Keywords: nerve regeneration, brain injury, brain-derived neurotrophic factor, liposomes, targeting, vector, transfection, hippocampus, cortex, encapsulation efficiency, blood-brain barrier, transferrin, glial fibrillary acidic protein, polyethylene glycol, neural regeneration
Abstract
Brain-derived neurotrophic factor (BDNF) plays an important role in the repair of central nervous system injury, but cannot directly traverse the blood-brain barrier. Liposomes are a new type of non-viral vector, able to carry macromolecules across the blood-brain barrier and into the brain. Here, we investigate whether BDNF could be transported across the blood-brain barrier by tail-vein injection of liposomes conjugated to transferrin (Tf) and polyethylene glycol (PEG), and carrying BDNF modified with cytomegalovirus promoter (pCMV) or glial fibrillary acidic protein promoter (pGFAP) (Tf-pCMV-BDNF-PEG and Tf-pGFAP-BDNF-PEG, respectively). Both liposomes were able to traverse the blood-brain barrier, and BDNF was mainly expressed in the cerebral cortex. BDNF expression in the cerebral cortex was higher in the Tf-pGFAP-BDNF-PEG group than in the Tf-pCMV-BDNF-PEG group. This study demonstrates the successful construction of a non-virus targeted liposome, Tf-pGFAP-BDNF-PEG, which crosses the blood-brain barrier and is distributed in the cerebral cortex. Our work provides an experimental basis for BDNF-related targeted drug delivery in the brain.
Introduction
Brain-derived neurotrophic factor (BDNF) was the second member of the neurotrophin factor family to be discovered, after nerve growth factor (Binder, 2008; Zhang, 2012). It is widely expressed in the central nervous system, especially in the hippocampus and cortex (Bertrand, 2010; Qin, 2011). BDNF and its receptor TrkB play an important role in regulating various biological properties, including neuronal survival, differentiation, growth and plasticity (Ploughman et al., 2009; Zhang et al., 2009; Tuinstra, 2012; Zhang, 2012; Mayara, 2014). In the developing brain, endogenous BDNF levels are very low; therefore, in cases of brain injury (such as ischemia, degeneration and inflammation) during development, administration of exogenous BDNF is a promising treatment approach. However, as a macromolecular protein, exogenous BDNF cannot directly traverse the blood-brain barrier (BBB) or other biomembranes to exert its therapeutic function (Tapas, 2012; Vijender, 2015).
Despite the promise of viral vectors in drug delivery, their application is restricted by issues such as toxicity, and the search for a safe and effective treatment is ongoing. Liposomes are a new non-viral vector for the delivery of drugs to the brain, such as doxorubicin, which can be encased in the aqueous phase of the liposome (Schnyder et al., 2005; Xie et al., 2005; Afergan et al., 2008; Xu, 2011; Gao et al., 2013).
Transferrin (Tf) is a single glycoprotein synthesized in the liver, and is the main iron-containing protein in blood plasma. Upon combination with Fe2+, its molecular structure changes and it acquires binding affinity for the transferrin receptor (TfR) (Widera et al., 2003; Yang et al., 2009). TfR is widely expressed in capillary endothelial cells of the BBB, and targeted administration via the Tf-TfR pathway is a promising method of drug delivery.
In a preliminary experiment, we encased exogenous genes encoding Tf-BDNF in liposomes conjugated to polyethylene glycol (PEG), to increase the stability and targeting of the liposomes in vivo.
Glial fibrillary acidic protein (GFAP) is an intermediate filament protein, and transcriptional regulation of this protein has attracted considerable research attention. The GFAP promoter (pGFAP) can induce BDNF gene expression. pGFAP is used widely in the study of gene therapy (Girah et al. 2010; Yeo et al., 2013).
