Abstract
The G/C single-nucleotide polymorphism in the serotonin 1a receptor promoter, rs6295, has previously been linked with depression, suicide and antidepressant responsiveness. In vitro studies suggest that rs6295 may have functional effects on the expression of the serotonin 1a receptor gene (HTR1A) through altered binding of a number of transcription factors. To further explore the relationship between rs6295, mental illness and gene expression, we performed dual epidemiological and biological studies. First, we genotyped a cohort of 1412 individuals, randomly split into discovery and replication cohorts, to examine the relationship between rs6295 and five psychiatric outcomes: history of psychiatric hospitalization, history of suicide attempts, history of substance or alcohol abuse, current posttraumatic stress disorder (PTSD), current depression. We found that the rs6295G allele is associated with increased risk for substance abuse, psychiatric hospitalization and suicide attempts. Overall, exposure to either childhood or non-childhood trauma resulted in increased risk for all psychiatric outcomes, but we did not observe a significant interaction between rs6295 and trauma in modulating psychiatric outcomes. In conjunction, we also investigated the potential impact of rs6295 on HTR1A expression in postmortem human brain tissue using relative allelic expression assays. We found more mRNA produced from the C versus the G-allele of rs6295 in the prefrontal cortex (PFC), but not in the midbrain of nonpsychiatric control subjects. Further, in the fetal cortex, rs6295C allele exhibited increased relative expression as early as gestational week 18 in humans. Finally, we found that the C:G allelic expression ratio was significantly neutralized in the PFC of subjects with major depressive disorder (MDD) who committed suicide as compared with controls, indicating that normal patterns of transcription may be disrupted in MDD/suicide. These data provide a putative biological mechanism underlying the association between rs6295, trauma and mental illness. Moreover, our results suggest that rs6295 may affect transcription during both gestational development and adulthood in a region-specific manner, acting as a risk factor for psychiatric illness. These findings provide a critical framework for conceptualizing the effects of a common functional genetic variant, trauma exposure and their impact on mental health.
Introduction
Substantial evidence supports the role of serotonin 1a receptors (5-HT1A) in modulating anxiety and depression-related traits and impacting antidepressant efficacy.1 5-HT1A receptors are highly expressed in areas with strong involvement in emotional and affective processes, including the hippocampus, cingulate cortex, orbitofrontal cortex, insula, amygdala and midbrain raphe.2, 3 Positron emission tomography imaging studies have found altered 5-HT1A levels in major depressive disorder (MDD),4, 5, 6, 7, 8 bipolar disorder9 and anxiety disorders.10, 11, 12 Postmortem data also point toward altered 5-HT1A density in depression and suicide.13, 14, 15, 16, 17, 18 Clinically, the 5-HT1A partial agonist, buspirone, is used in the treatment of anxiety disorders19 and vilazodone, a partial 5-HT1A agonist and serotonin reuptake inhibitor, is used in the treatment of depression.20
Work in animal models has also highlighted the distinct behavioral contributions of 5-HT1A receptors across different brain regions and at different developmental time points.21 Pharmacological studies have indicated that postnatal but not adult administration of a 5-HT1A antagonist leads to increased anxiety later in life,22, 23 and genetic studies suggest that this effect is mediated by 5-HT1A autoreceptors localized on serotonergic neurons in the raphe.24, 25 In contrast, manipulations of 5-HT1A in the raphe selectively during adulthood has no effects on anxiety, but does impact stress-coping behaviors and antidepressant responsiveness.26 In the forebrain, whole-life suppression of 5-HT1A levels may affect depression-related behavior.27 Thus, the forebrain and midbrain 5-HT1A populations modulate distinct behaviors, and their contribution to behavior evolves across the course of development. Further, relatively small alterations in receptor levels are sufficient to impact behavior,24, 26 indicating that naturally occurring variation in receptor levels may contribute to individual differences in behavior. Given the multiple lines of evidence implicating variation in 5-HT1A levels in mental illness, there is a clear need to identify the factors that contribute to variation in 5-HT1A expression across the lifetime to guide individualized treatment and intervention.
Studies of the transcriptional regulation of the serotonin 1a receptor gene (HTR1A) have identified a putatively functional G/C single-nucleotide polymorphism (SNP) located 1019 bp upstream of the translation start site, designated rs6295.28 The G-allele of this SNP was initially found to be overrepresented in suicide,29 and was also associated with a decreased responsiveness to antidepressants.30 As with many gene associations, replications of these results have been inconsistent, but a series of recent meta-analyses supports an association between rs6295 and mood disorders, specifically MDD and bipolar disorder.31, 32, 33 In addition, this SNP has also been associated with a variety of other illnesses, including panic disorder,16 substance abuse,16 premenstrual dysphoric disorder,34, 35 eating disorders36 and schizophrenia16 (reviewed in ref. 28). These findings suggest that rs6295 may impact intermediate phenotypes that are common to multiple disorders.37
In vitro studies indicate that rs6295 may functionally impact HTR1A transcription through the altered binding of transcription factors.28 Specifically, the G-allele fails to bind the transcription factor, NUDR/Deaf1. This leads to higher levels of HTR1A transcription in raphe-derived neurons where NUDR/Deaf1 acts as a repressor, but lower levels in forebrain-derived neurons where it acts as a transcriptional activator.38, 39 However, this dual activity has never been demonstrated in human tissue. In addition, rs6295 impacts the binding of the factors, Hes1 and Hes5, which are expressed predominantly during neuronal differentiation.29, 38, 40 As such, the impact of this polymorphism on transcription during early development may differ from its effects during adulthood.
To clarify the relationship between rs6295, gene transcription and mental illness, we undertook dual lines of research. First, we investigated the hypotheses that the rs6295G-allele may be associated with an increased risk for psychiatric illness, and that it may interact with exposure to trauma in a previously characterized cohort studied for the effects of trauma on anxiety and depression-related traits.41, 42, 43 In separate studies, we also tested the hypothesis that rs6295 may impact transcription in the human brain, using relative allelic expression assays to examine rs6295-associated transcriptional differences across the different brain regions and in early development compared with adulthood. Finally, we investigated the hypothesis that HTR1A transcription may be altered in disease states. Together, these studies further indicate that naturally occurring common variation in HTR1A may contribute to transcriptional differences and risk for psychiatric illness.
Materials and methods
rs6295, trauma exposure and psychiatric outcomes
Participants
This study was part of a larger study undertaken by the Grady Trauma Project investigating the genetic and trauma-related risk factors for posttraumatic stress disorder (PTSD) and depression in an urban, highly traumatized, low-income, predominantly African American population in Atlanta, Georgia.41, 42, 43 The participants were recruited while waiting for outpatient appointments at the primary care or obstetrical-gynecological clinics of Grady Memorial Hospital. Both phenotype data and rs6295 genotype data were available for 1367 subjects. Of these, a subset of 1233 self-identified African American individuals were included in the final analyses because of clear genetic stratification that corresponded with self-reported ethnicity (Supplementary Methods and Supplementary Figure 1). Given the high rates of non-replication of gene association studies, these individuals were randomly split into a discovery cohort (n=822) and a replication cohort (n=411) when assessing the relationship between rs6295 and psychiatric outcomes (Supplementary Methods). These subjects provided informed consent, completed a battery of self-report measures and provided a salivary sample for DNA extraction. Owing to variation in literacy across subjects, all self-report measures were obtained by interview. The study was approved by the institutional review boards for Emory University School of Medicine and Grady Memorial Hospital. Subsequent analysis was performed on de-identified data. Sociodemographic information included sex, age, self-identified race/ethnicity, education, employment status, income, marital status and disability status (Table 1). The mean (s.d.) age in the sample was 40.11 (13.4, range 18–77). Details of trauma and psychiatric assessments, genetic methods and data analysis are provided in Supplementary Materials and Methods.
