Abstract
The four genetically divergent dengue virus (DENV) types are traditionally classified as serotypes. Antigenic and genetic differences among the DENV types influence disease outcome, vaccine-induced protection, epidemic magnitude, and viral evolution. We characterized antigenic diversity in the DENV types by antigenic maps constructed from neutralizing antibody titers obtained from African green monkeys and after human vaccination and natural infections. Genetically, geographically, and temporally, diverse DENV isolates clustered loosely by type, but we found many are as similar antigenically to a virus of a different type as to some viruses of the same type. Primary infection antisera did not neutralize all viruses of the same DENV type any better than other types did up to two years after infection and did not show improved neutralization to homologous type isolates. That the canonical DENV types are not antigenically homogenous has implications for vaccination and research on the dynamics of immunity, disease, and the evolution of DENV.
Dengue virus (DENV) infects up to 390 million people each year, and of the 96 million individuals who develop an acute systemic illness, approximately 500,000 experience potentially life-threatening complications, including hemorrhage and shock (1, 2). The four genetic DENV types have long been thought to exist as four serotypes, and the antigenic differences between the types are believed to have a key role in the severity of disease, epidemic magnitude, viral evolution, and design of vaccines (3–5).
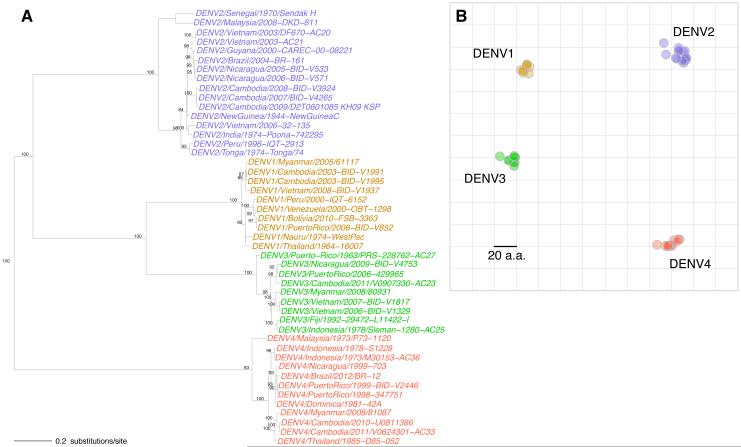
The description of DENV types as serotypes originated with the observation that the human immune response following primary DENV infection fully protected against challenge with viruses of the homologous type but only partially, and transiently, protected against challenge by viruses of a heterologous type (6). This finding was supported by in vitro neutralization experiments in which each DENV type was on average better neutralized by homologous than heterologous DENV infection antisera (7). The immune response immediately after a primary D ENV infection varied from individual to individual, but generally was characterized by high levels of neutralizing antibody titers to multiple DENV types. The neutralizing response was observed to become more DENV type-specific over time (8). It was later shown that antibodies to a heterologous DENV type could enhance infection in vivo and were associated with increased risk of severe disease in nature (9, 10). Although antigenic variability was observed within DENV types from the earliest studies, this variation is generally considered to be substantially less than the differences between types, and not thought to modify type-specific protection (11, 12). Together, the DENV types clearly form an antigenic subgroup within the Flaviviridae (13, 14). Analyses of envelope (E) proteins, and later full genomes, showed that the four types are as genetically divergent among themselves as sequences assigned to different viruses within the genus Flavivirus (15). These deep evolutionary divergences between DENV types were evident in the phylogenetic tree of the genetically diverse E-gene sequences of the viruses we investigated here (Fig. 1A; fig. S1; and table S1) (16). Similarly, a map of amino acid differences between the E proteins revealed four compact, segregated types (Fig. 1B and fig. S2), as the number of amino acid substitutions between heterologous types far exceeded the maximum difference within a type.
Fig. 1.
Genetic analyses of the DENV panel (n=47). (A) Phylogenetic tree showing the evolutionary relationships of DENV E gene sequences. Sequences were aligned with MAFFT, and a maximum likelihood tree (ML) was estimated using a general time reversible model, accounting for both among site rate variation and invariant sites (GTR+G4+I). Bootstrap support values of at least 75% are shown. (B) Amino acid map of dengue E protein sequences (493-495 amino acids in length). The total amino acid differences between pairs of E sequences correspond to distances between points on the geometric display.
However, investigations that rely on the classification of DENV into serotypes do not fully explain clinical and epidemiological phenomena. Despite this, antigenic properties are still thought to play a critical role in the biology of DENV infections. One hypothesis is that antigenic differences are critical, but that categorization by serotype alone is too coarse a measure. For example, differences in epidemic magnitude might be determined not only by the serotype but also by the antigenic differences between the particular infecting viruses that populations experience during sequential epidemics. Antigenic variation within and among the DENV types has also been hypothesized, in addition to intrinsic viral fitness and other factors, to explain phenomena including extinction and replacement of previously successful lineages and variation in disease outcome caused by genetically similar viruses (17–19). Here, we empirically test the antigenic relationships among a panel of diverse DENV isolates and re-examine the serotype concept.
