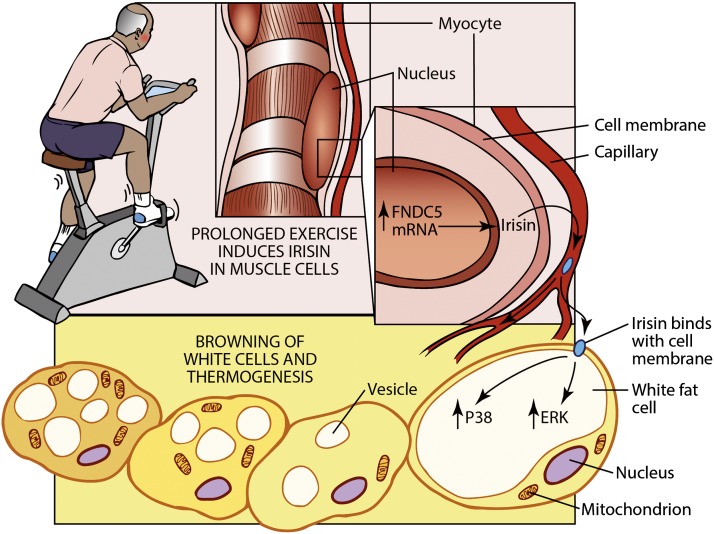
Increasing energy expenditure is an attractive approach to fighting the worldwide epidemic in obesity and type 2 diabetes. Exercise is an important component of good health and represents the first line of therapy for humans with a variety of metabolic disorders: obesity, diabetes, and nonalcoholic hepatic steatosis. Recent data has shown that exercise, besides using calories to do physical work, also causes an increase in energy expenditure through augmentation in brown fat and the browning of white fat (Fig. 1) (1,2). Indeed, these effects on brown fat could represent part of the longer-lasting benefits of exercise.
Figure 1.
Recombinant irisin regulates the thermogenic program in fat through ERK and p38 pathways. Recombinant irisin produced in yeast is glycosylated and active. It induces the thermogenic gene program in 3T3-L1 cells and primary subcutaneous adipocytes. In vivo treatments of this recombinant protein in mice show strong anti-obesity effects and improve systematic glucose homeostasis.
That brown fat, in all of its dimensions, can improve type 2 diabetes and metabolic health seems to be settled science, at least in experimental animals (3). These cells express UCP1 and have a high mitochondrial content, thereby dissipating chemical energy in the form of heat. In fact, the improvements seen in glucose tolerance observed with “browning” of white fat and the formation of “beige” or “brite” cells might be greater than expected solely from their effects on body weight and adiposity (4). The confirmed presence of UCP1+ brown fat in humans has added to the interest in finding methods and molecules that can augment energy expenditure through browning of beige fat cells (5–7). Several polypeptides, including FGF21, BMP7/8b, BNP/ANP, and orexin, all have interesting browning effects (8–12). Irisin was of interest because it is induced during exercise in rodents and is at least partially responsible for the browning response observed in white fat during chronic exercise (2). The parent polypeptide, FNDC5, is synthesized as a type 1 membrane protein and is then cleaved and shed into the circulation as a highly glycosylated polypeptide of roughly 12kDa. Irisin appears to act preferentially on the browning of white fat deposits when elevated in the blood of obese mice via viral vectors. This correlates with improvements in glucose tolerance in obese mice. Regarding human irisin, it is clear that FNDC5 mRNA is increased in skeletal muscle in some exercise paradigms but not others (2,13,14). Interestingly, two articles report that human patients with diabetes are deficient in irisin compared with normal counterparts (15,16). Because the human irisin mRNA has an AUA start codon in the precise location where other species have a classical ATG start codon, the possibility that the human gene might not encode a protein has been raised (17), though the large number of studies measuring human irisin in blood with different antibodies and methods would seem to close this issue (15,16,18–22).
