Abstract
Airway pressure release ventilation (APRV) was first described in 1987 and defined as continuous positive airway pressure (CPAP) with a brief release while allowing the patient to spontaneously breathe throughout the respiratory cycle. The current understanding of the optimal strategy to minimize ventilator-induced lung injury is to “open the lung and keep it open”. APRV should be ideal for this strategy with the prolonged CPAP duration recruiting the lung and the minimal release duration preventing lung collapse. However, APRV is inconsistently defined with significant variation in the settings used in experimental studies and in clinical practice. The goal of this review was to analyze the published literature and determine APRV efficacy as a lung-protective strategy. We reviewed all original articles in which the authors stated that APRV was used. The primary analysis was to correlate APRV settings with physiologic and clinical outcomes. Results showed that there was tremendous variation in settings that were all defined as APRV, particularly CPAP and release phase duration and the parameters used to guide these settings. Thus, it was impossible to assess efficacy of a single strategy since almost none of the APRV settings were identical. Therefore, we divided all APRV studies divided into two basic categories: (1) fixed-setting APRV (F-APRV) in which the release phase is set and left constant; and (2) personalized-APRV (P-APRV) in which the release phase is set based on changes in lung mechanics using the slope of the expiratory flow curve. Results showed that in no study was there a statistically significant worse outcome with APRV, regardless of the settings (F-ARPV or P-APRV). Multiple studies demonstrated that P-APRV stabilizes alveoli and reduces the incidence of acute respiratory distress syndrome (ARDS) in clinically relevant animal models and in trauma patients. In conclusion, over the 30 years since the mode’s inception there have been no strict criteria in defining a mechanical breath as being APRV. P-APRV has shown great promise as a highly lung-protective ventilation strategy.
Keywords: APRV, Ventilator-induced lung injury, ARDS, Lung protection
Review
Introduction
Stock and Downs first defined airway pressure release ventilation (APRV) as maintenance of a continuous positive airway pressure (CPAP) that allows for spontaneous breaths without significant airway pressure fluctuation and a brief cyclic release phase for efficient ventilation (i.e., CPAP with release) [1]. However, the flexibility of this definition has become an Achilles heel of sorts since a wide variety of APRV settings have all been used in the literature making comparison between studies impossible. Therefore, the current acronym “APRV” is a nebulous term identifying a ventilator mode without a precisely defined mechanical breath structure or ventilator settings. Variability in settings, and thus mechanical breath structure, significantly changes how the lung “sees” the breath being delivered. The first description of APRV published by Stock et al. in 1987 was, “APRV is a new way to administer simultaneously a supportive level of CPAP and assist CO2 elimination” [2]. Thus, the original definition of APRV was simply CPAP with a release to eliminate CO2. CPAP with a release is a very general description of a mechanical breath and can define all of the mechanical breaths seen in Fig. 1. Indeed, all of these mechanical breaths were defined as APRV [2–5]. As can be readily seen, the biggest difference between these APRV breaths is the duration at inspiration and expiration. Since the original definition of APRV was CPAP with a release, which did not specify the duration of the CPAP or release time, all of these breaths fit the original definition. Over the 30 years since APRV was originally described, many combinations of CPAP and release times have been used and all defined as APRV.
Fig. 1.
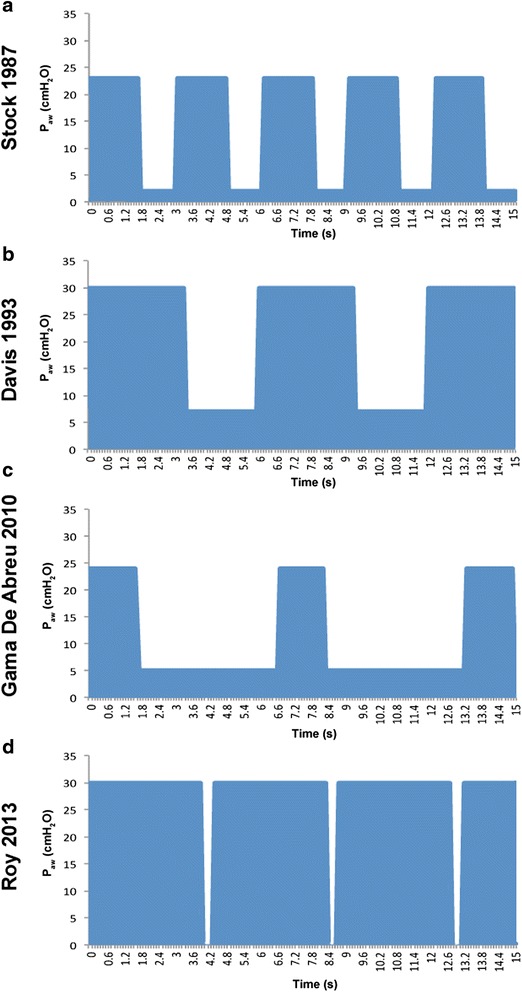
Comparison of APRV pressure waveforms. Artistic depiction of airway pressure waveforms, all of which were defined as APRV, illustrating the significant variability in what has been defined as an APRV breath. Stock in 1987 used 60 % CPAP with TLow of 1.27 s and a respiratory rate (RR) of 20 [2]. Davis in 1993 used a similar %CPAP, but decreased the RR by prolonging THigh and TLow [3]. Gama de Abreau in 2010 simulated conventional ventilation with a prolonged TLow and short THigh [4]. Finally, Roy in 2013 used a very brief adaptive TLow and large THigh with 90 % CPAP [5]. Of note, though the ventilator pressure is set at zero, this does not reflect true pressure as the brief TLow prevents full deflation of the lung, and thus prevents end-expiratory pressure from reaching zero. Figures a–c are examples of fixed-APRV (F-APRV) and figure d of personalized APRV (P-APRV)
In this review, we examine published original research in both animal and human trials in which the authors stated they utilized APRV, or a comparable but differently named ventilator mode. Our goals with this review were as follows: (1) to examine the historical evolution of APRV methodology; (2) to evaluate the efficacy of the APRV methodologies versus conventional positive pressure ventilation (CPPV); and (3) to determine if there are optimal APRV settings for maximal lung protection.
The evolution of APRV mostly revolves around modifying the CPAP and release time durations (time at expiration—TLow) (Tables 1, 2, 3, and 4, %CPAP and TLow). However, the most significant evolution in APRV has been the development of the ability to personalize the expiratory duration to precisely meet the needs of the patient’s changing lung physiology. The advantage of this method is that expiratory duration is set to maintain and open and stable the lung, regardless the level of lung pathology. Since there was no consistency in the CPAP and release time duration in the published literature, we chose to separate APRV strategies into two categories: fixed- (F-APRV) and personalized (P-APRV) APRV. If the expiratory duration or release time was not adjusted by mechanical changes in the lung, regardless of duration of CPAP or release time, it was categorized as F-APRV. If the APRV strategy sets expiratory duration by changes in lung mechanics, using the slope of the expiratory flow curve, it was categorized as P-APRV (Fig. 1).
Table 1.
