During development and regeneration, axonal growth depends on a rapid response to extracellular growth and guidance molecules. One mechanism underlying this rapid response is local protein synthesis (Jung et al., 2012). Local protein synthesis is a highly tuned, multi-step process by which mRNA binding proteins bind to specific sequences within the untranslated regions of particular mRNAs in the soma; mRNAs are then transported to axons and growth cones and translated at the appropriate spatial location in response to specific guidance cues. Many studies have now demonstrated that stimulus-induced local translation is critical to appropriate axon growth, guidance and regeneration. Multiple types of proteins are locally translated within axons, including those regulating the cytoskeleton, retrograde signaling, regeneration and cellular survival.
Neural cell adhesion molecules are transmembrane proteins that mediate a variety of processes, including cell recognition, axon growth and guidance, and regeneration. mRNAs encoding a number of cell adhesion molecules (CAMs), such as L1CAM (L1 cell adhesion molecule), NCAM (neural cell adhesion molecule), CHL1 (cell adhesion molecule L1 like) (Zivraj et al., 2010), DSCAM (Down syndrome cell adhesion molecule), ALCAM (activated leukocyte cell adhesion molecule), and PECAM (platelet/endothelial cell adhesion molecule) (Minis et al., 2014), are enriched in axons and growth cones from a variety of neuronal types. Therefore, it is logical that cell adhesion molecules are locally translated in axons in order to mediate stimulus-induced cellular signaling and axon growth. In fact, recent studies have demonstrated that three cell adhesion molecules, NF-protocadherin (NFPC), ALCAM and DSCAM, are locally translated in axons. As a result, it is becoming evident that the local translation of cell adhesion molecules is a critical mechanism regulating axon growth and guidance.
NFPC is a homophilic cell adhesion molecule and member of the cadherin superfamily. In addition, NFPC is known to regulate numerous processes including neural tube closure and axon growth of retinal ganglion cells (RGCs). NFPC mRNA and protein are found in the axons and growth cones of Xenopus RGCs as well as the optic tract (Piper et al., 2008), suggesting a role for NFPC in the formation of the retinotectal projection. During formation of this projection, the repulsive guidance cue Semaphorin 3A mediates a caudal turn in the mid-optic tract that is required for proper pathfinding of RGC axons to the tectum. Interestingly, knockdown of NFPC results in disruption of RGC axon growth and guidance at this same caudal turn in the mid-optic tract. Taken together, these findings suggest that there might be a connection between semaphorin 3A and NFPC signaling at this caudal turn to mediate appropriate pathfinding. A recent study elegantly linked these findings by demonstrating that this caudal turn is mediated through semaphorin 3A-stimulated local translation of NFPC in RGC axonal growth cones (Leung et al., 2013). Specifically, semaphorin 3A stimulation causes a rapid increase in NFPC protein in RGC axonal growth cones in vitro; this increase is blocked in the presence of a translational inhibitor. Using a fluorescent photoconvertible translation reporter, this study also demonstrated that there is a significant increase in translation of NFPC in RGC axonal growth cones at the caudal turn in vivo. This increase in NFPC translation is dependent on neuropilin-1, which is the receptor for semaphorin 3A. Taken together, this study demonstrates that local translation of NFPC in response to regionally expressed guidance cues, such as semaphorin 3A, is necessary for appropriate axon growth and guidance in the optic tract.
ALCAM is a cell adhesion molecule and member of the immunoglobulin superfamily that can interact either in a homophilic or heterophilic manner. ALCAM protein is found on growing axons of RGCs and is necessary for their appropriate growth and guidance (Avci et al., 2004). A recent study demonstrated that ALCAM mRNA is also present in RGC axons and axonal growth cones (Thelen et al., 2012), prompting an investigation into its potential local translation. This study demonstrated that ALCAM is locally translated within growth cones and this local translation is required for maintaining appropriate physiological levels of ALCAM in the axonal plasma membrane. Specifically, experimentally induced internalization of ALCAM combined with inhibition of translation results in decreased ALCAM levels. However, experimentally induced internalization of ALCAM but preserving ALCAM local translation allows for the maintenance of physiological levels of ALCAM in the axonal plasma membrane. This study went on to demonstrate that ERK and TOR kinase are required for the local translation of ALCAM. Furthermore, ERK kinase and the local translation of ALCAM are necessary for appropriate axon growth, which means that ALCAM local translation is functionally relevant. Finally, either decreased or increased levels of axonal ALCAM (via knockdown and overexpression, respectively) lead to a reduction in axonal growth on an ALCAM substrate, suggesting that a very specific level of ALCAM is required for axonal growth and guidance. Overall, this study demonstrates that local translation is a critical mechanism to maintain physiological levels of ALCAM in axonal growth cones and proper axon pathfinding.
