Summary
This review challenges the use of solutions of dissolved exogenous H2S in the literature as a tool to determine the potential physiological functions of endogenous H2S as well as its putative therapeutic applications.
Our major point of contention is that solutions of dissolved H2S are used in vitro at concentrations, within the high microM range, which are above the concentrations of dissolved H2S found in blood and tissues during H2S lethal exposure in vivo. In addition, since the levels of toxicity are extremely variable among cell types, a property that is seldom acknowledged, the physiological relevance of data obtained after local or in-vitro administrations of H2S at concentrations of few microM is far from certain. Conversely, the rate of disappearance of the dissolved pool of H2S in the body (being trapped or oxidized), which we found to be at least of several micromoles/kg/min, is so rapid in vivo that if relatively low quantities of H2S, i.e. few micromoles for instance, are administered, no change in H2S concentrations in the body is to be expected, unless toxic levels are used. Protocols looking at the effects of compounds slowly releasing H2S must also resolve a similar conundrum, as their effects must be reconciled with the unique ability of the blood and tissues to get rid of H2S and the steepness of the dose-toxic effects relationship.
Only by developing a comprehensive framework in which H2S metabolism and toxicity will be used as a rationale to develop and justify any experimental approach will we be able to bring definitive evidence supporting a protective role for exogenous H2S, if any, and its putative function as an endogenous mediator.
Keywords: Hydrogen sulfide, gaseous transmitter, Toxicology
What effects can we expect to be produced by a 5 microliter solution containing dissolved hydrogen sulfide (H2S/HS−) at the concentration of 2.5 milliM directly administered into the cerebro-ventricles of a rat? It is, in substance, one of the fundamental questions raised by Li et al. (Li et al., 2016) in their recent study published in this journal.
The approach consisting in injecting high microM or even milliM solutions of dissolved H2S directly into an organ or in the milieu bathing cells in culture has been repeatedly used in the literature (see for list of references (Nicholson and Calvert, 2010; Szabo, 2007; Szabo et al., 2011) with 2 main objectives: (1) determine whether exogenous H2S possess therapeutic properties, such as for instance limiting the consequences of an hypoxic/anoxic insult or (2) speculate about the possible role of “endogenous” H2S or its products of oxidation.
For all intents and purposes, the argument presented in this viewpoint is that solutions of exogenous H2S above few microM, when in direct contact with cells or tissues produce effects that are toxic or even lethal in vivo (cardiac arrest, coma with neuronal necrosis). In contrast, cells or tissues containing large amount of metallo-compounds or expressing high level of sulfide quinone reductase activity, e.g. colonocytes or heptacoytes, are able to trap or oxidize very large amount of H2S. These specific cells are immune to both the toxicity as well as “physiological” effects produced by sulfide in other cells or tissues. Conversely, low doses injected in vivo are probably not able to increase H2S in a significant manner, since the blood (hemoglobin and proteins) can prevent any free sulfide to diffuse in a measurable manner into the tissues, unless toxic levels are used. Perhaps more importantly, due to the multifarious effects of H2S, it is, for now, very difficult to define a specific function for H2S in vivo based on any given defined H2S-target interaction described from in vitro experiments.
1- What concentration of H2S is toxic?
Solutions containing H2S, prepared from NaSH or Na2S, have been used as a source of sulfide to test the effects of hydrogen sulfide in various in vitro or in vivo studies [see for general review and discussion (Deleon et al., 2011; Furne et al., 2008; Levitt et al., 2010; Olson, 2011a, b). The sulfides dissolved in these solutions are, at a physiological pH of about 7, composed in large part of HS− in equilibrium with a smaller portion of dissolved/free gaseous H2S (Almgren et al., 1976.; Carroll and Mather, 1989; Douabul and Riley, 1979). When used in cell cultures or isolated tissues, as well as when directly injected into an organ, the amount of H2S able to diffuse into cells is therefore proportional to the partial pressure of the gaseous form of H2S and thus the concentration of total free/dissolved H2S/HS−. Impurities and products of sulfide oxidation may be present in solution, their roles remain to be clarified (Nagy, 2015). Of note, when prepared without agitation and immediately before the experiment using a sealed container, concentration of gaseous H2S remains relatively stable over time (at least for one hour) (Van de Louw and Haouzi, 2013). As soon as the solution of H2S is added into a dish, evaporation of gaseous H2S takes place and the concentration of soluble H2S/HS− exposed to the cells or tissue decreases in the milieu depending on many factors including the temperature, the pH, the surface of exchange and the level of agitation. In most experimental conditions, if the solution is neither agitated nor ventilated, a concentration in high microM or milliM range is going to remain present within the same order of magnitude for many minutes, a time long enough to produce a lethal effect (Judenherc-Haouzi et al., 2016). Many of these points have been discussed in details in recent reviews (Olson, 2011a; Olson et al., 2014).
