Abstract
The important roles of retinols and their metabolites have recently been emphasized in the interactions between hepatic stellate cells (HSCs) and natural killer (NK) cells. Nevertheless, the expression and role of retinol metabolizing enzyme in both cell types have yet to be clarified. Thus, we investigated the expression of retinol metabolizing enzyme and its role in liver fibrosis. Among several retinol metabolizing enzymes, only alcohol dehydrogenase (ADH) 3 expression was detected in isolated HSCs and NK cells, whereas hepatocytes express all of them. In vitro treatment with 4-methylpyrazole (4-MP), abroad ADH inhibitor, or depletion of the ADH3 gene downregulated collagen and transforming growth factor-β1 (TGF-β1) gene expression, but did not affect α-smooth muscle actin gene expression in cultured HSCs. Additionally, in vitro, treatments with retinol suppressed NK cell activities, whereas inhibition of ADH3 enhanced interferon-γ (IFN-γ) production and cytotoxicity of NK cells against HSCs. In vivo, genetic depletion of the ADH3 gene ameliorated bile duct ligation- and carbon tetrachloride-induced liver fibrosis, in which a higher number of apoptotic HSCs and an enhanced activation of NK cells were detected. Freshly isolated HSCs from ADH3-deficient mice showed reduced expression of collagen and TGF-β1, but enhanced expression of IFN-γ was detected in NK cells from these mice compared with those of control mice. Using reciprocal bone marrow transplantation of wild-type and ADH3-deficient mice, we demonstrated that ADH3 deficiency in both HSCs and NK cells contributed to the suppressed liver fibrosis.
Conclusion
ADH3 plays important roles in promoting liver fibrosis by enhancing HSC activation and inhibiting NK cell activity, and could be used as a potential therapeutic target for the treatment of liver fibrosis.
Keywords: retinol, retinaldehyde, retinoic acid, 4-methylpyrazole, interferon-gamma
Introduction
In normal conditions, quiescent hepatic stellate cells (qHSCs) store up to 80% of body retinols (vitamin A lipid droplets) and contribute to retinol homeostasis.1 During the activation of HSCs, retinol can be oxidized into retinaldehyde and then further metabolized into retinoic acid (RA) by alcohol dehydrogenases (ADHs) and retinaldehyde dehydrogenases (Raldhs), respectively.2 Then, RA binds to nuclear receptors such as RA receptors (RARs) and retinoid X receptors (RXRs), which directly regulate gene expression.3 In addition to ADHs, microsomal short-chain dehydrogenase/reductase (SDR) is also involved in retinol metabolism.3, 4 However, ADHs are more effective in retinol metabolism compared with SDRs because of the cytosolic location of ADHs.4 Among the 6 different types in the ADH family, class I (ADH1), class IV (ADH4) and class III (ADH3) are responsible for retinol metabolism in mouse tissues. ADH1 and ADH4 are tissue-specifically expressed in the liver and the stomach, respectively, whereas ADH3 is ubiquitously expressed.3-5
Interestingly, retinol and its metabolites play a variety of roles in the differentiation of organs and tissues, obesity-related metabolic disease (insulin resistance) and the regulation of immune cells.6-9 Recent emerging evidence has suggested that retinol metabolites of HSCs are involved in liver inflammation and fibrosis via the modulation of immune cells. Specifically, RAs of HSCs have been reported to mediate the differentiation of naïve T cells into regulatory T cells (Tregs) in vitro.10 In vivo experiments have shown that RAs of HSCs increase the anti-fibrotic effects of natural killer (NK) cells and Gr1+CD11b+ bone marrow cells via enhanced production of interferon-γ (IFN-γ) and interleukin (IL)-10, respectively.11, 12 In contrast, direct treatment of RAs negatively regulates IFN-γ secretion in T cells and cytotoxic activities in the human NK cell line 92.13, 14 Moreover, elevated levels of retinol metabolites have been reported not only in acute ethanol-fed liver toxicity but also in carbon tetrachloride (CCl4)- and thioacetamide-induced liver fibrosis.15, 16 Furthermore, several reports have suggested that retinol metabolites play an important role in liver fibrosis via activating HSCs to increase latent transforming growth factor (TGF)-β activation and the expression of pro-collagen Iα and suppressor of cytokine signaling 1 (SOCS1).17-19 Nevertheless, neither the type of ADHs involved in the retinol metabolism of HSCs nor the bidirectional roles of retinols and their metabolites during the interaction between HSCs and NK cells have been clarified. In the present study, we investigated the expression of ADHs and their functions in HSCs and NK cells in liver fibrosis.
Material and Methods
Animals
Male C57BL/6 wild-type (WT) and green fluorescence protein (GFP)-transgenic mice were purchased from the Jackson Laboratory (Bar Harbor, ME). ADH1 knock-out (ADH1−/−) and ADH3−/− mice on a C57BL/6 background (8-10 weeks) were graciously provided by Dr. Gregg Duester (Sanford-Burnham Medical Research Institute, CA, USA) and Dr. Takeshi Haseba (Nippon Medical School, Tokyo, Japan). All animals were maintained in a specific pathogen-free animal facility at the Korea Advanced Institute of Science and Technology (KAIST). Chimeric mice were prepared by bone marrow transplantation as previously reported.20 All animals received humane care according to the criteria outlined in the Guide for the Care and Use of Laboratory Animals published by NIH, and all experimental procedures were approved by the Institutional Animal Care and Use Committee of KAIST.
CCl4- or Bile Duct Ligation-Induced Liver Fibrosis in Mice
Liver fibrosis was induced by CCl4 injection (0.4 ml/kg, 3 times per week) or bile duct ligation (BDL) for 2 weeks.
Serum Biochemical Measurements
Serum was collected and assayed for alanine aminotransferase (ALT), aspartate aminotransferase (AST) total bilirubin using kits purchased from IDEXX Laboratories (ME, USA). Serum or supernatant levels of IL-6, MCP-1 and IFN-γ were measured using an ELISA kit (Biosource International Inc, CA).
Cell Isolation and Co-culturing
As described previously,2, 11, 12 HSCs and NK cells were isolated by collagenase perfusion followed by differential centrifugation on an Opti-Prep (Sigma) density gradient and an NK cell isolation kit (Miltenyi), respectively. Isolated HSCs were cultured with 10% fetal bovine serum plus 10% horse serum in RPMI medium and co-cultured with NK cells in serum-free medium. Liver mononuclear cells (MNCs) were also isolated by Percoll gradients (Sigma).