Antigenic differences among viruses are caused by amino acid differences that lead to structural changes on viral proteins that modify antibody binding. The structural effect of such amino acid substitutions is difficult to predict from genetic sequences alone. In some instances substitutions have no antigenic effect, sometimes single substitutions cause substantial antigenic change, and other times it takes multiple substitutions (20, 21). Thus today, antigenic differences must be determined by phenotype, including by an antibody neutralization assay (13). Most often, viruses are measured against multiple sera to form a table of neutralization data from which antigenic relationships are inferred (22). However, such inferences are notoriously difficult to make, and this has hindered the reliable systematic antigenic characterization of DENV. The difficulties are caused by random error, the use of diverse methods among laboratories, and the intrinsic variability among immune sera due to differences in hosts and infection histories (23, 24). Moreover, neutralization data often contain apparent contradictions that are difficult to interpret, such as higher-than-homologous titers and sera that similarly neutralize multiple DENV types.
Previous antigenic analyses of DENV have addressed such challenges by using monoclonal antibodies, averaging responses of many individuals, or excluding sera with unusual patterns of reactivity. Despite careful work, these approaches have not produced a unified framework for understanding patterns across large neutralization data sets. Antigenic cartography is a method that positions viruses and antisera as points in a map, such that the distance between each virus and antiserum is derived from the corresponding neutralization titer in the tabular data. This method exploits variation in host responses to better triangulate the map, reduces the effect of some measurement errors because each virus is measured against multiple antisera (and vice versa), and has been shown to accurately interpret apparent contradictions in the data (25).
We formed the Dengue Antigenic Cartography Consortium, an open collaboration of international research laboratories, to establish empirically how DENV types relate to one another antigenically. Thirty-six African green monkeys (Chlorocebus sabaeus, hereafter NHP) were experimentally inoculated with diverse DENV isolates, and their sera were tested for neutralizing antibody potency against the genetically (all known genotypes), temporally (1944-2012), and geographically (20 countries) diverse panel of DENV isolates shown in Fig. 1 (table S1). Serum samples were taken three months post-inoculation, and titrations were conducted using an immunofocus reduction neutralization test on mosquito cells (C6/36, Aedes albopictus) (tables S2-S7 and fig. S3) (16, 26). A conventional interpretation of the raw antibody neutralization titers was consistent with previous observations, both for DENV and for other flaviviruses: antisera were generally able to neutralize viruses of the infecting type better than heterologous types.
The cartographic analyses fit these data with low error and were internally consistent (figs. S4, S6, and S7). Only 1% of map distances differed by more than four-fold from the measured titer (table S8). The positions of viruses and antisera were robust to different methods of calculating neutralization titers and to the exclusion of outliers (figs. S5, S8-S12 and table S10). Maps made with random subsets of the data set could predict excluded titers within two-fold error (r=0.90 for the relation between all measured and predicted titers) (table S9).
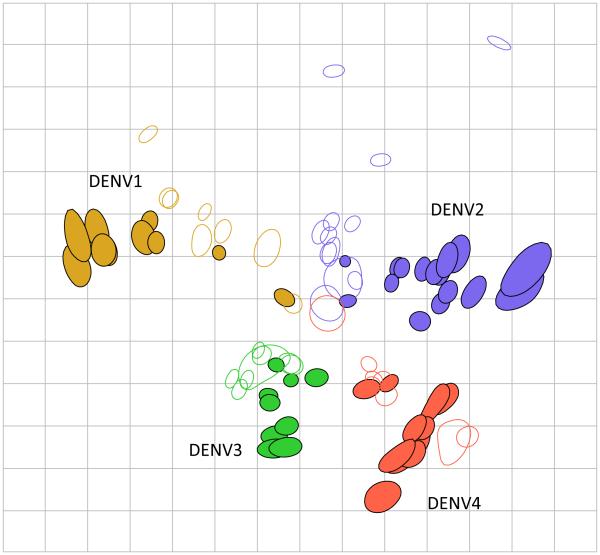
Our analyses showed that the DENV isolates in our panel did group according to current serotype classification (Fig. 2), and the majority of viruses neighboring any given virus are of the same DENV type. However, many of the viruses were positioned as close to a virus of another DENV type as to some viruses of their own type, and the distance within and between types was comparable. Similarly, while neutralizing antisera responses clustered closely to viruses of the homologous type, almost all were at least as close to a heterologous-type isolate (table S11, table S12).
Fig. 2.
Antigenic map of the DENV panel (n=46) titrated against three-month post-infection African green monkey antisera (n=36). Each unit of antigenic distance (length of one grid-square side, measured in any direction) is equivalent to a two-fold dilution in the neutralization assay. Each antiserum (open shape) and virus (closed shape) is colored according to the infecting genetic type (16). The size and shape of each point is the confidence area of its position.
To examine these findings in detail, we evaluated whether the observed antigenic diversity of the virus types was also observed with human antisera and over time, and whether the neutralizing responses of individual antisera became increasingly type-specific over time.