Table 1. Sample demographics for epidemiological cohort.
|
Participants |
||
|---|---|---|
| N | % | |
| Education | ||
| Less than 12th grade | 294 | 23.9 |
| High school or equivalent | 550 | 44.7 |
| More than high school | 385 | 31.2 |
| Employment | ||
| Currently employed | 348 | 28.3 |
| Unemployed | 881 | 71.7 |
| Current disability status | ||
| Current receiving support | 249 | 20.3 |
| Not receiving support | 975 | 79.7 |
| Household income (monthly) | ||
| $0–249 | 324 | 27.2 |
| $250–499 | 120 | 10.1 |
| $500–999 | 336 | 28.2 |
| $1000 or more | 412 | 25.6 |
rs6295 and transcription in postmortem human tissue
Samples
Adult tissue samples were obtained from the Postmortem Brain Core Facility at the University of Mississippi Medical Centre and from Columbia University/New York State Psychiatric Institute (Table 2). Samples from the University of Mississippi were genotyped as outlined above with Taqman genotyping assay for rs6295. Samples from Columbia University had been previously genotyped for rs6295.16 The procedures used for brain tissue collection, toxicology, psychological autopsy and stereology have been published elsewhere.14, 17, 44, 45, 46 Fetal tissue samples were obtained from the NICHD Brain and Tissue Bank for Developmental Disorders at the University of Maryland. Initially, we genotyped 27 fetal samples from peripheral tissue using the Wizard Genomic DNA Purification Kit (Promega #A1120, Madison, WI, USA) according to the manufacturer's instructions followed by genotyping as outlined above. This yielded five individuals with usable haplotypes for allelic expression assays (below), and cortical tissue was subsequently obtained for these five samples. An overview of sample characteristics is shown in Table 3.
Table 2. Distribution of genotypes.
|
rs6295 genotype |
|||
|---|---|---|---|
| GG | CG | CC | |
| Number (%) | Number (%) | Number (%) | |
| Male | 145 (30.2) | 243 (50.6) | 92 (19.2) |
| Female | 251 (33.3) | 363 (48.2) | 139 (18.5) |
| Total | 396 (32.1) | 606 (49.1) | 231 (18.7) |
Genotypes are in Hardy–Weinberg equilibrium for all the groups. P>0.05.
Table 3. Postmortem sample demographics.
| Control | MDD/suicide | |
|---|---|---|
| Age (year) | 52.7 (±18.7) | 54.9 (±17.6) |
| Gender | 13 F, 26 M | 8 F, 13 M |
| PMI (h) | 19.3 (±9.8) | 20.0 (±6.06) |
| Brain pH | 6.5 (±0.33) | 6.6 (±0.30) |
| RIN | 7.2 (±1.5) | 6.9 (±1.7) |
Abbreviations: MDD, major depressive disorder; PMI, postmortem interval; RIN, RNA integrity number.
Identification of SNPs for allelic expression assays
To identify an SNP in the HTR1A mRNA that could reasonably serve as a proxy for rs6295, which is located upstream of the transcription start sites,47 we cloned and sequenced a region encompassing rs6295, rs6294, rs878567 and rs6449693 from 50 rs6295GC heterozygous individuals (13 fetal, 37 adults). The latter three SNPs are located in the HTR1A mRNA. We used primers 5′-GAGTAAGGCTGGACTGTTAGATG-3′ and 5′-AGCAGATTCGTGCATAAGGATGG-3′ in failsafe PCR premix D (Epicentre Technologies, Madison, WI, USA) and Taq polymerase using the following PCR conditions: 95 °C for 5 min, (95 °C for 15 s, 55 °C for 15 s, 72 °C for 4 min) × 35° cycles, 72 °C for 5 min, 4 °C forever. The PCR products were analyzed by Sanger sequencing using the following primers: 5′-GAGTAAGGCTGGACTGTTAGATG-3′, 5′-CGTGGCCAATTATCTTATTGGC-3′ and 5′-AGCAGATTCGTGCATAAGGATGG-3′. The presence of overlapping peaks at the SNP locations was used to confirm heterozygosity. In samples heterozygous for rs6295 and one of the other SNPs, we cloned the subsequent PCR product into the PCR-TopoII vector according to the manufacturer's instructions (Invitrogen #K4610-20, Carlsbad, CA, USA). Three clones were initially picked for each sample and sent for sequencing. In a subset of cases, sequencing results that were inconsistent with those of the PCR product itself were observed, presumably due to mispaired annealing and extension of a partially completed PCR product. In those instances, an additional five clones were picked and sent for sequencing and the majority haplotype from all clones was used to assign haplotypes for an individual.
RNA extraction
RNA from frozen tissue punches or slide scrapings were collected directly into lysis buffer and pulverized by hand using a disposable pestle (Fisher #NC9719656). RNA was extracted using the Norgen RNA/DNA Purification Kit (Norgen Biotek #48700, Thorold, ON, Canada) according to the manufacturer's instructions with the exception of 600 μl rather than 300 μl of lysis buffer. RNA quality and concentration were assessed with an RNA nano chip using a 2100 Bioanalyzer (Agilent Technologies, Santa Clara, CA, USA).
Assessment of allelic imbalance
Because rs6295 was found to be in much stronger LD with rs878567 and rs6449693 than with rs6294 in our samples, we investigated allelic imbalance in individuals who were heterozygous for both rs6295 and rs878567 (see Results). We chose to use rs878567 instead of rs6449693 because the T-allele of this SNP creates a trinucleotide string that appears as a single peak in the pyrogram, making it difficult to calculate relative allelic expression. The complementary DNA (cDNA) was generated with 250 ng RNA template and random primers using the high-capacity reverse transcription kit (Applied Biosystems, Foster City, CA, USA) according to the manufacturer's instructions. 1 μl cDNA was used as template for subsequent pyrosequencing reactions, which were performed in triplicate. The region surrounding rs6295 was amplified using primers 5′- TGGCTCAGACTTTGCCTGTAT-3′ and biotin 5′-AACATCCACCGCAAAGATTTAG-3′ in a 25 μl reaction containing 200 nm primers, Taq polymerase (New England Biolabs, Ipswitch, MA, USA) and premix F (Epicentre Biotechnologies) using the following reaction conditions: 95 °C for 1 min; 35 cycles: 72 °C for 15 s, 53 °C for 15 s, 72 °C for 30 s; 72 °C for 7 min. Pyrosequencing was performed on a PyroMark Q24 pyrosequencer using sequencing primer 5′-CATCAGTTTTGATCCCAG-3′ with the following order of nucleotide addition: GTCATGCT. Genomic DNA from rs878567 and cDNA from rs878567TT and CC homozygotes were used to validate the pyrosequencing assay (representative pyrograms are shown in Supplementary Figure 3). Relative percent of T and C were quantified using PyroMark Q24 2.0.4 software (Qiagen, Hilden, Germany). Allelic imbalance was determined by a one-sample t-test relative to an expected value of 0.5, representing equal expression from the C- and T-allele. In addition, group differences were assessed using a two- tailed Student's t-test.
Results
rs6295, trauma exposure and psychiatric outcomes
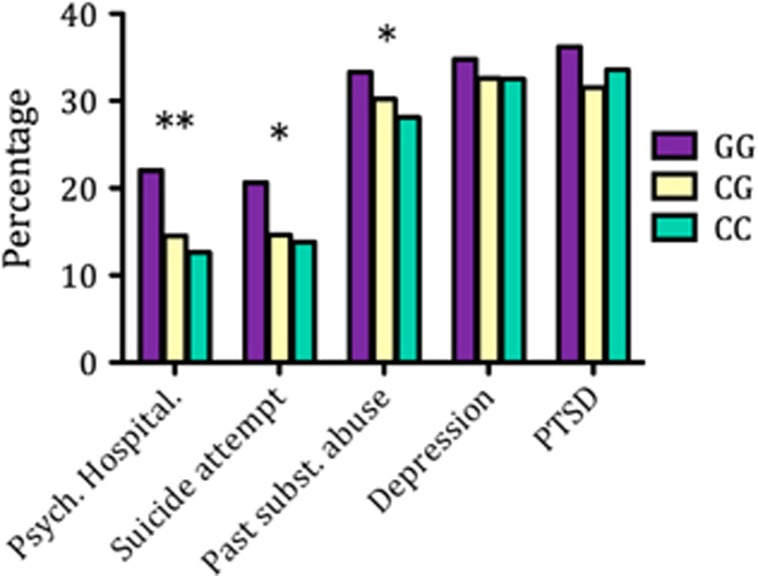
Our initial investigation in our discovery cohort revealed a significant association between rs6295GG genotype and psychiatric hospitalization, prior suicide attempts and previous substance abuse after controlling for sex, age, age2 and current disability status (Figure 1 and Table 4). In particular, rs6295GG individuals had an increased likelihood of past drug or alcohol abuse (odds ratio (OR)=1.506, P=0.02, confidence interval (CI)=1.068–2.122), prior hospitalization for psychiatric reasons (OR=1.571, P=0.025, CI=1.058–2.332) and increased likelihood of attempting suicide (OR=1.555, P=0.031, CI=1.040–2.323). We subsequently tested for these associations in our discovery cohort, where we confirmed a significant relationship between rs6295 and psychiatric hospitalization with a greater odds ratio than in our discovery cohort (OR=2.397, P=0.003, CI=1.355–4.240). In addition, although the reduced power in our replication sample failed to produce significance at the P<0.05 level, the odds ratios for the effect of rs6295GG and substance abuse (OR=1.126, P=0.645) and suicide attempts (OR=1.506, P=0.161) fell within the 95% CI of these associations in the discovery cohort, indicating that effect size did not differ between cohorts. The strongest and most consistent association existed between rs6295GG and psychiatric hospitalization, which may potentially reflect a pan-diagnostic impact on mental illness. We did not observe a significant association between rs6295 and current PTSD or depression.
