Abstract
The Banting Medal for Scientific Achievement is the highest scientific award of the American Diabetes Association (ADA). Given in memory of Sir Frederick Banting, one of the key investigators in the discovery of insulin, the Banting Medal is awarded annually for scientific excellence, recognizing significant long-term contributions to the understanding, treatment, or prevention of diabetes. Philipp E. Scherer, PhD, of the Touchstone Diabetes Center, The University of Texas Southwestern Medical Center, Dallas, TX, received the prestigious award at the ADA's 75th Scientific Sessions, 5–9 June 2015, in Boston, MA. He presented the Banting Lecture, “The Multifaceted Roles of Adipose Tissue—Therapeutic Targets for Diabetes and Beyond,” on Sunday, 7 June 2015.
A number of different cell types contribute to the cellular architecture of adipose tissue. Although the adipocyte is functionally making important contributions to systemic metabolic homeostatis, several additional cell types contribute a supportive role to bestow maximal flexibility on the tissue with respect to many biosynthetic and catabolic processes, depending on the metabolic state. These cells include vascular endothelial cells, a host of immune cells, and adipocyte precursor cells and fibroblasts. Combined, these cell types give rise to a tissue with remarkable flexibility with respect to expansion and contraction, while optimizing the ability of the tissue to act as an endocrine organ through the release of many protein factors, critically influencing systemic lipid homeostasis and biochemically contributing many metabolites. Using an example from each of these categories—adiponectin as a key adipokine, sphingolipids as critical mediators of insulin sensitivity, and uridine as an important metabolite contributed by the adipocyte to the systemic pool—I will discuss the emerging genesis of the adipocyte over the past 20 years from metabolic bystander to key driver of metabolic flexibility.
Fat cells—friends or foes? No other tissue has been more maligned than adipose tissue. We appreciate that an excess of adipose tissue is frequently associated with an increased risk to develop cardiovascular disease, diabetes, and, in fact, even cancer. This is despite the fact that our view of the adipocyte has changed significantly since the early 1990s. At that time, we valued the fat cell mostly for its ability to undergo insulin-mediated glucose uptake and to release and reesterify free fatty acids, depending on nutritional status (1). Now, we appreciate that the adipocyte is a very complex, metabolically highly active, and potent secretory cell (2). It is capable of releasing a large number of protein products, it is well equipped to manipulate local and systemic lipid levels, and it releases a number of key metabolites. We are going to touch on each of these emerging themes in this article.
Obviously, our fat stores are a reflection of the net difference between energy intake and energy expenditure. Energy intake reflects mostly the uptake of calorically dense foods, while the energy expenditure side reflects the energy required to meet basic energy demands, energy required for physical activity, and energy expended on adaptive thermogenesis (3,4). We know that the systemic ability to adjust to changing nutrient conditions—a phenomenon that we refer to as metabolic flexibility (5)—very much depends on the ability of the adipocyte and adipose tissue to expand in order to accommodate a potential excess of calories going into the system at any given time.
Adipose tissue expansion can occur under a couple of different scenarios, and depending on how effective this expansion process is, it leaves us with either metabolically fit, happy fat cells or dysfunctional, unhappy fat cells that display a large degree of metabolic dysregulation.
Metabolic flexibility is not exclusively a function of absolute fat mass. Plotting fat mass versus the degree of insulin resistance does not reveal a strictly linear relationship. Many of us fall into the category of metabolically unhealthy obese individuals. That means, the more adipose tissue we have, the more metabolically challenged we are. However, some individuals fall into the category of metabolically healthy obese: despite having relatively high BMIs, they display normal metabolic parameters, even though they may not be immune to future pathophysiological changes as a consequence of the increased fat mass (6). The other extreme is lipodystrophy, reflecting the metabolically unhealthy lean individual, displaying severe metabolic dysfunction despite the fact that these individuals have very little or, in some instances, almost no adipose tissue to cope with (7). Why can some of us maintain normal insulin sensitivity despite being overweight or even obese? And, what can go wrong in our adipose tissue as we gain weight?
The reasons why an excess of adipose tissue generally causes metabolic stress are certainly complex. However, we can dissect the path to dysfunction into a number of relatively simple steps. An expanding fat pad, just like any other rapidly expanding tissue mass, displays impaired angiogenesis that can lead to local hypoxia, in other words, an insufficiency in the local oxygen supply (8). This then results in fibrosis and the activation of a number of other downstream events, including the unfolded protein response in the secretory pathway. All of these phenomena ultimately trigger increased cellular stress, increased necrosis, and some degree of chronic, unresolved inflammation (9). We can visualize some of these steps in adipose tissue. Increased fibrosis, as a result of enhanced production of extracellular matrix components, can be visualized by trichrome staining as well as by a number of other methods. This fibrotic response is widely observed in obese adipose tissue from both humans and rodents. We know from both genetic and pharmacological studies that the modulation of the extracellular matrix environment plays a vital role in the health of adipose tissue, both at steady state and during expansion (10). Elegant studies by the Clément group in Paris, France, and the Smith group in Orlando, FL, have underlined the clinical implications of many of the pathological changes associated with fibrous adipose tissue (11,12).
In this context, we have studied the relevance of collagen 6 expression, one of the most abundant extracellular matrix constituents of adipose tissue (13). We can observe in a growing fat pad that there is a significant reduction in the degree of extracellular matrix accumulation in the context of mice lacking collagen 6. As a result, these mice retain normal metabolic function while challenged by a high-fat diet, despite displaying very large individual fat cells, with a reduced incidence of adipocyte cell death. This is apparent when we subject the mice to an oral glucose gavage and observe an improved oral glucose tolerance. Even though these mice have mechanically less stable fat tissue in the absence of this key extracellular building block (14), they are metabolically healthier because they can bypass the usual constraints imposed by the fibrous extracellular milieu that the fat pads experience during expansion.
Extending the proangiogenic component in adipose tissue during expansion can also significantly stave off the fibrotic response, reduce ensuing cell death, and translate into a reduced level of local insulin resistance as a result of reduced inflammation (15). The accumulation of extracellular matrix is an early event and offers an opportunity for pharmacological interventions with antifibrotic agents. The increased rate of cell death in the context of a disproportionate accumulation of extracellular matrix constituents leads to the attraction of macrophages. These macrophages surround lipid droplets resulting from a remnant dead adipocyte and start to engulf lipids over the course of several weeks. It is this process that is associated with a chronic inflammatory response (8). There is no doubt that chronic, unresolved inflammation leads to chronic insulin resistance, a phenomenon that we frequently see epidemiologically in patients with metabolic syndrome. This has also proven to be relevant in a host of rodent models (16). However, is it invariably a negative process to have some low level of inflammation in adipose tissue? This is an important question as anti-inflammatory interventions have proven to be of limited benefit with respect to clinical improvements in insulin sensitivity.