Summary of animal studies utilizing F-APRV
| First author | Year | n | Animal | Study design | % CPAP | TLow | Findings |
|---|---|---|---|---|---|---|---|
| Stock [2] | 1987 | 10 | Mongrel dog | Crossover CPPV vs. APRV |
58 % | 1.27 s | APRV improved oxygenation with lower PIP and without cardiopulmonary compromise |
| Rasanen [21] | 1988 | 10 | Mongrel dog | Crossover CPPV vs. CPAP vs. APRV |
50 % | 1.5 s | CPPV impaired circulatory function and tissue oxygen balance, APRV had higher systemic vascular resistance and decreased pulmonary vascular resistance |
| Martin [17] | 1991 | 7 | Neonatal sheep | Crossover Spont vs. CPAP vs. CPPV vs. APRV |
50 % | 1 s | APRV augmented alveolar ventilation vs. CPAP, and had lower Paw than PPV without compromised cardiovascular function |
| Smith [23] | 1995 | 5 | Swine | Crossover CPAP vs. APRV |
80 % | 1.1 s exp flow 0 | APRV maintains oxygenation without hemodynamic compromise |
| Neumann [19] | 2001 | 9 | Swine | Crossover CPAP vs. APRV +/− PEEP |
67 % | 1 s | APRV decreased O2 compared with CPAP, No difference with PEEP |
| Hering [13] | 2003 | 12 | Swine | Crossover APRV +/− SB |
50 % | N/A | APRV + SB increased oxygenation and cardiovascular function |
| Wrigge [24] | 2003 | 24 | Swine | Randomized prospective APRV +/− SB |
50 % | 1.5–2 s | APRV + SB increased oxygenation and cardiovascular function |
| Neumann [20] | 2005 | 20 | Swine | Randomized prospective APRV +/− SB |
50 % | 1.5 s | APRV + B increased ventilation in dependent lung and decreased shunt |
| Hering [14] | 2005 | 12 | Swine | Crossover APRV vs. SB |
50 % | ~1.7 s | APRV + SB improved oxygenation after lung injury |
| Wrigge [25] | 2005 | 22 | Swine | Randomized Prospective APRV +/− SB |
50 % | 1.5–2 s | APRV + SB redistributes ventilation to dependent lung regions and counters cyclic collapse |
| Hering [12] | 2008 | 12 | Swine | Crossover APRV +/− SB |
50 % | N/A | APRV + SB improved oxygenation and splanchnic blood flow |
| Gama de Abreu [9] | 2008 | 12 | Swine | Crossover BiPAP + SB, PSV +/− sighs, “noisy” PSV |
N/A | exp flow 0 | “Noisy” CPPV improved oxygenation by redistributing perfusion |
| Carvalho [7] | 2009 | 5 | Swine | Crossover PSV vs. BiPAP + SB |
Titrated by Paw | N/A | BiPAP + SB and pressure support had similar oxygenation improvement and did not improve aeration of dependent lung |
| Gama de Abreu [4] | 2010 | 10 | Swine | Crossover PSV vs. BiPAP + SB |
25 % | N/A | BiPAP + SB had lower tidal volume with comparable oxygenation and ventilation distribution |
| Henzler [11] | 2010 | 20 | Swine | Randomized prospective APRV +/− SB |
42 % | ~1.2 s | Elevated IAH impaired respiratory mechanics regardless of SB |
| Kreyer [16] | 2010 | 12 | Swine | Randomized Prospective APRV +/− SB |
50 % | 1.5–2 s exp flow 0 |
APRV + SB improved systemic blood flow and cerebrospinal blood flow |
| Matsuzawa [18] | 2010 | 21 | Rabbit | Randomized prospective CPPV vs. LTV vs. APRV |
95 % | 0.15 s | APRV reduced HMGB1 levels and lung water |
| Slim [22] | 2011 | 7 | Swine | Case series APRV |
80 % | N/A | Increased Paw increased pulmonary capillary wedge pressure and left atrial pressure, but these may not correlate with end diastolic volume |
| Xia [26] | 2011 | 24 | Rabbit | Randomized prospective APRV +/− SB |
50 % | N/A | APRV + SB improved oxygenation and attenuated VILI |
| Carvalho [8] | 2014 | 36 | Swine | Randomized prospective APRV +/− SB |
50 % | N/A | APRV + SB improved oxygenation and reduced lung injury |
| Guldner [10] | 2014 | 12 | Swine | Crossover APRV +/− SB |
50 % | ~1 s | Higher levels of SB reduce global lung stress and strain with minimal changes in perfusion |
| Kill [15] | 2014 | 24 | Swine | Randomized prospective CPPV vs. Bilevel vs. Compression synchronized ventilation |
40 % | 3.6 s | CPPV and Bilevel usable during CPR, though compression synchronized ventilation was best |
Number of studies: 22
T Low time at low pressure, CPPV conventional positive pressure ventilation, LTV low tidal volume ventilation, CPAP continuous positive airway pressure, SB spontaneous breathing, PEEP positive end-expiratory pressure, PIP peak inspiratory pressure, P aw airway pressure, PSV pressure support ventilation, BiPAP biphasic positive airway pressure
Table 2.
Summary of animal studies utilizing P-APRV
| First author | Year | n | Animal | Study design | %CPAP | TLow | Findings |
|---|---|---|---|---|---|---|---|
| Albert [49] | 2011 | 22 | Swine | Randomized prospective, CPPV vs. LTV vs. APRV vs. HFOV | 90 % | 50–75 % PEF | APRV increased oxygenation and ventilation with reduced cytokines compared to LTV |
| Roy [57] | 2012 | 8 | Swine | Randomized prospective, CPPV vs. APRV | 90 % | 75 % PEF | Early APRV prevented ARDS with improved oxygenation, histopathology, and surfactant protein preservation |
| Emr [62] | 2013 | 16 | Rat | Randomized prospective, spont vs. CPPV vs. APRV | 90 % | 75 % PEF | Early APRV prevented ARDS with improved oxygenation and histopathology |
| Roy [5] | 2013 | 12 | Swine | Randomized prospective, sham vs. LTV vs. APRV | 90 % | 75 % PEF | Early APRV prevented ARDS with improved oxygenation and histopathology with reduced inflammatory markers |
| Roy [61] | 2013 | 9 | Rat | Randomized prospective, CPPV vs. APRV | 90 % | 75 % PEF | Early APRV prevented ARDS with improved oxygenation and histopathology |
| Kollisch-Singule [55] | 2014 | 8 | Rat | Randomized prospective, CPPV vs. APRV | 90 % | 10 or 75 % PEF | APRV with low expiratory time reduced conducting airway microstrain |
| Kollisch-Singule [54] | 2014 | 6 | Rat | Randomized prospective, CPPV with PEEP vs. APRV | 90 % | 10 or 75 % PEF | APRV with low expiratory time reduced alveolar microstrain |
| Davies [52] | 2015 | 22 | Swine | Randomized prospective, LTV vs. APRV | 90 % | 75 % PEF | APRV increased oxygenation compared with LTV, APRV had a “trend towards” increased cerebral ischemia. |
| Arrindell [51] | 2015 | 19 | Preterm swine | Randomized prospective, CPPV vs. APRV | N/A | 75 % PEF | APRV increased oxygenation without change in histopathology or inflammatory markers |
| Kollisch-Singule [56] | 2015 | 14 | Rat | Randomized prospective, uninjured vs. LTV vs. APRV | 90 % | 75 % PEF | APRV approximated control lungs best with increased homogeneity compared to LTV. LTV with high PEEP reduced heterogeneity. |
Number of studies: 10
T Low time at low pressure, CPPV conventional positive pressure ventilation, LTV low tidal volume ventilation, CPAP continuous positive airway pressure, PEF peak expiratory flow, SB spontaneous breathing, PEEP positive end-expiratory pressure, PIP peak inspiratory pressure
Table 3.
Summary of human trials using F-APRV
| First author | Year | n | Study design | %CPAP | TLow | Findings |
|---|---|---|---|---|---|---|
| Garner [31] | 1988 | 14 | Crossover CPPV baseline with APRV wean |
N/A | 1.5 s | APRV maintained similar oxygenation with >50 % reduced PIP |
| Rasanen [40] | 1991 | 50 | Crossover PEEP titrated CPPV vs. APRV |
50 % | 1.5 s | APRV maintained similar oxygenation with >50 % reduced PIP |
| Cane [28] | 1991 | 18 | Crossover CPPV vs. APRV |
67 % | 1.5 s | APRV maintained similar oxygenation and cardiopulmonary function with reduced PIP |
| Davis [3] | 1993 | 15 | Crossover CPPV vs. APRV |
32 % | 2.6 ± 0.6 | APRV maintained similar oxygenation with >50 % reduced PIP and reduced PEEP |
| Chiang [29] | 1994 | 18 | Crossover CPPV vs. APRV |
66 % | 1.5 s | APRV maintained similar oxygenation with >50 % reduced PIP |
| Sydow [42] | 1994 | 18 | Crossover CPPV vs. APRV |
80 % | 0.5–0.7 | APRV maintained similar oxygenation with decreased A-a gradient after 8 h and reduced PIP |
| Bratzke [27] | 1998 | 20 | Crossover CPPV vs. APRV |
88 % | 1 | APRV maintained similar oxygenation with reduced PIP |
| Kaplan [36] | 2001 | 12 | Crossover Inverse ratio PPV vs. APRV |
85 % | 0.8 | APRV is safe, decreases PIP and need for sedation/paralytics/pressors, increases CI |
| Putensen [39] | 2001 | 30 | Randomized prospective CPPV vs. APRV |
Identical to CPPV | Exp flow 0 | APRV + SB maintained increased oxygenation, CI, and pulmonary compliance with reduced ALI/ARDS incidence and sedative requirements |
| Schultz [41] | 2001 | 15 | Crossover CPPV vs. APRV |
N/A | N/A | APRV maintained similar oxygenation with >50 % reduced PIP |
| Wrigge [45] | 2001 | 14 | Randomized prospective APRV +/− automatic tube compensation |
N/A | N/A | APRV with tube compensation increased end-expiratory lung volume and minute ventilation without affecting oxygenation or cardiopulmonary status |
| Hering [34] | 2002 | 12 | Crossover APRV +/− SB |
N/A | N/A | APRV + SB had increased renal blood flow and glomerular filtration rate |
| Varpula [43] | 2003 | 33 | Randomized prospective CPPV vs. APRV |
N/A | Exp flow 0 | APRV feasible in prone positioning and increased oxygenation at 24 h |
| Varpula [44] | 2004 | 58 | Randomized prospective CPPV vs. APRV |
80 % | 1 | APRV had similar mortality and ventilator free days |
| Dart [30] | 2005 | 46 | Crossover CPPV vs. APRV |
N/A | 40–50 % PEF | APRV reduced PIP and increased oxygenation |
| Liu [37] | 2009 | 58 | Retrospective case-control CPPV vs. APRV |
67 % | ~1.5 | APRV reduced pressor use/A-a gradient and increased oxygenation |
| Kamath [35] | 2010 | 11 | Retrospective cohort CPPV vs. APRV |
70 % | 1.2 ± 0.9 | APRV had no adverse effects on blood pressure or urine output |
| Gonzalez [32] | 2010 | 468 | Case matched retrospective CPPV vs. APRV |
70 % | N/A | APRV maintained similar oxygenation with reduced PIP and associated increased tracheostomy rate |
| Maxwell [38] | 2010 | 63 | Randomized prospective LTV vs. APRV |
N/A | 25–75 % PEF | APRV had similar physiological parameters despite increased disease severity at baseline |
| Hanna [33] | 2011 | 45 | Retrospective case series CPPV vs. APRV |
N/A | N/A | APRV had increased P/F Ratio, lung procurement rate with similar graft survival rate |
| Maung [46] | 2012 | 38 | Retrospective case series APRV |
85 % | 0.8–1 | Switching from CPPV to APRV improved oxygenation and decreased PCO2 without hemodynamic compromise |
| Maung [47] | 2012 | 362 | Retrospective case series CPPV vs. APRV |
N/A | N/A | APRV had increased ventilator days. |
| Testerman [48] | 2013 | 48 | Case-matched retrospective APRV; obese vs. nonobese |
N/A | N/A | APRV in morbidly obese similar to nonobese, though morbidly obese required extended care after discharge more often |
Number of studies: 23
T Low time at low pressure, CPPV conventional positive pressure ventilation, LTV low tidal volume ventilation, CPAP continuous positive airway pressure, PEF peak expiratory flow, SB spontaneous breathing, PEEP positive end-expiratory pressure, PIP peak inspiratory pressure
Table 4.
