Abstract
Background
Placental growth factor (PlGF) is a member of the vascular endothelial growth factor family that acts as a pleiotropic cytokine capable of stimulating angiogenesis and accelerating atherogenesis. Soluble fms-like tyrosine kinase-1 (sFlt-1) antagonizes PlGF action. Higher levels of PlGF and sFlt-1 have been associated with cardiovascular events in patients with chronic kidney disease, yet little is known about their relationship with adverse outcomes in patients on peritoneal dialysis (PD). The aim of this study was to investigate the association of PlGF and sFlt-1 with technique survival and cardiovascular events.
Methods
We measured serum levels of PlGF and plasma levels of sFlt-1 in 40 PD patients at Nara Medical University.
Results
PlGF and sFlt-1 levels were significantly correlated with the dialysate-to-plasma ratio of creatinine (r = 0.342, p = 0.04 and r = 0.554, p < 0.001) although PlGF and sFlt-1 levels were not correlated with total creatinine clearance and total Kt/V. Additionally, both PlGF and sFlt-1 levels were significantly higher in patients with high transport membranes compared to those without (p = 0.039 and p < 0.001, respectively). Patients with PlGF levels above the median had lower technique survival and higher incidence of cardiovascular events than patients with levels below the median, with hazard ratios of 11.9 and 7.7, respectively, in univariate Cox regression analysis. However, sFlt-1 levels were not associated with technique survival or cardiovascular events (p = 0.11 and p = 0.10, respectively).
Conclusion
Elevated PlGF and sFlt-1 are significantly associated with high transport membrane status. PlGF may be a useful predictor of technique survival and cardiovascular events in PD patients.
Key Words: Peritoneal dialysis, Technique survival, Cardiovascular events, Placental growth factor, Soluble fms-like tyrosine kinase-1
Introduction
Vascular endothelial growth factor (VEGF) and placental growth factor (PlGF) are pleiotropic cytokines that stimulate intramural angiogenesis and accelerated atherogenesis by recruiting and activating macrophages via binding to VEGF receptor-1, also known as fms-like tyrosine kinase-1 (Flt-1) [1,2,3]. A circulating soluble isoform of Flt-1 (sFlt-1), produced by a splice variant of full-length Flt-1 mRNA, acts as an endogenous inhibitor of PlGF and VEGF [4]. Studies of molecular mechanisms underlying angiogenesis and atherogenesis have recently focused on the balance or imbalance between such angiogenic factors and their soluble receptors. Using a heparin loading test, we found that the post-heparin level of sFlt-1 estimates the total concentration of sFlt-1 in the body and is negatively associated with atherosclerosis in patients with advanced chronic kidney disease (CKD), while an elevated pre-heparin (i.e. baseline) level of sFlt-1 may be associated with vascular endothelial dysfunction [5,6].
Several clinical studies have shown that baseline levels of PlGF and sFlt-1 are useful predictors of mortality and cardiovascular events in patients with acute coronary syndrome and chronic heart failure [7,8,9]. Elevated levels of sFlt-1 and VEGF are independently associated with all-cause mortality in patients on dialysis [10,11]. In addition, our recent studies have shown that PlGF is strongly associated with mortality and cardiovascular events among patients with CKD, including dialysis patients [5,12].
However, the association between angiogenic factors and hard clinical end points such as technique survival and cardiovascular events in patients on peritoneal dialysis (PD) is less clear. Our aim, therefore, was to investigate the association of PlGF and sFlt-1 levels with PD technique survival and the incidence of cardiovascular events in PD patients.
Patients and Methods
Patients
In this study, 40 consecutive patients on PD for >3 months were analyzed at Nara Medical University between January 1, 2010 and December 31, 2011. All participants had regular follow-up visits at Nara Medical University approximately once per month. Clinical data including age, gender, hypertension, diabetes, dyslipidemia, smoking, laboratory parameters, dialysis-related parameters, and current medications were assessed through patient interviews and medical records. Hypertension was defined as systolic blood pressure ≥140 mm Hg, systolic blood pressure ≥90 mm Hg, or use of oral antihypertensive medications. Diabetes was defined as fasting glucose ≥126 mg/dl or use of oral hypoglycemic agents. Dyslipidemia was defined as low-density lipoprotein cholesterol ≥140 mg/dl or use of lipid-lowering medications.
Blood Samples and Peritoneal Equilibration Test
Plasma and serum blood samples collected from all patients were immediately centrifuged and kept frozen at −80°C until assayed. Plasma levels of sFlt-1 and serum levels of PlGF and VEGF-A were measured using commercial sandwich enzyme-linked immunosorbent assay kits (R&D Systems, Minneapolis, Minn., USA).