We previously obtained the gene sequence for BDNF by PCR amplification. After recovery, purification and macrorestriction, we conjugated this with the cytomegalovirus promoter (pCMV) and pGFAP carrier, to construct the expression vectors pCMV-BDNF and pGFAP-BDNF (Hou et al., 2013; Chen et al., 2014). We then in turn conjugated each vector with thiolated Tf and PEG-liposomes.
The aim of the present study was to determine whether the modified BDNF traversed the BBB after peripheral administration through the tail vein, and to investigate the expression pattern of the different liposomes in the brain at high and low doses. Our results provide an experimental basis for targeted gene therapy.
Materials and Methods
Animals
Sixty male Sprague-Dawley rats, aged 6–8 weeks and weighing 180 ± 30 g, were provided by the Public Health College of Jilin University, China (animal license No. SCXK (Ji) 2013-0003). Surgery was performed under anesthesia, and all efforts were made to minimize pain and distress in the experimental animals. All animal experiments were carried out in accordance with the United States National Institutes of Health Guide for the Care and Use of Laboratory Animals (NIH Publication No. 85-23, revised 1986).
Construction of targeting liposomes
In a preliminary experiment (Hou et al., 2013), we constructed two expression vectors: pCMV-BDNF and pGFAP-BDNF. We conjugated each of these with Tf and long-chain PEG to improve targeting and increase the peripheral circulation time. We performed non-isotope measurement of the liposome envelopment ratio, targeting ligand coupling ratio and molecular biological features to construct the required vectors: Tf-pGFAP-BDNF-PEG, Tf-pCMV-BDNF-PEG and the control, Tf-H2O-PEG.
Group assignment
Rats were acclimated in individual ventilated cages for three days, and assigned randomly to six groups (n = 10 rats per group), to receive low (2 μg) or high (10 μg) doses of Tf-pGFAP-BDNF-PEG, Tf-pCMV-BDNF-PEG, or Tf-H2O-PEG through the tail vein. The rats were sacrificed 48 hours later and the brains were removed, fixed in 4% paraformaldehyde, and stored in liquid nitrogen.
Immunofluorescence for BDNF protein expression in the brain
The fixed brain tissue was dehydrated in alcohol, cleared in xylene, embedded in paraffin and cut into sections 3 μm thick. Dewaxing and epitope retrieval was carried out. All samples were incubated in rabbit anti-rat BDNF polyclonal antibody (1:300; Beijing Biosynthesis Biotechnology Co., Ltd., Beijing, China) at 4°C overnight, rinsed three times with PBS for 5 minutes each time, incubated with Cy5-labeled goat anti-rabbit IgG at 37°C for 30 minutes in the dark, and rinsed as before. The sections were mounted with glycerin and observed under a fluorescence microscope (BX51; Olympus, Tokyo, Japan). Images were captured and the immunoreactive cells were counted in sections through the cortex, hippocampal CA1 and white matter. The number of positive cells was recorded in each specimen from five different visual fields, and the mean was calculated.
Reverse transcription-polymerase chain reaction (RT-PCR) for BDNF mRNA expression in the cerebral cortex
Thirty samples of brain tissue were stored in liquid nitrogen. The samples were then homogenized and repeatedly centrifuged to obtain the RNA. Using an ultraviolet spectrophotometer, the purity of the RNA was measured at 280 and 260 nm to ensure the ratio fell between 1.8 and 2.1. High-quality RNA had a ratio of 2.0. Primers were designed using BLAST (http://blast.ncbi.nlm.nih.gov/Blast.cgi) and synthesized by Sangon Biotech, Shanghai, China, and included a semi-quantitative primer (Table 1).
Table 1.
Primer information

The primers were used for one-step RT-PCR amplification of gene fragments (Qiagen, Frankfurt, Germany), which were then electrophoresed, and the intensity of the bands measured to estimate the expression level in each group.
Statistical analysis
Data were analyzed using SPSS 17.0 software (SPSS, Chicago, IL, USA) and followed a normal distribution. All values are presented as the mean ± SD. One-way analysis of variance and the least significant difference t-test were used to compare group means. P < 0.05 was considered statistically significant.
