test (C). Error bars represent the standard error of the mean (SEM) and ‘*’ denotes significant differences between conditions. At the conclusion of training (A), P (n=16) demonstrated positive-patterning by lever pressing more during compound (black bars) than elemental trials (white bars) while N (n=16) performed negative-patterning by lever pressing less to compound than elemental trials. (B) P rats responded similarly to A-alone tests when B was Covered (gray bars) and Uncovered (white bars) while N responded significantly less when B was Covered compared to Uncovered. Importantly, N and P did not respond differently when B was Covered. Despite committing equivalent lever presses during test, N-Covered (n=8) showed significantly elevated cFos expression in dentate gyrus (DG) compared to all other conditions (C). Representative samples of cFos expression in DG for each test group D) P-Uncovered E) P-Covered F) N-Uncovered G) N-Covered; (excluding Homecage controls). CFos positive cells appear brown (DAB), all others appears blue or purple (Hematoxylin QS).
Because P rats in this experiment were not exposed to negative-patterning, they were not expected to respond differently during uncovered and covered tests. Therefore, P rats controlled for neural behavior related to sensory aspects of the test. N rats, on the other hand, were expected to respond less when B was covered compared to unlit. Although a reduction in lever presses contrasts with the increased responses demonstrated in the above experiments, it remains consistent with the rat inferring the presence of the hidden light. In the above experiments, rats that distinguished between the explicit and ambiguous tests responded more because compound AB trials were always reinforced during training (positive-patterning with visual cues). However, compound AB trials were never reinforced during training for N rats in the current experiment (negative-patterning with visual cues). Importantly, instrumental behavior was not expected to differ between P and N rats tested with B covered in the current experiment. Thus, comparison of cFos expression profiles between these conditions controlled for any contribution from motor activity while providing an index of neural involvement in inference behavior during the ambiguous test situation.
Further supporting our hypothesis that negative-patterning is necessary for inferences about hidden cues, an ANOVA computed on test responses revealed a Training X Test condition interaction, F(1, 30) = 5.277, p = .029, with planned comparison t-tests indicating that only N rats treated the test situations differently by responding less when B was covered than unlit (t(14) = −2.446, p = .028, d = 1.307, Fig. 2B). This behavior is consistent with the rats inferring the presence of the hidden light because compound AB trials were never reinforced during training. Importantly, behavior between P-Covered and N-Covered did not differ, t(14) = −1.443, p = .171, d = 0.771, thus differences in cFos cannot be attributed to motor activity.
A labeling index, calculated by taking the percentage of DAB-stained (cFos+) area over total nuclear area (DAB + Hematoxylin QS) within regions of interest (ROI) was analyzed with a mixed ANOVA between test conditions. ROIs included CA1, CA2, CA3, and dentate gyrus (DG) of the DH. An ROI X Condition interaction, F(28, 203) = 2.273, p = 5.8 × 10−4, was further analyzed with univariate ANOVAs between conditions for each ROI. There was a main effect of training in CA1, F(1, 33) = 5.654, p = .023, with N rats showing a greater labeling index (M = 9.421, SD = 2.373) than P rats (M = 7.719, SD = 1.521), regardless of test condition; suggesting experience with negative-, but not positive-patterning, engages CA1. No significant differences were found in CA2 or CA3; however, a main effect of Test was found in DG, F(2, 33) = 4.804, p = .015, along with a Train X Test interaction, F(1, 33) = 6.620, p = .015. Planned contrasts with a Bonferroni correction for the five comparisons between training and test conditions revealed that, although behavior did not differ, N-Covered displayed a higher labeling index than P-Covered, t(14) = −2.747, p = .005, d = 1.468 (Fig. 2C). This result indicates DG involvement in the inference process engaged during the ambiguous situation.
