Abstract
Papillary craniopharyngioma is an intracranial tumor that results in high levels of morbidity. We recently demonstrated that the vast majority of these tumors harbor the oncogenic BRAF V600E mutation. The pathologic diagnosis of papillary craniopharyngioma can now be confirmed using mutation specific immunohistochemistry and targeted genetic testing. Treatment with targeted agents is now also a possibility in select situations. We recently reported a patient with a multiply recurrent papillary craniopharyngioma in whom targeting both BRAF and MEK resulted in a dramatic therapeutic response with a marked anti-tumor immune response. This work shows that activation of the MAPK pathway is the likely principal oncogenic driver of these tumors. We will now investigate the efficacy of this approach in a multicenter phase II clinical trial. Post-treatment resection samples will be monitored for the emergence of resistance mechanisms. Further advances in the non-invasive diagnosis of papillary craniopharyngioma by radiologic criteria and by cell-free DNA testing could someday allow neo-adjuvant therapy for this disease in select patient populations.
Keywords: Craniopharyngioma, BRAF, dabrafenib, trametinib, VE1, targeted, Rathke’s, cell-free DNA
Background
Craniopharyngiomas are uncommon epithelial neoplasms that arise above the sella turcica of the skull base, in the suprasellar, infundibulotuberal and third ventricular areas of the brain1–3. Despite their benign histologic appearance, these tumors pose many clinical challenges4–6. The tumors arise in proximity to critical structures and can compress or infiltrate these vital neurological areas4–7. Visual defects, pan-hypopituitarism, cognitive deficits, personality changes, hyperphagia and morbid obesity are common complications that result not only from the growth of the tumor but also often as a consequence of treatment with surgery, radiation, or both4–11. Moreover, scarring and reactive changes occur following surgical resection and radiation treatment. As a result, resecting recurrent tumors is fraught with difficulties. Patient management is further complicated by a lack of effective systemic chemotherapies12 and by variations in clinical practice algorithms13.
There are two histopathologic variants of craniopharyngioma. Adamantinomatous craniopharyngioma (ACP) occur in both children and adults and papillary craniopharyngioma (PCP) occur almost exclusively in adults. These variants have distinct histologic features1, 14, 15.
When resection specimens are large and abundant and well-preserved tumor epithelium is present, classification is routine on H&E stained sections1, 14, 15. ACP have epithelium that grows in cords, lobules and whorls, with palisading peripheral columnar epithelium and loosely arranged epithelium called stellate reticulum. “Wet” keratin is a hallmark of this variant. PCP have well-differentiated monomorphic squamous epithelium covering fibrovascular cores with thin capillary blood vessels and scattered immune cells including macrophages and neutrophils. The epithelium lacks surface maturation and there is no “wet” keratin14, 15.
In some craniopharyngioma resection specimens, however, the epithelium is sparse or absent and establishing a definite diagnosis can be challenging16. Some specimens for instance have prominent reactive changes with marked granulomatous inflammation, cholesterol clefts and prominent lymphocytic inflammation. On small biopsies, some PCP can be difficult to distinguish from other suprasellar and infundibulotuberal masses such as non-neoplastic Rathke’s cleft cysts16, 17. These cysts are often lined by ciliated cuboidal or columnar epithelium, but prominent squamous metaplasia can also occur, resembling the epithelium of PCP17.
Our recent genomic characterization of ACP and PCP revealed that each subtype of craniopharyngioma harbors highly recurrent activating mutations18. We observed that over 90% of ACP have mutations in CTNNB118 consistent with other studies demonstrating that mutations in exon 3 of the gene encoding beta-catenin and activation of the WNT pathway are important in the tumorigenesis of ACP19–23. Unexpectedly, we found that over 90% of PCP have BRAF V600E mutations18. CTNNB1 and BRAF alterations were mutually exclusive, clonal and specific to each subtype. This propitious finding has important implications for the diagnosis of PCP and the clinical management of some patients with this tumor.
Diagnostic evaluation of craniopharyngioma
The most immediate clinical impact of our findings is on everyday practical pathology diagnostics. Pathologists can now use immunohistochemistry (IHC) to guide diagnostic classification of suprasellar lesions. This is particularly helpful in specimens that are minute or have scant epithelium. In cells that lack mutations in CTNNB1 such as in PCP, the beta-catenin protein is localized at the cell membrane. In ACP that harbor mutations in CTNNB1, beta-catenin shifts into both the cytoplasm and nucleus of the neoplastic cells19–21, 24. The development of a mutation-specific antibody (VE1) that recognizes BRAF V600E mutant protein but not wild type BRAF protein provides pathologists with another IHC tool to discriminate PCP from ACP and from other entities that are in the differential diagnosis 25. Our study demonstrated a very high concordance between IHC results and genetic mutations in craniopharyngioma18. Thus, IHC information can be used in diagnostic decision making when needed and when available as pathologists formulate their final diagnostic reports.
