Abstract
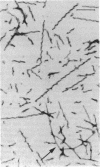
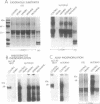
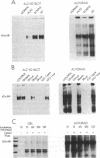
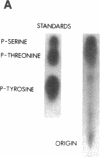
We have identified a protein kinase in immunoaffinity-purified preparations of paired helical filaments from brain tissue of individuals with Alzheimer disease. The kinase phosphorylates the filament proteins in vitro in a manner independent of second messenger regulation or of modulation by heparin and polyamines. Physiological concentrations of hemin, an oxidized heme porphyrin, inhibit the kinase and abolish Alz-50 immunoreactivity of the proteins. Since paired helical filaments are composed of hyperphosphorylated proteins, association of a protein kinase with the filaments provides a mechanism for abnormal processing of the proteins in disease.
Full text
PDF



Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Braak H., Braak E., Grundke-Iqbal I., Iqbal K. Occurrence of neuropil threads in the senile human brain and in Alzheimer's disease: a third location of paired helical filaments outside of neurofibrillary tangles and neuritic plaques. Neurosci Lett. 1986 Apr 24;65(3):351–355. doi: 10.1016/0304-3940(86)90288-0. [DOI] [PubMed] [Google Scholar]
- Ghanbari H. A., Miller B. E., Haigler H. J., Arato M., Bissette G., Davies P., Nemeroff C. B., Perry E. K., Perry R., Ravid R. Biochemical assay of Alzheimer's disease--associated protein(s) in human brain tissue. A clinical study. JAMA. 1990 Jun 6;263(21):2907–2910. [PubMed] [Google Scholar]
- Greenberg S. G., Davies P. A preparation of Alzheimer paired helical filaments that displays distinct tau proteins by polyacrylamide gel electrophoresis. Proc Natl Acad Sci U S A. 1990 Aug;87(15):5827–5831. doi: 10.1073/pnas.87.15.5827. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Greenberg S. G., Davies P., Schein J. D., Binder L. I. Hydrofluoric acid-treated tau PHF proteins display the same biochemical properties as normal tau. J Biol Chem. 1992 Jan 5;267(1):564–569. [PubMed] [Google Scholar]
- Gross M. The control of protein synthesis by hemin in rabbit reticulocytes. Mol Cell Biochem. 1980 May 28;31(1):25–36. doi: 10.1007/BF00817888. [DOI] [PubMed] [Google Scholar]
- Grundke-Iqbal I., Iqbal K., Tung Y. C., Quinlan M., Wisniewski H. M., Binder L. I. Abnormal phosphorylation of the microtubule-associated protein tau (tau) in Alzheimer cytoskeletal pathology. Proc Natl Acad Sci U S A. 1986 Jul;83(13):4913–4917. doi: 10.1073/pnas.83.13.4913. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hathaway G. M., Traugh J. A. Casein kinases--multipotential protein kinases. Curr Top Cell Regul. 1982;21:101–127. [PubMed] [Google Scholar]
- Hyman B. T., Van Hoesen G. W., Wolozin B. L., Davies P., Kromer L. J., Damasio A. R. Alz-50 antibody recognizes Alzheimer-related neuronal changes. Ann Neurol. 1988 Apr;23(4):371–379. doi: 10.1002/ana.410230410. [DOI] [PubMed] [Google Scholar]
- Iqbal K., Zaidi T., Thompson C. H., Merz P. A., Wisniewski H. M. Alzheimer paired helical filaments: bulk isolation, solubility, and protein composition. Acta Neuropathol. 1984;62(3):167–177. doi: 10.1007/BF00691849. [DOI] [PubMed] [Google Scholar]
- Ishiguro K., Omori A., Sato K., Tomizawa K., Imahori K., Uchida T. A serine/threonine proline kinase activity is included in the tau protein kinase fraction forming a paired helical filament epitope. Neurosci Lett. 1991 Jul 22;128(2):195–198. doi: 10.1016/0304-3940(91)90259-v. [DOI] [PubMed] [Google Scholar]