Figure 1.
Prevalence of psychiatric outcomes for rs6295 genotypes. *P<0.05; GG versus C-allele carriers in discovery cohort. Psych., psychiatric; PTSD, posttraumatic stress disorder; subst., substance.
Table 4. Effects of rs6295 on psychiatric outcomes.
|
Psychiatric hospitalization |
Suicide attempt |
Past substance abuse |
Depression PTSD |
PTSD |
||||||
|---|---|---|---|---|---|---|---|---|---|---|
| OR | P | OR | P | OR | P | OR | P | OR | P | |
| Discovery sample | ||||||||||
| rs6295GG | 1.571 | 0.025 | 1.555 | 0.031 | 1.506 | 0.020 | 1.234 | 0.203 | 1.282 | 0.128 |
| Male sex | 0.693 | 0.075 | 0.307 | <0.001 | 1.813 | <0.001 | 0.771 | 0.116 | 0.913 | 0.576 |
| Age | 1.228 | <0.001 | 1.237 | <0.001 | 1.427 | <0.001 | 1.128 | 0.004 | 1.103 | 0.019 |
| Age2 | 0.998 | 0.001 | 0.998 | 0.001 | 0.996 | <0.001 | 0.998 | 0.004 | 0.999 | 0.017 |
| Disability | 2.39 | <0.001 | 2.297 | <0.001 | 1.113 | 0.589 | 1.087 | 0.685 | 1.287 | 0.206 |
| Test sample | ||||||||||
| rs6295GG | 2.397 | 0.003 | 1.506 | 0.161 | 1.126 | 0.645 | ||||
| Male sex | 0.864 | 0.623 | 0.542 | 0.046 | 1.609 | 0.048 | ||||
| Age | 1.428 | <0.001 | 1.348 | <0.001 | 1.487 | <0.001 | ||||
| Age2 | 0.996 | <0.001 | 0.996 | <0.001 | 0.996 | <0.001 | ||||
| Disability | 3.515 | <0.001 | 2.107 | 0.028 | 1.521 | 0.142 | ||||
Abbreviation: PTSD, posttraumatic stress disorder.
Odds ratios (OR) and P-values (P) for P<0.05 are shown in bold.
Given the association of rs6295 with more than one psychiatric outcome, we undertook pairwise comparisons of the entire sample to determine the extent to which we observed comorbidity in our sample. Chi-squared analyses showed that all phenotypes were significantly associated with each other (P<0.001). Odds ratio calculations (Table 5) show that the strongest associations exist between PTSD and depression and between psychiatric hospitalization and suicide attempts. This reflects the fact that suicide attempts are the most common reason for psychiatric hospitalization.
Table 5. Pairwise relationships between psychiatric outcomes.
| Psychiatric hospitalization | Suicide attempt | Past substance abuse | Depression | |
|---|---|---|---|---|
| Odds ratio | ||||
| Suicide attempt | 13.563 | |||
| Substance abuse | 4.222 | 3.995 | ||
| Depression | 2.642 | 3.546 | 1.852 | |
| PTSD | 2.798 | 2.797 | 2.057 | 8.696 |
Abbreviation: PTSD, posttraumatic stress disorder.
All pairwise comparisons are significant (P<0.001). Odds ratios shown above.
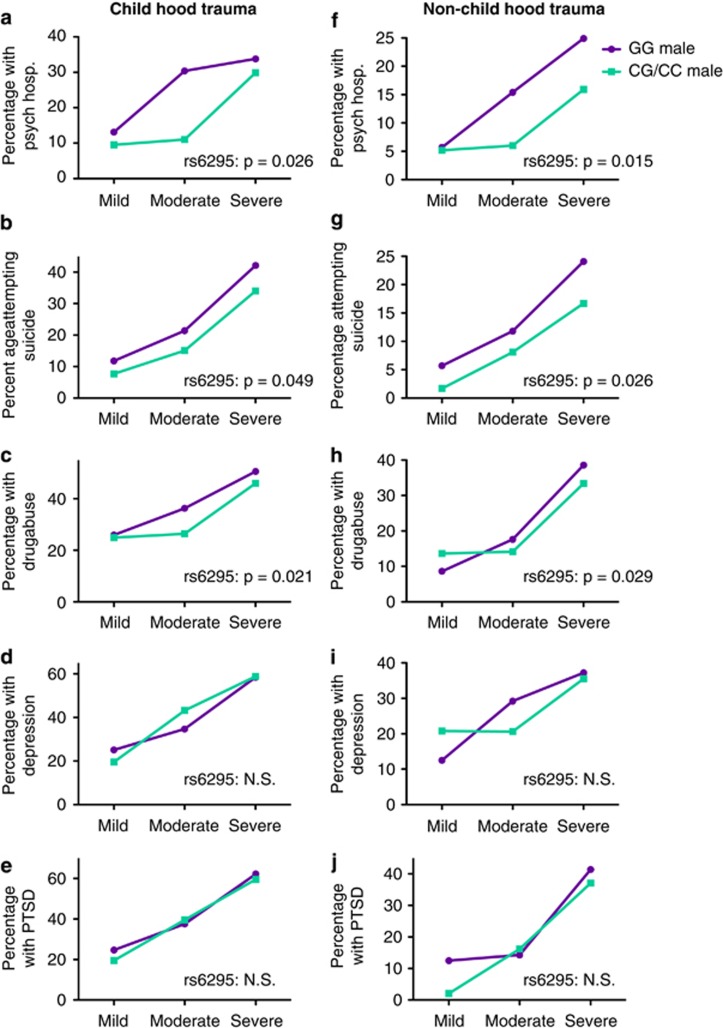
Having investigated the potential main effects of rs6295 on psychiatric outcomes, we were also interested in identifying potential interactions with childhood or non-childhood trauma that might moderate genetic effects. We assessed potential interactions between genotype, gender and exposure to trauma for our five outcome measures. Genotype, sex, age, age2 and current disability status were retained in all models with successive model selection for various interactions. No significant associations were observed, but both childhood and non-childhood trauma exposure were significantly associated with increased incidence in all five psychiatric outcomes that were assessed in this study (Table 6 and Table 7 and Figure 2).
