Infrared light represents a broad spectrum of light with wavelengths from 700 nm to 1 million nm (1,000 microns). At its shortest wavelengths (referred to as near-infrared), it merges with the red spectrum of visible light. At the longest end (referred to as far-infrared), it blends into the range of microwaves. Near-infrared light has gained increased attention for its ability to activate anti-inflammatory processes and is now widely used in veterinary medicine to treat sprains, bone fractures, and to speed the healing of wounds. Over the last ten years, there has been an assemblage of animal studies focusing on the use of transcranial near-infrared light therapy (NILT) to treat brain injury from stroke or trauma.
While not fully understood, the mechanisms underlying the therapeutic benefits of NILT appear to depend upon the absorption of NIR photons in the wavelength range of 600–1,200 nm by cytochrome c oxidase in the mitochondria (Karu and Kolyakov, 2005; Chung et al., 2012; Henderson and Morries, 2015a). In addition to increasing adenosine triphosphate (ATP) production, NILT can modulate reactive oxygen species, activate mitochondrial DNA replication, increase early-response genes, increase growth factor expression, induce synaptogenesis, and stimulate cell proliferation (Vacca et al., 1993; Leung et al., 2002; Huang et al., 2009; Chen et al., 2011; Chung et al., 2012; Henderson and Morries, 2015a). Animal studies of low-level infrared light therapy (LLLT) have shown demonstrable neuroregeneration and repair (Oron et al., 2006; Lapchak, 2010; Xuan et al., 2014; Henderson and Morries, 2015a). In rodent models of stroke, light delivered with power densities of 0.9 – 36 J/cm2 applied at 24 hours after injury reduced neurological deficits by 32% and induced neuron proliferation and migration (Chung et al., 2012). In animal models of traumatic brain injury (TBI), the number of apoptotic neurons was reduced following LLLT using similar fluence (Oron et al., 2007; Xuan et al., 2014; Henderson and Morries, 2015a). In addition, decreased expression of pro-apoptotic genes, increased expression of anti-apoptotic genes, and increased expression of neurotrophic factors, such as nerve growth factor and brain-derived neurotrophic factor (BDNF), were induced by near-infrared light in a fluence range of 0.9 – 36 J/cm2 (Xuan et al., 2014; Henderson and Morries, 2015a; Morries et al., 2015) (see Figure 1).
Figure 1.
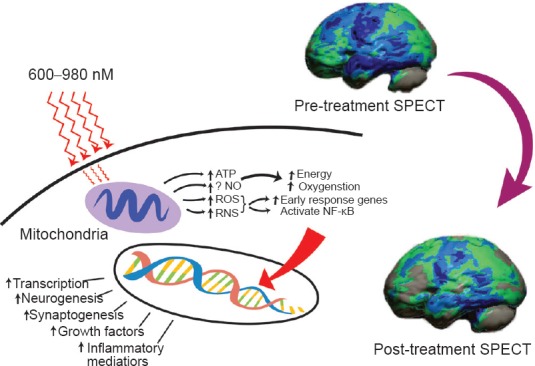
Hypothesized mechanism of action of near-infrared light therapy (NILT).
Near infrared light (600-980 nm) penetrates tissue and a fraction of the photonic energy reaches the mitochondria. The light of particular wavelengths is absorbed by cytochrome C oxidase and activates increased ATP production, increased production of reactive oxygen species (ROS), reactive nitrogen species (RNS), and possibly increases nitric oxide (Chung et al., 2012; Henderson and Morries, 2015). Downstream events include increased early response genes – c-fos, c-jun and activation of nuclear factor kappa B (NF-κB), which in turn induces increased transcription of gene products leading to synaptogenesis, neurogenesis, and increased production of inflammatory mediators and growth factors (Henderson and Morries, 2015a). Clinically, this results in decreased symptoms of headache, cognitive impairment, sleep disturbance, anxiety, and depression (Morries et al., 2015). Functional neuroimaging demonstrates increased cortical function. The case illustrated is of a patient with a 30 year old moderate traumatic brain injury (TBI). Shown is the contralateral hemisphere with marked diffuse hypoperfusion indicating widespread dysfunction. Following a course of NILT, the patient had demonstrable clinical improvement and significantly improved cerebral perfusion/function (Henderson and Morries, 2015b). SPECT scan data was processed and a map of statistically significant differences from a normative database was generated using the Oasis software by Segami, Inc. The color scale indicates gray for areas that do not differ significantly from the normative database. In contrast, areas of green, light blue, and dark blue represent areas of more than 2, 3, and 4 SD below the mean perfusion of the normative database, respectively. Statistically significant increases in perfusion are illustrated in the red color scale not seen in this patient's brain map.
