Abstract
Bone marrow-derived mesenchymal stromal stem cells (BMSCs) are a promising cell source for treating articular cartilage defects. The objective of this study was to assess the impact of cell seeding density within a collagen I scaffold on in vitro BMSC chondrogenesis following isolation and expansion in two-dimensional (2D) and three-dimensional (3D) environments. It was hypothesized that both expansion protocols would produce BMSCs capable of hyaline-like chondrogenesis with an optimal seeding density of 10 × 106 cells/cm3. Ovine BMSCs were isolated in a 2D environment by plastic adherence, expanded to passage two in flasks containing an expansion medium, and seeded within collagen I scaffolds at densities of 50, 10, 5, 1, and 0.5 × 106 BMSCs/cm3. For 3D isolation and expansion, aspirates containing known quantities of mononucleated cells (bone marrow-derived mononucleated cells [BMNCs]) were seeded on scaffolds at 50, 10, 5, 1, and 0.5 × 106 BMNCs/cm3 and cultured in the expansion medium for an equivalent duration to 2D expansion. Constructs were differentiated in vitro in the chondrogenic medium for 21 days and assessed with reverse-transcription quantitative polymerase chain reaction, safranin O staining, histological scoring using the Bern Score, collagen immunofluorescence, and glycosaminoglycan (GAG) quantification. Two-dimensional-expanded BMSCs seeded at all densities were capable of proteoglycan production and displayed increased expressions of aggrecan and collagen II messenger RNA (mRNA) relative to pre-differentiation controls. Collagen II deposition was apparent in scaffolds seeded at 0.5–10 × 106 BMSCs/cm3. Chondrogenesis of 2D-expanded BMSCs was most pronounced in scaffolds seeded at 5–10 × 106 BMSCs/cm3 based on aggrecan and collagen II mRNA, safranin O staining, Bern Score, total GAG, and GAG/deoxyribonucleic acid (DNA). For 3D-expanded BMSC-seeded scaffolds, increased aggrecan and collagen II mRNA expressions relative to controls were noted with all densities. Proteoglycan deposition was present in scaffolds seeded at 0.5–50 × 106 BMNCs/cm3, while collagen II deposition occurred in scaffolds seeded at 10–50 × 106 BMNCs/cm3. The highest levels of aggrecan and collagen II mRNA, Bern Score, total GAG, and GAG/DNA occurred with seeding at 50 × 106 BMNCs/cm3. Within a collagen I scaffold, 2D- and 3D-expanded BMSCs are capable of hyaline-like chondrogenesis with optimal cell seeding densities of 5–10 × 106 BMSCs/cm3 and 50 × 106 BMNCs/cm3, respectively.
Introduction
Bone marrow-derived mesenchymal stromal stem cells (BMSCs) are a promising cell source for treating articular cartilage defects.1 BMSCs seeded within biomaterial scaffolds and implanted into focal chondral defects are capable of resurfacing cartilage in animal and human joints, although inconsistent outcomes have been reported based on macroscopic assessment, histological analysis, magnetic resonance imaging, and clinical scoring.2–7 Repair tissue quality has been shown to correlate with functional outcome.6–8 Therefore, tissue-engineering variables, such as cell expansion environment and seeding density of scaffolds, are currently under investigation with the goal of improving neocartilage quality.
BMSCs have conventionally been isolated by plastic adherence and expanded in a two-dimensional (2D) environment within tissue culture flasks.9,10 Although this method has been shown to produce cells capable of chondrogenic differentiation,11–14 major drawbacks include loss of multi-potent differentiation, inability to produce cartilaginous extracellular matrix (ECM) proteins and cellular senescence during prolonged expansion periods.15–18 Three-dimensional (3D) isolation and expansion of BMSCs have been proposed as a method of mimicking the natural bone marrow microenvironment and maintaining multipotency and chondrogenic capacity.19,20 Cell collections containing bone marrow-derived mononucleated cells (BMNCs)—a small fraction of which are BMSCs—are seeded within biomaterials for 3D isolation, expansion, and subsequent differentiation.20,21
Cell seeding density is a transplantation variable that has not been evaluated in detail to date for either 2D- or 3D-expanded BMSCs. Healthy articular cartilage naturally contains 9.6 × 106 chondrocytes/cm3.22 The optimal BMSC seeding density required for cell organization, chondrogenic differentiation, and ECM production to create engineered tissue that resembles native cartilage is currently unknown. For implantation purposes in vivo, BMSC seeding densities of 1–50 × 106 cells/cm3 of biomaterial matrix have been used in preclinical animal studies.1,14,23–32 In clinical studies, a density of 5 × 106 cells/cm3 has been reported although the rationale for adopting this seeding density was not described.3,4
The objective of this study was to assess the impact of cell seeding density within a clinically relevant, collagen I scaffold on in vitro BMSC chondrogenesis following 2D and 3D isolation and expansion. Two-dimensional isolation was performed by seeding whole bone marrow aspirates (BMAs) into tissue culture flasks, while 3D isolation involved seeding whole BMAs within collagen scaffolds. Collagen was used as a biomaterial given that it is used routinely in preclinical animal and human studies.3–5,14 Ovine cells were studied as sheep are emerging as a useful animal model for the study of cell transplantation techniques for cartilage repair.14 It was hypothesized that hyaline-like cartilage would be produced within collagen scaffolds by 2D- and 3D-expanded BMSCs with an optimal seeding density of 10 × 106 cells/cm3.
Materials and Methods
Bone marrow aspiration and BMNC counting
BMAs were obtained from the iliac crest of six female Suffolk sheep (mean age ± standard error of the mean [SEM] of 3.3 ± 0.8 years) as previously described following ethical approval from the University of Alberta’s Animal Care and Use Committee (Table 1).13 Staining with crystal violet (Sigma-Aldrich, Oakville, Canada) and hemocytometer counting were used to determine the number of BMNCs in each BMA.
Table 1.
Bone Marrow Donor Information
| Donor | Gender | Age (years) | Mass (kg) |
|---|---|---|---|
| Z28 | Female | 2.0 | 66 |
| Z01 | Female | 2.2 | 71 |
| Z33 | Female | 2.3 | 81 |
| Y19 | Female | 3.2 | 63 |
| Y08 | Female | 3.3 | 94 |
| T10 | Female | 7.0 | 74 |
Culture of 2D-expanded BMSCs
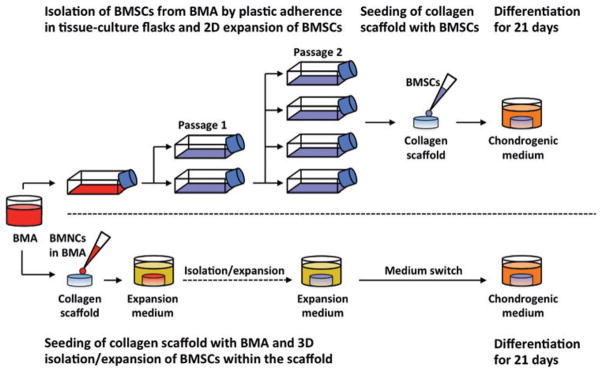
BMSCs were isolated in a 2D environment by plastic adherence from BMAs and expanded in tissue culture flasks to passage two (P2) within the expansion medium (Fig. 1). Expanded BMSCs were then seeded onto porous collagen scaffolds at 50, 10, 5, 1, or 0.5 × 106 BMSCs/cm3. Thereafter, BMSC–scaffold constructs were differentiated within the chondrogenic medium for 21 days.
FIG. 1.
In vitro cartilage engineering from two-dimensional (2D)-expanded and three-dimensional (3D)-expanded bone marrow-derived mesenchymal stromal stem cells (BMSCs). BMSCs were isolated in a 2D environment by plastic adherence from bone marrow aspirates (BMAs) and expanded in tissue culture flasks to passage two (P2) within expansion medium. BMSCs were then seeded onto porous collagen scaffolds at 50, 10, 5, 1, or 0.5 × 106 BMSCs/cm3. For 3D isolation and expansion, BMAs containing known numbers of bone marrow-derived mononucleated cells (BMNCs) were seeded onto porous collagen scaffolds at densities of 50, 10, 5, 1, or 0.5 × 106 BMNCs/cm3 and cultured in expansion medium. Thereafter, all constructs were differentiated within chondrogenic medium for 21 days. Color images available online at www.liebertpub.com/tec
Two-dimensional isolation and expansion involved seeding whole BMA collections containing 8 × 107 BMNCs within each 150 cm2 tissue culture flask and submersing each BMA collection in the expansion medium containing the alpha-minimal essential medium (α-MEM; Corning-Mediatech, Manassas, VA) supplemented with 8.8% vol/vol heat-inactivated fetal bovine serum (FBS), 88.5 units/mL penicillin, 88.5 μg/ mL streptomycin, 258.4 μg/mL L-glutamine, 8.8 mM 4-(2-hydroxyethyl)-1-piperazineethanesulfonic acid (HEPES), 885.0 μM sodium pyruvate (all from Life Technologies, Burlington, Canada), and 5 ng/mL fibroblast growth factor-2 (FGF-2; Neuromics, Edina, MN).
Cells were allowed to adhere and grow statically for 7 days in a humidified incubator. The humidified incubator used throughout the entirety of the study for cell and tissue culture contained air with 3% O2 and 5% CO2 heated to 37°C. After 7 days, the media were changed twice per week. Once 80% cell confluence was attained, adherent BMSCs were washed with phosphate-buffered saline, detached using 0.05% wt/vol trypsin-ethylenediaminetetraacetic acid (EDTA; Corning-Mediatech), and expanded to P2.
