Abstract
Objectives
To evaluate the effect of functional overloading on the transplantation of muscle derived stem cells (MDSCs) into dystrophic muscle and the ability of transplanted cells to increase dystrophic muscle’s ability to resist overloading-induced weakness.
Design
Cross-sectional.
Setting
Laboratory.
Animals
Male mice (N=10) with a dystrophin gene mutation.
Interventions
MDSCs were intramuscularly transplanted into the extensor digitorum longus muscle (EDL). Functional overloading of the EDL was performed by surgical ablation of the EDL’s synergist.
Main Outcome Measures
The total number of dystrophin-positive fibers/cross-section (as a measure of stem cell engraftment), the average number of CD31+ cells (as a measure of capillarity), and in vitro EDL contractile strength. Independent t tests were used to investigate the effect of overloading on engraftment, capillarity, and strength. Paired t tests were used to investigate the effect of MDSC engraftment on strength and capillarity.
Results
MDSC transplantation protects dystrophic muscles against overloading-induced weakness (specific twitch force: control 4.5N/cm2±2.3; MDSC treated 7.9N/cm2±1.4) (P=.02). This improved force production following overloading is concomitant with an increased regeneration by transplanted MDSCs (MDSC: 26.6±20.2 dystrophin-positive fibers/cross-section; overloading + MDSC: 170.6±130.9 dystrophin-positive fibers/cross-section [P=.03]). Overloading-induced increases in skeletal muscle capillarity is significantly correlated with increased MDSC engraftment (R2=.80, P=.01).
Conclusions
These findings suggest that the functional contribution of transplanted MDSCs may rely on activity-dependent mechanisms, possibly mediated by skeletal muscle vascularity. Rehabilitation modalities may play an important role in the development of stem cell transplantation strategies for the treatment of muscular dystrophy.
Keywords: Contractile function, Duchenne muscular dystrophy, Skeletal muscle, Stem cells
Unlike many other forms of adult tissue, healthy skeletal muscle displays a remarkable ability for regeneration after injury. This regenerative capacity relies primarily on a population of progenitor cells, muscle satellite cells, which reside between the basement membrane and the sarcolemma of muscle fibers.1 After injury, these normally quiescent satellite cells become activated, proliferate, and fuse to form myofibers.2 Both satellite cell activation and proliferation are necessary for skeletal muscle regeneration. Muscles that show declines in the absolute numbers of satellite cells, as seen with aging3 and 4 and DMD,5 and 6 are also characterized by a decreased regenerative capacity.
In healthy muscle, the protein dystrophin helps maintain membrane integrity during muscle contraction. In dystrophic muscle, an X-linked mutation in the dystrophin gene leads to a critical or complete loss of this membrane-bound protein.7, 8, 9 and 10 Dystrophic muscle fibers have a decreased resistance to muscle injury and become inflamed, necrotic, and fibrotic, ultimately resulting in severe muscle wasting and muscle fatigue.11 and 12 Therefore, dystrophic muscle becomes unable to tolerate the loading typical of everyday activities. Clinically, the decreased resistance to mechanical loading of dystrophic muscle is manifest by severe muscle weakness that renders children with DMD unable to ambulate by their early teens.
Although the past 20 years have seen significant efforts dedicated to developing biologic and cell-based therapies to ameliorate the decreased muscle function seen in DMD, there is currently no effective treatment available. Investigations performed in animal models of DMD provide important insights for a greater understanding of the pathogenesis of this disorder in humans and a vehicle for evaluating the efficacy of regenerative medicine approaches.13
Among the most promising methods investigated, the transplantation of healthy, activated satellite cells (myoblasts) into dystrophic mouse skeletal muscle offers the possibility of restoring the dystrophin protein into the host muscle. Transplanted myoblasts are believed to fuse together, thereby forming new, dystrophin-positive fibers, or fuse with damaged host myofibers, thereby aiding in regeneration. However, this treatment has been met with limited success, in part because of the massive cell death after transplantation of myoblasts.14 and 15 MDSCs, on the other hand, appear to show an enhanced transplantation capacity, primarily because of their strong capacity for self-renewal, their ability to undergo multipotent differentiation, their immune-privileged behavior, and their ability to survive because of an increased resistance to stress.16,17 and 18 These characteristics may favor the survival of the MDSCs after transplantation and, therefore, their use in the development of biologic therapies. Previous results performed in animal models of dystrophy show that the transplantation of MDSCs may, at least partially, restore normal histology of female dystrophic muscle for up to 3 months after transplantation.16 In humans, the intramuscular transplantation of MDSCs has been shown to be safe and feasible.19 Still undetermined, however, is the contribution of these engrafted stem cells to the overall physiologic functioning of the host muscle and how well transplanted stem cells respond to increased mechanical demands, such as muscle loading.
Healthy skeletal muscle is known to hypertrophy in response to overloading and stretching. It is widely accepted that environmental cues resulting from muscle injury or exercise stimulate resident satellite and stem cells to proliferate and differentiate to form multinucleated myotubes. However, how exercise and/or mechanical loading affect the proliferation and differentiation of transplanted stem cells has received much less attention. In the face of increased muscle degeneration resulting from overloading of dystrophic muscles, are transplanted MDSCs able to enhance muscle regeneration and ultimately improve muscle functioning?
