Abstract
OBJECTIVE
Appropriate glycemic control is fundamental to diabetes care, but aggressive glucose targets and intensive therapy may unintentionally increase episodes of hypoglycemia. We quantified the burden of severe hypoglycemia requiring medical intervention in a well-defined population of insured individuals with diabetes receiving care in integrated health care delivery systems across the U.S.
RESEARCH DESIGN AND METHODS
This observational cohort study included 917,440 adults with diabetes receiving care during 2005 to 2011 at participating SUrveillance, PREvention, and ManagEment of Diabetes Mellitus (SUPREME-DM) network sites. Severe hypoglycemia rates were based on any occurrence of hypoglycemia-related ICD-9 codes from emergency department or inpatient medical encounters and reported overall and by age, sex, comorbidity status, antecedent A1C level, and medication use.
RESULTS
Annual rates of severe hypoglycemia ranged from 1.4 to 1.6 events per 100 person-years. Rates of severe hypoglycemia were higher among those with older age, chronic kidney disease, congestive heart failure, cardiovascular disease, depression, and higher A1C levels, and in users of insulin, insulin secretagogues, or β-blockers (P < 0.001 for all). Changes in severe hypoglycemia occurrence over time were not clinically significant in the cohort as a whole but were observed in subgroups of individuals with chronic kidney disease, congestive heart failure, and cardiovascular disease.
CONCLUSIONS
Risk of severe hypoglycemia in clinical settings is considerably higher in identifiable patient subgroups than in randomized controlled trials. Strategies that reduce the risk of hypoglycemia in high-risk patients are needed.
Introduction
Appropriate glycemic control is a cornerstone of diabetes care (1). The value of good glycemic control in reducing microvascular complications of diabetes was demonstrated for type 1 diabetes in 1993 by the Diabetes Control and Complications Trial (2) and for type 2 diabetes in 1998 by the United Kingdom Prospective Diabetes Study (UKPDS) (3–5). In response to these findings, early national standards of diabetes care defined the acceptable level of glycated hemoglobin (A1C) as <8% for most patients with diabetes (6). In 2002, the American Diabetes Association decreased the target A1C for adults to <7% (7). Changes in national guidelines and the addition of new glucose-lowering medications resulted in a dramatic decrease in A1C levels in the U.S. from a mean A1C of 8.2% in 1996 (8) to 7.2% in 2006 (9). In 2008, results of the Action to Control Cardiovascular Risk in Diabetes (ACCORD) (10), Action in Diabetes and Vascular Disease: Preterax and Diamicron Modified Release Controlled Evaluation (ADVANCE) (11), and Veterans Affairs Diabetes Trial (VADT) (12) trials, follow-up from the Steno-2 (13) and UKPDS (14) trials, and multiple epidemiological studies of A1C level and treatment intensification (15,16) began to raise concerns about the risks of aggressive treatment and suggested that A1C goals should be individually customized (10,17) based on anticipated benefits and risk of therapy, including increased risk of severe hypoglycemia with more aggressive glucose and A1C goals (18–20).
Hypoglycemia can be a life-threatening complication of diabetes as well as a major source of anxiety and a threat to quality of life (21,22). A recent study of 33 million Medicare patients demonstrated that although hospital admission for hyperglycemia events declined ∼39% from 1999 to 2011, the admission rates for hypoglycemia increased ∼12%, and hypoglycemia is now the most common acute metabolic event leading to hospitalization (23). Most estimates of severe hypoglycemia risk are derived from clinical trials, in which event rates range from 0.3 to 1 per 100 person-years of observation (11,18,24). However, the Medicare data described above and other observational studies suggest that severe hypoglycemia in community-treated patients is much more common than indicated by clinical trial data (25–27). Additional evidence from clinical trials suggests that the increased risk of hypoglycemia in the absence of the strict oversight present in clinical trials may also be related to a variety of other factors, including age, cardiovascular and kidney function, cognition, body weight, diabetes duration, and glucose-lowering agent use (28,29).
Having established the SUrveillance, PREvention, and ManagEment of Diabetes Mellitus (SUPREME-DM) DataLink, which represents the largest insured diabetes patient cohort with data from electronic health records in the U.S. outside of the Veterans Administration (30), we sought to quantify the burden of severe hypoglycemia requiring medical intervention in a well-defined community population. Specific goals of the current study were to quantify the occurrence of severe hypoglycemia among insured, community-treated adults with diabetes; to identify subgroups of individuals at high risk of severe hypoglycemia; and to report trends in hypoglycemia over time as a whole and among different subgroups. On the basis of experiences in clinical practice and the clinical trial findings described above, we hypothesized that rates of severe hypoglycemia would be higher among patients treated with insulin who are elderly and have multiple comorbid conditions.
Research Design and Methods
Study Design and Location
This study was conducted as part of the SUPREME-DM network, which brings together diabetes researchers from 11 member organizations of the Health Maintenance Organization Research Network and university partners. Health systems participating in SUPREME-DM include Marshfield Clinic (Wisconsin), Geisinger Health System (Pennsylvania), Group Health (Washington), HealthPartners (Minnesota), Henry Ford Health System (Michigan), and Kaiser Permanente Colorado, Northern California, Southern California, Hawaii, Georgia, and Northwest (Oregon and Washington). Researchers embedded within these health systems extracted information on demographic characteristics and outpatient and inpatient health care encounters from their clinical and administrative data systems to identify persons with diabetes. The SUPREME-DM diabetes cohort includes more than 1 million individuals with diabetes drawn from SUPREME-DM health system members between 1 January 2005 and 31 December 2011.