- KIDD M. Paired helical filaments in electron microscopy of Alzheimer's disease. Nature. 1963 Jan 12;197:192–193. doi: 10.1038/197192b0. [DOI] [PubMed] [Google Scholar]
- Kosik K. S., Orecchio L. D., Binder L., Trojanowski J. Q., Lee V. M., Lee G. Epitopes that span the tau molecule are shared with paired helical filaments. Neuron. 1988 Nov;1(9):817–825. doi: 10.1016/0896-6273(88)90129-8. [DOI] [PubMed] [Google Scholar]
- Ksiezak-Reding H., Binder L. I., Yen S. H. Alzheimer disease proteins (A68) share epitopes with tau but show distinct biochemical properties. J Neurosci Res. 1990 Mar;25(3):420–430. doi: 10.1002/jnr.490250320. [DOI] [PubMed] [Google Scholar]
- Lee V. M., Balin B. J., Otvos L., Jr, Trojanowski J. Q. A68: a major subunit of paired helical filaments and derivatized forms of normal Tau. Science. 1991 Feb 8;251(4994):675–678. doi: 10.1126/science.1899488. [DOI] [PubMed] [Google Scholar]
- Love S., Burrola P., Terry R. D., Wiley C. A. Immunoelectron microscopy of Alzheimer and Pick brain tissue labelled with the monoclonal antibody Alz-50. Neuropathol Appl Neurobiol. 1989 May-Jun;15(3):223–231. doi: 10.1111/j.1365-2990.1989.tb01224.x. [DOI] [PubMed] [Google Scholar]
- Saitoh T., Masliah E., Jin L. W., Cole G. M., Wieloch T., Shapiro I. P. Protein kinases and phosphorylation in neurologic disorders and cell death. Lab Invest. 1991 May;64(5):596–616. [PubMed] [Google Scholar]
- Sloboda R. D., Rudolph S. A., Rosenbaum J. L., Greengard P. Cyclic AMP-dependent endogenous phosphorylation of a microtubule-associated protein. Proc Natl Acad Sci U S A. 1975 Jan;72(1):177–181. doi: 10.1073/pnas.72.1.177. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Spira G., Bargellesi A., Teillaud J. L., Scharff M. D. The identification of monoclonal class switch variants by sib selection and an ELISA assay. J Immunol Methods. 1984 Nov 30;74(2):307–315. doi: 10.1016/0022-1759(84)90298-9. [DOI] [PubMed] [Google Scholar]
- TERRY R. D., GONATAS N. K., WEISS M. ULTRASTRUCTURAL STUDIES IN ALZHEIMER'S PRESENILE DEMENTIA. Am J Pathol. 1964 Feb;44:269–297. [PMC free article] [PubMed] [Google Scholar]
- Tabaton M., Whitehouse P. J., Perry G., Davies P., Autilio-Gambetti L., Gambetti P. Alz 50 recognizes abnormal filaments in Alzheimer's disease and progressive supranuclear palsy. Ann Neurol. 1988 Sep;24(3):407–413. doi: 10.1002/ana.410240309. [DOI] [PubMed] [Google Scholar]
- Trachsel H., Ranu R. S., London I. M. Regulation of protein synthesis in rabbit reticulocyte lysates: purification and characterization of heme-reversible translational inhibitor. Proc Natl Acad Sci U S A. 1978 Aug;75(8):3654–3658. doi: 10.1073/pnas.75.8.3654. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vincent I. J., Davies P. Phosphorylation characteristics of the A68 protein in Alzheimer's disease. Brain Res. 1990 Oct 29;531(1-2):127–135. doi: 10.1016/0006-8993(90)90765-4. [DOI] [PubMed] [Google Scholar]
- Wolozin B. L., Pruchnicki A., Dickson D. W., Davies P. A neuronal antigen in the brains of Alzheimer patients. Science. 1986 May 2;232(4750):648–650. doi: 10.1126/science.3083509. [DOI] [PubMed] [Google Scholar]
- Wolozin B., Davies P. Alzheimer-related neuronal protein A68: specificity and distribution. Ann Neurol. 1987 Oct;22(4):521–526. doi: 10.1002/ana.410220412. [DOI] [PubMed] [Google Scholar]
- Wood J. G., Mirra S. S., Pollock N. J., Binder L. I. Neurofibrillary tangles of Alzheimer disease share antigenic determinants with the axonal microtubule-associated protein tau (tau) Proc Natl Acad Sci U S A. 1986 Jun;83(11):4040–4043. doi: 10.1073/pnas.83.11.4040. [DOI] [PMC free article] [PubMed] [Google Scholar]