It should be kept in mind that H2S is one of the most toxic mitochondrial poisons, even more toxic than cyanide on a mole-to-mole basis. In vitro, the activity of the mitochondrial cytochrome c oxidase is abolished by a solution of H2S at concentrations of H2S/HS− ranging from 10 to 30 microM (Cooper and Brown, 2008; Leschelle et al., 2005; Yong and Searcy, 2001). This effect appears however to develop at much lower concentrations in certain tissues (neurons), while cells like the colonocytes can survive exposure to milliM concentrations of free H2S for long period of time. In vivo, a depression in respiratory medullary neurons (leading to a fatal apnea within minutes) and a severe depression in cardiac contractility (leading to a terminal asystole within seconds) can be produced in rodents and in large mammals (figure 1) by infusing or inhaling H2S at levels yielding blood concentrations of gaseous H2S between 2–5 microM (Haouzi et al., 2014; Klingerman et al., 2013; Sonobe and Haouzi, 2016), corresponding to level of total dissolved/free sulfide of less than 20 microM (Almgren et al., 1976.; Carroll and Mather, 1989; Douabul and Riley, 1979). For instance, whenever solutions of H2S at concentrations above 50 microM are used on an isolated heart or isolated cardiac cells, the effect is always a depression in cardiac contractility (Geng et al., 2004; Sun et al., 2008; Yong et al., 2008; Zhang et al., 2012). This depression is an obvious pathological and toxic change in the function at both the level of individual cardiac cells and the whole heart (Judenherc-Haouzi et al., 2016), akin to the effects observed in vivo during life threatening H2S exposure (figure 2). In a recent study, we found that the ejection fraction of the left ventricle started to decrease for an average level of gaseous H2S of 7.09 ± 3.53 microM, while blood pressure is already decreased around 5 microM. This corresponds to a total level dissolved H2S/HS− of about 15–20 microM (assuming that 2/3 of total dissolved sulfide present is in the form of HS− at physiological pH). Finally, Extensive necrosis of the cortical and subcortical neurons is observed in about 30% of animals surviving H2S induced coma (Sonobe et al., 2015), which can develop after exposure to concentrations of free H2S in the blood in the microM range for few minutes only. Interpretation of the effects of exogenous H2S must consider the fact that effects of H2S on, for instance, neurons, cardiomyocytes, hepatocytes or cancer cells - the latter relying on a glycolytic activity for ATP production- are not interchangeable. In addition, the level of metabolism of cells in culture and in vivo can be very different. A model allowing us to translate the effects of H2S in a dish to those in vivo is therefore cruelly missing, but few microM concentrations of dissolved H2S does produce toxic effects in most tissues in vivo, affecting vital respiratory and cardio-vascular functions.
FIGURE 1.
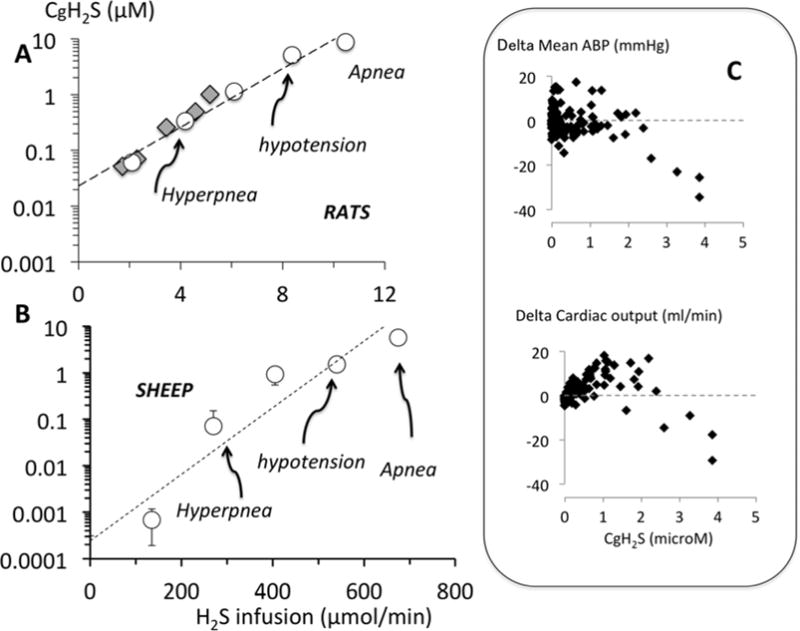
A: Concentration of gaseous H2s in the arterial blood (CgH2S) estimated from alveolar H2S as a function of the rate of H2S infused (only mean values are shown) in a group of rats. Breathing was stimulated at concentrations of gaseous H2S ranging between 0.34 and 1.14 μM while the highest values corresponding to the lethal level ranged from 5.09 to 8.80 μM. The data recomputed form the study of Insko et al (Insko et al., 2009) (diamonds) fits with the relationship established in the present study. Modified from (Klingerman et al., 2013). Of note these concretion represents 1/3 of total Free H2S, i.e. H2S/HS−, present in the blood. B: Concentration of gaseous H2S in the arterial blood (CgH2S) estimated from alveolar H2S as a function of the rate of H2S infused in a group of sheep. Breathing stimulation and apnea were produced at levels similar to those found in the rats. Modified from (Haouzi et al., 2014) C: Individual data points obtained in a group of rats during infusion of NaHS. Note that when CgH2S reached concentrations of about 2–3 microM (less than 10 microM total free sulfide), a progressive drop in blood pressure associated with a reduction in cardiac output was observed. Note the very narrow range of H2S between trivial and life-threatening effects. Modified from (Sonobe and Haouzi, 2016).
