Abstract
Degeneration of dopamine neurons in the midbrain causes symptoms of the movement disorder, Parkinson disease. Dopamine neurons are generated from proliferating progenitor cells localized in the embryonic ventral midbrain. However, it remains unclear for how long cells with dopamine progenitor character are retained and if there is any potential for reactivation of such cells after cessation of normal dopamine neurogenesis. We show here that cells expressing Lmx1a and other progenitor markers remain in the midbrain aqueductal zone beyond the major dopamine neurogenic period. These cells express dopamine receptors, are located in regions heavily innervated by midbrain dopamine fibres and their proliferation can be stimulated by antagonizing dopamine receptors, ultimately leading to increased neurogenesis in vivo. Furthermore, treatment with dopamine receptor antagonists enhances neurogenesis in vitro, both from embryonic midbrain progenitors as well as from embryonic stem cells. Altogether our results indicate a potential for reactivation of resident midbrain cells with dopamine progenitor potential beyond the normal period of dopamine neurogenesis.
Midbrain dopamine neurons play key roles in neurotransmission controlling locomotion, cognition and reward. Major motor symptoms in Parkinson’s disease are caused by degeneration of dopamine neurons and an important research aim has therefore been to develop methodology for engineering dopamine neurons from stem cells in vitro for cell replacement therapy in Parkinson’s patients1,2,3, reviewed in ref. 4. Detailed understanding of the normal process of dopamine neurogenesis has been essential in these efforts, reviewed in ref. 5. During development, spatially organized signalling events lead to the expression of transcription factors, including the LIM-homeodomain protein Lmx1a in early proliferating neural progenitors localized close to the ventricular wall of the midbrain aqueduct6,7. Lmx1a, together with the related transcription factor Lmx1b, specify neural progenitors and is essential for the initiation of a molecular program for dopamine neurogenesis8,9. As Lmx1a-specified progenitor cells exit the cell cycle additional transcription factors are induced, including Nurr1 and Pitx35. These factors promote dopamine neuron differentiation and the acquisition of dopaminergic characteristics as cells migrate first radially and then tangentially towards the prospective ventral tegmental area and substantia nigra10,11.
The bulk of dopamine neurogenesis occurs normally between embryonic days (E) 10–14 in mice12. An exciting possibility would be if substantial dopamine neurogenesis could be induced after the main period of embryonic dopamine neurogenesis. The potential for late dopamine neurogenesis has previously been investigated. Some studies indicate that loss of dopamine neurons in the adult brain evokes responses that could lead to de novo generation of dopamine neurons, while others find no evidence for such events13,14,15,16. However, both the identity of potential progenitor cells as well as the mechanisms regulating their fate have remained elusive.
Here we addressed the duration of Lmx1a expression in midbrain ventricular cells, and whether Lmx1a-expressing cells could constitute a cell population with progenitor potential for reactivation at later stages of embryogenesis. We also asked if neurogenesis in the developing ventral midbrain could be regulated by the neurotransmitter dopamine itself since previous findings revealed that in aquatic salamanders, dopamine negatively controls the production of dopamine neurons both during homeostasis and regeneration17. Salamanders are the only vertebrates known to date with the ability to fully restore the dopaminergic system by a process that is driven by reactivation of dopamine neurogenesis18,19. Together our analyses indicate a potential for neurogenesis from persisting Lmx1a-expressing cells.
Results
Ventral midbrain ventricular cells maintained expression of Lmx1a and other progenitor markers
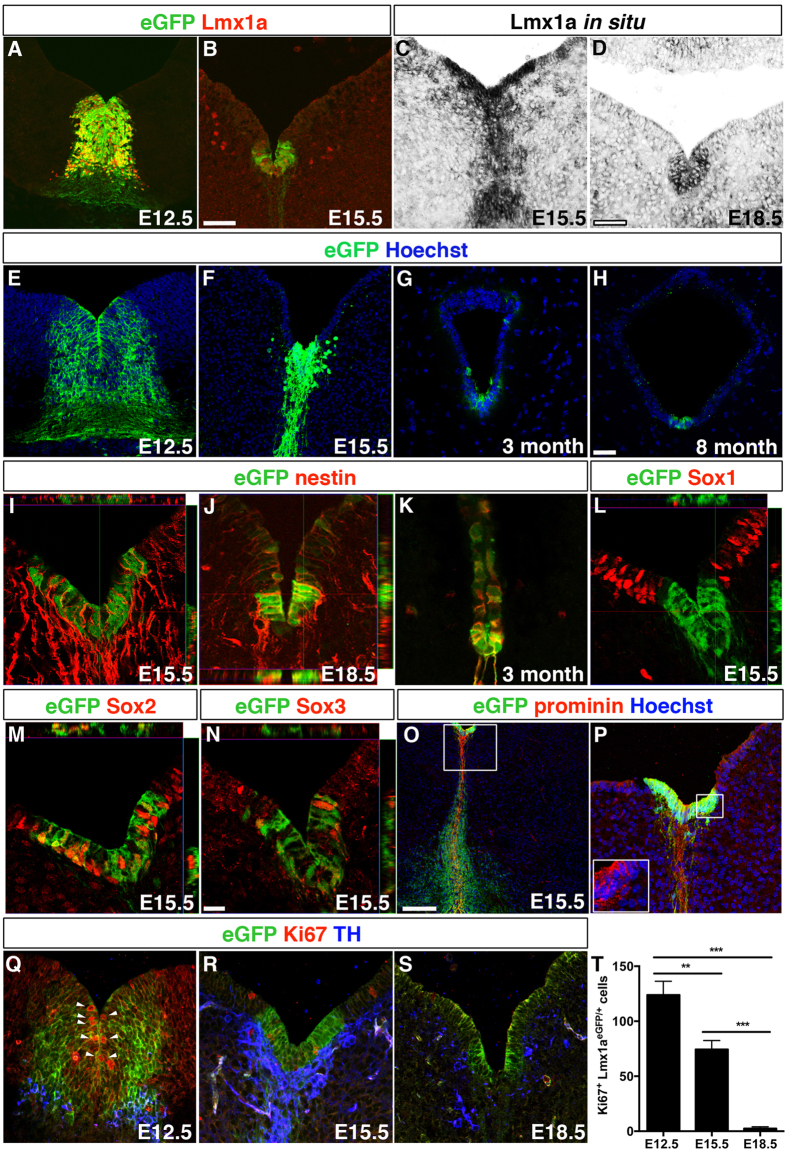
