Abstract
Interstitial granulomatous dermatitis (IGD), a rare disease, is well known to be associated with connective tissue disorders, malignancies and several drugs. We describe this first case of IGD in association with chronic lymphocytic leukaemia/small lymphocytic lymphoma (CLL/SLL). A 66-year-old woman with a 6-year history of untreated CLL/SLL, presented with a 2-month history of progressively worsening eruption of the left thigh, along with fatigue, lymphadenopathy and night sweats. Skin biopsy showed findings consistent with IGD and infiltration of CLL. The eruption was non-responsive to treatment with antibiotics and local steroids. There was a significant improvement in the rash after an initial cycle of chemotherapy (combination therapy with bendamustine and rituximab) and complete resolution by the third cycle, for the treatment of her CLL. We suggest that the possibility of an underlying haematological malignancy should be investigated in patients with a skin rash non-responsive to conventional therapy.
Background
Interstitial granulomatous dermatitis (IGD) is a rare form of non-infectious dermatitis. It was first described by Ackerman in 1993 as a predominantly histiocytic infiltrate of collagen bundles in the dermis.1 2 It is classically associated with autoimmune and connective tissue disorders such as rheumatoid arthritis (RA).3 4 However, it has also been described in association with haematological malignancies such as myelodysplastic syndrome (MDS) and acute promyelocytic leukaemia (APL) (table 1), non-haematological malignancies such as gynaecological cancers5 and several drugs.6–9 In this report, we describe the first case of IGD in association with chronic lymphocytic leukaemia/small lymphocytic lymphoma (CLL/SLL). We suggest that possibility of an underlying haematological malignancy should be investigated in patients with non-responsive dermatosis.
Table 1.
Previously reported cases of IGD associated with haematological malignancies
| Author(s) | Year | Age (years) | Gender | Haematological malignancy | Timing of presentation | Treatment | Outcomes |
|---|---|---|---|---|---|---|---|
| Balin et al13 | 2011 | 71 | Male | MDS | Before malignancy | Lenalidomide | Clearance of IGD |
| Patsinakidis et al8 | 2014 | 73 | Male | MDS | Before malignancy | 5-Azacytidine | Clearance of IGD |
| Sangueza et al3 |
2008 | 28 | Female | APL | Before malignancy | Chemotherapy and antibiotics |
No clearance of IGD |
APL, acute promyelocytic leukaemia; IGD, granulomatous dermatitis; MDS, myelodysplastic syndrome.
Case presentation
A 66-year-old Caucasian woman with a 6-year history of mainly asymptomatic untreated CLL/SLL presented with an eruption of the left thigh that had started 2 months before presentation. The skin eruption began as a coin sized annular pruritic area, initially attributed, by the patient, to a bug bite. The eruption progressed to involve a bigger area on the anterior thigh and lower abdomen, despite empiric treatment with antibiotics such as doxycycline and cephalexin. The rash was preceded by a few weeks of worsening cervical lymphadenopathy, new onset of fatigue and night sweats. The fatigue and lymphadenopathy worsened gradually along with the enlarging skin lesions; however, the patient denied any weight loss. She was known to be allergic to erythromycin, meperidine, penicillin, sulfonamide antibiotics, shellfish, latex, pork and pollen. Her only current medication was cetirizine, which was stopped due to suspicion of drug-induced rash. Medical history was positive for a long-standing history of fibrocystic disease of the breast, a 40-year-old history of a suspicious coccidioidomycosis lung lesion—seen on a chest X-ray taken for employment screening—which was never treated, as she had shown no respiratory symptoms at the time, asymptomatic mitral valve prolapse, history of rectal prolapse, stress incontinence, osteoarthritis and anxiety. Surgical history was positive for hysterectomy, orthopaedic surgery on the left ankle after a traumatic fracture and left total hip arthroplasty after a fall 5 years prior to this presentation.
Regarding the patient's CLL/SLL diagnosed 6 years prior, she had not received therapy because of the indolent course of the disease; she was mainly asymptomatic, except for mild neck adenopathy, which remained stable through the years. The diagnosis of coccidioidomycosis was revisited prior to chemotherapy. The coccidioidomycosis serology results were indeterminate and CT scan of the chest was negative for active coccidioidomycosis infection. Currently a reformed smoker, she had a history of 12 pack years of smoking.