Results
Comparison of BDNF content in different brain regions
Forty-eight hours after injection of 10 μg of different liposomes through the tail vein, BDNF was observed in the Tf-pGFAP-BDNF-PEG and Tf-pCMV-BDNF-PEG groups, but not the Tf-H2O-PEG group, as expected. There was no difference in BDNF expression between the two hemispheres. However, the distribution of BDNF varied across the brain, with the greatest expression in the cortex. Less BDNF expression was detectable in the hippocampus or white matter (Figure 1).
Figure 1.
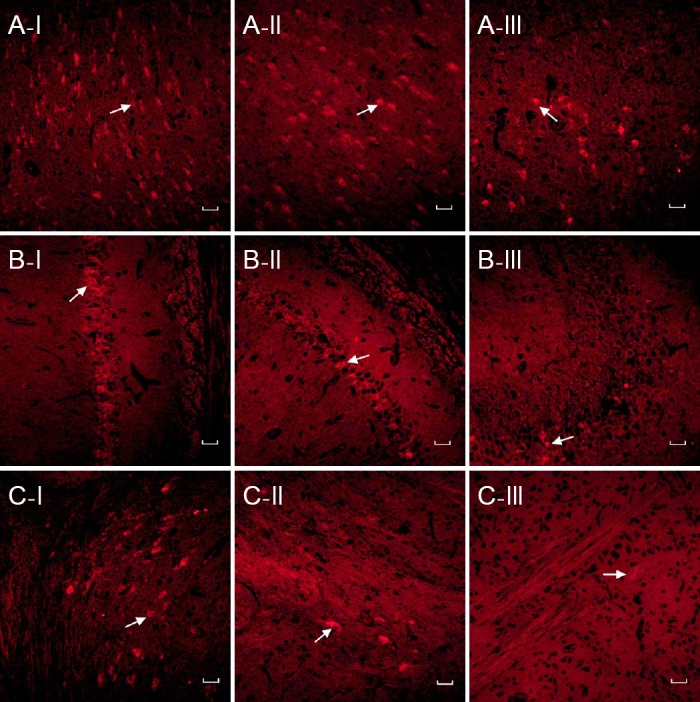
BDNF expression in different brain regions after high dose (10 μg) liposome injection (immunofluorescence staining, fluorescence microscope, × 200).
Red-stained cells (arrows) are BDNF-positive and labeled with Cy5. BDNF expression was observed in the cortex (A) after injection of Tf-pGFAP-BDNF-PEG (I) and Tf-pCMV-BDNF-PEG (II), but not Tf-H2O-PEG (III). Less BDNF expression was visible in the hippocampal CA1 (B) or white matter (C). Scale bars: 50 μm. BDNF: Brain-derived neurotrophic factor; Tf: transferrin; pGFAP: glial fibrillary acidic protein promoter; PEG: polyethylene glycol; pCMV: cytomegalovirus promoter.