In this issue, Zhang et al. (23) addressed the signal transduction pathways by which irisin drives the browning of white fat cells. This article used the mammalian irisin produced in yeast cells and found that it is both heavily glycosylated and biologically active when placed on either 3T3-L1 cells or primary cultures from the rat inguinal depot. The effects on the 3T3-L1 cultures are especially impressive because these cells are generally viewed as very “white,” or not prone to the induction of mRNA encoding UCP1 and other thermogenic genes. The article shows rather convincingly that these browning effects depend on the activation of extracellular signal–related kinase (ERK) and p38 protein kinase signaling cascades. While both of these kinases have been implicated previously in the thermogenic actions of other agents on brown fat, including β-adrenergic agonists and FGF21, the role in irisin action was not known (11,24,25). The signal transduction through ERK and p38 occurs within 20 min after irisin is added to the cell culture. The swift response and the evidence that irisin directly binds to the cell membrane alludes to a yet-to-be-identified irisin receptor present in both primary inguinal cells and 3T3-L1 cells. Further studies will illustrate how the expression and activation of this receptor is regulated under physiological (exercise) and/or pathological (metabolic diseases) conditions. Of importance, Zhang et al. further demonstrate that mutation of either glycosylation site of irisin compromised its activity; whether this is due to a strict requirement of these modifications for (putative) receptor binding or whether they influence protein folding/solubility was not addressed.
Last and of import, Zhang et al. (23) gave irisin by injection daily for 2 weeks and saw strong changes in body weight, browning of the adipose tissues, and improvements in glucose tolerance. While these data are consistent with our earlier studies using viral vectors, showing these effects with a stable version of the protein is a very substantial step in the direction of human therapeutics.
Scale up and production of recombinant proteins in yeast is well established, so this new irisin reagent will be of great interest to the fields of diabetes, metabolism, and exercise science. Exercise, of course, benefits other disorders of the liver, heart, muscle, and brain. It will be of great interest to apply these and other irisin preparations to models of other disease states. Identification of the irisin receptor will also open new possibilities for activation of these areas.
Article Information
Duality of Interest. B.M.S. is a consultant to and shareholder in Ember Therapeutics, Inc. No other potential conflicts of interest relevant to this article were reported.
Footnotes
See accompanying original article, p. 514.
References
- 1.Xu X, Ying Z, Cai M, et al. Exercise ameliorates high-fat diet-induced metabolic and vascular dysfunction, and increases adipocyte progenitor cell population in brown adipose tissue. Am J Physiol Regul Integr Comp Physiol 2011;300:R1115–R1125 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Boström P, Wu J, Jedrychowski MP, et al. A PGC1-α-dependent myokine that drives brown-fat-like development of white fat and thermogenesis. Nature 2012;481:463–468 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Enerbäck S. Adipose tissue metabolism in 2012: Adipose tissue plasticity and new therapeutic targets. Nat Rev Endocrinol 2013;9:69–70 [DOI] [PubMed] [Google Scholar]
- 4.Seale P, Conroe HM, Estall J, et al. Prdm16 determines the thermogenic program of subcutaneous white adipose tissue in mice. J Clin Invest 2011;121:96–105 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Cypess AM, Lehman S, Williams G, et al. Identification and importance of brown adipose tissue in adult humans. N Engl J Med 2009;360:1509–1517 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.van Marken Lichtenbelt WD, Vanhommerig JW, Smulders NM, et al. Cold-activated brown adipose tissue in healthy men. N Engl J Med 2009;360:1500–1508 [DOI] [PubMed] [Google Scholar]