To investigate if Lmx1a expression was temporally restricted to the period of dopamine neurogenesis (E10-E14)12 we first performed immunostaining and in situ hybridizations at various time points during development. We found that Lmx1a expression was maintained in ventricular cells of the ventral midbrain at E15.5. In situ hybridization also demonstrated persistent Lmx1a mRNA expression at E15.5 and E18.5 (Fig. 1A–D). However, while Lmx1a protein was expressed at E15.5, no protein expression could be detected by immunohistochemistry at E18.5 and at postnatal stages. We confirmed these findings by examining heterozygous Lmx1aeGFP/+ reporter mouse embryos8, which in accordance with immunostaining and in situ hybridization showed declining expression, but persistent presence of eGFP+ cells throughout development and also in the adult animal at three and eight months of age (Fig. 1E–H).
Figure 1. Ventral midbrain Lmx1a-expressing ventricular cells maintained progenitor properties.
Lmx1a is expressed in ventral midline cells that generate midbrain dopamine neurons at E12.5, shown by staining against Lmx1a protein and using Lmx1aGFP/+ reporter mice (A). The expression of Lmx1a was maintained in ventricular progenitors at E15.5 (B,C) and E18.5 (D) shown by immunofluorescence and in situ hybridization. Lmx1aGFP/+ reporter mice revealed a persistent presence of eGFP+ ventricular cells during development and in the adult animals, depicted at E12.5 (E), E15.5 (F), 3 months (G) and 8 months of age (H). However, the eGFP+ cells decreased in numbers with time and changed in morphology with a gradual shortening of the processes (E–H). Ventral eGFP+ ventricular cells expressed nestin both in the embryo and the adult animal (I–K). At E15.5 the majority of cells lining the aqueduct were nestin+ (I), while at E18.5, nestin was restricted mainly to the eGFP+ cells (J). eGFP+ cells were distinguished from other ventricular cells by their lack of Sox1 expression (L), but showed overlapping expression with Sox2 (M), Sox3 (N) and prominin (O,P). The number of proliferating eGFP+ ventricular cells steadily decreased during development, with 123.9 ± 12.5 Ki67+ cells at E12.5 (Q,T, n = 6 mice), 74.4 ± 9.1 Ki67+ cells at E15.5 (R,T, n = 8 mice) and 2.5 ± 1.7 Ki67+ cells at E18.5 (S,T, n = 5 mice) (**P < 0.01, ***P < 0.001, ANOVA, Tukey’s post hoc test). Scale bars: 50 μM in (B) (applicable to A,K), 50 μM in (D) (applicable to C), 50 μM in (H) (applicable to E–G,P,Q–S), 20 μM in (N) (applicable to I,J,L,M) and 150 μM in (O).
To characterize ventral eGFP+ ventricular cells we analysed the expression of stem and progenitor markers during late embryogenesis. We found that eGFP+ cells expressed nestin both in the embryo and the adult animal (Fig. 1I–K). At E15.5 most ventricular cells lining the aqueduct were nestin+ (Fig. 1I), while at E18.5, the expression of nestin was restricted mainly to eGFP+ cells in the floor plate (FP) (Fig. 1J) and the roof plate (RP) (data not shown). Although nestin was maintained in the adult animal, the morphology of the eGFP+ cells changed markedly with age, displaying gradual shortening of their processes, as visualised by eGFP (Fig. 1G,H) and nestin expression (Fig. 1K). In contrast to the neighboring ventricular cells, eGFP+ cells were devoid of Sox1 (Fig. 1L), but showed an overlapping expression with Sox2 (Fig. 1M) and Sox3 (Fig. 1N). The Sox1, Sox2 and Sox3 expression patterns were maintained at E18.5 (Supplementary Fig. 1A–F). Notably, the eGFP+ cells also expressed the stem cell marker prominin (Fig. 1O,P). Adult ventricular eGFP+ cells also displayed a robust expression of Sox2 (Supplementary Fig. 2A–E), indicative of their maintained progenitor potential. In addition, a subset of the adult eGFP+ cells expressed Sox3 and prominin (Supplementary Fig. 2F–M). Ki67 staining showed that the number of proliferating eGFP+ ventricular cells steadily decreased with developmental age (Fig. 1Q–T) indicating that these cells exited the cell cycle concomitant with the termination of dopamine neurogenesis. Collectively, our data demonstrate that cells of the Lmx1a-lineage maintain progenitor markers during late embryogenesis until E18.5. Thus, dopamine progenitor properties may extend beyond the main period of dopamine neurogenesis.