Statistical Analysis
Data are presented as the means ± SEM. To compare values obtained from two or more groups, Student’s t test or one-way analysis of variance was performed. A value of P < 0.01 or 0.05 was considered significant statistically. All other materials and methods including staining, retinoid measurements, isolation techniques, reverse transcription-polymerase chain reaction (RT-PCR) or real-time PCR analyses, western blotting, cytotoxicity assay, small interfering ribonucleic acid (siRNA) targeting ADH3 and fluorescence activated cell sorting (FACS) analyses are described in the supporting information.
Results
Suppression of ADH3 Inhibits HSC Activation
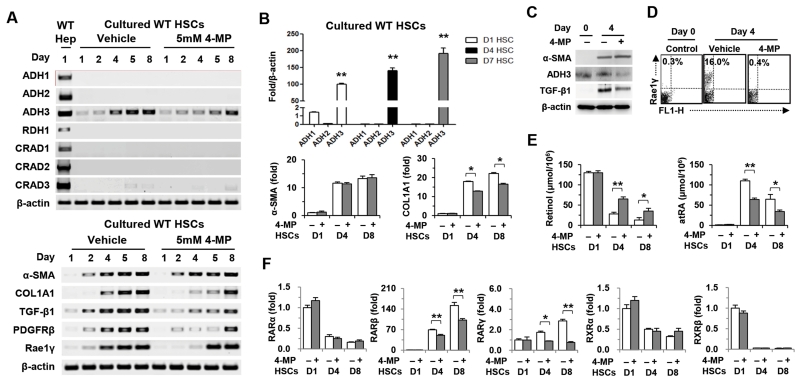
In RT-PCR analyses, we demonstrated that among several retinol metabolizing enzymes, only ADH3 was detected in HSCs, whereas normal hepatocytes expressed most of them, including ADH1, ADH2, retinol dehydrogenase 1 (RDH1) and cis-retinol/androgen dehydrogenases (CRADs) and the ADH3 expression of HSCs gradually increased during in vitro activation (Fig. 1A). In real-time PCR analyses, the expression of ADH1 and ADH2 was negligibly detected compared to the ADH3 expression in mouse HSCs and human HSC lines such as LX-2 and hTERT (Fig. 1B and Supporting Fig. 1A). In an in vitro culturing of HSCs, 4-methylpyrazole (4-MP) treatment, a competitive inhibitor of ADH, suppressed the ADH3 expression, whereas all-trans RA (atRA) treatment increased the ADH3 expression in mouse HSCs and hTERT (Fig. 1A and Supporting Fig. 1B). In addition, in vitro 4-MP treatment attenuated gene expression or protein levels of type I collagen alpha 1(COL1A1), TGF-β1, platelet-derived growth factor receptor β, retinoic acid-early inducible gene 1 (Rae1), IL-6, monocyte chemotactic protein (MCP)-1 and ADH3 in cultured HSCs from wild-type mice (Fig. 1A-D and Supporting Fig. 1C,D). Intriguingly, there was no difference in the expression of alpha-smooth muscle actin (α-SMA), a cytoskeletal protein gene, and fatty acyl CoA synthetase 1 in either group (Fig. 1A-C and Supporting Fig. 1E). In parallel with our previous in vitro study that demonstrated suppressed retinol metabolism of HSCs by 4-MP treatment,19 the production of retinol metabolites in cultured HSCs was significantly suppressed by 4-MP (Fig. 1E). In accordance with decreased levels of RAs by 4-MP, the expression of RARβ and RARγ was significantly suppressed in 4-MP-treated HSCs compared to that of vehicle-treated HSCs (Fig. 1F). Moreover, in vitro 4-MP treatment did not alter the gene expression of α-SMA, COL1A1, TGF-β1, IL-6 and MCP-1 in ADH3-deficient HSCs, suggesting that 4-MP-mediated suppression of HSC activation is dependent on ADH3 (Supporting Fig. 1F). In RT- and real-time PCR analyses, similar to the WT HSCs, ADH1−/− HSCs only expressed ADH3 among several retinol metabolizing enzymes, and the expression of COL1A1 and TGF-β1 in 4-MP-treated ADH1−/− HSCs was suppressed, but α-SMA expression was not affected (Supporting Fig. 1G, H). In addition, 4-MP treatment did not affect apoptosis of WT HSCs (Supporting Fig. 1I). Collectively, these data suggest that ADH3-mediated retinol metabolism plays an important role in promoting HSC activation.
Fig. 1.
4-MP treatment inhibits activation of HSCs. (A) RT-PCR analyses of wild type (WT) hepatocytes and WT HSCs treated with 4-MP. (B) Real-time PCR analyses of ADH3, α-SMA and COL1A1 in WT HSCs. (C) Western blotting for α-SMA, ADH3 and TGF-β1 in cultured HSCs. (D) Flow cytometry for Rae1 expression in cultured HSCs. (E) Intracellular concentration of retinol and atRA from cultured HSCs. (F) Real-time PCR analyses on retinoic acid receptors (RARs) and retinoic X receptors (RXRs) of 4-MP-treated WT HSCs. Data are expressed as the mean ± SEM. *P < 0.05, **P < 0.01 in comparison with the corresponding controls.
Depletion of ADH3 Attenuates Activation and Proliferation in HSCs
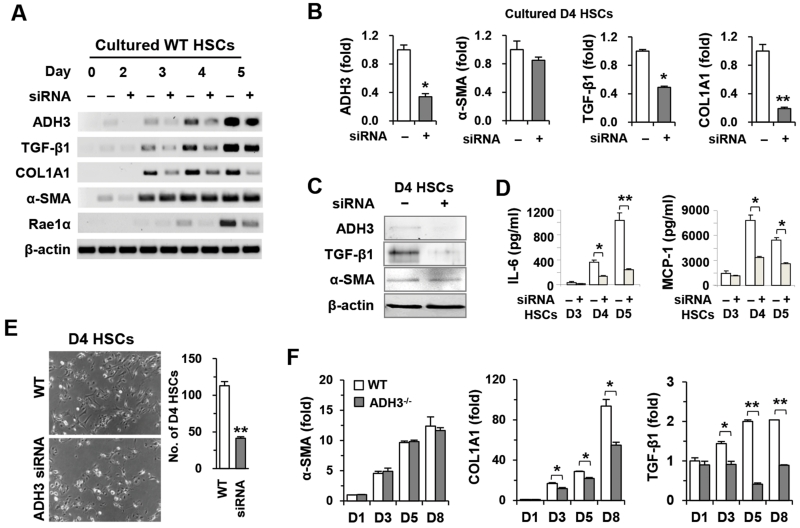
To confirm ADH3-mediated activation and proliferation of HSCs, we performed siRNA targeting ADH3. As shown in Fig. 2A, ADH3 expression of HSCs was successfully depleted or inhibited up to day 4 and was restored at day 5. In concordance with ADH3 inhibition by 4-MP, the expression of TGF-β1, COL1A1 and Rae1 was suppressed, whereas α-SMA expression was not affected in siRNA-treated HSCs compared with that in vehicle-treated HSCs (Fig. 2A-C). Moreover, cytokine production of IL-6 and MCP-1 and proliferation in siRNA-treated HSCs were significantly reduced compared to those of vehicle-treated HSCs (Fig. 2D,E). Furthermore, suppressed expression of COL1A1 and TGF-β1 was confirmed in ADH3−/− HSCs compared to that in WT HSCs (Fig. 2F).