Summary of human trial utilizing P-APRV
| First author | Year | n | Study design | %CPAP | TLow | Findings |
|---|---|---|---|---|---|---|
| Yoshida [60] | 2009 | 18 | Retrospective case-control LTV vs. APRV |
N/A | 50–75 % PEF | APRV + SB had increased oxygenation and MAP and decreased atelectasis |
| Walsh [58] | 2011 | 20 | Retrospective case series CPPV vs. APRV |
>80 % | 50–75 % PEF | APRV improved pulmonary blood flow after tetralogy of fallot repair or cavopulmonary shunt |
| Andrews [50] | 2013 | 66,099 | Retrospective review CPPV vs. early APRV |
90 % | 75 % PEF | Early APRV decreased ARDS incidence tenfold and mortality threefold |
| Kawaguchi [53] | 2014 | 13 | Retrospective case series CPPV vs. APRV |
90 % | 50–75 % PEF | APRV safe in pediatric ARDS without hemodynamic compromise |
| Yehya [59] | 2014 | 104 | Retrospective cohort HFOV vs. APRV |
N/A | 50–75 % PEF | APRV had no mortality effect compared to oscillatory ventilation as rescue treatments |
Number of studies: 5
T Low time at low pressure, CPPV conventional positive pressure ventilation, LTV low tidal volume ventilation, PEF peak expiratory flow, SB spontaneous breathing
Methodology
A PubMed search of the terms “Airway Pressure Release Ventilation”, “APRV”, “Bi-Vent”, “APRV/Biphasic” and “Bi-Level” and “Mechanical Ventilation” OR “Ventilator” was conducted alone or in combination. English language studies between the years 1987 and 2015 were included, and studies testing non-invasive ventilator strategies alone, reviews, editorials, and case studies were excluded, yielding 52 articles. Of the 60 articles, 32 were animal studies and 28 human studies (Tables 1, 2, 3, and 4). APRV methodologies were analyzed by examining the following settings: (1) pressure during inspiration/CPAP phase (PHigh); (2) time during inspiration/CPAP phase (THigh); (3) pressure during expiration/release phase (PLow); (4) time during expiration/release phase (TLow); and (5) percent CPAP (%CPAP) to reflect the time spent at PHigh relative to the entire breath duration . In addition, we assessed the factors used to titrate these settings including respiratory rate or partial pressure of carbon dioxide (PCO2) for THigh and TLow and oxygenation or plateau pressure for PHigh.
Reviewing the evolution of APRV from its inception in 1987, we noted a major paradigm shift in the way APRV is set. Initially, the variations in settings were to the inspiratory and expiratory duration time that were fixed and not adjusted to changes in lung mechanics (Fig. 1a–c). In 2005, Habashi published a paper with a novel method of setting the expiratory duration based on changing lung mechanics identified by the slope of the expiratory flow curve (Figs. 1d and 2) [6]. This novel method of setting expiratory duration sets this personalized APRV (P-APRV) strategy (Figs. 1d and 2) apart from all other fixed setting-APRV (F-APRV) strategies.
Fig. 2.
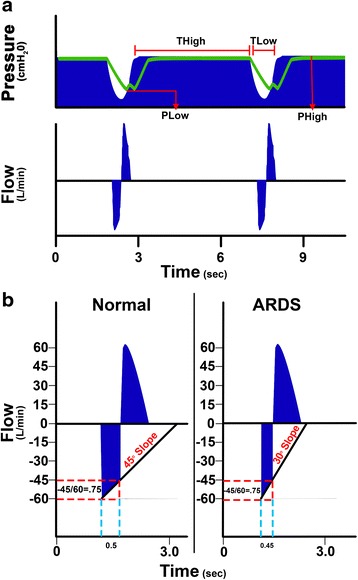
Method of setting expiratory duration (TLow). a Typical personalized airway pressure release ventilation (P-APRV) airway pressure and flow curves. Correctly set P-APRV has a very brief release phase (time at low pressure—TLow) and CPAP phase (time at high pressure—THigh) [6]. The THigh is ~90 % of each breath. The two other P-ARPV settings are the pressure at inspiration (PHigh) and at expiration (PLow). TLow is sufficiently brief such that end-expiratory pressure (PLow) never reaches 0 cmH2O measured by the tracheal pressure (green line). b Maintain alveolar stability by adaptively adjusting the expiratory duration as directed by the expiratory flow curve. The rate of lung collapse is seen in the normal (slope 45°) and acutely injured lung (ARDS, slope 30°). ARDS causes a more rapid lung collapse due to decreased lung compliance. Our preliminary studies have shown that if the end-expiratory flow (EEF; −45 L/min) to the peak expiratory flow (PEF; −60 L/min) ratio is equal to 0.75, the resultant TLow (0.5 s) is sufficient to stabilize alveoli [54, 55]. The lung with ARDS collapses more rapidly such that the EEF/PEF ratio of 75 % identifies an expiratory duration of 0.45 s as necessary to stabilize alveoli. Thus, this method of setting expiratory duration is adaptive to changes in lung pathophysiology and personalizes the mechanical breath to each individual patient
Thus, we chose to divide our review of APRV efficacy into these two basic categories. The F-APRV breath with a relatively short inspiratory time (THigh) occupying <90 % of total cycle time (Fig. 1a–c) of each breath with a fixed expiratory time (TLow) that is not adjusted based on changing lung mechanics. The second category originally described by Habashi [6] was a P-APRV breath with the following settings: (1) the inspiratory pressure (PHigh) is set to the desired plateau pressure,; (2) the THigh is typically set to occupy 90 % of the total cycle time of each breath (Fig. 2a); (3) the TLow set based on changes in lung mechanics by analyzing the slope of the expiratory flow curve (Fig. 2b); and (4) the expiratory pressure (PLow) is set at 0 cmH2O to minimize resistance to convective expiratory gas flow and maximize ventilation. The short duration at end expiration prevents the airway pressure from reaching atmospheric pressure, thus maintaining a positive end-expiratory pressure. Based on the above criteria, the studies reviewed were placed into either the F-APRV (Tables 1 and 3) or P-APRV (Tables 2 and 4) category.