Lever-presses at test were subtracted from training lever-presses as an index of decision-making strategy engaged during the test (excluding Homecage). This measure was positively correlated with cFos expression in CA2, r = 0.376, p = .022, indicating that changes in instrumental behavior at test, compared to training, were associated with a selective increase in cFos expression in CA2. Only group N-Covered demonstrated significant changes in behavior between training and test, suggesting the correlation with CA2 was driven by subjects in this condition.
To characterize expression profiles, we examined the correlation in cFos expression between all ROIs for each test condition, with a Bonferroni correction for the six ROI comparisons. CFos expression in DG and CA1 were correlated, r = 0.865, p = .003 for N-Covered, but not N-Uncovered (expression normalized between conditions), suggesting an increased concordance in activity between DG and CA1 may mediate sensitivity to ambiguity. Additionally, N-Uncovered (but not N-Covered) showed a correlation between cFos expression in CA2 and CA1, r = 0.874, p = .005. Given that CA2 activity was positively correlated with change in behavior during test, it is possible that CA2 influences the correspondence between DG and CA1 under conditions of uncertainty (Kohara et al., 2014).
DH cholinergic modulation mediates inference
Rats learned a visual positive-patterning discrimination along with an auditory negative-patterning (PN), or pseudo-discrimination (C±, D±, CD±; PS). When training concluded, lever presses to visual A-alone trials did not differ between the conditions, F(3, 23) = 0.947, p = .434, thus any differences during subsequent tests could only be attributable to the test conditions themselves.
Tests involved probe trials of A while B either was covered or unlit. Each subject experienced a series of three separate tests (Fig. 3A) following microinfusion of phosphate buffered saline (PBS; Tests 1 and 3) or scopolamine (Test 2) into the DH (Gale et al., 2001). A univariate ANOVA computed on lever press responses between the first and second PBS tests failed to reveal any significant differences, ps > .06. An additional mixed ANOVA with session (training, vehicle test, and scopolamine test) as a within-subject factor and test condition as a between-subject factor revealed a significant Session X Test interaction, F(6, 46) = 2.756, p = .023. Subsequent t-test comparisons explored this interaction with a Bonferroni correction for multiple comparisons adjusting alpha to .0167. As reported above, only PN rats responded differently when positive-patterning cue B was covered during vehicle (PBS) tests (Fig. 2B) compared to A-alone training trials when B was explicitly absent, t(6) = −3.639, p = .011, d = 2.971. Specifically, these rats lever-pressed more when B was covered compared to training and test trials with B unlit. Again, this behavior is consistent with inference about the perceptually-absent cue B when it was covered because lever-presses were always reinforced when they occurred during compound AB training trials, but never during A-alone trials. As described above, PS rats trained with the auditory pseudo-discrimination did not respond differently from training, regardless if B was covered or not, p > .05.
Fig 3. DH cholinergic modulation mediates inference of hidden cues.
Rats received three tests, across separate days (A). Mean lever presses occurring during 30-s A-alone tests minus baseline responses are illustrated, error bars represent the standard error of the mean and ‘*’ denotes significant differences between conditions. White bars reflect tests with B Uncovered and gray bars illustrate tests with B Covered. (B) The cover position did not affect PS rats’ responses (n=16), while PN rats responded more to A when B was Covered (n=8) compared to when B was Uncovered (n=8) or A-alone training trials following microinfusions of phosphate buffered saline (PBS), but not scopolamine (Scop, C).
Importantly, scopolamine administered prior to test significantly reduced PN test responses with B covered compared to vehicle microinfusions, t(6) = 3.610, p = .011, d = 2.948, effectively returning behavior to levels demonstrated during A-alone training trials, t(6) = 1.211, p = .272. That is, scopolamine eliminated the ability of PN rats to infer the presence of the hidden light. Scopolamine had no effect on lever-presses when B was unlit, or in Group PS, ps > .05. Thus, scopolamine selectively disrupted the rats’ ability to infer the presence of the hidden light when it was covered at test.