For chondrogenic differentiation, 2D-expanded BMSCs were suspended in the chondrogenic medium consisting of Dulbecco’s modified Eagle’s medium containing 4.5 mg/mL D-glucose, 110 μg/mL sodium pyruvate and L-glutamine (Sigma-Aldrich) supplemented with 9.6 mM HEPES, 95.6 units/mL penicillin, 95.6 μg/mL streptomycin, 279.2 μg/mL L-glutamine (all from Life Technologies), 1× insulin–transferrin–selenium (ITS)+ premix (BD Biosciences, Mississauga, Canada), 365 μg/mL ascorbic acid 2-phosphate, 40 μg/mL L-proline, 100 nM dexamethasone, 125 μg/mL human serum albumin (all from Sigma-Aldrich), and 10 ng/mL transforming growth factor-beta three (TGF-β3; ProSpec, East Brunswick, NJ).
Cylindrical collagen scaffolds (3.5 mm thickness; 6 mm diameter) were created using a biopsy punch on sheets of type I collagen sponge (125 × 100 × 3.5 mm3 dimension; 115 ± 20 μm pore size; Integra LifeSciences, Plainsboro, NJ). Cell counts were calculated using trypan blue staining and hemocytometer counting, and BMSCs were micro-pipetted onto each scaffold within a 20 μL chondrogenic medium suspension. Scaffold seeding was performed at five BMSC densities (Table 2). Seeded scaffolds were incubated for 15 min followed by addition of 100 μL of chondrogenic medium to the base of each scaffold. Thereafter, constructs were incubated for an additional 30 min to promote cell adhesion and then submersed in 1 mL of chondrogenic medium. Constructs were cultured statically for 21 days within a humidified incubator. Media were changed twice per week.
Table 2.
Cell Seeding Density
| Density (×106 cells/cm3) | Number of cells seeded |
|---|---|
| 50 | 4,948,000 |
| 10 | 989,600 |
| 5 | 494,800 |
| 1 | 98,960 |
| 0.5 | 49,480 |
Culture of 3D-expanded BMSCs
BMAs containing known numbers of BMNCs were seeded onto collagen scaffolds at densities of 50, 10, 5, 1, or 0.5 × 106 BMNCs/cm3 and cultured in the expansion medium to foster 3D isolation and expansion of BMSCs. Following the expansion period, all constructs were differentiated within the chondrogenic medium for 21 days (Fig. 1).
For 3D isolation and expansion, scaffolds were prepared in an identical manner to the protocol used for seeding of 2D-expanded BMSCs. During seeding, BMAs containing known numbers of BMNCs were centrifuged and suspended within the expansion medium to create 20 μL seeding collections that were micropipetted onto scaffolds. Scaffold seeding was performed at five BMNC densities (Table 2).
Seeded scaffolds were incubated for 15 min followed by addition of 100 μL of expansion medium to the base of each scaffold. Constructs were incubated for an additional 30 min to promote cell adhesion and were subsequently immersed in 1 mL of expansion medium and cultured within a humidified incubator. In an identical manner to the 2D expansion group, BMA-seeded scaffolds were cultured statically for 7 days undisturbed. Media were changed twice weekly thereafter. For each donor, 3D expansion was carried out for the equivalent amount of time as 2D expansion.
To induce chondrogenic differentiation, the medium used for culture of 3D-expanded BMSC-seeded scaffolds was switched from 1 mL of expansion medium to 1 mL of chondrogenic medium at the same time point as chondrogenic culture commenced for 2D-expanded BMSC-seeded scaffolds. Constructs were cultured statically for 21 days within a humidified incubator. Media were changed twice per week.
Cell count and population doubling during isolation and expansion
A colony-forming unit fibroblastic (CFU-F) assay of P0 BMSCs was performed as previously described.13 Each colony was considered to have derived from a single isolated BMSC, which enabled calculation of the number of BMSCs isolated from 1 × 105 BMNCs that were seeded. The ratio of isolated BMSCs to seeded BMNCs was used to calculate the number of BMSCs arising from 8 × 107 BMNCs plated in each tissue culture flask at P0. At the end of P0, P1, and P2, cell counts of BMSCs were determined using trypan blue staining and hemocytometer counting.
In the 3D expansion group, crystal violet staining and hemocytometer counting were used to calculate the quantity of BMNCs per volume of BMA. Known numbers of BMNCs were seeded within BMA collections onto scaffolds before expansion. The number of seeded BMSCs was estimated using the BMSC-to-BMNC ratio calculated from the CFU-F assay. Postexpansion cell counts were calculated from a standard curve (Supplementary Fig. S1; Supplementary Data are available online at www.liebertpub.com/tec) based on deoxyribonucleic acid (DNA) content derived from a Cy-QUANT Cell Proliferation Assay (Life Technologies). Population doublings were determined using the method described by Solchaga et al.33
Trilineage differentiation potential of BMSCs
To confirm the presence of cells with properties of mesenchymal stem cells within the BMAs, plastic adherent P2 BMSCs were cultured in osteogenic, adipogenic, and chondrogenic media and trilineage differentiation potential was demonstrated as previously described.13
Assessment of chondrogenesis
Tissue-engineered constructs were assessed with reverse-transcription quantitative polymerase chain reaction for gene expression (primers are shown in Table 3), safranin O staining of ECM proteoglycans, and biochemical quantification of glycosaminoglycan (GAG) and DNA as described previously.13 Histological scoring was also performed, and depositions of collagens I and II were assessed with immunofluorescence.
Table 3.
Ovine Primer Sequences Used in RT-qPCR Analysis
| Gene | Primer sequences | NCBI reference | |
|---|---|---|---|
| β-actin (ACTB) | 5′-CGGCGGGACCACCAT-3′ | Forward | NM_001009784.1 |
| 5′-GCAGTGATCTCTTTCTGCATCCT-3′ | Reverse | ||
| Aggrecan (ACAN) | 5′-TGGAATGATGTCCCATGCAA-3′ | Forward | XM_004018048.1 |
| 5′-GCCACTGTGCCCTTTTTACAG-3′ | Reverse | ||
| Collagen I (COL1A1) | 5′-CGCCCCAGACCAGGAATT-3′ | Forward | XM_004012773.1 |
| 5′-GTGGAAGGAGTTTACAGGAAGCA-3′ | Reverse | ||
| Collagen II (COL2A1) | 5′-ACCTCACGTCTCCCCATCA-3′ | Forward | XM_004006408.1 |
| 5′-CTGCTCGGGCCCTCCTAT-3′ | Reverse | ||
| Collagen X (COL10A1) | 5′-CAGGCTCGAATGGGCTGTAC-3′ | Forward | XM_004011185.1 |
| 5′-CCACCAAGAATCCTGAGAAAGAG-3′ | Reverse | ||
| SRY-Box 9 (SOX9) | 5′-GCTGCTGGCCGTGATGA-3′ | Forward | XM_004013527.1 |
| 5′-GGGTCGCGCGTTTGTTT-3′ | Reverse |
β-actin, beta-actin; RT-qPCR, reverse-transcription quantitative polymerase chain reaction; SOX9, sex determining region Y (SRY)-box 9.
Histological scoring using the Bern Score
Constructs containing BMSCs from five donors were fixed in 10% wt/vol buffered formalin, processed into paraffin wax, and sectioned at a thickness of 5 μm. Sections were stained with safranin O and fast green, as previously described,13 and graded using the Bern Score based on ECM staining, intercellular distance and ECM accumulation, and cell morphology.34 Each category was scored out of three with a combined maximal Bern Score of nine. Nonhomogeneous constructs were evaluated through assessing distinct regions and combining scores based on the percentage area represented as described by Grogan et al.34
Immunofluorescence
Sections with a thickness of 5 μm were treated with Protease XXV (Fisher Scientific, Ottawa, Canada) and hyaluronidase (Sigma-Aldrich) and incubated with rabbit anti-collagen I (NBP1-30054, 1:250 dilution; Novus Biologicals, Oakville, Canada) and mouse anti-collagen II (II-II6B3, 1:200 dilution; Developmental Studies Hybridoma Bank, Iowa City, IA). Immune localized antigens were visualized with Texas Red anti-rabbit IgG (Vector Laboratories, Burlington, Canada) and fluorescein isothiocyanate (FITC) goat anti-mouse Ig (BD Biosciences). Sections were mounted using the Biotium EverBrite Mounting Medium with DAPI (4′,6-diamidino-2-phenylindole; Cedarlane Laboratories, Burlington, Canada).
Images were captured using an Eclipse Ti-S microscope (Nikon Canada, Mississauga, Canada) fitted with NIS Elements (version 4.20; Nikon Canada) and assembled in Photoshop (Adobe Systems, San Jose, CA).
Statistical analysis
Analyses were performed using SPSS Statistics 22 (IBM, Armonk, NY) and significance was concluded when p < 0.05.
Repeated measures analysis of variance (ANOVA) was used for assessment of cell count and doubling over time during expansion and differentiation. For assessment of cell count over time within specific seeding density groups, a paired t-test was used. When different constructs were used at different time points, as in the analysis of peri-differentiation cell count of 3D-expanded BMSC-seeded scaffolds, a t-test was used given that pairing was not possible. Comparison of gene expressions, biochemical quantities, cell counts, and histological scores following chondrogenic differentiation between seeding groups required a Kruskal–Wallis one-way ANOVA with pairwise comparisons. A Mann–Whitney U test was used to compare predifferentiation to post-differentiation gene expressions.