Fig. 2.
Suppression of the ADH3 gene inhibits the activation and proliferation of HSCs. WT and ADH3−/− HSCs were isolated and cultured. (A,B) RT- and real-time PCR analyses of HSCs after the addition of ADH3 siRNA. (C) Western blot analyses of 4-day cultured HSCs (D4 HSCs) after ADH3 siRNA. (D) Supernatant levels of IL-6 and MCP-1 after ADH3 siRNA in HSCs. (E) Cultured D4 HSCs of WT mice with ADH3 siRNA and their numbers (original magnification ×100). (F) Cultured HSCs of WT and ADH3−/− mice were subjected to real-time PCR analyses. Data are expressed as the mean ± SEM. *P < 0.05, **P < 0.01 in comparison with the corresponding controls.
ADH3 inhibition Enhances Cytotoxicity of NK cells against HSCs by Increasing IFN-γ Production
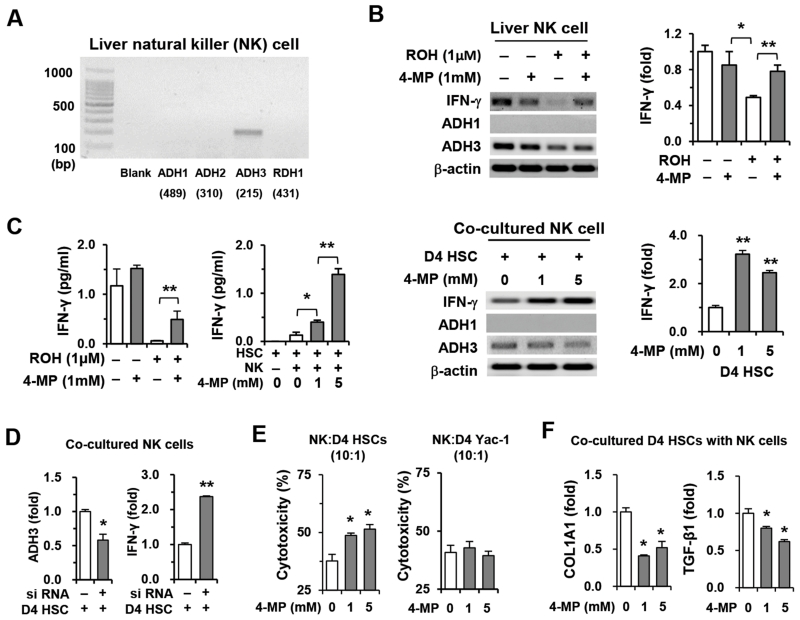
In RT-PCR analyses, among several retinol metabolizing genes, only the ADH3 gene was detected in liver NK cells (Fig. 3A and Supporting Fig. 2A). To assess the effects of retinol and ADH3 on NK cells in vitro, we directly treated NK cells with retinols or co-cultured NK cells with HSCs because HSCs are natural sources of retinol. In PCR analyses, IFN-γ expression of NK cells was significantly suppressed by retinol, whereas its expression was restored by 4-MP-mediated inhibition of retinol metabolism (upper panels of Fig. 2B). In co-culturing with HSCs, ADH3 inhibition by 4-MP increased IFN-γ expression in NK cells (lower panels of Fig. 3B). In addition, increased IFN-γ levels in their supernatants supported the data presented above (Fig. 3C and Supporting Fig. 2B). To confirm the inhibitory role of retinol through ADH3 in the IFN-γ production of NK cells, we co-cultured NK cells with HSCs after ADH3 siRNA treatment of NK cells in vitro. In PCR analyses, the ADH3 expression of NK cells was suppressed by ADH3 siRNA, whereas IFN-γ expression was significantly increased in siRNA-treated NK cells compared with controls (Fig. 3D). In addition, the expression of COL1A1 and MCP-1 in co-cultured HSCs was significantly suppressed by siRNA-treated NK cells (Supporting Fig. 2C). Furthermore, 4-MP-mediated ADH3 inhibition showed not only an increased killing rate but also suppressive activities of NK cells against HSCs (Fig. 3E,F). Interestingly, however, there was no significant difference in the Yac-1 cytotoxicity of NK cells after 4-MP treatment. These data demonstrate that retinol released from HSCs plays an inhibitory role in NK cell cytotoxicity through ADH3-mediated suppression of IFN-γ production.
Fig. 3.
Retinol suppresses IFN-γ production and cytotoxicity of NK cells against HSCs through ADH3. Freshly isolated NK cells from WT mice were used for in vitro experiments. (A) Expression of retinol metabolizing enzymes was analyzed using liver NK cells. (B) NK cells were treated with retinol or co-cultured with D4 HSCs for 3 hours in the presence or absence of 4-MP and then subjected to RT- and real-time PCR analyses. (C) IFN-γ levels of supernatants were measured. (D) Liver NK cells treated with ADH3 siRNA or scramble siRNA were co-cultured with D4 HSCs and subjected to real-time PCR analyses. (E) Liver NK cells were used as effector cells for a cytotoxicity assay against D4 HSCs and Yac-1 cells in the presence or absence of 4-MP. (F) Co-cultured D4 HSCs with NK cells were subjected to real-time PCR analyses. Data are expressed as the mean ± SEM. *P < 0.05, **P < 0.01 in comparison with the corresponding controls.