Study Population
The eligible source population for each calendar year between 2005 and 2011 consisted of adults with diabetes who were enrolled in a participating health insurance plan from January 1 through December 31 of that year, without any enrollment gaps or lapses in health insurance coverage greater than 90 days, and who had at least 1 year of follow-up. We used inpatient and outpatient diagnosis codes, laboratory tests and associated results, and information on medications dispensed to identify adults with diabetes from among those meeting enrollment inclusion criteria (31). Importantly, our electronic algorithm is unable to distinguish between type 1 and type 2 diabetes. Because the population is limited to adults, ∼95% have type 2 diabetes (32). Subjects were classified as having diabetes if, within a 24-month period, they had either one or more inpatient diabetes diagnostic code (ICD-9 250.x, 357.2, 366.41, 362.01–362.07) or any combination of two or more of the following criteria:
1) outpatient visit with a diabetes diagnosis code (see inpatient codes);
2) A1C ≥6.5%;
3) fasting plasma glucose ≥126 mg/dL (7.0 mmol/L);
4) random plasma glucose of ≥200 mg/dL (11.1 mmol/L); or
5) any filled prescription for a glucose-lowering medication (31).
When the two criteria used for identification were of the same source (e.g., two outpatient diagnoses or two elevated laboratory values), we required them to occur on separate days, but no more than 2 years apart. Two dispenses of metformin or a thiazolidinedione with no other indication of diabetes were not counted because these agents could be used as treatment for other conditions, such as prediabetes, polycystic ovarian syndrome, or HIV lipodystrophy. Women with evidence of diabetes only during pregnancy were excluded. Similar methods of diabetes identification have been evaluated previously (30,33).
Information on diabetes status was available from 2005 through 2011 and from as early as 2000 at some sites. For individuals identified as having diabetes, an index date was defined as the latter of 1 January 2005 or the date on which they were identified as having diabetes. An individual who was identified as having diabetes remained in the diabetes cohort until the date of censoring (the earliest of disenrollment from the health plan, death, or 31 December 2011). To be eligible for inclusion in study analyses, subjects had to 1) be age 20 years or older on the index date, 2) be enrolled at the index date, and 3) have at least 1 year of follow-up.
Data Sources, Extraction, and Validation
Data were derived from each member institution’s Virtual Data Warehouse, a standardized database that unifies clinical and administrative data for patients and members of the participating health care systems, extensively described elsewhere (30,34). A coordinating site distributed a program to all participating sites that extracted Virtual Data Warehouse data to create the SUPREME-DM DataLink. All data were evaluated for range, distribution, implausible values, and missing data by DataLink programmers at the local site and then transferred to a centralized DataLink site. The centralized site performed additional data validation procedures, generated required analytic files, and performed data analyses. All data files were constructed to meet definitions of Limited Data Sets and shared only under strict governance of Data Use Agreements among the SUPREME-DM DataLink sites.
During SUPREME-DM DataLink construction, eight sites ceded institutional review board review authority to a ninth participating system (Kaiser Permanente Colorado), and two sites retained local oversight (34). This study was reviewed in advance, approved, and monitored by these institutional review boards.
Definition and Measurement of Key Variables
For study purposes, severe hypoglycemia was defined as any primary or secondary occurrence of a hypoglycemia-related ICD-9 code extracted from an emergency department or inpatient encounter. On the basis of previous validation studies (31), we included 1) any occurrence of ICD-9 codes 251.0 (hypoglycemic coma), 251.1 (other specified hypoglycemia), 251.2 (hypoglycemia, unspecified), and 962.3 (poisoning by insulins and antidiabetic agents), and 2) occurrences of ICD-9 code 250.8x (diabetes with other specified manifestations) that were not associated with a codiagnosis code unrelated to hypoglycemia. The excluded codiagnosis ICD-9 codes were 258.9 (secondary diabetic glycogenosis), 272.7 (diabetic lipidosis), 681.xx, 682.xx, 686.9x (cellulitis), 707.1–707.9 (ulcers of the lower extremity), 709.3 (Oppenheim-Urbach syndrome), 730.0–730.2, and 731.8 (osteomyelitis). This method of hypoglycemia identification has been demonstrated to have a positive predictive value of 89% (31) and has been used in previous studies (35).
Demographic data included age and sex. Age was defined in years at baseline, which was the latter of 1 January 2005 and the index date of diabetes diagnosis. The last A1C value in the quarter preceding the event was extracted. If there was no A1C value in that quarter, we identified the most recent A1C value in the 12 months preceding the event. When the most recent A1C was measured more than 12 months before the event date of interest, A1C was treated as missing.
Pharmacy dispensing records were used to identify exposure to glucose-lowering medications and β-blockers in the quarter before the event. We determined whether there was a pharmacy fill for each of these classes of medication, including long- and intermediate-acting insulin, short- and rapid-acting insulin, secretagogues (sulfonylureas and meglitinides), and other drugs (metformin, thiazolidinediones, α-glucosidase inhibitors, GLP-1 receptor agonists, and dipeptidyl peptidase-4 inhibitors). Patients on premixed insulin were considered to have received both long- and short-acting insulin. Antihyperglycemic therapy use in the cohort is further described by Raebel et al. (36). Also recorded was additional information regarding certain comorbid conditions with the potential to influence hypoglycemia or hypoglycemia awareness, including chronic kidney disease (CKD; ICD-9 codes 585.x), congestive heart failure (CHF; ICD-9 codes 428.x), cardiovascular disease (CVD, excluding CHF; ICD-9 codes 410–414.x or 429.4), and depression (ICD-9 codes 296.2, 296.3, or 311.x). Depression was assessed at cohort entry only. All other comorbidities were assessed annually and correlated with the rate of hypoglycemia in that year.
Statistical Analysis
Planned statistical analyses were established a priori. For each calendar year, we calculated the rate of severe hypoglycemia overall and by prespecified subgroups based on age, sex, comorbidities of interest (i.e., CKD, CHF, CVD, and depression), A1C level, and medication exposure (i.e., glucose-lowering agents and β-blockers). We also calculated the average rate of severe hypoglycemia during the entire 7-year study period (2005–2011), weighted by person-time in each year. For our analysis of medication and A1C groups, we calculated the rate of severe hypoglycemia per quarter and linked it to the medication data or A1C values from the previous quarter to allow for changes in A1C and medication use. Annual rates were then calculated by taking the average over the quarters, weighted by the amount of person-time in each quarter. Poisson regression models were used to examine the time trend from 2005 to 2011 for the overall study population and within subgroups of risk factors (age; sex; medication exposure; comorbidity status for CKD, CVD, CHF, and depression; and A1C levels), and to test the difference in average hypoglycemia rates between subgroups. Analyses were conducted using SAS 9.2 software.