Results
Isolation and expansion within 2D and 3D environments
The duration of time from plating of BMNCs to reach 80% confluence at P2 during 2D expansion was 23.8 ± 0.8 days. The mean number (±SEM) of BMSCs present per flask at the completion of P0 was 7.9 ± 1.4 × 106 and this number increased to 16.1 ± 1.6 × 106 and 27.6 ± 4.4 × 106 at the completion of P1 and P2, respectively (p < 0.001; Supplementary Fig. S2). BMSC count following 2D expansion was 34.2% ± 4.5% of the BMNC count initially seeded. There were 12.3 ± 1.0 cumulative BMSC population doublings during expansion. Based on the CFU-F assay, 18.0 ± 8.5 BMSCs were isolated from 1 × 105 seeded BMNCs. Therefore, 0.018% ± 0.009% of all seeded BMNCs was BMSCs.
Following 3D expansion, the number of BMSCs present was significantly lower than the initial number of BMNCs seeded in scaffolds at densities of 50, 10, and 5 × 106 BMNCs/cm3 (p < 0.001), while there was not a significant difference noted for densities of 1 and 0.5 × 106 BMNCs/cm3 (p = 0.87 and 1.0, respectively; Supplementary Fig. S2). For seeding densities of 50, 10, 5, 1, and 0.5 BMNCs/cm3, BMSC counts following 3D expansion were 0.29 ± 0.05, 0.15 ± 0.06, 0.12 ± 0.05, 0.09 ± 0.06, and 0.05 ± 0.05 × 106 BMSCs. These BMSC counts equated to 5.8% ± 0.9%, 14.9% ± 6.2%, 24.2% ± 9.9%, 89.5% ± 59.2%, and 100.3% ± 92.2% of initial BMNC counts, respectively.
Following expansion, cell densities were calculated to be 2.9 ± 0.5, 1.5 ± 0.6, 1.2 ± 0.5, 0.9 ± 0.6, and 0.5 ± 0.5 BMSCs/cm3, respectively. Cumulative BMSC population doublings were 9.3 ± 1.1, 9.7 ± 1.4, 10.2 ± 1.2, 11.1 ± 1.2, and 10.7 ± 1.2, respectively.
Trilineage differentiation of BMSCs
Spindle-shaped BMSCs demonstrated plastic adherence and were differentiated into cells capable of producing cartilaginous proteoglycans, adipose droplets, and calcified bone matrix (Supplementary Fig. S2).
Seeding density of 2D- and 3D-expanded BMSCs affected chondrogenic gene expression
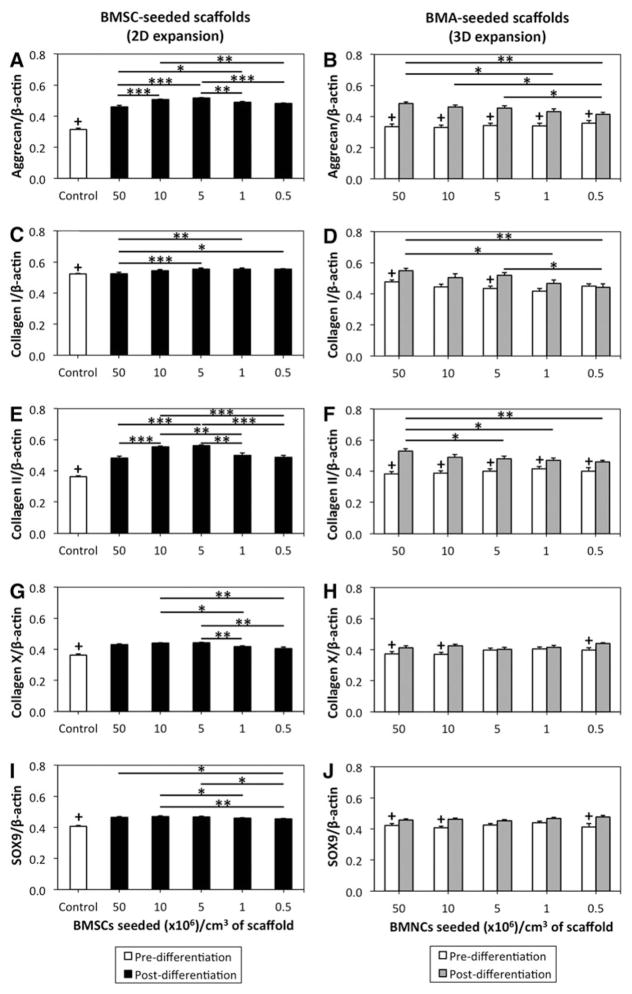
Constructs containing 2D- and 3D-expanded BMSCs had varying levels of cartilage-related genes that were dependent on seeding density following chondrogenic culture. In 2D-expanded BMSC-seeded scaffolds, messenger RNA (mRNA) expressions of hyaline cartilage-related collagen II and aggrecan were highest in scaffolds seeded at 5–10 × 106 BMSCs/cm3 (Fig. 2A, E).
FIG. 2.
Reverse-transcription quantitative polymerase chain reaction (RT-qPCR) for gene expression analysis of 2D- and 3D-expanded BMSCs seeded within collagen scaffolds. BMSCs were isolated and expanded within 2D and 3D environments, differentiated within collagen scaffolds for 21 days and assessed with RT-qPCR using SYBR Green detection. (A, C, E, G, I) Data represent the mean ± standard error of the mean (SEM) of 2D-expanded BMSCs in doublets based on seeding density group. (B, D, F, H, J) Data represent the mean ± SEM of 3D-expanded BMSCs in doublets based on seeding density group. Statistical analysis is represented by unlabeled, not significant; *p < 0.05; **p < 0.01; ***p < 0.001; +predifferentiation control value that is significantly different than differentiated BMSC-seeded scaffold (p < 0.05).
For aggrecan mRNA expression, constructs containing 10 and 5 × 106 BMSCs/cm3 had significantly enhanced aggrecan mRNA expression in comparison to scaffolds seeded with more cells (50 × 106 BMSCs/cm3; p < 0.001) and less cells (0.5 × 106 BMSCs/cm3; p < 0.01; Fig. 2A). Seeding at 5 × 106 BMSCs/cm3 also resulted in improved aggrecan mRNA expression relative to 1 × 106 BMSCs/cm3 (p < 0.01).
Densities of 10 and 5 × 106 BMSCs/cm3 corresponded to a greater collagen II mRNA expression than densities of 50, 1, and 0.5 × 106 BMSCs/cm3 (p < 0.01; Fig. 2E). Ratios of collagen II/I and collagen II/X also followed this pattern (p < 0.05). Expression of transcription factor sex determining region Y (SRY)-box 9 (SOX9) was not different between scaffolds seeded at 50, 10, and 5 × 106 BMSCs/cm3 (p ≥ 0.29), but these densities led to increased expression relative to 0.5 × 106 BMSCs/cm3 (p < 0.05; Fig. 2I).
With respect to fibrocartilage-related collagen I mRNA expression, constructs seeded at 5, 1, and 0.5 × 106 BMSCs/cm3 had a greater collagen I mRNA expression than 50 × 106 BMSCs/cm3 (p < 0.05; Fig. 2C). Expression of hypertrophic cartilage-associated collagen X was greater in constructs containing 10 and 5 BMSCs/cm3 than those containing 1 and 0.5 × 106 BMSCs/cm3 (p < 0.05; Fig. 2G).
In 3D-expanded BMSC-seeded scaffolds, aggrecan and collagen II mRNA expressions were highest in scaffolds seeded with BMA collections containing 50 × 106 BMNCs/cm3 (Fig. 2B, F). A significantly higher aggrecan mRNA expression was found in scaffolds seeded at 50, 10, and 5 × 106 BMNCs/ cm3 than 0.5 × 106 BMNCs/cm3 (p < 0.05), although the difference was most significant between 50 and 0.5 × 106 BMNCs/ cm3 (p < 0.01; Fig. 2B). Hyaline cartilage-related collagen II mRNA expression was significantly higher in scaffolds seeded at 50 × 106 BMNCs/cm3 than 5, 1, or 0.5 × 106 BMNCs/cm3 (p < 0.05) and approached a significantly higher level in scaffolds seeded at 50 × 106 BMNCs/cm3 relative to 10 × 106 BMNCs/cm3 (p = 0.07; Fig. 2F). Postdifferentiation collagen II mRNA was significantly greater than predifferentiation collagen II mRNA in all seeding density groups (p < 0.05).
For fibrocartilage-related collagen I, mRNA expression was more pronounced in constructs containing 50 × 106 BMNCs/ cm3 than 1 or 0.5 × 106 BMNCs/cm3 (p < 0.05; Fig. 2D). Significant differences were not noted between seeding density groups for collagen X and SOX9 mRNA expressions (p = 0.13 and 0.25; respectively; Fig. 2H, J). The collagen II/X ratio was higher in scaffolds seeded at 50 × 106 BMNCs/cm3 than 10, 1, and 0.5 × 106 BMNCs/cm3 (p < 0.05). The collagen II/I ratio was higher in scaffolds seeded at 0.5 × 106 BMNCs/cm3 than 50 and 5 × 106 BMNCs/cm3 (p < 0.05).
To determine whether BMNC seeding density during 3D isolation and expansion primed chondrogenic gene expression before onset of differentiation, 3D-expanded BMSC-seeded scaffolds were also assessed for gene expression directly following expansion. Seeding density did not significantly impact mRNA expressions of aggrecan, collagen I, collagen II, collagen X, or SOX9 (p = 0.82, 0.09, 0.64, 0.40, 0.40, respectively; Fig. 2 right column).