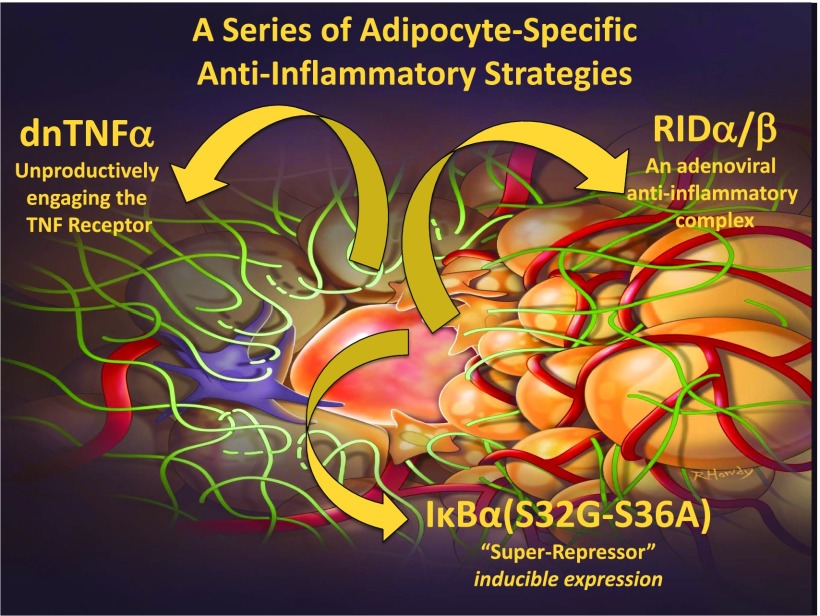
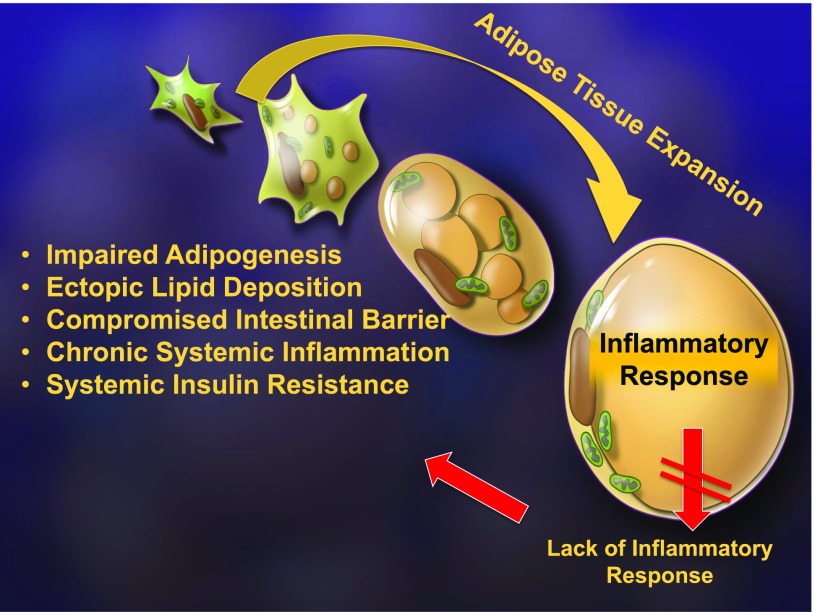
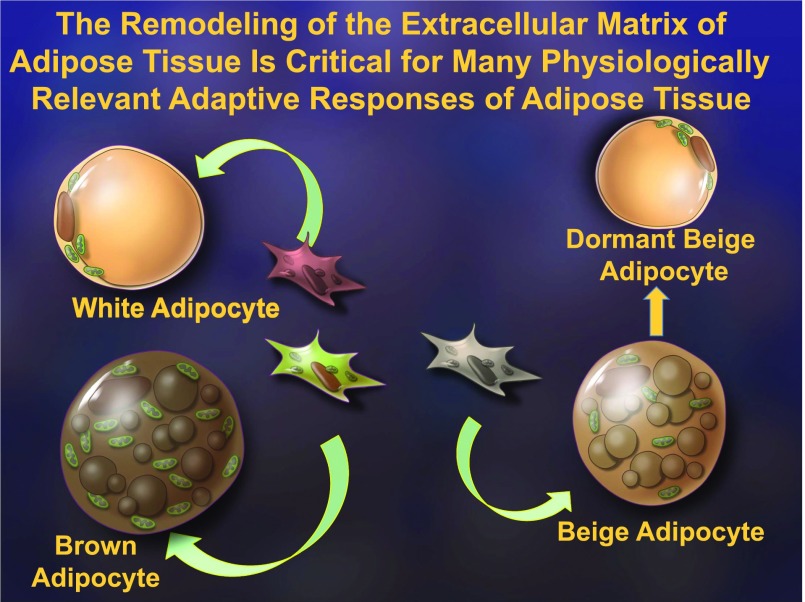
To probe further into the role of the inflammatory response in adipose tissue physiology, we took a series of genetic approaches in which we generated mice that retain a high level of anti-inflammatory activity, exclusively at the level of the adipocyte (17). We have done this in three different ways. We have expressed either a dominant-negative tumor necrosis factor-α (TNFα) ligand that unproductively engages the TNF receptors or we have expressed a potent adenoviral anti-inflammatory molecule called RID. In addition, we have taken advantage of a traditional “super-repressor” of inflammation in the form of a constitutively active inhibitor of NFκB (IκBα) (Fig. 1). The specific details of each approach are not critical, but in all three cases, we have fat cells expressing potent anti-inflammatory factors. We were very surprised to see that in the process of adipose tissue expansion an inflammatory response is initiated as part of the healthy expansion process. By inhibiting this inflammatory response, we noticed that we impair adipogenesis, thereby reducing the number of fat cells available to accommodate any excess lipids, leading to ectopic lipid deposition in the liver. This also led to a compromised intestinal barrier and the release of more endotoxin into the system. Therefore, despite an early anti-inflammatory action at the level of the adipocyte, chronic systemic inflammation and severe systemic insulin resistance ensue (Fig. 2). As a result, we reached the surprising conclusion that a transient, low level of inflammation is beneficial and, in fact, is required for the remodeling of adipose tissue, again highlighting the central role of the extracellular matrix in the process of tissue expansion (17). There are additional physiological settings, beyond mere expansion, in which the importance of extracellular matrix remodeling of adipose tissue is apparent. Fat cells can assume a number of different specialized functions and can, in fact, be recruited to do so by a number of different interventions. This leads to a surprising complexity in terms of the nature of the different adipocytes in our fat pads (Fig. 3). There are classic white adipocytes and brown adipocytes that are specialized in the generation of heat, with high expression of the marker uncoupled protein 1. A third cell type, the “beige adipocyte,” is found in white adipose tissue but has the capacity to develop many of the functional characteristics of brown fat under certain conditions, including cold exposure (18). An active beige adipocyte is a cell that can convert back to a dormant beige adipocyte that resembles a white adipocyte. All of these adipose tissue transitions critically rely on the remodeling of the extracellular matrix.
Figure 1.
The role of inflammation in the adipocyte. Genetic approaches to suppress local inflammatory responses in the adipocyte include a dominant-negative (dn) TNFα ligand that unproductively engages the TNF receptors, a potent adenoviral anti-inflammatory molecule called RID, and a traditional “super-repressor” of inflammation in the form of a mutant IκBα.