Ablation of ADH3 Gene Impedes Liver Fibrosis by Up-regulating IFN-γ Production of NK Cells and Down-regulating Activation of HSCs
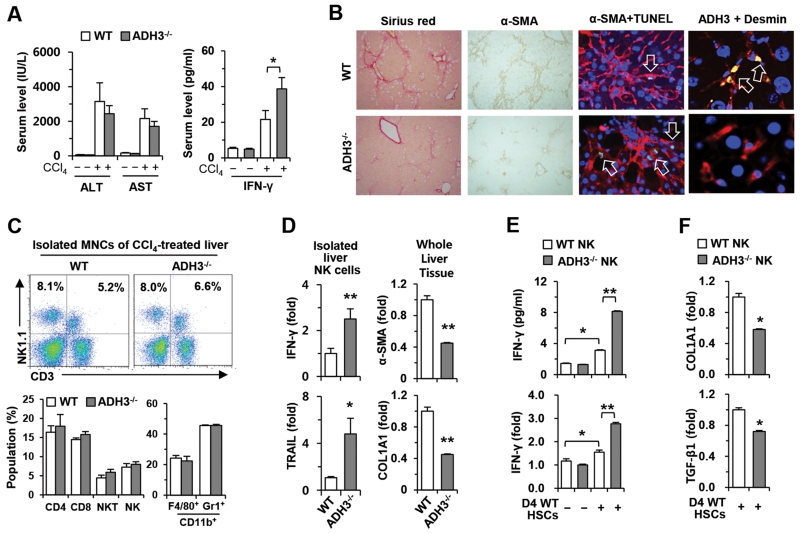
Based on the data described above, ADH3 may have cell-type specific roles, such as positive effects, on the activation of HSCs but inhibitory effects on NK cells. To confirm this concept in vivo, we injected CCl4 into WT and ADH3−/− mice for 2 weeks. Blood chemistry analyses revealed that levels of alanine aminotransferase (ALT) and aspartate aminotransferase (AST) were similar in the two groups, but IFN-γ levels were significantly increased in ADH3−/− mice compared with those in WT mice (Fig. 4A). In addition, collagen deposition and α-SMA expression were significantly reduced, whereas apoptotic HSCs were increased in livers of ADH3−/− mice compared with those of WT mice (Fig. 4B and Supporting Fig. 3A,B). Interestingly, the increased expression of ADH3 in HSCs was detected in fibrotic liver of WT mice (Fig. 4B). Furthermore, the gene expression of ADH3 and Raldh1 was significantly up-regulated in isolated HSCs of CCl4-treated WT mice compared to that of HSCs isolated from control WT mice (Supporting 3C). In FACS analyses, there were no significant liver immune-cell population changes between the groups (Fig. 4C). However, the expression of IFN-γ and TNF-related apoptosis-inducing ligand (TRAIL) was significantly increased in ADH3−/− NK cells, whereas the expression of α-SMA and COL1A1 was remarkably decreased in ADH3−/− liver tissues compared to those of WT mice (Fig. 4D). In an in vitro co-culturing system, co-cultured ADH3−/− NK cells produced more IFN-γ than did WT NK cells (Fig. 4E), which subsequently suppressed the expression of COL1A1 and TGF-β1 in co-cultured WT HSCs compared with that in co-cultured WT HSCs with WT NK cells (Fig. 4F). Similarly, ADH3−/− mice showed less accumulation of collagen fibers and activation of HSCs than WT mice in BDL-induced liver fibrosis, although there was no significant liver injury in the two groups (Supporting Fig. 3D-G). However, serum levels of IFN-γ were significantly increased in ADH3−/− mice (Supporting Fig. 3D). In accordance with data of CCl4 treatment, the increased gene expression of ADH3 and Raldh1 was detected in isolated HSCs of BDL-treated WT mice compared to that of HSCs isolated from control WT mice (Supporting 3H).
Fig. 4.
Ablation of the ADH3 gene inhibits liver fibrosis by up-regulation of IFN-γ production of NK cells and down-regulation of the activation of HSCs. WT and AD3−/− mice were injected with CCl4 for 2 weeks. (A) Levels of ALT, AST and IFN-γ were measured in collected sera. (B) Liver sections were stained with Sirius red and α-SMA antibody (original magnification, ×100), co-stained with α-SMA antibody (red) and TUNEL (green) or with ADH3 (green) and desmin (red) (original magnification, ×800, ×1200). Nuclei were stained with DAPI (blue) (C) Liver MNCs were subjected to flow cytometry. (D) Isolated liver NK cells and whole liver tissues were subjected to real-time PCR analyses. (E) IFN-γ production and its expression were analyzed by co-culturing NK cells of WT and ADH3−/− mice with D4 WT HSCs. (F) Co-cultured D4 WT HSCs with NK cells of WT and ADH3−/− mice were subjected to real-time PCR analyses. Data are expressed as the mean ± SEM. *P < 0.05, **P < 0.01 in comparison with the corresponding controls.