Seeding density of 2D- and 3D-expanded BMSCs affected chondrogenic ECM deposition
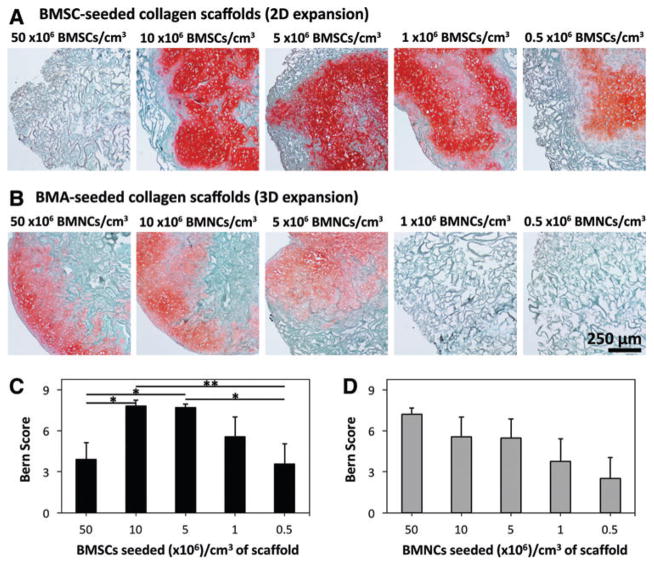
In 2D-expanded BMSC-seeded scaffolds, safranin O staining of proteoglycans appeared to be most pronounced in scaffolds seeded at 10 and 5 × 106 BMSCs/cm3, although abundant staining was also apparent in scaffolds seeded at other densities (Fig. 3A and Supplementary Fig. S3). Accordingly, Bern Scores were highest in scaffolds seeded at 10 and 5 × 106 BMSCs/cm3 (7.8 ± 0.4 and 7.7 ± 0.3, respectively; Fig. 3C and Supplementary Fig. S4). Scores for these densities were significantly larger than those for 50 and 0.5 × 106 BMSCs/cm3 (p < 0.05).
FIG. 3.
Histological analysis and scoring of 2D- and 3D-expanded BMSC-seeded collagen scaffolds. BMSCs were isolated and expanded within 2D and 3D environments, and differentiated within collagen scaffolds for 21 days. Thereafter, constructs were fixed, sectioned at 5 μm thickness, and stained with safranin O and fast green. Presented photomicrographs represent cell–scaffold constructs derived from (A) 2D-expanded BMSCs seeded at 50, 10, 5, 1, or 0.5 × 106 BMSCs/cm3, and (B) 3D-expanded BMSCs seeded at 50, 10, 5, 1, or 0.5 × 106 BMNCs/cm3 (cells from donor Z28; 10× magnification). (C) Mean (±SEM) Bern Score of 2D-expanded BMSCs from five donors based on seeding density group. (D) Mean (±SEM) Bern Score of 3D-expanded BMSCs from five donors based on seeding density group. Statistical analysis is represented by unlabeled, not significant; *p < 0.05; **p < 0.01. Color images available online at www.liebertpub.com/tec
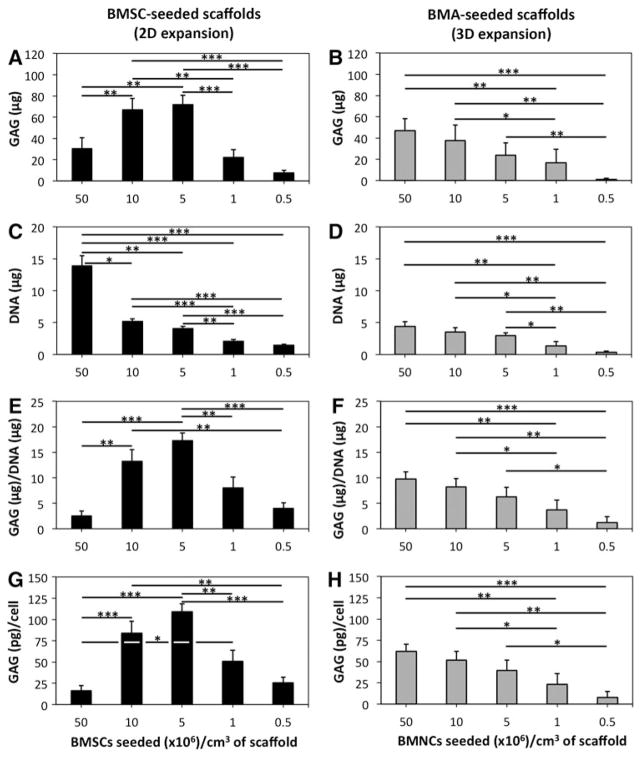
Analysis of proteoglycan quantity demonstrated that total GAG, GAG/DNA, and GAG/cell were highest in scaffolds seeded at 10 and 5 × 106 BMSCs/cm3 (Fig. 4A, E, G). There was not a significant difference between 10 and 5 × 106 BMSCs/cm3 for these parameters (p = 0.72, 0.18, and 0.21, respectively). Constructs containing 5 × 106 BMSCs/cm3 had significantly higher GAG/DNA than constructs containing 50, 1, and 0.5 × 106 BMSCs/cm3 (p < 0.01; Fig. 4E). Constructs containing 10 × 106 BMSCs/cm3 had significantly higher GAG/DNA than scaffolds seeded with 50 and 0.5 × 106 BMSCs/cm3 (p < 0.01), but were marginally different than 1 × 106 BMSCs/cm3 (p = 0.50; Fig. 4E).
FIG. 4.
Glycosaminoglycan (GAG) and deoxyribonucleic acid (DNA) quantification of 2D- and 3D-expanded BMSC-seeded collagen scaffolds. BMSCs were isolated and expanded within 2D and 3D environments and differentiated within collagen scaffolds for 21 days. Thereafter, constructs were assessed for GAG and DNA quantities. (A, C, E, G) Data represent the mean ± SEM of GAG quantity, DNA quantity, GAG/DNA, and GAG/ cell within constructs from six donors in doublets based on BMSC seeding density in 2D-expanded BMSC-seeded scaffolds. (B, D, F, H) Data represent the mean ± SEM of GAG quantity, DNA quantity, GAG/DNA, and GAG/ cell within constructs from six donors in doublets based on BMNC seeding density in 3D-expanded BMSC-seeded scaffolds. Statistical analysis is represented by unlabeled, not significant; *p < 0.05; **p < 0.01; ***p < 0.001.
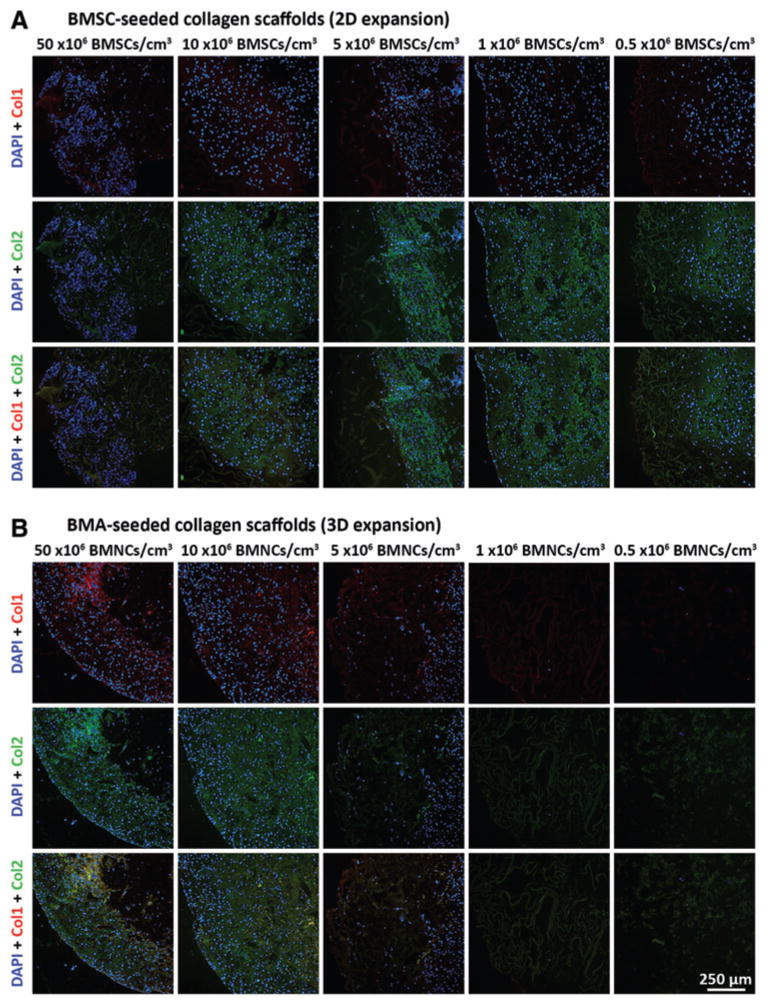
Collagen II deposition was pronounced in scaffolds seeded at 10, 5, 1, and 0.5 × 106 BMSCs/cm3, while scaffolds seeded at all densities appeared to lack abundant collagen I deposition (Fig. 5A; see Supplementary Fig. S5 for controls).
FIG. 5.