In addition to situations where specimens are minute, BRAF VE1 IHC may be useful in the routine evaluation of Rathke’s cleft cysts (RCCs)17, 26, 27. A recent re-evaluation of 33 suprasellar mass that were diagnosed as RCCs showed that three cases harbored BRAF V600E mutations27. These cases had an atypical clinical presentation and two had squamous metaplasia. Upon reevaluation of the pathology and clinical information, these three cases were re-classified as PCP. Hence, determining the BRAF status is likely of significant value when evaluating epithelial suprasellar lesions17, 26, 27.
One caveat and challenge of using IHC to identify specimens harboring BRAF V600E mutations is that the VE1 antibody can cross-react with certain BRAF wild-type tissues. For example, endocrine tissues such as normal pituitary are immunoreactive with the VE1 antibody despite these tissues having wild-type BRAF28. The VE1 antibody also cross-reacts with cilia17, 26, 29 through recognition of epitopes in the axonemal dyneins of cilia that resemble the BRAF V600E peptide sequence used to generate the VE1 antibody26. The cytosol of ciliated cells shows variable degrees of positivity26. Thus, VE1 IHC of Rathke’s cleft cysts which have ciliated epithelium should be interpreted cautiously. In some cases where interpretation of the staining may be uncertain, allele-specific genetic testing for BRAF V600E mutation may be required to support the IHC results17, 26, 27. In some institutions allele specific genetic testing may be the preferred diagnostic modality. Because over 90% of papillary craniopharyngiomas harbor BRAF V600E mutations, some institutions may find it sufficient to make diagnoses and guide therapy decisions based on review of H&E stained sections alone.
Currently, the WHO classification of craniopharyngioma does not require mutation assessment using surrogates like IHC or direct genetic testing1. In time though, the classification of craniopharyngioma could have at least four groups: Adamantinomatous craniopharyngioma CTNNB1 mutated, adamantinomatous craniopharyngioma CTNNB1 wild type, papillary craniopharyngioma BRAF V600E mutated and papillary craniopharyngioma BRAF wild type. It is possible that the wild-type tumors may have different ways of activating BRAF or beta-catenin that have not yet identified. Uncovering the genetic drivers of the few CTNNB1 and BRAF wild-type craniopharyngioma will allow for further refinement of these categories.
Interestingly, a study has suggested that BRAF V600E mutations and mutations in CTNNB1 may co-exist in approximately 10% of ACP30. While targeted sequencing from the validation set from our original genomic study did not detect ACP samples with BRAF V600E mutations18, the possibility is very intriguing and requires further exploration. If co-existence of mutations in craniopharyngioma is confirmed, it will be important to determine if either mutation is clonal or sub-clonal, the clinical course of such tumors as well as the optimal treatment.
Systemic treatment for BRAF V600e mutated papillary craniopharyngioma
While targeting and inhibiting beta-catenin directly remains an unsolved challenge, considerable advances in the treatment of BRAF V600E mutant melanoma provide a paradigm for the targeted therapy of PCP31, 32. Targeted therapy has been successful for treating patients with other BRAF V600E mutated tumors33 including hairy cell leukemia33–39, Erdheim Chester Disease33, 40, ameloblastoma41–43 and pleomorphic xanthoastrocytoma33, 44–47.
Using targeted agents that inhibit BRAF and MEK, we recently achieved a dramatic response in a patient with a multiply recurrent BRAF V600E mutated PCP 48. Prior to therapy with BRAF and MEK inhibitors, the patient required several urgent neurosurgical decompressions for a rapidly growing tumor, which had a very large cystic component. The patient suffered from panhypopituitarism and chronic bilateral optic neuropathy. We first treated the patient with the BRAF inhibitor dabrafenib alone. In 17 days, the solid part of the tumor decreased by 50% and the cystic portion by 70%. Because concomitant inhibition of BRAF and MEK has been shown to reduce the emergence of resistance in melanoma31, we added trametinib for an additional 14 days. During the treatment the solid part of the tumor decreased by 85% and the cystic portion by 81%. The size of the cyst may have decreased as the treatment compromised the tumor epithelium and presumably diminished cyst fluid secretion. The residual tumor was resected and then three weeks later the patient was administered radiation therapy. Eight months following the radiation therapy, the patient remains without new symptoms 48.