To ensure scopolamine had not influenced motor function, visual processing, instrumental motivation, or underlying positive-patterning abilities, three patterning assessment sessions (identical to training) followed microinfusions of PBS or scopolamine (Fig. 4A). A univariate ANOVA computed on discrimination ratios (DRs) for the positive-patterning discrimination between the two PBS vehicle sessions failed to reveal any significant differences, ps > .06. A mixed ANOVA computed on positive-patterning DRs with vehicle and scopolamine sessions as a within-subject factor and auditory training (negative-patterning or pseudo-discrimination), revealed a main effect of Session, F(1, 24) = 6.803, p = .015; however, post-hoc t-test comparisons, with alpha adjusted to .0167 using the Bonferroni method to correct for multiple comparisons, revealed that DRs did not differ from test criterion (.65) and all rats continued to respond significantly more to compound trials than during elemental trials following microinfusions of both vehicle, t(26) = 16.247, p < .001, d = 6.373, and scopolamine, t(26) = 5.764, p < .001, d = 2.261. Importantly, these results indicate that rats successfully performed positive-patterning following localized microinfusion of scopolamine (Fig. 4C), thus scopolamine’s disruption of test performance (reported above) was not driven by an underlying deficit in visual positive-patterning discrimination.
Fig 4. Scopolamine localized in DH disrupts negative-patterning but not visual processing, instrumental motivation, or motor skills.
(A) Three patterning assessment tests were conducted after rats met performance criterion during “Refresh” sessions (identical to training). These “Patterning Sessions” occurred 20 minutes after microinfusion of phosphate buffered saline (PBS) or scopolamine (Scop). Rats did not participate in experimental procedures for 1 week following scop infusion. Mean lever presses during the 30-s visual and auditory discriminative cues, minus baseline, are presented in panels b–e. Error bars represent the standard error of the mean (SEM) and ‘*’ denotes significant differences between conditions. Both PN (n=16) and PS rats (n=16) successfully solved the visual-positive-patterning discrimination following microinfusion of PBS (B) and Scop (C). PS rats maintained non-biased responding to elemental and compound auditory trials after PBS and Scop (D, E, respectively). PN rats successfully performed auditory-negative-patterning following microinfusion of PBS (D) but not following microinfusion of Scop (E). Given visual and auditory trials were intermixed within a single 60 minute session, these results indicate that the disruption to negative-patterning was not due to altered motor abilities or motivation because these same rats showed intact visual positive-patterning (C).
A second univariate ANOVA was computed on DRs for the auditory discrimination (negative-patterning for Group PN and pseudo-discrimination for PS) between the two training conditions, failing to reveal any differences between the first and second vehicle session (p > .07). A mixed ANOVA on auditory DRs with infusion type (vehicle or scopolamine) as a within-subject factor and auditory training as a between-subjects factor failed to reveal an interaction between Infusion type and Auditory training. Because the appropriately low DRs for the pseudo-discrimination may have masked any effect of scopolamine on negative-patterning abilities, paired-samples t-tests were conducted to compare DRs following vehicle and scopolamine microinfusions separately for PN and PS rats. Scopolamine did not influence performance on the pseudo-discrimination for PS, t(12) = 1.663, p = .122, d = 0.960; however, similar to systemic administration (Moran, 1992; Richmond et al., 1997), scopolamine microinfused into the DH significantly impaired negative-patterning, t(12) = 2.477, p = .029, d = 1.430 (Fig. 4E). Given that both visual and auditory trials occurred intermixed within each patterning assessment session, and PN rats successfully discriminated visual positive-patterning cues, we can conclude that the disruption to negative-patterning was not produced by changes in motor function or instrumental motivation caused by scopolamine. That is, blocking cholinergic modulation within the DH (Fig. 5) selectively disrupted negative-patterning abilities in addition to inferences about the hidden positive-patterning light B.
Fig 5. DH cannulae placement.
Coronal images from Paxinos and Watson (2005) with coordinates indicating distance (mm) from bregma. Closed circles represent cannulae placement for subjects trained on positive- and negative- patterning with visual and auditory cues, respectively (Group PN). Open circles represent cannulae placement of subjects trained with visual positive-patterning and an auditory pseudo-discrimination (Group PS).