A peritoneal equilibration test (PET) was performed within 3 months of collecting blood samples using standard protocols [13,14]. The dialysate-to-plasma ratio of creatinine (D/P creatinine) was evaluated at 4 h and patients were classified as low transporters (0.34-0.5), low to average transporters (0.6-0.65), high to average transporters (0.66-0.81), or high transporters (0.82-1.03). Peritoneal urea clearance (Kt/V) and creatinine clearance (CCr) was estimated from a 24-hour collection of dialysate urea and creatinine, and residual renal Kt/V and CCr was estimated from a 24-hour collection of urine urea and creatinine. Total weekly Kt/V was calculated as the sum of renal and peritoneal Kt/V. Total weekly CCr was calculated as the sum of renal and peritoneal CCr and was normalized by 1.73 m2 of body surface area. Of the 40 enrolled participants, complete PET data were available for 34.
In echocardiographic findings, left ventricular mass index (LVMI) was calculated using the Devereux equation: LVMI = {0.8 × [1.04 × (left ventricular end-diastolic diameter + interventricular septum thickness + posterior wall thickness)3 - (left ventricular end-diastolic diameter)3] + 0.6}/body surface area.
Study End Points
Cumulative PD technique survival and the incidence of cardiovascular events were determined from the day of examination with a median (interquartile range) follow-up period of 18 (4-25) months. PD technique failure was defined as discontinuation of PD because of uncontrolled volume overload with 2.5% dextrose solution or icodextrin solution, progressive uremic anemia resistant to erythropoiesis-stimulating agents, or refractory peritonitis despite antibiotics according to the International Society for Peritoneal Dialysis (ISPD) guidelines [15]. Cardiovascular events were defined as a composite of the following individual events during the study period: new-onset fatal or non-fatal coronary artery disease, sudden cardiac death, congestive heart failure, and cerebrovascular disease.
Statistical Analyses
All variables were expressed as means ± standard deviation or medians with interquartile range, as appropriate. Spearman's rank correlation coefficients were used to assess the correlation between biomarkers and dialysis adequacy. Cumulative PD technique survival and the incidence of cardiovascular events were estimated using the Kaplan-Meier method, with the median value as the cutoff. Differences were assessed using the log-rank test. Univariate Cox regression models were used to determine the association between various biomarkers and time to PD technique failure and the incidence of cardiovascular events. A two-sided p value <0.05 was considered statistically significant. JMP 10.0.02 (SAS, Cary, N.C., USA) was used to perform all statistical analyses.
Results
Baseline Demographics
A total of 40 PD patients, consisting of 25 males and 15 females, with a median age (interquartile range) of 60 (56-72) years, were analyzed in the present study. Table 1 shows the baseline demographics of all participants. Eleven patients (28%) were on automated PD and the rest were on continuous ambulatory PD. Most were taking antihypertensive medications (calcium channel blockers n = 28, renin-angiotensin system inhibitors n = 30) and vitamin D (n = 17). Sixteen patients (40%) were treated with icodextrin solution. The underlying kidney disease was suspected to be diabetic nephropathy in 43%, benign nephrosclerosis in 20%, chronic glomerulonephritis in 20%, and other in 17%.
Table 1.
Baseline demographics (patients, n = 40)
| Clinical data | |
| Age, years | 60 (56–72) |
| Male gender | 25 (63%) |
| Dialysis vintage, months | 25 (9–57) |
| Urine volume, ml/day | 750 (23–1,000) |
| Ultrafiltration, ml/day | 300 (25–838) |
| Use of icodextrin | 16 (40%) |
| Body mass index | 23.0 (21.1–24.9) |
| Diabetes | 18 (45%) |
| Hypertension | 36 (90%) |
| Dyslipidemia | 12 (30%) |
| Smoking | 19 (48%) |
| Laboratory data | |
| Hemoglobin, g/dl | 10.7 (9.6–11.8) |
| Albumin, g/dl | 3.4 (2.8–3.7) |
| Urea nitrogen, mg/dl | 53 (42–64) |
| Creatinine, mg/dl | 8.01 (6.02–11.4) |
| Calcium, mg/dl | 9.5 (9.1–9.8) |
| Phosphorus, mg/dl | 4.9 (4.0–5.8) |
| Intact PTH, pg/ml | 115 (66–198) |
| HbA1c, % | 5.4 (5.1–5.9) |
| LDL cholesterol, mg/dl | 115 (88–141) |
| CRP, mg/dl | 0.1 (0.03–0.48) |
| BNP, pg/ml | 166 (50–421) |
| Cystatin C, mg/l | 6.06 (5.08–7.27) |
| β2-Microglobulin, mg/l | 18.2 (15.2–28.6) |
| VEGF, pg/ml | 270 (161–489) |
| PlGF, pg/ml | 15.5 (13.6–21.6) |
| sFlt-1, pg/ml | 122 (95–163) |
BNP = Brain natriuretic peptide; CRP = C-reactive protein; HbA1c = glycated hemoglobin; LDL = low-density lipoprotein; PTH = parathyroid hormone.