Results
At baseline, adult patients with diabetes in the SUPREME-DM DataLink who met entry criteria for this study (n = 917,440) were slightly more likely to be male than female and were a mean age of 57.9 years (Table 1). The prevalence of comorbidities ranged from 4.9% for CHF to 18.7% for CVD. Patients who experienced one or more severe hypoglycemic events tended to be slightly older and had more comorbidities than those who did not.
Table 1.
Baseline characteristics of the 917,440 patients with diabetes in the study sample by hypoglycemia status: the SUPREME-DM studya
| Total | Severe hypoglycemic events/year |
||||
|---|---|---|---|---|---|
| None |
1 severe |
≥2 severe |
|||
| (N = 917,440) | (n = 875,062) | (n = 41,063) | (n = 1,315) | P valueb | |
| Person-years of time at risk | 4,322,786 | 4,085,965 | 232,747 | 4,075 | |
| Female (%) | 47.9 | 47.8 | 49.0 | 47.5 | <0.001 |
| Age, mean (SD), years | 57.9 (13.2) | 57.7 (13.2) | 61.0 (13.2) | 60.5 (15.2) | <0.001 |
| Comorbidities (%) | |||||
| CKD | 10.4 | 9.5 | 29.1 | 49.0 | <0.001 |
| CVD | 18.7 | 18.0 | 34.2 | 49.0 | <0.001 |
| CHF | 4.9 | 4.5 | 12.9 | 23.3 | <0.001 |
| Depression | 11.1 | 11.1 | 11.0 | 16.0 | <0.001 |
| Comorbidity index, mean (SD)c | 2.70 (1.98) | 2.70 (1.96) | 2.96 (2.31) | 3.98 (2.97) | <0.001 |
| Follow-up, mean (SD), years | 4.71 (2.18) | 4.67 (2.18) | 5.67 (1.81) | 3.10 (1.97) | <0.001 |
aStudy includes patients with a minimum of 1 year of follow-up after diabetes identification.
bThree-way comparison between 0, 1, and ≥2 severe hypoglycemic events/year.
cQuan modification of the Elixhauser comorbidity index (37).
Annual rates of severe hypoglycemia ranged from a high of 1.59 events per 100 person-years in 2006 to a low of 1.37 events per 100 person-years in 2010 (Table 2). There was no clinically discernible trend in the annual rate of severe hypoglycemia during the study period. There was no significant difference in the rate of severe hypoglycemia by sex (P = 0.15). Severe hypoglycemia was most common in individuals of younger as well as older age (P < 0.001); the highest rates of severe hypoglycemia were observed in patients aged 75 years and older, whereas individuals aged 20–44 years had higher rates of hypoglycemia than those 45–64 years of age.
Table 2.
Rates of severe hypoglycemic events by year for adult participants in the SUPREME-DM
| Study | Annual rate (per 100 person-years) |
2005–2011 weighted rate | P value for time trendf | P value across the groupsg | ||||||
|---|---|---|---|---|---|---|---|---|---|---|
| 2005 | 2006 | 2007 | 2008 | 2009 | 2010 | 2011 | ||||
| Total person-years | 570,931 | 594,929 | 613,894 | 629,894 | 645,210 | 662,035 | 674,906 | |||
| Overall | ||||||||||
| Unadjusteda | 1.44 | 1.58 | 1.55 | 1.55 | 1.50 | 1.48 | 1.46 | 1.51 | 0.448 | |
| Adjustedb | 1.47 | 1.59 | 1.51 | 1.49 | 1.36 | 1.37 | 1.47 | 1.47 | 0.011 | |
| Demographicsa | ||||||||||
| Age (years) | <0.001 | |||||||||
| 20–44 | 1.35 | 1.26 | 1.32 | 1.15 | 1.22 | 1.06 | 1.15 | 1.22 | 0.016 | |
| 45–54 | 0.96 | 1.03 | 0.99 | 0.99 | 1.02 | 0.93 | 0.93 | 0.98 | 0.357 | |
| 55–64 | 1.09 | 1.15 | 1.11 | 1.14 | 1.06 | 1.11 | 1.11 | 1.11 | 0.852 | |
| 65–74 | 1.58 | 1.81 | 1.76 | 1.71 | 1.63 | 1.60 | 1.52 | 1.66 | 0.034 | |
| 75–84 | 2.34 | 2.72 | 2.64 | 2.70 | 2.57 | 2.51 | 2.39 | 2.55 | 0.393 | |
| ≥85 | 2.90 | 2.80 | 2.89 | 3.09 | 2.72 | 2.68 | 2.70 | 2.81 | 0.314 | |
| Sex | 0.154 | |||||||||
| Female | 1.47 | 1.61 | 1.60 | 1.60 | 1.54 | 1.51 | 1.41 | 1.53 | 0.173 | |
| Male | 1.42 | 1.55 | 1.50 | 1.51 | 1.47 | 1.45 | 1.51 | 1.49 | 0.970 | |
| Comorbiditiesb | ||||||||||
| CKD (cohort year) | <0.001 | |||||||||
| Yes | 7.16 | 6.20 | 4.95 | 5.77 | 5.04 | 4.48 | 4.51 | 5.26 | <0.001 | |
| No | 0.96 | 1.05 | 0.98 | 0.94 | 0.97 | 0.85 | 0.88 | 0.95 | 0.083 | |
| CHF (cohort year) | <0.001 | |||||||||
| Yes | 8.30 | 7.32 | 7.64 | 7.54 | 6.45 | 6.73 | 6.52 | 7.19 | 0.002 | |
| No | 1.28 | 1.40 | 1.32 | 1.33 | 1.33 | 1.19 | 1.22 | 1.29 | 0.021 | |
| CVD (cohort year) | <0.001 | |||||||||
| Yes | 6.69 | 5.46 | 6.39 | 5.72 | 5.03 | 4.59 | 4.18 | 5.37 | <0.001 | |
| No | 1.03 | 1.15 | 1.04 | 1.04 | 1.07 | 0.95 | 0.97 | 1.03 | 0.190 | |
| Depression (baseline) | <0.001 | |||||||||
| Yes | 2.25 | 2.24 | 2.38 | 2.12 | 2.13 | 1.76 | 2.03 | 2.12 | 0.041 | |
| No | 1.39 | 1.49 | 1.40 | 1.44 | 1.39 | 1.29 | 1.28 | 1.38 | 0.003 | |
| Glycemic controlb | ||||||||||
| A1C | <0.001 | |||||||||