eGFP+ cells expressed dopamine receptors and were localized within a domain innervated by midbrain dopamine neurons
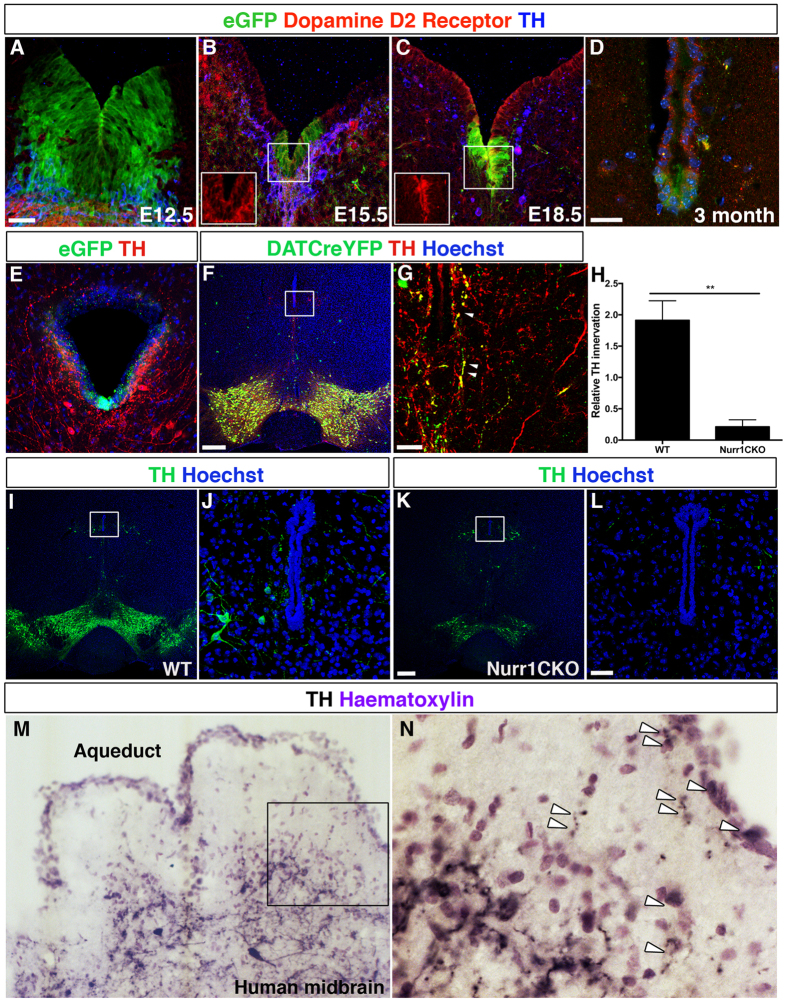
We previously showed that mature dopamine neurons are in close contact with neural stem cells in the adult salamander, a highly regenerative species in which the dopaminergic system can be completely restored following chemical ablation of dopamine neurons, and in which dopamine negatively regulates midbrain dopamine neurogenesis17. We set out to address to what extent such a regulatory mechanism might be evolutionary conserved. First, we found that eGFP+ cells expressed dopamine D2 receptors (D2R) both during embryogenesis and in the adult brain (Fig. 2A–D; orthogonal view of E18.5 shown in Supplementary Fig. 3A,B). Next, immunofluorescent analyses revealed that tyrosine hydroxylase (TH+) fibers projected onto the ventral aqueduct in the midbrain (Fig. 2E). High magnification confocal images and orthogonal sections indicated that TH fibers partially surrounded a proportion of the eGFP+ progenitor cells (Supplementary Fig. 3C,D). To investigate the origin of the TH+ fibers projecting to the midbrain aqueduct, we analysed DATCreYFP mice, in which YFP is exclusively expressed in dopamine transporter (DAT)-expressing midbrain dopamine neurons. We found that TH+ fibers surrounding the ventricular Lmx1a-expressing cells expressed YFP in these embryos, indicating their midbrain origin (Fig. 2F,G). Periaqueductal TH+ neurons, located just lateral to the ventral aqueduct, do not express DAT and for this reason did not express YFP in transgenic embryos. These periaqueductal TH+ neurons also extended processes in close proximity to the aqueduct (Fig. 2F,G). Histological analysis of conditional Nurr1DATCre (Nurr1CKO) mice, in which most dopamine neurons are lost20, showed a 7.9-fold reduction in TH+ fiber innervation (Fig. 2H–L) suggesting that a majority of fibers originated from midbrain dopamine neurons in the ventral midbrain area. As periaqueductal TH+ neurons are not lost in Nurr1-ablated mice (Fig. 2H,K,L), it is possible that some of the remaining fibers originated from this neuronal subpopulation. Furthermore, we found that eGFP+ cells lining the fourth ventricle in the hindbrain were also in close proximity to TH+ fibers originating from the locus coeruleus (Supplementary Fig. 3E,F). Notably, analysis showed that TH+ fibers were in close proximity with the outer layers of the midbrain aqueductal cells also in the human brain (Fig. 2M,N). Together these data indicated a possibility for dopamine regulation of Lmx1a-expressing ventricular cells.