The purpose of this study was to evaluate the effect of functional overloading on the efficacy of MDSCs transplantation and to investigate a possible role of skeletal muscle vascularity on stem cell engraftment. In addition, we investigated whether transplanted stem cells were able to increase dystrophic muscle’s ability to withstand functional overloading.
Methods
Animals
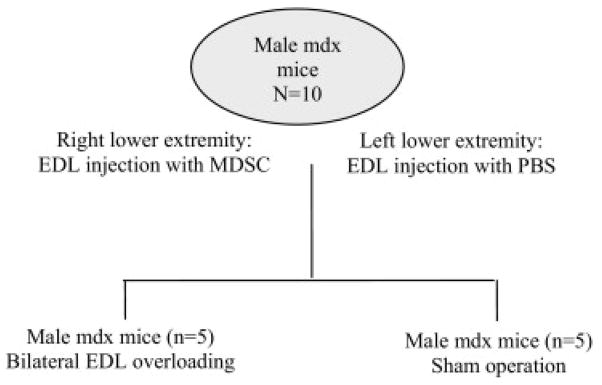
A total of 10 mdx mice, an animal model of DMD (C57BL/10ScSc-DMDmdx), that were 6 months of age were used in this study (fig 1). Animals were obtained from Jackson Laboratories, Bar Harbor, Maine, or were bred in-house. Mdx mice lack the protein dystrophin because of a spontaneously occurring point mutation in the dystrophin gene.20 These animals not only provide a useful model of muscular dystrophy but also provide a working model for tracking the fate of transplanted stem cells. Because engrafted donor cells differentiate into myotubes and muscle fibers expressing dystrophin, they can be easily distinguished from host cells. All animal procedures used in this study were approved by the Institutional Animal Care and Use Committee of Children’s Hospital of Pittsburgh.
Fig 1.
An animal grouping schematic.
Muscle-Derived Stem Cells Isolation
MDSCs were isolated from skeletal muscle biopsies obtained from female 3-week-old healthy mice by using a previously described modified preplate technique.16 and 17 MDSCs were cultured in normal growth medium consisting of Dulbecco’s Modified Eagle Medium 11995-073a supplemented with 10% fetal bovine serum, 10% HS, 1% penicillin/streptomycin, and 0.5% chick embryo extract. The MDSCs were grown and maintained in growth medium to approximately 30% confluence and were subsequently passaged.
On the day of injection, MDSCs were bathed in 20% trypsin and spun at 2000rpm for 5 minutes. The resulting pellet was resuspended in PBS + 0.1% microsphere beads solution at a concentration of 1.0 to 1.4 × 107 cells/mL.
Muscle-Derived Stem Cells Transplantation and Muscle Overloading
All animals were anesthetized with 2% isoflurane administered by inhalation. Bilateral lower extremities were shaved, and the skin was sterilized with iodine. An anterior longitudinal skin incision was performed to permit exposure of the entire anterior crural muscles. Right-side EDL muscles were injected locally with 0.7 to 1.0 × 105 MDSCs suspended in 7 μL PBS + 0.1% green fluorescent microsphere beads. Left-side control EDLs were injected with an equal volume of PBS + 0.1% green fluorescent microsphere beads. The track of the needle was oriented along the direction of the EDL muscle fibers for injection. To induce muscle hypertrophy, we used tenotomy of the synergistic muscle, which increases the loads applied on the intact muscle. Bilateral EDL muscles of the animals in the OL group (n=5) were functionally overloaded by surgical ablation of the tibialis anterior muscle, as described by Tsika et al.21 EDL functional overloading has been previously shown to result in a decreased twitch and tetanic tensions of dystrophic muscles of comparably aged animals.22 Briefly, at the time of MDSC or PBS injection, the tibialis anterior muscle was excised completely by cutting the distal tendon and cutting away the muscle from the tibialis bone. Bilateral EDLs were overloaded to minimize favoring of a single hind limb. After removal of the tibialis anterior, the skin was closed with 4.0 silk sutures. Animals then received a subcutaneous injection of ketorolac (1–5mg/kg body weight) to relieve pain. Animals was observed until recovery from anesthesia, at which time they were returned to their home cage with access to ad libitum food and water. Even immediately after the overloading surgery, animals showed minimal gait alterations. Two to 3 days after injury, no impairment in the locomotion of the overloaded animals was observed when compared with nonoverloaded control animals. The remaining 5 animals that received only MDSC and PBS injections (MDSC-only and PBS-only groups) were anesthetized and subjected to a sham operation, in which the fascia encasing the tibialis anterior was bluntly separated from the EDL. The skin was then sutured closed, and animals were returned to their cages.