| <7% | 1.00 | 1.20 | 1.06 | 0.92 | 0.86 | 0.92 | 0.79 | 0.96 | <0.001 | |
| 7–8% | 1.57 | 1.56 | 1.73 | 1.69 | 1.61 | 1.58 | 1.49 | 1.60 | 0.492 | |
| 8–9% | 2.20 | 1.91 | 2.23 | 2.46 | 2.35 | 2.17 | 1.89 | 2.17 | 0.748 | |
| ≥9% | 2.44 | 2.53 | 2.54 | 2.60 | 2.77 | 2.48 | 2.69 | 2.58 | 0.442 | |
| Medication exposureb | ||||||||||
| Diabetes medications | <0.001 | |||||||||
| Short-acting insulinc | 4.90 | 5.49 | 5.23 | 5.01 | 5.09 | 4.95 | 4.55 | 5.03 | 0.030 | |
| Long-acting insulinc | 3.79 | 3.30 | 3.54 | 2.99 | 3.28 | 3.03 | 2.72 | 3.18 | <0.001 | |
| Secretagoguesd | 0.97 | 0.88 | 0.91 | 0.97 | 0.87 | 0.88 | 1.01 | 0.93 | 0.764 | |
| Other drugse | 0.42 | 0.39 | 0.42 | 0.41 | 0.44 | 0.42 | 0.39 | 0.41 | 0.999 | |
| β-Blockers | <0.001 | |||||||||
| Yes | 2.46 | 2.47 | 2.38 | 3.29 | 2.99 | 2.04 | 1.65 | 2.46 | 0.173 | |
| No | 1.36 | 1.45 | 1.38 | 1.36 | 1.35 | 1.23 | 1.25 | 1.34 | 0.417 | |
aUnadjusted.
bAdjusted for age and sex. Categories are not mutually exclusive.
cShort-acting insulin category includes patients additionally on long-acting insulin or on premixed insulin. Long-acting insulin category also includes patients on combination or premixed insulin. Includes NPH insulin.
dInsulin secretagogues include sulfonylurea, combined sulfonylurea/biguanide, and meglitinides.
eOther drugs include metformin, thiazolidinediones, α-glucosidase inhibitors, dipeptidyl peptidase-4 inhibitors, and GLP-1 agonists.
fTest for monotonic trend across the years within each group from 2005 to 2011.
gTest for whether average weighted rates differ between subgroups (e.g., female vs. male).
Patients with one or more comorbid conditions were significantly more likely to experience a severe hypoglycemic episode. Unadjusted and age- and sex-adjusted rates were similar. Individuals with CKD, CHF, and/or CVD had four- to eightfold higher rates of severe hypoglycemia than individuals without these comorbidities (P < 0.001 for all). The risk of severe hypoglycemia was ∼50% greater in patients with depression at cohort entry compared with those without (P < 0.001). Patients with two or more episodes of severe hypoglycemia per year had a higher prevalence of comorbidities than those with one or fewer episodes of severe hypoglycemia (comorbidity index of 3.98 vs. 2.96 and 2.70, respectively). Interestingly, there was a significant downward trend in the incidence of severe hypoglycemia over time for each of four specific comorbidities examined. This downward trend appeared clinically significant for CKD, CHF, and CVD, but was less convincing in the case of depression.
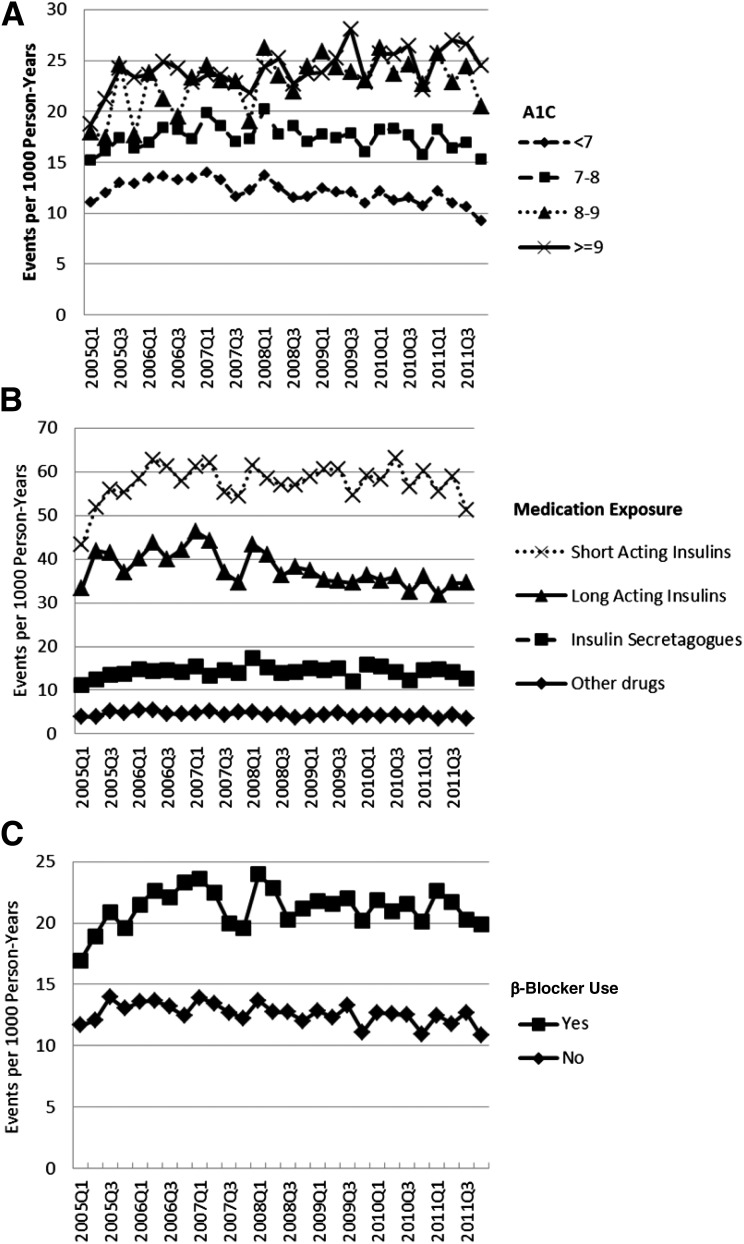
Glycemic control and medication exposure were significantly associated with the rate of severe hypoglycemia (Fig. 1). Higher A1C levels were associated with higher rates of severe hypoglycemia (P < 0.001), although the difference between those with an A1C of 8–9% and those with A1C >9% was modest (Fig. 1A). Patients filling prescriptions for insulin had the highest rates of severe hypoglycemia, with rates 10- to 12-times higher than those observed in patients on nonsecretagogue medications, and 3- to 5-times higher than those observed in patients on secretagogues (Fig. 1B). β-Blocker exposure was also associated with a higher rate of severe hypoglycemia (P < 0.001) (Fig. 1C). Severe hypoglycemia rates did not change in a clinically significant manner over time in subjects by glycemic control categories or drug class. Results suggest that risk factors for severe hypoglycemia include CHD, CKD, CVD, depression, advancing age, and treatment with insulin, sulfonylureas, or β-blockers (Table 2).
Figure 1.
Quarterly rates of severe hypoglycemic events by the level of glycemic control and medication exposure (2005–2011). Quarterly rates of severe hypoglycemic events age- and sex-standardized to the 2010 U.S. Census population for various levels of hemoglobin A1C (A), exposure to glucose-lowering medications (B), and β-blocker exposure (C). For each calendar quarter, A1C and medication exposure were determined in the preceding quarter. Categories labeled as short-acting insulin and long-acting insulin also include basal-bolus and premixed insulin users.
Conclusions