Both levels of cystatin C and β2-microglobulin were significantly correlated with dialysis vintage (r = 0.635, p < 0.001 and r = 0.576, p < 0.001, respectively), urine volume (r = −0.473, p = 0.002 and r = −0.686, p < 0.001, respectively), and ultrafiltration (r = −0.496, p = 0.001 and r = −0.736, p < 0.001, respectively). Serum levels of PlGF were directly correlated with brain natriuretic peptide (r = 0.366, p = 0.02) and low-density lipoprotein cholesterol (r = 0.319, p = 0.04), and plasma levels of sFlt-1 was inversely correlated with albumin (r = −0.358, p = 0.02). However, we did not find any correlation between VEGF and clinical factors.
Using PET data, serum levels of cystatin C and β2-microglobulin were significantly correlated with residual renal Kt/V urea (r = −0.607, p < 0.001 and r = −0.739, p < 0.001, respectively) and residual renal CCr (r = −0.670, p < 0.001 and r = −0.773, p < 0.001, respectively), but no significant relationship between angiogenic factors and residual renal Kt/V urea and CCr was observed (table 2). PlGF and sFlt-1 levels were directly correlated with D/P creatinine (r = 0.342, p = 0.04 and r = 0.554, p < 0.001, respectively), but VEGF levels were not associated with dialysis adequacy. In addition, both PlGF and sFlt-1 levels in patients with high transport status were significantly higher compared with those in patients without high transport status (p = 0.021 and p < 0.001, respectively).
Table 2.
Correlation between dialysis adequacy and biomarkers
| Variables | Correlation (r) |
||||
|---|---|---|---|---|---|
| PlGF | sFlt-l | VEGF | β2-microglobulin | cystatin C | |
| Urine volume | 0.114 | −0.197 | 0.199 | −0.686*** | −0.473** |
| Total Kt/V | −0.108 | −0.290 | 0.042 | −0.363* | −0.406* |
| Residual renal Kt/V | −0.076 | −0.238 | 0.224 | −0.739*** | −0.607*** |
| Peritoneal Kt/V | −0.040 | −0.068 | −0.230 | 0.472** | 0.250 |
| Total CCr | −0.082 | −0.269 | −0.222 | −0.673*** | −0.640*** |
| Residual renal CCr | −0.130 | −0.322 | 0.196 | −0.759*** | −0.670*** |
| Peritoneal CCr | 0.198 | 0.309 | −0.027 | 0.622*** | 0.423* |
| D/P creatinine | 0.342* | 0.554*** | 0.294 | −0.117 | −0.027 |
p < 0.05;
p < 0.01;
p < 0.001.
Echocardiography was performed within 2 weeks of blood collection in 36 patients. LVMI was significantly correlated with PlGF (r = 0.418, p = 0.02), but not with sFlt-1 and VEGF levels (r = 0.06, p = 0.91 and r = 0.01, p = 0.93, respectively), although ejection fraction was not correlated with all biomarkers.