Figure 2. Lmx1a-expressing ventricular cells expressed dopamine receptors and their domain was innervated by midbrain dopamine neurons.
There was a gradual increase in the fraction of eGFP+ cells expressing D2 receptor during embryonic development (A–C). eGFP+ cells expressed D2 receptors in adult animals (D). Immunofluorescent analysis of 3-month-old mice showed that the ventral aqueduct in the midbrain was innervated by tyrosine hydroxylase (TH+) fibers (E). Analysis of DATCrexROSA26YFP (DATCreYFP) mice demonstrated that TH+ fibers surrounding the ventricular eGFP+ cells were YFP+ and thus of a midbrain dopamine neuron origin (F, enlarged in G). Periaqueductal TH+ neurons, which lack DAT, also extended processes in close proximity to the aqueduct (F,G). Nurr1CKO mice, which lack SNc dopamine neurons, and have reduced numbers of VTA dopamine neurons, showed a 7.9-fold reduction in TH+ fiber innervation at 5 months of age (P = 0.0021, n = 3 + 4, unpaired Student’s t test) (H–L). Human midbrain sections demonstrated that TH+ fibers were in contact with the outer layers of the aqueductal cells (M,N). Scale bars: 50 μM in (A) (applicable to B,C), 50 μM in (D), 200 μM in (F), 50 μM in (G), 200 μM in (K) (applicable to I), 50 μM in (L) (applicable to E,J).
Haloperidol increased eGFP+ cell proliferation and neurogenesis
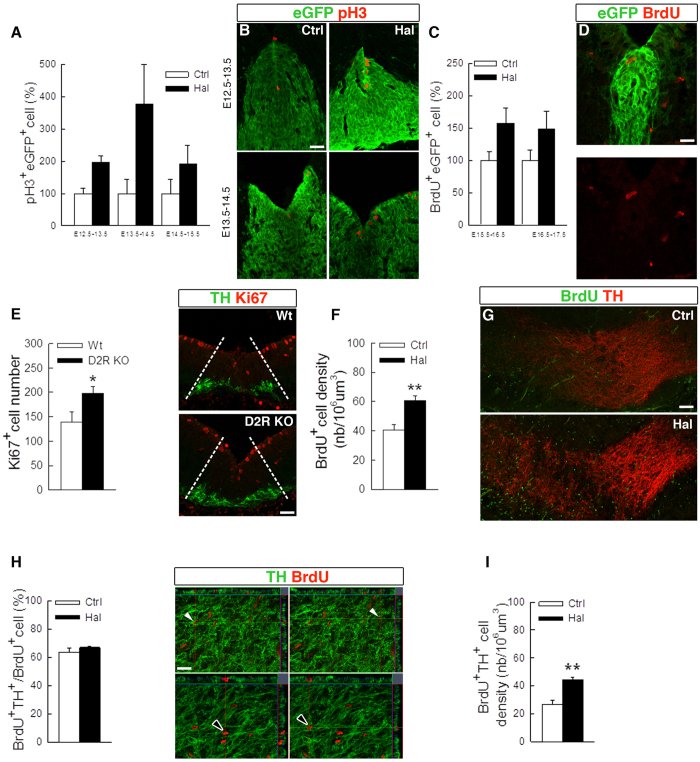
To examine if eGFP+ cells may be regulated by dopamine, we administered the broad-spectrum dopamine receptor antagonist, haloperidol, to pregnant Lmx1aeGFP/+ females. Animals received two daily injections of haloperidol or vehicle and were sacrificed the following day. We first carried out this series of experiments during E12.5–E14.5 and found that haloperidol administration led to an increased number of eGFP+ cells that were also positive for the mitotic marker pH3 (Fig. 3A,B). Interestingly, haloperidol induced a particularly strong increase in the number of pH3+ eGFP+ cells in the caudal portions of the midbrain (Supplementary Fig. 4A). This is in concordance with the more prominent TH innervation in caudal midbrain (Supplementary Fig. 5). We also administered the nucleotide analogue BrdU to animals via osmotic pumps, but positive cells could not be counted because virtually all cells had incorporated the analogue due to the intense proliferation during this active neurogenic period (data not shown). In contrast, when haloperidol was administered after the decline of normal dopamine neurogenesis, we could evaluate BrdU-incorporation and found a 1.5-fold increase of BrdU+ eGFP+ cells both at E16.5 and E17.5 (Fig. 3C,D). Similarly to the pH3-index, we noted a marked shift towards the caudal portions of the midbrain harbouring more BrdU+ eGFP+ cells (Supplementary Fig. 4B). We also found that haloperidol appeared to increase the proliferation of the eGFP-negative cells (Supplementary Fig. 4C). In agreement with these data we noted a marked increase in the number of cycling cells in the midbrain progenitor zone at E11.5 in gene targeted mice lacking both alleles of the D2R gene (Fig. 3E).