FIGURE 2.
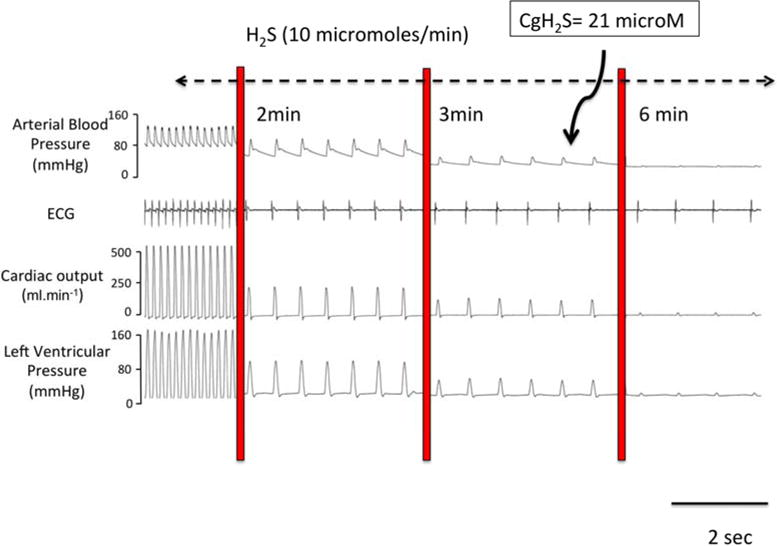
Example of a recording obtained in an anesthetized and mechanically ventilated rat, showing the effects of a continuous infusion of a solution of dissolved H2S at 10 μmol/min on hemodynamics and cardiac function. Blood pressure, EKG signal, cardiac output and left ventricular pressure are displayed. Within few minutes only, H2S led to a complete pulseless electrical activity or electromechanical dissociation, as the heart becomes incapable of contracting despite a persistent electrical activity visible of the EKG signal, while CgH2S in the blood reached about 20 microM (60 microM total free H2S). Modified from (Sonobe and Haouzi, 2015).
2- Why are toxic levels of H2S used in physiological studies?
The rational for using high levels, i.e. microM or even mM, of H2S to study and predict physiological effects relies on initial reports, wherein H2S concentrations were found to be in the high micromolar range in the blood or in tissues under conditions assumed to be endogenous H2S production only [see for discussion (Furne et al., 2008)]. Furne and Levitt (Furne et al., 2008; Levitt et al., 2010) followed by works from Olson’s group (Whitfield et al., 2008) have shown that these figures, i.e. high microM concentrations of free/soluble H2S, are unrealistically high by several orders of magnitude. Two main reasons have been put forward to explain why high microM levels of “endogenous” H2S were initially (and are still) reported in the blood and tissues. The first reason relates to the nature of the pools of sulfide present in the blood and in tissues found in post mortem conditions. No relevant (or trivial) levels of free/dissolved H2S (Furne et al., 2008; Levitt et al., 2010), which should smell like rotten eggs, can be found in the blood, the brain (Furne et al., 2008; Ishigami et al., 2009; Levitt et al., 2011) or heart (Levitt et al., 2011; Ubuka et al., 2001) homogenates. However, significant amount of H2S can be mobilized after exposing these tissues to a strong acid (bringing down pH of the tissues below 2) and can be measured after evaporating from the brain (Goodwin et al., 1989; Ishigami et al., 2009; Levitt et al., 2011; Ogasawara et al., 1994) or the heart (Levitt et al., 2011 Ogasawara, 1994 #1383; Ubuka et al., 2001). This pool of H2S represents several micromoles of sulfide per kg of tissue (Goodwin et al., 1989; Ishigami et al., 2009; Levitt et al., 2011; Ogasawara et al., 1994; Ubuka et al., 2001), but is “trapped”, literally fossilized, in the form for instance of metalo-sulfide (including Fe, Zn, Co etc..). Similarly, strong reducing agents can release H2S from a solution of proteins or from various tissues, where H2S is present under the form protein-bound thiols (Ogasawara et al., 1994; Warenycia et al., 1990). This effect can be produced by using large concentrations of DTT and TCEP (although DTT sometimes used as a reducing agent can per se also release H2S (Togawa et al., 1992)). The presence of H2S evaporating after such reactions should not be confused with the normal presence of dissolved H2S, able to freely diffuse and to potentially act as a gaseous-transmitter. The conditions leading to the release of free H2S by a powerful reducing agent or a strong acid are utterly artificial and can obviously never be spontaneously reproduced in vivo. The source of these “hidden” pools of H2S is far from being understood and could originate from numerous extra and intracellular thiol compounds and cysteine residues of almost all proteins (when exposed to a reducing agent) or sulfide combined with