Clinical Outcomes
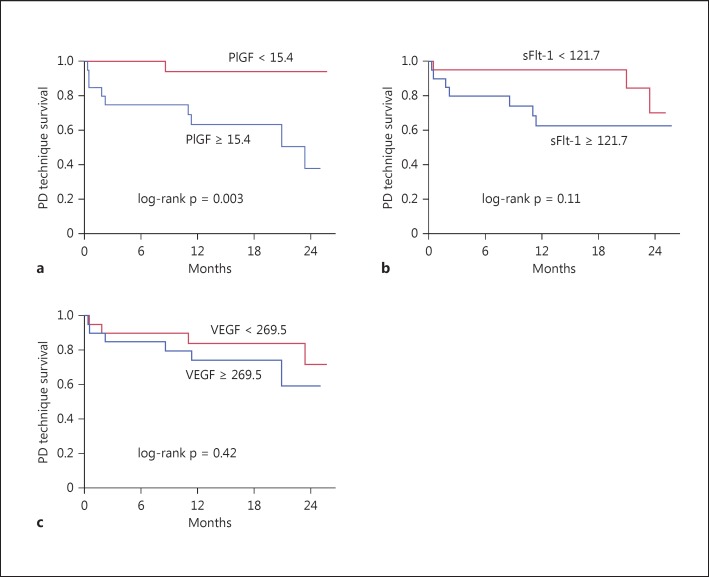
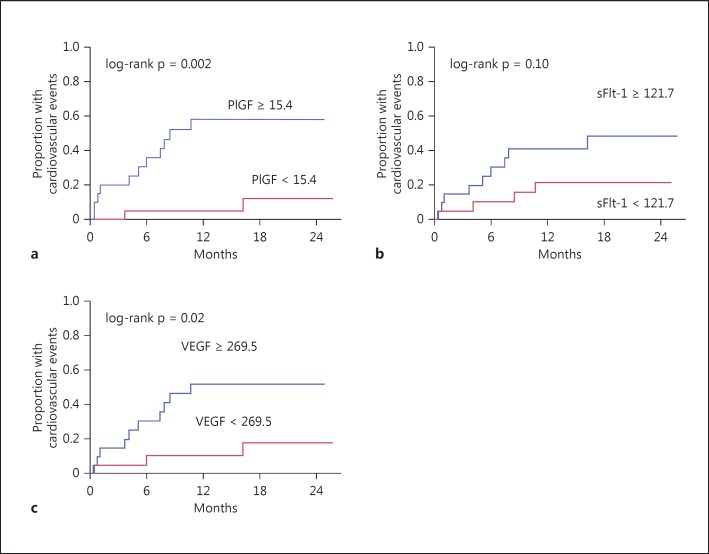
During the study period, 10 patients switched to hemodialysis (HD) and 13 patients experienced a new cardiovascular event, mostly congestive heart failure. Unadjusted Kaplan-Meier analysis showed that patients with above median levels of PlGF had significantly worse PD technique survival and a higher incidence of cardiovascular events than those with below median levels (p = 0.003 and p = 0.002, respectively, by log-rank test for trend) (fig. 1a, 2a). However, we did not find any association between sFlt-1 and PD technique survival or cardiovascular events (p = 0.11 and p = 0.10, respectively) (fig. 1b, 2b). VEGF was significantly associated with cardiovascular events but not PD technique survival (fig. 1c, 2c).
Fig. 1.
Kaplan-Meier analysis of PD technique survival using the median levels of PlGF (a), sFlt-1 (b) and VEGF (c) as the cutoff.
Fig. 2.
Kaplan-Meier analysis of cardiovascular events using the median levels of PlGF (a), sFlt-1 (b) and VEGF (c) as the cutoff.
As shown in table 3, PlGF but not sFlt-1 was significantly associated with both PD technique survival and cardiovascular events in univariate Cox proportional hazards models (hazard ratio [95% confidence interval] 11.9 [2.2-219.4] and 7.7 [2.1-49.9], respectively).
Table 3.
Univariate Cox regression analysis for PD technique failure and cardiovascular events
| Variables | Technique survival |
Cardiovascular events |
||
|---|---|---|---|---|
| HR (95% CI) | p | HR (95% CI) | p | |
| PlGF (≥15.4 pg/ml) | 11.9 (2.2–219.4) | 0.002 | 7.70 (2.1–49.9) | 0.002 |
| sFlt-1 (≥121.7 pg/ml) | 2.81 (0.77–13.1) | 0.12 | 2.66 (0.86–9.85) | 0.09 |
| VEGF (≥269.5 pg/ml) | 1.68 (0.48–6.64) | 0.42 | 4.05 (1.24–18.1) | 0.02 |
| Age (10-year difference) | 0.94 (0.51–1.65) | 0.82 | 1.04 (0.62–1.69) | 0.87 |
| Male gender | 2.69 (0.67–17.8) | 0.17 | 1.49 (0.48–5.52) | 0.50 |
| Dialysis vintage (>25 months) | 1.60 (0.46–6.27) | 0.17 | 1.22 (0.39–3.70) | 0.72 |
| Urine volume (within 750 ml/day) | 2.23 (0.61–10.4) | 0.23 | 1.65 (0.55–5.48) | 0.37 |
| Use of icodextrin | 0.82 (0.21–2.89) | 0.76 | 1.57 (0.52–4.87) | 0.42 |
| Diabetes | 0.57 (0.12–2.11) | 0.41 | 1.22 (0.41–3.81) | 0.72 |
| Hypertension | 1.31 (0.24–24.5) | 0.80 | 0.63 (0.17–4.09) | 0.57 |
| Dyslipidemia | 0.82 (0.18–2.95) | 0.77 | 0.62 (0.14–2.04) | 0.45 |
| Smoking | 1.96 (0.55–7.75) | 0.30 | 0.65 (0.20–1.94) | 0.44 |
| Cystatin C (≥6.06 mg/l) | 2.50 (0.69–11.6) | 0.17 | 1.21 (0.40–3.70) | 0.73 |
| β2-Microglobulin (≥18.1 mg/l) | 1.49 (0.42–5.84) | 0.54 | 0.87 (0.28–2.62) | 0.80 |
| BNP (≥166.2 pg/ml) | 4.22 (1.06–28.0) | 0.04 | 6.78 (1.82–43.8) | 0.003 |
BNP = Brain natriuretic peptide; CI = confidence interval; HR = hazard ratio.