DISCUSSION
Rats not only distinguish between the explicit and ambiguous absence of relevant events, they also draw on previous experience with the event that is hidden to make behavioral decisions. Our rats learned to discriminate between elemental (A or B) and compound (AB) presentations of two light cues. They distinguished between A-alone probe trials when light B was explicitly absent (unlit; resembling A-alone training trials) and ambiguous (covered by an opaque shield) at test if and only if they had experience with negative-patterning. Exposure to multiple discriminations, reinforcement history, experience with cue uncertainty, and training duration were insufficient factors to change behavior when a relevant positive-patterning cue was subsequently occluded from view. The behavioral pattern of rats tested with ambiguous positive- and negative-patterning cues is consistent with the rat inferring the presence of the hidden light B. Simultaneous presentations of A and B were always reinforced during positive-patterning training (AB+) but not during negative-patterning (AB−). Likewise, tests with an occluded positive-patterning cue increased behavior (similar to AB+) while an occluded negative-patterning cue reduced behavior (similar to AB−) compared to when B was unlit (explicitly absent).
Importantly, while our rats treated the explicit absence of a cue differently than the ambiguous absence of the same cue, we report elsewhere (Fast et al., 2015) that rats do not respond differently to a cue that is occluded (ambiguous) compared to explicitly present (lit) during extinction trials. In Experiment 1, rats were trained with an appetitive Pavlovian compound stimulus (AB+) before receiving A-alone probe trials while B was either uncovered (explicitly absent) or covered (ambiguous). As a control, subjects also received extinction trials with the trained AB compound. Although extinction may represent the phenomenon most closely linked to ambiguity, rats in this experiment treated extinction trials of AB identically to training trials but had a pronounced decrement in responding during A-alone (with B Uncovered) trials. The generalization decrement between AB and A-alone was attenuated when A occurred while B was Covered (ambiguous). These results are internally consistent with the results reported here, and further demonstrate that rats respond differently when a relevant stimulus is blocked from physical detection compared to when the status (both present and absent) of the same stimulus is explicit. This behavioral distinction between extinction and ambiguity corresponds to the observation that lesions of the hippocampus do not typically affect extinction. Thus, the behavioral differences reported here are consistent with the rats inferring the status of the ambiguous light rather than simply distinguishing between reinforced training trials and nonreinforced test trials. Collectively, our results support the hypothesis that the rule-based or configural approach recruited by tasks, such as negative-patterning, is necessary for rats to infer the presence of a hidden cue.
One way the rat might make this inference is through a retrieved representation (Konorski, 1967; Holland, 1981) of the hidden cue. Contemporary research on representation-mediated conditioning procedures (Pickens and Holland, 2004) demonstrate the rat’s ability to retrieve representations of events that are not physically present. These retrieved-representations are capable of substituting for the physical occurrence of a corresponding event to drive learning. Although our results suggest negative-patterning is necessary for rats to make a behavioral inference under ambiguity, representation-mediated conditioning procedures typically do not involve, nor require, negative-patterning training for representation retrieval to occur. Interestingly, other work in our lab (Blaisdell et al., 2009; Waldmann et al., 2012) indicates rats are similarly sensitive to ambiguity after learning simple Pavlovian associations. Importantly, these Pavlovian (Nicholson and Freeman, 2000) and representation-mediated-conditioning procedures rely on within-compound or cue-cue associations (Liljeholm and Balleine, 2009). Similar within-compound associations are believed to influence configural strategies (Rescorla, 1973; Pearce, 1994; Liljeholm and Balleine, 2009) such as those recruited by negative- (but not positive-) patterning (Rudy and Sutherland, 1995). Cue-cue associations could enable presentation of one cue to retrieve a representation of the second cue. Under normal conditions, the evoked representation may direct attention toward this second cue in the environment, whose explicit absence could adjust expectancies. However, when the status of the modulatory second cue is ambiguous (covered), the evoked representation (and related expectancies) remain active. Thus, the ability for rats to flexibly behave according to an inference under conditions of uncertainty may depend on retrieval of a representation that is mediated by cue-cue associations. Our results further suggest that the dorsal hippocampus plays a crucial role in this ability. Interestingly, others (Iordanova et al., 2011, Zeithamova et al., 2012b) have also implicated the dorsal hippocampus in some forms of representation-mediated-learning.