metallo-proteins (when exposed to a strong acid). The second reason accounting for these artificially high levels of free H2S reported in the blood and tissues relate to errors produced by the methodology of determination of the pool of diffusible/dissolved H2S in a biological milieu, either directly or after trapping it with Zn, using for instance techniques relying on the production of methylene blue based spectrophotometry method, which level of detection is relatively low and is affected by the turbidity of the milieu (Van de Louw and Haouzi, 2012).
3- Low levels of H2S administered in vivo are unlikely to increase concentrations of H2S in tissues
In contrast to the fate of a solution of H2S in vitro, the majority of H2S present in the blood during an intravenous (IV), or following an intra-peritoneal (IP), injections in vivo is immediately oxidized or combined with metallo-proteins (hemoglobin), dropping the level of free H2S to almost zero, unless lethal levels are used (Haouzi and Klingerman, 2013; Insko et al., 2009; Klingerman et al., 2013; Wintner et al., 2010). There is a very fine margin between the concentrations producing no effect and concentrations capable to kill. More specifically until the level of IV infusion reaches 3–5 micromol/kg/min in rat (Haouzi and Klingerman, 2013; Insko et al., 2009; Klingerman et al., 2013; Wintner et al., 2010) or large mammals (Haouzi et al., 2014), soluble H2S concentration barely increases in the blood remaining in the nanoM range. Whenever the amount of H2S administered IV at a rate that would be less than 5 micromole/kg/min, trivial diffusion of H2S into the tissues is therefore to be expected. More importantly, as all free H2S disappears from the body within one minute after an infusion of exogenous H2S is stopped (Haouzi and Klingerman, 2013; Insko et al., 2009; Klingerman et al., 2013; Wintner et al., 2010) due to the high rate of H2S oxidation in the mitochondria, it is unlikely that any IV injection of few micromoles/kg of H2S could significantly affect the levels of free H2S in the tissues in any given organ. Conversely, since the relationship dose effect is extremely steep for H2S, studies using a rate of H2S higher than 20 micromole/kg/min should be regarded as extremely toxic producing the dreadful effects on most physiological functions, this level of toxicity can be produced by an IP injection of 300 micromol/kg (Sonobe et al., 2015; Sonobe and Haouzi, 2015). As displayed in the lists of studies reviewed for instance by Szabo et al (Szabo, 2007; Szabo et al., 2011), the concentrations of H2S used among experiments are so variable that their results in toxic levels or levels of H2S unable to change H2S concentration in tissues. This issue is extremely relevant to the use of compounds which are slowly releasing H2S, for which no change in H2S in the blood and tissue might be expected if the rate of H2S “release” is too slow, while if higher rates of release are present, toxic levels can be reached in certain tissues like neurons for instance, with almost no transition from innocuous concentrations. The very short life (less than one minute) of free sulfide in the blood and the tissues {Haouzi, 2014 #1370} makes this argument even more compelling. This is not to say that no “therapeutic” window can not be found, but with respect to H2S, this window is expected to be extremely narrow varying form tissue to tissue in keeping with multiple factors and interactions much too complex to be inventoried yet (from content in metallo-proteins to sulfide quinone reductase activity)
4- Can we separate wheat from chaff?