Discussion
We first investigated the association of angiogenic factors with technique survival and cardiovascular events in PD patients. We found that PlGF but not sFlt-1 was significantly associated with PD technique survival and cardiovascular events in unadjusted models, which suggests that PlGF might be a more useful biomarker than sFlt-1 in PD patients. In addition, both PlGF and sFlt-1 levels were directly correlated with D/P creatinine, whereas VEGF levels did not have any correlation with dialysis adequacy.
PlGF, which has sequence homology with VEGF, plays a critical role in the pathophysiology of angiogenesis via the promotion of new tube formation by endothelial cells and destabilization of plaques through mediating macrophage accumulation and activation, thus promoting atherogenesis [16,17]. These biological actions of PlGF are mediated through its specific receptor, Flt-1, although VEGF has several receptors, including Flt-1. sFlt-1, produced by alternative splicing of full-length Flt-1, lacks the transmembrane and cytoplasmic domains. It binds PlGF and VEGF and acts as a decoy receptor, inhibiting the effects of both PlGF and VEGF. PlGF and sFlt-1 have emerged as practical clinical predictors of mortality and cardiovascular disease in patients with chronic heart failure, ischemic heart disease, diabetes, and CKD [5,12,18,19,20,21]. In addition, as shown by Guo et al. [10], sFlt-1 is independently associated with all-cause mortality in HD patients; however, the association between PlGF and adverse outcomes has not yet been investigated in PD patients.
Of note, both PlGF and sFlt-1 levels were directly correlated with D/P creatinine, based on PET data within 3 months of blood sample collection. Patients with high transport membranes had significantly higher levels of PlGF and sFlt-1 compared to those without. Taking these findings into account, PlGF and sFlt-1 might play important biological roles in regulating the peritoneal vasculature in PD patients, and an increase in circulating levels could serve as an important indicator of peritoneal deterioration. Zhang et al. [22] have recently reported that experimentally Flt-1 is generally expressed in peritoneal membranes in PD patients, which supports this hypothesis.
We found that PD patients with higher levels of PlGF have a greater risk of technique failure and cardiovascular diseases than those with lower levels of PlGF. PD patients have increased LVMI and cardiovascular risk if not adequately monitored [23] and in the study, the most common reason for switching to HD and incident cardiovascular events was congestive heart failure with poor fluid management. We and others have recently shown that PlGF is a strong predictor of elevated LVMI and a greater risk of heart failure requiring hospitalization in CKD patients [12,24]. PlGF transgenic mice exhibited a greater cardiac hypertrophic response, a greater increase in capillary density, and increased fibroblast content in the heart in response to pressure overload stimulation [25]. In addition, higher PlGF levels in our patients were significantly correlated with high peritoneal membrane transport rates and impaired net ultrafiltration. These findings suggest that PlGF may be involved in the development of not only atherosclerotic disease but also heart failure in PD patients. On the other hand, in spite of a closer relation between sFlt-1 and peritoneal function, elevated sFlt-1 levels were not associated with technique failure and cardiovascular events. Unlike PlGF, sFlt-1 levels were not correlated with brain natriuretic peptide levels and LVMI, which suggests that sFlt-1 was a surrogate marker of peritoneal angiogenesis rather than of cardiac hypertrophy and failure due to volume overload in PD patients. However, further research is needed to understand the association of PlGF and sFlt-1 with heart failure.
By contrast, increased expression of another powerful ligand for Flt-1, VEGF-A, in cultured human peritoneal mesothelial cells is responsible for the development and progression of peritoneal membrane hyperpermeability and fibrosis [26,27]. Additionally, dialysate VEGF predicts future withdrawal from PD in patients receiving PD [28]. In the study, however, VEGF-A was not significantly associated with PD technique survival and peritoneal function, and sFlt-1 antagonizes not only PlGF but also VEGF, possibly resulting in the prognostic differences between PlGF and sFlt-1 in PD patients.