Muscle Contractile Testing
Four weeks from the time of overloading and/or injection, animals were anesthetized intraperitoneally by using 40 to 80mg/kg body weight pentobarbital, and EDL muscles were removed for physiologic testing. In vitro contractile properties were measured in Reese-Simpson Mammalian Ringers bath (135mM Na+, 5mM K+, 2mM Ca2+, 1mMMg2+, 121mM C1−, 25mM HCO3−, 11mM glucose, 0.3mM glutamic acid, 0.4mM glutamine, 5mM N,N-bis-(2-hydroxyethyl)-2-aminoethanesulfonic acid (BES) buffer, and 0.008% d-tubocurarine chloride) maintained at 25°C by using a protocol as described by Mueller et al.23 Muscles were placed vertically in the bath secured at each end via a Michel clipb (fig 2). The pH of the bathing solution was stabilized at 7.4 by equilibration with a gas mixture of 95% O2/5% CO2. One tendon was attached to an isometric force transducer and length servo system (model 305Bc); the other tendon was connected to a mounting pin of a glass tissue support rodd connected in series to a micropositioner at the bottom of the bath. Muscles were stimulated (Grass model S-88e stimulator and current amplifier) by 1.0-ms duration monophasic rectangular pulses of cathodal current delivered through 2 platinum field electrodes (spaced approximately 1cm apart on either side of the muscle, with the EDL positioned vertically in between both electrodes). The current necessary to obtain peak twitch force for each muscle was between 250 and 300mA. By using a micropositioner, muscles were first adjusted to their Lo, defined as the length at which maximum isometric twitch tension was produced. Pt, TPT (the time from the onset of the stimulus to the peak force of the TPT), and ½RT (TPT to one-half of the relaxation) were first obtained from a twitch contraction. Po was assessed by using a stimulation frequency of 75 pulses a second delivered in a 500-ms duration train. Output was recorded on a digital oscilloscope.c
Fig 2.
A contractile testing schematic representation.
The lengthening activation, or eccentric contraction, paradigm consisted of one 500-ms tetanic contraction initiated at Lo. This protocol was designed to induce mechanical stress to the muscle and to determine the effect of functional overloading on the ability of the muscle to resist eccentric contraction injuries.24 After the first 300ms of the stimulus train, the muscle was lengthened from 100 to 110% Lo at a constant velocity of 1.0Lo/s. Stimulus trains performed by using this protocol were repeated for a total of 10 cycles separated by 2 minutes of rest (at Lo). After the eccentric contraction protocol, muscles were removed from the chamber and weighed. The mean CSA of each muscle was estimated by dividing the muscle mass by fiber length and the density of mammalian skeletal muscle (1.056g/cm3). Force (N) was then normalized to the CSA for the determination of specific twitch (Pt/CSA) and tetanic forces (Po/CSA). The investigator collecting contractile testing data was blinded to treatment group.
Immunofluorescent Staining
Immediately after contractile testing, muscles were snap-frozen in liquid nitrogen–cooled 2-methyl butane and cryosectioned at 10μm thickness. For dystrophin and CD31 (as a measure of muscle capillarity) staining, muscle sections were fixed in 4% formalin for 5 minutes and then washed twice in PBS. Nonspecific binding was then blocked for 1 hour by using PBS/10% DS, 5% HS for dystrophin, or CD31 stains, respectively. Sections were then incubated for 1 hour at room temperature with either a rabbit anti-mouse polyclonal dystrophin antibody (1:300 dilution in 10% DS) for dystrophin staining or with a rat anti-mouse primary antibody (1:300 dilution in 5% HS) for CD31 staining. After 3 PBS washes, antidystrophin sections were treated for 1 hour with a Cy3-labeled donkey anti-rabbit secondary antibody (diluted 1:200 in 10% DS), whereas anti-CD31 sections were treated for 1 hour with a 555-labeled goat anti-rat secondary antibody (diluted 1:300 in 5% HS). All samples were then again washed 3 times in PBS and incubated with 4′-6-diamidino-2-phenylindole (diluted 1:1000 in PBS) for 10 minutes. Muscle sections were evaluated by using fluorescent microscopy.
The total number of dystrophin-positive fibers/cross-section was counted by using Northern Eclipse software.f The total number of dystrophin-positive fibers within the cross-section containing the greatest number of green fluorescent beads (as an indication of the injection site) was photographed (20×) and manually counted. Similarly, the number of capillaries was manually counted as the total number of CD31-positive cells averaged over 3 areas within the single muscle cross-section of maximal engraftment. All analyses were performed by a blinded investigator.
Statistical Analysis
Data were analyzed by using statistical software package (SPSS 14.0).g Because most of our data showed no violations of assumptions (equality of variance and normality of distribution), parametric tests were used for comparison between injection and treatment groups. However, we used a heteroscedastic t test to determine group differences between MDSC engraftment in OL/MDSC versus MDSC groups because the data violated the assumption of equality of variances.
Paired t tests were used to compare muscle characteristics between MDSC- and PBS-treated muscles because the same animals for these analyses were used. Independent t-tests were used to compare between-group (ie, OL vs injection only) differences. A Spearman ρ correlation was used to determine the relationship between the number of dystrophin-positive fibers and the number of CD31 cells. A 2-way repeated-measures analysis of variance was used to compare contractile data after eccentric contraction across experimental groups, with a subsequent multiple comparison Scheffé post hoc analysis. Statistical significance was established, a priori, at P less than or equal to .05.
Results
Animal and Muscle Characteristics
All animals used in these experiments were age and sex matched for groups. Table 1 provides a summary of animal age, body weight, EDL mass, EDL Lo, TPT, and ½RT. We found no significant difference in the overall body weights of animals that had been exposed to functional overload of the EDL (OL/PBS or OL/MDSC) when compared with injection only (PBS only or MDSC only) control counterparts. However, OL/MDSC muscles weighed significantly more when compared with PBS-only muscles (P=.01), MDSC-only muscles (P=.01), and OL/PBS muscles (P=.04). Muscles that had been overloaded (OL/PBS and OL/MDSC) had significantly increased Lo in comparison to PBS-only counterparts.