In this study of nearly 1 million adults with diabetes, we observed rates of severe hypoglycemia requiring medical intervention of 1.4–1.6 per 100 person-years. Consistent with reports from clinical trials, we also noted associations between higher rates of severe hypoglycemia and older age, the presence of specific comorbidities, higher A1C levels, and use of insulin, insulin secretagogues, or β-blockers. Changes in severe hypoglycemia occurrence over time were not clinically significant in the cohort as a whole but were particularly striking in subgroups of individuals with CKD, CHF, and CVD.
Rates of severe hypoglycemia observed in this and other community-based populations are higher than the rates reported in clinical trials among subjects with type 2 diabetes treated by protocol to very low A1C levels (11,12,18,25,28,38). Of particular interest, however, is that our observed rates of severe hypoglycemia were also higher than the rates seen in the standard care arms of the ACCORD, ADVANCE, and Outcome Reduction With Initial Glargine Intervention (ORIGIN) trials (0.3–1 events per 100 person-years) (11,18,28). The differences in severe hypoglycemia rates are likely conservative, because identification by our electronic algorithm required an emergency department visit or inpatient encounter, whereas the definition of severe hypoglycemia in large trials is typically based on requiring the assistance of another person. Potential explanations for this finding may include significant differences in the means of ascertaining the patient populations, exclusion of individuals with a recent or frequent history of hypoglycemia in clinical trials, and the close attention paid to all enrollees in such clinical trials, even individuals within the standard care arms. In addition, our population does include a small percentage of individuals with type 1 diabetes, whereas clinical trials were limited to individuals with type 2 diabetes, or in the case of the ORIGIN trial, also included a minority of patients with prediabetes.
The rates of severe hypoglycemia observed in the current study are considerably higher than those reported in 2011 by Quilliam et al. (26), but similar to those more recently reported in 2014 by Geller et al. (27). Quilliam et al. (26) also assessed a community-treated population but examined younger patients with fewer comorbidities by focusing on those of “working age,” which may explain the lower incidence of hypoglycemia. Quilliam et al. (26) also excluded the 250.x1 diagnostic code, which is for type 1 diabetes but is often erroneously applied to patients with type 2 diabetes on insulin and, therefore, may have inadvertently excluded a substantial proportion of the patients with type 2 diabetes most likely to experience hypoglycemia (26). Geller et al. (27) used data related to emergency department visits and hospitalization from the National Electronic Injury Surveillance System–Cooperative Adverse Drug Event Surveillance Project and data related to insulin use captured by household survey through the National Health Interview Survey to estimate rates of insulin-related hypoglycemia nationwide. In this nationally representative sample, Geller et al. (27) demonstrated rates of severe hypoglycemia ranging from 1.3 to 3.5 per 100 person-years receiving insulin treatment, with the highest rates of hypoglycemia occurring in those older than the age of 80.
The concern over hypoglycemia in patients with diabetes was recently addressed by a working group of the American Diabetes Association and the Endocrine Society (39). In the resulting scientific statement, the working group delineated several important implications of severe hypoglycemia for short- and long-term outcomes among several subgroups of the population with diabetes, including those with type 1 and type 2 diabetes, the elderly, hospitalized patients, and pregnant women. The consequences of hypoglycemia can be more severe in the elderly and those with long-duration diabetes and multiple comorbid conditions (40,41). Of particular concern is the link between recurrent hypoglycemic events and cardiovascular risk, cognitive impairment, and depression (35,39,42,43). In addition, an increased risk of mortality in the 12 months after a severe hypoglycemic event in a large community-dwelling population was recently reported (44). These outcomes affect the cost of care as well as patient quality of life and activities of daily living. Our data support these concerns and confirm that subgroups at greater risk of severe hypoglycemia may require adjustment of A1C goals and additional monitoring.