Table 6. Logistic regression modeling of rs6295 and childhood trauma.
| Childhood trauma |
Psychiatric hospitalization |
Suicide attempt |
Past substance abuse |
Depression |
PTSD |
|||||
|---|---|---|---|---|---|---|---|---|---|---|
| OR | P | OR | P | OR | P | OR | P | OR | P | |
| Discovery sample | ||||||||||
| rs6295GG | 1.586 | 0.026 | 1.531 | 0.049 | 1.515 | 0.021 | 1.179 | 0.338 | 1.265 | 0.175 |
| Male sex | 0.722 | 0.123 | 0.34 | <0.001 | 2.091 | <0.001 | 0.855 | 0.364 | 1.073 | 0.684 |
| Age | 1.219 | 0.001 | 1.2 | 0.002 | 1.428 | <0.001 | 1.093 | 0.044 | 1.065 | 0.157 |
| Age2 | 0.998 | 0.003 | 0.998 | 0.007 | 0.996 | <0.001 | 0.999 | 0.053 | 0.999 | 0.168 |
| Disability | 2.32 | <0.001 | 2.186 | 0.001 | 1.127 | 0.565 | 0.999 | 0.998 | 1.207 | 0.378 |
| Moderate traumaa | 1.569 | 0.073 | 1.870 | 0.019 | 1.568 | 0.036 | 2.585 | <0.001 | 2.589 | <0.001 |
| Severe traumaa | 2.432 | <0.001 | 4.900 | <0.001 | 2.865 | <0.001 | 3.596 | <0.001 | 5.086 | <0.001 |
| Test sample | ||||||||||
| rs6295GG | 2.526 | 0.003 | 1.507 | 0.184 | 1.241 | 0.419 | ||||
| Male sex | 1.196 | 0.585 | 0.752 | 0.378 | 1.999 | 0.008 | ||||
| Age | 1.281 | 0.005 | 1.243 | 0.009 | 1.416 | <0.001 | ||||
| Age2 | 0.997 | 0.006 | 0.997 | 0.010 | 0.996 | <0.001 | ||||
| Disability | 3.185 | 0.001 | 1.893 | 0.074 | 1.507 | 0.167 | ||||
| Moderate traumaa | 2.019 | 0.071 | 1.879 | 0.086 | 1.126 | 0.698 | ||||
| Severe traumaa | 7.338 | <0.001 | 5.586 | <0.001 | 3.446 | <0.001 | ||||
Abbreviation: PTSD, posttraumatic stress disorder.
Relative to mild trauma.
Odds ratios (OR) and P-values (P) for P<0.05 are shown in bold.
Table 7. Logistic regression modeling of rs6295 and non-childhood trauma.
| Non-childhood trauma |
Psychiatric hospitalization |
Suicide attempt |
Past substance abuse |
Depression |
PTSD |
|||||
|---|---|---|---|---|---|---|---|---|---|---|
| OR | P | OR | P | OR | P | OR | P | OR | P | |
| Discovery sample | ||||||||||
| rs6295GG | 1.655 | 0.015 | 1.591 | 0.026 | 1.484 | 0.029 | 1.209 | 0.258 | 1.328 | 0.095 |
| Male sex | 0.615 | 0.022 | 0.300 | <0.001 | 1.703 | 0.002 | 0.742 | 0.078 | 0.851 | 0.336 |
| Age | 1.201 | 0.002 | 1.229 | 0.001 | 1.437 | <0.001 | 1.109 | 0.018 | 1.077 | 0.094 |
| Age2 | 0.998 | 0.005 | 0.998 | 0.001 | 0.996 | <0.001 | 0.999 | 0.015 | 0.999 | 0.072 |
| Disability | 2.454 | <0.001 | 2.178 | 0.001 | 1.136 | 0.534 | 0.998 | 0.993 | 1.178 | 0.429 |
| Moderate traumaa | 1.964 | 0.332 | 4.601 | 0.053 | 1.361 | 0.510 | 1.238 | 0.586 | 1.862 | <0.001 |
| Severe traumaa | 4.246 | 0.018 | 7.502 | 0.006 | 2.485 | 0.020 | 2.016 | 0.035 | 7.276 | <0.001 |
| Test sample | ||||||||||
| rs6295GG | 2.231 | 0.007 | 1.436 | 0.230 | 1.163 | 0.574 | ||||
| Male sex | 0.830 | 0.540 | 0.506 | 0.029 | 1.620 | 0.056 | ||||
| Age | 1.386 | <0.001 | 1.307 | 0.001 | 1.434 | <0.001 | ||||
| Age2 | 0.996 | <0.001 | 0.997 | 0.002 | 0.996 | <0.001 | ||||
| Disability | 3.163 | 0.001 | 1.946 | 0.064 | 1.396 | 0.274 | ||||
| Moderate traumaa | 1.578 | 0.613 | 1.055 | 0.966 | 1.177 | 0.862 | ||||
| Severe traumaa | 2.254 | 0.291 | 5.799 | 0.090 | 5.311 | 0.031 | ||||
Abbreviation: PTSD, posttraumatic stress disorder.
Relative to no trauma.
Odds ratios (OR) and P-values (P) for P<0.05 are shown in bold.
Figure 2.
Rates of psychiatric outcomes by genotype with increasing trauma severity. Childhood trauma (a–e) and non-childhood trauma (f–j) were binned into severity categories based on the number and types of trauma encountered. Because we identified a genotype × gender interaction for PTSD, males (solid lines) and females (dashed lines) are shown separately. Significant P-values from the discovery cohort for main effects of rs6295G-allele are indicated on the appropriate graph. NS, not significant; PTSD, posttraumatic stress disorder.
rs6295 and transcription
To identify SNPs in the HTR1A gene that existed in linkage disequilibrium with rs6295 in our sample, we cloned alleles from 50 rs6295GC individuals and determined haplotypes for rs6295, rs6294, rs6449693 and rs878567, the latter three of which exist in the HTR1A mRNA. Table 8 shows our observed haplotype frequencies.
Table 8. Distribution of haplotypes in rs6295GC samples.
| rs6295 | rs6294 | rs6449693 | rs878567 | Frequency |
|---|---|---|---|---|
| C | G | T | C | 50% |
| G | A | T | C | 9% |
| G | G | T | C | 3% |
| G | G | C | T | 38% |
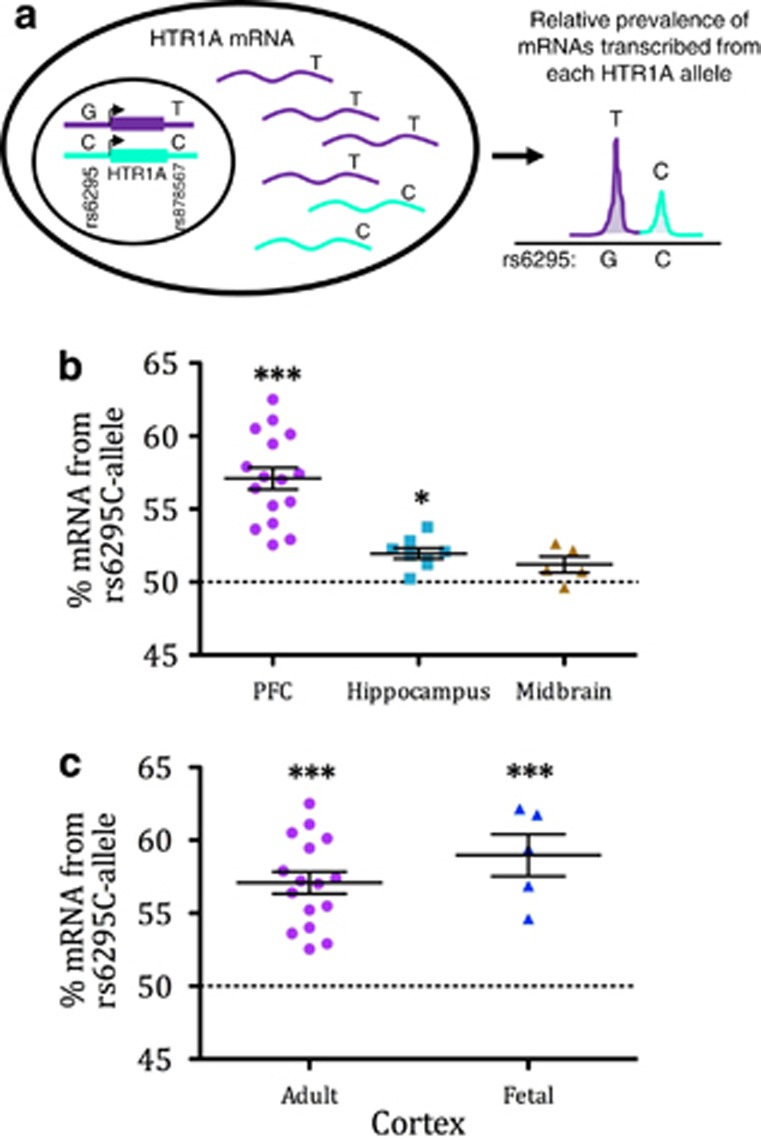
We observed strongest linkage disequilibrium between rs6295 and rs878567/rs6449693. Thus, we used rs878567 to determine the relative amount of mRNA produced from the G- versus C-allele of rs6295 in individuals heterozygous for both SNPs. Using this technique, we found more mRNA is produced from the C-allele as compared with the G-allele of rs6295 in the prefrontal cortex (PFC, Broadman areas 8 and 9) of control individuals (t (15)=6.092, P<0.001) and in the hippocampus (t (7)=5.194, P=0.003), although the effect size was much smaller in the latter (Figure 3). In addition, allelic transcription was not significantly biased in the midbrain (t (4)=2.881, P=0.135), indicating region-specific associations between allelic transcriptional differences and rs6295.
