Abstract
Sepsis is a complex, heterogeneous physiologic condition that represents a significant public health concern. While many insights into the pathophysiology of sepsis have been elucidated over the past decades of research, important questions remain. This article serves as a review of several important areas in sepsis research. Understanding the innate immune response has been at the forefront as of late, especially in the context of cytokine-directed therapeutic trials. Cellular bioenergetic changes provide insight into the development of organ dysfunction in sepsis. Autophagy and mitophagy perform crucial cell housekeeping and stress response functions. Finally, age-related changes and their potential impact on the septic response are reviewed.
Sepsis poses a considerable burden to the health care system, with nearly 750,000 cases each year in the United States alone and billions in annual costs [1,2]. Case fatality estimates range from 20% to 50%, with increasing incidence and greater proportions of severe sepsis representing a serious public health concern [1–4]. Despite the frequency with which the disease process is encountered, treatment for severe sepsis and septic shock remains limited essentially to supportive measures. The increase of early goal-directed therapy in the past two decades has allowed advances to be made in the resuscitation of patients with sepsis, leading to advantages in mortality and some improvements in long-term organ dysfunction. Recently, the benefits of these measures have been called into question. However, as noted, these treatments are directed at supporting failing organ systems and awaiting recovery of function, rather than addressing directly the root cause of organ dysfunction [5,6]. Recent research has led to a wide array of discoveries in areas related to the biology and immunometabolism of sepsis. Herein we review relevant mechanisms of innate immunity, cellular bioenergetics, and autophagy in sepsis.
Innate Immunity: Pathogen and Cell Damage Pattern Recognition
The body's immune response to infectious insult is divided into two components: The adaptive and innate immunity. Innate immunity functions through a variety of pattern recognition receptors (PRR) that recognize microbial as well as host-derived molecules and subsequently ignite a cascade of immune responses within the cell. Pattern recognition receptors have been characterized as either membrane-bound or cytoplasmic with regards to their orientation on or within the cell: Toll-like receptor (TLR), C-type lectin receptor (CLR), nucleotide-binding oligomerization domain (NOD)-like receptors (NLR), retinoic acid-inducible gene I (RIG-I)–like receptors (RLR), and others [7,8]. This organization is based, in part, on the pathogen-associated molecular pattern (PAMP) to which they are cognate. Toll-like receptor 4 responding to lipopolysaccharide (LPS) of gram-negative organisms is embedded in the cell membrane surface. In contrast, other PRR are located in endosomal membranes to detect nucleic acids or the cytosol to detect viral-associated nucleic acid PAMPs [7,9]. There are numerous examples of PAMPs, ranging from viral deoxyribonucleic acid/ribonucleic acid (DNA/RNA) to bacterial cell surface lipoproteins, LPS, bacterial nucleic acids, and many other molecules [7,8]. Recognition of these conserved molecular patterns is key for the activation of the innate immune response by pathogens and vital for survival.
The functions of PRR on immune and non-immune cells are only partially understood. Toll-like receptor 4 is one of the best studied because of its role in LPS recognition. Mice rendered genetically deficient in TLR4 expression have been shown to be “resistant” to LPS administration yet susceptible to gram-negative sepsis [10]. Toll-like receptor 4 has been shown to have a key role in regulating LPS and bacterial clearance in models of sepsis, although the role of TLR4 in inflammatory regulation varies by cell type [11]. In experiments by Deng et al. [11], hepatocytes were found to clear LPS in a TLR4-dependent manner, whereas leukocytes participated in the septic response by phagocytosis and clearance of bacteria after TLR4 activation. Interestingly, TLR4 has been shown to induce not only pro-inflammatory but also anti-inflammatory responses and may thus serve a role in “balancing” the inflammatory response to LPS [12]. It is possible to pharmacologically block the TLR4 receptor in experimental models of sepsis, which in turn blocks activation of nuclear factor kappa B (NF-κB) and downregulates interleukin (IL)-6 and tumor necrosis factor-α (TNF-α) production both in vitro and in vivo. Efforts have been made to test anti-TLR4 agents in patients with severe sepsis using the investigational drug eritoran to block LPS binding at the TLR4 receptor; however, a large multi-national clinical trial unfortunately did not yield any difference in 28-d mortality between experimental and control groups [13–15]. This is explained potentially by varied and cell-specific roles of TLR4 in sepsis and by the contributions of many other PRR and PAMP/damage-associated molecular patterns (DAMPs) in driving the host response to sepsis.
No discussion of PAMPs and activation of the innate immunity would be complete without also mentioning the topic of endogenous DAMPs. These molecules are “normally” unavailable to the immune system and originate from various intra-cellular and extra-cellular compartments in response to systemic stresses including sepsis, trauma, or ischemia. Damage-associated molecular patterns originate from the nucleus (e.g., DNA, high-mobility group box 1 protein [HMGB1]), cytoplasm (RNA), the extra-cellular matrix (hyaluronic acid, heparin sulfate), or from other organelles (mitochondrial deoxyribonucleic acid [mtDNA], andenosine triphosphate [ATP]), either from direct damage to the cell or from cellular secretion during periods of stress [7,16–18]. These molecules have many shared receptors with PAMPs and are known to stimulate TLR, CLR, and receptors of advanced glycosylation end products (RAGE), among others [7,16–18]. Thus, the inflammatory response in sepsis can be attributed to PAMP stimulation of the innate immune system initially, but it stands to reason that the host gains additional signaling from DAMPs after stress and tissue damage has occurred.
One of the best-studied DAMPs is the nuclear-derived HMGB1. High-mobility group box 1 protein is a DAMP released as a late mediator of sepsis or endotoxemia and is conserved highly in mammals. High-mobility group box 1 protein is also released after cells sustain damage in sterile inflammatory states such as trauma [19,20]. High-mobility group box 1 protein is capable of producing a robust inflammatory response independent of LPS when administered to experimental subjects [20,19]. Similar effects have been reported in both rodent sepsis models, as well as human patients with sepsis [19]. High-mobility group box 1 protein activates signaling through TLR4, TLR2, TLR9, and RAGE receptors and serves to stimulate cytokine production and function as a chemo-attractant [20]. The receptor specificity of extra-cellular HMGB1 is determined by the redox states of its three conserved cysteines, which in turn can regulate its potential binding partners [20]. The similar inflammatory responses when TLR4 is stimulated either by LPS (a PAMP) or disulfide HMGB1 (a DAMP) suggests that a final common pathway exists between infectious and non-infectious (i.e., trauma, ischemia) etiologies of cellular stress [21].