Results
Animal (Tables 1 and 2) and human (Tables 3 and 4) studies were examined for APRV settings and efficacy. As described above, the APRV methodologies were subdivided into two categories: F-APRV; (Tables 1 and 3) and P-APRV (Tables 2 and 4). The majority of the animal studies (69 % of total) [2, 4, 7–26] and human studies (82 % of total) [3, 27–48] were in the F-APRV category.
How expiratory duration was personalized
The expiratory flow curve is analyzed, and the ratio of the end-expiratory flow (EEF) to the peak expiratory flow (PEF) is set so that the EEF/PEF ratio is 75 % [5, 49–62] (Fig. 2b) based on the methodology described by Habashi [6] (Tables 2 and 4). The TLow is thus personalized based on alterations in lung mechanics, identified by changes in the slope of the expiratory flow curve (Fig. 2b). Using EEF/PEF ratio of 75 % results in a brief TLow ranging from 0.3 to 0.6 s (Fig. 1d). However, recent animal experiments have shown the optimal EEF/PEF ratio necessary to open and stabilize the lung is 75 % [54–56].
F-APRV: inspiratory duration
There was considerable variability in the F-APRV settings (i.e., duration at inspiration and expiration) in the papers reviewed, and in multiple papers, the settings were not defined, and thus not included in this review. Swine, dogs, and rabbits were used in the animals studies with the majority of studies using swine (Table 1). In only two of the animal studies were the %CPAP set at ≤80 % of the total cycle time (Table 1) [22, 23]. In one study, %CPAP was set very short at 25 % of the breath cycle time [5]. When both the animal and human studies were analyzed 23/45 studies set %CPAP ≤67 % of the breath and 13/45 had an indeterminate %CPAP (Tables 1 and 3).
Expiratory duration
In addition, the TLow in both animal and human studies was prolonged to levels seen in conventional mechanical ventilation (Fig. 1) [3, 9, 15, 16, 24, 25, 43]. Aside from two studies [30, 38], the TLow remained fixed or was titrated based on the PCO2 as opposed to being adjusted based on changes in lung mechanics using the expiratory flow curve (Fig. 2). The PHigh was titrated very differently in the studies reviewed, with a range between 10 cmH2O [2, 21, 28, 29, 31] and 35 cmH2O [3, 37] depending on whether the target was maximal oxygenation, maximal lung recruitment, or a specific tidal volume. In addition, most of the studies in the F-APRV group set a positive PLow > 0 cmH2O [2, 4, 7–11, 13, 14, 16, 18–23, 26, 40].
Outcome
The majority of the studies in the F-APRV group were crossover experiments, representing 59 % of the animal studies (Table 1) and 48 % of the human trials (Table 3) [2, 4, 7, 9, 10, 12–14, 17, 19, 21, 23, 32], with the primary goal to demonstrate that APRV could be safely used without adverse effects on lung function or hemodynamic characteristics. Many crossover trials showed similar or increased oxygenation with lower peak pressures and no negative effect on hemodynamics with APRV as compared with CPPV [2, 3, 17, 27–32, 39–42], and some studies showed hemodynamic improvement with APRV [13, 24, 36, 39]. Prospective randomized trials comparing APRV and CPPV showed that APRV is safe and potentially beneficial (Table 3). In none of the studies included in this review did APRV cause a significant negative impact on the physiologic process being studied, and in some prospective randomized studies, APRV was shown beneficial (Tables 1 and 3).
P-APRV
Similar to the F-APRV studies, P-APRV either resulted in improved outcomes or no change as compared with CPPV and in none of the studies was APRV shown to be harmful. Unlike the F-APRV studies that were mainly crossover studies, all of the P-APRV animal studies were randomized prospective cohort trials that compare non-protective CPPV or low tidal volume (LTV) with APRV [5, 49, 52, 54, 55, 57, 61, 62]. P-APRV was shown to reduce heterogeneity and both alveolar and alveolar duct micro-strain (i.e., change in alveolar size with applied stress) [54–56] suggesting a mechanism for the improvement in the efficacy experiments (Table 3) [5, 49, 57, 61, 62].
Discussion
There has been no consensus on what parameters are essential to define a mechanical breath as being APRV, and thus, APRV settings have been inconsistent over the three decades since it was first described. However, no studies have shown that APRV is harmful or significantly inferior as compared with conventional mechanical ventilation. In 2005, Habashi clearly defined the settings for what we have termed personalized APRV or P-APRV [6]. P-APRV has a prolonged THigh and very brief TLow duration, which is set by lung mechanics using the change in the slope of the expiratory flow curve [6]. Not only is the TLow very brief in P-APRV, it is set by analyzing the slope of the expiratory flow curve and therefore adaptive to changes in the patient’s lung mechanics (Fig. 2b). Using these precisely controlled APRV settings, a number of studies have shown that P-APRV recruits and stabilizes the alveoli and alveolar ducts [54–56] and reduces the incidence of ARDS in multiple animal models [5, 57, 62]. A meta-analysis has shown a reduction in ARDS incidence in trauma patients [53]. Given this lack of consensus on how APRV should be set, the remainder of the “Discussion” will be divided into the results obtained from the F-APRV and P-APRV subgroups, rather than discuss the findings from each experiment with a different %CPAP, TLow, and/or PHigh settings.
F-APRV studies
Since, as previously mentioned, most of the animal and human studies were crossover experiments (i.e., switching from CPPV or spontaneous breathing to F-APRV in the same animal or patient), the majority of published APRV studies do not address efficacy (i.e., is APRV superior to conventional ventilation). It is important to note that APRV did not cause a significant negative impact, as compared with CPPV or spontaneous breathing, on the physiologic parameters that were measured in any of the crossover studies (Table 1 and 3). In the randomized prospective animal studies, F-APRV was shown to be beneficial. It was shown that APRV reduced lung water and HMGB1 in rabbits [18], improved systemic and cerebrospinal blood flow in swine [16], improved oxygenation and attenuated ventilator-induced lung injury (VILI) in rabbits [26], and improved oxygenation and reduced lung injury in swine [7].
In humans, Putsenen et al. showed that APRV with spontaneous breathing increased oxygenation, cardiac index, and pulmonary compliance, with reduced sedative requirements compared with CPPV in humans [39]. Varpula et al. reported similar mortality and ventilator-free days for APRV and CPPV, and also demonstrated that it is feasible to utilize APRV with prone positioning. In that study, TLow was set specifically to allow expiratory flow to reach zero, which would allow the lung to collapse during expiration [43, 44]. Maxwell et al. showed no difference between low tidal volume ventilation (LTV) and APRV regarding mortality, ventilator days, ICU length of stay, or complication rates despite increased baseline disease severity in the APRV group [38]. In human retrospective trials, the APRV methodology used was not consistent among the studies analyzed (Table 3). Despite these inconsistencies, Gonzalez et al. showed that APRV reduced peak inspiratory pressure (PIP) while maintaining similar oxygenation levels [32]. Hanna et al. showed increased PaO2/FiO2 (P/F) ratio, lung procurement rate, and graft survival when used in organ donors where the lungs were transplanted [33].
A retrospective study examining trauma patients by Maung et al. demonstrated increased ventilator days on APRV compared with an unspecified method of CPPV [47]. However, this study excluded all mortality in the interest of investigating weaning and the baseline characteristics of the groups were different, with more severe chest trauma and decreased P/F ratio at the start in the APRV group. Further, this study used respiratory therapist-guided protocol weaning only in the CPPV group, whereas the APRV group had random, non-protocolized weaning based on physician guidance.
P-APRV
Despite defining the role of tidal volume (Vt) and plateau pressure on VILI in ARDS patients [63], our current understanding of the parameters comprising the mechanical breath that either propagate or impede progressive acute lung injury (ALI) is incomplete. The mechanism of VILI is believed to be the evolution from a normal homogenously ventilated lung into a heterogeneously ventilated lung, with collapse and edema-filled alveoli adjacent to open alveoli. This heterogeneity results in stress concentrators and recurrent alveolar collapse and reopening with each tidal breath that amplify lung tissue injury, instigated by the initial insult such as sepsis, trauma, or pneumonia [64–66]. Thus, the ventilation strategy that restores or maintains homogeneity would minimize VILI and obstruct progressive ALI [64, 66, 67].
P-APRV uses a 90 % CPAP phase that recruits alveoli resulting in homogeneous lung inflation (Fig. 2a) and a brief release phase with the TLow personalized to the mechanics of the lung (Fig. 2b) producing a nearly static ventilated lung, which prevents alveolar collapse and reopening, thereby reducing dynamic tissue strain [54, 56]. The question is, do these APRV settings that stabilize alveoli protect the lung?