An important distinction needs to be made between amount of energy delivered to the skin surface and amount of energy which reaches the target tissue. For example, 0.03 – 0.3 J/cm2 of energy delivered to a monolayer of cells in tissue culture was sufficient to activate transcription factors (Chen et al., 2011; Henderson and Morries, 2015a; Morries et al., 2015), while two orders of magnitude greater energy (30 J/cm2) was required to effect change in a rodent joint inflammation model (Castano et al., 2007; Morries et al, 2015). This distinction is because only a very small percentage of the fluence delivered to the skin surface can penetrate tissues. Indeed, Esnouf and colleagues reported that only 34% of an 850 nm continuous light source at 100 mW could penetrate 0.784 mm of human skin. Approximately 89% of 820 nm light will penetrate 0.4 mm of epidermis (Kolari, 1985) and about 13.5% traverses 2 mm of skin (Henderson and Morries, 2015a). Our laboratory research shows considerably less penetration by low level NIR. We found energy from a 50 mW 810 nm LED did not penetrate 2 mm of human or sheep skin (Henderson and Morries, 2015a). Similarly, no energy could be detected penetrating either human skin or sheep skin from a 200 mW LED (650 + 880 nm). Using higher energy devices, we found 9% of the energy from the 10 W combined 810/980 nm continuous wave infrared laser passed through 2 mm of skin (human or sheep).
The penetration of 810 nm energy through the entire depth of a mouse brain (3–5 mm) is reportedly 1–4% (Khuman et al., 2012). Hudson and colleagues (2013) measured 1 W 808/980 nm emitter photonic energy at varying depths of bovine muscle. They found the attenuation of 808 nm light at 3 cm was 99%. Similarly, we have found only 0.6% of 810 nm light delivered at 13.5W to the dorsum of the human hand could be detected penetrating the 25 mm of living tissue (Henderson and Morries, 2015a). Jagdeo and colleagues (2012) made similar observations - only 0.01–0.09% of 830 nm light from a 0.5 W emitter penetrated this distance of 25 mm.
Fitzgerald and colleagues (2013) modeled light penetration into the human brain using a 670 nm LED emitting 28 mW/cm2. Using a diffusion simulation computer model with assumptions of 10 mm of overlying scalp and skull, they derived that at the center of the brain (estimated at 56 mm), power density would be approximately 1.2 × 10–11 W/cm2 for 670 nm light. This would be insufficient fluence to exert a neuroregenerative effect. In our laboratory, we have modeled the human head by use of bisected, freshly slaughtered sheep heads (Henderson and Morries, 2015a). We found that NIR devices generating less than 1 W could not produce sufficient fluence at the surface to penetrate to 3 cm of depth. The energy from a 6 W LED system showed a 99.998% drop across 3 cm of tissue. In contrast, 0.14% of the energy from a 10 W 810/980 nm device penetrated 3 cm of tissue. At 15 W, an 810 nm emitter in continuous mode delivered 1.26% of the surface power density to the depth of 3 cm depth. Using pulsed NIR emission settings, a lower overall power density was delivered to the surface; however, similar penetration was achieved. Anders and colleagues have explored infrared light penetration in human cadaver heads and demonstrate measurable penetration of 808 nm light to the depth of 4 cm at 5 W (Tedford et al., 2015). The components features of the human head have also been studied. The penetration of 808–830 nm NIR light through dissected human skull ranges from 4.5% (Lapchak et al., 2015) to 11.7% (Jagdeo et al., 2012). When the combination of human scalp and skull were studied, penetration of an 810 nm NIR light at 1 W was reduced to 0.54% (Lychagov et al., 2006).