Immuno-fluorescence analysis of collagen I and collagen II content within 2D- and 3D-expanded BMSC-seeded collagen scaffolds. BMSCs were isolated and expanded within 2D and 3D environments and differentiated within collagen scaffolds for 21 days in chondrogenic medium. Thereafter, constructs were fixed, sectioned at 5 μm thickness, and processed for visualization of cells (DAPI), collagen I (Col I; Texas Red) and collagen II (Col II; FITC). Presented photomicrographs represent cell–scaffold constructs derived from (A) 2D-expanded BMSCs seeded at 50, 10, 5, 1, or 0.5 × 106 BMSCs/cm3, and (B) 3D-expanded BMSCs seeded at 50, 10, 5, 1, or 0.5 × 106 BMNCs/cm3 (cells from donor Z28; 10× magnification). DAPI, 4′,6-diamidino-2-phenylindole. FITC, fluorescein isothiocyanate. Color images available online at www.liebertpub.com/tec
During chondrogenic differentiation of 2D-expanded BMSCs, cell counts decreased in scaffolds that were seeded at densities of 50 and 10 × 106 BMSCs/cm3 (p < 0.05), while cell counts increased in scaffolds that were seeded at densities of 5, 1, and 0.5 × 106 BMSCs/cm3 (p < 0.05; Fig. 6A). At the end of the differentiation period, cell counts were not significantly different between scaffolds seeded at 10 and 5 × 106 BMSCs/cm3 (p = 0.19). There was also not a significant difference between 1 and 0.5 × 106 BMSCs/cm3 (p = 0.27). Cell counts between other seeding groups were notably different (p < 0.05).
FIG. 6.
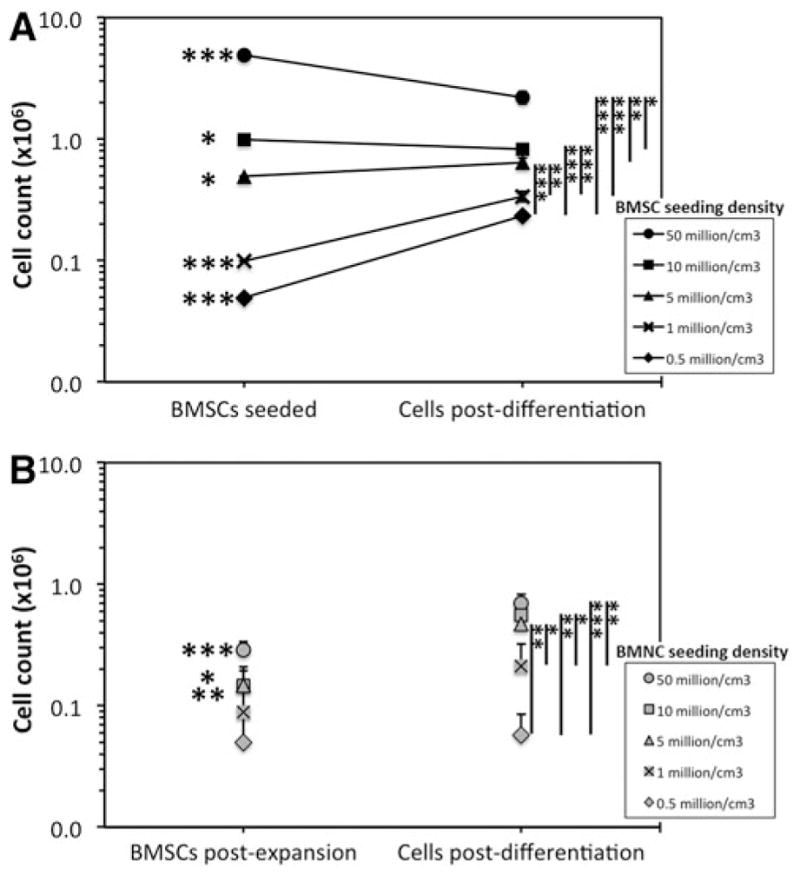
Peri-differentiation cell count of 2D- and 3D-expanded BMSC-seeded collagen scaffolds. (A) BMSCs were isolated and expanded within tissue culture flasks containing expansion medium (2D environment), seeded at specific cell counts within collagen scaffolds to create five seeding densities and differentiated for 21 days in chondrogenic medium. DNA levels were then quantified and used to calculate cell count following differentiation. (B) BMAs were seeded onto collagen scaffolds (3D environment) to create five BMNC densities, cultured in expansion medium, and subsequently differentiated within chondrogenic medium for 21 days. DNA levels were quantified in one group of scaffolds after expansion and another group of scaffolds after differentiation. Data represent the mean ± SEM of cell counts from six donors. Statistical analysis is represented by unlabeled, not significant; *p < 0.05; **p < 0.01; ***p < 0.001.
Scaffolds seeded with 3D-expanded BMSCs contained cartilaginous proteoglycans following differentiation. Proteoglycan staining appeared to be most pronounced in scaffolds seeded at 50, 10, and 5 × 106 BMNCs/cm3 (Fig. 3B), although variability was noted between donors, and cells from one donor (Z33) produced large depositions of proteoglycans at 1 × 106 BMNCs/cm3 (Supplementary Fig. S3). Bern Scores were highest in scaffolds seeded at 50 × 106 BMNCs/cm3 (7.2 ± 0.5; Fig. 3). However, a significant difference in scores between seeding densities was not present based on Kruskal–Wallis ANOVA (p = 0.16).
Quantitative analysis of proteoglycan deposition demonstrated that seeding at 50 × 106 BMNCs/cm3 led to higher total GAG, GAG/DNA, and GAG/cell than scaffolds seeding at 1 × 106 BMNCs/cm3 (p < 0.01) and 0.5 × 106 BMNCs/cm3 (p < 0.001; Fig. 4B, F, H). For these parameters, less significant differences were noted between 10 × 106 BMNCs/cm3 and lower densities of 1 × 106 BMNCs/cm3 (p < 0.05) and 0.5 × 106 BMNCs/cm3 (p < 0.01). Scaffolds seeded at 5 × 106 BMNCs/cm3 also had higher total GAG, GAG/DNA, and GAG/cell than scaffolds seeded at 0.5 × 106 BMNCs/cm3 (p < 0.05). Immunofluorescence indicated deposition of both collagens I and II, particularly in scaffolds seeded at 50 and 10 × 106 BMNCs/cm3 (Fig. 5B).
Following chondrogenic culture of 3D-expanded BMSC-seeded scaffolds, cell counts were not significantly different between scaffolds seeded at 50 and 10 × 106 BMNCs/cm3, or between scaffolds seeded at 1 and 0.5 × 106 BMNCs/cm3 (p ≥ 0.22; Fig. 6B). Seeding at 1 and 0.5 × 106 BMNCs/cm3 resulted in postdifferentiation BMSC counts of 0.21 ± 0.11 and 0.06 ± 0.03 × 106. Considerable variation in BMSC counts between donors was noted at these lowest BMNC seeding densities as some scaffolds appeared to have very low cell numbers present following differentiation, while others contained sufficient cell numbers to produce significant levels of cartilaginous ECM (Supplementary Fig. S3 and Fig. 5B).
Discussion
The major finding of this study is that isolation and expansion of ovine BMSCs under 2D and 3D conditions yielded cell populations capable of producing hyaline-like neocartilage in vitro, and chondrogenic differentiation of these cells was dependent on seeding density within collagen I scaffolds. Optimal seeding densities were identified for 2D- and 3D-expanded BMSCs.
Two-dimensional isolation and expansion were performed using a conventional protocol within tissue culture flasks.9,10 Whole BMA was seeded into flasks containing the expansion medium in an attempt to maintain the natural bone marrow microenvironment on the 2D surface of the tissue culture flask during isolation and the first week of expansion,11,13,35 while avoiding the loss of cells and mediators that could be discarded through the use of cell separation techniques.9,10 An extended expansion period beyond P2 was not used in this study as it was previously shown that prolonged expansion leads to dedifferentiation, senescence, and loss of multipotential capacity of BMSCs.17,18
Three-dimensional isolation and expansion were performed by seeding BMA collections containing defined numbers of BMNCs within scaffolds followed by culture in the expansion medium. The goal of this technique was to maintain BMSCs within a 3D environment and to avoid subjecting cells to the artificial environment within tissue culture flasks. Presumably, BMSCs adhered to the scaffold through integrin-mediated binding to type I collagen,36 while other cellular and chemical components of bone marrow37–40 were entrapped within scaffold pores but washed away over the course of the expansion period with media changes. BMA collections were seeded within scaffolds to create seeding densities of 50, 10, 5, 1, and 0.5 × 106 BMNCs/cm3.
The total number of BMSCs present following isolation and expansion was 5.8% of the initial BMNC count in the scaffolds seeded at 50 × 106 BMNCs/cm3. The percentage of postexpansion BMSCs increased as BMNC seeding density decreased and was measured to be 100.3% in scaffolds seeded at the lowest density of 0.5 × 106 BMNCs/cm3. Increased proliferation may have played a role in this effect, as population doublings were higher in constructs seeded at lower densities. Augmented proliferation at lower density was reported previously during both 2D and 3D expansion,41–44 and could involve reduced contact inhibition and increased nutrient availability per cell.45 Higher probability of attachment at the time of seeding due to a relatively increased availability of scaffold binding sites could also increase BMSC count at lower BMNC seeding densities.
The impact of seeding density on chondrogenesis was evaluated after 21 days of in vitro culture. To date, multiple parameters have been used in the literature to assess hyaline-like chondrogenic capacity, including proteoglycan content, histological scoring, GAG/DNA, collagen II quantity, and collagen II/I ratio.9,10,12,13,33,34 These parameters involve either gene expression or measurement of macromolecule deposition in the ECM.
A number of assays were used in the study at hand to provide a rigorous and multimodal evaluation of engineered tissue. Neocartilage was considered hyaline like if (1) collagen II was present based on collagen II mRNA expression, collagen II/I mRNA ratio and collagen II immunofluorescence; and (2) proteoglycan was abundant as measured through aggrecan mRNA expression, safranin O staining, Bern Score, total GAG, and GAG/DNA. Neocartilage was considered to be superior if these parameters were more pronounced in one construct relative to another construct.