On examination, there were multiple palpable lymph nodes in the posterior cervical chain bilaterally, left supraclavicular, pretrochlear areas, and bilateral axillary and inguinal areas. Chest and abdominal examinations were normal. There were no clinical symptoms or signs and no joint deformities to suggest an autoimmune disease such as RA, systemic lupus erythaematosus or Sjögren's syndrome.
Skin examination before systemic therapy showed a 20×20 cm geographic annular plaque on the anterior aspect of the left thigh, with a violaceous centre, and a firm, pink peripheral border. Several pink, non-contiguous 4–5 mm papules were also seen at the periphery of the primary lesion. Firm, faintly pink subcutaneous plaques were visible on the lower abdomen and the inner aspect of the left upper arm (satellite lesion) (figure 1A).
Figure 1.
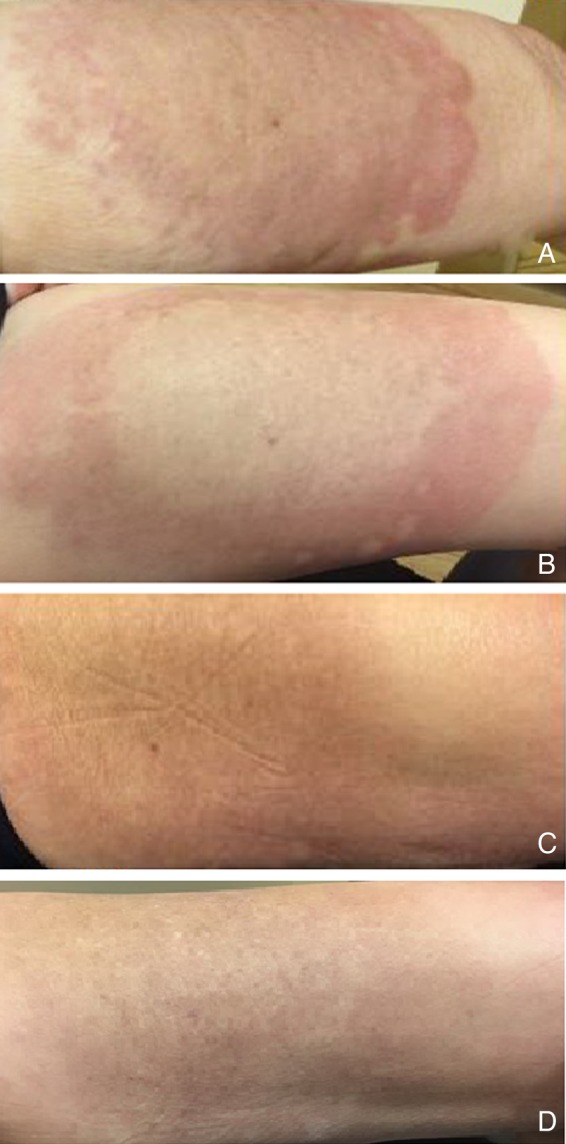
Interstitial granulomatous dermatitis rash: (A) erythaematous geographic annular plaque on the anterior aspect of the left thigh, with a violaceous centre and a firm, pink peripheral border, before starting chemotherapy. (B–D) Marked improvement in erythaematous plaque after 1st (16 July 2015), 2nd (13 August 2015) and 3rd (28 September 2015) cycles of chemotherapy, respectively.
Investigations
White cell count was 2400/µL, with absolute neutrophil count of 1330/µL and absolute lymphocyte count of 1270/µL. There was mild macrocytosis but there was no anaemia. Other blood cell counts, renal and liver function tests, serum alkaline phosphatase, lactate dehydrogenase and serum calcium were within normal range. β-2 microglobulin level was elevated at 4.41 mg/L (0.97–2.64 mg/L). Autoantibody screen (antinuclear antibodies, RA factor, anti-dsDNA antibody, anti-Ro (SSA 52) antibody, anti-Ro (SSA 60) antibody, anti-La (SSB) antibody and anti-SCL-70 antibody) was negative. CT scan of the chest, abdomen and pelvis showed bilateral extensive axillary, mediastinal, portahepatic, portacaval, retroperitoneal and common iliac lymphadenopathy. Most of the enlarged lymph nodes were in the range of 3–4 cm.