Human studies of NILT have been inconsistent, but promising. Three randomized, placebo-controlled clinical trials of low power NILT for the treatment of stroke yielded mixed results. The first trial demonstrated efficacy (Lampl et al., 2007). The subsequent clinical trials failed to show change in infarct size (Zivin et al., 2009; Kasner et al., 2013) or clinical efficacy (Hacke et al., 2014). Lapchak (2010) reported that the physical parameters of NILT in these clinical trials may have delivered insufficient fluence to cortical tissues to be effective. In these clinical trials, continuous wave NIR light of 808 nm wavelength with infrared energy densities of 0.9 J/cm2 was applied to the human scalp for a total of 40 minutes (Lapchak, 2010).
The limitations of low power NILT may include yielding only a transient effect. In a case report of two patients with TBI, a prolonged course of treatment was conducted with an 870 nm LED instrument at a fluence of 13.3 J/cm2, with 10 minutes per site over 4–60 months (Naesser et al., 2011). The patients experienced improvement in sustained attention, memory, and executive functions, but only transiently. If the patients stopped treatment for even a few days, they began to regress. A later case series of eleven patients treated with a similar LED device which delivered NIR wavelength 870 nm and 633 nm (red light) at a fluence of 13 J/cm2, for 20 minutes per session demonstrated improved attention, inhibition, and verbal memory based on psychological testing after 18 sessions (Naesser et al., 2014). It is not clear if the benefits in this case series were transient.
Our experience with multi-watt NIR therapy has been somewhat more encouraging. Using NIR lasers with a power range of 8–13 W, we have been treating TBI for the past five years. To date, we have treated over 25 patients and currently are preparing to begin a double-blind, sham-controlled clinical trial. We recently published data on our first eleven patients in a case series (Morries et al., 2015) and a case report (Henderson and Morries, 2015b). The case series of ten patients included civilians with TBI from motor vehicle accidents and veterans with TBI from impact and from blast injuries. Over 90% of the patients had complaints of irritability, insomnia, anxiety, and depression. Six of the ten patients had persistent headaches and 50% had cognitive difficulties, attention problems, and other signs of executive dysfunction. After a course of ten treatments of NILT (20 treatments in four patients), each patient experienced significant clinical improvement. Many of their symptoms resolved. Sixty percent had no residual symptoms after treatment. The remainder had mild or greatly reduced symptoms. Cognitive function appeared to improve since all disabled or impaired patients had returned to work, although cognitive tests were not performed. The quality of life dramatically improved in all cases. Based on the power densities of 14–28 J/cm2 delivered to the skin of patients in our clinic, our laboratory data indicate that at the depth of 3 cm into the human brain, we should be delivering 0.8–2.4 J/cm2. This is exactly within the range which has been shown to activate growth factors, other genes, and neuroregenerative processes within animal models.
These patients have been followed clinically since their treatment. They have maintained the clinical benefits that they initially experienced after a relatively short course of 10 or 20 treatments. Symptoms such as headache, insomnia, irritability, anxiety, depression, suicidal ideation, fatigue, and memory or concentration difficulties were resolved or greatly reduced and did not recur. Some patients are now four years since they completed treatment and they have continued to do well, according to follow-up interviews and reports from the referring physicians. Another important feature of these patients is that the majority had brain injuries which were 3 – 20 years old. Similarly, the case study described a patient with a 30-year old moderate TBI (see Figure 1). Nevertheless, there were significant changes in symptoms, function, and neuroimaging findings (Henderson and Morries, 2015b).