Consistent with recent findings by Huang et al. (41), we found that rates of severe hypoglycemia were highest in those patients older than 75 years. Young adults between the ages of 20 and 44 also had more severe hypoglycemic incidents than those 45 years and older. We postulate that this younger group is likely to include a higher percentage of individuals with type 1 diabetes than other age-groups, which may account for the higher rate of severe hypoglycemia. We also observed higher rates of severe hypoglycemia in patients with CKD, CVD, CHF, or depression. Among the possible causes of these associations may be a cyclic pattern of persistent health decline, whereby patients with comorbidities may be more likely to suffer from hypoglycemia, only to have their health status further worsened by continued bouts of hypoglycemia (40). Consistent with this finding, Zoungas et al. (45) also found associations between the occurrence of hypoglycemia and numerous adverse clinical outcomes, suggesting that severe hypoglycemia may serve as a marker of vulnerability. Alternatively, this finding could be related to complex medication regimens in the presence of multiple comorbidities or to physiological effects of these health conditions on insulin metabolism. Subjects with CKD, CHF, and CVD had statistically and clinically significant decreases in rates of severe hypoglycemia over time. Potential explanations for this downward trend include increased attention to monitoring for hypoglycemia in susceptible individuals and the development of new drug classes or treatment paradigms that may have mitigated hypoglycemia risk in such patients.
Although β-blockers modify the symptom pattern associated with hypoglycemia, a review of the literature in the early 2000s demonstrated no increased risk of hypoglycemia in the context of cardioselective β-blocker treatment (46). We observed that severe hypoglycemia occurred more often in patients on β-blockers. Although the possibility of confounding by comorbidities (CHF, CHD) is likely and β-blocker type was not assessed, treating physicians may need to take β-blocker therapy into consideration when selecting glucose-lowering therapy and the A1C target. For many patients with diabetes and CVD, hypoglycemia concerns related to β-blockers may be outweighed by their cardiovascular benefits (46).
Several factors limit the interpretation of these data. Although the study population was large, with robust subgroup representation, results may not generalize to other patient populations or care delivery systems, and the observational study design precludes causal inference. Several issues related to accurate identification of hypoglycemic events warrant consideration. We assessed only severe hypoglycemia, defined by a visit to the emergency department or a hospital admission, which admittedly misses many events (e.g., those cared for at home). Nearly 80% of severe hypoglycemia codes were from emergency department encounters, and 20% were from inpatient encounters. Because of the bundling of emergency department and resulting inpatient encounters by some hospitals, some of the codes from inpatient encounters represent codes that originated in the emergency department. We did not attempt to capture less severe hypoglycemic events, making comparisons to studies using other definitions difficult. We also did not capture clinical hypoglycemia events that may have occasionally occurred outside of the SUPREME-DM systems if no bill was submitted. These exclusions may have resulted in underestimating the rates of severe hypoglycemia. Conversely, by including codes from hospital admissions, we are likely to have included some cases of severe hypoglycemia that occurred as a consequence of treatment initiated after hospitalization.
Use of electronic data also limited our ability to distinguish between type 1 and type 2 diabetes, but we assume that the prevalence of type 1 diabetes in the adult population examined here is ∼5%, as previously reported (32). Because the proportion of adults with type 1 diabetes is likely to decrease with age, this may be particularly relevant for findings related to the 20- to 44-year-old age-group. The patients with the highest frequency of hypoglycemic events (≥2 events) had the shortest follow-up, suggesting that rates in the later years of our follow-up could be partially influenced by loss of follow-up of the patients experiencing the most events. Subgroup bias may also have occurred, because the likelihood of emergency department visits may vary by age or other factors. Finally, we collected A1C values and pharmacy data quarterly only, so our linkage of a hypoglycemic event to prior A1C and medication use focuses only on the most recent A1C and medication use and is subject to misclassification.
Despite these limitations, our findings based on nearly 1 million adult patients with diabetes observed over a 7-year study period provide important new information for clinicians and public health experts. We found higher-than-expected rates of severe hypoglycemia in community-treated patients, noted no consistent improvement in trend during the study period, and quantified the excess risk of hypoglycemia in high-risk subgroups, including those on insulin therapy, the elderly, and those with common comorbid conditions. These data can inform the development of clinical management strategies to more effectively reduce the occurrence of severe hypoglycemia in community-treated patients (25,27,41).
Article Information
Acknowledgments. The authors thank Rachel V. Stankowski, PhD, and Marie Fleisner, of the Marshfield Clinic Research Foundation Office of Scientific Writing and Publication, for assistance with manuscript preparation, and Andrea Paolino, of Kaiser Permanente Colorado, for project management. The authors thank the programmers and site investigators at Kaiser Permanente Colorado, Northwest, Northern California, Southern California, Georgia, and Hawaii, Geisinger Health System, Marshfield Clinic Research Foundation, HealthPartners Institute for Education and Research, Henry Ford Health System, and Group Health Research Institute for their efforts at developing and refining the SUPREME-DM data sets that contributed to this work.
Funding. This project was supported by the Agency for Healthcare Research and Quality (AHRQ) grant R01HS019859. The content is solely the responsibility of the authors and does not necessarily represent the official views of the AHRQ. The AHRQ had no role in the design, conduct, or reporting of this work.
Duality of Interest. E.R.S. served as a consultant for Sanofi. G.A.N. received industry-sponsored research support from Novartis, Merck Sharp & Dohme, Boehringer Ingelheim, AstraZeneca, and Bristol-Myers Squibb. No other potential conflicts of interest relevant to this article were reported.