Following 2D expansion, BMSCs seeded at all densities were shown to be capable of hyaline-like cartilaginous tissue formation based on increased collagen II and aggrecan mRNA expressions relative to controls, and proteoglycan deposition. Collagen scaffolds seeded at 10 and 5 × 106 BMSCs/cm3 had the highest mRNA expressions of hyaline cartilage-related collagen II, aggrecan, and SOX9, although expressions of collagens I and X that relate to fibrocartilage and hypertrophic cartilage, respectively, were also highest in these constructs.
Scaffolds seeded at 10 and 5 × 106 BMSCs/cm3 produced the highest levels of ECM proteoglycan deposition and were found to be superior based on histological scoring. Collagen II deposition was apparent in scaffolds seeded at 0.5–10 × 106 BMSCs/cm3, while collagen I was lacking. Constructs containing 50 × 106 BMSCs/cm3 at the time of seeding displayed stunted chondrogenesis in comparison to 10 and 5 × 106 BMSCs/cm3, which could relate to cell crowding, nutrient limitation, and relatively less volume available for ECM deposition.45,46 These findings suggest that the optimal seeding density for chondrogenesis of 2D-expanded BMSCs within collagen scaffolds is 5–10 × 106 BMSCs/cm3.
In 3D-expanded BMSC-seeded scaffolds, constructs seeded at all densities produced increased collagen II and aggrecan mRNA expressions relative to controls, and proteoglycan deposition. Constructs seeded at 50 × 106 BMNCs/ cm3 had the highest levels of aggrecan mRNA expression and ECM proteoglycan deposition, although scaffolds seeded at 10 and 5 × 106 BMNCs/cm3 also had higher levels of proteoglycans in comparison to lower seeding densities. The Bern Score was highest in scaffolds seeded at 50 × 106 BMNCs/cm3, but a significant difference was not demonstrated between seeding groups. Collagen II mRNA expression was highest in scaffolds seeded at 50 × 106 BMNCs/cm3.
Based on immunofluorescence, deposition of collagen II was particularly notable within scaffolds seeded at both 50 and 10 × 106 BMNCs/cm3. Collagen I mRNA expressions was highest in scaffolds seeded at 50, 10, and 5 × 106 BMNCs/cm3, while immunofluorescence showed significant deposition in scaffolds seeded at 50 and 10 × 106 BMNCs/cm3. The results of this study suggest that a seeding density of 50 × 106 BMNCs/cm3 leads to the highest level of hyaline-like chondrogenesis, although it is possible that higher densities that were not assessed in this study could further augment chondrogenesis.
At present, there is a paucity of literature examining the impact of BMSC seeding density on chondrogenesis. Several in vitro studies have assessed the impact of mature chondrocyte density on chondrogenesis. Hansen et al. demonstrated that seeding at 1.2 × 106 chondrocytes/cm3 augmented chondrogenesis within a porous methoxypolyethylene glycol-polylactic-co-glycolic acid scaffold relative to higher densities of 4–20 × 106 chondrocytes/cm3.32
Mahmoudifar and Doran and Vunjak-Novakovic et al. showed superior chondrogenesis within polyglycolic acid scaffolds seeded with 25–27 × 106 chondrocytes/cm3 in comparison to lower densities of 5–15 × 106 chondrocytes/cm3.47,48 Francioli et al. reported that chondrogenesis improved by increasing cell density within porous collagen II scaffolds from 25 to 66 × 106 chondrocytes/cm3.49 In agarose hydrogels, Huang et al. demonstrated that a density of 9 × 106 chondrocytes/cm3 was superior to 3–6 × 106 chondrocytes/cm3, while Mauck et al. found that 60 × 106 chondrocytes/cm3 augmented chondrogenesis relative to 20 × 106 chondrocytes/cm3.50,51
Cell density analysis in this study was based on the number of cells and volume of scaffold present during seeding, as this is the time point at which these parameters would be known in a clinical setting.4 It was demonstrated that cell count following differentiation varied in comparison to predifferentiation cell count, and this could have involved a balance between multiple factors, including cell loss during scaffold seeding and culture, and ongoing proliferation during the differentiation period.52 With the assumption that construct volume was maintained during chondrogenesis, final cell densities at the end of in vitro chondrogenesis were 22.3, 8.4, 6.5, 3.4, and 2.4 × 106 BMSCs/ cm3 for 2D-expanded BMSCs seeded at 50, 10, 5, 1, and 0.5 × 106 BMSCs/cm3, respectively, and 7.1, 5.6, 4.8, 2.1, and 0.6 × 106 BMSCs/cm3 for 3D-expanded BMSCs seeded at 50, 10, 5, 1, and 0.5 × 106 BMNCs/cm3, respectively.
Cell-seeded collagen scaffolds have been shown previously to decrease in volume during chondrogenesis as a result of cell-mediated contraction and degradation of scaffold biomaterials.53 Therefore, cell densities at the end of chondrogenic culture were potentially higher than calculated.
Although both 2D- and 3D-expanded BMSC-seeded collagen scaffolds were assessed and shown to produce bioengineered tissue with properties of hyaline cartilage, a direct comparison of these methods was not performed given that the properties of the cells populating scaffolds at the time of isolation and following culture may have differed. Two-dimensional-expanded BMSCs were shown to have qualities of plastic adherence and trilineage differentiation. Previously, our group characterized cell surface markers of BMSCs isolated and expanded using this isolation method.11
Although 3D-expanded BMSCs were derived from the same BMA collections as 2D-expanded BMSCs, the cells that adhered to the collagen scaffold during 3D isolation were not characterized and may have derived from a different subpopulation of BMSCs than those that adhered to plastic during 2D isolation. It was shown previously that different subpopulations of BMSCs are present based on adherence and cell surface markers.20,54 Future study is required to characterize cells derived from 3D isolation and to compare these cells to those derived from 2D methods utilizing whole BMA and cell fractions (BMNCs).
This study has some limitations. Static culture conditions were used, which may have led to less uniform densities throughout scaffolds relative to dynamic conditions.
Second, ovine rather than human BMSCs were used. Sheep are routinely involved as animal models in cartilage engineering studies.14,55,56 However, differences between species could impact the clinical applicability of our findings. Aspirates were derived from sheep at an age that is consistent with skeletal maturity and cartilage maturity based on zonal architecture, presence of a continuous calcified cartilage layer, and lack of spontaneous cartilage repair.57 Given that sheep have a life expectancy of 10–12 years, the sheep in this study (mean age of 3.3 years) could correspond to an adult human population that would be amenable to cell-based cartilage restoration techniques.
Only female sheep were included as donors. A difference in in vitro chondrogenesis between cells from male and female animals and humans has been demonstrated previously with a higher chondrogenic capacity demonstrated in males.58,59 Although the use of female sheep potentially reduced variability that may have been caused through the use of a mixed sample of animals, the impact of seeding density on chondrogenesis could be different in male animals.
Third, an in vitro model was used, and the applicability of the conclusions to clinical use is unclear. It is possible that remodeling that occurs in vivo over time could offset differences that are apparent within this in vitro model.32,60
Fourth, seeding of cells within biomaterial scaffolds was considered to be an effective process in this study and the number of cells that did not adhere was not determined.
Finally, during the calculation of population doublings during 3D expansion, the initial number of BMSCs present following isolation required the use of a ratio derived from the CFU-F assay. Given that the CFU-F involved plastic adherence, it is possible that the fraction of BMSCs present from the BMNC pool following 3D isolation was different than that calculated from the CFU-F assay.
In conclusion, BMSCs isolated and expanded in 2D and 3D environments are capable of producing hyaline-like cartilaginous tissue within a porous collagen I scaffold. Chondrogenesis appeared to be most pronounced with seeding densities of 5–10 × 106 BMSCs/cm3 and 50 × 106 BMNCs/ cm3 for 2D and 3D expansion protocols, respectively. Accordingly, these densities could be considered when seeding collagen I scaffolds in BMSC transplantation protocols.
Supplementary Material
Acknowledgments
Financial support was provided by the University Hospital Foundation at the University of Alberta Hospital, Canadian Institutes of Health Research (CIHR MOP 287058) and the Edmonton Orthopaedic Research Committee. Integra LifeSciences Corp. supplied the collagen scaffold for this study. The authors thank Dr. Thomas Churchill for providing us with photography and histology equipment, Mr. Joshua Hahn for his expertise in microscopy, and Dr. Craig Wilkinson and Ms. Brenda Tchir for their involvement in animal monitoring and care.
Footnotes
Disclosure Statement
No competing financial interests exist.