Table 1.
Animal and Extensor Digitorum Longus Characteristics
| Group | Age (d) | Body Weight (g) | EDL Mass (mg) | EDL Length (Lo [cm]) | TPT (ms) | ½RT (ms) |
|---|---|---|---|---|---|---|
| OL/PBS | 219.6±1.3 | 35.5±3.1 | 19.0±3.0 | 1.53±0.02* | 21.2±1.1 | 22.8±5.4 |
| OL/MDSC | 219.6±1.3 | 35.5±3.1 | 21.0±2.01*†‡ | 1.53±0.02* | 22.8±1.1† | 23.0±3.7 |
| PBS only | 222.6±0.5 | 34.1±1.6 | 18.0±2.00 | 1.49±0.02 | 22.8±1.8 | 23.4±2.5 |
| MDSC only | 222.6±0.5 | 34.1±1.6 | 18.0±1.00 | 1.51±0.05 | 22.4±2.6 | 21.4±3.1 |
NOTE. Mean ± SD. n=5 muscles per group.
Significantly different than PBS only (P ≤ 0.05).
Significantly different than OL/PBS (P ≤ 0.05).
Significantly different than MDSC only (P ≤ 0.05).
Contribution of Transplanted Muscle-Derived Stem Cells to the Overall Contractile Functioning of Dystrophic Muscle
We observed a trend for an increased specific tetanic force (P=.06) for MDSC-only EDLs (36.5N/cm2±4.2) when compared with PBS-only controls (25.5 N/cm2±10.2) (fig 3). There was no significant difference in the specific twitch force of MDSC-only and PBS-only muscles (9.2N/cm2±1.3 and 7.0N/cm2±1.9, respectively). Similarly, when considering resistance to eccentric contraction-induced injury, MDSC-only muscles initiated the protocol with a greater force generation, but by the tenth contraction, there was no difference between MDSC-only muscles and PBS-only muscles. At the end of the eccentric contraction protocol, PBS-only muscles only produced an average of 35.4%±0.09% twitch force relative to baseline values. Similarly, MDSC-only muscles produced only 34.2%±0.17% twitch force relative to baseline values. In fact, a sharp decline in force production for both groups was evident after only a few contractions. There was no significant difference in ½RT or TPT between PBS-only and MDSC-only groups (see table 1).
Fig 3.
Contractile testing: specific twitch (Pt/CSA) and tetanic (Po/CSA) forces among 4 groups. NOTE. Mean N/cm2 ± SD, n=5 per group. *Denotes statistically significant from OL/PBS (P≤.05). †Denotes statistically different from OL/MDSC (P≤.05).
Ability of Muscle-Derived Stem Cells to Protect Dystrophic Muscle From Overloading
There was a significant decrease in the specific twitch force of OL/PBS muscles (4.5N/cm2±2.3) when compared with PBS-only muscles (7.0N/cm2±1.9) (P=.05); there was a trend for a decrease in the specific tetanic force of OL/PBS muscle (14.9N/cm2±9.1) compared with PBS-only controls (25.5N/cm2±10.2) (P=.06) (see fig 3), suggesting that the increased loading imposed on the EDL contributed to muscle weakness and decreased force producing capacity. In comparison, OL/MDSC muscles produced significantly greater specific twitch force (7.9N/cm2±1.4) (P=.02) and specific tetanic force (26.4N/cm2±7.2) (P=.02) compared with OL/PBS muscles. In fact, there was no significant difference between OL/MDSC muscles and PBS-only controls. We also investigated whether coupling functional overload with MDSC transplantation could increase the resistance of dystrophic muscle to eccentric contraction-induced muscle injury. OL/PBS muscles showed a more gradual decline in force production compared with both PBS-only and MDSC-only muscles. At the end of the eccentric contraction protocol, OL/PBS muscles produced 50.8%±0.03% twitch force relative to baseline values. OL/MDSC muscles showed the greatest resistance to eccentric contraction injury at the end of the protocol and produced an average of 57.7%±0.17% of the baseline twitch force value. OL/MDSC muscles also showed significantly longer TPT when compared with OL/PBS (P=.05). Post hoc analysis revealed that data showed a large effect size, and our achieved power was greater than 93% for contractile variables, therefore greatly decreasing the likelihood for type II error even though our sample size was small.
Effect of Overloading on Muscle-Derived Stem Cells Engraftment
Immediately after contractile testing, muscles were analyzed for the extent of stem cell engraftment as determined by the number of dystrophin-positive fibers. MDSC-only muscles showed an average of 26.6±20.2 dystrophin-positive fibers/cross-section. On the other hand, OL/MDSC muscles showed a 6-fold increase in the number of dystrophin-positive fibers when compared with PBS-only controls, and the average number of dystrophin-positive fibers/cross-section in OL/MDSC muscles was 170.6±130.9. Four weeks of EDL overloading significantly enhanced the efficacy of stem cell transplantation into dystrophic skeletal muscle (P=.03) ( Fig 4 and Fig 5).
Fig 4.
Immunofluorescence of dystrophin (red) and nuclei (blue) in the EDL of (A) MDSC-only and (B) OL/MDSC muscles. Green represents fluorescent microspheres used to localize injection site (20× magnification).
Fig 5.