Author Contributions. R.D.P., E.B.S., and C.Z. analyzed data, wrote the manuscript, reviewed and edited the manuscript, and contributed to the discussion. J.D., G.A.N., J.M.L., J.F.S., J.S., and P.J.O. researched data, reviewed and edited the manuscript, and contributed to the discussion. E.R.S., J.E.L., A.T., B.W., and A.J.K. reviewed and edited the manuscript and contributed to the discussion. R.D.P., C.Z., and G.A.N. are the guarantors of this work and, as such, had full access to all the data in the study and take responsibility for the integrity of the data and the accuracy of the data analysis.
References
- 1.American Diabetes Association Standards of medical care in diabetes—2013. Diabetes Care 2013;36(Suppl. 1):S11–S66 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.The Diabetes Control and Complications Trial Research Group The effect of intensive treatment of diabetes on the development and progression of long-term complications in insulin-dependent diabetes mellitus. N Engl J Med 1993;329:977–986 [DOI] [PubMed] [Google Scholar]
- 3.Nathan DM. Some answers, more controversy, from UKPDS. United Kingdom Prospective Diabetes Study. Lancet 1998;352:832–833 [DOI] [PubMed] [Google Scholar]
- 4.Turner RC, Millns H, Neil HA, et al. Risk factors for coronary artery disease in non-insulin dependent diabetes mellitus: United Kingdom Prospective Diabetes Study (UKPDS: 23). BMJ 1998;316:823–828 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.UK Prospective Diabetes Study (UKPDS) Group Intensive blood-glucose control with sulphonylureas or insulin compared with conventional treatment and risk of complications in patients with type 2 diabetes (UKPDS 33). Lancet 1998;352:837–853 [PubMed] [Google Scholar]
- 6.American Diabetes Association Standards of medical care for patients with diabetes mellitus. Diabetes Care 1994;17:616–623 [DOI] [PubMed] [Google Scholar]
- 7.American Diabetes Association Standards of medical care for patients with diabetes mellitus. Diabetes Care 2003;26(Suppl. 1):S33–S50 [DOI] [PubMed] [Google Scholar]
- 8.Blumenthal KJ, Larkin ME, Winning G, Nathan DM, Grant RW. Changes in glycemic control from 1996 to 2006 among adults with type 2 diabetes: a longitudinal cohort study. BMC Health Serv Res 2010;10:158. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Nguyen NT, Nguyen XM, Lane J, Wang P. Relationship between obesity and diabetes in a US adult population: findings from the National Health and Nutrition Examination Survey, 1999-2006. Obes Surg 2011;21:351–355 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Ismail-Beigi F, Craven T, Banerji MA, et al.; ACCORD trial group . Effect of intensive treatment of hyperglycaemia on microvascular outcomes in type 2 diabetes: an analysis of the ACCORD randomised trial. Lancet 2010;376:419–430 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Patel A, MacMahon S, Chalmers J, et al.; ADVANCE Collaborative Group . Intensive blood glucose control and vascular outcomes in patients with type 2 diabetes. N Engl J Med 2008;358:2560–2572 [DOI] [PubMed] [Google Scholar]
- 12.Duckworth W, Abraira C, Moritz T, et al.; VADT Investigators . Glucose control and vascular complications in veterans with type 2 diabetes. N Engl J Med 2009;360:129–139 [DOI] [PubMed] [Google Scholar]
- 13.Gaede P, Lund-Andersen H, Parving HH, Pedersen O. Effect of a multifactorial intervention on mortality in type 2 diabetes. N Engl J Med 2008;358:580–591 [DOI] [PubMed] [Google Scholar]
- 14.Holman RR, Paul SK, Bethel MA, Matthews DR, Neil HA. 10-year follow-up of intensive glucose control in type 2 diabetes. N Engl J Med 2008;359:1577–1589 [DOI] [PubMed] [Google Scholar]
- 15.Chew EY, Ambrosius WT, Davis MD, et al.; ACCORD Study Group; ACCORD Eye Study Group . Effects of medical therapies on retinopathy progression in type 2 diabetes. N Engl J Med 2010;363:233–244 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Turnbull FM, Abraira C, Anderson RJ, et al.; Control Group . Intensive glucose control and macrovascular outcomes in type 2 diabetes. Diabetologia 2009;52:2288–2298 [DOI] [PubMed] [Google Scholar]
- 17.Qaseem A, Vijan S, Snow V, Cross JT, Weiss KB, Owens DK; Clinical Efficacy Assessment Subcommittee of the American College of Physicians . Glycemic control and type 2 diabetes mellitus: the optimal hemoglobin A1c targets. A guidance statement from the American College of Physicians. Ann Intern Med 2007;147:417–422 [DOI] [PubMed] [Google Scholar]
- 18.Action to Control Cardiovascular Risk in Diabetes Study Group; Gerstein HC, Miller ME, Byington RP, et al. Effects of intensive glucose lowering in type 2 diabetes. N Engl J Med 2008;358:2545–2559 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Genuth S, Ismail-Beigi F. Clinical implications of the ACCORD trial. J Clin Endocrinol Metab 2012;97:41–48 [DOI] [PubMed] [Google Scholar]
- 20.Ma J, Yang W, Fang N, Zhu W, Wei M. The association between intensive glycemic control and vascular complications in type 2 diabetes mellitus: a meta-analysis. Nutr Metab Cardiovasc Dis 2009;19:596–603 [DOI] [PubMed] [Google Scholar]
- 21.Lee SJ. So much insulin, so much hypoglycemia. JAMA Intern Med 2014;174:686–688 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Laiteerapong N, Karter AJ, Liu JY, et al. Correlates of quality of life in older adults with diabetes: the diabetes & aging study. Diabetes Care 2011;34:1749–1753 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Lipska KJ, Ross JS, Wang Y, et al. National trends in US hospital admissions for hyperglycemia and hypoglycemia among Medicare beneficiaries, 1999 to 2011. JAMA Intern Med 2014;174:1116–1124 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Gerstein HC, Bosch J, Dagenais GR, et al.; ORIGIN Trial Investigators . Basal insulin and cardiovascular and other outcomes in dysglycemia. N Engl J Med 2012;367:319–328 [DOI] [PubMed] [Google Scholar]