In prospective outcome animal experiments, P-APRV resulted in an increase in oxygenation along with a decrease in histopathologic injury as compared with CPPV and LTV ventilation strategies [5, 52, 57, 61, 62]. Several studies showed that P-APRV resulted in an increase in surfactant protein concentration [5, 57, 62], while two showed a reduction of inflammatory markers (Table 2) [49, 57].
Kollisch-Singule et al. [54–56] conducted three micro-anatomic studies (i.e., alveoli and alveolar ducts) that demonstrated reduced alveolar and conducting airway micro-strain as well as increased alveolar homogeneity using P-APRV in which the TLow was set to maintain an EEF/PEF ratio 75 or 10 %. The TLow set using an EEF/PEF ratio of 75 % was sufficiently short to stabilize alveoli and prevent alveolar collapse, whereas extending the TLow (EEF/PEF ratio 10 %) resulted in alveolar collapse and instability. These studies add mechanistic support to efficacy studies showing that preemptive P-APRV reduced ARDS incidence in a clinically applicable porcine ARDS model [5, 57].
Davies et al. [52] showed increased oxygenation with P-APRV as compared with LTV, with no significant differences in cerebral ischemia in a swine model of concomitant brain and lung injury (Table 2). In patients, Yoshida et al. [60] demonstrated increased oxygenation and mean arterial pressure (MAP) with decreased atelectasis using P-APRV as compared to LTV ventilation. Walsh et al. [58] showed improved pulmonary blood flow when using P-APRV versus CPPV ventilation post-operatively after tetratology of Fallot repair or cavopulmonary shunt in neonates (Table 4).
Furthermore, Andrews et al. [50] in a meta-analysis showed a tenfold decrease in ARDS incidence as well as a threefold decrease in mortality when compared to trauma patients with similar injuries that were treated with standard of care ventilation in 15 trauma intensive care units (Table 4). However, all of the human trials testing the P-APRV method [50, 53, 58–60] are retrospective studies, and as such, it is not clear that the precise P-APRV settings were followed, aside from the study by Andrews et al. [50] that strictly adhered to the P-APRV protocol throughout the study in trauma patients. Despite this, the human results are in concordance with multiple animal studies [5, 49, 54–57, 61, 62] that support the clinical data [50] that P-APRV may be used to reduce the incidence of acute respiratory distress syndrome (ARDS) more effectively than conventional LTV ventilation applied early or after the onset of ARDS.
A note on mechanical ventilators
An often under-analyzed aspect of mechanical breath delivery is the ventilator used to deliver the breath. Every ventilator has a specific design with differing resistances built into the ventilator gas path, responses to spontaneous ventilation, as well as software quirks that affect breath delivery. APRV may be named in various ways based on the ventilator in use such as (1) APRV (Drӓger Evita, Savina and V series, Hamilton G5), (2) Bi-Vent (Maquet Servo-i), (3) BiLevel (Engström Carestation, Puritan Bennett 840 & 980), and (4) APRV/Biphasic (Viasys Avea). However, even more problematic than the different names is the wide variation in their implementation of APRV.
In particular, the ability to control key APRV parameters such as the TLow varies, and fine control of this parameter is critical to properly set P-APRV (Fig. 2b). APRV strategies on some ventilators have deviated from the original concept of a timed CPAP with a release phase, by adding pressure support (PS). This requires a trigger that creates a need for synchronization with the ventilator resulting in automated changes to both the inspiratory and expiratory duration. More specifically, the TLow, which is critical to control end-expiratory lung volume and prevent airway closure, spontaneously adjusts when PS is added to this mode on these ventilators, regardless of the TLow setting, producing large and variable tidal volumes leading to lung volume loss and alveolar instability. So, even if APRV was set accurately by the clinician, the ventilator may automatically adjust the duration of the release phase (TLow) ultimately allowing the lung to collapse. This effect would be similar to conventional ventilation automatically adjusting Vt or PEEP without physician input.
Furthermore, many researchers define APRV as “extreme” inverse-ratio pressure-control ventilation (IR-PCV). However, unlike IR-PCV, P-APRV does not set a defined I:E ratio. Rather, the CPAP phase is briefly interrupted by a “release phase” (i.e., TLow) that is established by independently setting a TLow based upon analysis of the expiratory flow curve. Further, patients can breathe spontaneously throughout the entire respiratory cycle in APRV, superseding the set I:E ratio. Unfortunately, many times the exact make and model of the ventilator was not reported in the papers reviewed, and thus, we could not categorize studies by the ventilator used, but it is an important factor to consider if the ventilator being used can accurately deliver P-APRV.
Current state of APRV and clinical implications
As described previously, there is a paucity of data testing APRV with identical settings, especially in clinical trials almost all of which were retrospective or crossover studies. Rose et al. [68] reviewed the literature in 2008 and concluded that there is a lack of consistency in APRV settings making comparison with conventional ventilator strategies difficult. Another recent review by Facchin et al. [69] examined the current literature for both APRV and high-frequency oscillatory ventilation (HFOV) for treatment and prevention of ARDS; the authors concluded that there is inconsistent evidence and a lack of high quality trials to make conclusions regarding APRV or HFOV efficacy.
It is clear from the studies examined in this review that APRV has evolved from a mechanical breath defined as a prolonged CPAP phase with a brief release phase [1] into a highly sophisticated, dynamic mechanical breath with precise settings that are responsive to changes in lung physiology (i.e., P-APRV) [6]. Although the mechanical breath used by both Downs and Habashi are referred to as APRV, the actual mechanical breath created by each is very different (Fig. 1a, d) [2, 57] as is the breath created and studied in other experiments [3, 9] (Fig. 1b, c). The current use of the APRV acronym remains an imprecise term that demands a specific definition of parameter settings to be utilized consistently. Obviously, some APRV settings are going to superior to others in their ability to protect the lung.
P-APRV appears to be an exciting and novel open lung strategy that may significantly reduce ARDS incidence, morbidity, and mortality of established ARDS. Data suggests that rather than overdistending alveoli, the extended THigh/PHigh redistributes gas from the alveolar ducts to the alveoli, where it belongs [54, 55] and changes heterogeneous to homogeneous alveolar ventilation [56]. The calculated strain on alveoli was significantly reduced demonstrating that mechanical stretch on the alveolar wall was decreased. These data have led us to conclude that the extended inspiratory duration has a powerful positive impact on reducing strain at the alveolar level. We hypothesize that the mechanism of this protection (i.e., reduce alveolar strain) is secondary to increased lung volume and the number of recruited alveoli. The extended THigh “nudges” open alveoli over a several hours in a non-pathologic manner resulting in open lung ventilation. Since the lung becomes fully recruited with significantly improved compliance, even with relatively high tidal volumes (10–14 cm3/kg), the driving pressure is not increased (unpublished observations).
Conclusions
Although many settings have been used, none of the studies reviewed showed a worse outcome using APRV as compared with CPPV, with many studies showing significant benefits in cardiopulmonary variables. The evolution of APRV methodology has been drastic from Downs and Stock (F-APRV) [1] to the P-APRV method of Habashi [6]. P-APRV allows for a personalized control of lung stability on a breath-to-breath basis that is not possible with other modes of ventilation. P-APRV is an adaptive, flow directed, duration dependent ventilation strategy that adapts the setting to each patient regardless of their lung pathophysiology. This personalized, adaptive mechanical breath may prove more efficacious at treating and preventing ARDS than the current standard of care. Ultimately, more studies are needed using consistent and well-defined settings to identify the optimal APRV breath necessary to maximize lung protection.
Acknowledgments
Authors’ contributions
SVJ, MKS, BS, GFN, PA, and NMH contributed to the conception and design of this review. SVJ, LD, PA, and NMH contributed to data acquisition. SVJ, JS, GFN, and NMH helped draft the manuscript. All authors critically reviewed, read, and approved the final manuscript.
Competing interests
GFN/NMH/PLA have presented and received honoria and travel reimbursement at events sponsored by DrägerMedical outside of the submitted work.
GFN/NMH/PLA/SJ have lectured for Intensive Care Online (ICON).
PLA is employed by ICON.
NMH hold patents and is the founder of ICON.
Industry had no role in the design and conduct of the study; the collection, management, analysis, or interpretation of the data; or preparation, review, or approval of the manuscript.