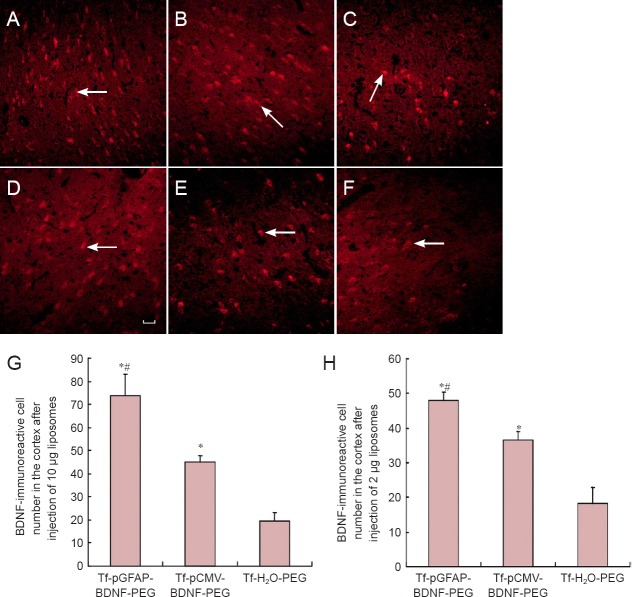
BDNF immunoreactivity in the cerebral cortex with different liposomes at high and low doses
Cortical BDNF immunoreactivity was significantly higher in the Tf-pGFAP-BDNF-PEG group than in the Tf-pCMV-BDNF-PEG (P < 0.05) or Tf-H2O-PEG (P < 0.05) groups, at both doses. High dose liposome administration resulted in significantly higher BDNF immunoreactivity than the low dose in the Tf-pGFAP-BDNF-PEG (73.6 vs. 44.8, P < 0.05) and Tf-pCMV-BDNF-PEG (48 vs. 36.4, P < 0.05) groups. No effect of dose was observed in the number of BDNF-immunoreactive cells after administration of Tf-H2O-PEG (P > 0.05; Figure 2).
Figure 2.
Comparison of BDNF-immunoreactive cells in the cerebral cortex after tail-vein injection of different liposomes at high and low doses (immunofluorescence staining).
(A–F) BDNF-immunoreactive cells (arrows) in the cortex at 10 μg (A–C) and 2 μg (D–F). (A, D) Tf-pGFAP-BDNF-PEG; (B, F) Tf-pCMVBDNF-PEG; (C, F) Tf-H2O-PEG. (G, H) Number of BDNF-immunoreactive cells in the cortex after injection of 10 μg (G) and 2 μg (H) liposomes. Red-stained cells (arrows) are BDNF-immunoreactive and labeled with Cy5. *P < 0.05, vs. Tf-H2O-PEG; #P < 0.05, vs. Tf-pCMV-BDNF-PEG. Data are presented as the mean ± SD (10 rats per group; one-way analysis of variance followed by the least significant difference t-test). Scale bar: 50 μm. BDNF: Brain-derived neurotrophic factor; Tf: transferrin; pGFAP: glial fibrillary acidic protein promoter; PEG: polyethylene glycol; pCMV: cytomegalovirus promoter.
Expression of BDNF mRNA in the cerebral cortex
BDNF mRNA expression was higher in the Tf-pGFAP-BDNF-PEG and Tf-pCMV-BDNF-PEG groups than in the Tf-H2O-PEG group at both doses, and higher in the high-dose groups than in the low-dose groups for each liposome (P < 0.05; Figure 3).
Figure 3.
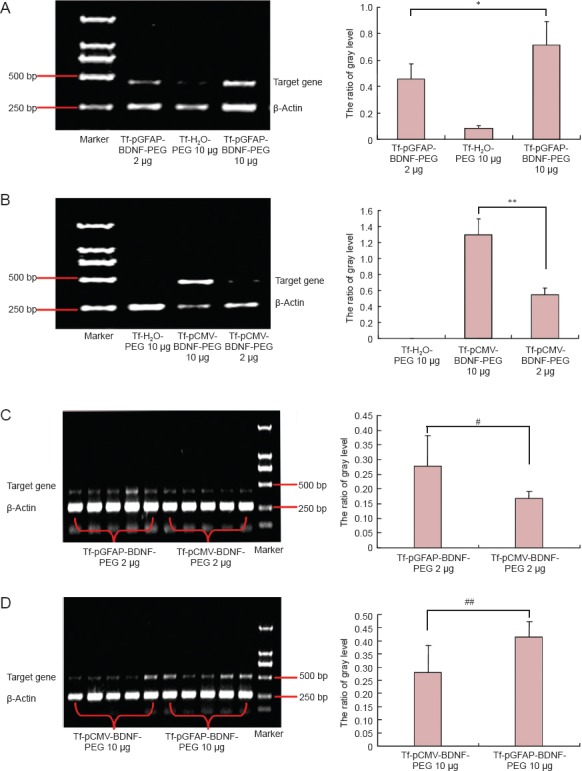
Expression of BDNF mRNA in cerebral cortex after tail-vein injection of different liposomes at high and low doses (reverse transcription-polymerase chain reaction).