Figure 3.
Relative allelic expression from the rs6295C-allele. (a) Diagrammatic representation of relative allelic expression of human HTR1A gene. (b) More mRNA was produced from the C-allele than the G-allele of rs6295 in the adult prefrontal cortex (PFC), but this was not observed in the midbrain. (c) Biased transcription from the C-allele was also observed in the fetal cortex. ***P<0.001, *P<0.05.
Further, because different transcription factors are thought to interact with rs6295 during neuronal differentiation, as compared with adulthood, we also investigated relative allelic expression in cortex from gestational week 18–19 fetus. Hippocampus and midbrain were not available at these early time points. As in the adult PFC, we found that more mRNA was produced from the C-allele of rs6295 (t (4)=6.24, P=0.003).
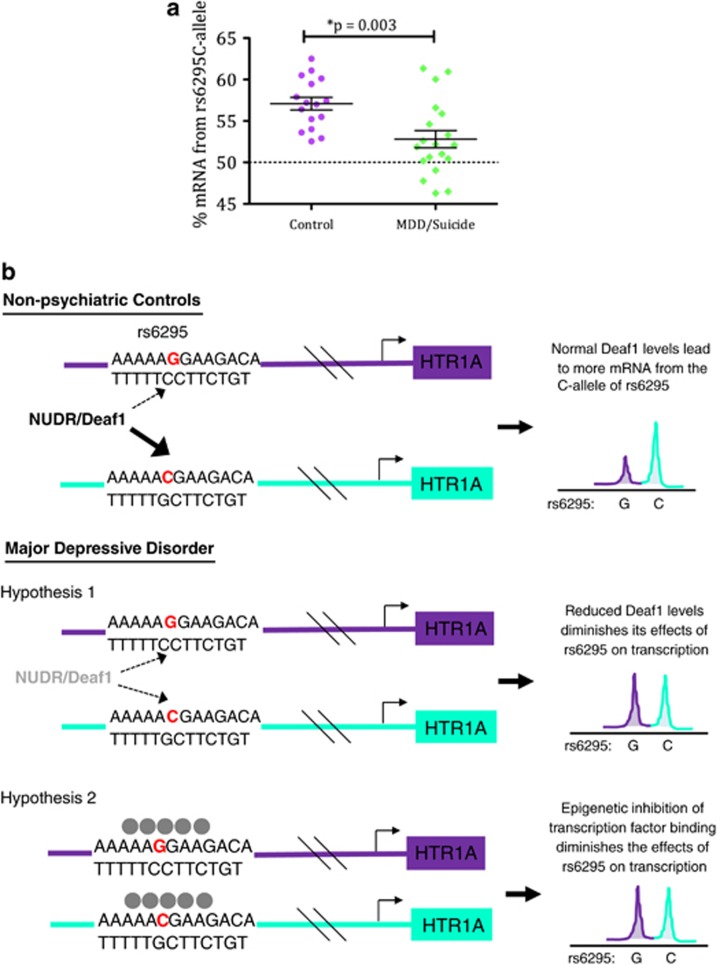
Finally, as differences in 5-HT1A levels have been observed in the individuals who died by suicide, we also assessed relative allelic expression in a subset of individuals who had MDD (with or without suicide; Figure 4). Because MDD with (n=14) and without (n=5) suicide did not differ (t (17)=−1.980, P=0.064), these were subsequently grouped together. Individuals with MDD were significantly different in their allelic transcription profile (% mRNA produced from C-allele) as compared with controls (t (33)=3.235, P=0.003).
Figure 4.
Relative allelic expression from the rs6295C-allele in the PFC of controls and MDD/suicide patients. (a) Relatively less HTR1A mRNA is produced from the C-allele of MDD patients compared with nonpsychiatric controls. (b) Two models that may explain the reduced penetrance of rs6295 in MDD. Previous work suggests that Deaf1 levels are impacted by stress, and decreased Deaf1 would be expected to preferentially decrease expression from the C-allele. Alternatively, epigenetic changes that inhibit access of transcription factors to the rs6295 locus would also neutralize the effects of this SNP. MDD, major depressive disorder; PFC, prefrontal cortex; SNP, single-nucleotide polymorphism.
Discussion
Our data provide biological support for the functional contribution of rs6295 to mental illness. In accordance with previous findings, we found that rs6295GG individuals had an increased incidence of substance and alcohol abuse, and an increased likelihood of both psychiatric hospitalization and attempting suicide. As evidence that rs6295 may directly impact these traits or their underlying intermediate phenotypes, we found that the G-allele was associated with lower levels of transcription in the PFC both during early gestation and in adulthood. These effects on transcription, which were more penetrant in nonpsychiatric controls than in patients with MDD, may thus serve as a risk factor for mental illness.
In addition to the main effects of rs6295 on psychiatric outcomes, we also explored the role of trauma. We found that exposure to either childhood or non-childhood trauma substantially increased the risk for all the psychiatric outcomes examined by us. However, we did not observe a significant interaction between rs6295 genotype and trauma exposure. It is possible that this may be owing to insufficient power, and it would be interesting to investigate potential interactions in a larger data set.
Together, these data confirm previous findings that the rs6295G allele is associated with mental illness. Furthermore, the strong association of rs6295GG genotype with increased rates of hospitalization may indicate that this genotype impacts intermediate phenotypes that span diagnostic categories and contribute to hospitalization, such as psychosis, impulsivity, aggression or disease severity. The 5-HT1A agonism is a feature of multiple pharmacotherapies, including aripiprizole, vilazadone and buspirone for schizophrenia, depression and anxiety disorders, respectively, suggesting complex and diverse behavioral effects resulting from the activation of this receptor.19, 20, 48, 49
We were initially surprised that we did not observe increased rates of depression in G-allele carriers, especially as GG individuals had higher likelihood of suicide attempts. However, both PTSD and depression symptoms were assessed only for 2 weeks before the data collection, while the other outcomes were retrospectively assessed across the lifespan. We do not have access to cumulative rates of PTSD or depression, and lifetime incidence of these disorders may show a stronger and/or clearer association with rs6295.
We also sought to establish a potential biological mechanism that could explain the association between rs6295 and mental illness. Because previous in vitro work indicated that rs6295G allele impairs the binding of a variety of transcription factors,38, 50 we examined the relationship between rs6295 and HTR1A transcription. There is often inherently high inter-individual variability in human samples that can result both from differences in life history and from differences in tissue handling, such as postmortem interval (time from death to freezing of brain tissue). As a control for this, we examined relative allelic expression in rs6295GC heterozygotes, a technique that compares the relative amount of mRNA produced from each allele in the same individual. This intra-individual comparison also has the advantage of assessing gene expression in the same transcription factor environment such that any differences in transcription are attributable to cis- regulatory sequence variants, such as rs6295.
Using this technique, our data indicate that rs6295, or a nearby linked variant, contributes to differences in HTR1A mRNA levels in vivo. Specifically, we found that more mRNA was produced from the C-allele of rs6295 as compared with the G-allele in the PFC of nonpsychiatric controls, but that this relative difference in expression did not occur in the raphe, although the sample sizes for hippocampus and raphe should be considered exploratory. These data support previous in vitro studies that found that NUDR/Deaf1 acts as a transcriptional activator in forebrain-derived cell lines.38 In this case, its failure to bind the G-allele would lead to decreased HTR1A mRNA from this allele. Although we did not observe an inversion of the C:G expression ratio in the raphe, this may be owing to insufficient spatial or cellular resolution, or it may also reflect our relatively limited sample size.