Comparison of the efficacy of stem cell engraftment (as determined by the number of dystrophin-positive fibers/cross-sectional area) for MDSC-only and OL/MDSC muscles (n=5). NOTE. Mean ± SD. *Denotes statistically significant P≤.05.
Role of Skeletal Muscle Capillarity on Stem Cell Engraftment
The transplantation of MDSCs into dystrophic muscle resulted in a significantly increased skeletal muscle capillarity (P=.000) when compared with PBS-injected counterparts. The addition of overloading also increased skeletal muscle capillarity after injection with PBS (P=.026) and trended toward an increased capillarity after MDSC injection (P=.055). There was significant direct correlation between the number of dystrophin-positive fibers and the number of CD31-positive cells (R2=0.80) (P=.01).
Discussion
A limited understanding of environmental factors controlling stem cell transplantation capacity and how these factors relate to meaningful improvements in skeletal muscle function has represented a major barrier in the clinical translation of cell therapies for the treatment of DMD. In this article, we presented data to show that in vivo mechanical loading enhances the efficacy of stem cell transplantation and that this increased transplantation capacity is related to increased skeletal muscle vascularity. We have also presented evidence suggesting that local injection of MDSCs can increase the resistance of dystrophic skeletal muscle to weakness caused by overloading.
Histologic analysis of MDSC-only muscle versus OL/MDSC muscle revealed a significant increase in the total number of dystrophin-positive myofibers. Similar findings have been shown by Palermo et al.25 Although the systemic delivery of bone marrow–derived stem cells results in minimal muscle contribution in the absence of an overt injury,26 they showed that there was a 10-fold increase in the percentage of GFP-positive fibers (an indication of stem cell engraftment) in animals that had been exposed to a forced treadmill running protocol and a 32-fold increase in GFP-positive fibers in functionally overloaded muscles.27 However, the number of regenerated fibers in these studies appeared to be functionally irrelevant, and, unlike our study, measures of force producing capacity of the treated muscles were not investigated. Nonetheless, after increased mechanical loading, some physiologic cues accompanying microdamage and inflammation of the myofibers were likely responsible for increasing the mobilization of stem cells from the bloodstream. Similarly, Bouchentouf et al28 found that the addition of a 4-week swimming protocol after the local injection of myoblast into the tibialis anterior of mdx mice improved graft success 1.9-fold (as determined by the number of dystrophin positive fibers). Increased transplantation of the myoblasts was again attributed to increased myofiber breakage, which stimulated increased donor cell recruitment and fusion. Therefore, inflammatory infiltrate through enhanced vascularity may be an important mechanism for donor cell participation in myofiber regeneration. Indeed, in our study, the increase in dystrophin-positive fibers was directly significantly correlated with an increased number of CD31-positive cells, indicating a relationship between skeletal muscle vascularity in stem cell proliferation and/or differentiation.
Stem cell engraftment efficacy is clearly dependent on a myriad of factors, the most important of which may be the secretion of local growth factors.29 and 30 One particular cytokine, VEGF, has been of particular interest in the past few years because it enhances stem cell proliferation both in vitro31 and in vivo.17 Importantly, exercise has been shown to increase growth factor secretion of important cytokines, such as VEGF, and increases satellite cell pools in normal skeletal muscle.32 Therefore, it is possible that the well-known effect of increased VEGF secretion that results from muscle loading may result in an increased proliferation of transplanted cells, ultimately allowing for an increased number of cells available to differentiate into myofibers. Alternatively, it is possible that the mechanical loading results in an increased secretion of VEGF by transplanted cells, therefore promoting angiogenesis. Payne et al33 recently showed that the addition of mechanical stimulation upregulates the secretion of VEGF by MDSCs. Taken together, it is likely that the addition of muscle loading increases VEGF secretion, which stimulates MDSC proliferation, thereby further increasing VEGF secretion by transplanted cells. Future studies that investigate the relationship between VEGF and MDSC transplantation and how these factors can be noninvasively manipulated to improve the function of dystrophic skeletal muscle are warranted. To determine the efficacy of transplantation, measures, such as contractile strength, that can be related to improvement in functional outcomes are critical. In our study, stem cell injection alone did not improve the contractile function of the treated muscle, and we did not observe a significant difference in the specific twitch or tetanic force of MDSC-only muscles when compared with PBS-only muscles. Similarly, Mueller et al23 showed that even 9 weeks after intramuscular MDSC transplantation, there was no significant improvement in the contractile function of dystrophic mouse skeletal muscle, despite marked engraftment of the transplanted cells. In their study, histologic analysis revealed that the dystrophin expression was limited to only the injected site along the muscle belly (distance, 60–900μm). Authors suggested that this may help explain the failure of cellular therapies to result in marked improvements in the muscle’s overall physiologic functioning. This is an important consideration, given that traditionally successful stem cell transplantation into skeletal muscle had been based on histologic findings alone. These current findings further suggest that the extent of stem cell engraftment and, specifically, the absolute number of dystrophin-positive fibers does not necessarily coincide with improvements in muscle contractile strength and functioning. The beneficial effect of transplanted stem cells may be independent of the engraftment of the cells themselves. Instead, the benefit may be more paracrine in nature and lie in promoting the regenerative capacity of the host itself. This should be the topic of future investigations.