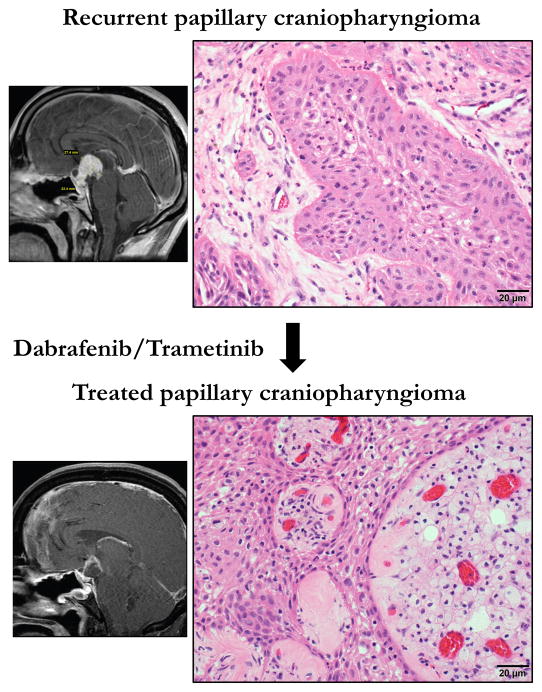
Review of the histology of the specimens that were resected before and after dabrafenib/trametinib treatment revealed a remarkable effect (Figure 1). The Ki67 proliferation index decreased from over 20% in the pre-treatment tumor to less than 0.5% in the on-treatment tumor. Radiation therapy was administered three weeks after this final on-treatment tumor resection. The combined dabrafenib and trametinib treatment led to a prominent immune response with foamy macrophages engorging the fibrovascular cores and CD8-positive T cells infiltrating throughout the tumor. This suggests that targeted therapy unleashes a strong anti-tumor immune response in PCP, a phenomenon that also appears to be elicited by BRAF inhibition in melanoma49–51.
Figure 1.
H&E stained sections of pre- and post-treatment papillary craniopharyngioma from case reported in Brastianos et al., JNCI 2015 48. Top panel shows the recurrent tumor. The lower panel shows the tumor following treatment with dabrafenib and trametinib. Side panels are sketch renditions of MRI images from our exceptional responder patient 48.
Development of resistance to BRAF and MEK inhibitors is common in patients with melanoma. Whole exome sequencing data from the pre- and on-treatment tumors from our patient did not identify the emergence of any known genetic drivers of BRAF resistance18. The low frequency of mutations and limited genomic complexity of these tumors suggests that combined therapy may effectively limit the emergence of resistance but high vigilance for the emergence of treatment resistance mechanisms will be required.
The rationale of concomitantly targeting BRAF and MEK for PCP treatment is supported by a recently published report using single agent vemurafenib treatment in a patient with a BRAF V600E mutant PCP52. Similar to the response observed in our patient, that tumor was also exceptionally responsive to targeted treatment, with a near complete radiological response after three months. When vemurafenib was held, however, the tumor regrew in six weeks. Tumor growth was stabilized when vemurafenib was re-administered but tumor progression subsequently ensued. The tumor progression seen in that patient treated with single agent vemurafenib suggests combining BRAF and MEK inhibition will be preferable for prolonged and durable control of tumor growth.
Phase II clinical trial study evaluating the combination of BRAF and MEK inhibition in patients with papillary craniopharyngioma
Given these exceptional tumor responses and the consistent occurrence of the BRAF V600E mutation in the vast majority of papillary craniopharyngiomas, we are now designing a multicenter phase II study evaluating the combination of BRAF and MEK inhibition in patients with papillary craniopharyngiomas. We will study the effect of dual inhibition because of the improved efficacy of the combination over single agent BRAF inhibitors in other BRAF-mutant tumors. As most resistance to single-agent BRAF inhibitors occurs because of reactivation of the RAF-MEK-ERK (MAPK) pathway, the addition of MEK inhibition delays the emergence of resistant clones. Furthermore, the major complication of RAF inhibitor treatment is the development of cutaneous squamous-cell carcinoma. This complication is significantly reduced in patients receiving the combination of dabrafenib and trametinib compared to those receiving single agent treatment alone31. Systemic treatment will be administered until definitive therapy with surgery or radiation therapy is indicated. Correlative studies will be performed with a focus on obtaining pre- and post-treatment tissue which will be characterized with whole exome sequencing and RNA-sequencing in an attempt to identify potential mutations, genomic aberrations or transcriptional mechanisms that might render PCP tumors either refractory or resistant to treatment.