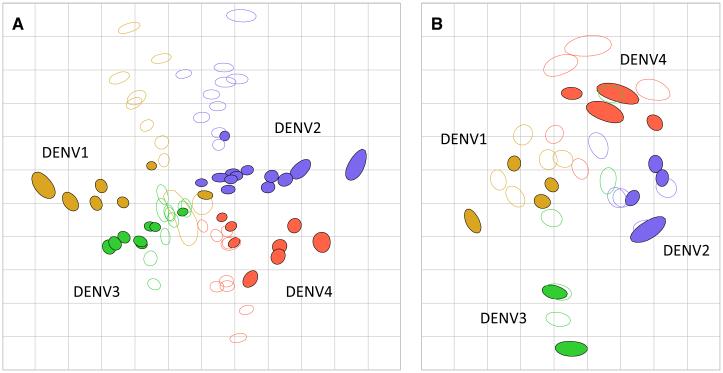
We titrated human antisera derived from vaccination with a live-attenuated chimeric DENV vaccine against the genetically diverse DENV panel. Individuals lacking detectable neutralizing antibodies against DENV or other flaviviruses were each inoculated with one monovalent component of the National Institutes of Health DENV vaccine (n=40 in total, 10 per DENV type). Antisera drawn 42 days post-injection were titrated against the DENV panel (n=36) using the neutralization test on mosquito cells. The resulting antigenic map is consistent with the NHP map in that the distance between DENV types was equivalent to the spread within type, and the overall orientation of DENV1-4 was the same (Fig. 3A).
Fig. 3.
Human primary infection antigenic maps. (A) Antisera from individuals inoculated with each monovalent component of the NIH live vaccine (10 per group) were drawn 42 days post-infection and titrated against 36 viruses in the DENV panel. (B) Antisera from 20 Nicaraguan children drawn in the year after their first DENV infections were titrated against an antigenically diverse subset of the DENV panel (n=14).
We measured the antigenic relationships among the DENV panel as recognized by antisera drawn from naturally-infected individuals, who had neutralizing responses representative of the cohort study from which they were selected. Serum samples drawn from 20 Nicaraguan children in the year following their first DENV infection were titrated, using the neutralization test on mosquito cells, against 14 viruses that captured the breadth of variation seen in the DENV panel in Fig. 2. Again, the antigenic distances among the DENV types were similar to those observed with NHP and human vaccine antisera, although the DENV4 cluster was positioned adjacent to DENV1 and DENV2 (Fig. 3B).
We also analyzed neutralization data from other studies that had used antisera from monovalent vaccine recipients and naturally infected human travelers, as well as different neutralization assays (22, 27, 28). Again, the antisera from these studies also recognized the antigenic relationships among the DENV isolates similarly to the three-month NHP antisera (figs. S23-S25).
The early antibody response is assumed to broadly neutralize all DENV types, but over time cross-type neutralization is thought to be lost so that the antibody response remaining in the months to years after infection only potently neutralizes isolates of the infecting type (8, 29, 30). We compared how antisera taken at various time points after infection recognize antigenic relationships among the DENV panel. The human antisera used to make the antigenic maps described above were taken at various times following infection, ranging from 42 days for the monovalent vaccine antisera to more than one year for the natural infection antisera. We also made an antigenic map of a published neutralization data set of 44 DENV isolates titrated with one-year post-inoculation monkey antisera and found a similar range of antigenic variants among the four DENV types (fig. S26) (12). Thus, in maps made with early (one month) as well as late convalescent (three months to one year) antisera, the antigenic relationships among diverse DENV isolates were similar to those observed with three-month NHP antisera.
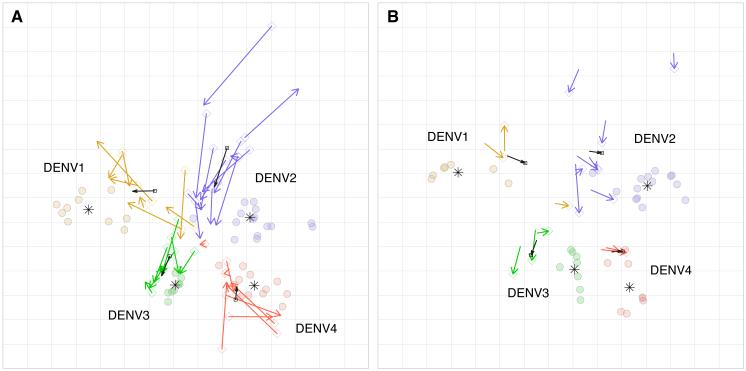
We tested if the patterns of antigenic recognition of the antisera from serially sampled individuals changed with time. We titrated antisera from the experimentally inoculated NHPs one month (n=36) and five months (n=16) post-infection against the DENV panel. As expected, the magnitude of the neutralizing titers generally dropped between one, three, and five months (table S14). However, viruses on the one and five-month antigenic maps showed the same orientation of types as the three-month antisera. At one month after infection, 55%, and at five months after infection, 41% of the viruses, respectively, clustered as closely to a virus in a heterologous type as to some viruses of the same type (Fig. 4A and B; table S11; table S13; and table S15). The antigenic relationships among isolates were conserved across time-points (fig. S13). We thus found that the antigenic relationships among the isolates in the DENV panel were recognized similarly by early and late convalescent antisera from the same individuals.
Fig. 4.