Other examples to be considered are nucleic acid receptors such as TLR3 and TLR9, which recognize microbial nucleic acids and RNA or DNA from cell necrosis to drive an inflammatory response [22–24]. Just as PAMP-induced inflammation can lead to tissue damage and DAMP release, it is also reasonable to consider that DAMP-modulated inflammation can cause epithelial and endothelial “barrier failure,” which may lead to invasion of pathogens and subsequent additional immune stimulation from PAMPs [18]. A final area to be explored is the anti-inflammatory regulatory response. Immune cells have been shown to become tolerant of PAMP/DAMP stimulation, and intra-cellular activation of anti-inflammatory pathways equally serves to decrease cytokine secretion and stimulate the release of anti-inflammatory mediators such as resolvins [18,25]. Neuroendocrine stimulation by inflammatory cytokines and PAMPs can activate anti-inflammatory pathways such as the hypothalamic-pituitary-adrenal axis to stimulate release of cortisol and catecholamines, which modulate immunosuppression [18,25]. In addition, vagus nerve release of acetylcholine can stimulate cholinergic receptors on macrophages to inhibit the release of pro-inflammatory cytokines [25]. It is important to bear in mind that regulation of the pro-inflammatory and anti-inflammatory homeostatic balance is essential; dysregulation of this process has been attributed by many to be the driving force behind the development of sepsis and septic shock. These neural anti-inflammatory pathways are also important to consider when examining in vitro models of sepsis, cell cultures lack the complex feedback loops that are present in vivo and result in considerably elevated non-physiologic cytokine responses when stimulated by PAMPs/DAMPs [18].
Cellular Bioenergetics in Sepsis
A variety of energetic alterations have been noted in sepsis and other inflammatory states. Sepsis is characterized by an initial hypermetabolic state with elevated respiration, ATP production, and endocrine hormone release. However, a hypometabolic state eventually ensues that manifests with decreased mitochondrial respiration, ATP production, and downregulated and/or hormone-resistant endocrine pathways [26,27]. Singer et al. [28–31] propose a hypometabolic theory of sepsis wherein cells enter a “hibernation”-like state that may serve as a protective mechanism that is vital for recovery. This hibernation may protect the cell from ATP depletion by lowering the cell's metabolic demands in cellular environments in which ATP production is impaired [29].
Several mechanisms are known to contribute to the cell's decreased ability to produce ATP during sepsis. Mitochondria are responsible for the production of the vast majority of ATP in the resting state. Inflammatory molecules present during sepsis such as nitric oxide and other reactive oxygen species (ROS) impair the mitochondrial respiratory protein complex and mitochondrial respiration [29,32]. Additionally, lower metabolic rates in sepsis have been associated with decreased quantities of mitochondrial DNA and a resulting deficit in oxidative phosphorylation capacity [33]. Gene transcription of proteins necessary for mitochondrial respiration is downregulated [31,34,35]. Furthermore, hormonal alterations may also contribute to the decreased capacity to produce ATP via oxidative phosphorylation [28]. Lee and Hüttemann [36] have proposed that mitochondrial dysfunction in inflammation and sepsis is driven by successive changes in the phosphorylation states of cytochrome c oxidase and other mitochondrial proteins that decrease protein activity and impair oxidative phosphorylation.
Although respiration is impaired, cells retain other mechanisms of maintaining energy production. In cecal ligation and puncture models of sepsis, hepatocytes show increases in anaerobic metabolism, especially in pathways processing proteins and fatty acids [37]. Overall ATP concentrations were unchanged in hepatocytes during these experiments, but a notable increase in adenosine monophosphate (AMP) was observed, which may be important in cellular signaling from both a metabolic and inflammatory standpoint [37]. Also noted were increases in tricarboxylic acid (TCA) cycle metabolites [37]. These TCA cycle intermediates play a role in the regulation of inflammation and production of pro-inflammatory molecules [38]. In immune cells there have been well-characterized shifts in metabolic pathways, based on substrate availability, which promote maintenance of immune system function under stress [39].
Tissue ATP concentrations have been shown to be variable in patients with sepsis. Patients who died from severe sepsis were shown to have decreased muscle ATP content as opposed to survivors who maintained or increased their ATP concentrations [40]. Although in the past it has been proposed that decreases in oxygen or metabolic substrate supply were the rate-limiting factors in energy production, this does not appear to be the case, because mitochondria retain the ability to utilize a variety of substrates in energy production [41]. Additionally, cells do not suffer from a lack of oxygen supply at the tissue level during sepsis and tissue consumption of oxygen has been shown to decrease as sepsis severity increases [31,42]. Further studies by Fink et al. [32] indicate that the oxygen supply is not the issue in sepsis, rather the problem lies in the cell/mitochondria's ability to utilize the oxygen that is present, which has been coined “cytopathic hypoxia.” Fink et al. [32] further detail that this process is typically not present in the early stages of sepsis and tends to appear after hours have passed. They propose that this may explain some of the therapeutic benefits observed in the “early goal-directed therapy” for sepsis [32].
Disruptions in cell bioenergetics have been implicated in the pathogenesis of sepsis-induced organ failure, which itself is a strong predictor of mortality [2]. The exact mechanism behind the observed hypometabolic state is still a subject of debate, but may involve a combination of impairment of the oxidative phosphorylation apparatus, damage to mtDNA, and increases in cellular protective pathways such as autophagy.
Autophagy and its Role in Cell Maintenance
Autophagy is a cytoprotective process that allows cells to degrade and recycle biomolecules, especially under times of stress [43–48]. Literally meaning “self-eating,” it has been described classically as a response to times of nutrient starvation wherein the cell can maintain critical cell processes by recycling cell components [43–45]. The process is evolutionarily conserved across all eukaryotes, and the materials processed range from individual molecules to entire organelles [43,44]. In addition to its role in energy conservation during stress, autophagy also serves an important role in immune activation via both the innate and adaptive immune systems [43,48]. Autophagy regulates the immune response in multiple ways, from assisting in antigen presentation to lymphocyte differentiation to regulation of cytokine production [43,48]. Autophagy also serves a role in removal of intra-cellular pathogens [43,46,48].