Figure 3. Haloperidol treatment increases the proliferation of eGFP+ cells and neurogenesis in vivo.
Haloperidol treatment increased the percentage of pH3+ eGFP+ ventricular cells (A,B, Haloperidol during E12.5–13.5, n = 8 (Ctrl), n = 9 (Hal); Haloperidol during E13.5–14.5, n = 11 (Ctrl), n = 9 (Hal); Haloperidol during E14.5–15.5, n = 13 (Ctrl), n = 10 (Hal), 2-way-ANOVA, treatment effect P = 0,003). Haloperidol treatment increased the percentage of BrdU+ eGFP+ cells at E16.5 in the ventral part of the third ventricle (C,D, Haloperidol during E15.5–16.5, n = 10 (Ctrl), n = 11 (Hal); Haloperidol during E16.5–17.5, n = 12 (Ctrl), n = 12 (Hal), 2-way-ANOVA, treatment effect P = 0,016). The number of Ki67+ ventricular cells in the ventral midbrain was increased in D2R KO mice at E11.5 compared to wild-type littermates (E, n = 5 (Wt), n = 5 (KO), P = 0.04, unpaired Student’s t test). Haloperidol treatment at E15.5–E17.5 increased the number of BrdU+ cells in the prospective substantia nigra and ventral tegmental area (F,G, n = 6 (Ctrl), n = 7 (Hal), P = 0.0014, unpaired Student’s t test). Haloperidol treatment at E15.5–E17.5 did not change the percentage of BrdU+ cells expressing TH in the prospective substantia nigra and ventral tegmental area (H, n = 4 (Ctrl), n = 4 (Hal), P = 0.32, unpaired Student’s t test). Orthogonal sections showing BrdU+ TH+ (top panel, arrow) and BrdU+ TH− (bottom panel, arrow) (I). Haloperidol treatment at E15.5–E17.5 increased the density of BrdU+ TH+ cells in the prospective substantia nigra and ventral tegmental area (P = 0.002, unpaired Student’s t test). Scale bars: 50 μM in (B,D,E), 100 μM in (G) and 20 μM in (H).
Next we tested whether haloperidol treatment could increase the proliferation of the eGFP+ cells and the production of new dopamine neurons after E14.5. We thus administered haloperidol and BrdU between E15.5 to E17.5, as described above, but this time evaluated the number BrdU+ cells in the prospective substantia nigra and ventral tegmental area instead of the ventricular zone. A 1.5-fold increase in the density of BrdU+ cells was detected in haloperidol treated animals (Fig. 3F,G). These BrdU+ cells were equally distributed between VTA and SNc (Supplementary Fig. 4D). Next we analysed the identity of BrdU+ cells and found that the percentage of BrdU+ cells expressing TH was unaffected, indicating that dopamine neuron differentiation was similar in both groups (Fig. 3H). Hence, the total number of newborn TH+ neurons showed a 1.5-fold increase in haloperidol compared to vehicle treated animals (Fig. 3I). These data collectively show that antagonizing dopamine signalling increases the proliferation of ventral aqueductal cells and ultimately the number of newborn TH+ neurons in the midbrain.