(A, B) Tf-pGFAP-BDNF-PEG and Tf-pCMV-BDNF-PEG at different doses: BDNF mRNA expression was greater in the high-dose groups than in the low-dose groups (*P < 0.05, **P < 0.01). (C, D) Tf-pGFAP-BDNF-PEG and Tf-pCMV-BDNF-PEG groups at 2 μg (C) or 10 μg (D); BDNF mRNA expression was higher in the Tf-pGFAP-BDNF-PEG group than in the Tf-pCMV-BDNF-PEG group at both doses (#P < 0.05, ##P < 0.05). Data are presented as the mean ± SD (10 rats in each group; one-way analysis of variance followed by the least significant difference-t test). BDNF: Brain-derived neurotrophic factor; Tf: transferrin; pGFAP: glial fibrillary acidic protein promoter; PEG: polyethylene glycol; pCMV: cytomegalovirus promoter.
Discussion
In the present study, we used liposomes as vectors to wrap BDNF, and modified the liposomes with PEG, which can effectively increase peripheral circulation time. Tf is biodegradable, non-toxic, and shows good targeting properties (Daniels et al., 2006; Gregoriadis et al., 2008; Müller et al., 2008; Yeo et al., 2009). Gao et al. (2013) conjugated liposomes with both folate and transferrin to produce dual-targeting doxorubicin liposomes, which effectively crossed the BBB and targeted tumors. We therefore conjugated Tf with PEG liposomes and improved the targeting and stability of the liposomes by receptor-mediated methods in order to target drugs to the brain more effectively. Tf targeting PEG2000 lipid surface has a long circulation time and low absorption. The blood half-life of PEG-liposomes made by this method is extended to 90 hours (Sun et al., 2012). However, Tf receptors are abundant in the peripheral tissues where the encapsulated BDNF may be further destroyed. Therefore, when preparing the liposomes in the preliminary experiment, we also used the brain-specific promoter GFAP as a marker for specific expression of the targeted genes.
GFAP, an intermediate filament protein, is mainly expressed in glial cells for stability of the normal astrocyte cytoskeleton (Restrepo et al., 2011). Because of the specificity of GFAP for astrocytes and its involvement in the response to central nervous system damage, the regulation of GFAP gene transcription has attracted widespread attention (Lv et al., 2013; Yeo et al., 2013). pGFAP can successfully guide BDNF gene targeting, and is widely used in the study of gene therapy. Virta et al. (2006) demonstrated functional expression of the pGFAP gene in astrocytes. Chen et al. (2012) also confirmed that pGFAP was more effective than a general virus promoter in achieving therapeutic gene expression.
Here, we used immunofluorescence staining to show that injection of three different liposomes resulted in different numbers of BDNF-immunoreactive cells in the brain. The greatest BDNF expression occurred in the cerebral cortex, and there was no obvious expression in the hippocampus or white matter. Furthermore, the number of BDNF-immunoreactive cells in the cerebral cortex was greater in the Tf-pGFAP-BDNF-PEG group than in the Tf-pCMV-BDNF-PEG group. In addition, BDNF expression levels in the high-dose groups were higher than in the low-dose groups of the same vector, illustrating that increasing the dose of liposomes can enhance the brain delivery efficiency. Our results demonstrate that liposomes can target more effectively when combined with pGFAP.