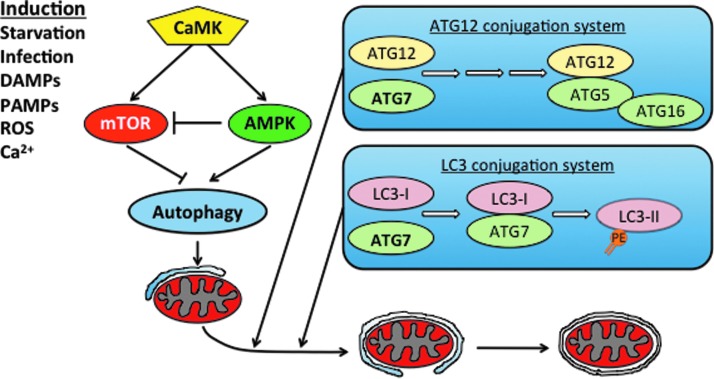
The morphologic hallmark of autophagy in the cell is the formation of a double membrane vesicle termed an autophagosome (Fig. 1) [43,44,48]. These structures form de novo, as opposed to budding off of existing membranes, and the process is initiated at sites called phagopore assembly sites. After initiation, a growth step of phagopore elongation occurs during which the membrane grows around the desired target of degradation [43,47]. Molecular signaling to initiate autophagy involves the target of rapamycin (TOR) protein, a serine/threonine kinase that is intrinsically active and inhibits autophagy under nutrient-rich conditions [43,46] This molecule serves as a vital sensor of the nutritional state of the cell and becomes inhibited under nutrient-poor conditions, at which point autophagy is promoted [46]. The mammalian target of rapamycin (mTOR) is known to be a sensor of nitrogen/amino acids and is also regulated by protein kinase A and adenosine monophosphate-activated protein kinase [47]. Adenosine monophosphate-activated protein kinase (AMPK) serves as an energy rheostat and is sensitive to the ratio of AMP to ATP in the cell. Adenosine monophosphate-activated protein kinase functions to inhibit mTOR directly and indirectly [47]. Inhibiting mTOR decreases the phosphorylation concentration of autophagy-specific protein (Atg) 13, which then interacts with Atg1 and a series of additional Atg proteins to initiate autophagy [46]. The formation of phagopores is also upregulated by phosphatidylinositol 3-kinase VPS34, which forms a complex with beclin-1 (another autophagy-inducing factor) to promote formation of phagopores [43].
FIG. 1.
Autophagy is inhibited by the metabolic growth rheostat mammalian target of rapamycin (mTOR) that inhibits autophagy when adenosine triphosphate (ATP) and nutrients are plentiful. Sepsis and metabolic stress lead to inhibition of mTOR and initiation of the isolation membrane. Atg12-Atg5-Atg16L and LC3-II localize to the phagopore to capture selected targets for degradation. Upon completion of autophagosome formation, the Atg12-Atg5-Atg16L complex dissociates while LC3-II remains on it. The autophagosome ultimately fuses with the lysosome to form an autolysosome, where its contents are degraded. DAMPs = damage-associated molecular patterns; PAMPs = pathogen-associated molecular patterns; ROS = reactive oxygen species; CaMK = calcium/calmodulin-dependent protein kinases; AMPK = adenosine monophosphate-activated protein kinase. Color image is available online at www.liebertpub.com/sur
After initiation of autophagy, two molecular pathways have been described that promote phagopore elongation: (1) the Atg12-Atg5-Atg16 complex and (2) the Atg8-LC3 system [47]. Both of these pathways are dependent on Atg7 and involve ubiquitin-like proteins (UBL) during phagopore expansion [47]. The phosphatidylethanolamine-conjugated LC3-II protein is a marker associated with completed autophagosomes and can be a useful experimental marker of autophagy [49]. The expanding membrane eventually fuses with itself and envelops the material to be degraded [47].
The completed autophagosome will be directed toward a lysosome in a microtubule-dependent manner and then will fuse to form an autolysosome, within which the biomaterials are degraded [47]. The fusion mechanism of the autophagosome with a lysosome is incompletely understood, but is known to be dependent on the protein VTI1B [47]. The autolysosome maturation process is also enhanced by VPS34 [43].
Alternative pathways of autophagy activation have been described. One of particular interest is that of the calcium/calmodulin-dependent protein kinases (CaMK), which are responsive to varying concentrations of intra-cellular calcium [50]. Stress-induced endoplasmic reticulum calcium release stimulates autophagy by increasing cytosolic calcium concentrations [51,52]. CaMKIα and CaMKIV have been shown previously to regulate inflammation in LPS models of sepsis through HMGB1 shuttling and release [53,54]. Evidence suggests that CaMKIα regulates autophagy in macrophages exposed to LPS via the CaMKKα/β-CaMKIα-AMPK pathway in a manner that is independent of mTOR inhibition [55].
In models of sepsis, autophagy is induced by LPS stimulation of TLR4, suggesting that PRR activation serves both pro- and anti-inflammatory roles while also upregulating cytoprotective pathways [12,43]. Other TLR such as TLR3 and TLR7 can also be activated by their respective ligands to induce autophagy [43]. After TLRs are stimulated, they activate both NF-κB and the mitogen-activated protein kinases (MAPK), the former serving to augment the inflammatory response whereas the latter have varying pro- or anti-autophagy effects.
NF-κB upregulates pro-inflammatory cytokine production while down-regulating autophagy [43]. The MAPK JNK, also activated by TLR, phosphorylates Bcl-2, releasing it from the Bcl-2/beclin-1 autophagy inhibitory complex and promoting autophagy [43,56]. Lipopolysaccharide stimulation of TLR4 also induces autophagy via a p38 MAPK/VPS34 signaling pathway that activates heme oxygenase-1 (HO-1) [57]. The upregulation during sepsis of HO-1 as an anti-inflammatory molecule not only induces autophagy as described above but also produces carbon monoxide, which has been shown to decrease nitric oxide (NO) synthesis by inhibiting inducible nitric oxide synthase (iNOS) dimer formation [58]. Nitric oxide has been shown to be an inhibitor of autophagy by several mechanisms [59]. Thus, activation of TLRs of the innate immune system serves to initiate inflammation but also simultaneously induces autophagy pathways to aid in cell recovery.