Investigating the role of a given molecule as a neuro-mediator or a gaseous-transmitter, relies on two prerequisites: 1- the effect of this molecule is only dictated by the limited numbers of accessible receptors or targets and the affinity for these targets; 2- the molecule of interest has no relevant effects outside its interaction with its receptors or its targets, at least within the range of concentrations of interest. As a fundamental corollary, the responses observed in vivo must be predicted or anticipated from those produced in vitro, and vice versa. For instance, applying dopamine or morphine directly into a tissue or in cell cultures will produce effects that can be understood in light of the interactions between dopamine and dopaminergic and adrenergic receptors or between morphine and opioid receptors, in a dose-dependent manner. These responses can be translated in vivo into a series of physiological effects and functions. However, H2S does not fit into this paradigm. Indeed at any given concentration, exogenously applied H2S is going to interact with a large number of receptors and metabolic pathways comprising interactions with cysteine residues of proteins and a pool of countless metallo-proteins; ATP regeneration will be impeded (even at very low levels) while producing free radicals and affecting the activity of many ion channels, with an alarming overlap between concentrations producing toxic or “non-toxic” effects. This makes extremely difficult, even in reduced preparations, to establish a causal link between a perturbation and a H2S-target interaction, unless all these interactions are understood and could be dissociated into elementary responses, which is, for now, totally unrealistic. This notion remains valid whether the described mode of action is a receptor-molecule interaction or a cascade of signaling pathways initiated or modulated by soluble H2S or any products of sulfide oxidation. Another level of confusion has aroused from the fact that the frame of reference used to rationalize experimental findings about hydrogen sulfide has been constantly changing (Haouzi et al., 2011). For instance, H2S has been claimed to be a gaseous-transmitter in large part based on the premise that free H2S is released from tissues and can be measured in conditions susceptible to increase its concentration in tissues (see (Olson, 2008) for discussion on the effects of hypoxia). If true, it should be possible to decrease H2S concentrations in cells by increasing the ability of H2S to diffuse more rapidly through for instance the trapping of H2S as soon as it diffuses in the blood or even directly into tissues (Haouzi, 2011). However the above approach becomes obsolete, if data supporting the diffusion of free H2S in and outside cells or tissues result from an artifact (Olson, 2011a), if H2S would be trapped as soon as it diffuses in the blood in baseline conditions. The implications of such a debate are rarely acknowledged but are quite simple, rejecting data obtained from the measurement of H2S diffusing outside various tissues should make us refute the original hypothesis, which was the rational for measuring H2S in the first place, that is H2S is a gaseous transmitter. The confusion here does not result from a difference in opinion but from a constantly evolving framework to understand what level of H2S is required to produce a claimed effect. This process, which should be regarded as a self-correcting process in any scientific endeavor, has resulted in the creation of an untestable hypothesis. The analogy made between the presence and role of H2S and the search for “an invisible, incorporeal, floating dragon who spits heatless fire hired in Sagan’s garage (Sagan, 1997)” still holds (Haouzi, 2011). A “dragon” with such characteristics makes no difference with “no dragon at all”. Until each experiment using exogenous H2S can define what concentrations of H2S are really present in the tissues and what effects are expected to be produced in keeping with sulfide toxicity and its rapid disappearance from the body or in vitro solution, no proper case can be made to support the role of H2S as a neuro-mediator.
5 - Final words
Finding a compound/molecule, which possesses genuine protective properties against the effects of anoxia, is certainly the Holy Grail of critical care research. However, if one wants to offer a mitochondrial poison like H2S as a possible treatment of the dreadful consequences of hypoxia-induced neuronal injury for instance, not only should the “limitations” of such an idea be presented in a comprehensive way, but undisputable evidence supporting this disturbing claim should be provided, unless by protection we imply that repetitive anoxic insults produced by H2S toxicity may, through some form of preconditioning, limit the effects of future hypoxia or ischemia.
A large body of evidence supporting the “physiological” and protective roles of H2S has been obtained using solution of free H2S in the high microM range - levels only found during a lethal intoxication in vivo. Despite the fact that such concentrations of free H2S, as physiological levels, have been repeatedly shown to be unrealistically high due to possible errors on the nature of the sulfide compounds that could be released from the blood or tissues, high microM or milliM levels of free sulfide are still used to try to elucidate the properties of endogenous H2S.
As an answer to the question raised in the introduction, we could offer the following proposition, which, one should admit, remains a pure speculation: injecting 5 μl of a solution with a concentration of 2.5 mM of H2S/HS− in the cerebro-ventricles - assuming a total CSF volume in rat brain of approximately 90 μl- could seriously intoxicate all the neurons surrounding the site of injection within seconds. In addition, since the cerebrospinal fluid, unlike the blood, contains little metallo-proteins to rapidly “trap” free sulfide, the effect of such a high concentration of H2S/HS− on more distant neuronal populations is quite worrisome and must be understood.
Finally, the effect of H2S cannot be reduced to well-defined physiological compound-target interactions. The reactivity of H2S with a multitude of critical biological processes is so pervasive at the dose typically used to produce a so-called physiological effect that the data obtained from these studies should in many instances belong to the field of toxicology.