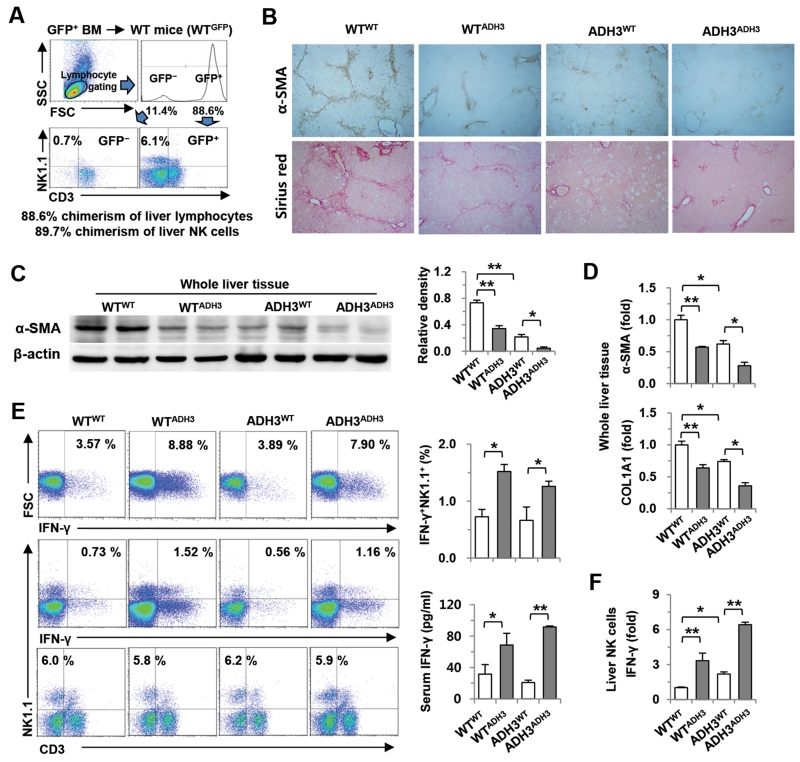
ADH3-Deficient Bone Marrow Transplantation Ameliorates Liver Fibrosis by Enhanced IFN-γ Production of NK cells
To investigate the effects of ADH3-depleted NK cells in liver fibrosis, we produced chimeric mice by reciprocal bone marrow transplantation using WT and ADH3−/− mice. At 8 weeks after the transplantation of WT GFP-expressing bone marrow, approximately 88.6% and 89.7% of liver MNCs and NK cells were replaced with donor bone marrow (Fig. 5A). In addition, the number and population of non-parenchymal immune cells were similar after reciprocal bone marrow transplantation (Supporting Fig. 4). Chimeric mice prepared by reciprocal transplantation of WT and ADH3−/− bone marrow (WTWT, WTADH3, ADH3WT or ADH3ADH3) were injected with CCl4 for 2 weeks. Collagen deposition and expression of α-SMA were significantly decreased in chimeric mice containing ADH3-depleted NK cells (WTADH3, ADH3ADH3) or ADH3-depleted HSCs (ADH3WT) compared with WTWT in liver sections and tissues (Fig. 5B,C). Real-time PCR analyses of whole liver tissues showed that the expression of α-SMA and COL1A1 was remarkably suppressed in WTADH3, ADH3WT and ADH3ADH3 compared with their expression in WTWT (Fig. 5D). Specifically, chimeric ADH3ADH3 mice showed remarkably reduced expression of α-SMA and COL1A1 compared with the three other groups (Fig. 5C,D). In FACS analyses, the IFN-γ positive population and its expression of liver NK cells from chimeric mice transplanted with ADH3-depleted bone marrow (WTADH3 and ADH3ADH3) were higher than those from WTWT and ADH3WT, although there were no significant changes in NK cell population among the four groups (Fig. 5E,F).
Fig. 5.
ADH3-deficient bone marrow transplantation ameliorates liver fibrosis. (A) At week 8 after the transplantation of GFP-producing WT bone marrow into WT mice. Chimerism of NK cells was assessed by flow cytometry. (B) After reciprocal bone marrow transplantation between WT and ADH3-deficient mice, mice were injected with CCl4 for 2 weeks. Liver sections were stained with α-SMA antibody and Sirius red (original magnification, ×100). (C, D) Liver tissues were removed and used for Western blotting and real-time PCR analyses. (E) IFN-γ production of liver NK cells was analyzed by flow cytometry, and serum levels of IFN-γ were measured. (F) IFN-γ expression in isolated liver NK cells was analyzed by real-time PCR analyses. Data are expressed as the mean ± SEM. *P < 0.05, **P < 0.01 in comparison with the corresponding controls.
Discussion
Retinoids (retinol and its derivatives) are known as a critical factor for not only immune responses but also metabolic disorders.7-9 Although their metabolizing enzyme activity and their roles in liver fibrosis have not yet been clarified, it is known that HSCs store vast amounts of retinol lipid droplets. Thus, the present study demonstrates that the ADH3 gene is mainly expressed in both HSCs and NK cells. In addition, by using the ADH3 gene deletion or ADH inhibitor, we have demonstrated that ADH3 acts as a positive regulator in the activation of HSCs, whereas ADH3 negatively regulates NK cell activity during liver fibrogenesis (Fig. 6). Furthermore, although 4-MP is a broad inhibitor of ADHs, we provided several lines of in vitro evidence supporting that the effects of 4-MP were mediated through the inhibition of ADH3. First, we showed that direct RA treatment increased the ADH3 gene expression in mouse HSCs and human hTERT, suggesting 4-MP-mediated suppression of the ADH3 gene expression is due to decreased metabolism of retinols into RAs in HSCs (Supporting Fig. 1B). Second, in vitro 4-MP treatment suppressed WT HSC activation but had no effect on ADH3-deficient HSCs, indicating that 4-MP inhibits the activation of HSCs via an ADH3-dependent manner (Supporting Fig. 1F). Finally, a previous report showed that 4-MP inhibited fatty acyl coenzyme synthetase during alcohol metabolism.21 By analyzing the expression of fatty acyl coenzyme synthetase, we found that 4-MP treatment did not affect the expression of these enzymes significantly in HSCs (Supporting Fig. 1E).
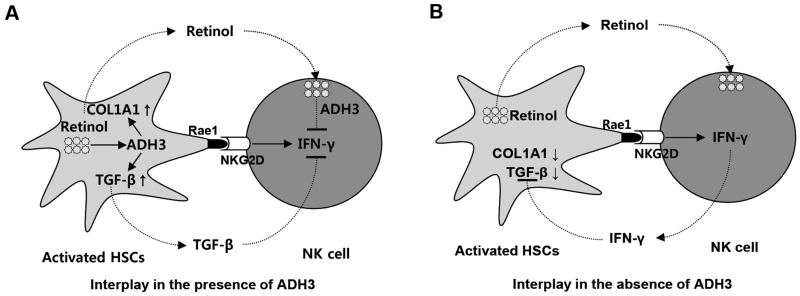
Fig. 6.
Cell type-specific roles of ADH3 in liver fibrosis. During liver injury, retinols are metabolized by ADH3 in HSCs and NK cells, which affects the interaction of HSCs and NK cells in 2 ways. (A) In the presence of ADH3 in HSCs and NK cells, ADH3 induces activation of HSCs by expressing COL1A1 or activating latent TGF-β. In the meantime, Rae1 expression in activated HSCs recruits liver NK cells, which is followed by the suppression of IFN-γ production of NK cells by activated TGF-β. In addition, released retinols from activated HSCs are metabolized by the ADH3 of NK cells, resulting in the inhibition of IFN-γ production in NK cells. (B) In the absence of ADH3 in HSCs and NK cells, the expression of COL1A1 and TGF-β was significantly reduced in HSCs, whereas activated NK cells by HSCs produce more IFN-γ, resulting in the inhibition of HSC activation. Therefore, ADH3 is a positive regulator in the activation of HSCs but a negative regulator in IFN-γ production of NK cells.