Although we have presented autophagy primarily as a beneficial adaptive mechanism, it is important to note that its prolonged or inappropriate activation may have deleterious effects as well. Autophagy can serve as a cytoprotective process against apoptosis, however, it can also degrade a large enough portion of the cellular components that cell death ensues, a process called pyroptosis [60]. In cancer research, autophagy has been documented to have roles in both oncogenesis and tumor suppression [48]. Defective autophagy has been implicated in the development of neurodegenerative diseases, cystic fibrosis, and some myopathies [48].
Hypometabolism and Organ Failure in Sepsis
Organ failure is the leading cause of death because of severe sepsis, and the risk of mortality increases with each successive system failure [2]. Patients with organ dysfunction or failure who do not die may still not have a rapid return to normal function; these patients are at risk of developing a state of “persistent critical illness” with organ dysfunction that can persist up to a month or longer from the initial presentation [18,61]. Organ failure may in part be because of impaired perfusion, with alterations in vascular tone and capillary plugging by circulating erythrocytes, leukocytes, and platelets. However, the early organ failure of sepsis typically occurs without grossly evident histologic changes [31,32]. Histologic examination of tissue samples from failing organs in patients with sepsis demonstrate preservation of the architecture with minimal amounts of apoptosis, necrosis, or cell damage [18,28–31,62]. It has been proposed that the tissue changes leading to organ failure are “functional rather than structural” [28]. As noted previously, Singer et al. [28–31] have published extensively regarding the theory that cells enter a hibernation-like state as a protective mechanism, which clinically manifests as organ dysfunction. Others have also reported that cells under the stress of sepsis tend to revert to behaving similar to “unicellular organisms.” During periods of stress the cells decrease their metabolic activity, and thus their phenotype, to preserve themselves at the expense of no longer maintaining specialized tissue function [18]. As discussed earlier, metabolic substrates and oxygen are readily available in sepsis, making it unlikely that the organ dysfunction of sepsis is a consequent of inadequate substrate supply [30,32,41,42]. Noted is the observation that eventually oxidative phosphorylation is decreased after the onset of sepsis. However, it has not been proven whether this is because of oxidative damage to mitochondria or a decrease in mitochondrial activity because of increased cytoprotective pathways (e.g., autophagy/mitophagy) [30,32,63]. Long-term organ dysfunction/failure may be a result of exceeding the maximum duration that cells can tolerate a hypometabolic state; however, the exact mechanism requires further experimental delineation. The balance between mitochondrial damage/removal and mitochondrial biogenesis is likely to play an important role.
Mitophagy: Specialized Autophagy
Mitochondria are the primary consumers of cellular oxygen and produce the vast majority of ATP under aerobic circumstances [31]. However, their role extends beyond the classically taught “powerhouse” function. They also serve to generate ROS, produce heat, initiate cell apoptosis or necrosis, and play a vital role in cellular calcium homeostasis [31,64]. In previous sections we discussed the bioenergetic alterations that occur during sepsis, including hypometabolism and autophagy. Mitophagy is a specialized form of autophagy for mitochondria that serves roles in normal organelle turnover, cell maturation, and removal of damaged mitochondria during periods of stress [47]. As mitochondria become damaged they become a potential source of harm for the cell, as the mitochondrial membrane potential (MMP) can be lost, which leads to opening of mitochondrial transition permeability pores (MTP) [18,65,66]. Release of mitochondrial DAMPs such as mitochondrial DNA drives the innate immune response and can contribute to further inflammation and injury [66–68]. Other DAMPs, such as cytochrome c, can be released that can trigger apoptotic pathways [18,65,68].
Depolarization of the MMP, as occurs in periods of increased oxidative stress such as sepsis, is a strong signal for mitophagy to occur [65,69]. Calcium concentrations in the cell can vary because of oxidative stress, and the loss of the MMP because of this stress impairs the mitochondria's ability to regulate calcium homeostasis [70–72]. Increased cytoplasmic concentrations of calcium can then induce retrograde signaling to the nucleus, upregulating NF-κB in a calcineurin-dependent fashion, which enhances the pro-inflammatory cascade [71]. Mitophagy has also been shown to be activated by Sirt1, a NAD+-dependent deacetylase from the sirtuin family of proteins, which is in turn is activated by the TCA cycle intermediate NAD+ [38].
Two pathways of mitophagy regulation merit further discussion: The PINK1/Parkin pathways and the Nix/BNIP3 pathway [47,65,69]. The PINK1/Parkin pathway is activated during times of oxidative stress and damage to mitochondria [47,65,69]. The mechanism involves the binding of PINK1 to the outer mitochondrial membrane after the MMP has been reduced [47,65,69]. PINK1 then interacts with Parkin after accumulating on the outer mitochondrial membrane, and Parkin then facilitates ubiquitination of the damaged mitochondria and mitophagy [69]. In contrast, Nix/BNIP3L and BNIP3 are proteins that are located on the outer mitochondrial membrane, which can bind to LC3 or gamma-aminobutyric acid receptor-associated protein (GABARAP) on the developing autophagosomes to initiate mitophagy [69]. These mitophagy receptors on the outer mitochondrial membrane are believed to serve more of a role in steady-state mitochondrial maintenance and turnover and in cell maturation, such as with maturing erythrocytes [47,69].
Although it will not be discussed in detail here, it should be noted that mitochondrial fission and fusion also play a role in maintenance of a healthy mitochondrial population [69]. These pathways can protect against excessive mitophagy and perhaps provide some safeguard against cell death [43,69]. The removal of damaged or dysfunctional mitochondria by mitophagy may be part of the explanation for the observed hypometabolic state in sepsis, which is in turn counteracted by the process of generating new mitochondria for the cell: Mitochondrial biogenesis.