Despite the high demand to make decisions under ambiguity, this ability deteriorates disproportionately with age and may correspond to deficits in cue-cue associations (Mutter et al., 2012) in humans. Also consistent with the human condition, we found expression of the immediate early gene cFos was correlated between dentate gyrus (DG) and CA1 for rats demonstrating behavioral sensitivity to the ambiguous test, similar to CA1 activity found when humans make an inference (Schlichting et al., 2014). Elevated cFos expression in DG-alone suggests DG may be critical to inferences committed during ambiguous situations. Importantly, DG involvement was not driven by motor responses or sensory experiences during the ambiguous test situation, but was specific to the inference process engaged. Finally, cFos in CA2 was positively correlated with our behavioral index of inference. Coupled with the discovery that CA2 (Wintzer et al., 2014) may be sensitive to small environmental changes, our results suggest CA2 may play an important role in detecting ambiguous situations to enable flexible adaptations in behavior, a function that warrants further empirical scrutiny.
Aging related cognitive deficits may be mitigated by enhancing cholinergic function (Jack et al., 1998; Fisher, 2011; Boyle et al., 2012; Grothe et al., 2012; Haense et al., 2012; Jacus et al., 2013), which is prevalent in the hippocampus (Frotscher and Léránth, 1985; Woolf et al., 1984). Our results also mirror this relationship. Scopolamine localized in the DH disrupted the rats’ sensitivity to ambiguity (without affecting absolute lever-pressing) and likewise disrupted negative-patterning performance. Importantly, scopolamine did not affect performance on visual positive-patterning, indicating the disrupted test performance was not due to an underlying deficit in visual discrimination but was specific to the inference process invoked during the ambiguous test. These results also suggest that similar strategies recruited by negative-patterning may mediate sensitivity to ambiguity and rely on cholinergic and hippocampal integrity. Collectively, our results suggest that deficits in decision making during conditions of uncertainty may be alleviated, or protected from erosion, through behavioral interventions that emphasize configural or rule-based processing or treatments that otherwise enhance cholinergic modulation in the hippocampus.
Acknowledgments
Support was provided by Grant Sponsor: NSF, Grant number: BCS-0843027 (APB), Grant Sponsor: NIA, Grant number: R36AG045410 (CDF), Grant Sponsor: NIMH, Grant Number: R01MH062122 (MSF), and an American Psychological Association Dissertation Award (CDF). The authors wish to thank Michelle Cho, Maria Cornejo, Patricia Stan, Esther Yang, and James Yang for their assistance with data collection.