Antigenic maps of the DENV panel made with antisera drawn from NHPs one and five months post-infection. (A) An antigenic map of 47 DENV isolates titrated against 36 NHP antisera drawn one month post-infection. Colored arrows (DENV1=yellow, DENV2=blue, DENV3=green, DENV4=red) show the change in antiserum positions between one and three months. The black arrows show the average shift in serum position for each DENV type. The star denotes the antigenic center for each DENV type. (B) An antigenic map of 37 DENV isolates titrated against 16 NHP antisera drawn five months post-infection. Arrows point from positions of antisera at three months to the corresponding five-month positions.
We measured changes in neutralizing type-specificity for each NHP by comparing the antiserum positions in the one, three, and five-month antigenic maps. The antiserum positions shifted (on average, greater than four-fold) between one month and three months, consistent with the period of somatic hypermutation and selection for affinity matured B cells (Fig. 4A and fig. S14). However, few antisera showed improved neutralization of the infecting DENV type relative to heterologous types between one and three months. The antiserum positions changed minimally between three months and five months, despite a significant decline in the magnitude of titers over that period, in some cases below the assay limit of detection (Fig. 4B and table S14). Thus, we did not observe a systematic shift toward increasing neutralizing specificity to viruses of the infecting type nor decreasing specificity toward heterotypic viruses (fig. S15 and fig. S21).
Published studies of neutralizing responses in the first year after experimental inoculation also reported stability of neutralization specificity. In one study, the ratio between homologous and heterologous neutralizing titers for 16 Rhesus monkeys between 4-13 months after experimental inoculation was remarkably consistent. NHPs that were initially type-specific remained so, while those that exhibited early cross-type titers maintained titers to those types to the end of the study period (fig. S28) (31). A second study following the neutralizing responses of Aotus nancymae monkeys for 1-4 months to DENV1 and DENV2 isolates showed similarly stable neutralization specificity to the infecting type and heterologous types (fig. S29) (32).
We further analyzed the neutralizing responses in the natural human infection data set to look at the type-specificity of antisera obtained during the first two years after infection. The antisera in the map in Fig. 3B ranged in neutralizing type-specificity, with 55% of antisera responses clustering as closely to a heterologous isolate as some homologous isolates. For each individual, the serum position in Fig. 3B, made with titrations conducted on mosquito cells, closely corresponded to the serum position in the map made with titrations using human cells expressing the DENV attachment factor, DC-SIGN (Fig. 3B and fig. S16). The position of the DENV4 cluster was between DENV1 and DENV2 on both maps (Fig. 3B and fig. S16). We compared the antibody titrations after one and two years for each individual, and found that all maintained the pattern of neutralization, including cross-neutralization, observed in the first year after infection (fig. S17 and S18). Thus, neutralizing antibody responses in natural human DENV infections did not show a trend toward increasing type-specificity even two years after infection.
Type-specific and cross-reactive neutralizing antibodies are thought to target distinct viral structures, and thus potentially may produce different antigenic maps (33). We therefore tested whether cross-reactive neutralizing antisera recognized different antigenic relationships among the DENV panel than type-specific neutralizing responses, using the serum positions of the monovalent vaccine map (Fig. 3A). Despite the fact that all ten individuals for each DENV type were inoculated with the same vaccine component, the antisera responses to the isolates varied. Collectively, the antisera provided a coherent description of antigenic patterns among the isolates (fig. S19). The relationships among the DENV panel changed minimally between maps made with only the most central, cross-reactive 20 antisera or only the most peripheral, type-specific 20 antisera (fig. S20 and fig. S22). Thus, the DENV type-specific and cross-reactive neutralizing responses recognized the same antigenic relationships among the DENV panel.
The antigenic characterization of any pathogen relies on the biological relevance of the assay used to generate the data. Both recent and historical studies have found significant associations between pre-infection neutralization titers and DENV viremia or infection outcome (34–37); however, other studies have been inconclusive (38, 39). Thus, the identification of immune correlates of protection including, but not exclusively, potently neutralizing antibodies, is an active area of research for DENV (40–42). Notably, the antigenic patterns in our data are similar to those in antigenic maps we made of DENV antibody neutralization data from other published studies using different cell lines, virus preparations, methods for detecting infected cells, and plaque or immunofocus reduction end-points (figs. S23-S27) (12, 19, 22, 27, 28). We also found that the human antisera from natural infections titrated on mosquito cells showed similar neutralization profiles to those titrated on human cells (fig. S16 and S18). The antigenic variation we observed is thus not limited to the assay or samples that we used.
While overall, prior immunity to a heterologous DENV type still remains the strongest risk factor for disease, there is evidence that neutralizing responses to the particular DENV lineages circulating in a population modifies the magnitude and severity of epidemics caused by subsequent infecting lineages (17, 18). In one study, cross-type neutralization provided by prior DENV1 immunity correlated with a mild epidemic caused by one lineage of DENV2, but showed no neutralization of other DENV2 lineages that in immunologically similar populations caused severe epidemics (fig. S27) (19). These, and our, studies highlight the importance of studying the specific relationship between antigenic distances as measured with neutralizing antibody titers and protection. The approach described here, in combination with global surveillance of the genetic, antigenic, and clinical features of DENVs as well as further detailed studies of natural infection and vaccination-derived protection, has the potential to inform whether vaccination protects against circulating isolates as well as recognize gaps in vaccine-induced protection should they emerge over time.