- 7.Virtanen KA, Lidell ME, Orava J, et al. Functional brown adipose tissue in healthy adults. N Engl J Med 2009;360:1518–1525 [DOI] [PubMed] [Google Scholar]
- 8.Fisher FM, Kleiner S, Douris N, et al. FGF21 regulates PGC-1α and browning of white adipose tissues in adaptive thermogenesis. Genes Dev 2012;26:271–281 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Tseng YH, Kokkotou E, Schulz TJ, et al. New role of bone morphogenetic protein 7 in brown adipogenesis and energy expenditure. Nature 2008;454:1000–1004 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Whittle AJ, Carobbio S, Martins L, et al. BMP8B increases brown adipose tissue thermogenesis through both central and peripheral actions. Cell 2012;149:871–885 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Bordicchia M, Liu D, Amri EZ, et al. Cardiac natriuretic peptides act via p38 MAPK to induce the brown fat thermogenic program in mouse and human adipocytes. J Clin Invest 2012;122:1022–1036 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Sellayah D, Bharaj P, Sikder D. Orexin is required for brown adipose tissue development, differentiation, and function. Cell Metab 2011;14:478–490 [DOI] [PubMed] [Google Scholar]
- 13.Pekkala S, Wiklund P, Hulmi JJ, et al. Are skeletal muscle FNDC5 gene expression and irisin release regulated by exercise and related to health? J Physiol 2013;591:5393–5400 [DOI] [PMC free article] [PubMed]
- 14.Lecker SH, Zavin A, Cao P, et al. Expression of the irisin precursor FNDC5 in skeletal muscle correlates with aerobic exercise performance in patients with heart failure. Circ Heart Fail 2012;5:812–818 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Park KH, Zaichenko L, Brinkoetter M, et al. Circulating irisin in relation to insulin resistance and the metabolic syndrome. J Clin Endocrinol Metab. 20 September 2013 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Choi YK, Kim MK, Bae KH, et al. Serum irisin levels in new-onset type 2 diabetes. Diabetes Res Clin Pract 2013;100:96–101 [DOI] [PubMed] [Google Scholar]
- 17.Raschke S, Elsen M, Gassenhuber H, et al. Evidence against a beneficial effect of irisin in humans. PLoS One 2013;8:e73680. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Vamvini MT, Aronis KN, Panagiotou G, et al. Irisin mRNA and circulating levels in relation to other myokines in healthy and morbidly obese humans. Eur J Endocrinol 2013;169:829–834 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Huh JY, Panagiotou G, Mougios V, et al. FNDC5 and irisin in humans: I. Predictors of circulating concentrations in serum and plasma and II. mRNA expression and circulating concentrations in response to weight loss and exercise. Metabolism 2012;61:1725–1738 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Gouni-Berthold I, Berthold HK, Huh JY, et al. Effects of lipid-lowering drugs on irisin in human subjects in vivo and in human skeletal muscle cells ex vivo. PLoS One 2013;8:e72858. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Swick AG, Orena S, O’Connor A. Irisin levels correlate with energy expenditure in a subgroup of humans with energy expenditure greater than predicted by fat free mass. Metabolism 2013;62:1070–1073 [DOI] [PubMed] [Google Scholar]
- 22.Stengel A, Hofmann T, Goebel-Stengel M, Elbelt U, Kobelt P, Klapp BF. Circulating levels of irisin in patients with anorexia nervosa and different stages of obesity—correlation with body mass index. Peptides 2013;39:125–130 [DOI] [PubMed] [Google Scholar]
- 23.Zhang Y, Li R, Meng Y, et al. Irisin stimulates browning of white adipocytes through mitogen-activated protein kinase p38 MAP kinase and ERK MAP kinase signaling. Diabetes 2014;63:514–525 [DOI] [PubMed] [Google Scholar]
- 24.Ye L, Kleiner S, Wu J, et al. TRPV4 is a regulator of adipose oxidative metabolism, inflammation, and energy homeostasis. Cell 2012;151:96–110 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Cao W, Daniel KW, Robidoux J, et al. p38 Mitogen-activated protein kinase is the central regulator of cyclic AMP-dependent transcription of the brown fat uncoupling protein 1 gene. Mol Cell Biol 2004;24:3057–3067 [DOI] [PMC free article] [PubMed] [Google Scholar]