References
- Abramoff MD, Magalheas PJ, Ram SJ. Image processing with ImageJ. Biophotonics International. 2004;11:36–42. [Google Scholar]
- Alvarado MC, Rudy JW. A comparison of kainic acid plus colchicines and ibotenic acid-induced hippocampal formation damage in four configural tasks in rats. Behav Neurosci. 1995;109:1052–1062. doi: 10.1037//0735-7044.109.6.1052. [DOI] [PubMed] [Google Scholar]
- Barron HC, Dolan RJ, Behrens TEJ. Online evaluation of novel choices by simultaneous representation of multiple memories. Nat Neurosci. 2013;16:1492–1498. doi: 10.1038/nn.3515. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Blaisdell AP, Leising KJ, Stahlman WD, Waldmann MR. Rats distinguish between absence and lack of information. Int J Comp Psychol. 2009;22:1–18. [Google Scholar]
- Ghirlanda S, Enquist M. A century of generalization. Anim Behav. 2003;66:15–36. [Google Scholar]
- Boyle PA, Yu L, Wilson RS, Gamble K, Buchman AS, Bennett DA. Poor decision making is a consequence of cognitive decline among older persons without Alzheimer’s disease or mild cognitive impairment. PLoS ONE. 2012;7:1–5. doi: 10.1371/journal.pone.0043647. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Butt AE, Noble MM, Rogers JL, Rea TE. Impairments in negative patterning, but not simple discrimination learning, in rats with 192 IgG-Saporin lesions of the nucleus basalis magnocellularis. Behav Neurosci. 2002;116:241–255. doi: 10.1037//0735-7044.116.2.241. [DOI] [PubMed] [Google Scholar]
- Fanselow MS. From contextual fear to a dynamic view of memory systems. Trends Cogn Sci. 2009;14:7–15. doi: 10.1016/j.tics.2009.10.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fast CD, Blaisdell AP. Rats are sensitive to ambiguity. Psychon Bull Rev. 2011;18:1230–1237. doi: 10.3758/s13423-011-0171-0. [DOI] [PubMed] [Google Scholar]
- Fast CD, Biederman T, Blaisdell AP. Imagine that! Cue-evoked representations guide rat behavior during ambiguous situations. J Exp Psychol [Anim Learn Cog] doi: 10.1037/xan0000093. in press. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fisher A. Cholinergic modulation of amyloid precursor protein processing with emphasis on M1 muscarinic receptor: Perspective and challenges in treatment of Alzheimer’s disease. J Neurochem. 2011;120:22–33. doi: 10.1111/j.1471-4159.2011.07507.x. [DOI] [PubMed] [Google Scholar]
- Frotscher M, Léránth C. Cholinergic innervations of the rat hippocampus as revealed by choline acetyltransferase immunocytochemistry: A combined light and electron microscopic study. J Comp Neurol. 1985;239:237–246. doi: 10.1002/cne.902390210. [DOI] [PubMed] [Google Scholar]
- Gale GD, Anagnostaras SG, Fanselow MS. Cholinergic modulation of Pavlovian fear conditioning: Effects of intrahippocampal scopolamine infusion. Hippocampus. 2001;11:371–376. doi: 10.1002/hipo.1051. [DOI] [PubMed] [Google Scholar]
- Grothe M, Heinsen H, Teipel SJ. Atrophy of the cholinergic basal forebrain over the adult age range and in early stages of Alzheimer’s disease. Biol Psychiatry. 2012;71:805–813. doi: 10.1016/j.biopsych.2011.06.019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Haense C, Kalbe E, Herholz K, Hohmann C, Neumaier B, Krais R, Heiss W-D. Cholinergic system function and cognition in mild cognitive impairment. Neurobiol Aging. 2012;33:867–877. doi: 10.1016/j.neurobiolaging.2010.08.015. [DOI] [PubMed] [Google Scholar]
- Hagmayer Y, Waldmann MR. Inferences about unobserved causes in human contingency learning. Q J Exp Psychol. 2007;60:330–355. doi: 10.1080/17470210601002470. [DOI] [PubMed] [Google Scholar]
- Harris JA, Gharaei S, Moore CA. Representations of simple and compound stimuli in negative and positive-patterning. Learn Behav. 2009;37:230–245. doi: 10.3758/LB.37.3.230. [DOI] [PubMed] [Google Scholar]