Only the accumulation of experimental evidence based on well validated methodologies and a proper framework allowing us to give meaning to all the effects produced in vivo and in vitro by exogenous H2S will eventually bring a final answer to the question of the protective role, if any, of H2S and its potential functions as a endogenous mediator.
Acknowledgments
The author would like to thank Dr. Takashi Sonobe and Dr. Matthew Rannals for their comments and suggestions. This work has been supported by the CounterACT Program, National Institutes of Health Office of the Director (NIH OD), and the National Institute of Neurological Disorders and Stroke (NINDS), Grant Number 1R21NS080788-01 and 1R21NS090017-01.
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
References
- Almgren T, Dyrssen D, Elgquist B, Johannsson O. Dissociation of hydrogen sulfide in seawater and comparison of pH scales. Marine Chemistry. 1976;4:289–297. [Google Scholar]
- Carroll JJ, Mather AE. The solubility of hydrogen sulfide in water from 0 to 90°C and pressures to 1 MPa. Geochim Cosmochim Acta. 1989;53:1163–1170. [Google Scholar]
- Cooper CE, Brown GC. The inhibition of mitochondrial cytochrome oxidase by the gases carbon monoxide, nitric oxide, hydrogen cyanide and hydrogen sulfide: chemical mechanism and physiological significance. J Bioenerg Biomembr. 2008;40:533–539. doi: 10.1007/s10863-008-9166-6. [DOI] [PubMed] [Google Scholar]
- Deleon ER, Stoy GF, Olson KR. Passive loss of hydrogen sulfide in biological experiments. Anal Biochem. 2011 doi: 10.1016/j.ab.2011.10.016. [DOI] [PubMed] [Google Scholar]
- Douabul AA, Riley JP. The solubility of gases in distilled water and seawater - V. Hydrogen sulphide. Deep-Sea Research. 1979;26A:259–268. 1979. 26A, 259–268. [Google Scholar]
- Furne J, Saeed A, Levitt MD. Whole tissue hydrogen sulfide concentrations are orders of magnitude lower than presently accepted values. American journal of physiology. Regulatory, integrative and comparative physiology. 2008;295:R1479–1485. doi: 10.1152/ajpregu.90566.2008. [DOI] [PubMed] [Google Scholar]
- Geng B, Yang J, Qi Y, Zhao J, Pang Y, Du J, Tang C. H2S generated by heart in rat and its effects on cardiac function. Biochemical and biophysical research communications. 2004;313:362–368. doi: 10.1016/j.bbrc.2003.11.130. [DOI] [PubMed] [Google Scholar]
- Goodwin LR, Francom D, Dieken FP, Taylor JD, Warenycia MW, Reiffenstein RJ, Dowling G. Determination of sulfide in brain tissue by gas dialysis/ion chromatography: postmortem studies and two case reports. J Anal Toxicol. 1989;13:105–109. doi: 10.1093/jat/13.2.105. [DOI] [PubMed] [Google Scholar]
- Haouzi P. Sulfide and methemoglobinemia. Respiratory physiology & neurobiology. 2011;179:119–120. doi: 10.1016/j.resp.2011.09.009. [DOI] [PubMed] [Google Scholar]
- Haouzi P, Bell H, Van de Louw A. Hypoxia-induced arterial chemoreceptor stimulation and hydrogen sulfide: too much or too little? Respiratory physiology & neurobiology. 2011;179:97–102. doi: 10.1016/j.resp.2011.09.009. [DOI] [PubMed] [Google Scholar]
- Haouzi P, Klingerman CM. Fate of intracellular H2S/HS(−) and metallo-proteins. Respiratory physiology & neurobiology. 2013;188:229–230. doi: 10.1016/j.resp.2013.05.029. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Haouzi P, Sonobe T, Torsell-Tubbs N, Prokopczyk B, Chenuel B, Klingerman CM. In vivo interactions between cobalt or ferric compounds and the pools of sulphide in the blood during and after H2S poisoning. Toxicological sciences: an official journal of the Society of Toxicology. 2014;141:493–504. doi: 10.1093/toxsci/kfu140. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Insko MA, Deckwerth TL, Hill P, Toombs CF, Szabo C. Detection of exhaled hydrogen sulphide gas in rats exposed to intravenous sodium sulphide. Br J Pharmacol. 2009;157:944–951. doi: 10.1111/j.1476-5381.2009.00248.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ishigami M, Hiraki K, Umemura K, Ogasawara Y, Ishii K, Kimura H. A source of hydrogen sulfide and a mechanism of its release in the brain. Antioxidants & redox signaling. 2009;11:205–214. doi: 10.1089/ars.2008.2132. [DOI] [PubMed] [Google Scholar]