Future outlook
The current standard of care for treating PCP involves surgery and radiation and can lead to substantial morbidity. Therefore, a neo-adjuvant approach for treating these tumors could be of use in select patient populations. Such strategies are commonly used for prolactin producing pituitary adenomas which are treated with bromocriptine as well as for germ cell tumors. Of note, we were able to detect mutant BRAF V600E DNA circulating in the blood of our exceptional responder patient 48. This finding is encouraging and suggests that confirming the presence of PCP may be achievable through non-invasive “liquid biopsy” methods such as cell-free DNA (cfDNA) detection testing. Our trial will include explorative objectives designed to carefully assess such capabilities. Moreover, the development of validated radiological criteria for discriminating PCP and ACP from one another and from other tumors will be important for evaluating patients with suprasellar masses3, 53–56. A combination of improved non-invasive diagnostics coupled with effective targeted therapy could provide a new treatment paradigm that in molecularly selected patient populations reduces the morbidities associated with surgery and radiation and improves the outcomes of patients with PCP and other rare brain tumors57.
Acknowledgments
Funding: This work was supported by the National Institutes of Health (K08 NS064168 and K12 CA090354-11), the Brain Science Foundation, the Conquer Cancer Foundation, the American Brain Tumor Association, the Jared Branfman Sunflowers for Life Fund for Pediatric Brain and Spinal Cancer Research, A Kid’s Brain Tumor Cure Foundation, Pedals for Pediatrics and the Clark Family, the Stahl Family Charitable Foundation, the Stop&Shop Pediatric Brain Tumor Program and the Pediatric Brain Tumor Clinical and Research Fund.
Footnotes
Declaration of interest: We declare that there is no conflict of interest that could be perceived as prejudicing the impartiality of the research reported.
References
- 1.Louis DN, Ohgaki H, Wiestler OD, Cavenee WK. WHO Classification of Tumours of the Central Nervous System. 4. International Agency for Research on Cancer; 2007. pp. 238–240. [Google Scholar]
- 2.Ostrom QT, Gittleman H, Liao P, Rouse C, Chen Y, Dowling J, Wolinsky Y, Kruchko C, Barnholtz-Sloan J. CBTRUS statistical report: primary brain and central nervous system tumors diagnosed in the United States in 2007–2011. Neuro Oncol. 2014;16(Suppl 4):iv1–63. doi: 10.1093/neuonc/nou223. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Pascual JM, Prieto R, Carrasco R. Infundibulo-tuberal or not strictly intraventricular craniopharyngioma: evidence for a major topographical category. Acta Neurochir (Wien) 2011;153:2403–2425. doi: 10.1007/s00701-011-1149-4. discussion 2426. [DOI] [PubMed] [Google Scholar]
- 4.Duff J, Meyer FB, Ilstrup DM, Laws ER, Jr, Schleck CD, Scheithauer BW. Long-term outcomes for surgically resected craniopharyngiomas. Neurosurgery. 2000;46:291–302. doi: 10.1097/00006123-200002000-00007. discussion 302–295. [DOI] [PubMed] [Google Scholar]
- 5.Barkhoudarian G, Laws ER. Craniopharyngioma: history. Pituitary. 2013;16:1–8. doi: 10.1007/s11102-012-0402-z. [DOI] [PubMed] [Google Scholar]
- 6.Sterkenburg AS, Hoffmann A, Gebhardt U, Warmuth-Metz M, Daubenbuchel AM, Muller HL. Survival, hypothalamic obesity, and neuropsychological/psychosocial status after childhood-onset craniopharyngioma: newly reported long-term outcomes. Neuro Oncol. 2015;17:1029–1038. doi: 10.1093/neuonc/nov044. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Stache C, Holsken A, Fahlbusch R, Flitsch J, Schlaffer SM, Buchfelder M, Buslei R. Tight junction protein claudin-1 is differentially expressed in craniopharyngioma subtypes and indicates invasive tumor growth. Neuro Oncol. 2014;16:256–264. doi: 10.1093/neuonc/not195. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Roemmler-Zehrer J, Geigenberger V, Stormann S, Losa M, Crippa V, Otto B, Bidlingmaier M, Dimopoulou C, Stalla GK, Schopohl J. Food intake regulating hormones in adult craniopharyngioma patients. Eur J Endocrinol. 2014;170:627–635. doi: 10.1530/EJE-13-0832. [DOI] [PubMed] [Google Scholar]