Figure 2.
The consequences of a suppressed inflammatory response of the adipocyte. The process of adipose tissue expansion requires an inflammatory response as part of the healthy expansion process. By inhibiting these inflammatory events, adipogenesis is impaired, thereby reducing the number of fat cells available to accommodate any excess lipids, leading to ectopic lipid deposition in the liver. This also leads to a compromised intestinal barrier and the enhanced release of endotoxin into the system. Therefore, despite an early anti-inflammatory action at the level of the adipocyte, chronic systemic inflammation and severe systemic insulin resistance ensue in this setting.
Figure 3.
The different types of adipocytes. Distinct precursor cells give rise to the different types of adipocytes, the classic white and brown adipocytes and the beige adipocytes that may transition to a “dormant” cell resembling a white adipocyte when their presence is not required. All these events require extensive remodeling of the extracellular matrix microenvironment of these cells.
Let us leave the tissue-autonomous effects behind at this point and take a broader view of the adipocyte and its systemic contributions. I want to highlight three specific areas in which the adipocyte plays a major role in systemic energy homeostasis. These areas include the adipocyte as a source of protein factors, the so-called adipokine, that exerts important effects on systemic metabolism. Here, we will focus on adiponectin, an adipokine that our laboratory has studied extensively over the past 20 years (19). Some of these factors play a major role in lipid homeostasis, and as such, the adipocyte also serves as an important buffer for critical lipid mediators. We discuss a specific class of lipids, the sphingolipids. A third area that is just beginning to be unraveled at present is the topic of the adipocyte as an important source of critical metabolites. In the context of this review, we will focus specifically on uridine.
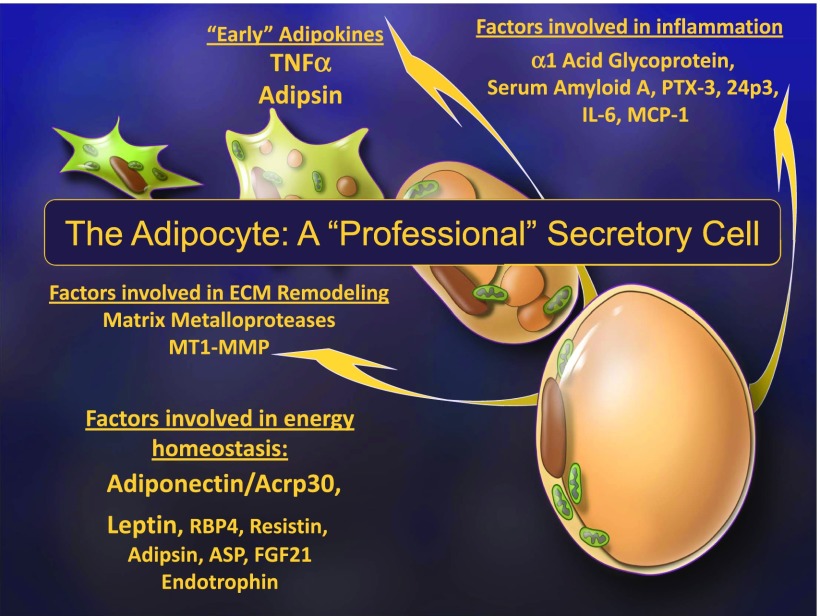
Historically, Spiegelman and colleagues (20,21) started to look at the adipocyte as a source of enzymes and secreted factors. In the late 1980s and early 1990s, they described components of the complement fixation cascade and the inflammatory factor TNFα as adipocyte-derived products. Our own contributions toward establishing the concept that the adipocyte is a highly active “professional” secretory cell expanded the notion that adipocytes release many factors affecting the extracellular matrix, such as matrix metalloproteases, and also increase the release of proinflammatory factors in the obese state (22). More importantly, the adipocyte produces a series of factors highly enriched in adipose tissue that are released into the system and that have profound effects on systemic metabolism. These factors include proteins, such as adiponectin, that we first discovered in the early 1990s (23), around the same time that leptin, another key adipokine, was reported by Jeff Friedman’s laboratory (24). Additional molecules, such as resistin (25,26), fibroblast growth factor 21 (FGF21) (27), and endotrophin (28), were added to that list of adipocyte-enriched secretory products at later points by us and others (Fig. 4).
Figure 4.
The adipocyte as a professional secretory cell. Many distinct categories of secretory proteins have been reported to be released from adipocytes. Acrp30, adipocyte complement-related protein of 30 kDa; ASP, acylation-stimulating protein; ECM, extracellular matrix; IL-6, interleukin 6; MT1-MMP, membrane type 1 matrix metalloproteinase; PTX-3, pentraxin 3; RBP4, retinol-binding protein 4.
For the past 20 years, work in my laboratory has focused on elucidating the physiological function and the systemic impact of adiponectin, the most adipocyte-specific of all secretory factors described to date. The original data obtained for this gene indicated that both the mRNA and protein for adiponectin are highly enriched in fat tissue and dramatically induced in tissue culture over the course of adipogenic differentiation. Since its original description, adiponectin has been a wonderful source of inspiration to learn much about not only the physiological impact of the protein itself but also the adipose tissue physiology in general. Despite its exclusive expression at the level of the fat cell, adiponectin concentrations in plasma are actually inversely proportional to fat mass, an observation that, at least at first sight, is somewhat counterintuitive and in contrast to essentially every single other adipocyte-derived secretory protein (29,30). We have also learned through comparing metabolically healthy and metabolically unhealthy obese individuals that metabolically healthy individuals display higher levels of adiponectin despite identical BMIs (31,32). This suggests that adiponectin levels more closely correlate with insulin sensitivity rather than merely reflecting fat mass. As we increase our fat mass under normal conditions, adiponectin levels are decreased. We have genetically manipulated mice, such that they maintain high adiponectin levels similar to lean mice despite gaining weight, reminiscent of what the metabolically healthy obese individual does. We have done this in rodents on several occasions, either through a direct overexpression of adiponectin (33) or through the manipulation of other critical cellular pathways, including interventions at the level of mitochondria (34). We noticed that the mice dramatically expand their fat mass under these conditions to become the rodent equivalent of an 800-lb human subject. However, despite being the heaviest mice ever reported, they retain full metabolic flexibility, as the subcutaneous fat of these mice effectively absorbs all excess calories, thereby sparing all other tissues from any adverse lipotoxic effects. In fact, the adipose tissue of these mice is a paradigm for healthy adipose tissue expansion. Histologically comparing these metabolically healthy mice to their leaner unhealthy counterparts, we notice that metabolically healthy mice have many more, but smaller, fat cells, with widespread adipocyte hyperplasia. They do not display the conventional elevated levels of inflammatory markers as judged by the reduced number of immune cells infiltrating the tissue. They are better vascularized and show a reduced degree of fibrosis. Healthy fat pads translate into insulin-sensitive livers that have less steatosis, and β-cell function is also spared to maintain fully functional islets (34).