Mitochondrial Biogenesis
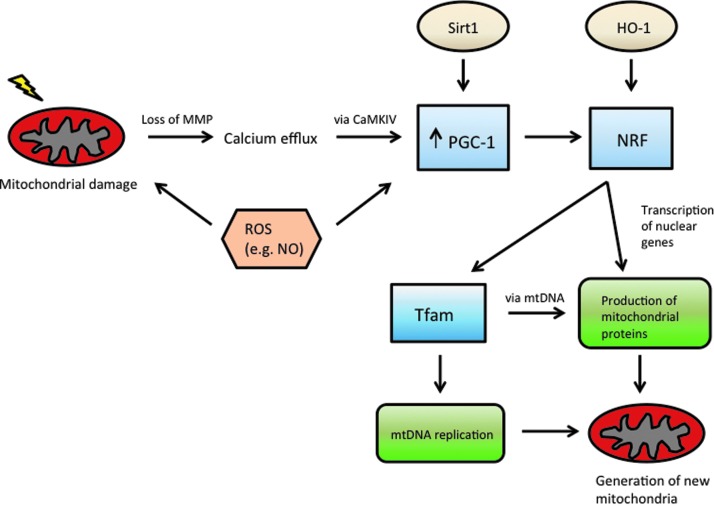
Mitochondrial biogenesis (Fig. 2) is the process of generating new mitochondria and occurs as a response to sepsis-induced mitochondrial damage [33]. Furthermore, the recovery of respiratory capability and increased metabolic function is linked with molecular activation of mitochondrial biogenesis [33,63]. Regeneration of mitochondria requires the collective and integrated function of proteins encoded in the cell's nuclear genome, proteins encoded in the separate mitochondrial DNA, and signaling between the nucleus and mitochondria [33,67].
FIG. 2.
Mitochondrial biogenesis. Damage to mitochondria by reactive oxygen species sets off a cascade to generate new mitochondria, which is dependent on activation of both nuclear and mitochondrial genes. MMP = mitochondrial membrane potential; ROS = reactive oxygen species; HO-1 = heme oxygenase-1; NRF = nuclear respiratory factor; Tfam = mitochondrial transcription factor A; mtDNA = mitochondrial deoxyribonucleic acid. Color image is available online at www.liebertpub.com/sur
Peroxisome proliferator-activated receptor-γ coactivator-1 alpha (PGC-1α) is a protein essential in regulating mitochondrial biogenesis, and its upregulation accelerates recovery of mitochondrial function [73]. Indeed, PGC-1α has been regarded as a “master regulator” of mitochondrial biogenesis and it stimulates nuclear respiratory factor 1 (NRF-1) and NRF-2 expression [74,75]. It has also been noted that greater capability for mitochondrial biogenesis, measured by PGC-1α concentrations, is associated with increased survival in sepsis [31,35]. The nuclear respiratory factors in turn promote transcription of nuclear mitochondrial genes and production of mitochondrial transcription factor A (Tfam) and B, on which mitochondrial DNA (mtDNA) replication and transcription are dependent [33,74,75].
Mitochondrial biogenesis is induced by nitric oxide, which increases PGC-1α and NRF-1 [29,71,75]. In an LPS model of sepsis, mice injected with LPS demonstrated a TLR4-mediated increase in iNOS [76]. Interestingly, NO plays a role both in inflammatory damage to mitochondria, as well as, signaling the upregulation of mitochondrial biogenesis; therefore, it is at least partially responsible for both the decrease in mitochondria populations because of oxidative stress as well as in recovery from the injury [31,63,76]. In addition to TLR4, mitochondrial biogenesis is dependent on TLR9 recognition of mitochondrial DNA in hepatocytes [77]. Other mediators from the TCA cycle such as NAD+ induce Sirt1, which upregulates mitochondrial biogenesis in addition to its previously discussed role of inducing mitophagy [38]. Induction of HO-1 has also been associated with upregulation of mitochondrial biogenesis, adding to its roles in regulating autophagy and the inflammatory response [31]. Calcium concentration fluctuations because of cell stress also regulate mitochondrial biogenesis via CaMKIV-induced expression of PGC-1α [78]. Thus, many of the previously mentioned molecules that serve regulatory functions in the inflammatory response also serve as an impetus for the cell to initiate cellular repair via mitochondrial biogenesis.
Aging and the Response to Sepsis
Increasing age is a risk factor for the development of sepsis as well as mortality because of sepsis. The elderly represent a large proportion of patients admitted with sepsis in the United States (approximately two thirds) [1]. Age-related decline in mitochondrial respiratory function has been well described [79]. Analysis of transcriptional profiles in Drosophila, mice, and human beings reveals decreased expression of the electron transport chain with age [80]. Others have examined mitochondrial numbers directly and noted a similar decrease with age, which correlates with the decreased capacity for oxidative phosphorylation and decreased gene expression [81]. Why this occurs remains to be determined.
A proposed theory of the mechanisms of aging centers on mitochondria sustaining damage from ROS, thereby accumulating mitochondrial DNA mutations in a repetitive fashion. This has been termed the “mitochondrial free radical theory of aging” [79,81,82]. This theory has been discussed and explored over the past half-century, leading to several notable modifications or alternate theories. One study suggests that one way cells deal with ROS is a mild level of uncoupling of the oxidative phosphorylation chain, which decreases ROS production. Uncoupling was noted to occur at a greater concentration in muscle tissues that were more active and maintained function longer into old age; this contrasted with less active muscle tissues, which showed less uncoupling and a greater decrease in respiratory function with increasing age [83]. However, other recent reviews have minimized the role of ROS as a driving factor in aging and instead point to clonal expansion of somatic mitochondrial DNA mutations creating mosaic respiratory chain dysfunction in aging tissues [81]. Still others have concluded that a low concentration of heteroplasmy of mitochondrial DNA exists at baseline and natural selection occurs in mitochondria as a consequence of stress events [84]. It is suggested that non-lethal insults select mitochondria that are more resilient to subsequent challenges. This leads to the hypothesis that intermittent insults benefit mitochondrial health and promote healthy aging and increased longevity [84].