- Judenherc-Haouzi A, Zhang XQ, Sonobe T, Song J, Rannals MD, Wang J, Tubbs N, Cheung JY, Haouzi P. METHYLENE BLUE COUNTERACTS H2S TOXICITY INDUCED CARDIAC DEPRESSION BY RESTORING L-TYPECa CHANNEL ACTIVITY. American journal of physiology. Regulatory, integrative and comparative physiology. 2016 doi: 10.1152/ajpregu.00527.2015. ajpregu 00527 02015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Klingerman CM, Trushin N, Prokopczyk B, Haouzi P. H2S concentrations in the arterial blood during H2S administration in relation to its toxicity and effects on breathing. American journal of physiology. Regulatory, integrative and comparative physiology. 2013;305:R630–638. doi: 10.1152/ajpregu.00218.2013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Leschelle X, Goubern M, Andriamihaja M, Blottiere HM, Couplan E, Gonzalez-Barroso MD, Petit C, Pagniez A, Chaumontet C, Mignotte B, Bouillaud F, Blachier F. Adaptative metabolic response of human colonic epithelial cells to the adverse effects of the luminal compound sulfide. Biochim Biophys Acta. 2005;1725:201–212. doi: 10.1016/j.bbagen.2005.06.002. [DOI] [PubMed] [Google Scholar]
- Levitt MD, Abdel-Rehim MS, Furne J. Free and Acid-labile Hydrogen Sulfide Concentrations in Mouse Tissues: Anomalously High Free Hydrogen Sulfide in Aortic Tissue. Antioxidants & redox signaling. 2010 doi: 10.1089/ars.2010.3525. [DOI] [PubMed] [Google Scholar]
- Levitt MD, Abdel-Rehim MS, Furne J. Free and acid-labile hydrogen sulfide concentrations in mouse tissues: anomalously high free hydrogen sulfide in aortic tissue. Antioxidants & redox signaling. 2011;15:373–378. doi: 10.1089/ars.2010.3525. [DOI] [PubMed] [Google Scholar]
- Li H, Chen L, Hou X, Zhou H, Zheng Y. Hydrogen sulfide attenuates hypoxia-induced respiratory suppression in anesthetized adult rats. Respiratory physiology & neurobiology. 2016;220:1–9. doi: 10.1016/j.resp.2015.09.001. [DOI] [PubMed] [Google Scholar]
- Nagy P. Mechanistic chemical perspective of hydrogen sulfide signaling. Methods in enzymology. 2015;554:3–29. doi: 10.1016/bs.mie.2014.11.036. [DOI] [PubMed] [Google Scholar]
- Nicholson CK, Calvert JW. Hydrogen sulfide and ischemia-reperfusion injury. Pharmacological research. 2010;62:289–297. doi: 10.1016/j.phrs.2010.06.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ogasawara Y, Isoda S, Tanabe S. Tissue and subcellular distribution of bound and acid-labile sulfur, and the enzymic capacity for sulfide production in the rat. Biological & pharmaceutical bulletin. 1994;17:1535–1542. doi: 10.1248/bpb.17.1535. [DOI] [PubMed] [Google Scholar]
- Olson KR. Hydrogen sulfide and oxygen sensing: implications in cardiorespiratory control. J Exp Biol. 2008;211:2727–2734. doi: 10.1242/jeb.010066. [DOI] [PubMed] [Google Scholar]
- Olson KR. A Practical Look at the Chemistry and Biology of Hydrogen Sulfide. Antioxidants & redox signaling. 2011a doi: 10.1089/ars.2011.4401. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Olson KR. The therapeutic potential of hydrogen sulfide: separating hype from hope. American journal of physiology. Regulatory, integrative and comparative physiology. 2011b;301:R297–312. doi: 10.1152/ajpregu.00045.2011. [DOI] [PubMed] [Google Scholar]
- Olson KR, DeLeon ER, Liu F. Controversies and conundrums in hydrogen sulfide biology. Nitric oxide: biology and chemistry/official journal of the Nitric Oxide Society. 2014;41:11–26. doi: 10.1016/j.niox.2014.05.012. [DOI] [PubMed] [Google Scholar]
- Sagan C. The Demon-Haunted World: Science As A Candle In the Dark. Ballantine Books; New York: 1997. [Google Scholar]