DSCAM is a homophilic cell adhesion molecule, member of the immunoglobulin superfamily, and located in the trisomic region of human chromosome 21 that is involved in Down syndrome pathogenesis. DSCAM is also a netrin-1 receptor that is localized to axonal growth cones and plays an important role in axon growth and guidance (Zhu et al., 2011). We recently found that Dscam mRNA is localized to mouse hippocampal axons, prompting an investigation into Dscam mRNA localization and local translation in response to extracellular stimuli. We found that Dscam mRNA is localized and locally translated in response to netrin-1, an attractive guidance cue, in axonal growth cones (Jain and Welshhans, 2015). Specifically, netrin-1 regulates both Dscam mRNA and protein localization in growth cones in a dynamic, time dependent manner. Dscam mRNA localization in growth cones is slightly increased 10 minutes following netrin-1 stimulation and significantly decreased 20 minutes following netrin-1 stimulation. Furthermore, stimulation for 20 or 30 minutes with netrin-1 induces significantly increased DSCAM protein localization in hippocampal axonal growth cones. We investigated DSCAM protein localization following netrin-1 stimulation in the presence of anisomycin, a translational inhibitor, and nocodazole, which deploymerizes microtubules and thus prevents axonal transport of DSCAM. Anisomycin significantly blocked the netrin-1 induced DSCAM increased localization in axonal growth cones, whereas nocodazole treatment did not abolish the netrin-1 induced increase in DSCAM localization. Taken together these experiments confirm that Dscam mRNA is locally translated in axonal growth cones in response to netrin-1. Hence, we propose a model that netrin-1 stimulation may initially increase Dscam mRNA in growth cones and cause a burst of translation, which in turn activates a feedback loop leading to Dscam mRNA degradation after sufficient protein has been translated. Interestingly, after further investigation of the mechanism underlying netrin-1 induced local translation of DSCAM, we found that deleted in colorectal cancer (DCC) is required for netrin-1 induced DSCAM local translation in axons. This finding is significant because DCC is one of the major receptors for netrin-1 and known to associate with translational machinery to regulate netrin-1 induced translation (Tcherkezian et al., 2010). Finally, we examined the potential functional relevance of increased DSCAM protein, and found that overexpression of DSCAM results in severely stunted axon length. Taken together, these data suggest that netrin-1 induces local translation of Dscam in axonal growth cones via DCC, and that maintaining appropriate levels of this cell adhesion molecule is critical for proper axon growth (Jain and Welshhans, 2015).
Local translation of cell adhesion molecules in axons is emerging as a critical mechanism to regulate diverse processes (Figure 1). Semaphorin 3A stimulation induces local translation of NFPC in RGCs to mediate the caudal turn in the mid-optic tract (Leung et al., 2013). Homophilic interactions of ALCAM in RGCs induce local translation of ALCAM through ERK signaling, and this process is necessary for appropriate axon growth and fasciculation (Thelen et al., 2012). Netrin-1 stimulation induces local translation of DSCAM in hippocampal axons to mediate axon growth (Jain and Welshhans, 2015). Local translation is one of the mechanisms by which physiological levels of cell adhesion molecules is maintained, and thereby mediates appropriate axon growth and guidance in the nervous system. Taken together, these studies suggest that local translation of cell adhesion molecules in axons provides a dynamic spatiotemporal mechanism to regulate pathfinding at intermediate targets.
Figure 1.