According to the literature, decreased retinol contents but increased RAs in cases of liver injury have been implicated in the activation of HSCs and liver fibrosis via the enhanced activation of latent TGF-β.16-18 However, whether increased retinol metabolites act as activators or suppressors of liver fibrosis remains controversial. Several studies have reported that exogenous retinoid treatments suppressed the activation of HSCs and liver fibrosis in vitro and in vivo.22, 23 In contrast, some studies showed that different sources or types of retinoids induced divergent results in the proliferation and collagen production of HSCs.24-26 Moreover, in our previous study, the levels of retinaldehydes and the expression of collagen fibers continuously increased in cultured HSCs for 6 days, whereas cytosolic levels of RAs within activating HSCs peaked at day 3 and then sharply decreased at day 6.19 Furthermore, depletion of retinol storing enzyme lecithin-retinol acyltransferase (LRAT) in mice neither accelerates nor suppresses liver fibrosis compared with that in WT mice; however, LRAT-depleted mice showed elevated expression of retinoid-response genes in liver tumors,27 suggesting that retinol metabolism still occurs even though retinol storage is absent in LRAT-depleted mice. To clarify these controversial results, it is necessary to first know which ADH enzyme is involved in the oxidation of retinol into retinaldehyde in HSCs. Next, the function of the enzyme in the activation of HSCs and liver fibrosis needs to be understood. Therefore, in the present study, we investigated the major enzyme responsible for retinol metabolism in HSCs and found that ADH3-mediated retinol metabolism is, in part, related to the acceleration of liver fibrosis by regulating the activation of HSCs. In addition, ADH3-deficient mice and WT mice had comparable degree of liver injury and inflammation induced by CCl4 and BDL (Fig. 4A and Supporting Fig. 3D). This suggests that ADH3 promotion of liver fibrosis is not mediated via the regulation of liver injury and inflammation. Lack of decrease in liver injury in ADH3-deficient mice is likely because CCl4-induced liver toxicity is mainly mediated via the metabolism of CCl4 by cytochrome P4502E1, and BLD-induced liver injury and fibrosis is mediated by secreted profibrotic factors of proliferating cholangiocytes.28, 29 Moreover, the ADH3 gene depletion by reciprocal bone marrow transplantation did not change the number and population of hepatic immune cells (Supporting Fig. 4).
Recently, the liver has been considered an immunologic organ because of enriched hepatic immune cells, including both innate (e.g., NK cells) and adaptive (e.g., T cells) immune cells.30, 31 Of the many hepatic immune cells, NK cell activation has been highlighted recently as a novel candidate for therapy of liver fibrosis.11, 19, 32, 33 In a fibrotic liver, NK cells can recognize and kill activated HSCs via receptor activation-mediated cytokine production.11, 33 However, a recent study by our group demonstrated that activated HSCs can be resistant to NK cell killing.19 Activated HSCs can metabolize retinols into RAs that induce enhanced expression of TGF-β1 and SOCS1, leading to the suppression of NK cell activity or the inhibition of IFN-γ signaling, respectively.19 However, HSCs do not metabolize all of the retinol during activation, and some retinol is released from HSCs.2, 34 Thus, it is necessary to identify which effect of released retinol from HSCs is associated with hepatic immune cells, especially NK cells. According to the literature, RAs have been implicated in a wide range of NK cell responses including proliferation, maturation, differentiation and activity.2, 14, 35, 36 However, it is not clear yet which enzymes of ADHs are involved in the oxidation of retinol into RAs in NK cells and what function of RAs on NK cells upon liver fibrosis. In the present study, we observed that NK cells expressed only ADH3 among several retinol metabolizing enzymes and that ADH3 played a negative role in NK cell activities. As shown in Fig 3B,C, direct treatment of retinol suppressed NK cell killing against HSCs by down-regulating IFN-γ production, whereas NK cell killing was restored when ADH3 was inhibited by 4-MP. This finding implies that ADH3-mediated retinol metabolism plays an important role in inhibiting NK cell cytotoxicity. In addition, in co-culturing with WT HSCs (a source of natural retinol), IFN-γ production of NK cells with suppressed or depleted ADH3 was more enhanced than that of control NK cells (Figs. 3D, 4E). In parallel with our data, all-trans RA has been shown to negatively regulate cytotoxic activities of the human NK cell line 92 without affecting their proliferation.14 All of these results suggest that ADH3 is a negative regulator for IFN-γ production and cytotoxicity of NK cells against HSCs (Fig. 6). However, retinol deficiency reduces the number of NK cells and impairs their activities in peripheral blood in non-fibrotic conditions.35 Thus, the effects of retinol on NK cell activity remain obscure.
The importance of RAs with respect to hepatic tumorigenesis and metabolic disorders has been presented. A recent report suggested that fibrotic changes can reduce tumor surveillance of NK and NKT cells, which contributes to tumor progression.37 Additionally, suppressed innate immunity (NK cell/IFN-γ) was found in the advanced stages of liver fibrosis.19 Moreover, HSCs are involved in the induction of myeloid-derived suppressor cells (MDSCs) that can suppress anti-tumor immune responses.38 In a previous study by our group, RAs of HSCs played the role of a positive regulator for increased IL-10 production of MDSCs in fibrotic liver.12 In contrast, exogenous treatments of acyclic retinoids inhibit human hepatoma cell growth by down-regulating fibroblast growth factor-mediated signaling.39 One of the most important aspects of RAs in metabolic disorders is that genetically depleted production of RAs reduces fat accumulation and insulin resistance of the liver in high fat diet-induced obese rodent models, whereas exogenous dietary treatment of RAs improves insulin sensitivity by up-regulating leptin receptor expression.7, 8 Therefore, retinoids with endogenous or exogenous origins may have different roles and signaling mechanisms in tumorigenesis and metabolic liver disease. Thus, further studies are needed to clarify the effects of endogenously generated or exogenously treated retinoids on liver disease.
In conclusion, our study suggests that ADH3 is a main enzyme for retinol metabolism in both HSCs and NK cells, but it plays different roles. For example, ADH3 is a positive regulator for the activation of HSCs but a negative regulator for NK cells in cases of liver fibrosis. Therefore, ADH3 could be a novel therapeutic target for improving liver fibrosis.