In summary, we have used liposomes conjugated with PEG and Tf to carry BDNF across the BBB via a receptor-mediated pathway after intravenous injection to achieve expression of exogenous macromolecules in the brain. Furthermore, transfection efficiency in the brain was enhanced by increasing the dose of injected liposomes. The use of Tf targeting PEG liposomes and pGFAP can reduce distribution into peripheral tissues, prolong the action of the drug, achieve specific expression of the exogenous gene in the brain and improve the ability of drugs to cross the BBB, resulting in the targeted delivery of genes. Further research is necessary to determine the safety of gene therapy and to improve drug targeting, but liposome-mediated gene transfer is a promising avenue of research into the treatment of central nervous system diseases.
Acknowledgments
We thank Professor Xiang-fu Song from the Pathology Teaching and Research Section, College of Public Health, Jilin University, China for technical support.
Footnotes
Funding: This work was funded by a grant from Jilin Province Development and Reform Commission of China, No. JF2012C008-3, Jilin Province Industrial Innovation Special Fund Project of China, No. JF2016C050-2, and the Joint Project between Jilin University and Jilin You-bang Pharmaceutical Co. Ltd., No. 2015YX323.
Conflicts of interest: None declared.
Plagiarism check: This paper was screened twice using Cross-Check to verify originality before publication.
Peer review: This paper was double-blinded and stringently reviewed by international expert reviewers.
Copyedited by Slone-Murphy J, Wysong S, Wang J, Qiu Y, Li CH, Song LP, Zhao M
References
- Afergan E, Epstein H, Dahan R. Delivery of serotonin to the brain by monocytes following phagocytosis of liposomes. J Control Release. 2008;132:84–90. doi: 10.1016/j.jconrel.2008.08.017. [DOI] [PubMed] [Google Scholar]
- Bertrand N, Simard P, Leroux JC. Serum-stable long-circulating pH-sensitive PEGylated liposomes. Methods Mol Biol. 2010;605:545–558. doi: 10.1007/978-1-60327-360-2_36. [DOI] [PubMed] [Google Scholar]
- Binder DK, Scharfman HE. Brain-derived neurotrophic factor. Growth Factors. 2008;22:123–131. doi: 10.1080/08977190410001723308. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen JJ, Jin MX, Zhu SL, Li F, Xing Y. The synthesis and characteristic study of transferrin-conjugated liposomes carrying brain-derived neurotrophic factor. Biomed Mater Eng. 2014;24:2089–2099. doi: 10.3233/BME-141019. [DOI] [PubMed] [Google Scholar]
- Chen Y, Liu LH. Modern methods for delivery of drugs across the blood-brain barrier. Adv Drug Deliv Rev. 2012;64:640–665. doi: 10.1016/j.addr.2011.11.010. [DOI] [PubMed] [Google Scholar]
- Daniels TR, Delgado T, Rodriguez JA, Helguera G, Penichet ML. The transferrin receptor part II: targeted delivery of therapeutic agents into cancer cells. Clin Immunol. 2006;121:159–176. doi: 10.1016/j.clim.2006.06.006. [DOI] [PubMed] [Google Scholar]
- Faria MC, Gonçalves GS, Rocha NP, Moraes EN, Bicalho MA, Gualberto Cintra MT, Jardim de Paula J, José Ravic de Miranda LF, Clayton de Souza Ferreira A, Teixeira AL, Gomes KB, Carvalho Md, Sousa LP. Increased plasma levels of BDNF and inflammatory markers in Alzheimer's disease. J Psychiatr Res. 2014;53:166–172. doi: 10.1016/j.jpsychires.2014.01.019. [DOI] [PubMed] [Google Scholar]