Autophagy changes and their association with aging is another area that has been investigated. Augmenting or promoting autophagy in experimental models has enhanced fitness and longevity, whereas diminishing autophagy yields an increase in pro-aging pathways [48]. Decreases in autophagy and mitochondrial biogenesis have been noted to occur with age [68]. Our group has noted that experimental animals of increasing age exhibit a decreased capacity for autophagy/mitophagy, with insufficient induction of cytoprotective pathways under periods of stress [85]. We observed that young and old mice to have similar concentrations of basal autophagy, but after septic insult younger mice were more likely to recover from acute kidney injury and showed greater experimental markers of autophagy [85]. Inhibiting autophagy in young mice caused them to have a pattern of acute kidney injury similar to old mice [85]. Older mice treated pharmacologically with temsirolimus to augment autophagy had patterns of autophagy and acute kidney injury that were similar to that of the young mice [85]. Certainly there are great opportunities for research and discovery of potential therapeutics that augment autophagy or mitochondrial biogenesis in an effort to relieve some of the physiologic stresses of sepsis.
Acknowledgment
This work was supported by NIH Grant ROI GM082852.
Author Disclosure Statement
No competing financial interests exist.
References
- 1.Angus DC, Linde-Zwirble WT, Lidicker J, et al. Epidemiology of severe sepsis in the United States: Analysis of incidence, outcome, and associated costs of care. Crit Care Med 2001;29:1303–1310 [DOI] [PubMed] [Google Scholar]
- 2.Martin GS, Mannino DM, Eaton S, Moss M. The epidemiology of sepsis in the United States from 1979 through 2000. N Engl J Med 2003;348:1546–1554 [DOI] [PubMed] [Google Scholar]
- 3.Dombrovskiy VY, Martin AA, Sunderram J, Paz H. Rapid increase in hospitalization and mortality rates for severe sepsis in the United States: A trend analysis from 1993 to 2003. Crit Care Med 2007;35:1244–1250 [DOI] [PubMed] [Google Scholar]
- 4.Kumar G, Kumar N, Taneja A, et al. Nationwide trends of severe sepsis in the 21st century (2000–2007). Chest 2011;140:1223–1231 [DOI] [PubMed] [Google Scholar]
- 5.Rivers E, Nguyen B, Havstad S, et al. Early goal-directed therapy in the treatment of severe sepsis and septic shock. N Engl J Med 2001;345:1368–1377 [DOI] [PubMed] [Google Scholar]
- 6.Otero RM, Nguyen HB, Huang DT, et al. Early goal-directed therapy in severe sepsis and septic shock revisited. Chest 2006;130:1579–1595 [DOI] [PubMed] [Google Scholar]
- 7.Iwasaki A, Medzhitov R. Regulation of adaptive immunity by the innate immune system. Science 2010;327:291–295 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Broz P, Monack DM. Newly described pattern recognition receptors team up against intracellular pathogens. Nat Rev Immunol 2013;13:551–565 [DOI] [PubMed] [Google Scholar]
- 9.Carpenter S, O'Neill LA. How important are toll-like receptors for antimicrobial responses? Cell Microbiol 2007;9:1891–1901 [DOI] [PubMed] [Google Scholar]
- 10.Poltorak A, He X, Smirnova I, et al. Defective LPS signaling in C3H/HeJ and C57BL/10ScCr mice: Mutations in Tlr4 gene. Science 1998;282:2085–2088 [DOI] [PubMed] [Google Scholar]
- 11.Deng M, Scott MJ, Loughran P, et al. Lipopolysaccharide clearance, bacterial clearance, and systemic inflammatory responses are regulated by cell type-specific functions of TLR4 during sepsis. J Immunol 2013;190:5152–5160 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Waltz PK, Carchman EH, Young AC, et al. Lipopolysaccharide induces autophagic signaling in macrophages via a TLR4, heme oxygenase-1 dependent pathway. Autophagy 2011;7:315–320 [DOI] [PubMed] [Google Scholar]
- 13.Rossignol DP, Wasan KM, Choo E, et al. Safety, pharmacokinetics, pharmacodynamics, and plasma lipoprotein distribution of eritoran (E5564) during continuous intravenous infusion into healthy volunteers. Antimicrob Agents Chemother 2004;48:3233–3240 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Barochia A, Solomon S, Cui X, et al. Eritoran tetrasodium (E5564) treatment for sepsis: Review of preclinical and clinical studies. Expert Opin Drug Metab Toxicol 2011;7:479–494 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Opal SM, Laterre PF, Francois B, et al. Effect of eritoran, an antagonist of MD2-TLR4, on mortality in patients with severe sepsis: The ACCESS randomized trial. JAMA 2013;309:1154–1162 [DOI] [PubMed] [Google Scholar]
- 16.Rubartelli A, Lotze MT. Inside, outside, upside down: Damage-associated molecular pattern molecules (DAMPs) and redox. Trends Immunol 2007;28:429–436 [DOI] [PubMed] [Google Scholar]
- 17.Lotze MT, Zeh HJ, Rubartelli A, et al. The grateful dead: Damage-associated molecular pattern molecules and reduction/oxidation regulate immunity. Immunol Rev 2007;220:60–81 [DOI] [PubMed] [Google Scholar]
- 18.Deutschman CS, Tracey KJ. Sepsis: Current dogma and new perspectives. Immunity 2014;40:463–475 [DOI] [PubMed] [Google Scholar]
- 19.Venereau E, Schiraldi M, Uguccioni M, Bianchi M. HMGB1 and leukocyte migration during trauma and sterile inflammation. Mol Immunol 2013;55:76–82 [DOI] [PubMed] [Google Scholar]
- 20.Wang H, Bloom O, Zhang M, et al. HMG-1 as a late mediator of endotoxin lethality in mice. Science 1999;285:248–251 [DOI] [PubMed] [Google Scholar]
- 21.Andersson U, Tracey KJ. HMGB1 is a therapeutic target for sterile inflammation and infection. Annu Rev Immunol 2011;29:139–162 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Cavassani KA, Ishii M, Wen H, et al. TLR3 is an endogenous sensor of tissue necrosis during acute inflammatory events. J Exp Med 2008;205:2609–2621 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Hemmi H, Takeuchi O, Kawai T, et al. A toll-like receptor recognizes bacterial DNA. Nature 2000;408:740–745 [DOI] [PubMed] [Google Scholar]