Skin biopsy from the left anterior thigh lesions showed findings consistent with IGD. Positive findings were infiltration of CD68 positive histiocytes around collagen bundles with a few neutrophils in the dermis. Predominant septal panniculitis was noted along with focal/patchy infiltration of CD5 and CD20 positive lymphocytic infiltrate, consistent with CLL, representing neoplastic seeding of the inflammatory infiltrate (figure 2). Cultures of the skin biopsy were negative for fungal and mycobacterial infection. Nasal swab was negative for Staphylococcus aureus. The bone marrow biopsy showed 70% cellularity with a diffuse interstitial small lymphocytic infiltrate with coarse chromatin, scant cytoplasm and inconspicuous nucleoli, comprising ∼90% of the marrow cellularity. As compared with previous bone marrow (2009), the cellularity had increased from 20% to 70% and the lymphoid infiltrate had increased from <5% to 60%. There was no fibrosis, no granulomata and no large cell component. The flow cytometry identified a dim κ restricted CD5+, subset CD23+ population of B cells consistent with SLL/CLL. Other markers on flow cytometry were dim (CD11c, CD19, CD20, CD22) and the following markers were negative (CD2, CD3, CD4, CD7, CD8, CD10, CD16, CD38, CD56, surface λ). Fluorescence in situ hybridisation showed trisomy of chromosome 12. Chromosome analysis by conventional cytogenetics showed normal female karyotyping (46, XX) in all 20 cells.
Figure 2.
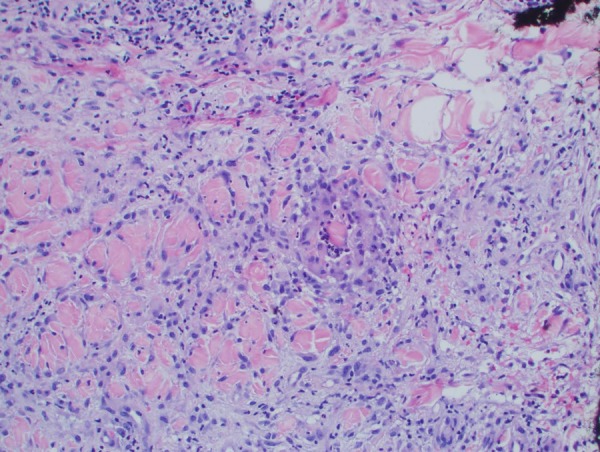
Interstitial granulomatous dermatitis. Photomicrograph showing CD68 positive histiocytes around collagen bundles with a few neutrophils in the dermis. There is predominant septal panniculitis along with a patchy infiltration of CD5 and CD20 positive lymphocytic infiltrate consistent with underlying chronic lymphocytic leukaemia.
Differential diagnosis
A number of differential diagnoses were considered including primary skin diseases, cutaneous infections, drug reaction and malignancy-associated rash. Infection was less likely as the rash did not respond to empirical antibiotics; nasal swab was negative for methicillin-resistant S. aureus and skin cultures were unremarkable. Drug reaction was excluded because the rash persisted despite stopping a suspected drug, namely, cetirizine. The eruption of dermatosis along with simultaneous worsening of symptoms associated with underlying haematological malignancy and improvement of rash after initiating the chemotherapy unveiled the possible association between the rash and CLL/SLL. The rash was confirmed as IGD on skin biopsy.
Treatment
There was no improvement in the rash after 4 weeks of administration of intralesional injectable triamcinolone and topical application of high-potency corticosteroids (clobetasol propionate). However, there was significant improvement in the severity of the rash after initiation of chemotherapy (combination therapy with bendamustine and rituximab), and within 4 weeks of systemic therapy, the rash started to clear from the central area and (figure 1B) completely resolved after the third cycle of chemotherapy. To date, our patient has completed the intended course of chemotherapy, and has shown complete resolution of the IGD and achieved complete remission of the underlying CLL for more than 6 months. The timeline for chemotherapy is outlined in table 2.
Table 2.