Unlike humans, mouse models of muscular dystrophy show few symptoms of the disease, despite the lack of the dystrophin gene.22 and 34 However, in muscles of the mouse exposed to continuous use and an increased rate of work production, such as the diaphragm, increased skeletal muscle deterioration is evident.35 and 36 Similarly, functional overloading through ablation of the EDL synergist of mdx mice has been previously shown to result in a significant decrease in the twitch and tetanic tensions of 5- to 8-month-old animals and increased evidence of myofibrillar damage, suggesting that these muscles were unable to withstand long-term mechanical overloading.22 Therefore, functional overloading, as performed in our study, may provide a more comparable animal model for the chronic skeletal muscle deterioration observed in children with DMD.
By using a working model of loading-induced muscle damage in the mdx mouse, the contractile characteristics of overloaded muscles that had been treated with MDSCs were evaluated. Our results show that intramuscular transplantation of stem cells appears to protect the muscle from the overload-induced muscle weakness characterizing dystrophic muscle. MDSC transplanted muscles that were overloaded were significantly stronger than overloaded counterparts that were treated with PBS. It has been suggested that the transplantation of stem cells obtained from normal animals has the potential to create a reservoir of normal stem cells that have the capability of restoring dystrophin expression and enhancing muscle regeneration after injury.16 When exposed to increased mechanical demands and likely myofiber microdamage, it is possible that transplanted stem cells become recruited to contribute to the overall muscle repair. More importantly, this enhanced repair appears to result in an overall increased functional capacity of the treated muscle. This is supported by the increased myogenic regeneration of transplanted MDSCs that we observed histologically. Whether these stem cells, in the face of muscle damage, assist in regeneration through the secretion of growth factors necessary for myofiber repair and/or if they are themselves stimulated toward increased myofiber differentiation remains to be investigated in future studies.
Study Limitations
The animal model used in this study is a limitation, given that mdx mice do not show a skeletal muscle pathology comparable to the symptomatic presentation of DMD in humans. In fact, mdx mice show a regenerative capacity similar to normal controls.22 and 34 Double dystrophin and utrophin knockout mice may serve as a better model of DMD because they show significant muscle pathology. However, these mice have a much shorter lifespan (3–5mo), which limits the ability to evaluate the effects of MDSC transplantation over time. In addition, physiologic contractile testing is not a clinically relevant measure of skeletal muscle function. However, because we are only treating single muscles and not muscle groups, changes in overall function of the mice are not likely to be observed. Along these lines, field electric stimulation, as used in this study, does not provide information as to whether the MDSC-derived muscle fibers are innervated via a neuromuscular junction. Further investigations should investigate the contractile strength of MDSC-treated muscles by using direct nerve stimulation.
Conclusions
Taken together, the findings of the current study suggest that transplanted stem cells can respond, both histologically and physiologically, to in vivo mechanical stimulation. The maintenance of force-producing capacity of MDSC-treated muscles after 4 weeks of overloading suggests that transplanted stem cells become activated and fuse to form newly regenerated myofibers in the face of muscle damage, as supported by the increased expression of dystrophin-positive fibers in OL/MDSC muscle. The strong relationship between skeletal muscle capillarity and stem cell engraftment provides some direction as to the possible mechanism by which muscle loading enhances stem cell engraftment and preserves skeletal muscle function. The present findings suggest that rehabilitation modalities may play an important role in the development of regenerative medicine strategies for the treatment of DMD. Future studies should seek to elucidate exercise-related growth factors that may play a critical role in transplanted stem cell recruitment toward a myogenic lineage.
Acknowledgments
We thank Terrence L. O’Day, MS, for his help with contractile testing; Mary Synnott for graphic assistance; and Christian Niyonkuru, MS, for statistical assistance.
Supported in part by the Competitive Medical Research Fund of the University of Pittsburgh; the K12 for Physical and Occupational Therapists–A Comprehensive Opportunities in Rehabilitation Research Training program (grant no. 1K12HD055931-01); the Henry J. Mankin Endowed Chair for the Orthopedic Research at the University of Pittsburgh; the William F. and Jean W. Donaldson Chair at Children’s Hospital of Pittsburgh, Pittsburgh, PA; and the NIH (grant no. 5R01 AR49684).
List of Abbreviations
- CSA
cross-sectional area
- DS
donkey serum
- DMD
Duchenne muscular dystrophy
- mdx
dystrophin gene mutation
- EDL
extensor digitorum longus
- GFP
green fluorescent protein
- ½RT
half relaxation time
- HS
horse serum
- Lo
optimal length
- MDSC
muscle-derived stem cell
- OL
overload
- PBS
phosphate-buffered saline
- Pt
peak twitch tension
- Po
peak tetanic tension
- TPT
time to peak twitch tension
- VEGF
vascular endothelial growth factor
Footnotes
Invitrogen Corp, 1600 Faraday Ave, Carlsbad, CA 92008.
Fine Science Tools Inc, 373-G Vintage Park Dr, Foster City, CA 94404-1139.
Aurora Scientific Inc, 360 Industrial Parkway S, Unit 4, Aurora, ON, Canada L4G 3V7.
Radnoti Glass Technology Inc, 227 W Maple Ave, Monrovia, CA 91016.
Grass Technologies, Astro-Med Industrial Park, 600 E Greenwich Ave, West Warwick, RI 02893.
Northern Eclipse Software (Empix Imaging), Cheektowaga, NY.