Treatment options for TBI have been painfully limited. While there have been some encouraging reports of improvement with the use of hyperbaric oxygen, published military studies show no benefit (Morries et al., 2015). Pharmacology has been limited to symptomatic amelioration (Morries et al., 2015). Recent multi-site studies of pharmacological agents to directly treat concussion and brain injury have failed (Willyard, 2015). The overlap in symptoms between TBI and post-traumatic stress disorder has contributed to the use of medications which have been shown to be potentially harmful to the injured brain (Raji et al., 2015) in those with a TBI. For example, anti-psychotics have been shown to be neurotoxic and can potentially impede any neurological regeneration which might otherwise occur (Phelps et al., 2014).
These data presented herein make an intriguing case for the potential of multi-watt NIR laser therapy as a safe and effective modality for the treatment of TBI and possibly other neurological insults. This multi-Watt modality provides sufficient energy delivery to the skin to yield biologically meaningful fluence at the site of brain injury (Henderson and Morries, 2015a, b; Morries et al., 2015; Tedford et al., 2015), which can be 1–7 cm from the scalp surface. Moreover, our data indicate that multi-Watt NIR laser therapy appears to induce a persistent change in neurological function (Morries et al., 2015), given patients have maintained their clinical improvements for years after treatment. Indeed, it is possible that multi-watt NILT induces a neuroregenerative change, based on the evidence of neurotrophin induction in animal models (e.g., Xuan et al., 2014) and the improvement of neurological function revealed with serial functional neuroimaging (Henderson and Morries, 2015b).
References
- Castano AP, Dai T, Yaroslavsky I, Cohen R, Apruzzese WA, Smotrich MH, Hamblin MR. Low-level laser therapy for zymosan-induced arthritis in rats: Importance of illumination time. Lasers Surg Med. 2007;39:543–550. doi: 10.1002/lsm.20516. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen AC, Arany PR, Huang YY, Tomkinson EM, Sharma SK, Kharkwal GB, Saleem T, Mooney D, Yull FE, Blackwell TS, Hamblin MR. Low-level laser therapy activates NF-kB via generation of reactive oxygen species in mouse embryonic fibroblasts. PLoS One. 2011;6:e22453. doi: 10.1371/journal.pone.0022453. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chung H, Dai T, Sharma SK, Huang YY, Carroll JD, Hamblin MR. The nuts and bolts of low-level laser (light) therapy. Ann Biomed Eng. 2012;40:516–533. doi: 10.1007/s10439-011-0454-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Esnouf A, Wright PA, Moore JC, Ahmed S. Depth of penetration of an 850nm wavelength low level laser in human skin. Acupunct Electrother Res. 2007;32:81–86. doi: 10.3727/036012907815844165. [DOI] [PubMed] [Google Scholar]
- Fitzgerald M, Hodgetts S, Van Den Heuvel C, Natoli R, Hart NS, Valter K, Harvey AR, Vink R, Provis J, Dunlop SA. Red/near-infrared irradiation therapy for treatment of central nervous system injuries and disorders. Rev Neurosci. 2013;24:205–226. doi: 10.1515/revneuro-2012-0086. [DOI] [PubMed] [Google Scholar]
- Hacke W, Schellinger PD, Albers GW, Bornstein NM, Dahlof BL, Fulton R, Kasner SE, Shuaib A, Richieri SP, Dilly SG, Zivin J, Lees KR. NEST 3 Committees and Investigators (2014) Transcranial laser therapy in acute stroke treatment: results of neurothera effectiveness and safety trial 3, a phase III clinical end point device trial. Stroke. 45:3187–3193. doi: 10.1161/STROKEAHA.114.005795. [DOI] [PubMed] [Google Scholar]