Timeline for chemotherapy
| Cycle of chemotherapy | Date |
|---|---|
| 1st | 07/16/2015 |
| 2nd | 08/13/2015 |
| 3rd | 09/28/2015 |
| 4th | 10/26/2015 |
| 5th | 11/23/2015 |
| 6th | 12/22/2015 |
| 7th | 01/18/2016 |
| 8th | 02/15/2016 |
Outcome and follow-up
The patient is actively under follow-up in oncology clinic. The post chemotherapy course has remained uncomplicated with complete resolution of the rash and complete remission of the underlying CLL/SLL.
Discussion
IGD is an uncommon skin disease known to be associated with a wide variety of rheumatological diseases and autoimmune conditions such as RA, Sjögren's syndrome (SS) and systemic lupus erythaematosus (SLE). It is also associated with drug reactions, solid organ cancers such as breast cancer, hypopharyngeal squamous cell carcinoma and lung cancer, and haematological malignancies such as myeloproliferative disorders and MDS.9–12 The eruption of IGD may be the only presenting manifestation of a haematological malignancy.7 8 13 The timeline of this association is not clearly established in haematological disorders; it may develop before or after the diagnosis of the primary malignancy. In our case, the IGD developed 6 years after initial diagnosis of CLL, and the patient was mainly asymptomatic; the rash roughly coincided with worsening generalised lymphadenopathy, and symptoms of fatigue and night sweating.
The clinical manifestations of IGD are variable. It may present as symmetrically distributed erythaematous polycyclic plaques, violaceous indurated linear cords (rope sign) or erythaematous papules and nodules localised to the axilla, trunk and inner thighs.1 The typical histology includes necrotising granulomatous inflammation with diffuse, palisading infiltration of the reticular dermis by histiocytes around degenerating collagen. The histiocytic infiltrate is usually accompanied by variable numbers of neutrophils and eosinophils.3 Although the pathophysiology is not well understood, the role of immune complex deposition in the reticular dermal vessels has been hypothesised; this can activate the complement system, leading to neutrophil infiltration resulting in damage to dermal collagen fibres.4
The typical autoimmune manifestations of CLL are limited to the haematopoietic system and include autoimmune thrombocytopaenia, Evans syndrome and pure red cell aplasia. The uncommon non-haematological associations of CLL include RA, SS, SLE, ulcerative colitis, pernicious anaemia, pemphigus, nephrotic syndrome, myopathy and neuropathy.14 15 IGD is not known to be associated with autoimmune dermatological manifestations. The IGD likely occurred in this patient secondary to SLL/CLL, as evidenced by the appearance of a rash around the time the CLL worsened, and the appearance of symptoms and later resolution of the rash in response to systemic treatment of the CLL. Association of an IGD rash in the setting of untreated SLL/CLL, to the best of our knowledge, has never been described in the literature. Although this first case report does not unequivocally establish the association between the IGD and CLL, and must be confirmed by subsequent reports, there are several features reported here that reasonably point towards this association making any alternative explanations less likely. This case demonstrates that physicians should consider the possibility of underlying SLL/CLL along with other haematological malignancies in patients with IGD in the absence of a usual cause. IGD cannot be diagnosed with clinical examination alone and requires biopsy for histopathological confirmation. Moreover, it should be noted that IGD might appear many years before or after the diagnosis of an underlying malignancy.
Learning points.
Interstitial granulomatous dermatitis (IGD) is a rare skin disease commonly associated with connective tissue disorders, drugs, non-haematological and haematological malignancies (ie, myelodysplastic syndrome and acute promyelocytic leukaemia.
This case is the first reported association of IGD with chronic lymphocytic leukaemia (CLL). It should be noted that IGD can appear many years after the diagnosis of CLL/small lymphocytic lymphoma and may hint towards disease progression.
Patients with non-responsive skin rash should be screened for an underlying malignancy, and progression/worsening of the underlying malignancy may be considered in patients with known diagnosis of malignancy.
Footnotes
Contributors: IBR was involved in the conception and design, drafting of manuscript and final approval of manuscript. MUK was involved in the conception and design, drafting of manuscript. RJS was involved in drafting of manuscript. FA was involved in the conception and design, final approval of manuscript.
Competing interests: None declared.
Patient consent: Obtained.
Provenance and peer review: Not commissioned; externally peer reviewed.