SPSS Inc, 233 S Wacker Dr, 11th Fl, Chicago, IL 60606.
Presented as an abstract to Engineering Tissues: Replace, Repair, Regenerate, March 8–11, 2007, Hilton Head, SC.
An organization with which 1 or more of the authors is associated has received or will receive financial benefits from a commercial party having a direct financial interest in the results of the research supporting this article.
References
- 1.Mauro A. Satellite cell of skeletal muscle fibers. J Biophys Biochem Cytol. 1961;9:493–495. doi: 10.1083/jcb.9.2.493. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Schultz E, Jaryszak DL, Valliere CR. Response of satellite cells to focal skeletal muscle injury. Muscle Nerve. 1985;8:217–222. doi: 10.1002/mus.880080307. [DOI] [PubMed] [Google Scholar]
- 3.Kadi F, Charifi N, Denis C, Lexell J. Satellite cells and myonuclei in young and elderly women and men. Muscle Nerve. 2004;29:120–127. doi: 10.1002/mus.10510. [DOI] [PubMed] [Google Scholar]
- 4.Snow MH. The effects of aging on satellite cells in skeletal muscles of mice and rats. Cell Tissue Res. 1977;185:399–408. doi: 10.1007/BF00220299. [DOI] [PubMed] [Google Scholar]
- 5.Webster C, Blau HM. Accelerated age-related decline in replicative life-span of Duchenne muscular dystrophy myoblasts: implications for cell and gene therapy. Somat Cell Mol Genet. 1990;16:557–565. doi: 10.1007/BF01233096. [DOI] [PubMed] [Google Scholar]
- 6.Heslop L, Morgan JE, Partridge TA. Evidence for a myogenic stem cell that is exhausted in dystrophic muscle. J Cell Sci. 2000;113(Pt 12):2299–2308. doi: 10.1242/jcs.113.12.2299. [DOI] [PubMed] [Google Scholar]
- 7.Hoffman EP, Brown RH, Jr, Kunkel LM. Dystrophin: the protein product of the Duchenne muscular dystrophy locus. Cell. 1987;51:919–928. doi: 10.1016/0092-8674(87)90579-4. [DOI] [PubMed] [Google Scholar]
- 8.Watkins SC, Hoffman EP, Slayter HS, Kunkel LM. Immunoelectron microscopic localization of dystrophin in myofibres. Nature. 1988;333:863–866. doi: 10.1038/333863a0. [DOI] [PubMed] [Google Scholar]
- 9.Bonilla E, Samitt CE, Miranda AF, et al. Duchenne muscular dystrophy: deficiency of dystrophin at the muscle cell surface. Cell. 1988;54:447–452. doi: 10.1016/0092-8674(88)90065-7. [DOI] [PubMed] [Google Scholar]
- 10.Zubrzycka-Gaarn EE, Bulman DE, Karpati G, et al. The Duchenne muscular dystrophy gene product is localized in sarcolemma of human skeletal muscle. Nature. 1988;333:466–469. doi: 10.1038/333466a0. [DOI] [PubMed] [Google Scholar]
- 11.Ervasti JM, Campbell KP. Membrane organization of the dystrophin-glycoprotein complex. Cell. 1991;66:1121–1131. doi: 10.1016/0092-8674(91)90035-w. [DOI] [PubMed] [Google Scholar]
- 12.Ibraghimov-Beskrovnaya O, Ervasti JM, Leveille CJ, Slaughter CA, Sernett SW, Campbell KP. Primary structure of dystrophin-associated glycoproteins linking dystrophin to the extracellular matrix. Nature. 1992;355:696–702. doi: 10.1038/355696a0. [DOI] [PubMed] [Google Scholar]
- 13.Vainzof M, Ayub-Guerrieri D, Onofre PC, et al. Animal models for genetic neuromuscular diseases. J Mol Neurosci. 2008;34:241–248. doi: 10.1007/s12031-007-9023-9. [DOI] [PubMed] [Google Scholar]
- 14.Fan Y, Maley M, Beilharz M, Grounds M. Rapid death of injected myoblasts in myoblast transfer therapy. Muscle Nerve. 1996;1919:853–860. doi: 10.1002/(SICI)1097-4598(199607)19:7<853::AID-MUS7>3.0.CO;2-8. [DOI] [PubMed] [Google Scholar]
- 15.Hodgetts SI, Beilharz MW, Scalzo AA, Grounds MD. Why do cultured transplanted myoblasts die in vivo?: DNA quantification shows enhanced survival of donor male myoblasts in host mice depleted of CD4+ and CD8+ cells or Nk1.1+ cells. Cell Transplant. 2000;9:489–502. doi: 10.1177/096368970000900406. [DOI] [PubMed] [Google Scholar]
- 16.Qu-Petersen Z, Deasy B, Jankowski R, et al. Identification of a novel population of muscle stem cells in mice: potential for muscle regeneration. J Cell Biol. 2002;157:851–864. doi: 10.1083/jcb.200108150. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Oshima H, Payne TR, Urish KL, et al. Differential myocardial infarct repair with muscle stem cells compared to myoblasts. Mol Ther. 2005;12:1130–1141. doi: 10.1016/j.ymthe.2005.07.686. [DOI] [PubMed] [Google Scholar]