Abbreviations
- ALI
acute lung injury
- APRV
airway pressure release ventilation
- ARDS
acute respiratory distress syndrome
- BiPAP
biphasic positive airway pressure
- CPAP
continuous positive airway pressure
- CPPV
conventional positive pressure ventilation
- F-APRV
fixed-setting airway pressure release ventilation
- HFOV
high frequency oscillatory ventilation
- LTV
low tidal volume
- MAP
mean arterial pressure
- P/F
PaO2/FiO2
- P-APRV
personalized airway pressure release ventilation
- Paw
airway pressure
- PCO2
partial pressure of carbon dioxide
- PEEP
positive end-expiratory pressure
- PEF
peak expiratory flow
- PHigh
pressure during CPAP phase
- PIP
peak inspiratory pressure
- PLow
pressure during release phase
- PS
pressure support
- PSV
pressure support ventilation
- SB
spontaneous breathing
- THigh
time at CPAP/PHigh
- TLow
time at release/PLow
- VILI
ventilator-induced lung injury
- Vt
tidal volume
References
- 1.Downs JB, Stock MC. Airway pressure release ventilation: a new concept in ventilatory support. Crit Care Med. 1987;15:459–461. doi: 10.1097/00003246-198701000-00018. [DOI] [PubMed] [Google Scholar]
- 2.Stock MC, Downs JB, Frolicher DA. Airway pressure release ventilation. Crit Care Med. 1987;15:462–466. doi: 10.1097/00003246-198705000-00002. [DOI] [PubMed] [Google Scholar]
- 3.Davis K, Jr, Johnson DJ, Branson RD, Campbell RS, Johannigman JA, Porembka D. Airway pressure release ventilation. Arch Surg. 1993;128:1348–1352. doi: 10.1001/archsurg.1993.01420240056010. [DOI] [PubMed] [Google Scholar]
- 4.Gama de Abreu M, Cuevas M, Spieth PM, Carvalho AR, Hietschold V, Stroszczynski C, Wiedemann B, Koch T, Pelosi P, Koch E. Regional lung aeration and ventilation during pressure support and biphasic positive airway pressure ventilation in experimental lung injury. Crit Care. 2010;14:R34. doi: 10.1186/cc8912. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Roy S, Habashi N, Sadowitz B, Andrews P, Ge L, Wang G, Roy P, Ghosh A, Kuhn M, Satalin J, Gatto LA, Lin X, Dean DA, Vodovotz Y, Nieman G. Early airway pressure release ventilation prevents ARDS—a novel preventive approach to lung injury. Shock. 2013;39:28–38. doi: 10.1097/SHK.0b013e31827b47bb. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Habashi NM. Other approaches to open-lung ventilation: airway pressure release ventilation. Crit Care Med. 2005;33:S228–S240. doi: 10.1097/01.CCM.0000155920.11893.37. [DOI] [PubMed] [Google Scholar]
- 7.Carvalho AR, Spieth PM, Pelosi P, Beda A, Lopes AJ, Neykova B, Heller AR, Koch T, Gama de Abreu M. Pressure support ventilation and biphasic positive airway pressure improve oxygenation by redistribution of pulmonary blood flow. Anesth Analg. 2009;109:856–865. doi: 10.1213/ane.0b013e3181aff245. [DOI] [PubMed] [Google Scholar]
- 8.Carvalho NC, Guldner A, Beda A, Rentzsch I, Uhlig C, Dittrich S, Spieth PM, Wiedemann B, Kasper M, Koch T, Richter T, Rocco PR, Pelosi P, de Abreu MG. Higher levels of spontaneous breathing reduce lung injury in experimental moderate acute respiratory distress syndrome. Crit Care Med. 2014;42:e702–e715. doi: 10.1097/CCM.0000000000000605. [DOI] [PubMed] [Google Scholar]
- 9.Gama de Abreu M, Spieth PM, Pelosi P, Carvalho AR, Walter C, Schreiber-Ferstl A, Aikele P, Neykova B, Hubler M, Koch T. Noisy pressure support ventilation: a pilot study on a new assisted ventilation mode in experimental lung injury. Crit Care Med. 2008;36:818–827. doi: 10.1097/01.CCM.0000299736.55039.3A. [DOI] [PubMed] [Google Scholar]
- 10.Guldner A, Braune A, Carvalho N, Beda A, Zeidler S, Wiedemann B, Wunderlich G, Andreeff M, Uhlig C, Spieth PM, Koch T, Pelosi P, Kotzerke J, de Abreu MG. Higher levels of spontaneous breathing induce lung recruitment and reduce global stress/strain in experimental lung injury. Anesthesiology. 2014;120:673–682. doi: 10.1097/ALN.0000000000000124. [DOI] [PubMed] [Google Scholar]
- 11.Henzler D, Hochhausen N, Bensberg R, Schachtrupp A, Biechele S, Rossaint R, Kuhlen R. Effects of preserved spontaneous breathing activity during mechanical ventilation in experimental intra-abdominal hypertension. Intensive Care Med. 2010;36:1427–1435. doi: 10.1007/s00134-010-1827-3. [DOI] [PubMed] [Google Scholar]
- 12.Hering R, Bolten JC, Kreyer S, Berg A, Wrigge H, Zinserling J, Putensen C. Spontaneous breathing during airway pressure release ventilation in experimental lung injury: effects on hepatic blood flow. Intensive Care Med. 2008;34:523–527. doi: 10.1007/s00134-007-0957-8. [DOI] [PubMed] [Google Scholar]
- 13.Hering R, Viehofer A, Zinserling J, Wrigge H, Kreyer S, Berg A, Minor T, Putensen C. Effects of spontaneous breathing during airway pressure release ventilation on intestinal blood flow in experimental lung injury. Anesthesiology. 2003;99:1137–1144. doi: 10.1097/00000542-200311000-00021. [DOI] [PubMed] [Google Scholar]
- 14.Hering R, Zinserling J, Wrigge H, Varelmann D, Berg A, Kreyer S, Putensen C. Effects of spontaneous breathing during airway pressure release ventilation on respiratory work and muscle blood flow in experimental lung injury. Chest. 2005;128:2991–2998. doi: 10.1378/chest.128.4.2991. [DOI] [PubMed] [Google Scholar]
- 15.Kill C, Hahn O, Dietz F, Neuhaus C, Schwarz S, Mahling R, Wallot P, Jerrentrup A, Steinfeldt T, Wulf H, Dersch W. Mechanical ventilation during cardiopulmonary resuscitation with intermittent positive-pressure ventilation, bilevel ventilation, or chest compression synchronized ventilation in a pig model. Crit Care Med. 2014;42:e89–e95. doi: 10.1097/CCM.0b013e3182a63fa0. [DOI] [PubMed] [Google Scholar]
- 16.Kreyer S, Putensen C, Berg A, Soehle M, Muders T, Wrigge H, Zinserling J, Hering R. Effects of spontaneous breathing during airway pressure release ventilation on cerebral and spinal cord perfusion in experimental acute lung injury. J Neurosurg Anesthesiol. 2010;22:323–329. doi: 10.1097/ANA.0b013e3181e775f1. [DOI] [PubMed] [Google Scholar]
- 17.Martin LD, Wetzel RC, Bilenki AL. Airway pressure release ventilation in a neonatal lamb model of acute lung injury. Crit Care Med. 1991;19:373–378. doi: 10.1097/00003246-199103000-00016. [DOI] [PubMed] [Google Scholar]
- 18.Matsuzawa Y, Nakazawa K, Yamamura A, Akashi T, Kitagaki K, Eishi Y, Makita K. Airway pressure release ventilation reduces the increase in bronchoalveolar lavage fluid high-mobility group box-1 levels and lung water in experimental acute respiratory distress syndrome induced by lung lavage. Eur J Anaesthesiol. 2010;27:726–733. doi: 10.1097/EJA.0b013e328333c2b0. [DOI] [PubMed] [Google Scholar]
- 19.Neumann P, Hedenstierna G. Ventilatory support by continuous positive airway pressure breathing improves gas exchange as compared with partial ventilatory support with airway pressure release ventilation. Anesth Analg. 2001;92:950–958. doi: 10.1097/00000539-200104000-00030. [DOI] [PubMed] [Google Scholar]
- 20.Neumann P, Wrigge H, Zinserling J, Hinz J, Maripuu E, Andersson LG, Putensen C, Hedenstierna G. Spontaneous breathing affects the spatial ventilation and perfusion distribution during mechanical ventilatory support. Crit Care Med. 2005;33:1090–1095. doi: 10.1097/01.CCM.0000163226.34868.0A. [DOI] [PubMed] [Google Scholar]
- 21.Rasanen J, Downs JB, Stock MC. Cardiovascular effects of conventional positive pressure ventilation and airway pressure release ventilation. Chest. 1988;93:911–915. doi: 10.1378/chest.93.5.911. [DOI] [PubMed] [Google Scholar]