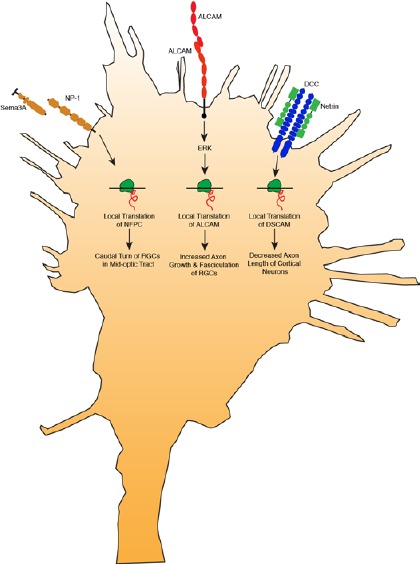
Model showing that local translation of cell adhesion molecules in axons is emerging as a critical mechanism to regulate diverse processes.
Semaphorin 3A binds to neuropilin-1 and induces local translation of NF-protocadherin (NFPC) in retinal ganglion cells (RGCs) to mediate the caudal turn in the mid-optic tract. Homophilic interactions of activated leukocyte cell adhesion molecule (ALCAM) in RGCs induce local translation of ALCAM through extracellular regulated protein kinases (ERK) signaling, and this process is necessary for appropriate axon growth and fasciculation. Netrin-1 binds to deleted in colorectal cancer (DCC) and induces local translation of Down syndrome cell adhesion molecule (DSCAM) to mediate axon growth of hippocampal neurons.
The studies described herein employed fluorescent and photoconvertible translation reporters to demonstrate local translation of CAMs. These reporter constructs consist of a fluorescent or photoconvertible protein and the 3’UTR of the mRNA of interest. Although this is a powerful method to visualize local translation in real time in living cells, it cannot demonstrate whether the locally translated proteins are then inserted into the plasma membrane. Thus, future studies will need to determine whether locally translated CAMs are properly trafficked and inserted in the axonal membrane. Furthermore, it is worth mentioning that the classical mechanisms and structures for the secretory pathway have not been identified in axons (Deglincerti and Jaffrey, 2012). There is immunostaining for some components of this pathway in axons, however tracking dyes have demonstrated that the labeling pattern of these structures is discontinuous and thus different from that which is seen in the soma (Merianda et al., 2009). However, there is evidence from two previous studies that proteins that are locally translated in vertebrate axons can be properly inserted into the axonal plasma membrane (Brittis et al., 2002; Merianda et al., 2013). Thus, it seems as though the classical pathway for protein synthesis and secretion does not exist in the axonal compartment, but there is a “functional equivalent” to secrete locally synthesize proteins (Merianda et al., 2009).
Cell adhesion molecules play a significant role in axonal growth, fasciculation and guidance. Specifically, cell adhesion molecules can activate intracellular signaling cascades and act as receptors for guidance cues, which then mediates selective fasciculation of growing axons and directs axon growth and guidance. Thus, cell adhesion molecules regulate axon physiology and behavior in both a spatial and temporal manner. A few recent studies, highlighted herein, have begun to establish local translation of cell adhesion molecules as an important mechanism to ensure appropriate axon growth and navigation. However, the mRNAs for some CAMs may be axonally localized, but not locally translated. For example, L1CAM mRNA has been localized to axonal growth cones using microarray (Zivraj et al., 2010), but a direct examination of L1CAM protein suggests that it is not locally translated in growth cones (Thelen et al., 2012). Then again, it is important to note that different species were employed in these studies. Furthermore, the temporal aspect of local translation must also be considered. It is likely that ALCAM is locally translated only before axons reach the optic chiasm because ALCAM protein is not found in axons after that point (Thelen et al., 2012). In addition, NFPC may be translated only during the caudal turn in the optic tract (Leung et al., 2013). Thus L1CAM may be locally translated, but in a different spatial or temporal manner than what has been tested thus far.
Future studies will determine whether local translation of cell adhesion molecules is a more widespread mechanism than previously realized to regulate axon growth during development and regeneration of the nervous system. It will also be of interest to determine which RNA binding proteins mediate the localization and local translation of the mRNAs described herein. Finally, future investigations will also shed light on how local translation maintains a critical physiological level of cell adhesion molecules to regulate the spatiotemporal dynamics of axon navigation during development and regeneration. We have only begun to elucidate the molecules and mechanisms underlying local translation of cell adhesion molecules in axons. Increasing our understanding of this field is critical and will allow us to gain knowledge that can be used to create treatments for neurodevelopmental disorders and promote regenerative axonal growth.