- 24.Yasuda K, Yu P, Kirschning CJ, et al. Endosomal translocation of vertebrate DNA activates dendritic cells via TLR9-dependent and -independent pathways. J Immunol 2005;174:6129–6136 [DOI] [PubMed] [Google Scholar]
- 25.Angus DC, van der Poll T. Severe sepsis and septic shock. N Engl J Med 2013;369:840–851 [DOI] [PubMed] [Google Scholar]
- 26.Singer M, Brealey D. Mitochondrial dysfunction in sepsis. Biochem Soc Symp 1999;66:149–166 [DOI] [PubMed] [Google Scholar]
- 27.Van den Berghe G, de Zegher F, Bouillon R. Acute and prolonged critical illness as different neuroendocrine paradigms. J Clin Endocrinol Metab 1998;83:1827–1834 [DOI] [PubMed] [Google Scholar]
- 28.Singer M, De Santis V, Vitale D, Jeffcoate W. Multiorgan failure is an adaptive, endocrine-mediated, metabolic response to overwhelming systemic inflammation. Lancet 2004;364:545–548 [DOI] [PubMed] [Google Scholar]
- 29.Singer M. Mitochondrial function in sepsis: Acute phase versus multiple organ failure. Crit Care Med 2007;35:S441–S448 [DOI] [PubMed] [Google Scholar]
- 30.Carre JE, Singer M. Cellular energetic metabolism in sepsis: The need for a systems approach. Biochim Biophys Acta 2008;1777:763–771 [DOI] [PubMed] [Google Scholar]
- 31.Singer M. The role of mitochondrial dysfunction in sepsis-induced multi-organ failure. Virulence 2014;5:66–72 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Fink MP. Bench-to-bedside review: Cytopathic hypoxia. Crit Care 2002;6:491–499 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Haden DW, Suliman HB, Carraway MS, et al. Mitochondrial biogenesis restores oxidative metabolism during Staphylococcus aureus sepsis. Am J Respir Crit Care Med 2007;176:768–777 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Calvano SE, Xiao W, Richards DR, et al. A network-based analysis of systemic inflammation in humans. Nature 2005;437:1032–1037 [DOI] [PubMed] [Google Scholar]
- 35.Carre JE, Orban JC, Felsmann K, et al. Survival in critical illness is associated with early activation of mitochondrial biogenesis. Am J Respir Crit Care Med 2010;182:745–751 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Lee I, Hüttemann M. Energy crisis: The role of oxidative phosphorylation in acute inflammation and sepsis. Biochim Biophys Acta 2014;1842:1579–1586 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Whelan SP, Carchman EH, Kautza B, et al. Polymicrobial sepsis is associated with decreased hepatic oxidative phosphorylation and an altered metabolic profile. J Surg Res 2014;186:297–303 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Mills E, O'Neill LA. Succinate: A metabolic signal in inflammation. Trends Cell Biol 2014;24:313–320 [DOI] [PubMed] [Google Scholar]
- 39.Pearce EL, Pearce EJ. Metabolic pathways in immune cell activation and quiescence. Immunity 2013;38:633–643 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Brealey D, Brand M, Hargreaves I, et al. Association between mitochondrial dysfunction and severity and outcome of septic shock. Lancet 2002;360:219–223 [DOI] [PubMed] [Google Scholar]
- 41.Leverve XM. Mitochondrial function and substrate availability. Crit Care Med 2007;35:S454–S460 [DOI] [PubMed] [Google Scholar]
- 42.Kreymann G, Grosser S, Buggisch P, et al. Oxygen consumption and resting metabolic rate in sepsis, sepsis syndrome, and septic shock. Crit Care Med 1993;21:1012–1019 [DOI] [PubMed] [Google Scholar]
- 43.Delgado M, Singh S, De Haro S, et al. Autophagy and pattern recognition receptors in innate immunity. Immunol Rev 2009;227:189–202 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Klionsky DJ, Emr SD. Autophagy as a regulated pathway of cellular degradation. Science 2000;290:1717–1721 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Klionsky DJ. Autophagy. Curr Biol 2005;15:R282–R283 [DOI] [PubMed] [Google Scholar]
- 46.Yorimitsu T, Klionsky DJ. Autophagy: Molecular machinery for self-eating. Cell Death Differ 2005;12:1542–1552 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Parzych KR, Klionsky DJ. An overview of autophagy: Morphology, mechanism, and regulation. Antioxid Redox Signal 2014;20:460–473 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Wirawan E, Vanden Berghe T, Lippens S, et al. Autophagy: For better or for worse. Cell Res 2012;22:43–61 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Kabeya Y, Mizushima N, Ueno T, et al. LC3, a mammalian homologue of yeast Apg8p, is localized in autophagosome membranes after processing. EMBO J 2000;19:5720–5728 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Means AR. The year in basic science: Calmodulin kinase cascades. Mol Endocrinol 2008;22:2759–2765 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Nakayama Y, Endo M, Tsukano H, et al. Molecular mechanisms of the LPS-induced non-apoptotic ER stress-CHOP pathway. J Biochem 2010;147:471–483 [DOI] [PubMed] [Google Scholar]
- 52.Høyer-Hansen M, Bastholm L, Szyniarowski P, et al. Control of macroautophagy by calcium, calmodulin-dependent kinase kinase-beta, and Bcl-2. Mol Cell 2007;25:193–205 [DOI] [PubMed] [Google Scholar]