Positive changes in adiponectin levels are also a reflection of therapeutic improvements in systemic insulin sensitivity. For example, upon exposure to a peroxisome proliferator–activated receptor γ (PPARγ) agonist, we reported that the relative increase in adiponectin is proportional to the effectiveness of the drug to improve systemic insulin sensitivity (35). In fact, in the absence of adiponectin, PPARγ agonists reduce their potency to induce improvements in insulin sensitivity (36). This type of correlation has been seen in dozens of different studies with distinct classes of antidiabetes drugs. We conclude therefore that adiponectin levels are excellent integrators of adipose tissue health and systemic metabolic flexibility (37). Most importantly, changes in adiponectin levels, induced by either weight loss or pharmacological intervention, reflect improvements in systemic insulin sensitivity. As a result, many laboratories have studied the effects of adiponectin on a large number of different target cells and tissues. There is not a tissue in the system that is not directly—or minimally indirectly—affected by the actions of adiponectin (19). In many instances, improvements at the level of the tissue are not merely correlational to increases in adiponectin levels, but rather they are a direct consequence of it (38).
In order to analyze the function of adiponectin in different target tissues, we have constructed a series of models that allow us to challenge specific cell types in target tissues with a proapoptotic stimulus. We refer to these mice as the ATTAC mice, for Apoptosis Through Triggered Activation of Caspase-8 (39). These mice have a transgenic cassette encoding a suicide gene that we can specifically activate in the respective target cell. In this way, we can probe for the ability of the specific cell type to resist cell death as a function of adiponectin concentration. Once we have ablated the target cell population and stopped the apoptotic stimulus, we can also use the same system to test the capacity of the tissue to regenerate as a function of adiponectin concentrations in plasma. We have done this for a number of different tissues, including the heart (40), the pancreatic β-cell (41), the podocyte (a kidney cell critically affected by diabetes) (42), and for many other tissues. In the cell ablation model for the cardiomyocyte, the so-called “Heart-ATTAC mouse,” we activated the suicide gene and monitored the number of animals that survive as a function of time after the initiation of the treatment, and we reported that the higher the adiponectin levels, the higher the survival rate (40). In order to focus on the pancreatic β-cell, we use the pancreatic islet cell ATTAC mice, a system that we refer to as “PANIC-ATTAC mice.” After ablation of the β-cells, we found islets that almost exclusively contain α-cells, as judged by immunohistochemistry with anti-glucagon antibodies, and contain very few remaining insulin-positive cells. In the absence of adiponectin, these β-cells are a lot more susceptible to the proapoptotic stimulus induced by the PANIC-ATTAC transgene. In fact, we can monitor the systemic blood glucose levels and see that the glucose levels are higher, reflecting reduced β-cell viability and an increased loss of β-cell mass in the absence of adiponectin (40). If we look at the regenerating β-cell mass once we uniformly ablate insulin-positive cells in all genotypes to the same extent, we notice that the presence of increased levels of adiponectin in the system significantly improves the ability of the pancreas to regenerate β-cell mass and reach near-euglycemic conditions, an observation with profound implications for both type 1 and type 2 diabetes (43). Similarly, in the kidney, we can induce widespread elimination of the podocyte in the “POD-ATTAC mouse” and notice that we improve the functional recovery of the kidney dramatically with elevated adiponectin in the system, reflecting the profound impact of adiponectin on improvements in kidney function in the context of chronic kidney disease and diabetic nephropathy (42).
Adiponectin therefore represents a protein that has potent antiapoptotic functions in a number of different cell types. In addition, it can be proangiogenic (44), making sure that expanding fat pads get appropriately vascularized. It can be antiatherogenic with important implications for cardiovascular disease (16). It is a potent anti-inflammatory factor (16). It can stimulate the generation of new small fat cells, so it is proadipogenic (33). It can cause potent improvements at the level of hepatic insulin sensitivity (45), and it exerts positive effects on wound healing. Adiponectin has given us many additional insights over the years. Adiponectin is one of the best biomarkers for existing metabolic disease—low levels of adiponectin reflect high degrees of insulin resistance (46,47). It is one of the best prospective biomarkers for future incidence of cardiovascular disease and diabetes (48). Its genetic dissection has provided us with a lot of valuable research tools. Mutations in the adiponectin gene in humans that lead to reduced adiponectin levels are closely tied to a metabolic syndrome phenotype (46). At the preclinical level, the adiponectin promoter cassette has allowed us to highly specifically manipulate the mature adipocyte and to generate a number of genetic tools that let us monitor the generation of new fat cells under many different conditions (49,50). And importantly, the analysis of the physiological impact of adiponectin has highlighted a number of new therapeutic areas. There is widespread interest in the adiponectin receptor as a drug target (51). A lot of effort currently goes into the identification of adiponectin receptor agonists that bear promise as therapeutic insulin sensitizers.
These efforts should be further facilitated by the availability of high-resolution structures for the adiponectin receptors that were recently published by Kadowaki and colleagues (52). Both the Kadowaki group in Tokyo, Japan (53), and the Matsuzawa group in Osaka, Japan (54), have contributed significantly to our understanding of adiponectin physiology over many years. With the concerted efforts among our three groups and many others in academia and industry, I am confident that there is ample room for the development of a clinically relevant class of adiponectin receptor agonists for the treatment of metabolic disease.
In addition to all of these aspects, adiponectin has also taught us much about the ability of the fat cell to manipulate lipid factors. The connection between adiponectin and lipid metabolism is particularly apparent when we look at the broad category of lipids referred to as ceramides (55). The ceramide subspecies of sphingolipids have been linked to inflammation, cell death, and insulin resistance. Ceramides can be degraded to generate sphingosine phosphates that have the exact opposite function of ceramides: they signal survival and proliferation. Degrading ceramides is therefore a very important step from a metabolic perspective. The ratio of ceramides versus sphingosines—we refer to it as the Ceramide-S1P Rheostat—is a critical determinant of the metabolic health of any given cell type. We were able to show that adiponectin has potent effects on this lipid class. Adiponectin has the ability to decrease ceramide levels in the liver and in many other tissues, thereby improving hepatic insulin sensitivity (40). Low adiponectin reduces the level of sphingosine-1-phosphate within cardiomyocytes and increases the levels of ceramides, thereby reducing the survival of these cardiomyocytes either in response to hypoxia or to a lipotoxic insult (40).