- 18.Péault B, Rudnicki M, Torrente Y, et al. Stem and progenitor cells in skeletal muscle development, maintenance, and therapy. Mol Ther. 2007;15:867–877. doi: 10.1038/mt.sj.6300145. [DOI] [PubMed] [Google Scholar]
- 19.Torrente Y, Belicchi M, Marchesi C, et al. Autologous transplantation of muscle-derived CD133+ stem cells in Duchenne muscle patients. Cell Transplant. 2007;16:563–577. doi: 10.3727/000000007783465064. [DOI] [PubMed] [Google Scholar]
- 20.Sicinski P, Geng Y, Ryder-Cook AS, Barnard EA, Darlison MG, Barnard PJ. The molecular basis of muscular dystrophy in the mdx mouse: a point mutation. Science. 1989;244:1578–1580. doi: 10.1126/science.2662404. [DOI] [PubMed] [Google Scholar]
- 21.Tsika RW, Herrick RE, Baldwin KM. Interaction of compensatory overload and hindlimb suspension on myosin isoform expression. J Appl Physiol. 1987;62:2180–2186. doi: 10.1152/jappl.1987.62.6.2180. [DOI] [PubMed] [Google Scholar]
- 22.Dick J, Vrbová G. Progressive deterioration of muscles in mdx mice induced by overload. Clin Sci. 1993;84:145–150. doi: 10.1042/cs0840145. [DOI] [PubMed] [Google Scholar]
- 23.Mueller GM, O’Day T, Watchko JF, Ontell M. Effect of injecting primary myoblasts versus putative muscle-derived stem cells on mass and force generation in mdx mice. Hum Gene Ther. 2002;13:1081–1090. doi: 10.1089/104303402753812485. [DOI] [PubMed] [Google Scholar]
- 24.Watchko JF, O’Day TL, Hoffman EP. Functional characteristics of dystrophic skeletal muscle: insights from animal models. J Appl Physiol. 2002;93:407–417. doi: 10.1152/japplphysiol.01242.2001. [DOI] [PubMed] [Google Scholar]
- 25.Palermo AT, Labarge MA, Doyonnas R, Pomerantz J, Blau HM. Bone marrow contribution to skeletal muscle: a physiological response to stress. Dev Biol. 2005;279:336–344. doi: 10.1016/j.ydbio.2004.12.024. [DOI] [PubMed] [Google Scholar]
- 26.Wagers AJ, Sherwood RI, Christensen JL, Weissman IL. Little evidence for developmental plasticity of adult hematopoietic stem cells. Science. 2002;297:2256–2259. doi: 10.1126/science.1074807. [DOI] [PubMed] [Google Scholar]
- 27.Conboy IM, Conboy MJ, Wagers AJ, Girma ER, Weissman IL, Rando TA. Rejuvenation of aged progenitor cells by exposure to a young systemic environment. Nature. 2005;433:760–764. doi: 10.1038/nature03260. [DOI] [PubMed] [Google Scholar]
- 28.Bouchentouf M, Benabdallah BF, Mills P, Tremblay JP. Exercise improves the success of myoblast transplantation in mdx mice. Neuromuscul Disord. 2006;16:518–529. doi: 10.1016/j.nmd.2006.06.003. [DOI] [PubMed] [Google Scholar]
- 29.Lavasani M, Lu A, Peng H, Cummins J, Huard J. Nerve growth factor improves the muscle regeneration capacity of muscle stem cells in dystrophic muscle. Hum Gene Ther. 2006;17:180–192. doi: 10.1089/hum.2006.17.180. [DOI] [PubMed] [Google Scholar]
- 30.Deasy BM, Lu A, Tebbets JC, et al. A role for cell sex in stem cell-mediated skeletal muscle regeneration: female cells have higher muscle regeneration efficiency. J Cell Biol. 2007;177:73–86. doi: 10.1083/jcb.200612094. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Deasy BM, Qu-Peterson Z, Greenberger JS, Huard J. Mechanisms of muscle stem cell expansion with cytokines. Stem Cells. 2002;20:50–60. doi: 10.1634/stemcells.20-1-50. [DOI] [PubMed] [Google Scholar]
- 32.Prior BM, Yang HT, Terjung RL. What makes vessels grow with exercise training? J Appl Physiol. 2004;97:1119–1128. doi: 10.1152/japplphysiol.00035.2004. [DOI] [PubMed] [Google Scholar]
- 33.Payne TR, Oshima H, Okada M, et al. A relationship between vascular endothelial growth factor, angiogenesis, and cardiac repair after muscle stem cell transplantation into ischemic hearts. J Am Coll Cardiol. 2007;50:1677–1684. doi: 10.1016/j.jacc.2007.04.100. [DOI] [PubMed] [Google Scholar]
- 34.Dangain J, Vrbova G. Muscle development in mdx mutant mice. Muscle Nerve. 1984;7:700–704. doi: 10.1002/mus.880070903. [DOI] [PubMed] [Google Scholar]
- 35.Stedman HH, Sweeney HL, Shrager JB, et al. The mdx mouse diaphragm reproduces the degenerative. doi: 10.1038/352536a0. [DOI] [PubMed] [Google Scholar]
- 36.Dupont-Versteegden EE, McCarter RJ. Differential expression of muscular dystrophy in diaphragm versus hindlimb muscles of mdx mice. Muscle Nerve. 1992;15:1105–1110. doi: 10.1002/mus.880151008. [DOI] [PubMed] [Google Scholar]