Our data further indicate that the relative increase in cortical expression from the C-allele is also present in early gestational development in our small sample set, indicating that rs6295 may impact HTR1A mRNA both during neuronal differentiation and later in life. HTR1A expression levels are high during this gestational period, and it has been hypothesized that 5-HT1A may be involved in the neurotrophic actions of serotonin.51 Because Hes1, Hes5, Hes6 and Deaf1/NUDR may all putatively have an impact on expression,38, 40 we used publicly available data sets to examine the relative levels of these genes during gestational week 18–19 (refs 52, 53; Supplementary Figure 4). At gestational week 17–18, Hes1 levels are falling, Hes5 levels are low, and Hes6 and Deaf1/NUDR levels are high. Thus, Hes1 and Deaf1 are most likely to impact HTR1A expression at this time. However, the extremely high levels of Hes 6 may counteract the repressive effects of Hes1.40 Thus, the increased expression from the C-allele at this time point may reflect a Deaf1/NUDR-dependent effect.
Finally, we also compared the C:G expression ratio in depressed suicide cases as compared with controls. Previous studies have identified an overall difference in 5-HT1A levels in depression.13, 15, 18, 54, 55 We focused on the PFC, as this is where we observed the largest effect for rs6295. Overall, we observed significantly less bias in the C:G relationship in MDD patients as compared with controls. Because the effects of rs6295 on transcription are stronger in nonpsychiatric controls, this SNP likely contributes to disease vulnerability rather than representing a disease state per se.
The blunted C:G transcriptional ratio in MDD patients suggests a change in transcription factor and/or epigenetic milieu at this locus in the disease state. Reduced Deaf1 protein has previously been observed in the PFC of depressed subjects and was correlated with reduced 5-HT1A protein levels.56 A reduction in Deaf1 would be expected to preferentially reduce the expression of the C-allele to neutralize allelic imbalance (Figure 4b). Alternatively, differences in epigenetic factors, such as methylation, at the locus could inhibit transcriptional access to the rs6295-containing region and thereby neutralize the effect of this polymorphism. Numerous studies have shown stress-induced differences in neural 5-HT1A levels,57, 58, 59, 60, 61, 62, 63 and a preliminary study in primary blood leukocytes found a correlation between HTR1A promoter DNA methylation levels and HTR1A mRNA levels,64 suggesting that epigenetic mechanisms may contribute to HTR1A transcriptional regulation. It is also plausible that altered Deaf1 regulation may interact with epigenetic changes in a complex, stress-dependent manner. For instance, chronic mild stress in male rats led to reduced PFC levels of 5-HT1A receptor as wellas of Deaf1,65 whereas more severe chronic restraint stress increased PFC HTR1A mRNA and 5-HT1A protein levels with no effect on Deaf1.66 Furthermore, different types of stress affected Deaf1 and 5-HT1A receptor expression differently and gender-specifically.65
There are a number of ways in which rs6295-associated alterations in 5-HT1A expression could potentially affect PFC neural function and behavior. 5-HT1A is an inhibitory receptor that is expressed both on pyramidal cells and on inhibitory interneurons in the PFC. It remains unclear whether one or both of these populations are sensitive to the putative effects of rs6295, and it has previously been suggested that the relative expression of 5-HT1A receptors on these two types of neurons may have important implications for overall effects of serotonin on limbic circuitry.67 In addition, there are two primary means by which the PFC has been implicated in serotonin-related behavior. First, the PFC is thought to have an important role in top-down control of limbic circuits.68 Alterations in 5-HT1A may impact such top-down control. In addition, the PFC provides important feedback that modulates the firing of the serotonergic raphe neurons.69 This feedback is sensitive to 5-HT1A activation. Thus, increased levels of 5-HT1A in the PFC may indirectly affect serotonergic modulation of other brain regions through this feedback mechanism. For instance, previous genetic imaging studies have found that rs6295 is associated with differences in amygdala activation, which the authors propose may be owing to differences in serotonin release in the amygdala.70, 71 Additional studies are needed to determine both the cell-type-specific effects of rs6295 in the PFC and clarify these potential circuit-level effects. Existing mouse models that enable both region-specific and temporally specific suppression of 5-HT1A levels, may be particularly well suited for this task.26, 27, 72
Our studies have focused on the effects of rs6295 on transcription with the assumption that this is the most direct method for investigating potential functional effects of this SNP. However, other factors beyond transcription likely also have a role in generating functional diversity within the 5-HT1A system. For instance, functional desensitization of these receptors, such as that induced by chronic antidepressant administration, may also have an important role in generating individual variation in the 5-HT1A system.73 Ultimately, a more thorough understanding of the diverse mechanisms impacting 5-HT1A levels and function will be important for understanding the contribution of this system to disease.
These findings provide an important advance in our understanding of a putatively functional promoter polymorphism in a psychiatrically relevant gene by linking genetic association data with an in vivo mechanism. The strongest association we observed was between rs6295 and psychiatric hospitalization. This may suggest that rs6295 impacts one or more intermediate phenotypes relevant to a variety of illnesses. In addition, we have provided important evidence that this SNP may have a functional role in disease risk by modulating transcription of HTR1A in a region-specific manner in humans. These studies provide an important framework for future studies of the complex mechanisms underlying HTR1A-mediated disease risk and add to the growing literature highlighting the complex mechanisms underlying individual sensitivity to stress and mental illness.
Acknowledgments
This work was supported by a grant from the Robert Wood Johnson Foundation, American Foundation for Suicide Prevention, T32MH015144 and K99MH102352 (ZRD); NIMH (R01 MH088542), NARSAD and HDRF (RH); CIHR (PRA): MH071537 (KJR.); P30 GM103328 (CAS); MH40210 (VA); P50 MH090964 (FAC). We deeply appreciate the invaluable contributions made by the families consenting to donate brain tissue and be interviewed. The support of the Cuyahoga County Medical Examiner's Office, Cleveland, OH, USA, is also acknowledged.
The authors declare no conflict of interest.
Footnotes
Supplementary Information accompanies the paper on the Translational Psychiatry website (http://www.nature.com/tp)
Supplementary Material
References
- Savitz J, Lucki I, Drevets WC. 5-HT1A receptor function in major depressive disorder. Prog Neurobiol 2009; 88: 17–31. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hornung J. Handbook of the Behavioral Neurobiology of Serotonin. Academic Press: London, UK, 2010. [Google Scholar]
- Kranz GS, Kasper S, Lanzenberger R. Reward and the serotonergic system. Neuroscience 2010; 166: 1023–1035. [DOI] [PubMed] [Google Scholar]
- Drevets WC, Thase ME, Moses-Kolko EL, Price J, Frank E, Kupfer DJ et al. Serotonin-1A receptor imaging in recurrent depression: replication and literature review. Nucl Med Biol 2007; 34: 865–877. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Drevets WC, Frank E, Price JC, Kupfer DJ, Holt D, Greer PJ et al. PET imaging of serotonin 1A receptor binding in depression. Biol Psychiatry 1999; 46: 1375–1387. [DOI] [PubMed] [Google Scholar]
- Hirvonen J, Karlsson H, Kajander J, Lepola A, Markkula J, Rasi-Hakala H et al. Decreased brain serotonin 5-HT1A receptor availability in medication-naive patients with major depressive disorder: an in-vivo imaging study using PET and [carbonyl-11C]WAY-100635. Int J Neuropsychopharmacol 2008; 11: 465–476. [DOI] [PubMed] [Google Scholar]