The antigenic analyses shown here using one, three, and five-month NHP antisera, human monovalent vaccine antisera, late-convalescent human natural infection antisera, and published neutralization data show that the DENV types do not fall into order as distinct serotypes. We have found that while DENV isolates are usually located closer to other viruses of the same type, some viruses, both modern and historical, have greater antigenic resemblance to viruses of a different type than to some viruses of the same type. We find that primary infection neutralizing antibody titers, although they drop in magnitude, do not systematically become more type-specific in the year after primary infection. As expected, individuals infected with the same or different antigens have variable patterns of neutralization, but cross-neutralizing responses consistently recognize the same antigenic relationships within the DENV panel as do the neutralizing responses that are most type-specific. These findings shift our understanding of the antigenic properties of DENV, enable more detailed study of the antigenic determinants of clinical severity, epidemic magnitude, and DENV evolution, and provide additional methods for the selection of future vaccine strains and global surveillance of the antigenic dynamics of dengue viruses.
Supplementary Material
Acknowledgments
We wish to express our gratitude to the members of the Dengue Antigenic Cartography Consortium, named in the supplementary materials, for their advice and contributions to the Consortium to date. We thank D. Burke, N. Lewis, E. Selkov, E. Skepner, A. Mosterín, R. Mögling, S. Wilks, T. Kotarba, and V. Duong for their technical expertise. M. Melendrez, J. Hang, R. Jarman, S.M. Cave, S.G. Widen, T.G. Wood, and V. Duong assisted with virus sequencing. C. Firestone and M. Galvez assisted with neutralization assay titrations. This research was supported in part by the Intramural Research Program of the US NIH, National Institute of Allergy and Infectious Diseases, European Union (EU) FP7 programs EMPERIE (223498) and ANTIGONE (278976), Human Frontier Science Program (HFSP) program grant P0050/2008, the NIH Director’s Pioneer Award DP1-OD000490-01, the FIRST program from the Bill and Melinda Gates Foundation and the Instituto Carlos Slim de la Salud (E.H.). The antigenic cartography toolkit was in part supported by NIAID-NIH Centers of Excellence for Influenza Research and Surveillance contracts HHSN266200700010C and HHSN272201400008C for use on influenza virus. L.C.K. was supported by the Gates Cambridge Scholarship and the NIH Oxford Cambridge Scholars Program. J.M.F. was supported by an MRC Fellowship (MR/K021885/1) and a Junior Research Fellowship from Homerton College Cambridge. E.C.H. was supported by an NHMRC Australia Fellowship. N.V. and R.B.T were supported by NIH contract HHSN272201000040I/HHSN27200004/D04. The viruses and sera used in this study are covered by standard material transfer agreements at the home institutions of S.S.W., C.P.S., E.H., P.B., J.G.A., J.L.M.J., N.V., and R.B.T. A.D.M.E.O. is Professor/Director Artemis One Health Utrecht, The Netherlands; CSO Viroclinics Biosciences BV, The Netherlands Board Member Protein Sciences USA. P.B. performed this work while at the Institut Pasteur in Cambodia, but since June 2015, is with GlaxoSmithKline vaccines in Singapore, and has stock options with GSK. C.P.S. is a paid consultant to GSK Pharma, GSK Vaccines, and Merck, and has received a grant and consulting payments to his institution from Sanofi Pasteur. The sequences used in this study are available from GenBank (http://www.ncbi.nlm.nih.gov/genbank/) and are listed in Table S1. Files used for genetic analyses are available as supplementary data files. The NIH monovalent DENV vaccines trials (ClinicalTrials.gov identifiers: NCT00473135 NCT00920517, NCT00831012, NCT00831012) were performed under an investigational new drug application reviewed by the US Food and Drug Administration and approved by the Institutional Review Board at the University of Vermont and Johns Hopkins University. Informed consent was obtained in accordance federal and international regulations (21CFR50, ICHE6). The Pediatric Dengue Cohort Study in Managua, Nicaragua was approved by the Institutional Review Boards of the Nicaraguan Ministry of Health and the University of California, Berkeley. Parents or legal guardians of all subjects provided written informed consent, and subjects 6 years of age and older provided assent.