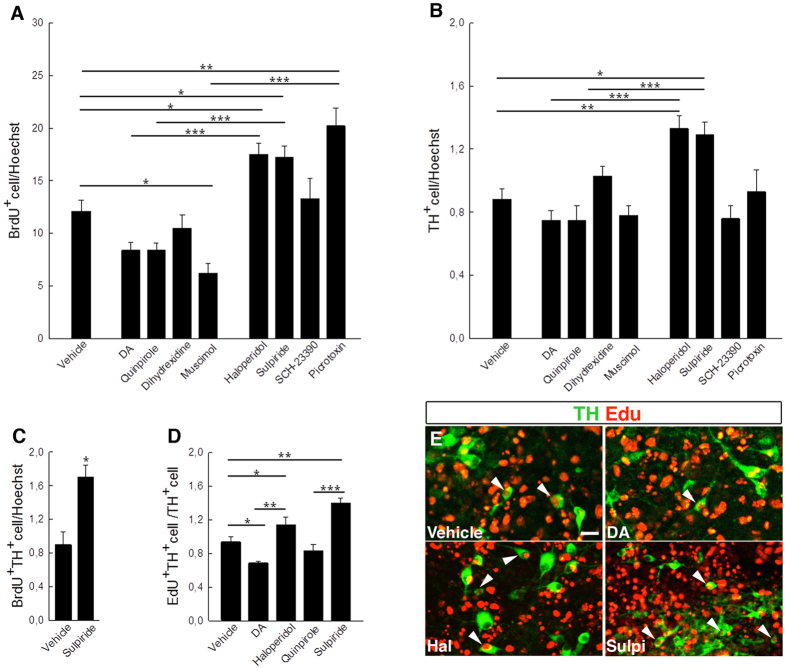
To corroborate these findings we tested the effect of various neurotransmitters on primary cultures of embryonic midbrain cells from animals undergoing dopamine neurogenesis. We took advantage of the fact that these cultures contained a mixture of precursor, including both dopamine and γ-aminobutyric acid (GABA) progenitors and used an array of neurotransmitter agonists and antagonists targeting dopamine and GABA receptors. In accordance with the in vivo observations we found that haloperidol treatment increased the proliferation of primary embryonic midbrain cells 1.5 fold as assayed by BrdU-incorporation (Fig. 4A). Moreover treatment with the dopamine 2 receptor (D2R) antagonist, sulpiride, but not the dopamine 1 receptor (D1R) antagonist SCH-23390, increased proliferation (Fig. 4A). In contrast, neither dopamine itself nor the dopamine receptor agonists quinpirole or dihydrexidine increased proliferation. We also found that the GABAA receptor agonist muscimol decreased proliferation, while the GABAA receptor blocker, picrotoxin, increased proliferation (Fig. 4A, Supplementary Fig. 6). Consistently with these results we observed an increased fraction of TH+ cells in the cultures upon haloperidol and sulpiride treatment, while neither the dopamine receptor agonists nor SCH-23390 had this effect (Fig. 4B). Furthermore, sulpiride also increased the fraction of BrdU+ cells expressing TH (Fig. 4C). Although we found that the GABAA receptor blocker, picrotoxin, increased proliferation, the fraction of TH+ cells did not increase in the cultures (Fig. 4B), indicating a selective role for dopamine receptor signalling in dopamine neurogenesis. To further validate these observations and to analyse if dopamine signalling could regulate the number of dopamine neurons generated from mouse embryonic stem cells (mESCs) we induced neurogenesis in mESC-derived midbrain cultures and found that dopamine decreased the fraction of EdU+/TH+ cells whereas both haloperidol and sulpiride increased it (Fig. 4D,E). These in vitro data are consistent with the results obtained in vivo and indicate that dopamine receptor antagonists may serve as a tool to enhance dopamine neurogenesis.
Figure 4. Dopamine receptor antagonists increased cell proliferation and neurogenesis in embryonic midbrain and mESC cultures.
The fraction of primary midbrain cells increased significantly upon treatment with the dopamine receptor (DR) antagonists haloperidol and sulpiride, but not with SCH-23390. Dopamine and the DR agonist quinpirole did not increase cell proliferation. The GABAA receptor agonist muscimol decreased proliferation, while the GABAA receptor blocker, picrotoxin, increased proliferation (A). Only the DR antagonists haloperidol and sulpiride increased the fraction of TH+ cells in primary midbrain cultures (B). The DR antagonist sulpiride increased the fraction of BrdU+ TH+ cells in primary midbrain cultures (C). Dopamine (DA) decreased, while haloperidol (Hal) and sulpiride (Sulpi) increased the fraction of EdU+ TH+/TH+ cells derived from mESCs (D,E). (A,B,D, *P < 0.05; **P < 0.01; ***P < 0.001, 1-way ANOVA and Newman-Keuls post hoc test, (C), unpaired Student’s t test). Scale bars: 20 μM.
Discussion
In this work we characterized ventral midbrain ventricular cells in the Lmx1a lineage through embryogenesis into adulthood in mice. These cells retain expression of nestin, Sox2, Sox3 and prominin beyond the period of normal dopamine neurogenesis during embryonic development and even in the adult, suggesting that they might have sustained progenitor cell properties. We further showed that proliferation and subsequent neurogenic conversion of these cells was enhanced by dopamine receptor antagonist treatment, an effect that extended beyond the bulk of normal embryonic dopamine neurogenesis.
Several previous studies have shown that neurotransmitters influence neurogenesis both during embryonic development and in the adult brain and spinal cord in several species, e.g. in mammals, salamanders and zebrafish21,22,23,24. The role of dopamine signalling in neurogenesis in the mammalian brain was addressed in different experimental settings, which collectively indicated region- and cell-type specific effects. Dopamine depletion has been seen to decrease cell proliferation in the dentate gyrus and subventricular zone in some of these studies25,26,27,28,29,30, while others documented increased cell proliferation13,31,32. Furthermore, observations in gerbils and in rats showed increased cell proliferation in dentate gyrus33, thus revealing that haloperidol treatment can increase the number of stem cells and the proliferation of transient-amplifying progenitors in the SVZ34.