- Parsey RV, Oquendo MA, Ogden RT, Olvet DM, Simpson N, Huang YY et al. Altered serotonin 1A binding in major depression: a [carbonyl-C-11]WAY100635 positron emission tomography study. Biol Psychiatry 2006; 59: 106–113. [DOI] [PubMed] [Google Scholar]
- Parsey RV, Ogden RT, Miller JM, Tin A, Hesselgrave N, Goldstein E et al. Higher serotonin 1A binding in a second major depression cohort: modeling and reference region considerations. Biol Psychiatry 2010; 68: 170–178. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sullivan GM, Ogden RT, Oquendo MA, Kumar JS, Simpson N, Huang YY et al. Positron emission tomography quantification of serotonin-1A receptor binding in medication-free bipolar depression. Biol Psychiatry 2009; 66: 223–230. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lanzenberger RR, Mitterhauser M, Spindelegger C, Wadsak W, Klein N, Mien LK et al. Reduced serotonin-1A receptor binding in social anxiety disorder. Biol Psychiatry 2007; 61: 1081–1089. [DOI] [PubMed] [Google Scholar]
- Nash JR, Sargent PA, Rabiner EA, Hood SD, Argyropoulos SV, Potokar JP et al. Serotonin 5-HT1A receptor binding in people with panic disorder: positron emission tomography study. Br J Psychiatry 2008; 193: 229–234. [DOI] [PubMed] [Google Scholar]
- Neumeister A, Bain E, Nugent AC, Carson RE, Bonne O, Luckenbaugh DA et al. Reduced serotonin type 1A receptor binding in panic disorder. J Neurosci 2004; 24: 589–591. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Arango V, Underwood MD, Gubbi AV, Mann JJ. Localized alterations in pre- and postsynaptic serotonin binding sites in the ventrolateral prefrontal cortex of suicide victims. Brain Res 1995; 688: 121–133. [DOI] [PubMed] [Google Scholar]
- Boldrini M, Underwood MD, Mann JJ, Arango V. Serotonin-1A autoreceptor binding in the dorsal raphe nucleus of depressed suicides. J Psychiatr Res 2008; 42: 433–442. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hsiung SC, Adlersberg M, Arango V, Mann JJ, Tamir H, Liu KP. Attenuated 5-HT1A receptor signaling in brains of suicide victims: involvement of adenylyl cyclase, phosphatidylinositol 3-kinase, Akt and mitogen-activated protein kinase. J Neurochem 2003; 87: 182–194. [DOI] [PubMed] [Google Scholar]
- Huang YY, Battistuzzi C, Oquendo MA, Harkavy-Friedman J, Greenhill L, Zalsman G et al. Human 5-HT1A receptor C(-1019)G polymorphism and psychopathology. Int J Neuropsychopharmacol 2004; 7: 441–451. [DOI] [PubMed] [Google Scholar]
- Stockmeier CA, Shapiro LA, Dilley GE, Kolli TN, Friedman L, Rajkowska G. Increase in serotonin-1A autoreceptors in the midbrain of suicide victims with major depression-postmortem evidence for decreased serotonin activity. J Neurosci 1998; 18: 7394–7401. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Underwood MD, Kassir SA, Bakalian MJ, Galfalvy H, Mann JJ, Arango V. Neuron density and serotonin receptor binding in prefrontal cortex in suicide. Int J Neuropsychopharmacol 2012; 15: 435–447. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cohn JB, Rickels K. A pooled, double-blind comparison of the effects of buspirone, diazepam and placebo in women with chronic anxiety. Curr Med Res Opin 1989; 11: 304–320. [DOI] [PubMed] [Google Scholar]
- Wang SM, Han C, Lee SJ, Patkar AA, Masand PS, Pae CU. A review of current evidence for vilazodone in major depressive disorder. Int J Psychiatry Clin Pract 2013; 17: 160–169. [DOI] [PubMed] [Google Scholar]
- Donaldson ZR, Nautiyal KM, Ahmari SE, Hen R. Genetic approaches for understanding the role of serotonin receptors in mood and behavior. Curr Opin Neurobiol 2013; 23: 399–406. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vinkers CH, Oosting RS, van Bogaert MJ, Olivier B, Groenink L. Early-life blockade of 5-HT(1A) receptors alters adult anxiety behavior and benzodiazepine sensitivity. Biol Psychiatry 2010; 67: 309–316. [DOI] [PubMed] [Google Scholar]
- Lo Iacono L, Gross C. Alpha-Ca2+/calmodulin-dependent protein kinase II contributes to the developmental programming of anxiety in serotonin receptor 1A knock-out mice. J Neurosci 2008; 28: 6250–6257. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Donaldson ZR, Piel DA, Santos TL, Richardson-Jones J, Leonardo ED, Beck SG et al. Developmental effects of serotonin 1A autoreceptors on anxiety and social behavior. Neuropsychopharmacology 2013; 39: 291–302. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Garcia-Garcia AL, Meng Q, Richardson-Jones J, Dranovsky A, Leonardo ED. Disruption of 5-HT function in adolescence but not early adulthood leads to sustained increases of anxiety. Neuroscience; e-pub ahead of print 3 June 2015; doi: 10.1016/j.neuroscience.2015.05.076. [DOI] [PMC free article] [PubMed]
- Richardson-Jones JW, Craige CP, Guiard BP, Stephen A, Metzger KL, Kung HF et al. 5-HT1A autoreceptor levels determine vulnerability to stress and response to antidepressants. Neuron 2010; 65: 40–52. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Richardson-Jones JW, Craige CP, Nguyen TH, Kung HF, Gardier AM, Dranovsky A et al. Serotonin-1A autoreceptors are necessary and sufficient for the normal formation of circuits underlying innate anxiety. J Neurosci 2011; 31: 6008–6018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Albert PR. Transcriptional regulation of the 5-HT1A receptor: implications for mental illness. Philos Trans R Soc Lond B Biol Sci 2012; 367: 2402–2415. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lemonde S, Turecki G, Bakish D, Du L, Hrdina PD, Bown CD et al. Impaired repression at a 5-hydroxytryptamine 1A receptor gene polymorphism associated with major depression and suicide. J Neurosci 2003; 23: 8788–8799. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lemonde S, Du L, Bakish D, Hrdina P, Albert PR. Association of the C(-1019)G 5-HT1A functional promoter polymorphism with antidepressant response. Int J Neuropsychopharmacol 2004; 7: 501–506. [DOI] [PubMed] [Google Scholar]
- Kishi T, Yoshimura R, Fukuo Y, Okochi T, Matsunaga S, Umene-Nakano W et al. The serotonin 1A receptor gene confer susceptibility to mood disorders: results from an extended meta-analysis of patients with major depression and bipolar disorder. Eur Arch Psychiatry Clin Neurosci 2013; 263: 105–118. [DOI] [PubMed] [Google Scholar]
- Kishi T, Tsunoka T, Ikeda M, Kawashima K, Okochi T, Kitajima T et al. Serotonin 1A receptor gene and major depressive disorder: an association study and meta-analysis. J Hum Genet 2009; 54: 629–633. [DOI] [PubMed] [Google Scholar]
- Kishi T, Okochi T, Tsunoka T, Okumura T, Kitajima T, Kawashima K et al. Serotonin 1A receptor gene, schizophrenia and bipolar disorder: an association study and meta-analysis. Psychiatry Res 2011; 185: 20–26. [DOI] [PubMed] [Google Scholar]
- Yen JY, Tu HP, Chen CS, Yen CF, Long CY, Ko CH. The effect of serotonin 1A receptor polymorphism on the cognitive function of premenstrual dysphoric disorder. Eur Arch Psychiatry Clin Neurosci 2013; 264: 729–739. [DOI] [PubMed] [Google Scholar]
- Dhingra V, Magnay JL, O'Brien PM, Chapman G, Fryer AA, Ismail KM. Serotonin receptor 1A C(-1019)G polymorphism associated with premenstrual dysphoric disorder. Obstet Gynecol 2007; 110: 788–792. [DOI] [PubMed] [Google Scholar]
- Lim SW, Ha J, Shin DW, Woo HY, Kim KH. Associations between the serotonin-1A receptor C(-1019)G polymorphism and disordered eating symptoms in female adolescents. J Neural Transm (Vienna) 2010; 117: 773–779. [DOI] [PubMed] [Google Scholar]
- Smoller JW, Craddock N, Kendler K, Lee PH, Neale BM, Nurnberger JI et al. Identification of risk loci with shared effects on five major psychiatric disorders: a genome-wide analysis. Lancet 2013; 381: 1371–1379. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Czesak M, Lemonde S, Peterson EA, Rogaeva A, Albert PR. Cell-specific repressor or enhancer activities of Deaf-1 at a serotonin 1A receptor gene polymorphism. J Neurosci 2006; 26: 1864–1871. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Le Francois B, Czesak M, Steubl D, Albert PR. Transcriptional regulation at a HTR1A polymorphism associated with mental illness. Neuropharmacology 2008; 55: 977–985. [DOI] [PubMed] [Google Scholar]