More importantly, we now know that ceramide levels are frequently elevated in the context of type 2 diabetes in both plasma and tissues (56). Adiponectin potently lowers these ceramide levels. Several antidiabetes drug treatment regimens that lead to an increase of adiponectin ultimately cause a decrease in circulating ceramide levels. This includes, but is not limited to, the widely studied factor FGF21, which is one of the more recent antidiabetes targets under study. FGF21, through both central action in the brain and direct action on the adipocyte, potently induces adiponectin. It loses a lot of its insulin-sensitizing effects in the absence of adiponectin (57). Similarly, PPARγ agonists, such as the thiazolidinediones, lead to an effective reduction of ceramide levels by mediating an induction of FGF21, which in turn elevates adiponectin (57,58). The signaling events induced by adiponectin affecting the ceramide pathway at the cellular level have been worked out in detail. Despite a lot of data implicating ceramides as mediators of insulin resistance, there is still a sound scientific debate ongoing as to whether ceramides are truly directly responsible for reducing insulin sensitivity or whether they are simply an indirect by-product of insulin resistance. Other lipids, such as diacylglycerols, have also been implicated as insulin desensitizers by elegant work in the Shulman laboratory at Yale (59). In order to address the involvement of the ceramides more directly, we used an enzyme that targets ceramides for degradation, a so-called acid ceramidase (60). In parallel, we performed the same type of analysis with the adiponectin receptors that we have previously demonstrated to be associated with a potent ceramidase activity, leading to a cascade of downstream signaling events within a cell. We have taken advantage of powerful genetic mouse models that allow us to inducibly overexpress acid ceramidase in the adult mouse liver in the context of high-fat diet exposure and test the hypothesis that an acute lowering of ceramides through the action of acid ceramidase is associated with an improvement in insulin sensitivity (60). In fact, we can see that upon lowering ceramide species in the liver, we can detect potent improvements in insulin sensitivity, as judged by the glucose infusion rates and the hepatic glucose production rates during euglycemic clamp studies. Surprisingly, the hepatic improvements due to hepatic overexpression of acid ceramidase are also associated with a rapid improvement in insulin sensitivity at the level of the adipocyte, as judged by several different criteria, including an enhanced insulin-mediated glucose uptake. Vice versa, we can generate a model in which we induce acid ceramidase in the adipocyte. Under these conditions, we can see an acute improvement in glucose clearance in an oral glucose tolerance test in the context of significantly lower insulin levels, reflecting the improvement in systemic insulin sensitivity. Putting acid ceramidase into the adipocyte, we also noted that the hepatic lipid content is reduced, as judged by the minimal steatosis seen in the transgenic livers, with a concomitant improvement in insulin sensitivity. Therefore, either hepatic- or adipocyte-specific reduction of ceramides leads to rapid improvements in insulin sensitivity in the entire system, highlighting the potent communication axis between the adipocyte and the hepatocyte at the level of sphingolipids (60). Similar results across both tissues can also be observed upon overexpression of the adiponectin receptors. These observations invoke adiponectin as an effective antilipotoxic agent on the basis of its ability to reduce ceramides, thereby reducing inflammation, improving cell survival, and enhancing insulin sensitivity.
The effects of adiponectin on lipids go beyond ceramides. Adiponectin is one of only a handful of factors that effectively induces “beiging” of white adipose tissue, thereby enhancing β-oxidative capacity of the tissue (37). Other prominent factors that have the ability to do this in white adipose tissue are vascular endothelial growth factors (15,61); bone morphogenic proteins (62); a set of muscle-derived molecules, such as meteorin-like (63) and irisin (64); and the growth factor receptor Her3/ErbB3, a target that we are actively working on currently.
Collectively, because of all these effects on lipid and carbohydrate metabolism, adiponectin is clearly a driving force for maintaining metabolic flexibility. Whether under conditions of feeding or fasting, carbohydrate excess or increased levels of free fatty acids, adiponectin acts in a pivotal manner to help the cell, and in fact the entire system, to adjust appropriately to varied conditions and maintain normal glucose and fatty acid levels (65).
Having covered adipokines and sphingolipids, I want to now highlight a last important aspect of the adipocyte—the secretion of a series of key metabolites that unexpectedly originate in the adipocyte. Specifically, we want to discuss a series of unpublished observations focusing on a metabolite that plays a critical role in the feed/fasting adaptation that we mentioned in the context of metabolic flexibility. This key metabolite is uridine. Uridine is an important building block for a large number of biochemical reactions that include RNA and DNA biosynthesis and the hexosamine pathway. It can even serve under some conditions as a fuel source, particularly for neurons (66). Uridine biosynthesis takes place predominantly in the liver, at least in the fed state (67). However, we noticed that in the fasted state, when hepatic uridine biosynthesis is shut down, the adipocyte becomes the major systemic source of uridine. What is the driver of this process, and what are the physiological consequences of this observation? The upstream regulator turns out to be the transcription factor Xbp1s. Traditionally thought of as one of the key mediators of the cellular unfolded protein response in the secretory pathway, it turns out that Xbp1s does many more things than just initiate the unfolded protein response. We, along with a number of colleagues at The University of Texas Southwestern Medical Center, have shown that Xbp1s is a factor critically involved in the adaptation phase between feeding and fasting in a range of different tissues, including in the liver (68), the heart (69), key neurons in the hypothalamus (70), and adipocyte. Just like with ceramides, Xbp1s activity is highly coordinated between the adipocyte and the liver. After ingestion of a meal, we have a transient rise of Xbp1s in the liver, while we have very low levels of Xbp1s at the level of the adipocyte. In contrast, in the fasted state, we see very low levels of Xbp1s in the liver, whereas we find very high levels of Xbp1s in the adipocytes. The functional implications of this phenomenon are quite profound. In the fed state, Xbp1s tilts the balance between gluconeogenesis and uridine biosynthesis toward uridine biosynthesis, such that the liver produces high levels of uridine and releases uridine into circulation. In the fasting liver, however, low Xbp1s leads to low uridine biosynthesis, enabling the liver to focus on gluconeogenesis. In the fasting adipocyte, there are high levels of Xbp1s, and we found that Xbp1s is a potent inducer of lipolysis. The liberated free fatty acids are not only exported to the liver as a source of energy but also can be oxidized locally in the adipocyte. A key downstream target of Xbp1s is an enzyme called CAD (carbamoyl-phosphate synthetase 2, aspartate transcarbamylase, and dihydroorotase) (67). CAD is the rate-limiting enzyme for uridine biosynthesis. Xbp1s, when active in the adipocyte, induces CAD, and CAD uses the energy generated by lipolysis and enhanced β-oxidation to synthesize high levels of uridine in the adipocyte. We observed that plasma uridine concentrations are increased in the fasted state. We see the same thing in patients during fasting and after meal ingestion. Individuals who have fasted overnight display high uridine levels; after consuming breakfast, the levels of uridine in the plasma uniformly drop and a meal-induced lowering of uridine in circulation can be observed. At the same time, meal ingestion in these individuals prompts a reduction of the uridine content of the adipocyte, consistent with the suggestion that it is adipose tissue that is responsible for the high levels of uridine in circulation in the fasted state. We measured uridine under a number of different conditions and noticed that the mice that are very obese and metabolically dysfunctional and lack leptin action, such as the db/db mouse or the ob/ob mouse, have very high plasma uridine levels. Under the same conditions, we know that the core body temperature of these mice is significantly lower in the fed state and particularly in the fasted state. Food removal prompts a drop in core body temperature by several degrees. It turns out that when uridine is injected, there is a transient lowering of the core body temperature. Putting these observations together, we suggest that it is the fasting-induced increase in plasma uridine that mediates the well-established fasting-induced reduction in core body temperature. In fact, if we prevent uridine biosynthesis by injecting N-(phosphonacetyl)-L-aspartate acid (PALA) (71), an inhibitor of CAD, into fasted animals, we notice that these fasted animals no longer reduce their core body temperature to the same extent. We therefore suggest that the adipocyte is a very effective manipulator of core body temperature on the basis of manipulating its ability to synthesize and release uridine into the system. Lowering core body temperature is associated with a reduction in the metabolic rate, which serves to preserve energy in the fasted state.