- 9.Pickering L, Jennum P, Gammeltoft S, Poulsgaard L, Feldt-Rasmussen U, Klose M. Sleep-wake and melatonin pattern in craniopharyngioma patients. Eur J Endocrinol. 2014;170:873–884. doi: 10.1530/EJE-13-1025. [DOI] [PubMed] [Google Scholar]
- 10.Ogawa Y, Kawaguchi T, Tominaga T. Outcome and mid-term prognosis after maximum and radical removal of craniopharyngiomas with the priority to the extended transsphenoidal approach--a single center experience. Clin Neurol Neurosurg. 2014;125:41–46. doi: 10.1016/j.clineuro.2014.07.014. [DOI] [PubMed] [Google Scholar]
- 11.Jahangiri A, Wagner J, Han SW, Zygourakis CC, Han SJ, Tran MT, Miller LM, Tom MW, Kunwar S, Blevins LS, Jr, Aghi MK. Morbidity of repeat transsphenoidal surgery assessed in more than 1000 operations. J Neurosurg. 2014;121:67–74. doi: 10.3171/2014.3.JNS131532. [DOI] [PubMed] [Google Scholar]
- 12.Liubinas SV, Munshey AS, Kaye AH. Management of recurrent craniopharyngioma. J Clin Neurosci. 2011;18:451–457. doi: 10.1016/j.jocn.2010.10.004. [DOI] [PubMed] [Google Scholar]
- 13.Hankinson TC, Palmeri NO, Williams SA, Torok MR, Serrano CA, Foreman NK, Handler MH, Liu AK. Patterns of care for craniopharyngioma: survey of members of the american association of neurological surgeons. Pediatr Neurosurg. 2013;49:131–136. doi: 10.1159/000357783. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Crotty TB, Scheithauer BW, Young WF, Jr, Davis DH, Shaw EG, Miller GM, Burger PC. Papillary craniopharyngioma: a clinicopathological study of 48 cases. J Neurosurg. 1995;83:206–214. doi: 10.3171/jns.1995.83.2.0206. [DOI] [PubMed] [Google Scholar]
- 15.Giangaspero F, Burger PC, Osborne DR, Stein RB. Suprasellar papillary squamous epithelioma (“papillary craniopharyngioma”) Am J Surg Pathol. 1984;8:57–64. doi: 10.1097/00000478-198401000-00006. [DOI] [PubMed] [Google Scholar]
- 16.Zada G, Lin N, Ojerholm E, Ramkissoon S, Laws ER. Craniopharyngioma and other cystic epithelial lesions of the sellar region: a review of clinical, imaging, and histopathological relationships. Neurosurg Focus. 2010;28:E4. doi: 10.3171/2010.2.FOCUS09318. [DOI] [PubMed] [Google Scholar]
- 17.Kim JH, Paulus W, Heim S. BRAF V600E mutation is a useful marker for differentiating Rathke’s cleft cyst with squamous metaplasia from papillary craniopharyngioma. J Neurooncol. 2015;123:189–191. doi: 10.1007/s11060-015-1757-6. [DOI] [PubMed] [Google Scholar]
- 18.Brastianos PK, Taylor-Weiner A, Manley PE, Jones RT, Dias-Santagata D, Thorner AR, Lawrence MS, Rodriguez FJ, Bernardo LA, Schubert L, Sunkavalli A, Shillingford N, Calicchio ML, Lidov HG, Taha H, Martinez-Lage M, Santi M, Storm PB, Lee JY, Palmer JN, Adappa ND, Scott RM, Dunn IF, Laws ER, Jr, Stewart C, Ligon KL, Hoang MP, Van Hummelen P, Hahn WC, Louis DN, Resnick AC, Kieran MW, Getz G, Santagata S. Exome sequencing identifies BRAF mutations in papillary craniopharyngiomas. Nat Genet. 2014;46:161–165. doi: 10.1038/ng.2868. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Martinez-Barbera JP, Buslei R. Adamantinomatous craniopharyngioma: pathology, molecular genetics and mouse models. J Pediatr Endocrinol Metab. 2015;28:7–17. doi: 10.1515/jpem-2014-0442. [DOI] [PubMed] [Google Scholar]