- 53.Zhang X, Guo L, Collage RD, et al. Calcium/calmodulin-dependent protein kinase (CaMK) Iα mediates the macrophage inflammatory response to sepsis. J Leukoc Biol 2011;90:249–261 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Zhang X, Wheeler D, Tang Y, et al. Calcium/calmodulin-dependent protein kinase (CaMK) IV mediates nucleocytoplasmic shuttling and release of HMGB1 during lipopolysaccharide stimulation of macrophages. J Immunol 2008;181:5015–5023 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Guo L, Stripay JL, Zhang X, et al. CAMKIα regulates AMP kinase-dependent, TORC-1-independent autophagy during lipopolysaccharide-induced acute lung neutrophilic inflammation. J Immunol 2013;190:3620–3628 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Wei Y, Pattingre S, Sinha S, et al. JNK1-mediated phosphorylation of Bcl-2 regulates starvation-induced autophagy. Mol Cell 2008;30:678–688 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Carchman EH, Rao J, Loughran PA, et al. Heme oxygenase-1–mediated autophagy protects against hepatocyte cell death and hepatic injury from infection/sepsis in mice. Hepatology 2011;53:2053–2062 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Kim HS, Loughran PA, Billiar TR. Carbon monoxide decreases the level of iNOS protein and active dimer in IL-1beta-stimulated hepatocytes. Nitric Oxide 2008;18:256–265 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Sarkar S, Korolchuk VI, Renna M, et al. Complex inhibitory effects of nitric oxide on autophagy. Mol Cell 2011;43:19–32 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Maiuri MC, Zalckvar E, Kimchi A, Kroemer G. Self-eating and self-killing: Crosstalk between autophagy and apoptosis. Nat Rev Mol Cell Biol 2007;8:741–752 [DOI] [PubMed] [Google Scholar]
- 61.Guirgis FW, Khadpe JD, Kuntz GM, et al. Persistent organ dysfunction after severe sepsis: A systematic review. J Crit Care 2014;29:320–326 [DOI] [PubMed] [Google Scholar]
- 62.Hotchkiss RS, Swanson PE, Freeman BD, et al. Apoptotic cell death in patients with sepsis, shock, and multiple organ dysfunction. Crit Care Med 1999;27:1230–1251 [DOI] [PubMed] [Google Scholar]
- 63.Crouser ED, Julian MW, Huff JE, et al. Carbomoyl phosphate synthase-1: A marker of mitochondrial damage and depletion in the liver during sepsis. Crit Care Med 2006;34:2439–2446 [DOI] [PubMed] [Google Scholar]
- 64.Dedkova EN, Blatter LA. Mitochondrial Ca2+ and the heart. Cell Calcium 2008;44:77–91 [DOI] [PubMed] [Google Scholar]
- 65.Kubli DA, Gustafsson AB. Mitochondria and mitophagy: The yin and yang of cell death control. Circ Res 2012;111:1208–1221 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Nakahira K, Haspel JA, Rathinam VA, et al. Autophagy proteins regulate innate immune responses by inhibiting the release of mitochondrial DNA mediated by the NALP3 inflammasome. Nat Immunol 2011;12:222–230 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.West AP, Shadel GS, Ghosh S. Mitochondria in innate immune responses. Nat Rev Immunol 2011;11:389–402 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Zang QS, Wolf SE, Minei JP. Sepsis-induced cardiac mitochondrial damage and potential therapeutic interventions in the elderly. Aging Dis 2014;5:137–149 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Hammerling BC, Gustafsson AB. Mitochondrial quality control in the myocardium: Cooperation between protein degradation and mitophagy. J Mol Cell Cardiol 2014;75:122–130 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Ermak G, Davies KJ. Calcium and oxidative stress: From cell signaling to cell death. Mol Immunol 2002;38:713–721 [DOI] [PubMed] [Google Scholar]
- 71.Butow RA, Avadhani NG. Mitochondrial signaling: The retrograde response. Mol Cell 2004;14:1–15 [DOI] [PubMed] [Google Scholar]
- 72.Kawamata H, Manfredi G. Mitochondrial dysfunction and intracellular calcium dysregulation in ALS. Mech Ageing Dev 2010;131:517–526 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Rasbach KA, Schnellmann RG. PGC-1alpha over-expression promotes recovery from mitochondrial dysfunction and cell injury. Biochem Biophys Res Commun 2007;355:734–739 [DOI] [PubMed] [Google Scholar]
- 74.Wu Z, Puigserver P, Andersson U, et al. Mechanisms controlling mitochondrial biogenesis and respiration through the thermogenic coactivator PGC-1. Cell 1999;98:115–124 [DOI] [PubMed] [Google Scholar]
- 75.Nisoli E, Clementi E, Paolucci C, et al. Mitochondrial biogenesis in mammals: The role of endogenous nitric oxide. Science 2003;299:896–899 [DOI] [PubMed] [Google Scholar]
- 76.Suliman HB, Welty-Wolf KE, Carraway MS, et al. Toll-like receptor 4 mediates mitochondrial DNA damage and biogenic responses after heat-inactivated E. coli. FASEB J 2005;19:1531–1533 [DOI] [PubMed] [Google Scholar]
- 77.Carchman EH, Whelan S, Loughran P, et al. Experimental sepsis-induced mitochondrial biogenesis is dependent on autophagy, TLR4, and TLR9 signaling in liver. FASEB J 2013;27:4703–4711 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Wu H, Kanatous SB, Thurmond FA, et al. Regulation of mitochondrial biogenesis in skeletal muscle by CaMK. Science 2002;296:349–352 [DOI] [PubMed] [Google Scholar]
- 79.Chan DC. Mitochondria: Dynamic organelles in disease, aging, and development. Cell 2006;125:1241–1252 [DOI] [PubMed] [Google Scholar]
- 80.Zahn JM, Sonu R, Vogel H, et al. Transcriptional profiling of aging in human muscle reveals a common aging signature. PLoS Genet 2006;2:1058–1069 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Bratic A, Larsson NG. The role of mitochondria in aging. J Clin Invest 2013;123:951–957 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Harman D. Aging: A theory based on free radical and radiation chemistry. J Gerontol 1956;11:298–300 [DOI] [PubMed] [Google Scholar]
- 83.Amara CE, Shankland EG, Jubrias SA, et al. Mild mitochondrial uncoupling impacts cellular aging in human muscles in vivo. Proc Natl Acad Sci USA 2007;104:1057–1062 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Rodell A, Rasmussen LJ, Bergerson LH, Singh KK, Gjedde A. Natural selection of mitochondria during somatic lifetime promotes healthy aging. Front Neuroenergetics 2013;5:1–6 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Howell GM, Gomez H, Collage RD, et al. Augmenting autophagy to treat acute kidney injury during endotoxemia in mice. PLoS One 2013;8:1–7 [DOI] [PMC free article] [PubMed] [Google Scholar]