References
- 1.Wakitani S, Goto T, Pineda SJ, Young RG, Mansour JM, Caplan AI, Goldberg VM. Mesenchymal cell-based repair of large, full-thickness defects of articular cartilage. J Bone Joint Surg Am. 1994;76:579. doi: 10.2106/00004623-199404000-00013. [DOI] [PubMed] [Google Scholar]
- 2.Bornes TD, Adesida AB, Jomha NM. Mesenchymal stem cells in the treatment of traumatic articular cartilage defects: a comprehensive review. Arthritis Res Ther. 2014;16:432. doi: 10.1186/s13075-014-0432-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Wakitani S, Nawata M, Tensho K, Okabe T, Machida H, Ohgushi H. Repair of articular cartilage defects in the patello-femoral joint with autologous bone marrow mesenchymal cell transplantation: three case reports involving nine defects in five knees. J Tissue Eng Regen Med. 2007;1:74. doi: 10.1002/term.8. [DOI] [PubMed] [Google Scholar]
- 4.Kuroda R, Ishida K, Matsumoto T, Akisue T, Fujioka H, Mizuno K, Ohgushi H, Wakitani S, Kurosaka M. Treatment of a full-thickness articular cartilage defect in the femoral condyle of an athlete with autologous bone-marrow stromal cells. Osteoarthritis Cartilage. 2007;15:226. doi: 10.1016/j.joca.2006.08.008. [DOI] [PubMed] [Google Scholar]
- 5.Gobbi A, Karnatzikos G, Scotti C, Mahajan M, Mazzucco L, Grigolo B. One-step cartilage repair with bone marrow aspirate concentrated cells and collagen matrix in full-thickness knee cartilage lesions: results at 2-year follow-up. Cartilage. 2011;2:286. doi: 10.1177/1947603510392023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Buda R, Vannini F, Cavallo M, Grigolo B, Cenacchi A, Giannini S. Osteochondral lesions of the knee: a new one-step repair technique with bone-marrow-derived cells. J Bone Joint Surg Am. 2010;92(Suppl 2):2. doi: 10.2106/JBJS.J.00813. [DOI] [PubMed] [Google Scholar]
- 7.Giannini S, Buda R, Battaglia M, Cavallo M, Ruffilli A, Ramponi L, Pagliazzi G, Vannini F. One-step repair in talar osteochondral lesions: 4-year clinical results and t2-mapping capability in outcome prediction. Am J Sports Med. 2013;41:511. doi: 10.1177/0363546512467622. [DOI] [PubMed] [Google Scholar]
- 8.Enea D, Cecconi S, Calcagno S, Busilacchi A, Manzotti S, Kaps C, Gigante A. Single-stage cartilage repair in the knee with microfracture covered with a resorbable polymer-based matrix and autologous bone marrow concentrate. Knee. 2013;20:562. doi: 10.1016/j.knee.2013.04.003. [DOI] [PubMed] [Google Scholar]
- 9.Mackay AM, Beck SC, Murphy JM, Barry FP, Chichester CO, Pittenger MF. Chondrogenic differentiation of cultured human mesenchymal stem cells from marrow. Tissue Eng. 1998;4:415. doi: 10.1089/ten.1998.4.415. [DOI] [PubMed] [Google Scholar]
- 10.Pittenger MF, Mackay AM, Beck SC, Jaiswal RK, Douglas R, Mosca JD, Moorman MA, Simonetti DW, Craig S, Marshak DR. Multilineage potential of adult human mesenchymal stem cells. Science. 1999;284:143. doi: 10.1126/science.284.5411.143. [DOI] [PubMed] [Google Scholar]
- 11.Adesida AB, Mulet-Sierra A, Jomha NM. Hypoxia mediated isolation and expansion enhances the chondrogenic capacity of bone marrow mesenchymal stromal cells. Stem Cell Res Ther. 2012;3:9. doi: 10.1186/scrt100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Zscharnack M, Poesel C, Galle J, Bader A. Low oxygen expansion improves subsequent chondrogenesis of ovine bone-marrow-derived mesenchymal stem cells in collagen type I hydrogel. Cells Tissues Organs. 2009;190:81. doi: 10.1159/000178024. [DOI] [PubMed] [Google Scholar]
- 13.Bornes TD, Jomha NM, Mulet-Sierra A, Adesida AB. Hypoxic culture of bone marrow-derived mesenchymal stromal stem cells differentially enhances in vitro chondrogenesis within cell-seeded collagen and hyaluronic acid porous scaffolds. Stem Cell Res Ther. 2015;6:84. doi: 10.1186/s13287-015-0075-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Zscharnack M, Hepp P, Richter R, Aigner T, Schulz R, Somerson J, Josten C, Bader A, Marquass B. Repair of chronic osteochondral defects using predifferentiated mesenchymal stem cells in an ovine model. Am J Sports Med. 2010;38:1857. doi: 10.1177/0363546510365296. [DOI] [PubMed] [Google Scholar]
- 15.Banfi A, Muraglia A, Dozin B, Mastrogiacomo M, Cancedda R, Quarto R. Proliferation kinetics and differentiation potential of ex vivo expanded human bone marrow stromal cells: implications for their use in cell therapy. Exp Hematol. 2000;28:707. doi: 10.1016/s0301-472x(00)00160-0. [DOI] [PubMed] [Google Scholar]
- 16.Banfi A, Bianchi G, Notaro R, Luzzatto L, Cancedda R, Quarto R. Replicative aging and gene expression in long-term cultures of human bone marrow stromal cells. Tissue Eng. 2002;8:901. doi: 10.1089/107632702320934001. [DOI] [PubMed] [Google Scholar]
- 17.Tsai CC, Chen YJ, Yew TL, Chen LL, Wang JY, Chiu CH, Hung SC. Hypoxia inhibits senescence and maintains mesenchymal stem cell properties through down-regulation of E2A-p21 by HIF-TWIST. Blood. 2011;117:459. doi: 10.1182/blood-2010-05-287508. [DOI] [PubMed] [Google Scholar]
- 18.Wagner W, Horn P, Castoldi M, Diehlmann A, Bork S, Saffrich R, Benes V, Blake J, Pfister S, Eckstein V, Ho AD. Replicative senescence of mesenchymal stem cells: a continuous and organized process. PloS One. 2008;3:e2213. doi: 10.1371/journal.pone.0002213. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Walenda G, Hemeda H, Schneider RK, Merkel R, Hoffmann B, Wagner W. Human platelet lysate gel provides a novel three dimensional-matrix for enhanced culture expansion of mesenchymal stromal cells. Tissue Eng Part C Methods. 2012;18:924. doi: 10.1089/ten.TEC.2011.0541. [DOI] [PubMed] [Google Scholar]
- 20.Papadimitropoulos A, Piccinini E, Brachat S, Braccini A, Wendt D, Barbero A, Jacobi C, Martin I. Expansion of human mesenchymal stromal cells from fresh bone marrow in a 3D scaffold-based system under direct perfusion. PloS One. 2014;9:e102359. doi: 10.1371/journal.pone.0102359. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Wise JK, Alford AI, Goldstein SA, Stegemann JP. Comparison of uncultured marrow mononuclear cells and culture-expanded mesenchymal stem cells in 3D collagen-chitosan microbeads for orthopedic tissue engineering. Tissue Eng Part A. 2014;20:210. doi: 10.1089/ten.tea.2013.0151. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Hunziker EB, Quinn TM, Hauselmann HJ. Quantitative structural organization of normal adult human articular cartilage. Osteoarthritis Cartilage. 2002;10:564. doi: 10.1053/joca.2002.0814. [DOI] [PubMed] [Google Scholar]
- 23.Uematsu K, Hattori K, Ishimoto Y, Yamauchi J, Habata T, Takakura Y, Ohgushi H, Fukuchi T, Sato M. Cartilage regeneration using mesenchymal stem cells and a three-dimensional poly-lactic-glycolic acid (PLGA) scaffold. Biomaterials. 2005;26:4273. doi: 10.1016/j.biomaterials.2004.10.037. [DOI] [PubMed] [Google Scholar]
- 24.Wayne JS, McDowell CL, Shields KJ, Tuan RS. In vivo response of polylactic acid-alginate scaffolds and bone marrow-derived cells for cartilage tissue engineering. Tissue Eng. 2005;11:953. doi: 10.1089/ten.2005.11.953. [DOI] [PubMed] [Google Scholar]
- 25.Shao X, Goh JC, Hutmacher DW, Lee EH, Zigang G. Repair of large articular osteochondral defects using hybrid scaffolds and bone marrow-derived mesenchymal stem cells in a rabbit model. Tissue Eng. 2006;12:1539. doi: 10.1089/ten.2006.12.1539. [DOI] [PubMed] [Google Scholar]
- 26.Dragoo JL, Carlson G, McCormick F, Khan-Farooqi H, Zhu M, Zuk PA, Benhaim P. Healing full-thickness cartilage defects using adipose-derived stem cells. Tissue Eng. 2007;13:1615. doi: 10.1089/ten.2006.0249. [DOI] [PubMed] [Google Scholar]
- 27.Han SH, Kim YH, Park MS, Kim IA, Shin JW, Yang WI, Jee KS, Park KD, Ryu GH, Lee JW. Histological and biomechanical properties of regenerated articular cartilage using chondrogenic bone marrow stromal cells with a PLGA scaffold in vivo. J Biomed Mater Res A. 2008;87:850. doi: 10.1002/jbm.a.31828. [DOI] [PubMed] [Google Scholar]
- 28.Loken S, Jakobsen RB, Aroen A, Heir S, Shahdadfar A, Brinchmann JE, Engebretsen L, Reinholt FP. Bone marrow mesenchymal stem cells in a hyaluronan scaffold for treatment of an osteochondral defect in a rabbit model. Knee Surg Sports Traumatol Arthrosc. 2008;16:896. doi: 10.1007/s00167-008-0566-2. [DOI] [PubMed] [Google Scholar]
- 29.Dashtdar H, Rothan HA, Tay T, Ahmad RE, Ali R, Tay LX, Chong PP, Kamarul T. A preliminary study comparing the use of allogenic chondrogenic pre-differentiated and undifferentiated mesenchymal stem cells for the repair of full thickness articular cartilage defects in rabbits. J Orthop Res. 2011;29:1336. doi: 10.1002/jor.21413. [DOI] [PubMed] [Google Scholar]