Supplementary Material
Acknowledgments
Financial Support
This work was supported by the National Research Foundation of Korea (NRF) grant funded by the Bio & Medical Technology Development Program of the National Research Foundation funded by the Korean government (2012M3A9C7050093), the Intelligent Synthetic Biology Center of Global Frontier Project funded by the Ministry of Science, ICT & Future Planning (2011-0031955), grants of the Korean Health Technology R&D Project, Ministry of Health & Welfare, Republic of Korea (A111345 and A111498) and a grant from the Next-Generation BioGreen 21 Program (No. PJ009957), Rural Development Administration, Republic of Korea
List of Abbreviations
- HSCs
hepatic stellate cells
- RA
retinoic acid
- ADH
alcohol dehydrogenase
- Raldh
retinaldehyde dehydrogenase
- RAR
retinoic acid receptor
- RXR
retinoid X receptor
- SDR
microsomal short-chain dehydrogenase/reductase
- Tregs
regulatory T cells
- NK
natural killer
- IFN
interferon
- IL
interleukin
- CCl4
carbon tetrachloride
- TGF
transforming growth factor
- SOCS
suppressor of cytokine signaling
- WT
wild type
- GFP
green fluorescence protein
- BDL
bile duct ligation
- ALT
alanine aminotransferase
- AST
aspartate aminotransferase
- MNCs
mononuclear cells
- RT-PCR
reverse transcription-polymerase chain reaction
- siRNA
small interfering ribonucleic acid
- FACS
Fluorescence Activated Cell Sorting
- RDH
retinol dehydrogenase
- CRAD
cis-retinol/androgen dehydrogenase
- 4-MP
4-methylpyrazole
- COL1A1
type I collagen alpha 1
- Rae1
retinoic acid-early inducible gene 1
- MCP-1
monocyte chemotactic protein-1
- α-SMA
alpha-smooth muscle actin
- TRAIL
TNF-related apoptosis-inducing ligand
- LRAT
lecithin-retinol acyltransferase
- MDSC
myeloid-derived suppressor cell
References
- 1.Friedman SL. Hepatic stellate cells: protean, multifunctional, and enigmatic cells of the liver. Physiol Rev. 2008;88:125–172. doi: 10.1152/physrev.00013.2007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Radaeva S, Wang L, Radaev S, Jeong WI, Park O, Gao B. Retinoic acid signaling sensitizes hepatic stellate cells to NK cell killing via upregulation of NK cell activating ligand RAE1. Am J Physiol Gastrointest Liver Physiol. 2007;293:G809–816. doi: 10.1152/ajpgi.00212.2007. [DOI] [PubMed] [Google Scholar]
- 3.Pares X, Farres J, Kedishvili N, Duester G. Medium- and short-chain dehydrogenase/reductase gene and protein families : Medium-chain and short-chain dehydrogenases/reductases in retinoid metabolism. Cell Mol Life Sci. 2008;65:3936–3949. doi: 10.1007/s00018-008-8591-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Duester G, Mic FA, Molotkov A. Cytosolic retinoid dehydrogenases govern ubiquitous metabolism of retinol to retinaldehyde followed by tissue-specific metabolism to retinoic acid. Chem Biol Interact. 2003;143-144:201–210. doi: 10.1016/s0009-2797(02)00204-1. [DOI] [PubMed] [Google Scholar]
- 5.Crabb DW, Matsumoto M, Chang D, You M. Overview of the role of alcohol dehydrogenase and aldehyde dehydrogenase and their variants in the genesis of alcohol-related pathology. Proc Nutr Soc. 2004;63:49–63. doi: 10.1079/pns2003327. [DOI] [PubMed] [Google Scholar]
- 6.Duester G. Retinoic acid synthesis and signaling during early organogenesis. Cell. 2008;134:921–931. doi: 10.1016/j.cell.2008.09.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Tsuchiya H, Ikeda Y, Ebata Y, Kojima C, Katsuma R, Tsuruyama T, Sakabe T, et al. Retinoids ameliorate insulin resistance in a leptin-dependent manner in mice. Hepatology. 2012;56:1319–1330. doi: 10.1002/hep.25798. [DOI] [PubMed] [Google Scholar]
- 8.Ziouzenkova O, Orasanu G, Sharlach M, Akiyama TE, Berger JP, Viereck J, Hamilton JA, et al. Retinaldehyde represses adipogenesis and diet-induced obesity. Nat Med. 2007;13:695–702. doi: 10.1038/nm1587. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Mucida D, Park Y, Kim G, Turovskaya O, Scott I, Kronenberg M, Cheroutre H. Reciprocal TH17 and regulatory T cell differentiation mediated by retinoic acid. Science. 2007;317:256–260. doi: 10.1126/science.1145697. [DOI] [PubMed] [Google Scholar]
- 10.Dunham RM, Thapa M, Velazquez VM, Elrod EJ, Denning TL, Pulendran B, Grakoui A. Hepatic stellate cells preferentially induce Foxp3+ regulatory T cells by production of retinoic acid. J Immunol. 2013;190:2009–2016. doi: 10.4049/jimmunol.1201937. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Radaeva S, Sun R, Jaruga B, Nguyen VT, Tian Z, Gao B. Natural killer cells ameliorate liver fibrosis by killing activated stellate cells in NKG2D-dependent and tumor necrosis factor-related apoptosis-inducing ligand-dependent manners. Gastroenterology. 2006;130:435–452. doi: 10.1053/j.gastro.2005.10.055. [DOI] [PubMed] [Google Scholar]
- 12.Suh YG, Kim JK, Byun JS, Yi HS, Lee YS, Eun HS, Kim SY, et al. CD11b(+) Gr1(+) bone marrow cells ameliorate liver fibrosis by producing interleukin-10 in mice. Hepatology. 2012 doi: 10.1002/hep.25817. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Carman JA, Hayes CE. Abnormal regulation of IFN-gamma secretion in vitamin A deficiency. J Immunol. 1991;147:1247–1252. [PubMed] [Google Scholar]
- 14.Li A, He M, Wang H, Qiao B, Chen P, Gu H, Zhang M, et al. All-trans retinoic acid negatively regulates cytotoxic activities of nature killer cell line 92. Biochem Biophys Res Commun. 2007;352:42–47. doi: 10.1016/j.bbrc.2006.10.132. [DOI] [PubMed] [Google Scholar]
- 15.Kane MA, Folias AE, Wang C, Napoli JL. Ethanol elevates physiological all-trans-retinoic acid levels in select loci through altering retinoid metabolism in multiple loci: a potential mechanism of ethanol toxicity. FASEB J. 2010;24:823–832. doi: 10.1096/fj.09-141572. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Natarajan SK, Thomas S, Ramachandran A, Pulimood AB, Balasubramanian KA. Retinoid metabolism during development of liver cirrhosis. Arch Biochem Biophys. 2005;443:93–100. doi: 10.1016/j.abb.2005.09.008. [DOI] [PubMed] [Google Scholar]