The current study has some limitations. First, it was limited by a relatively small number of patients and events, and thus we cannot identify independent risk factors of technique failure and cardiovascular events. Second, we did not analyze the changes in circulating PlGF and sFlt-1 levels or measure dialysate PlGF and sFlt-1 levels.
In conclusion, both PlGF and sFlt-1 serve as surrogate markers of peritoneal deterioration. PlGF but not sFlt-1 is significantly associated with not only technique survival but also cardiovascular events in PD patients, suggesting that PlGF may be a promising biomarker in the clinical management of PD. However, larger studies in the future will be helpful for validating our results.
Statement of Ethics
This clinical study was approved by the ethics board of Nara Medical University and written informed consent was obtained from each study participant (No. 2002-009).
Disclosure Statement
Dr. Y. Saito received lecture fees from Merck, Takeda Pharmaceutical Company, Novartis Pharma KK, Daiichi Sankyo Company, Mitsubishi Tanabe Pharma Corp., Pfizer Japan, Otsuka Pharmaceutical and research funding from the Japan Heart Foundation and the Naito Foundation. He belongs to the Department of Regulatory Medicine of Blood Pressure sponsored by Merck. The other authors have no financial conflicts of interest to disclose.
Acknowledgements
We acknowledge S. Yoshimura and A. Okuda for assaying PlGF levels. This work was supported in part by Grants-in-Aid from the Ministry of Health, Labor and Welfare of Japan, and the Takeda Science Foundation.
References
- 1.Park JE, Chen HH, Winder J, Houck KA, Ferrara N. Placenta growth factor. Potentiation of vascular endothelial growth factor bioactivity, in vitro and in vivo, and high affinity binding to Flt-1 but not to Flk-1/KDR. J Biol Chem. 1994;269:25646–25654. [PubMed] [Google Scholar]
- 2.Luttun A, Tjwa M, Moons L, Wu Y, Angelillo-Scherrer A, Liao F, Nagy JA, Hooper A, Priller J, De Klerck B, Compernolle V, Daci E, Bohlen P, Dewerchin M, Herbert JM, Fava R, Matthys P, Carmeliet G, Collen D, Dvorak HF, Hicklin DJ, Carmeliet P. Revascularization of ischemic tissues by PlGF treatment, and inhibition of tumor angiogenesis, arthritis and atherosclerosis by anti-Flt1. Nat Med. 2002;8:831–840. doi: 10.1038/nm731. [DOI] [PubMed] [Google Scholar]
- 3.Khurana R, Moons L, Shafi S, Luttun A, Collen D, Martin JF, Carmeliet P, Zachary IC. Placental growth factor promotes atherosclerotic intimal thickening and macrophage accumulation. Circulation. 2005;111:2828–2836. doi: 10.1161/CIRCULATIONAHA.104.495887. [DOI] [PubMed] [Google Scholar]
- 4.Kendall RL, Thomas KA. Inhibition of vascular endothelial cell growth factor activity by an endogenously encoded soluble receptor. Proc Natl Acad Sci USA. 1993;90:10705–10709. doi: 10.1073/pnas.90.22.10705. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Matsui M, Takeda Y, Uemura S, Matsumoto T, Seno A, Onoue K, Tsushima H, Morimoto K, Soeda T, Okayama S, Somekawa S, Samejima K, Kawata H, Kawakami R, Nakatani K, Iwano M, Saito Y. Suppressed soluble Fms-like tyrosine kinase-1 production aggravates atherosclerosis in chronic kidney disease. Kidney Int. 2014;85:393–403. doi: 10.1038/ki.2013.339. [DOI] [PubMed] [Google Scholar]
- 6.Di Marco GS, Reuter S, Hillebrand U, Amler S, König M, Larger E, Oberleithner H, Brand E, Pavenstädt H, Brand M. The soluble VEGF receptor sFlt1 contributes to endothelial dysfunction in CKD. J Am Soc Nephrol. 2009;20:2235–2245. doi: 10.1681/ASN.2009010061. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Ky B, French B, Ruparel K, Sweitzer NK, Fang JC, Levy WC, Sawyer DB, Cappola TP. The vascular marker soluble fms-like tyrosine kinase 1 is associated with disease severity and adverse outcomes in chronic heart failure. J Am Coll Cardiol. 2011;19:386–394. doi: 10.1016/j.jacc.2011.03.032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Glaser R, Peacock WF, Wu AH, Muller R, Möckel M, Apple FS. Placental growth factor and B-type natriuretic peptide as independent predictors of risk from a multibiomarker panel in suspected acute coronary syndrome (Acute Risk and Related Outcomes Assessed With Cardiac Biomarkers [ARROW]) study. Am J Cardiol. 2011;107:821–826. doi: 10.1016/j.amjcard.2010.11.003. [DOI] [PubMed] [Google Scholar]