References and Notes
- 1.Bhatt S, et al. The global distribution and burden of dengue. Nature. 2013;496:504–7. doi: 10.1038/nature12060. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.WHO/TDR . Dengue: Guidelines for Diagnosis, Treatment, Prevention and Control. Geneva, Switzerland: 2009. [PubMed] [Google Scholar]
- 3.Chau TNB, et al. Dengue in Vietnamese infants--results of infection-enhancement assays correlate with age-related disease epidemiology, and cellular immune responses correlate with disease severity. J. Infect. Dis. 2008;198:516–524. doi: 10.1086/590117. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Lanciotti RS, Gubler DJ, Trent DW. Molecular evolution and phylogeny of dengue-4 viruses. 1997:2279–2286. doi: 10.1099/0022-1317-78-9-2279. [DOI] [PubMed] [Google Scholar]
- 5.Zhang C, et al. Clade Replacements in Dengue Virus Serotypes 1 and 3 Are Associated with Changing Serotype Prevalence †. 2005;79:15123–15130. doi: 10.1128/JVI.79.24.15123-15130.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Sabin AB. Research on dengue during World War II. Am. J. Trop. Med. Hyg. 1952;1:30–50. doi: 10.4269/ajtmh.1952.1.30. [DOI] [PubMed] [Google Scholar]
- 7.Hammon WM, Rudnick A, Sather GE. Viruses associated with epidemic hemorrhagic fevers of the Philippines and Thailand. Science. 1960;131:1102–1103. doi: 10.1126/science.131.3407.1102. [DOI] [PubMed] [Google Scholar]
- 8.Guzman MG, et al. Neutralizing antibodies after infection with dengue 1 virus. Emerg. Infect. Dis. 2007;13:282–6. doi: 10.3201/eid1302.060539. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Halstead SB. In vivo enhancement of dengue virus infection in rhesus monkeys by passively transferred antibody. J. Infect. Dis. 1979;140:527–533. doi: 10.1093/infdis/140.4.527. [DOI] [PubMed] [Google Scholar]
- 10.Sangkawibha N, et al. Risk factors in dengue shock syndrome: a prospective epidemiologic study in Rayong, Thailand. I. The 1980 outbreak. Am. J. Epidemiol. 1984;120:653–669. doi: 10.1093/oxfordjournals.aje.a113932. [DOI] [PubMed] [Google Scholar]
- 11.Gentry MK, Henchal EA, McCown JM, Brandt WE, Dalrymple JM. Identification of distinct antigenic determinants on dengue-2 virus using monoclonal antibodies. Am. J. Trop. Med. Hyg. 1982;31:548–555. doi: 10.4269/ajtmh.1982.31.548. [DOI] [PubMed] [Google Scholar]
- 12.Russell PK, Nisalak A. Dengue virus identification by the plaque reduction neutralization test. J. Immunol. 1967 [PubMed] [Google Scholar]
- 13.Calisher CH, et al. Antigenic relationships between flaviviruses as determined by cross-neutralization tests with polyclonal antisera. J. Gen. Virol. 1989;70:37–43. doi: 10.1099/0022-1317-70-1-37. Pt 1. [DOI] [PubMed] [Google Scholar]
- 14.Mansfield KL, et al. Flavivirus-induced antibody cross-reactivity. J. Gen. Virol. 2011;92:2821–2829. doi: 10.1099/vir.0.031641-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Holmes EC, Twiddy SS. The origin, emergence and evolutionary genetics of dengue virus. Infect. Genet. Evol. 2003;3:19–28. doi: 10.1016/s1567-1348(03)00004-2. [DOI] [PubMed] [Google Scholar]
- 16. “Supplementary Materials.”.
- 17.OhAinle M, et al. Dynamics of dengue disease severity determined by the interplay between viral genetics and serotype-specific immunity. Sci. Transl. Med. 2011;3:114ra128. doi: 10.1126/scitranslmed.3003084. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Adams B, et al. Cross-protective immunity can account for the alternating epidemic pattern of dengue virus serotypes circulating in Bangkok. Proc. Natl. Acad. Sci. 2006;103:14234–14239. doi: 10.1073/pnas.0602768103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Kochel TJ, et al. Effect of dengue-1 antibodies on American dengue-2 viral infection and dengue haemorrhagic fever. Lancet. 2002;360:310–2. doi: 10.1016/S0140-6736(02)09522-3. [DOI] [PubMed] [Google Scholar]
- 20.Koel BF, et al. Substitutions near the receptor binding site determine major antigenic change during influenza virus evolution. Science. 2013;342:976–9. doi: 10.1126/science.1244730. [DOI] [PubMed] [Google Scholar]
- 21.VanBlargan LA, et al. The type-specific neutralizing antibody response elicited by a dengue vaccine candidate is focused on two amino acids of the envelope protein. PLoS Pathog. 2013;9:e1003761. doi: 10.1371/journal.ppat.1003761. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Vasilakis N, et al. Short Report!: Antigenic Relationships between Sylvatic and Endemic Dengue Viruses. 2008;79:128–132. [PubMed] [Google Scholar]
- 23.van Panhuis WG, et al. Inferring the serotype associated with dengue virus infections on the basis of pre-and postinfection neutralizing antibody titers. J. Infect. Dis. 2010;202:1002–10. doi: 10.1086/656141. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Thomas SJ, et al. Dengue Plaque Reduction Neutralization Test (PRNT) in Primary and Secondary Dengue Virus Infections: How Alterations in Assay Conditions Impact Performance. Am. J. Trop. Med. Hyg. 2009;81:825–833. doi: 10.4269/ajtmh.2009.08-0625. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Smith DJ, et al. Mapping the antigenic and genetic evolution of influenza virus. Science. 2004;305:371–6. doi: 10.1126/science.1097211. [DOI] [PubMed] [Google Scholar]