- 22.Slim AM, Martinho S, Slim J, Davenport E, Castillo-Rojas LM, Shry EA. The effect of airway pressure release ventilation on pulmonary catheter readings: specifically pulmonary capillary wedge pressure in a swine model. Anesthesiology Res Prac. 2011;2011:371594. doi: 10.1155/2011/371594. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Smith RA, Smith DB. Does airway pressure release ventilation alter lung function after acute lung injury? Chest. 1995;107:805–808. doi: 10.1378/chest.107.3.805. [DOI] [PubMed] [Google Scholar]
- 24.Wrigge H, Zinserling J, Neumann P, Defosse J, Magnusson A, Putensen C, Hedenstierna G. Spontaneous breathing improves lung aeration in oleic acid-induced lung injury. Anesthesiology. 2003;99:376–384. doi: 10.1097/00000542-200308000-00019. [DOI] [PubMed] [Google Scholar]
- 25.Wrigge H, Zinserling J, Neumann P, Muders T, Magnusson A, Putensen C, Hedenstierna G. Spontaneous breathing with airway pressure release ventilation favors ventilation in dependent lung regions and counters cyclic alveolar collapse in oleic-acid-induced lung injury: a randomized controlled computed tomography trial. Crit Care. 2005;9:R780–R789. doi: 10.1186/cc3908. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Xia J, Sun B, He H, Zhang H, Wang C, Zhan Q. Effect of spontaneous breathing on ventilator-induced lung injury in mechanically ventilated healthy rabbits: a randomized, controlled, experimental study. Crit Care. 2011;15:R244. doi: 10.1186/cc10502. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Bratzke E, Downs JB, Smith RA. Intermittent CPAP: a new mode of ventilation during general anesthesia. Anesthesiology. 1998;89:334–340. doi: 10.1097/00000542-199808000-00008. [DOI] [PubMed] [Google Scholar]
- 28.Cane RD, Peruzzi WT, Shapiro BA. Airway pressure release ventilation in severe acute respiratory failure. Chest. 1991;100:460–463. doi: 10.1378/chest.100.2.460. [DOI] [PubMed] [Google Scholar]
- 29.Chiang AA, Steinfeld A, Gropper C, MacIntyre N. Demand-flow airway pressure release ventilation as a partial ventilatory support mode: comparison with synchronized intermittent mandatory ventilation and pressure support ventilation. Crit Care Med. 1994;22:1431–1437. doi: 10.1097/00003246-199409000-00013. [DOI] [PubMed] [Google Scholar]
- 30.Dart BW, Maxwell RA, Richart CM, Brooks DK, Ciraulo DL, Barker DE, Burns RP. Preliminary experience with airway pressure release ventilation in a trauma/surgical intensive care unit. J Trauma. 2005;59:71–76. doi: 10.1097/00005373-200507000-00010. [DOI] [PubMed] [Google Scholar]
- 31.Garner W, Downs JB, Stock MC, Rasanen J. Airway pressure release ventilation (APRV). A human trial. Chest. 1988;94:779–781. doi: 10.1378/chest.94.4.779. [DOI] [PubMed] [Google Scholar]
- 32.Gonzalez M, Arroliga AC, Frutos-Vivar F, Raymondos K, Esteban A, Putensen C, Apezteguia C, Hurtado J, Desmery P, Tomicic V, Elizalde J, Abroug F, Arabi Y, Moreno R, Anzueto A, Ferguson ND. Airway pressure release ventilation versus assist-control ventilation: a comparative propensity score and international cohort study. Intensive Care Med. 2010;36:817–827. doi: 10.1007/s00134-010-1837-1. [DOI] [PubMed] [Google Scholar]
- 33.Hanna K, Seder CW, Weinberger JB, Sills PA, Hagan M, Janczyk RJ. Airway pressure release ventilation and successful lung donation. Arch Surg. 2011;146:325–328. doi: 10.1001/archsurg.2011.35. [DOI] [PubMed] [Google Scholar]
- 34.Hering R, Peters D, Zinserling J, Wrigge H, von Spiegel T, Putensen C. Effects of spontaneous breathing during airway pressure release ventilation on renal perfusion and function in patients with acute lung injury. Intensive Care Med. 2002;28:1426–1433. doi: 10.1007/s00134-002-1442-z. [DOI] [PubMed] [Google Scholar]
- 35.Kamath SS, Super DM, Mhanna MJ. Effects of airway pressure release ventilation on blood pressure and urine output in children. Pediatr Pulmonol. 2010;45:48–54. doi: 10.1002/ppul.21058. [DOI] [PubMed] [Google Scholar]
- 36.Kaplan LJ, Bailey H, Formosa V. Airway pressure release ventilation increases cardiac performance in patients with acute lung injury/adult respiratory distress syndrome. Crit Care. 2001;5:221–226. doi: 10.1186/cc1027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Liu L, Tanigawa K, Ota K, Tamura T, Yamaga S, Kida Y, Kondo T, Ishida M, Otani T, Sadamori T, Tsumura R, Takeda T, Iwasaki Y, Hirohashi N. Practical use of airway pressure release ventilation for severe ARDS—a preliminary report in comparison with a conventional ventilatory support. Hiroshima J Med Sci. 2009;58:83–88. [PubMed] [Google Scholar]
- 38.Maxwell RA, Green JM, Waldrop J, Dart BW, Smith PW, Brooks D, Lewis PL, Barker DE. A randomized prospective trial of airway pressure release ventilation and low tidal volume ventilation in adult trauma patients with acute respiratory failure. J Trauma. 2010;69:501–510. doi: 10.1097/TA.0b013e3181e75961. [DOI] [PubMed] [Google Scholar]
- 39.Putensen C, Zech S, Wrigge H, Zinserling J, Stuber F, Von Spiegel T, Mutz N. Long-term effects of spontaneous breathing during ventilatory support in patients with acute lung injury. Am J Respir Crit Care Med. 2001;164:43–49. doi: 10.1164/ajrccm.164.1.2001078. [DOI] [PubMed] [Google Scholar]
- 40.Rasanen J, Cane RD, Downs JB, Hurst JM, Jousela IT, Kirby RR, Rogove HJ, Stock MC. Airway pressure release ventilation during acute lung injury: a prospective multicenter trial. Crit Care Med. 1991;19:1234–1241. doi: 10.1097/00003246-199110000-00004. [DOI] [PubMed] [Google Scholar]
- 41.Schultz TR, Costarino AJA, Durning SM, Napoli LA, Schears G, Godinez RI, Priestley M, Dominguez T, Lin R, Helfaer M. Airway pressure release ventilation in pediatrics. Pediatr Crit Care Med. 2001;2:243–246. doi: 10.1097/00130478-200107000-00010. [DOI] [PubMed] [Google Scholar]
- 42.Sydow M, Burchardi H, Ephraim E, Zielmann S, Crozier TA. Long-term effects of two different ventilatory modes on oxygenation in acute lung injury. Comparison of airway pressure release ventilation and volume-controlled inverse ratio ventilation. Am J Respir Crit Care Med. 1994;149:1550–1556. doi: 10.1164/ajrccm.149.6.8004312. [DOI] [PubMed] [Google Scholar]
- 43.Varpula T, Jousela I, Niemi R, Takkunen O, Pettila V. Combined effects of prone positioning and airway pressure release ventilation on gas exchange in patients with acute lung injury. Acta Anaesthesiol Scand. 2003;47:516–524. doi: 10.1034/j.1399-6576.2003.00109.x. [DOI] [PubMed] [Google Scholar]
- 44.Varpula T, Valta P, Niemi R, Takkunen O, Hynynen M, Pettila VV. Airway pressure release ventilation as a primary ventilatory mode in acute respiratory distress syndrome. Acta Anaesthesiol Scand. 2004;48:722–731. doi: 10.1111/j.0001-5172.2004.00411.x. [DOI] [PubMed] [Google Scholar]
- 45.Wrigge H, Zinserling J, Hering R, Schwalfenberg N, Stuber F, von Spiegel T, Schroeder S, Hedenstierna G, Putensen C. Cardiorespiratory effects of automatic tube compensation during airway pressure release ventilation in patients with acute lung injury. Anesthesiology. 2001;95:382–389. doi: 10.1097/00000542-200108000-00020. [DOI] [PubMed] [Google Scholar]
- 46.Maung AA, Luckianow G, Kaplan LJ. Lessons learned from airway pressure release ventilation. J Trauma Acute Care Surg. 2012;72:624–628. doi: 10.1097/TA.0b013e318247668f. [DOI] [PubMed] [Google Scholar]