- 17.Okuno M, Moriwaki H, Imai S, Muto Y, Kawada N, Suzuki Y, Kojima S. Retinoids exacerbate rat liver fibrosis by inducing the activation of latent TGF-beta in liver stellate cells. Hepatology. 1997;26:913–921. doi: 10.1053/jhep.1997.v26.pm0009328313. [DOI] [PubMed] [Google Scholar]
- 18.Okuno M, Sato T, Kitamoto T, Imai S, Kawada N, Suzuki Y, Yoshimura H, et al. Increased 9,13-di-cis-retinoic acid in rat hepatic fibrosis: implication for a potential link between retinoid loss and TGF-beta mediated fibrogenesis in vivo. J Hepatol. 1999;30:1073–1080. doi: 10.1016/s0168-8278(99)80262-1. [DOI] [PubMed] [Google Scholar]
- 19.Jeong WI, Park O, Suh YG, Byun JS, Park SY, Choi E, Kim JK, et al. Suppression of innate immunity (natural killer cell/interferon-gamma) in the advanced stages of liver fibrosis in mice. Hepatology. 2011;53:1342–1351. doi: 10.1002/hep.24190. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Seki E, De Minicis S, Osterreicher CH, Kluwe J, Osawa Y, Brenner DA, Schwabe RF. TLR4 enhances TGF-beta signaling and hepatic fibrosis. Nat Med. 2007;13:1324–1332. doi: 10.1038/nm1663. [DOI] [PubMed] [Google Scholar]
- 21.Bradford BU, Forman DT, Thurman RG. 4-Methylpyrazole inhibits fatty acyl coenzyme synthetase and diminishes catalase-dependent alcohol metabolism: has the contribution of alcohol dehydrogenase to alcohol metabolism been previously overestimated? Mol Pharmacol. 1993;43:115–119. [PubMed] [Google Scholar]
- 22.Davis BH, Kramer RT, Davidson NO. Retinoic acid modulates rat Ito cell proliferation, collagen, and transforming growth factor beta production. J Clin Invest. 1990;86:2062–2070. doi: 10.1172/JCI114943. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Senoo H, Wake K. Suppression of experimental hepatic fibrosis by administration of vitamin A. Lab Invest. 1985;52:182–194. [PubMed] [Google Scholar]
- 24.Hellemans K, Verbuyst P, Quartier E, Schuit F, Rombouts K, Chandraratna RA, Schuppan D, et al. Differential modulation of rat hepatic stellate phenotype by natural and synthetic retinoids. Hepatology. 2004;39:97–108. doi: 10.1002/hep.20015. [DOI] [PubMed] [Google Scholar]
- 25.Lee YS, Jeong WI. Retinoic acids and hepatic stellate cells in liver disease. J Gastroenterol Hepatol. 2012;27(Suppl 2):75–79. doi: 10.1111/j.1440-1746.2011.07007.x. [DOI] [PubMed] [Google Scholar]
- 26.Hellemans K, Grinko I, Rombouts K, Schuppan D, Geerts A. All-trans and 9-cis retinoic acid alter rat hepatic stellate cell phenotype differentially. Gut. 1999;45:134–142. doi: 10.1136/gut.45.1.134. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Kluwe J, Wongsiriroj N, Troeger JS, Gwak GY, Dapito DH, Pradere JP, Jiang H, et al. Absence of hepatic stellate cell retinoid lipid droplets does not enhance hepatic fibrosis but decreases hepatic carcinogenesis. Gut. 2011;60:1260–1268. doi: 10.1136/gut.2010.209551. [DOI] [PubMed] [Google Scholar]
- 28.Tsukamoto H, Matsuoka M, French SW. Experimental models of hepatic fibrosis: a review. Semin Liver Dis. 1990;10:56–65. doi: 10.1055/s-2008-1040457. [DOI] [PubMed] [Google Scholar]
- 29.Glaser SS, Gaudio E, Miller T, Alvaro D, Alpini G. Cholangiocyte proliferation and liver fibrosis. Expert Rev Mol Med. 2009;11:e7. doi: 10.1017/S1462399409000994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Racanelli V, Rehermann B. The liver as an immunological organ. Hepatology. 2006;43:S54–62. doi: 10.1002/hep.21060. [DOI] [PubMed] [Google Scholar]
- 31.Gao B, Jeong WI, Tian Z. Liver: An organ with predominant innate immunity. Hepatology. 2008;47:729–736. doi: 10.1002/hep.22034. [DOI] [PubMed] [Google Scholar]
- 32.Muhanna N, Abu Tair L, Doron S, Amer J, Azzeh M, Mahamid M, Friedman S, et al. Amelioration of hepatic fibrosis by NK cell activation. Gut. 2011;60:90–98. doi: 10.1136/gut.2010.211136. [DOI] [PubMed] [Google Scholar]
- 33.Melhem A, Muhanna N, Bishara A, Alvarez CE, Ilan Y, Bishara T, Horani A, et al. Anti-fibrotic activity of NK cells in experimental liver injury through killing of activated HSC. J Hepatol. 2006;45:60–71. doi: 10.1016/j.jhep.2005.12.025. [DOI] [PubMed] [Google Scholar]
- 34.Senoo H. Structure and function of hepatic stellate cells. Med Electron Microsc. 2004;37:3–15. doi: 10.1007/s00795-003-0230-3. [DOI] [PubMed] [Google Scholar]
- 35.Semba RD. The role of vitamin A and related retinoids in immune function. Nutr Rev. 1998;56:S38–48. doi: 10.1111/j.1753-4887.1998.tb01643.x. [DOI] [PubMed] [Google Scholar]
- 36.Ma Y, Ross AC. The anti-tetanus immune response of neonatal mice is augmented by retinoic acid combined with polyriboinosinic:polyribocytidylic acid. Proc Natl Acad Sci U S A. 2005;102:13556–13561. doi: 10.1073/pnas.0506438102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Zhang DY, Friedman SL. Fibrosis-dependent mechanisms of hepatocarcinogenesis. Hepatology. 2012;56:769–775. doi: 10.1002/hep.25670. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Chou HS, Hsieh CC, Yang HR, Wang L, Arakawa Y, Brown K, Wu Q, et al. Hepatic stellate cells regulate immune response by way of induction of myeloid suppressor cells in mice. Hepatology. 2011;53:1007–1019. doi: 10.1002/hep.24162. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Shao RX, Otsuka M, Kato N, Taniguchi H, Hoshida Y, Moriyama M, Kawabe T, et al. Acyclic retinoid inhibits human hepatoma cell growth by suppressing fibroblast growth factor-mediated signaling pathways. Gastroenterology. 2005;128:86–95. doi: 10.1053/j.gastro.2004.09.077. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.