- Sonobe T, Chenuel B, Cooper TK, Haouzi P. Immediate and Long-Term Outcome of Acute H2S Intoxication Induced Coma in Unanesthetized Rats: Effects of Methylene Blue. PLoS One. 2015;10:e0131340. doi: 10.1371/journal.pone.0131340. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sonobe T, Haouzi P. H2S induced coma and cardiogenic shock in the rat: Effects of phenothiazinium chromophores. Clin Toxicol (Phila) 2015;53:525–539. doi: 10.3109/15563650.2015.1043440. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sonobe T, Haouzi P. Sulfide Intoxication-Induced Circulatory Failure is Mediated by a Depression in Cardiac Contractility. Cardiovasc Toxicol. 2016;16(1):67–78. doi: 10.1007/s12012-015-9309-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sun YG, Cao YX, Wang WW, Ma SF, Yao T, Zhu YC. Hydrogen sulphide is an inhibitor of L-type calcium channels and mechanical contraction in rat cardiomyocytes. Cardiovasc Res. 2008;79:632–641. doi: 10.1093/cvr/cvn140. [DOI] [PubMed] [Google Scholar]
- Szabo C. Hydrogen sulphide and its therapeutic potential. Nat Rev Drug Discov. 2007;6:917–935. doi: 10.1038/nrd2425. [DOI] [PubMed] [Google Scholar]
- Szabo G, Veres G, Radovits T, Gero D, Modis K, Miesel-Groschel C, Horkay F, Karck M, Szabo C. Cardioprotective effects of hydrogen sulfide. Nitric oxide: biology and chemistry/official journal of the Nitric Oxide Society. 2011;25:201–210. doi: 10.1016/j.niox.2010.11.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Togawa T, Ogawa M, Nawata M, Ogasawara Y, Kawanabe K, Tanabe S. High performance liquid chromatographic determination of bound sulfide and sulfite and thiosulfate at their low levels in human serum by pre-column fluorescence derivatization with monobromobimane. Chemical & pharmaceutical bulletin. 1992;40:3000–3004. doi: 10.1248/cpb.40.3000. [DOI] [PubMed] [Google Scholar]
- Ubuka T, Abe T, Kajikawa R, Morino K. Determination of hydrogen sulfide and acid-labile sulfur in animal tissues by gas chromatography and ion chromatography. Journal of chromatography. B, Biomedical sciences and applications. 2001;757:31–37. doi: 10.1016/s0378-4347(01)00046-9. [DOI] [PubMed] [Google Scholar]
- Van de Louw A, Haouzi P. Oxygen deficit and H2S in hemorrhagic shock in rats. Crit Care. 2012;16:R178. doi: 10.1186/cc11661. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Van de Louw A, Haouzi P. Ferric Iron and Cobalt (III) compounds to safely decrease hydrogen sulfide in the body? Antioxidants & redox signaling. 2013;19:510–516. doi: 10.1089/ars.2012.4513. [DOI] [PubMed] [Google Scholar]
- Warenycia MW, Goodwin LR, Francom DM, Dieken FP, Kombian SB, Reiffenstein RJ. Dithiothreitol liberates non-acid labile sulfide from brain tissue of H2S-poisoned animals. Arch Toxicol. 1990;64:650–655. doi: 10.1007/BF01974693. [DOI] [PubMed] [Google Scholar]
- Whitfield NL, Kreimier EL, Verdial FC, Skovgaard N, Olson KR. Reappraisal of H2S/sulfide concentration in vertebrate blood and its potential significance in ischemic preconditioning and vascular signaling. American journal of physiology. Regulatory, integrative and comparative physiology. 2008;294:R1930–1937. doi: 10.1152/ajpregu.00025.2008. [DOI] [PubMed] [Google Scholar]
- Wintner EA, Deckwerth TL, Langston W, Bengtsson A, Leviten D, Hill P, Insko MA, Dumpit R, VandenEkart E, Toombs CF, Szabo C. A monobromobimane-based assay to measure the pharmacokinetic profile of reactive sulphide species in blood. Br J Pharmacol. 2010;160:941–957. doi: 10.1111/j.1476-5381.2010.00704.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yong QC, Pan TT, Hu LF, Bian JS. Negative regulation of beta-adrenergic function by hydrogen sulphide in the rat hearts. Journal of molecular and cellular cardiology. 2008;44:701–710. doi: 10.1016/j.yjmcc.2008.01.007. [DOI] [PubMed] [Google Scholar]
- Yong R, Searcy DG. Sulfide oxidation coupled to ATP synthesis in chicken liver mitochondria. Comp Biochem Physiol B Biochem Mol Biol. 2001;129:129–137. doi: 10.1016/s1096-4959(01)00309-8. [DOI] [PubMed] [Google Scholar]
- Zhang H, Guo C, Wu D, Zhang A, Gu T, Wang L, Wang C. Hydrogen sulfide inhibits the development of atherosclerosis with suppressing CX3CR1 and CX3CL1 expression. PLoS One. 2012;7:e41147. doi: 10.1371/journal.pone.0041147. [DOI] [PMC free article] [PubMed] [Google Scholar]