- 26.Durbin AP, et al. Attenuation and immunogenicity in humans of a live dengue virus type-4 vaccine candidate with a 30 nucleotide deletion in its 3’-untranslated region. Am. J. Trop. Med. Hyg. 2001;65:405–13. doi: 10.4269/ajtmh.2001.65.405. [DOI] [PubMed] [Google Scholar]
- 27.Durbin AP, et al. Emergence potential of sylvatic dengue virus type 4 in the urban transmission cycle is restrained by vaccination and homotypic immunity. Virology. 2013;439:34–41. doi: 10.1016/j.virol.2013.01.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Messer WB, et al. Development and characterization of a reverse genetic system for studying dengue virus serotype 3 strain variation and neutralization. PLoS Negl. Trop. Dis. 2012;6:e1486. doi: 10.1371/journal.pntd.0001486. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Gibbons RV, et al. Analysis of repeat hospital admissions for dengue to estimate the frequency of third or fourth dengue infections resulting in admissions and dengue hemorrhagic fever, and serotype sequences. Am. J. Trop. Med. Hyg. 2007;77:910–913. [PubMed] [Google Scholar]
- 30.Halstead SB, Papaevangelou G. Transmission of dengue 1 and 2 viruses in Greece in 1928. Am. J. Trop. Med. Hyg. 1980;29:635–637. doi: 10.4269/ajtmh.1980.29.635. [DOI] [PubMed] [Google Scholar]
- 31.Hickey AC, et al. Serotype-specific host responses in rhesus macaques after primary dengue challenge. Am. J. Trop. Med. Hyg. 2013;89:1043–57. doi: 10.4269/ajtmh.13-0145. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Kochel TJ, et al. Cross-serotype neutralization of dengue virus in Aotus nancymae monkeys. J. Infect. Dis. 2005;191:1000–4. doi: 10.1086/427511. [DOI] [PubMed] [Google Scholar]
- 33.Dejnirattisai W, et al. A new class of highly potent, broadly neutralizing antibodies isolated from viremic patients infected with dengue virus. Nat. Immunol. 2015;16:785. doi: 10.1038/ni0715-785a. [DOI] [PubMed] [Google Scholar]
- 34.Buddhari D, et al. Dengue virus neutralizing antibody levels associated with protection from infection in thai cluster studies. PLoS Negl. Trop. Dis. 2014;8:e3230. doi: 10.1371/journal.pntd.0003230. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Sariol CA, White LJ. Utility, limitations, and future of non-human primates for dengue research and vaccine development. Front. Immunol. 2014;5:452. doi: 10.3389/fimmu.2014.00452. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Kliks SC, Nimmanitya S, Nisalak a., Burke DS. Evidence that maternal dengue antibodies are important in the development of dengue hemorrhagic fever in infants. Am. J. Trop. Med. Hyg. 1988;38:411–419. doi: 10.4269/ajtmh.1988.38.411. [DOI] [PubMed] [Google Scholar]
- 37.Libraty DH, et al. A prospective nested case-control study of Dengue in infants: rethinking and refining the antibody-dependent enhancement dengue hemorrhagic fever model. PLoS Med. 2009;6:e1000171. doi: 10.1371/journal.pmed.1000171. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Endy TP, et al. Relationship of preexisting dengue virus (DV) neutralizing antibody levels to viremia and severity of disease in a prospective cohort study of DV infection in Thailand. J. Infect. Dis. 2004;189:990–1000. doi: 10.1086/382280. [DOI] [PubMed] [Google Scholar]
- 39.Sabchareon A, et al. Protective efficacy of the recombinant, live-attenuated, CYD tetravalent dengue vaccine in Thai schoolchildren: a randomised, controlled phase 2b trial. Lancet. 2012;380:1559–67. doi: 10.1016/S0140-6736(12)61428-7. [DOI] [PubMed] [Google Scholar]
- 40.Plotkin SA. Complex correlates of protection after vaccination. Clin. Infect. Dis. 2013;56:1458–1465. doi: 10.1093/cid/cit048. [DOI] [PubMed] [Google Scholar]
- 41.Mukherjee S, et al. Mechanism and Significance of Cell Type-Dependent Neutralization of Flaviviruses. J. Virol. 2014;88:7210–7220. doi: 10.1128/JVI.03690-13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Malavige GN, Ogg GS. T cell responses in dengue viral infections. J. Clin. Virol. 2013;58:605–611. doi: 10.1016/j.jcv.2013.10.023. [DOI] [PubMed] [Google Scholar]
- 43.Rico-Hesse R, et al. Origins of dengue type 2 viruses associated with increased pathogenicity in the Americas. Virology. 1997;230:244–51. doi: 10.1006/viro.1997.8504. [DOI] [PubMed] [Google Scholar]
- 44.Darriba D, Taboada GL, Doallo R, Posada D. jModelTest 2: more models, new heuristics and parallel computing. Nat. Methods. 2012;9:772–772. doi: 10.1038/nmeth.2109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Guindon S, et al. New algorithms and methods to estimate maximum-likelihood phylogenies: Assessing the performance of PhyML 3.0. Syst. Biol. 2010;59:307–321. doi: 10.1093/sysbio/syq010. [DOI] [PubMed] [Google Scholar]
- 46.Hordijk W, Gascuel O. Improving the efficiency of SPR moves in phylogenetic tree search methods based on maximum likelihood. Bioinformatics. 2005;21:4338–4347. doi: 10.1093/bioinformatics/bti713. [DOI] [PubMed] [Google Scholar]