- 25.Lipska KJ, Warton EM, Huang ES, et al. HbA1c and risk of severe hypoglycemia in type 2 diabetes: the Diabetes and Aging Study. Diabetes Care 2013;36:3535–3542 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Quilliam BJ, Simeone JC, Ozbay AB, Kogut SJ. The incidence and costs of hypoglycemia in type 2 diabetes. Am J Manag Care 2011;17:673–680 [PubMed] [Google Scholar]
- 27.Geller AI, Shehab N, Lovegrove MC, et al. National estimates of insulin-related hypoglycemia and errors leading to emergency department visits and hospitalizations. JAMA Intern Med 2014;174:678–686 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.ORIGIN Trial Investigators Predictors of nonsevere and severe hypoglycemia during glucose-lowering treatment with insulin glargine or standard drugs in the ORIGIN trial. Diabetes Care 2015;38:22–28 [DOI] [PubMed] [Google Scholar]
- 29.Mellbin LG, Malmberg K, Waldenström A, Wedel H, Rydén L. DIGAMI 2 investigators. Prognostic implications of hypoglcaemic episodes during hospitalization for myocardial infarction in patients with type 2 diabetes: a report from the DIGAMI 2 trial. Heart 2009;95:721–727 [DOI] [PubMed] [Google Scholar]
- 30.Nichols GA, Desai J, Elston Lafata J, et al.; SUPREME-DM Study Group . Construction of a multisite DataLink using electronic health records for the identification, surveillance, prevention, and management of diabetes mellitus: the SUPREME-DM project. Prev Chronic Dis 2012;9:E110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Ginde AA, Blanc PG, Lieberman RM, Camargo CA Jr. Validation of ICD-9-CM coding algorithm for improved identification of hypoglycemia visits. BMC Endocr Disord 2008;8:4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Centers for Disease Control and Prevention National Diabetes Statistics Report: Estimates of Diabetes and Its Burden in the United States, 2014. Atlanta, GA, U.S. Department of Health and Human Services, 2014 [Google Scholar]
- 33.O’Connor PJ, Rush WA, Peterson J, et al. Continuous quality improvement can improve glycemic control for HMO patients with diabetes. Arch Fam Med 1996;5:502–506 [DOI] [PubMed] [Google Scholar]
- 34.Go AS, Magid DJ, Wells B, et al. The Cardiovascular Research Network: a new paradigm for cardiovascular quality and outcomes research. Circ Cardiovasc Qual Outcomes 2008;1:138–147 [DOI] [PubMed] [Google Scholar]
- 35.Whitmer RA, Karter AJ, Yaffe K, Quesenberry CP Jr, Selby JV. Hypoglycemic episodes and risk of dementia in older patients with type 2 diabetes mellitus. JAMA 2009;301:1565–1572 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Raebel MA, Ellis JL, Schroeder EB, et al. Intensification of antihyperglycemic therapy among patients with incident diabetes: a Surveillance Prevention and Management of Diabetes Mellitus (SUPREME-DM) study. Pharmacoepidemiol Drug Saf 2014;23:699–710 [DOI] [PubMed] [Google Scholar]
- 37.Quan H, Li B, Couris CM, et al. Updating and validating the Charlson comorbidity index and score for risk adjustment in hospital discharge abstracts using data from 6 countries. Am J Epidemiol 2011;173:676–682 [DOI] [PubMed] [Google Scholar]
- 38.Wright AD, Cull CA, Macleod KM, Holman RR; UKPDS Group . Hypoglycemia in type 2 diabetic patients randomized to and maintained on monotherapy with diet, sulfonylurea, metformin, or insulin for 6 years from diagnosis: UKPDS73. J Diabetes Complications 2006;20:395–401 [DOI] [PubMed] [Google Scholar]
- 39.Seaquist ER, Anderson J, Childs B, et al. Hypoglycemia and diabetes: a report of a workgroup of the American Diabetes Association and the Endocrine Society. Diabetes Care 2013;36:1384–1395 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Zammitt NN, Frier BM. Hypoglycemia in type 2 diabetes: pathophysiology, frequency, and effects of different treatment modalities. Diabetes Care 2005;28:2948–2961 [DOI] [PubMed] [Google Scholar]
- 41.Huang ES, Laiteerapong N, Liu JY, John PM, Moffet HH, Karter AJ. Rates of complications and mortality in older patients with diabetes mellitus: the diabetes and aging study. JAMA Intern Med 2014;174:251–258 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Katon WJ, Young BA, Russo J, et al. Association of depression with increased risk of severe hypoglycemic episodes in patients with diabetes. Ann Fam Med 2013;11:245–250 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Fox CS, Golden SH, Anderson C, et al.; American Heart Association Diabetes Committee of the Council on Lifestyle and Cardiometabolic Health, Council on Clinical Cardiology, Council on Cardiovascular and Stroke Nursing, Council on Cardiovascular Surgery and Anesthesia, Council on Quality of Care and Outcomes Research, and the American Diabetes Association . Update on prevention of cardiovascular disease in adults with type 2 diabetes mellitus in light of recent evidence: a scientific statement from the American Heart Association and the American Diabetes Association. Circulation 2015;132:691–718 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Elwen FR, Huskinson A, Clapham L, et al. An observational study of patient characteristics and mortality following hypoglycemia in the community. BMJ Open Diabetes Res Care 2015;3:e000094. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Zoungas S, Patel A, Chalmers J, et al.; ADVANCE Collaborative Group . Severe hypoglycemia and risks of vascular events and death. N Engl J Med 2010;363:1410–1418 [DOI] [PubMed] [Google Scholar]
- 46.Sawicki PT, Siebenhofer A. Betablocker treatment in diabetes mellitus. J Intern Med 2001;250:11–17 [DOI] [PubMed] [Google Scholar]