- 47.Maung AA, Schuster KM, Kaplan LJ, Ditillo MF, Piper GL, Maerz LL, Lui FY, Johnson DC, Davis KA. Compared to conventional ventilation, airway pressure release ventilation may increase ventilator days in trauma patients. J Trauma Acute Care Surg. 2012;73:507–510. doi: 10.1097/TA.0b013e31825ff653. [DOI] [PubMed] [Google Scholar]
- 48.Testerman GM, Breitman I, Hensley S. Airway pressure release ventilation in morbidly obese surgical patients with acute lung injury and acute respiratory distress syndrome. Am Surg. 2013;79:242–246. [PubMed] [Google Scholar]
- 49.Albert S, Kubiak BD, Vieau CJ, Roy SK, DiRocco J, Gatto LA, Young JL, Tripathi S, Trikha G, Lopez C, Nieman GF. Comparison of “open lung” modes with low tidal volumes in a porcine lung injury model. J Surg Res. 2011;166:e71–e81. doi: 10.1016/j.jss.2010.10.022. [DOI] [PubMed] [Google Scholar]
- 50.Andrews PL, Shiber JR, Jaruga-Killeen E, Roy S, Sadowitz B, O'Toole RV, Gatto LA, Nieman GF, Scalea T, Habashi NM. Early application of airway pressure release ventilation may reduce mortality in high-risk trauma patients: a systematic review of observational trauma ARDS literature. J Trauma Acute Care Surg. 2013;75:635–641. doi: 10.1097/TA.0b013e31829d3504. [DOI] [PubMed] [Google Scholar]
- 51.Arrindell EL, Jr, Krishnan R, van der Merwe M, Caminita F, Howard SC, Zhang J, Buddington RK. Lung volume recruitment in a preterm pig model of lung immaturity. Am J Physiol Lung Cell Mol Physiol. 2015;309:L1088–L1092. doi: 10.1152/ajplung.00292.2015. [DOI] [PubMed] [Google Scholar]
- 52.Davies SW, Leonard KL, Falls RK, Jr, Mageau RP, Efird JT, Hollowell JP, Trainor WE, 2nd, Kanaan HA, Hickner RC, Sawyer RG, Poulin NR, Waibel BH, Toschlog EA. Lung protective ventilation (ARDSNet) versus airway pressure release ventilation: ventilatory management in a combined model of acute lung and brain injury. J Trauma Acute Care Surg. 2015;78:240–249. doi: 10.1097/TA.0000000000000518. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Kawaguchi A, Guerra GG, Duff JP, Ueta I, Fukushima R (2014) Hemodynamic changes in child acute respiratory distress syndrome with airway pressure release ventilation: a case series. Clin Respir J. doi:10.1111/crj.12155 [DOI] [PubMed]
- 54.Kollisch-Singule M, Emr B, Smith B, Roy S, Jain S, Satalin J, Snyder K, Andrews P, Habashi N, Bates J, Marx W, Nieman G, Gatto LA. Mechanical breath profile of airway pressure release ventilation: the effect on alveolar recruitment and microstrain in acute lung injury. JAMA Surg. 2014;149:1138–1145. doi: 10.1001/jamasurg.2014.1829. [DOI] [PubMed] [Google Scholar]
- 55.Kollisch-Singule M, Emr B, Smith B, Ruiz C, Roy S, Meng Q, Jain S, Satalin J, Snyder K, Ghosh A, Marx W, Andrews P, Habashi N, Nieman G, Gatto LA. Airway pressure release ventilation reduces conducting airway micro-strain in lung injury. J Am Coll Surg. 2014;219:9. doi: 10.1016/j.jamcollsurg.2014.09.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Kollisch-Singule M, Jain S, Andrews P, Smith BJ, Hamlington-Smith KL, Roy S, DiStefano D, Nuss E, Satalin J, Meng Q, Marx W, Bates JH, Gatto LA, Nieman GF, Habashi NM. Effect of airway pressure release ventilation on dynamic alveolar heterogeneity. JAMA Surg. 2016;151(1):64–72. doi: 10.1001/jamasurg.2015.2683. [DOI] [PubMed] [Google Scholar]
- 57.Roy S, Sadowitz B, Andrews P, Gatto LA, Marx W, Ge L, Wang G, Lin X, Dean DA, Kuhn M, Ghosh A, Satalin J, Snyder K, Vodovotz Y, Nieman G, Habashi N. Early stabilizing alveolar ventilation prevents acute respiratory distress syndrome: a novel timing-based ventilatory intervention to avert lung injury. J Trauma Acute Care Surg. 2012;73:391–400. doi: 10.1097/TA.0b013e31825c7a82. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Walsh MA, Merat M, La Rotta G, Joshi P, Joshi V, Tran T, Jarvis S, Caldarone CA, Van Arsdell GS, Redington AN, Kavanagh BP. Airway pressure release ventilation improves pulmonary blood flow in infants after cardiac surgery. Crit Care Med. 2011;39:2599–2604. doi: 10.1097/CCM.0b013e318228297a. [DOI] [PubMed] [Google Scholar]
- 59.Yehya N, Topjian AA, Lin R, Berg RA, Thomas NJ, Friess SH. High frequency oscillation and airway pressure release ventilation in pediatric respiratory failure. Pediatr Pulmonol. 2014;49:707–715. doi: 10.1002/ppul.22853. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Yoshida T, Rinka H, Kaji A, Yoshimoto A, Arimoto H, Miyaichi T, Kan M. The impact of spontaneous ventilation on distribution of lung aeration in patients with acute respiratory distress syndrome: airway pressure release ventilation versus pressure support ventilation. Anesth Analg. 2009;109:1892–1900. doi: 10.1213/ANE.0b013e3181bbd918. [DOI] [PubMed] [Google Scholar]
- 61.Roy SK, Emr B, Sadowitz B, Gatto LA, Ghosh A, Satalin JM, Snyder KP, Ge L, Wang G, Marx W, Dean D, Andrews P, Singh A, Scalea T, Habashi N, Nieman GF. Preemptive application of airway pressure release ventilation prevents development of acute respiratory distress syndrome in a rat traumatic hemorrhagic shock model. Shock. 2013;40:210–216. doi: 10.1097/SHK.0b013e31829efb06. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Emr B, Gatto LA, Roy S, Satalin J, Ghosh A, Snyder K, Andrews P, Habashi N, Marx W, Ge L, Wang G, Dean DA, Vodovotz Y, Nieman G. Airway pressure release ventilation prevents ventilator-induced lung injury in normal lungs. JAMA Surg. 2013;148:1005–1012. doi: 10.1001/jamasurg.2013.3746. [DOI] [PubMed] [Google Scholar]
- 63.(2000) Ventilation with lower tidal volumes as compared with traditional tidal volumes for acute lung injury and the acute respiratory distress syndrome. The acute respiratory distress syndrome network. N Engl J Med 342:1301–1308 [DOI] [PubMed]
- 64.Protti A, Andreis DT, Monti M, Santini A, Sparacino CC, Langer T, Votta E, Gatti S, Lombardi L, Leopardi O, Masson S, Cressoni M, Gattinoni L. Lung stress and strain during mechanical ventilation: any difference between statics and dynamics? Crit Care Med. 2013;41:1046–1055. doi: 10.1097/CCM.0b013e31827417a6. [DOI] [PubMed] [Google Scholar]
- 65.Gattinoni L, Protti A, Caironi P, Carlesso E. Ventilator-induced lung injury: the anatomical and physiological framework. Crit Care Med. 2010;38:S539–S548. doi: 10.1097/CCM.0b013e3181f1fcf7. [DOI] [PubMed] [Google Scholar]
- 66.Retamal J, Bergamini BC, Carvalho AR, Bozza FA, Borzone G, Borges JB, Larsson A, Hedenstierna G, Bugedo G, Bruhn A. Non-lobar atelectasis generates inflammation and structural alveolar injury in the surrounding healthy tissue during mechanical ventilation. Crit Care. 2014;18:505. doi: 10.1186/s13054-014-0505-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Caironi P, Cressoni M, Chiumello D, Ranieri M, Quintel M, Russo SG, Cornejo R, Bugedo G, Carlesso E, Russo R, Caspani L, Gattinoni L. Lung opening and closing during ventilation of acute respiratory distress syndrome. Am J Respir Crit Care Med. 2010;181:578–586. doi: 10.1164/rccm.200905-0787OC. [DOI] [PubMed] [Google Scholar]
- 68.Rose L, Hawkins M. Airway pressure release ventilation and biphasic positive airway pressure: a systematic review of definitional criteria. Intensive Care Med. 2008;34:1766–1773. doi: 10.1007/s00134-008-1216-3. [DOI] [PubMed] [Google Scholar]
- 69.Facchin F, Fan E. Airway pressure release ventilation and high-frequency oscillatory ventilation: potential strategies to treat severe hypoxemia and prevent ventilator-induced lung injury. Respir Care. 2015;60:1509–1521. doi: 10.4187/respcare.04255. [DOI] [PubMed] [Google Scholar]