- 20.Martinez-Barbera JP. Molecular and cellular pathogenesis of adamantinomatous craniopharyngioma. Neuropathol Appl Neurobiol. 2015 doi: 10.1111/nan.12226. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Gaston-Massuet C, Andoniadou CL, Signore M, Jayakody SA, Charolidi N, Kyeyune R, Vernay B, Jacques TS, Taketo MM, Le Tissier P, Dattani MT, Martinez-Barbera JP. Increased Wingless (Wnt) signaling in pituitary progenitor/stem cells gives rise to pituitary tumors in mice and humans. Proc Natl Acad Sci U S A. 2011;108:11482–11487. doi: 10.1073/pnas.1101553108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Sekine S, Shibata T, Kokubu A, Morishita Y, Noguchi M, Nakanishi Y, Sakamoto M, Hirohashi S. Craniopharyngiomas of adamantinomatous type harbor beta-catenin gene mutations. Am J Pathol. 2002;161:1997–2001. doi: 10.1016/s0002-9440(10)64477-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Buslei R, Nolde M, Hofmann B, Meissner S, Eyupoglu IY, Siebzehnrubl F, Hahnen E, Kreutzer J, Fahlbusch R. Common mutations of beta-catenin in adamantinomatous craniopharyngiomas but not in other tumours originating from the sellar region. Acta Neuropathol. 2005;109:589–597. doi: 10.1007/s00401-005-1004-x. [DOI] [PubMed] [Google Scholar]
- 24.Hofmann BM, Kreutzer J, Saeger W, Buchfelder M, Blumcke I, Fahlbusch R, Buslei R. Nuclear beta-catenin accumulation as reliable marker for the differentiation between cystic craniopharyngiomas and rathke cleft cysts: a clinico-pathologic approach. Am J Surg Pathol. 2006;30:1595–1603. doi: 10.1097/01.pas.0000213328.64121.12. [DOI] [PubMed] [Google Scholar]
- 25.Capper D, Preusser M, Habel A, Sahm F, Ackermann U, Schindler G, Pusch S, Mechtersheimer G, Zentgraf H, von Deimling A. Assessment of BRAF V600E mutation status by immunohistochemistry with a mutation-specific monoclonal antibody. Acta Neuropathol. 2011;122:11–19. doi: 10.1007/s00401-011-0841-z. [DOI] [PubMed] [Google Scholar]
- 26.Jones RT, Abedalthagafi MS, Brahmandam M, Greenfield EA, Hoang MP, Louis DN, Hornick JL, Santagata S. Cross-reactivity of the BRAF VE1 antibody with epitopes in axonemal dyneins leads to staining of cilia. Mod Pathol. 2015;28:596–606. doi: 10.1038/modpathol.2014.150. [DOI] [PubMed] [Google Scholar]
- 27.Schweizer L, Capper D, Holsken A, Fahlbusch R, Flitsch J, Buchfelder M, Herold-Mende C, von Deimling A, Buslei R. BRAF V600E analysis for the differentiation of papillary craniopharyngiomas and Rathke’s cleft cysts. Neuropathol Appl Neurobiol. 2014 doi: 10.1111/nan.12201. [DOI] [PubMed] [Google Scholar]
- 28.Mordes DA, Lynch K, Campbell S, Dias-Santagata D, Nose V, Louis DN, Hoang MP. VE1 antibody immunoreactivity in normal anterior pituitary and adrenal cortex without detectable BRAF V600E mutations. Am J Clin Pathol. 2014;141:811–815. doi: 10.1309/AJCP37TLZLTUAOJL. [DOI] [PubMed] [Google Scholar]
- 29.Bosmuller H, Fischer A, Pham DL, Fehm T, Capper D, von Deimling A, Bonzheim I, Staebler A, Fend F. Detection of the BRAF V600E mutation in serous ovarian tumors: a comparative analysis of immunohistochemistry with a mutation-specific monoclonal antibody and allele-specific PCR. Hum Pathol. 2013;44:329–335. doi: 10.1016/j.humpath.2012.07.010. [DOI] [PubMed] [Google Scholar]
- 30.Larkin SJ, Preda V, Karavitaki N, Grossman A, Ansorge O. BRAF V600E mutations are characteristic for papillary craniopharyngioma and may coexist with CTNNB1-mutated adamantinomatous craniopharyngioma. Acta Neuropathol. 2014;127:927–929. doi: 10.1007/s00401-014-1270-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Flaherty KT, Infante JR, Daud A, Gonzalez R, Kefford RF, Sosman J, Hamid O, Schuchter L, Cebon J, Ibrahim N, Kudchadkar R, Burris HA, 3rd, Falchook G, Algazi A, Lewis K, Long GV, Puzanov I, Lebowitz P, Singh A, Little S, Sun P, Allred A, Ouellet D, Kim KB, Patel K, Weber J. Combined BRAF and MEK inhibition in melanoma with BRAF V600 mutations. N Engl J Med. 2012;367:1694–1703. doi: 10.1056/NEJMoa1210093. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Flaherty KT, Puzanov I, Kim KB, Ribas A, McArthur GA, Sosman JA, O’Dwyer PJ, Lee RJ, Grippo JF, Nolop K, Chapman PB. Inhibition of mutated, activated BRAF in metastatic melanoma. N Engl J Med. 2010;363:809–819. doi: 10.1056/NEJMoa1002011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Hyman DM, Puzanov I, Subbiah V, Faris JE, Chau I, Blay JY, Wolf J, Raje NS, Diamond EL, Hollebecque A, Gervais R, Elez-Fernandez ME, Italiano A, Hofheinz RD, Hidalgo M, Chan E, Schuler M, Lasserre SF, Makrutzki M, Sirzen F, Veronese ML, Tabernero J, Baselga J. Vemurafenib in Multiple Nonmelanoma Cancers with BRAF V600 Mutations. N Engl J Med. 2015;373:726–736. doi: 10.1056/NEJMoa1502309. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Maevis V, Mey U, Schmidt-Wolf G, Schmidt-Wolf IG. Hairy cell leukemia: short review, today’s recommendations and outlook. Blood Cancer J. 2014;4:e184. doi: 10.1038/bcj.2014.3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Peyrade F, Re D, Ginet C, Gastaud L, Allegra M, Ballotti R, Thyss A, Zenz T, Auberger P, Robert G. Low-dose vemurafenib induces complete remission in a case of hairy-cell leukemia with a V600E mutation. Haematologica. 2013;98:e20–22. doi: 10.3324/haematol.2012.082404. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Munoz J, Schlette E, Kurzrock R. Rapid response to vemurafenib in a heavily pretreated patient with hairy cell leukemia and a BRAF mutation. J Clin Oncol. 2013;31:e351–352. doi: 10.1200/JCO.2012.45.7739. [DOI] [PubMed] [Google Scholar]
- 37.Follows GA, Sims H, Bloxham DM, Zenz T, Hopper MA, Liu H, Bench A, Wright P, Van’t Veer MB, Scott MA. Rapid response of biallelic BRAF V600E mutated hairy cell leukaemia to low dose vemurafenib. Br J Haematol. 2013;161:150–153. doi: 10.1111/bjh.12201. [DOI] [PubMed] [Google Scholar]
- 38.Dietrich S, Hullein J, Hundemer M, Lehners N, Jethwa A, Capper D, Acker T, Garvalov BK, Andrulis M, Blume C, Schulte C, Mandel T, Meissner J, Frohling S, von Kalle C, Glimm H, Ho AD, Zenz T. Continued response off treatment after BRAF inhibition in refractory hairy cell leukemia. J Clin Oncol. 2013;31:e300–303. doi: 10.1200/JCO.2012.45.9495. [DOI] [PubMed] [Google Scholar]
- 39.Dietrich S, Glimm H, Andrulis M, von Kalle C, Ho AD, Zenz T. BRAF inhibition in refractory hairy-cell leukemia. N Engl J Med. 2012;366:2038–2040. doi: 10.1056/NEJMc1202124. [DOI] [PubMed] [Google Scholar]
- 40.Haroche J, Cohen-Aubart F, Emile JF, Maksud P, Drier A, Toledano D, Barete S, Charlotte F, Cluzel P, Donadieu J, Benameur N, Grenier PA, Besnard S, Ory JP, Lifermann F, Idbaih A, Granel B, Graffin B, Hervier B, Arnaud L, Amoura Z. Reproducible and Sustained Efficacy of Targeted Therapy With Vemurafenib in Patients With BRAFV600E-Mutated Erdheim-Chester Disease. J Clin Oncol. 2014 doi: 10.1200/JCO.2014.57.1950. [DOI] [PubMed] [Google Scholar]
- 41.Sweeney RT, McClary AC, Myers BR, Biscocho J, Neahring L, Kwei KA, Qu K, Gong X, Ng T, Jones CD, Varma S, Odegaard JI, Sugiyama T, Koyota S, Rubin BP, Troxell ML, Pelham RJ, Zehnder JL, Beachy PA, Pollack JR, West RB. Identification of recurrent SMO and BRAF mutations in ameloblastomas. Nat Genet. 2014;46:722–725. doi: 10.1038/ng.2986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Kaye FJ, Ivey AM, Drane WE, Mendenhall WM, Allan RW. Clinical and radiographic response with combined BRAF-targeted therapy in stage 4 ameloblastoma. J Natl Cancer Inst. 2015;107:378. doi: 10.1093/jnci/dju378. [DOI] [PubMed] [Google Scholar]
- 43.Gomes CC, Diniz MG, Gomez RS. Progress towards personalized medicine for ameloblastoma. J Pathol. 2014;232:488–491. doi: 10.1002/path.4331. [DOI] [PubMed] [Google Scholar]
- 44.MacConaill LE, Campbell CD, Kehoe SM, Bass AJ, Hatton C, Niu L, Davis M, Yao K, Hanna M, Mondal C, Luongo L, Emery CM, Baker AC, Philips J, Goff DJ, Fiorentino M, Rubin MA, Polyak K, Chan J, Wang Y, Fletcher JA, Santagata S, Corso G, Roviello F, Shivdasani R, Kieran MW, Ligon KL, Stiles CD, Hahn WC, Meyerson ML, Garraway LA. Profiling critical cancer gene mutations in clinical tumor samples. PLoS One. 2009;4:e7887. doi: 10.1371/journal.pone.0007887. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Lee EQ, Ruland S, LeBoeuf NR, Wen PY, Santagata S. Successful Treatment of a Progressive BRAF V600E-Mutated Anaplastic Pleomorphic Xanthoastrocytoma With Vemurafenib Monotherapy. J Clin Oncol. 2014 doi: 10.1200/JCO.2013.51.1766. [DOI] [PubMed] [Google Scholar]