- Gao JQ, Lv Q, Li LM, Tang XJ, Li FZ, Hu LY, Han M. Glioma targeting and blood-brain barrier penetration by dual-targeting doxorubincin liposomes. Biomaterials. 2013;34:5628–5639. doi: 10.1016/j.biomaterials.2013.03.097. [DOI] [PubMed] [Google Scholar]
- Girah A, Friedman HC, Caneda FB. BDNF regulation under GFAP promoter provides engineered astrocytes as a new approach for long-term protection in Huntington's disease. Gene Ther. 2010;17:1294–1308. doi: 10.1038/gt.2010.71. [DOI] [PubMed] [Google Scholar]
- Giralt A, Friedman HC, Caneda-Ferrón B, Urbán N, Moreno E, Rubio N, Blanco J, Peterson A, Canals JM, Alberch J. BDNF regulation under GFAP promoter provides engineered astrocytes as a new approach for long-term protection in Huntington's disease. Gene Ther. 2010;17:1294–1308. doi: 10.1038/gt.2010.71. [DOI] [PubMed] [Google Scholar]
- Gregoriadis G. Liposome research in drug delivery: the early days. J Drug Target. 2008;16:520–524. doi: 10.1080/10611860802228350. [DOI] [PubMed] [Google Scholar]
- Hou L, Xia L, ZX Xing Y. Cloning of rat BDNF construction of recombinant targeted expression vector. J Pure App Microbiol. 2013;7:783–787. [Google Scholar]
- Li Y, He H, Jia XR, Lu WL, Lou JN, Wei Y. A dual-targeting nanocarrier based on poly(amidoamine) dendrimers conjugated with transferrin and tamoxifen for treating brain gliomas. Biomaterials. 2012;33:3899–3908. doi: 10.1016/j.biomaterials.2012.02.004. [DOI] [PubMed] [Google Scholar]
- Lv Q, Li LM, Han M, Tang XJ, Yao JN, Ying XY, Li FZ, Gao JQ. Characteristics of sequential targeting of brain glioma for transferrin-modified cisplatin liposome. Int J Pharm. 2013;444:1–9. doi: 10.1016/j.ijpharm.2013.01.025. [DOI] [PubMed] [Google Scholar]
- Makar TK, Nimmagadda VK, Patibandla GK, Le T, Judge SI, Trisler D, Bever CT. Use of engineered bone marrow stem cells to deliver brain derived neurotrophic factor under the control of a tetracycline sensitive response element in experimental allergic encephalomyelitis. J Neuroimmunol. 2012;252:1–15. doi: 10.1016/j.jneuroim.2012.07.005. [DOI] [PubMed] [Google Scholar]
- Müller HD, Hanumanthiah KM, Diederich K, Diederich K, Schwab S, Schabitz WR. Brain-derived neurotrophic factor but not forced arm use improves long-term outcome after photothrombotic stroke and transiently upregulates binding densities of excitatory glutamate receptors in the rat brain. Stroke. 2008;39:12–21. doi: 10.1161/STROKEAHA.107.495069. [DOI] [PubMed] [Google Scholar]
- Ploughman M, Windle V, MacLellan CL. Brain-derived neurotrophic factor contributes to recovery of skilled reaching after focal ischemia in rats. Stroke. 2009;40:1490–1495. doi: 10.1161/STROKEAHA.108.531806. [DOI] [PubMed] [Google Scholar]
- Qin Y, Chen H, Zhang Q. Liposome formulated with TAT-modified cholesterol for improving brain delivery and therapeutic efficacy on brain glioma in animals. Int J Pharm. 2011;420:304–312. doi: 10.1016/j.ijpharm.2011.09.008. [DOI] [PubMed] [Google Scholar]
- Restrepo A, Smith CA, Agnihotri S. Epigenetic regulation of glial fibrillary acidic protein by DNA methylation in human malignant gliomas. Neuro Oncol. 2011;13:42–50. doi: 10.1093/neuonc/noq145. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schnyder A, Krähenbühl S, Drewe J. Targeting of daunomycin using biotinylated immunoliposomes: pharmacokinetics tissue distribution and in vitro pharmacological effects. J Drug Target. 2005;13:325–335. doi: 10.1080/10611860500206674. [DOI] [PubMed] [Google Scholar]