- Jacobsen KX, Vanderluit JL, Slack RS, Albert PR. HES1 regulates 5-HT1A receptor gene transcription at a functional polymorphism: essential role in developmental expression. Mol Cell Neurosci 2008; 38: 349–358. [DOI] [PubMed] [Google Scholar]
- Ressler KJ, Mercer KB, Bradley B, Jovanovic T, Mahan A, Kerley K et al. Post-traumatic stress disorder is associated with PACAP and the PAC1 receptor. Nature 2011; 470: 492–497. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bradley RG, Binder EB, Epstein MP, Tang Y, Nair HP, Liu W et al. Influence of child abuse on adult depression: moderation by the corticotropin-releasing hormone receptor gene. Arch Gen Psychiatry 2008; 65: 190–200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mehta D, Klengel T, Conneely KN, Smith AK, Altmann A, Pace TW et al. Childhood maltreatment is associated with distinct genomic and epigenetic profiles in posttraumatic stress disorder. Proc Natl Acad Sci USA 2013; 110: 8302–8307. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Boldrini M, Underwood MD, Hen R, Rosoklija GB, Dwork AJ, John Mann J et al. Antidepressants increase neural progenitor cells in the human hippocampus. Neuropsychopharmacology 2009; 34: 2376–2389. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mann JJ, Huang YY, Underwood MD, Kassir SA, Oppenheim S, Kelly TM et al. A serotonin transporter gene promoter polymorphism (5-HTTLPR) and prefrontal cortical binding in major depression and suicide. Arch Gen Psychiatry 2000; 57: 729–738. [DOI] [PubMed] [Google Scholar]
- Stockmeier CA, Howley E, Shi X, Sobanska A, Clarke G, Friedman L et al. Antagonist but not agonist labeling of serotonin-1A receptors is decreased in major depressive disorder. J Psychiatr Res 2009; 43: 887–894. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Parks CL, Shenk T. The serotonin 1a receptor gene contains a TATA-less promoter that responds to MAZ and Sp1. J Biol Chem 1996; 271: 4417–4430. [DOI] [PubMed] [Google Scholar]
- Bantick RA, Deakin JF, Grasby PM. The 5-HT1A receptor in schizophrenia: a promising target for novel atypical neuroleptics? J Psychopharmacol 2001; 15: 37–46. [DOI] [PubMed] [Google Scholar]
- Pierz KA, Thase ME. A review of vilazodone, serotonin, and major depressive disorder. Prim Care Companion CNS Disord 2014; 16: PCC.13r01554. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lemonde S, Turecki G, Bakish D, Du L, Hrdina PD, Bown CD et al. Impaired repression at a 5-hydroxytryptamine 1A receptor gene polymorphism associated with major depression and suicide. J Neurosci 2003; 23: 8788–8799. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hillion J, Milne-Edwards JB, Catelon J, de Vitry F, Gros F, Hamon M. Prenatal developmental expression of rat brain 5-HT1A receptor gene followed by PCR. Biochem Biophys Res Commun 1993; 191: 991–997. [DOI] [PubMed] [Google Scholar]
- ABI. Brainspan: Atlas of the developing human brain. 2014. Allen Institute for Brain Sciencehttp://www.brainspan.org/static/atlas.
- Colantuoni C, Lipska BK, Ye T, Hyde TM, Tao R, Leek JT et al. Temporal dynamics and genetic control of transcription in the human prefrontal cortex. Nature 2011; 478: 519–523. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Parsey RV, Oquendo MA, Ogden RT, Olvet DM, Simpson N, Huang Y-y et al. Altered serotonin 1A binding in major depression: a [carbonyl-C-11]WAY100635 positron emission tomography study. Biol Psychiatry 2006; 59: 106–113. [DOI] [PubMed] [Google Scholar]
- Shrestha S, Hirvonen J, Hines CS, Henter ID, Svenningsson P, Pike VW et al. Serotonin-1A receptors in major depression quantified using PET: controversies, confounds, and recommendations. Neuroimage 2012; 59: 3243–3251. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Szewczyk B, Albert PR, Burns AM, Czesak M, Overholser JC, Jurjus GJ et al. Gender-specific decrease in NUDR and 5-HT1A receptor proteins in the prefrontal cortex of subjects with major depressive disorder. Int J Neuropsychopharmacol 2009; 12: 155–168. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Choi JY, Shin S, Lee M, Jeon TJ, Seo Y, Kim CH et al. Acute physical stress induces the alteration of the serotonin 1A receptor density in the hippocampus. Synapse 2014; 68: 363–368. [DOI] [PubMed] [Google Scholar]
- Franklin TB, Linder N, Russig H, Thony B, Mansuy IM. Influence of early stress on social abilities and serotonergic functions across generations in mice. PLoS One 2011; 6: e21842. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jovanovic H, Perski A, Berglund H, Savic I. Chronic stress is linked to 5-HT(1A) receptor changes and functional disintegration of the limbic networks. Neuroimage 2011; 55: 1178–1188. [DOI] [PubMed] [Google Scholar]
- Leventopoulos M, Russig H, Feldon J, Pryce CR, Opacka-Juffry J. Early deprivation leads to long-term reductions in motivation for reward and 5-HT1A binding and both effects are reversed by fluoxetine. Neuropharmacology 2009; 56: 692–701. [DOI] [PubMed] [Google Scholar]
- Lopez JF, Chalmers DT, Little KY, Watson SJ. A.E. Bennett Research Award. Regulation of serotonin1A, glucocorticoid, and mineralocorticoid receptor in rat and human hippocampus: implications for the neurobiology of depression. Biol Psychiatry 1998; 43: 547–573. [DOI] [PubMed] [Google Scholar]
- Spinelli S, Chefer S, Carson RE, Jagoda E, Lang L, Heilig M et al. Effects of early-life stress on serotonin(1A) receptors in juvenile Rhesus monkeys measured by positron emission tomography. Biol Psychiatry 2010; 67: 1146–1153. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang SH, Zhang ZJ, Guo YJ, Teng GJ, Chen BA. Decreased expression of serotonin 1A receptor in the dentate gyrus in association with chronic mild stress: a rat model of post-stroke depression. Psychiatry Res 2009; 170: 245–251. [DOI] [PubMed] [Google Scholar]
- Xu J, Zhang G, Cheng Y, Chen B, Dong Y, Li L et al. Hypomethylation of the HTR1A promoter region and high expression of HTR1A in the peripheral blood lymphocytes of patients with systemic lupus erythematosus. Lupus 2011; 20: 678–689. [DOI] [PubMed] [Google Scholar]
- Szewczyk B, Kotarska K, Daigle M, Misztak P, Sowa-Kucma M, Rafalo A et al. Stress-induced alterations in 5-HT1A receptor transcriptional modulators NUDR and Freud-1. Int J Neuropsychopharmacol 2014; 17: 1763–1775. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Iyo AH, Kieran N, Chandran A, Albert PR, Wicks I, Bissette G et al. Differential regulation of the serotonin 1 A transcriptional modulators five prime repressor element under dual repression-1 and nuclear-deformed epidermal autoregulatory factor by chronic stress. Neuroscience 2009; 163: 1119–1127. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rainnie DG. Serotonergic modulation of neurotransmission in the rat basolateral amygdala. J Neurophysiol 1999; 82: 69–85. [DOI] [PubMed] [Google Scholar]
- Meyer-Lindenberg A, Weinberger DR. Intermediate phenotypes and genetic mechanisms of psychiatric disorders. Nat Rev Neurosci 2006; 7: 818–827. [DOI] [PubMed] [Google Scholar]
- Celada P, Puig MV, Casanovas JM, Guillazo G, Artigas F. Control of dorsal raphe serotonergic neurons by the medial prefrontal cortex: involvement of serotonin-1A, GABA(A), and glutamate receptors. J Neurosci 2001; 21: 9917–9929. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fakra E, Hyde LW, Gorka A, Fisher PM, Munoz KE, Kimak M et al. Effects of HTR1A C(-1019)G on amygdala reactivity and trait anxiety. Arch Gen Psychiatry 2009; 66: 33–40. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sesack SR, Pickel VM. In the rat medial nucleus accumbens, hippocampal and catecholaminergic terminals converge on spiny neurons and are in apposition to each other. Brain Res 1990; 527: 266–279. [DOI] [PubMed] [Google Scholar]
- Donaldson ZR, Piel DA, Santos TL, Richardson-Jones J, Leonardo ED, Beck SG et al. Developmental effects of serotonin 1A autoreceptors on anxiety and social behavior. Neuropsychopharmacology 2014; 39: 291–302. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Blier P. Altered function of the serotonin 1A autoreceptor and the antidepressant response. Neuron 2010; 65: 1–2. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.