- Hata T, Kumai K, Okaichi H. Hippocampal acetylcholine efflux increases during negative-patterning and elemental discrimination in rats. Neurosci Lett. 2007;418:127–132. doi: 10.1016/j.neulet.2007.03.013. [DOI] [PubMed] [Google Scholar]
- Holland PC. Acquisition of representation mediated conditioned food aversions. Learn Motiv. 1981;12:1–18. doi: 10.1016/j.lmot.2008.08.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Iordanova MD, Good M, Honey RC. Retrieval-mediated learning involving episodes requires synaptic plasticity in the hippocampus. J Neurosci. 2011;31:7156–7162. doi: 10.1523/JNEUROSCI.0295-11.2011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jack CR, Jr, Peterson RC, Xu Y, O’Brien PC, Smith GE, Ivnik RJ, Tangalos EG, Kokmen E. Rate of medial temporal lobe atrophy in typical aging and Alzheimer’s disease. Neurol. 1998;51:993–999. doi: 10.1212/wnl.51.4.993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jacus J, Bayard SR, Gély-Nargeot MC. Decision-making and apathy in early stage of Alzheimer’s disease and in mild cognitive impairment. Geriatr Psychol Neuropsychiatr Vieil. 2013;11:215–223. doi: 10.1684/pnv.2013.0406. [DOI] [PubMed] [Google Scholar]
- Kohara K, Pignatelli M, Rivest AJ, Jung H-Y, Kitamura T, Suh J, Frank D, Kajikawa K, Mise N, Obata Y, Wickersham IR, Tonegawa S. Cell-type specific genetic and optogenetic tools reveal hippocampal CA2 circuits. Nat Neurosci. 2014;17:269–279. doi: 10.1038/nn.3614. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Konorski J. Integrative activity of the brain. Chicago: University of Chicago Press; 1967. [Google Scholar]
- Kumaran D. What representations and computations underpin the contribution of the hippocampus to generalization and inference? Front Hum Neurosci. 2012;6:1–11. doi: 10.3389/fnhum.2012.00157. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liljeholm M, Balleine BW. Mediated conditioning versus retrospective revaluation in humans: The influence of physical and functional similarity of cues. Q J Exp Psychol. 2009;62:470–482. doi: 10.1080/17470210802008805. [DOI] [PubMed] [Google Scholar]
- Mehta R, Williams DA. Elemental and configural processing of novel cues in deterministic and probabilistic tasks. Learn Motiv. 2002;33:456–484. [Google Scholar]
- Moran PM. Scopolamine deficits in negative-patterning discrimination: Evidence for a role of the central cholinergic system in retention but not acquisition of non-spatial configural association learning. Behav Brain Res. 1992;48:187–197. doi: 10.1016/s0166-4328(05)80156-1. [DOI] [PubMed] [Google Scholar]
- Mutter SA, Atchley AR, Plumlee LM. Aging and retrospective revaluation of causal learning. J Exp Psychol Learn Mem Cogn. 2012;38:102–117. doi: 10.1037/a0024851. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nicholson DA, Freeman JH., Jr Lesions of the perirhinal cortex impair sensory preconditioning in rats. Behav Brain Res. 2000;112:69–75. doi: 10.1016/s0166-4328(00)00168-6. [DOI] [PubMed] [Google Scholar]
- Paxinos G, Watson C. The rat brain in stereotaxic coordinates. 5. Academic Press; New York: NY: 2005. [DOI] [PubMed] [Google Scholar]
- Pearce JM. Similarity and discrimination: A selective review and a connectionist model. Psychol Rev. 1994;101:587–607. doi: 10.1037/0033-295x.101.4.587. [DOI] [PubMed] [Google Scholar]
- Phillips RG, LeDoux JE. Differential contribution of amygdala and hippocampus to cued and contextual fear conditioning. Behav Neurosci. 1992;106:274–285. doi: 10.1037//0735-7044.106.2.274. [DOI] [PubMed] [Google Scholar]
- Pickens CL, Holland PC. Conditioning and cognition. Neurosci Biobehav Rev. 2004;28:651–661. doi: 10.1016/j.neubiorev.2004.09.003. [DOI] [PubMed] [Google Scholar]