- Sun Y, Zhao Y. Enhanced pharmacokinetics and anti-tumor efficacy of PEGylated liposomal rhaponticin and plasma protein binding ability of rhaponticin. J Nanosci Nanotechnol. 2012;12:7677–7684. doi: 10.1166/jnn.2012.6599. [DOI] [PubMed] [Google Scholar]
- Tuinstra HM, Aviles MO, Shin S. Multifunctional multichannel bridges that deliver neurotrophin encoding lentivirus for regeneration following spinal cord injury. Biomaterials. 2012;33:1618–1626. doi: 10.1016/j.biomaterials.2011.11.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vijender S, Bhavuk G. Neuroplasticity and its relevance to psychiatric disorders. Cur Psychopharmacol. 2015;3:151–157. [Google Scholar]
- Virta S, Rapola J, Jalanko A. Use of nonviral promoters in adenovirus-mediated gene therapy: reduction of lysosomal storage in the aspartylglucosaminuria mouse. J Gene Med. 2006;8:699–706. doi: 10.1002/jgm.892. [DOI] [PubMed] [Google Scholar]
- Widera A, Norouziyan F, Shen WC. Mechanisms of TfR-mediated transcytosis and sorting in epithelial cells and applications toward drug delivery. Adv Drug Deliv Rev. 2003;55:39–66. doi: 10.1016/j.addr.2003.07.004. [DOI] [PubMed] [Google Scholar]
- Xie Y, Ye L, Zhang X, Cui W, Lou J, Nagai T, Hou X. Transport of nerve growth factor encapsulated into liposomes across the blood-brain barrier: in vitro and in vivo studies. J Control Release. 2005;105:106–119. doi: 10.1016/j.jconrel.2005.03.005. [DOI] [PubMed] [Google Scholar]
- Xu SL, Liu Y, Tai HC. Synthesis of transferrin (Tf) conjugated liposomes via Staudinger ligation. Int J Pharm. 2011;404:205–210. doi: 10.1016/j.ijpharm.2010.10.053. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yang X, Koh CG, Liu S. Transferrin receptor-targeted lipid nanoparticles for delivery of an antisense oligodeoxyribonucleotide against Bcl-2. Mol Pharm. 2009;6:221–230. doi: 10.1021/mp800149s. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yeo S, Bandyopadhyay S, Messing A. Stem cells modified by brain-derived neurotrophic factor to promote stem cells differentiation into neurons and enhance neurom- or function after brain injury. Chin J Traumatol. 2009;12:195–199. [PubMed] [Google Scholar]
- Yeo S, Bandyopadhyay S, Messing A, Brenner M. Transgenic analysis of GFAP promoter elements. Glia. 2013;61:1488–1499. doi: 10.1002/glia.22536. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang L, Xu TY, Wang S, Yu LQ, Liu DX, Zhan RZ, Yu SY. Curcumin produces antidepressant effects via activating MAPK/ERK-dependent brain-derived neurotrophic factor expression in the amygdala of mice. Behav Brain Res. 2012;235:67–72. doi: 10.1016/j.bbr.2012.07.019. [DOI] [PubMed] [Google Scholar]
- Zhang S, Liu XZ, Liu ZL. Stem cells modified by brain-derived neurotrophic factor to promote stem cells differentiation into neurons and enhance neurom- or function after brain injury. Chin J Traumatol. 2009;12:195–199. [PubMed] [Google Scholar]
- Zhang YG, Xiong KR. Effects of electroacupuncture combined with compound Salviae Miltiorrhizae tablet on the expressions of brain derived neurotrophic factor and vascular endothelial growth factor in hippocampus CA1 of chronic cerebral ischemia rats. Zhongguo Zhongxiyi Jiehe Zazhi. 2012;32:643–646. [PubMed] [Google Scholar]