- 46.Dias-Santagata D, Lam Q, Vernovsky K, Vena N, Lennerz JK, Borger DR, Batchelor TT, Ligon KL, Iafrate AJ, Ligon AH, Louis DN, Santagata S. BRAF V600E mutations are common in pleomorphic xanthoastrocytoma: diagnostic and therapeutic implications. PLoS One. 2011;6:e17948. doi: 10.1371/journal.pone.0017948. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Chamberlain MC. Salvage therapy with BRAF inhibitors for recurrent pleomorphic xanthoastrocytoma: a retrospective case series. J Neurooncol. 2013;114:237–240. doi: 10.1007/s11060-013-1176-5. [DOI] [PubMed] [Google Scholar]
- 48.Brastianos PK, Shankar GM, Gill CM, Taylor-Weiner A, Nayyar N, Panka DJ, Sullivan RJ, Frederick DT, Abedalthagafi M, Jones PS, Dunn IF, Nahed BV, Romero JM, Louis DN, Getz G, Cahill DP, Santagata S, Curry WT, Jr, Barker IFG. Dramatic Response of BRAF V600E Mutant Papillary Craniopharyngioma to Targeted Therapy. J Natl Cancer Inst. 2015 doi: 10.1093/jnci/djv310. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Frederick DT, Piris A, Cogdill AP, Cooper ZA, Lezcano C, Ferrone CR, Mitra D, Boni A, Newton LP, Liu C, Peng W, Sullivan RJ, Lawrence DP, Hodi FS, Overwijk WW, Lizee G, Murphy GF, Hwu P, Flaherty KT, Fisher DE, Wargo JA. BRAF inhibition is associated with enhanced melanoma antigen expression and a more favorable tumor microenvironment in patients with metastatic melanoma. Clin Cancer Res. 2013;19:1225–1231. doi: 10.1158/1078-0432.CCR-12-1630. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Cooper ZA, Juneja VR, Sage PT, Frederick DT, Piris A, Mitra D, Lo JA, Hodi FS, Freeman GJ, Bosenberg MW, McMahon M, Flaherty KT, Fisher DE, Sharpe AH, Wargo JA. Response to BRAF inhibition in melanoma is enhanced when combined with immune checkpoint blockade. Cancer Immunol Res. 2014;2:643–654. doi: 10.1158/2326-6066.CIR-13-0215. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Wilmott JS, Long GV, Howle JR, Haydu LE, Sharma RN, Thompson JF, Kefford RF, Hersey P, Scolyer RA. Selective BRAF inhibitors induce marked T-cell infiltration into human metastatic melanoma. Clin Cancer Res. 2012;18:1386–1394. doi: 10.1158/1078-0432.CCR-11-2479. [DOI] [PubMed] [Google Scholar]
- 52.Aylwin SJ, Bodi I, Beaney R. Pronounced response of papillary craniopharyngioma to treatment with vemurafenib, a BRAF inhibitor. Pituitary. 2015 doi: 10.1007/s11102-015-0663-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Sartoretti-Schefer S, Wichmann W, Aguzzi A, Valavanis A. MR differentiation of adamantinous and squamous-papillary craniopharyngiomas. AJNR Am J Neuroradiol. 1997;18:77–87. [PMC free article] [PubMed] [Google Scholar]
- 54.Pascual JM, Prieto R, Castro-Dufourny I, Carrasco R. Topographic Diagnosis of Papillary Craniopharyngiomas: The Need for an Accurate MRI-Surgical Correlation. AJNR Am J Neuroradiol. 2015 doi: 10.3174/ajnr.A4441. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Lee HJ, Wu CC, Wu HM, Hung SC, Lirng JF, Luo CB, Chang FC, Guo WY. Pretreatment diagnosis of suprasellar papillary craniopharyngioma and germ cell tumors of adult patients. AJNR Am J Neuroradiol. 2015;36:508–517. doi: 10.3174/ajnr.A4142. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Pascual JM, Gonzalez-Llanos F, Barrios L, Roda JM. Intraventricular craniopharyngiomas: topographical classification and surgical approach selection based on an extensive overview. Acta Neurochir (Wien) 2004;146:785–802. doi: 10.1007/s00701-004-0295-3. [DOI] [PubMed] [Google Scholar]
- 57.Brastianos PK, Horowitz PM, Santagata S, Jones RT, McKenna A, Getz G, Ligon KL, Palescandolo E, Van Hummelen P, Ducar MD, Raza A, Sunkavalli A, Macconaill LE, Stemmer-Rachamimov AO, Louis DN, Hahn WC, Dunn IF, Beroukhim R. Genomic sequencing of meningiomas identifies oncogenic SMO and AKT1 mutations. Nat Genet. 2013;45:285–289. doi: 10.1038/ng.2526. [DOI] [PMC free article] [PubMed] [Google Scholar]