- 9.Onoue K, Uemura S, Takeda Y, Somekawa S, Iwama H, Nishida T, Morikawa Y, Nakagawa H, Tsutsumi T, Sung JH, Takemoto Y, Soeda T, Okayama S, Ishigami K, Kawata H, Horii M, Nakajima T, Saito Y. Usefulness of soluble fms-like tyrosine kinase-1 as a biomarker of acute severe heart failure in patients with acute myocardial infarction. Am J Cardiol. 2009;104:1478–1483. doi: 10.1016/j.amjcard.2009.07.016. [DOI] [PubMed] [Google Scholar]
- 10.Guo Q, Carrero JJ, Yu X, Bárány P, Qureshi AR, Eriksson M, Anderstam B, Chmielewski M, Heimbürger O, Stenvinkel P, Lindholm B, Axelsson J. Associations of VEGF and its receptors sVEGFR-1 and −2 with cardiovascular disease and survival in prevalent haemodialysis patients. Nephrol Dial Transplant. 2009;24:3468–3473. doi: 10.1093/ndt/gfp315. [DOI] [PubMed] [Google Scholar]
- 11.Yuan J, Guo Q, Qureshi AR, Anderstam B, Eriksson M, Heimbürger O, Bárány P, Stenvinkel P, Lindholm B. Circulating vascular endothelial growth factor (VEGF) and its soluble receptor 1 (sVEGFR-1) are associated with inflammation and mortality in incident dialysis patients. Nephrol Dial Transplant. 2013;28:2356–2363. doi: 10.1093/ndt/gft256. [DOI] [PubMed] [Google Scholar]
- 12.Matsui M, Uemura S, Takeda Y, Samejima K, Matsumoto T, Hasegawa A, Tsushima H, Hoshino E, Ueda T, Morimoto K, Okamoto K, Okada S, Onoue K, Okayama S, Kawata H, Kawakami R, Maruyama N, Akai Y, Iwano M, Shiiki H, Saito Y, NARA-CKD Investigators Placental growth factor as a predictor of cardiovascular events in patients with CKD from the NARA-CKD study. J Am Soc Nephrol. 2015;26:2871–2881. doi: 10.1681/ASN.2014080772. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Twardowski ZJ. Clinical value of standardized equilibration tests in CAPD patients. Blood Purif. 1989;7:95–108. doi: 10.1159/000169582. [DOI] [PubMed] [Google Scholar]
- 14.Rebić DR, Rašić SR, Dervišević MD, Hamzić-Mehmedbašić AH, Muslimović AM, Hasanagić EH. Alteration of cardiovascular structure and function in patients undergoing peritoneal dialysis. Cardiorenal Med. 2015;5:135–144. doi: 10.1159/000380859. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Li PK, Szeto CC, Piraino B, Bernardini J, Figueiredo AE, Gupta A, Johnson DW, Kuijper EJ, Lye WC, Salzer W, Schaefer F, Struijk DG. Peritoneal dialysis-related infections recommendations: 2010 update. Perit Dial Int. 2010;30:393–423. doi: 10.3747/pdi.2010.00049. [DOI] [PubMed] [Google Scholar]
- 16.Pilarczyk K, Sattler KJ, Galili O, Versari D, Olson ML, Meyer FB, Zhu XY, Lerman LO, Lerman A. Placenta growth factor expression in human atherosclerotic carotid plaques is related to plaque destabilization. Atherosclerosis. 2008;196:333–340. doi: 10.1016/j.atherosclerosis.2006.10.038. [DOI] [PubMed] [Google Scholar]
- 17.Carmeliet P, Moons L, Luttun A, Vincenti V, Compernolle V, De Mol M, Wu Y, Bono F, Devy L, Beck H, Scholz D, Acker T, DiPalma T, Dewerchin M, Noel A, Stalmans I, Barra A, Blacher S, Vandendriessche T, Ponten A, Eriksson U, Plate KH, Foidart JM, Schaper W, Charnock-Jones DS, Hicklin DJ, Herbert JM, Collen D, Persico MG. Synergism between vascular endothelial growth factor and placental growth factor contributes to angiogenesis and plasma extravasation in pathological conditions. Nat Med. 2001;7:575–583. doi: 10.1038/87904. [DOI] [PubMed] [Google Scholar]