In the absence of adipose tissue, we observe low plasma uridine levels. As an example, in the lipodystrophic AGPAT2 knockout mouse (72), we see that uridine levels are very low in the fasting state and do not change very much in the feeding/fasting transition. Uridine serves as a master regulator of the systemic fasting response and has many additional effects. For instance, when we perfuse a pancreas with uridine, we observe a significantly increased release of the archetypal fasting hormone glucagon. So it is the adipocyte that is the major source of uridine during fasting, and as such, the adipocyte is the mastermind coordinating the systemic response to the fasting state. Interestingly, adiponectin and leptin are critically affected by feeding and fasting. Xbp1s potently suppresses both of these adipokines. We analyzed rodents in which we overruled feedback mechanisms and permanently activated Xbp1s expression at the level of the adipocyte. We noticed that this leads to an elevation of plasma uridine levels. Over time, this leads to a reduction of individual fat pad sizes and a reduction in overall adipose tissue mass. This is due to the fact that these mice expend a lot of energy in the adipocyte and convert that energy biosynthetically into uridine production and subsequent release. It is important to note that this phenomenon is completely independent of browning or beiging of adipose tissue!
Obviously, this phenomenon has profound implications in terms of how we think about potentially targeting weight-loss regimens pharmacologically at the level of the adipocyte. We have manipulated the adipocyte in a number of different ways. For instance, we have genetically reduced the level of β-oxidation in the adipocyte, but we did not achieve a reduction in weight. In fact, we created the fattest mice ever reported under these conditions (34). If we try the opposite and increase the β-oxidative capacity of white adipocytes, the energy generated by burning lipids in the mitochondria has nowhere to go, thereby leading to an increase in reactive oxygen species as a spin-off of the excessive production of oxidative capacity (34). A third possibility that many embrace at this point is the generation of heat by uncoupling mitochondria within white adipose tissue, technically turning the white adipocytes into beige adipocytes, enabling these cells to burn more energy and to release this excess energy as heat. That is a very powerful and a very effective way of dissipating excess energy, but we have to be very careful to have feedback mechanisms in place to avoid overheating (73,74).
Leveraging Xbp1s action in the adipocyte provides a very interesting and promising fourth option. In this case, the energy created by increased β-oxidation can be diverted into a biosynthetic pathway for a compound that can be used by other components of the system. Specifically, Xbp1s prompts an increase in the lipolytic rate, generating free fatty acids. These free fatty acids can be exported, but Xbp1s can also stimulate their oxidation locally. The energy generated by this increased β-oxidative activity can be funneled into uridine biosynthesis. If we constitutively elevate this process at all times, then we divert excess energy into a neutral biosynthetic pathway whose end products we can safely remove systemically.
From the perspective of maintaining metabolic homeostasis system-wide, it is clear that we need to keep our adipocytes fully functional and that full adipocyte functionality transpires into systemic metabolic flexibility that ensures overall metabolic health. At present, we have only scratched the surface of this fascinating cell type and that the cross talk of adipocytes with other tissues through the actions of adipokines, critical lipid factors, and other metabolites will teach us much more in the future.
When we think about the adipocyte, we have to think even more globally, beyond metabolic function. We suspect that many more disease areas are profoundly affected by these cells. We already appreciate that the adipocyte plays essential roles in the area of infectious diseases. As an example, the adipocyte is the target for several parasites, such as Trypanosoma cruzi, the causative agent of Chagas disease (75). We also value the critical role that adipose tissue plays in cancer through interactions between adipocytes and invading tumor cells. The infrastructure of the adipocyte can be taken over by an infiltrating tumor mass, particularly in the mammary gland, where tumor cells take advantage of local growth factors, cytokines, extracellular matrix modulators, and adipocyte-derived metabolites (28,76,77). This explains the epidemiological observation that increased BMI is associated with an increased rate of cancer incidence, particularly for postmenopausal breast cancer, endometrial cancer, and a few other cancer types. We are still hard-pressed to mechanistically explain the correlations between increased fat mass, increased tumor incidence, and worse prognosis, but the adipocyte will likely have a key role in this process. I believe we can expect many more surprising discoveries from the adipocyte in the future.
In conclusion, I am very grateful and proud of my highly talented fellow faculty members in the Touchstone Diabetes Center—the instructors, postdocs, and students, past and present, who shaped our approaches and our science and whose work I was privileged to share with you.
Article Information
Funding. P.E.S. has been supported by the National Institute of Diabetes and Digestive Kidney Diseases (grants R01-DK55758, R01-DK099110, and P01-DK088761) and a grant from the Cancer Prevention and Research Institute of Texas (CPRIT RP140412).
Duality of Interest. P.E.S. has served as an advisor or consultant within the past 12 months to Janssen Pharmaceuticals and has received research support from Merck, Eli Lilly, and Takeda Pharmaceuticals. Neither P.E.S. nor his family members hold stock directly or indirectly in any of these companies. No other potential conflicts of interest relevant to this article were reported.