- 30.Qi Y, Zhao T, Xu K, Dai T, Yan W. The restoration of full-thickness cartilage defects with mesenchymal stem cells (MSCs) loaded and cross-linked bilayer collagen scaffolds on rabbit model. Mol Biol Rep. 2012;39:1231. doi: 10.1007/s11033-011-0853-8. [DOI] [PubMed] [Google Scholar]
- 31.Chang NJ, Lam CF, Lin CC, Chen WL, Li CF, Lin YT, Yeh ML. Transplantation of autologous endothelial progenitor cells in porous PLGA scaffolds create a microenvironment for the regeneration of hyaline cartilage in rabbits. Osteoarthritis Cartilage. 2013;21:1613. doi: 10.1016/j.joca.2013.07.016. [DOI] [PubMed] [Google Scholar]
- 32.Hansen OM, Foldager CB, Christensen BB, Everland H, Lind M. Increased chondrocyte seeding density has no positive effect on cartilage repair in an MPEG-PLGA scaffold. Knee Surg Sports Traumatol Arthrosc. 2013;21:485. doi: 10.1007/s00167-012-1996-4. [DOI] [PubMed] [Google Scholar]
- 33.Solchaga LA, Penick K, Goldberg VM, Caplan AI, Welter JF. Fibroblast growth factor-2 enhances proliferation and delays loss of chondrogenic potential in human adult bone-marrow-derived mesenchymal stem cells. Tissue Eng Part A. 2010;16:1009. doi: 10.1089/ten.tea.2009.0100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Grogan SP, Barbero A, Winkelmann V, Rieser F, Fitzsimmons JS, O’Driscoll S, Martin I, Mainil-Varlet P. Visual histological grading system for the evaluation of in vitro-generated neocartilage. Tissue Eng. 2006;12:2141. doi: 10.1089/ten.2006.12.2141. [DOI] [PubMed] [Google Scholar]
- 35.Johnstone B, Hering TM, Caplan AI, Goldberg VM, Yoo JU. In vitro chondrogenesis of bone marrow-derived mesenchymal progenitor cells. Exp Cell Res. 1998;238:265. doi: 10.1006/excr.1997.3858. [DOI] [PubMed] [Google Scholar]
- 36.Gronthos S, Simmons PJ, Graves SE, Robey PG. Integrin-mediated interactions between human bone marrow stromal precursor cells and the extracellular matrix. Bone. 2001;28:174. doi: 10.1016/s8756-3282(00)00424-5. [DOI] [PubMed] [Google Scholar]
- 37.da Silva Meirelles L, Caplan AI, Nardi NB. In search of the in vivo identity of mesenchymal stem cells. Stem Cells. 2008;26:2287. doi: 10.1634/stemcells.2007-1122. [DOI] [PubMed] [Google Scholar]
- 38.Fortier LA, Barker JU, Strauss EJ, McCarrel TM, Cole BJ. The role of growth factors in cartilage repair. Clin Orthop Relat Res. 2011;469:2706. doi: 10.1007/s11999-011-1857-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Hoggatt J, Scadden DT. The stem cell niche: tissue physiology at a single cell level. J Clin Invest. 2012;122:3029. doi: 10.1172/JCI60238. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Fortier L, Cassano J, Kennedy J, Ross K, Fraser E, Goodale M. Chondrogenic molecules in bone marrow concentrate and platelet rich plasma. Abstract presented at the International Cartilage Repair Society World Congress; Chicago, IL. 2015. Abstract no. 2.1.2. [Google Scholar]
- 41.Both SK, van der Muijsenberg AJ, van Blitterswijk CA, de Boer J, de Bruijn JD. A rapid and efficient method for expansion of human mesenchymal stem cells. Tissue Eng. 2007;13:3. doi: 10.1089/ten.2005.0513. [DOI] [PubMed] [Google Scholar]
- 42.Lode A, Bernhardt A, Gelinsky M. Cultivation of human bone marrow stromal cells on three-dimensional scaffolds of mineralized collagen: influence of seeding density on colonization, proliferation and osteogenic differentiation. J Tissue Eng Regen Med. 2008;2:400. doi: 10.1002/term.110. [DOI] [PubMed] [Google Scholar]
- 43.Bartmann C, Rohde E, Schallmoser K, Purstner P, Lanzer G, Linkesch W, Strunk D. Two steps to functional mesenchymal stromal cells for clinical application. Transfusion. 2007;47:1426. doi: 10.1111/j.1537-2995.2007.01219.x. [DOI] [PubMed] [Google Scholar]
- 44.Colter DC, Class R, DiGirolamo CM, Prockop DJ. Rapid expansion of recycling stem cells in cultures of plastic-adherent cells from human bone marrow. Proc Natl Acad Sci U S A. 2000;97:3213. doi: 10.1073/pnas.070034097. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Fossett E, Khan WS. Optimising human mesenchymal stem cell numbers for clinical application: a literature review. Stem Cells Int. 2012;2012:465259. doi: 10.1155/2012/465259. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Foldager CB. Advances in autologous chondrocyte implantation and related techniques for cartilage repair. Dan Med J. 2013;60:B4600. [PubMed] [Google Scholar]
- 47.Mahmoudifar N, Doran PM. Effect of seeding and bioreactor culture conditions on the development of human tissue-engineered cartilage. Tissue Eng. 2006;12:1675. doi: 10.1089/ten.2006.12.1675. [DOI] [PubMed] [Google Scholar]
- 48.Vunjak-Novakovic G, Obradovic B, Martin I, Bursac PM, Langer R, Freed LE. Dynamic cell seeding of polymer scaffolds for cartilage tissue engineering. Biotechnol Prog. 1998;14:193. doi: 10.1021/bp970120j. [DOI] [PubMed] [Google Scholar]
- 49.Francioli SE, Candrian C, Martin K, Heberer M, Martin I, Barbero A. Effect of three-dimensional expansion and cell seeding density on the cartilage-forming capacity of human articular chondrocytes in type II collagen sponges. J Biomed Mater Res A. 2010;95:924. doi: 10.1002/jbm.a.32917. [DOI] [PubMed] [Google Scholar]
- 50.Huang CY, Reuben PM, D’Ippolito G, Schiller PC, Cheung HS. Chondrogenesis of human bone marrow-derived mesenchymal stem cells in agarose culture. Anat Rec A Discov Mol Cell Evol Biol. 2004;278:428. doi: 10.1002/ar.a.20010. [DOI] [PubMed] [Google Scholar]
- 51.Mauck RL, Seyhan SL, Ateshian GA, Hung CT. Influence of seeding density and dynamic deformational loading on the developing structure/function relationships of chondrocyte-seeded agarose hydrogels. Ann Biomed Eng. 2002;30:1046. doi: 10.1114/1.1512676. [DOI] [PubMed] [Google Scholar]
- 52.Kim DH, Kim DD, Yoon IS. Proliferation and chondrogenic differentiation of human adipose-derived mesenchymal stem cells in sodium alginate beads with or without hyaluronic acid. J Pharmaceut Inv. 2013;43:145. [Google Scholar]
- 53.Vickers SM, Squitieri LS, Spector M. Effects of cross-linking type II collagen-GAG scaffolds on chondrogenesis in vitro: dynamic pore reduction promotes cartilage formation. Tissue Eng. 2006;12:1345. doi: 10.1089/ten.2006.12.1345. [DOI] [PubMed] [Google Scholar]
- 54.Di Maggio N, Mehrkens A, Papadimitropoulos A, Schaeren S, Heberer M, Banfi A, Martin I. Fibroblast growth factor-2 maintains a niche-dependent population of self-renewing highly potent non-adherent mesenchymal progenitors through FGFR2c. Stem Cells. 2012;30:1455. doi: 10.1002/stem.1106. [DOI] [PubMed] [Google Scholar]
- 55.Schinhan M, Gruber M, Vavken P, Dorotka R, Samouh L, Chiari C, Gruebl-Barabas R, Nehrer S. Critical-size defect induces unicompartmental osteoarthritis in a stable ovine knee. J Orthop Res. 2012;30:214. doi: 10.1002/jor.21521. [DOI] [PubMed] [Google Scholar]
- 56.Marquass B, Schulz R, Hepp P, Zscharnack M, Aigner T, Schmidt S, Stein F, Richter R, Osterhoff G, Aust G, Josten C, Bader A. Matrix-associated implantation of predifferentiated mesenchymal stem cells versus articular chondrocytes: in vivo results of cartilage repair after 1 year. Am J Sports Med. 2011;39:1401. doi: 10.1177/0363546511398646. [DOI] [PubMed] [Google Scholar]
- 57.Hurtig MB, Buschmann MD, Fortier LA, Hoemann CD, Hunziker EB, Jurvelin JS, Mainil-Varlet P, McIlwraith CW, Sah RL, Whiteside RA. Preclinical studies for cartilage repair: recommendations from the International Cartilage Repair Society. Cartilage. 2011;2:137. doi: 10.1177/1947603511401905. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Matsumoto T, Kubo S, Meszaros LB, Corsi KA, Cooper GM, Li G, Usas A, Osawa A, Fu FH, Huard J. The influence of sex on the chondrogenic potential of muscle-derived stem cells: implications for cartilage regeneration and repair. Arthritis Rheum. 2008;58:3809. doi: 10.1002/art.24125. [DOI] [PubMed] [Google Scholar]
- 59.Payne KA, Didiano DM, Chu CR. Donor sex and age influence the chondrogenic potential of human femoral bone marrow stem cells. Osteoarthritis Cartilage. 2010;18:705. doi: 10.1016/j.joca.2010.01.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Chiang H, Liao CJ, Wang YH, Huang HY, Chen CN, Hsieh CH, Huang YY, Jiang CC. Comparison of articular cartilage repair by autologous chondrocytes with and without in vitro cultivation. Tissue Eng Part C Methods. 2010;16:291. doi: 10.1089/ten.tec.2009.0298. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.