Importantly, the present work is the first to address how modulating dopamine signalling influences cell proliferation and neurogenesis in the developing mammalian midbrain after the peak of dopamine neurogenesis. Although the effects in mice, as shown here, were rather modest, the results are in line with previous findings in aquatic salamanders, which demonstrated increased midbrain dopamine neurogenesis upon haloperidol treatment17. This indicates an evolutionary conserved regulatory mechanism by which dopamine suppresses the proliferation of dopamine progenitors and ultimately neurogenesis. Whether dopamine acts in a negative feedback-like manner in the mammalian midbrain remains to be proven. However, it is noteworthy that several reports identify regionally committed stem and progenitor cells that respond to varying levels of neurotransmitters. For example, the neurotransmitter (GABA) negatively controls proliferation of cells in the subventricular zone and in the rostral migratory stream35,36,37, and quiescent neural stem cells respond tonically to GABA by means of γ2-subunit-containing GABAA receptors in the adult dentate gyrus38. Hence, a plausible hypothesis is that a function of dopaminergic innervation of Lmx1a-expressing cells could be to maintain progenitors in quiescence, a state that can be transitioned into proliferation upon dopamine receptor antagonist treatment.
Dopamine neurogenesis in the adult salamander midbrain is critically dependent on reduced dopamine levels. Our observations raise the possibility that neurogenesis could be activated from resident Lmx1a-expressing cells by modulation of dopamine receptor signalling also in the postnatal mammalian brain. However, several significant ontogenetic changes occur as the region harbouring these cells transition beyond embryonic neurogenesis. The number of Lmx1a-expressing cells decreases, the expression level of Lmx1a in these cells is decreased and their processes become smaller and do not extend far beyond the aqueduct in the adult brain. As these processes likely support cell migration during development their disappearance may contribute to the diminution of neurogenesis in the adult midbrain. Nevertheless our findings warrant further experiments targeting the neurogenic potential of Lmx1a-expressing population in the adult midbrain.
Methods
Ethics statement
All the work involving animals or human subjects/tissues was carried out in accordance with the Code of Ethics of the World Medical Association (Declaration of Helsinki) and with national legislation and institutional guidelines. Animal procedures were approved by the regional animal ethics review board (Stockholms Norra Djurförsöksetiska nämnd) and the Florey Institute of Neuroscience and Mental Health animal ethics committee. Ethical approval for the use of human post mortem samples was obtained from the regional ethical review board in Stockholm, Sweden (Regionala Etikprövningsnämnden, Stockholm, EPN), ethical approval number 2012/111-31/1. All post mortem human tissues were obtained from the National Disease Research Interchange (NDRI, www.ndriresource.org) with the written informed consent from the donors or the next of kin.
Animals
All mouse lines were maintained on a C57Bl/6 background. The following transgenic mouse lines were utilized: Lmx1aeGFP/+ mice8; floxNurr1DATCre (Nurr1CKO) mice, generated by crossing floxed Nurr1 mice20 with mice carrying Cre inserted into the DAT gene locus39; and DATYFPxROSA26 mice, which were generated by crossing B6.129X1-Gt(ROSA)26Sortm1(EYFP)cos/J mice (Jackson laboratory #006148) with DAT-CreERT2 mice; D2R knock-out (D2R KO) mice and wild-type littermates40 (Jackson laboratory #003190).
BrdU delivery through subcutaneous pumps
The subcutaneous pump (Alzet, Model 2001) was filled with BrdU solution (0,08 mg/mL, Sigma), diluted in 60% DMSO and 40% dH2O.
Haloperidol in vivo administration
Haloperidol (Sigma) solution was prepared in acetic acid and saline and neutralized by addition of NaOH. For the proliferation experiments, pregnant dams received two daily injections of haloperidol (3 mg/kg) or vehicle at E12.5, E13.5, E14.5, E15.5 or E16.5, and were sacrificed the day after. For chase experiment performed from E15.5 to E18.5, animals were daily injected with vehicle or haloperidol (1.5 mg/kg).
Mouse tissue processing
Adult mice were anesthetized with avertin (tribromoethanol, Sigma) at 0.5–0.6 mg/g body weight i.p. and perfused intracardially with phosphate buffered saline (PBS) (Invitrogen) and subsequently 4% paraformaldehyde (Sigma). Brains were dissected, postfixed for 2–6 h in 4% PFA, cryoprotected in 20% and then 30% sucrose, sectioned (30–40 μm), serially collected and stored in antifreeze solution (30% glycerol, 30% ethoxyethanol, 40% PBS) at −20 °C until used. Pregnant dams were anesthetised by CO2 and sacrificed by cervical dislocation and embryos collected. For immunohistochemistry and in situ hybridization, collected embryos were postfixed in 4% PFA (1–3 h depending on the age of the embryo), cryoprotected in 20% and subsequently in 30% sucrose, embedded in OCT (TissueTek), sectioned at 12–20 μm, serially collected and stored at −80 °C.
Human tissue processing
The tissues were fixed in 4% PFA, sequentially placed through sucrose gradients (2%, 10%, 20% and 30%) for cryoprotection and sectioned on a freezing microtome (Microme HM430) at 40 μm thickness and serially collected. Tissues were subjected to antigen retrieval (0.01 M citric acid buffer, pH 6.0 for 20 min at 95 °C) and blocking of endogenous peroxidases (3% H2O2 in 50% methanol in PBS) prior to staining41.