References
- 1.Kasanicki MA, Pilch PF. Regulation of glucose-transporter function. Diabetes Care 1990;13:219–227 [DOI] [PubMed] [Google Scholar]
- 2.Rutkowski JM, Stern JH, Scherer PE. The cell biology of fat expansion. J Cell Biol 2015;208:501–512 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Unger RH, Scherer PE. Gluttony, sloth and the metabolic syndrome: a roadmap to lipotoxicity. Trends Endocrinol Metab 2010;21:345–352 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Lowell BB, Spiegelman BM. Towards a molecular understanding of adaptive thermogenesis. Nature 2000;404:652–660 [DOI] [PubMed] [Google Scholar]
- 5.Storlien L, Oakes ND, Kelley DE. Metabolic flexibility. Proc Nutr Soc 2004;63:363–368 [DOI] [PubMed] [Google Scholar]
- 6.Samocha-Bonet D, Dixit VD, Kahn CR, et al. Metabolically healthy and unhealthy obese--the 2013 Stock Conference report. Obes Rev 2014;15:697–708 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Patni N, Garg A. Congenital generalized lipodystrophies--new insights into metabolic dysfunction. Nat Rev Endocrinol 2015;11:522–534 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Sun K, Kusminski CM, Scherer PE. Adipose tissue remodeling and obesity. J Clin Invest 2011;121:2094–2101 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Rajala MW, Scherer PE. Minireview: The adipocyte--at the crossroads of energy homeostasis, inflammation, and atherosclerosis. Endocrinology 2003;144:3765–3773 [DOI] [PubMed] [Google Scholar]
- 10.Sun K, Tordjman J, Clément K, Scherer PE. Fibrosis and adipose tissue dysfunction. Cell Metab 2013;18:470–477 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Pasarica M, Gowronska-Kozak B, Burk D, et al. Adipose tissue collagen VI in obesity. J Clin Endocrinol Metab 2009;94:5155–5162 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Divoux A, Clément K. Architecture and the extracellular matrix: the still unappreciated components of the adipose tissue. Obes Rev 2011;12:e494–e503 [DOI] [PubMed] [Google Scholar]
- 13.Khan T, Muise ES, Iyengar P, et al. Metabolic dysregulation and adipose tissue fibrosis: role of collagen VI. Mol Cell Biol 2009;29:1575–1591 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Lackey DE, Burk DH, Ali MR, et al. Contributions of adipose tissue architectural and tensile properties toward defining healthy and unhealthy obesity. Am J Physiol Endocrinol Metab 2014;306:E233–E246 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Sun K, Wernstedt Asterholm I, Kusminski CM, et al. Dichotomous effects of VEGF-A on adipose tissue dysfunction. Proc Natl Acad Sci U S A 2012;109:5874–5879 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Berg AH, Scherer PE. Adipose tissue, inflammation, and cardiovascular disease. Circ Res 2005;96:939–949 [DOI] [PubMed] [Google Scholar]
- 17.Wernstedt Asterholm I, Tao C, Morley TS, et al. Adipocyte inflammation is essential for healthy adipose tissue expansion and remodeling. Cell Metab 2014;20:103–118 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Nedergaard J, Cannon B. The changed metabolic world with human brown adipose tissue: therapeutic visions. Cell Metab 2010;11:268–272 [DOI] [PubMed] [Google Scholar]
- 19.Ye R, Scherer PE. Adiponectin, driver or passenger on the road to insulin sensitivity? Mol Metab 2013;2:133–141 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Flier JS, Cook KS, Usher P, Spiegelman BM. Severely impaired adipsin expression in genetic and acquired obesity. Science 1987;237:405–408 [DOI] [PubMed] [Google Scholar]
- 21.Hotamisligil GS, Shargill NS, Spiegelman BM. Adipose expression of tumor necrosis factor-alpha: direct role in obesity-linked insulin resistance. Science 1993;259:87–91 [DOI] [PubMed] [Google Scholar]
- 22.Halberg N, Wernstedt-Asterholm I, Scherer PE. The adipocyte as an endocrine cell. Endocrinol Metab Clin North Am 2008;37:753–768, x–xi [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Scherer PE, Williams S, Fogliano M, Baldini G, Lodish HF. A novel serum protein similar to C1q, produced exclusively in adipocytes. J Biol Chem 1995;270:26746–26749 [DOI] [PubMed] [Google Scholar]
- 24.Zhang Y, Proenca R, Maffei M, Barone M, Leopold L, Friedman JM. Positional cloning of the mouse obese gene and its human homologue. Nature 1994;372:425–432 [DOI] [PubMed] [Google Scholar]
- 25.Steppan CM, Bailey ST, Bhat S, et al. The hormone resistin links obesity to diabetes. Nature 2001;409:307–312 [DOI] [PubMed] [Google Scholar]
- 26.Rajala MW, Lin Y, Ranalletta M, et al. Cell type-specific expression and coregulation of murine resistin and resistin-like molecule-alpha in adipose tissue. Mol Endocrinol 2002;16:1920–1930 [DOI] [PubMed] [Google Scholar]
- 27.Kharitonenkov A, Shiyanova TL, Koester A, et al. FGF-21 as a novel metabolic regulator. J Clin Invest 2005;115:1627–1635 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Park J, Scherer PE. Adipocyte-derived endotrophin promotes malignant tumor progression. J Clin Invest 2012;122:4243–4256 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Arita Y, Kihara S, Ouchi N, et al. Paradoxical decrease of an adipose-specific protein, adiponectin, in obesity. Biochem Biophys Res Commun 1999;257:79–83 [DOI] [PubMed] [Google Scholar]
- 30.Turer AT, Khera A, Ayers CR, et al. Adipose tissue mass and location affect circulating adiponectin levels. Diabetologia 2011;54:2515–2524 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Ahl S, Guenther M, Zhao S, et al. Adiponectin levels differentiate metabolically healthy vs unhealthy among obese and nonobese white individuals. J Clin Endocrinol Metab 2015;100:4172–4180 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Aguilar-Salinas CA, García EG, Robles L, et al. High adiponectin concentrations are associated with the metabolically healthy obese phenotype. J Clin Endocrinol Metab 2008;93:4075–4079 [DOI] [PubMed] [Google Scholar]
- 33.Kim JY, van de Wall E, Laplante M, et al. Obesity-associated improvements in metabolic profile through expansion of adipose tissue. J Clin Invest 2007;117:2621–2637 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Kusminski CM, Holland WL, Sun K, et al. MitoNEET-driven alterations in adipocyte mitochondrial activity reveal a crucial adaptive process that preserves insulin sensitivity in obesity. Nat Med 2012;18:1539–1549 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Pajvani UB, Hawkins M, Combs TP, et al. Complex distribution, not absolute amount of adiponectin, correlates with thiazolidinedione-mediated improvement in insulin sensitivity. J Biol Chem 2004;279:12152–12162 [DOI] [PubMed] [Google Scholar]
- 36.Nawrocki AR, Rajala MW, Tomas E, et al. Mice lacking adiponectin show decreased hepatic insulin sensitivity and reduced responsiveness to peroxisome proliferator-activated receptor gamma agonists. J Biol Chem 2006;281:2654–2660 [DOI] [PubMed] [Google Scholar]
- 37.Asterholm IW, Scherer PE. Enhanced metabolic flexibility associated with elevated adiponectin levels. Am J Pathol 2010;176:1364–1376 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Combs TP, Pajvani UB, Berg AH, et al. A transgenic mouse with a deletion in the collagenous domain of adiponectin displays elevated circulating adiponectin and improved insulin sensitivity. Endocrinology 2004;145:367–383 [DOI] [PubMed] [Google Scholar]
- 39.Pajvani UB, Trujillo ME, Combs TP, et al. Fat apoptosis through targeted activation of caspase 8: a new mouse model of inducible and reversible lipoatrophy. Nat Med 2005;11:797–803 [DOI] [PubMed] [Google Scholar]