- Henderson TA, Morries LD. Near-infrared photonic energy penetration: can infrared phototherapy effectively reach the human brain? Neuropsychiatr Dis Treat. 2015a;11:2191–2208. doi: 10.2147/NDT.S78182. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Henderson TA, Morries LD. SPECT perfusion imaging demonstrates improvement of traumatic brain injury with transcranial near-infrared laser phototherapy. Adv Mind Body Med. 2015b;29:27–33. [PubMed] [Google Scholar]
- Huang YY, Chen AC, Carroll JD, Hamblin MR. Biphasic dose response in low level light therapy. Dose Response. 2009;7:358–383. doi: 10.2203/dose-response.09-027.Hamblin. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hudson DE, Hudson DO, Wininger JM, Richardson BD. Penetration of laser light at 808 and 980 nm in bovine tissue samples. Photomed Laser Surg. 2013;31:163–168. doi: 10.1089/pho.2012.3284. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jagdeo JR, Adams LE, Brody NI, Siegel DM. Transcranial red and near infrared light transmission in a cadaveric model. PLoS One. 2012;7:e47460. doi: 10.1371/journal.pone.0047460. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Karu TI, Kolyakov SF. Exact action spectra for cellular responses relevant to phototherapy. Photomed Laser Surg. 2005;23:355–361. doi: 10.1089/pho.2005.23.355. [DOI] [PubMed] [Google Scholar]
- Kasner SE, Rose DZ, Skokan A, Walker MG, Shi J, Streeter J. NEST-2 Steering Committee and Investigators (2013) Transcranial laser therapy and infarct volume. Stroke. 44:2025–2027. doi: 10.1161/STROKEAHA.113.000870. [DOI] [PubMed] [Google Scholar]
- Kolari PJ. Penetration of unfocused laser light into the skin. Arch Dermatol Res. 1985;277:342–344. doi: 10.1007/BF00509097. [DOI] [PubMed] [Google Scholar]
- Khuman J, Zhang J, Park J, Carroll JD, Donahue C, Whalen MJ. Low-level laser light therapy improves cognitive deficits and inhibits microglial activation after controlled cortical impact in mice. J Neurotrauma. 2012;29:408–417. doi: 10.1089/neu.2010.1745. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lampl Y, Zivin JA, Fisher M, Lew R, Welin L, Dahlof B, Borenstein P, Andersson B, Perez J, Caparo C, Ilic S, Oron U. Infrared laser therapy for ischemic stroke: a new treatment strategy: results of the NeuroThera Effectiveness and Safety Trial-1 (NEST-1) Stroke. 2007;38:1843–1849. doi: 10.1161/STROKEAHA.106.478230. [DOI] [PubMed] [Google Scholar]
- Lapchak PA. Taking a light approach to treating acute ischemic stroke patients: transcranial near-infrared laser therapy translational science. Ann Med. 2010;42:576–586. doi: 10.3109/07853890.2010.532811. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lapchak PA, Boitano PD, Butte PV, Fisher DJ, Hölscher T, Ley EJ, Nuño M, Voie AH, Rajput PS. Transcranial near-infrared laser transmission (NILT) profiles (800 nm): systematic comparison in four common research species. PLoS One. 2015;10:e0127580. doi: 10.1371/journal.pone.0127580. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Leung MC, Lo SC, Siu FK, So KF. Treatment of experimentally induced transient cerebral ischemia with low energy laser inhibits nitric oxide synthase activity and up-regulates the expression of transforming growth factor-beta 1. Lasers Surg Med. 2002;31:283–288. doi: 10.1002/lsm.10096. [DOI] [PubMed] [Google Scholar]
- Lychagov VL, Tuchin VV, Vilensky MA, Reznik BN, Ichim T, De Taboada L. Experimental study of NIR transmittance of the human skull. Proc SPIE 6085, Complex Dynamics and Fluctuations in Biomedical Photonics III, 60850T. 2006 [Google Scholar]