- 47.Fraczkiewicz R, Braun W. Exact and efficient analytical calculation of the accessible surface areas and their gradients for macromolecules. J. Comput. Chem. 1998;19:319–333. [Google Scholar]
- 48.Innis BL, et al. An enzyme-linked immunosorbent assay to characterize dengue infections where dengue and Japanese encephalitis co-circulate. Am. J. Trop. Med. Hyg. 1989;40:418–27. doi: 10.4269/ajtmh.1989.40.418. [DOI] [PubMed] [Google Scholar]
- 49.Anderson KB, et al. A shorter time interval between first and second dengue infections is associated with protection from clinical illness in a school-based cohort in Thailand. J. Infect. Dis. 2014;209:360–8. doi: 10.1093/infdis/jit436. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Montoya M, et al. Symptomatic versus inapparent outcome in repeat dengue virus infections is influenced by the time interval between infections and study year. PLoS Negl. Trop. Dis. 2013;7:e2357. doi: 10.1371/journal.pntd.0002357. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Reich NG, et al. Interactions between serotypes of dengue highlight epidemiological impact of cross-immunity. J. R. Soc. Interface. 2013;10:20130414. doi: 10.1098/rsif.2013.0414. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Bhoomiboonchoo P, et al. Sequential dengue virus infections detected in active and passive surveillance programs in Thailand, –. BMC Public Health. 2015;15:1–10. doi: 10.1186/s12889-015-1590-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Blaney JE, Matro JM, Murphy BR, Whitehead SS. Recombinant , Live-Attenuated Tetravalent Dengue Virus Vaccine Formulations Induce a Balanced, Broad, and Protective Neutralizing Antibody Response against Each of the Four Serotypes in Rhesus Monkeys. 2005;79:5516–5528. doi: 10.1128/JVI.79.9.5516-5528.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.WHO . Guidelines on the quality, safety and efficacy of dengue tetravalent vaccines (live, attenuated) Geneva, Switzerland: 2011. [Google Scholar]
- 55.Kuan G, et al. The nicaraguan pediatric dengue cohort study: Study design, methods, use of information technology, and extension to other infectious diseases. Am. J. Epidemiol. 2009;170:120–129. doi: 10.1093/aje/kwp092. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Igarashi A. Isolation of a Singh’s Aedes albopictus cell clone sensitive to dengue and Chikungunya viruses. J. Gen. Virol. 1978;40:531–544. doi: 10.1099/0022-1317-40-3-531. [DOI] [PubMed] [Google Scholar]
- 57.Tesh RB. A method for the isolation and identification of dengue viruses, using mosquito cell cultures. Am. J. Trop. Med. Hyg. 1979;28:1053–1059. doi: 10.4269/ajtmh.1979.28.1053. [DOI] [PubMed] [Google Scholar]
- 58.Vasilakis N, et al. Mosquitoes put the brake on arbovirus evolution: Experimental evolution reveals slower mutation accumulation in mosquito than vertebrate cells. PLoS Pathog. 2009;5 doi: 10.1371/journal.ppat.1000467. doi:10.1371/journal.ppat.1000467. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Henchal EA, Gentry MK, McCown JM, Brandt WE. Dengue virus-specific and flavivirus group determinants identified with monoclonal antibodies by indirect immunofluorescence. Am. J. Trop. Med. Hyg. 1982;31:830–6. doi: 10.4269/ajtmh.1982.31.830. [DOI] [PubMed] [Google Scholar]
- 60.Sukupolvi-Petty S, et al. Structure and function analysis of therapeutic monoclonal antibodies against dengue virus type 2. J. Virol. 2010;84:9227–39. doi: 10.1128/JVI.01087-10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Wahala WMPB, et al. Natural strain variation and antibody neutralization of dengue serotype 3 viruses. PLoS Pathog. 2010;6:e1000821. doi: 10.1371/journal.ppat.1000821. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Gromowski GD, Barrett ND, Barrett ADT. Characterization of dengue virus complex-specific neutralizing epitopes on envelope protein domain III of dengue 2 virus. J. Virol. 2008;82:8828–37. doi: 10.1128/JVI.00606-08. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.de Alwis R, et al. Identification of human neutralizing antibodies that bind to complex epitopes on dengue virions. Proc. Natl. Acad. Sci. 2012;109:7439–7444. doi: 10.1073/pnas.1200566109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Koraka P, Benton S, van Amerongen G, Stittelaar KJ, Osterhaus ADME. Characterization of humoral and cellular immune responses in cynomolgus macaques upon primary and subsequent heterologous infections with dengue viruses. Microbes Infect. 2007;9:940–6. doi: 10.1016/j.micinf.2007.03.012. [DOI] [PubMed] [Google Scholar]
- 65.Toh YX, et al. Dengue serotype cross-reactive, anti-E protein antibodies confound specific immune memory for one year after infection. Immunol. Mem. 2014;5:1–12. doi: 10.3389/fimmu.2014.00388. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Fonville JM, et al. Antibody landscapes after influenza virus infection or vaccination. Science. 2014;346:996–1000. doi: 10.1126/science.1256427. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.