- 40.Holland WL, Miller RA, Wang ZV, et al. Receptor-mediated activation of ceramidase activity initiates the pleiotropic actions of adiponectin. Nat Med 2011;17:55–63 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Wang ZV, Mu J, Schraw TD, et al. PANIC-ATTAC: a mouse model for inducible and reversible beta-cell ablation. Diabetes 2008;57:2137–2148 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Rutkowski JM, Wang ZV, Park AS, et al. Adiponectin promotes functional recovery after podocyte ablation. J Am Soc Nephrol 2013;24:268–282 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Ye R, Holland WL, Gordillo R, et al. Adiponectin is essential for lipid homeostasis and survival under insulin deficiency and promotes β-cell regeneration. eLife 2014;3:3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Landskroner-Eiger S, Qian B, Muise ES, et al. Proangiogenic contribution of adiponectin toward mammary tumor growth in vivo. Clin Cancer Res 2009;15:3265–3276 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Berg AH, Combs TP, Du X, Brownlee M, Scherer PE. The adipocyte-secreted protein Acrp30 enhances hepatic insulin action. Nat Med 2001;7:947–953 [DOI] [PubMed] [Google Scholar]
- 46.Kondo H, Shimomura I, Matsukawa Y, et al. Association of adiponectin mutation with type 2 diabetes: a candidate gene for the insulin resistance syndrome. Diabetes 2002;51:2325–2328 [DOI] [PubMed] [Google Scholar]
- 47.Tonelli J, Li W, Kishore P, et al. Mechanisms of early insulin-sensitizing effects of thiazolidinediones in type 2 diabetes. Diabetes 2004;53:1621–1629 [DOI] [PubMed] [Google Scholar]
- 48.Pischon T, Girman CJ, Hotamisligil GS, Rifai N, Hu FB, Rimm EB. Plasma adiponectin levels and risk of myocardial infarction in men. JAMA 2004;291:1730–1737 [DOI] [PubMed] [Google Scholar]
- 49.Wang QA, Scherer PE. The AdipoChaser mouse: a model tracking adipogenesis in vivo. Adipocyte 2014;3:146–150 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Wang ZV, Deng Y, Wang QA, Sun K, Scherer PE. Identification and characterization of a promoter cassette conferring adipocyte-specific gene expression. Endocrinology 2010;151:2933–2939 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Okada-Iwabu M, Yamauchi T, Iwabu M, et al. A small-molecule AdipoR agonist for type 2 diabetes and short life in obesity. Nature 2013;503:493–499 [DOI] [PubMed] [Google Scholar]
- 52.Tanabe H, Fujii Y, Okada-Iwabu M, et al. Crystal structures of the human adiponectin receptors. Nature 2015;520:312–316 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Kadowaki T, Yamauchi T, Okada-Iwabu M, Iwabu M. Adiponectin and its receptors: implications for obesity-associated diseases and longevity. Lancet Diabetes Endocrinol 2014;2:8–9 [DOI] [PubMed] [Google Scholar]
- 54.Matsuzawa Y. Adiponectin: a key player in obesity related disorders. Curr Pharm Des 2010;16:1896–1901 [DOI] [PubMed] [Google Scholar]
- 55.Holland WL, Summers SA. Sphingolipids, insulin resistance, and metabolic disease: new insights from in vivo manipulation of sphingolipid metabolism. Endocr Rev 2008;29:381–402 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Chaurasia B, Summers SA. Ceramides - lipotoxic inducers of metabolic disorders. Trends Endocrinol Metab 2015;26:538–550 [DOI] [PubMed] [Google Scholar]
- 57.Holland WL, Adams AC, Brozinick JT, et al. An FGF21-adiponectin-ceramide axis controls energy expenditure and insulin action in mice. Cell Metab 2013;17:790–797 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Warshauer JT, Lopez X, Gordillo R, et al. Effect of pioglitazone on plasma ceramides in adults with metabolic syndrome. Diabetes Metab Res Rev 2015;31:734–744 [DOI] [PubMed] [Google Scholar]
- 59.Jornayvaz FR, Shulman GI. Diacylglycerol activation of protein kinase Cε and hepatic insulin resistance. Cell Metab 2012;15:574–584 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Xia JY, Holland WL, Kusminski CM, et al. Targeted induction of ceramide degradation leads to improved systemic metabolism and reduced hepatic steatosis. Cell Metab 2015;22:266–278 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Sun K, Kusminski CM, Luby-Phelps K, et al. Brown adipose tissue derived VEGF-A modulates cold tolerance and energy expenditure. Mol Metab 2014;3:474–483 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Xue R, Wan Y, Zhang S, Zhang Q, Ye H, Li Y. Role of bone morphogenetic protein 4 in the differentiation of brown fat-like adipocytes. Am J Physiol Endocrinol Metab 2014;306:E363–E372 [DOI] [PubMed] [Google Scholar]
- 63.Rao RR, Long JZ, White JP, et al. Meteorin-like is a hormone that regulates immune-adipose interactions to increase beige fat thermogenesis. Cell 2014;157:1279–1291 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Boström P, Wu J, Jedrychowski MP, et al. A PGC1-α-dependent myokine that drives brown-fat-like development of white fat and thermogenesis. Nature 2012;481:463–468 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Unger RH, Scherer PE, Holland WL. Dichotomous roles of leptin and adiponectin as enforcers against lipotoxicity during feast and famine. Mol Biol Cell 2013;24:3011–3015 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Choi JW, Yoo BK, Shin CY, et al. Uridine prevents the glucose deprivation-induced death of immunostimulated astrocytes via the action of uridine phosphorylase. Neurosci Res 2006;56:111–118 [DOI] [PubMed] [Google Scholar]
- 67.Jones ME. Pyrimidine nucleotide biosynthesis in animals: genes, enzymes, and regulation of UMP biosynthesis. Annu Rev Biochem 1980;49:253–279 [DOI] [PubMed] [Google Scholar]
- 68.Deng Y, Wang ZV, Tao C, et al. The Xbp1s/GalE axis links ER stress to postprandial hepatic metabolism. J Clin Invest 2013;123:455–468 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Wang ZV, Deng Y, Gao N, et al. Spliced X-box binding protein 1 couples the unfolded protein response to hexosamine biosynthetic pathway. Cell 2014;156:1179–1192 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Williams KW, Liu T, Kong X, et al. Xbp1s in Pomc neurons connects ER stress with energy balance and glucose homeostasis. Cell Metab 2014;20:471–482 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Swyryd EA, Seaver SS, Stark GR. N-(phosphonacetyl)-L-aspartate, a potent transition state analog inhibitor of aspartate transcarbamylase, blocks proliferation of mammalian cells in culture. J Biol Chem 1974;249:6945–6950 [PubMed] [Google Scholar]
- 72.Cortés VA, Curtis DE, Sukumaran S, et al. Molecular mechanisms of hepatic steatosis and insulin resistance in the AGPAT2-deficient mouse model of congenital generalized lipodystrophy. Cell Metab 2009;9:165–176 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Perry RJ, Zhang D, Zhang XM, Boyer JL, Shulman GI. Controlled-release mitochondrial protonophore reverses diabetes and steatohepatitis in rats. Science 2015;347:1253–1256 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Perry RJ, Kim T, Zhang XM, et al. Reversal of hypertriglyceridemia, fatty liver disease, and insulin resistance by a liver-targeted mitochondrial uncoupler. Cell Metab 2013;18:740–748 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Combs TP, Nagajyothi, Mukherjee S, de Almeida CJG, et al. The adipocyte as an important target cell for Trypanosoma cruzi infection. J Biol Chem 2005;280:24085–24094 [DOI] [PubMed] [Google Scholar]
- 76.Park J, Kusminski CM, Chua SC, Scherer PE. Leptin receptor signaling supports cancer cell metabolism through suppression of mitochondrial respiration in vivo. Am J Pathol 2010;177:3133–3144 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Park J, Euhus DM, Scherer PE. Paracrine and endocrine effects of adipose tissue on cancer development and progression. Endocr Rev 2011;32:550–570 [DOI] [PMC free article] [PubMed] [Google Scholar]