- 18.Matsumoto T, Uemura S, Takeda Y, Matsui M, Okada S, Nishida T, Soeda T, Okayama S, Somekawa S, Ishigami K, Onoue K, Kawata H, Kawakami R, Horii M, Saito Y. An elevated ratio of placental growth factor to soluble fms-like tyrosine kinase-1 predicts adverse outcomes in patients with stable coronary artery disease. Intern Med. 2013;52:1019–1027. doi: 10.2169/internalmedicine.52.9073. [DOI] [PubMed] [Google Scholar]
- 19.Lenderink T, Heeschen C, Fichtlscherer S, Dimmeler S, Hamm CW, Zeiher AM, Simoons ML, Boersma E, CAPTURE Investigators Elevated placental growth factor levels are associated with adverse outcomes at four-year follow-up in patients with acute coronary syndromes. J Am Coll Cardiol. 2006;47:307–311. doi: 10.1016/j.jacc.2005.08.063. [DOI] [PubMed] [Google Scholar]
- 20.Theilade S, Lajer M, Jorsal A, Tarnow L, Parving HH, Rossing P. Evaluation of placental growth factor and soluble Fms-like tyrosine kinase 1 as predictors of all-cause and cardiovascular mortality in patients with Type 1 diabetes with and without diabetic nephropathy. Diabet Med. 2012;29:337–344. doi: 10.1111/j.1464-5491.2011.03482.x. [DOI] [PubMed] [Google Scholar]
- 21.Rambod M, Heine GH, Seiler S, Dominic EA, Rogacev KS, Dwivedi R, Ramezani A, Wing MR, Amdur RL, Fliser D, Raj DS. Association of vascular endothelial factors with cardiovascular outcome and mortality in chronic kidney disease patients: a 4-year cohort study. Atherosclerosis. 2014;236:360–365. doi: 10.1016/j.atherosclerosis.2014.07.026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Zhang AH, Wang G, Zhang DL, Zhang QD, Liu S, Liao Y, Yin Y, Liu WH. Association between VEGF receptors and baseline peritoneal transport status in new peritoneal dialysis patients. Ren Fail. 2012;34:582–589. doi: 10.3109/0886022X.2012.669322. [DOI] [PubMed] [Google Scholar]
- 23.Lai S, Molfino A, Russo GE, Testorio M, Galani A, Innico G, Frassetti N, Pistolesi V, Morabito S, Rossi Fanelli F. Cardiac, inflammatory and metabolic parameters: hemodialysis versus peritoneal dialysis. Cardiorenal Med. 2015;5:20–30. doi: 10.1159/000369588. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Peiskerová M, Kalousová M, Danzig V, Míková B, Hodková M, Němeček E, Bani-Hani A, Ambrož D, Benáková H, Linhart A, Zima T, Tesař V. Placental growth factor may predict increased left ventricular mass index in patients with mild to moderate chronic kidney disease - a prospective observational study. BMC Nephrol. 2013;14:142. doi: 10.1186/1471-2369-14-142. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Accornero F, van Berlo JH, Benard MJ, Lorenz JN, Carmeliet P, Molkentin JD. Placental growth factor regulates cardiac adaptation and hypertrophy through a paracrine mechanism. Circ Res. 2011;109:272–280. doi: 10.1161/CIRCRESAHA.111.240820. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Ha H, Cha MK, Choi HN, Lee HB. Effects of peritoneal dialysis solutions on the secretion of growth factors and extracellular matrix proteins by human peritoneal mesothelial cells. Perit Dial Int. 2002;22:171–177. [PubMed] [Google Scholar]
- 27.De Vriese AS, Tilton RG, Stephan CC, Lameire NH. Vascular endothelial growth factor is essential for hyperglycemia-induced structural and functional alterations of the peritoneal membrane. J Am Soc Nephrol. 2001;12:1734–1741. doi: 10.1681/ASN.V1281734. [DOI] [PubMed] [Google Scholar]
- 28.Hazama T, Fukami K, Yamagishi S, Kusumoto T, Sakai K, Adachi T, Sonoda K, Kasuga S, Ueda S, Okuda S. Dialysate vascular endothelial growth factor is an independent determinant of serum albumin levels and predicts future withdrawal from peritoneal dialysis in uremic patients. Ther Apher Dial. 2014;18:391–397. doi: 10.1111/1744-9987.12120. [DOI] [PubMed] [Google Scholar]