This research is supported by a Jérôme Lejeune Foundation award and a Kent State University Innovation Research Seed Award to KW.
References
- Avci HX, Zelina P, Thelen K, Pollerberg GE. Role of cell adhesion molecule DM-GRASP in growth and orientation of retinal ganglion cell axons. Dev Biol. 2004;271:291–305. doi: 10.1016/j.ydbio.2004.03.035. [DOI] [PubMed] [Google Scholar]
- Brittis PA, Lu Q, Flanagan JG. Axonal protein synthesis provides a mechanism for localized regulation at an intermediate target. Cell. 2002;110:223–235. doi: 10.1016/s0092-8674(02)00813-9. [DOI] [PubMed] [Google Scholar]
- Deglincerti A, Jaffrey SR. Insights into the roles of local translation from the axonal transcriptome. Open Biol. 2012;2:120079. doi: 10.1098/rsob.120079. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jain S, Welshhans K. Netrin-1 induces local translation of down syndrome cell adhesion molecule in axonal growth cones. Dev Neurobiol. 2015 doi: 10.1002/dneu.22360. doi:10.1002/dneu.22360. [DOI] [PubMed] [Google Scholar]
- Jung H, Yoon BC, Holt CE. Axonal mRNA localization and local protein synthesis in nervous system assembly, maintenance and repair. Nat Rev Neurosci. 2012;13:308–324. doi: 10.1038/nrn3210. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Leung LC, Urbančič V, Baudet M-L, Dwivedy A, Bayley TG, Lee AC, Harris WA, Holt CE. Coupling of NF-protocadherin signaling to axon guidance by cue-induced translation. Nat Neurosci. 2013;16:166–173. doi: 10.1038/nn.3290. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Merianda TT, Vuppalanchi D, Yoo S, Blesch A, Twiss JL. Axonal transport of neural membrane protein 35 mRNA increases axon growth. J Cell Sci. 2013;126:90–102. doi: 10.1242/jcs.107268. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Merianda TT, Lin AC, Lam JSY, Vuppalanchi D, Willis DE, Karin N, Holt CE, Twiss JL. A functional equivalent of endoplasmic reticulum and Golgi in axons for secretion of locally synthesized proteins. Mol Cell Neurosci. 2009;40:128–142. doi: 10.1016/j.mcn.2008.09.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Minis A, Dahary D, Manor O, Leshkowitz D, Pilpel Y, Yaron A. Subcellular transcriptomics-dissection of the mRNA composition in the axonal compartment of sensory neurons. Dev Neurobiol. 2014;74:365–381. doi: 10.1002/dneu.22140. [DOI] [PubMed] [Google Scholar]
- Piper M, Dwivedy A, Leung L, Bradley RS, Holt CE. NF-protocadherin and TAF1 regulate retinal axon initiation and elongation in vivo. J Neurosci. 2008;28:100–105. doi: 10.1523/JNEUROSCI.4490-07.2008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tcherkezian J, Brittis PA, Thomas F, Roux PP, Flanagan JG. Transmembrane receptor DCC associates with protein synthesis machinery and regulates translation. Cell. 2010;141:632–644. doi: 10.1016/j.cell.2010.04.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Thelen K, Maier B, Faber M, Albrecht C, Fischer P, Pollerberg GE. Translation of the cell adhesion molecule ALCAM in axonal growth cones - regulation and functional importance. J Cell Sci. 2012;125:1003–1014. doi: 10.1242/jcs.096149. [DOI] [PubMed] [Google Scholar]
- Zhu K, Xu Y, Liu J, Xu Q, Ye H. Down syndrome cell adhesion molecule and its functions in neural development. Neurosci Bull. 2011;27:45–52. doi: 10.1007/s12264-011-1045-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zivraj KH, Tung YCL, Piper M, Gumy L, Fawcett JW, Yeo GSH, Holt CE. Subcellular profiling reveals distinct and developmentally regulated repertoire of growth cone mRNAs. J Neurosci. 2010;30:15464–15478. doi: 10.1523/JNEUROSCI.1800-10.2010. [DOI] [PMC free article] [PubMed] [Google Scholar]