- Morries LD, Cassano P, Henderson TA. Treatments for traumatic brain injury with emphasis on transcranial near-infrared laser phototherapy. Neuropsychiatr Dis Treat. 2015;11:2159–2175. doi: 10.2147/NDT.S65809. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Naeser MA, Saltmarche A, Krengel MH, Hamblin MR, Knight JA. Improved cognitive function after transcranial, light-emitting diode treatments in chronic, traumatic brain injury: two case reports. Photomed Laser Surg. 2011;29:351–358. doi: 10.1089/pho.2010.2814. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Naeser MA, Zafonte R, Krengel MH, Martin PI, Frazier J, Hamblin MR, Knight JA, Meehan WP, 3rd, Baker EH. Significant improvements in cognitive performance post-transcranial, red/near-infrared light-emitting diode treatments in chronic, mild traumatic brain injury: open-protocol study. J Neurotrauma. 2014;31:1008–1017. doi: 10.1089/neu.2013.3244. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Oron A, Oron U, Chen J, Eilam A, Zhang C, Sadeh M, Lampl Y, Streeter J, DeTaboada L, Chopp M. Low-level laser therapy applied transcranially to rats after induction of stroke significantly reduces long-term neurological deficits. Stroke. 2006;37:2620–2624. doi: 10.1161/01.STR.0000242775.14642.b8. [DOI] [PubMed] [Google Scholar]
- Oron A, Oron U, Streeter J, de Taboada L, Alexandrovich A, Trembovler V, Shohami E. Low-level laser therapy applied transcranially to mice following traumatic brain injury significantly reduces long-term neurological deficits. J Neurotrauma. 2007;24:651–656. doi: 10.1089/neu.2006.0198. [DOI] [PubMed] [Google Scholar]
- Phelps TI, Bondi CO, Ahmed RH, Olugbade YT, Kline AE. Divergent long-term consequences of chronic treatment with haloperidol, risperidone, and bromocriptine on traumatic brain injury-induced cognitive deficits. J Neurotrauma. 2015;32:590–597. doi: 10.1089/neu.2014.3711. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Raji CA, Willeumier K, Taylor D, Tarzwell R, Newberg A, Henderson TA, Amen DG. Functional neuroimaging with default mode network regions distinguishes PTSD from TBI in a military veteran population. Brain Imaging Behav. 2015;9:527–534. doi: 10.1007/s11682-015-9385-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tedford CE, DeLapp S, Jacques S, Anders J. Quantitative analysis of transcranial and intraparenchymal light penetration in human cadaver brain tissue. Lasers Surg Med. 2015;47:312–322. doi: 10.1002/lsm.22343. [DOI] [PubMed] [Google Scholar]
- Vacca RA, Marra E, Quagliariello E, Greco M. Activation of mitochondrial DNA replication by He-Ne laser irradiation. Biochem Biophys Res Commun. 1993;195:704–709. doi: 10.1006/bbrc.1993.2102. [DOI] [PubMed] [Google Scholar]
- Willyard C. A pill for concussion remains a brain busting challenge. Stat website. 2015. [Accessed December 29, 2015]. http://www.statnews.com/2015/12/28/concussion-pill .
- Xuan W, Agrawal T, Huang L, Gupta GK, Hamblin MR. Low-level laser therapy for traumatic brain injury in mice increases brain derived neurotrophic factor (BDNF) and synaptogenesis. J Biophotonics. 2015;8:502–511. doi: 10.1002/jbio.201400069. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zivin JA, Albers GW, Bornstein N, Chippendale T, Dahlof B, Devlin T, Fisher M, Hacke W, Holt W, Ilic S, Kasner S, Lew R, Nash M, Perez J, Rymer M, Schellinger P, Schneider D, Schwab S, Veltkamp R, Walker M, Streeter J. NeuroThera Effectiveness and Safety Trial-2 Investigators (2009) Effectiveness and safety of transcranial laser therapy for acute ischemic stroke. Stroke. 40:1359–1364. doi: 10.1161/STROKEAHA.109.547547. [DOI] [PubMed] [Google Scholar]