- Rasband WS. ImageJ. U. S. National Institute of Health; Bethesda: MD: 1997. [Google Scholar]
- Reber TP, Luechinger R, Boesiger P, Henke K. Unconscious relational inference recruits the hippocampus. J Neurosci. 2012;32:6138–6148. doi: 10.1523/JNEUROSCI.5639-11.2012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rescorla RA. Evidence for a “unique stimulus” account of configural conditioning. J Comp Physiol Psychol. 1973;79:307–317. doi: 10.1037/h0032553. [DOI] [PubMed] [Google Scholar]
- Richmond MA, Nichols BP, Deacon RMJ, Rawlins JNP. Effects of scopolamine and hippocampal lesions on negative-patterning performance in rats. Behav Neurosci. 1997;111:1217–1227. doi: 10.1037//0735-7044.111.6.1217. [DOI] [PubMed] [Google Scholar]
- Rudy JW, Sutherland RJ. Configural association theory and the hippocampal formation: An appraisal and reconfiguration. Hippocampus. 1995;5:375–389. doi: 10.1002/hipo.450050502. [DOI] [PubMed] [Google Scholar]
- Sakimoto Y, Okada K, Hattori M, Takeda K, Sakata S. Neural activity in the hippocampus during conflict resolution. Behav Brain Res. 2013;237:1–6. doi: 10.1016/j.bbr.2012.09.013. [DOI] [PubMed] [Google Scholar]
- Sakimoto Y, Sakata S. The decline in rat hippocampal theta activity during response inhibition for the compound stimulus of negative-patterning and simultaneous feature-negative tasks. Behav Brain Res. 2013;257:111–117. doi: 10.1016/j.bbr.2013.09.020. [DOI] [PubMed] [Google Scholar]
- Schlichting ML, Zeithamova D, Preston AR. CA1 subfield contributions to memory integration and inference. Hippocampus. 2014;24:1248–1260. doi: 10.1002/hipo.22310. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Taler V, Klepousniotou E, Phillips NA. Comprehension of lexical ambiguity in healthy aging, mild cognitive impairment, and Alzheimer’s disease. Neuropsychologia. 2009;47:1332–1343. doi: 10.1016/j.neuropsychologia.2009.01.028. [DOI] [PubMed] [Google Scholar]
- Tuominen VJ, Ruotoistenmäki S, Viitanen A, Jumppanen M, Isola J. ImmunoRatio: A publicly available web application for quantitative image analysis of estrogen receptor (ER), progesterone receptor (PR), and Ki-67. Breast Cancer Res. 2010;12:1–12. doi: 10.1186/bcr2615. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Waldmann MR, Schmid M, Wong J, Blaisdell AP. Rats distinguish between absence of events and lack of evidence in contingency learning. Anim Cogn. 2012;15:979–990. doi: 10.1007/s10071-012-0524-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Williams DA, Braker DS. Input coding in animal and human associative learning. Behav Processes. 2002;57:149–161. doi: 10.1016/s0376-6357(02)00011-6. [DOI] [PubMed] [Google Scholar]
- Wintzer ME, Boehringer R, Polygalov D, McHugh TJ. The hippocampal CA2 ensemble is sensitive to contextual change. J Neurosci. 2014;34:3056–3066. doi: 10.1523/JNEUROSCI.2563-13.2014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Woolf NJ, Eckenstein F, Butcher LL. Cholinergic systems in the rat brain: I. projections to the limbic telencephalon. Brain Res Bull. 1984;13:751–784. doi: 10.1016/0361-9230(84)90236-3. [DOI] [PubMed] [Google Scholar]
- Zeithamova D, Schlichting ML, Preston AR. The hippocampus and inferential reasoning: Building memories to navigate future decisions. Front Hum Neurosci. 2012a;6:1–14. doi: 10.3389/fnhum.2012.00070. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zeithamova D, Dominick AL, Preston AR. Hippocampal and ventral medial prefrontal activation during retrieval-mediated learning supports novel inference. Neuron. 2012b;75:168–179. doi: 10.1016/j.neuron.2012.05.010. [DOI] [PMC free article] [PubMed] [Google Scholar]