References
- 1.Chu P, Connolly MK, LeBoit PE. The histopathologic spectrum of palisaded neutrophilic and granulomatous dermatitis in patients with collagen vascular disease. Arch Dermatol 1994;130:1278–83. 10.1001/archderm.1994.01690100062010 [DOI] [PubMed] [Google Scholar]
- 2.Ackerman AB. Clues to diagnosis in dermatopathology. Am J Dermatopathol 1992;14:74. [Google Scholar]
- 3.Sangueza OP, Caudell MD, Mengesha YM et al. Palisaded neutrophilic granulomatous dermatitis in rheumatoid arthritis. J Am Acad Dermatol 2002;47:251–7. 10.1067/mjd.2002.124620 [DOI] [PubMed] [Google Scholar]
- 4.Verneuil L, Dompmartin A, Comoz F et al. Interstitial granulomatous dermatitis with cutaneous cords and arthritis: a disorder associated with autoantibodies. J Am Acad Dermatol 2001;45:286–91. 10.1067/mjd.2001.114577 [DOI] [PubMed] [Google Scholar]
- 5.Peroni A, Colato C, Schena D et al. Interstitial granulomatous dermatitis: a distinct entity with characteristic histological and clinical pattern. Br J Dermatol 2012;166:775–83. 10.1111/j.1365-2133.2011.10727.x [DOI] [PubMed] [Google Scholar]
- 6.Mason HR, Swanson JK, Ho J et al. Interstitial granulomatous dermatitis associated with darifenacin. J Drugs Dermatol 2008;7:895–7. [PubMed] [Google Scholar]
- 7.Swing DC Jr, Sheehan DJ, Sangueza OP et al. Interstitial granulomatous dermatitis secondary to acute promyelocytic leukemia. Am J Dermatopathol 2008;30:197–9. 10.1097/DAD.0b013e318162af78 [DOI] [PubMed] [Google Scholar]
- 8.Patsinakidis N, Susok L, Hessam S et al. Interstitial granulomatous dermatitis associated with myelodysplastic syndrome—complete clearance under therapy with 5-azacytidine. Acta Derm Venereol 2014;94:725–6. 10.2340/00015555-1827 [DOI] [PubMed] [Google Scholar]
- 9.Deng A, Harvey V, Sina B et al. Interstitial granulomatous dermatitis associated with the use of tumor necrosis factor alpha inhibitors. Arch Dermatol 2006;142:198–202. 10.1001/archderm.142.2.198 [DOI] [PubMed] [Google Scholar]
- 10.Perrin C, Lacour JP, Castanet J et al. Interstitial granulomatous drug reaction with a histological pattern of interstitial granulomatous dermatitis. Am J Dermatopathol 2001;23:295–8. 10.1097/00000372-200108000-00003 [DOI] [PubMed] [Google Scholar]
- 11.Schreckenberg C, Asch PH, Sibilia J et al. [Interstitial granulomatous dermatitis and paraneoplastic rheumatoid polyarthritis disclosing cancer of the lung]. Ann Dermatol Venereol 1998; 125:585–8. [PubMed] [Google Scholar]
- 12.Tomasini C, Pippione M. Interstitial granulomatous dermatitis with plaques. J Am Acad Dermatol 2002;46:892–9. 10.1067/mjd.2002.120532 [DOI] [PubMed] [Google Scholar]
- 13.Balin SJ, Wetter DA, Kurtin PJ et al. Myelodysplastic syndrome presenting as generalized granulomatous dermatitis. Arch Dermatol 2011;147:331–5. 10.1001/archdermatol.2011.39 [DOI] [PubMed] [Google Scholar]
- 14.Barcellini W, Capalbo S, Agostinelli RM et al. Relationship between autoimmune phenomena and disease stage and therapy in B-cell chronic lymphocytic leukemia. Haematologica 2006;91:1689–92. [PubMed] [Google Scholar]
- 15.Hassan S, Popalzai M, Yu E et al. A 61-year-old man presented with myopathy, neuropathy, and inflammatory dermatitis responsive to chronic lymphocytic leukemia treatment. Int J Gen Med 2012;5:647–53. 10.2147/IJGM.S32993 [DOI] [PMC free article] [PubMed] [Google Scholar]