Histological procedures for mouse and human tissues
For immunofluorescent staining of mouse tissues, sections were rinsed with PBS and incubated with blocking buffer (PBS, 5–10% normal donkey serum; NDS or normal goat serum; NGS (Jackson Laboratories), 0.1% Triton-X100) for 1 h prior to incubation with primary antibodies (Supplementary Table 1) in blocking buffer overnight at 4 °C. The tissue sections were subsequently incubated in Alexafluor secondary antibodies (1:500) for 1 h, and rinsed in PBS. Hoechst 33342 (4 μg/ml) was used for counterstaining and tissue sections were mounted onto slides in Mowiol 4–88 (Calbiochem). Confocal analysis was performed using a Zeiss LSM510/Meta Station (Thornwood, NY, http://www.zeiss.com). For identification of signal co-localization within a cell, optical thickness was kept to a minimum, and orthogonal reconstructions were obtained. Each experiment was performed in triplicate and all counts were done in a double-blind fashion.
Sections from human postmortem tissues were incubated with blocking buffer for 1 h. Subsequently, sections were incubated overnight at 4 °C using goat anti-tyrosine hydroxylase antibody (1:300, PelFreez). Sections were washed in PBS and incubated with a biotinylated secondary antibody (1:200; Vector Laboratories) for 1 h at room temperature, followed by incubation in streptavidin-biotin complex (Vectastain ABC kit Elite, Vector laboratories) for 1 h and visualized by incubation in 3,3′-diaminobenzidine solution (DAB, Vector Laboratories). Nuclei were counterstained using Myers hematoxylin (Histolab) for 1–2 min, rinsed in ddH20, quickly immersed in 70% ethanol containing 36 mM HCl to remove background staining, followed by another rinse in ddH20. Sections were subsequently dehydrated by sequential steps in increasing ethanol concentration accordingly; 25% ethanol (2 min), 70% ethanol (2 min), differentiator (75% ethanol containing 96 mM acetic acid) (1 min), 95% ethanol (2 min), 100% ethanol (2 min), xylene (2 × 2 min) and coverslipped using Mountex (Histolab)41. Brightfield images were captured using a Zeiss Axio Imager M1 Upright microscope. Control experiments were performed for all stainings by omitting either primary or secondary antibodies.
Quantification of cell numbers and intensity measurements in vivo
The signal intensity of tyrosine hydroxylase fiber innervation of the midbrain aqueduct was quantified using Image J (http://rsb.info.nih.gov/ij/). Values are displayed as relative intensity of signal in controls. Quantification of the number of Ki67+ eGFP+ cells, using immunofluorescent staining coupled with confocal analysis, was performed at E12.5 (1/5 sections counted), E15.5 (1/6 sections counted) and E18.5 (1/7.5 sections counted) (Fig. 1Q–T). The total number of Ki67+ cells in the ventral midbrain regions were retrieved by using the counts and taken into account the total number of sections, the average diameter of the cells (10 μm), by performing the Abercrombie correction, according to P = A • (M/L + M) • n, where P is the average number of nuclear points, A the number of nuclei identified/section, M is the thickness of the section, L the average length of the cell (in μm) and n the number of sections. TH innervation of the midbrain aqueduct in Nurr1CKO and wild-type littermate mice was conducted on confocal projection images derived from z-stacks of identical depth using the Image J software (https://imagej.nih.gov/ij/).
For quantification of Ki67+ cells in the D2R KO mice analysis was conducted at E11.5 (1/5 sections counted) (Fig. 3E). To evaluate the number ventral eGFP+ pH3+ cells or eGFP+ BrdU+ cells, a Zeiss upright microscope was used. The number of cells was quantified under 63X magnification with the optical fractionator method on a systematic random sampling of every fifth sections along the rostro-caudal axis. To evaluate the number of BrdU+ cells in the prospective VTA and SN, 20x images encompassing the entire midbrain were generated and cell numbers within the TH+ area counted (number of BrdU+ cells/TH+ area measured in μm3) in 3 or 4 sections. To analyse the percentage of BrdU+TH+/BrdU+ cells, 40x confocal images along the entire Z-axis (12 μm) using 1 μm intervals were counted. The co-labeled cells were analyzed with Imarys software.
In situ hybridization
In situ hybridization on sections was carried out essentially as previously described42 using a probe for Lmx1a6.
Mouse primary midbrain cultures and immunocytochemistry
Brains from E11.5 mice were obtained and the midbrain region was dissected, mechanically dissociated, plated on poly-d-lysine (150,000 cells/cm2), and grown in serum-free N2 media consisting of F12/DMEM (1:1) with insulin (10 ng/ml), apo-transferrin (100 μg/ml), putrescine (100 μM), progesterone (20 nM), selenium (30 nM), glucose (6 mg/ml), BSA (1 mg/ml) and FGF2 (10 ng/ml). Cells were treated for 3 days in vitro with either dopamine (10 μM, Sigma), haloperidol (1 μM, Sigma), quinpirole (10μM, Tocris Bioscience), dihydrexidine (10 μM, Tocris Bioscience), muscimol (30 μM, Sigma) sulpiride (10 μM, Tocris Bioscience), SCH-23390 (10 μM, Santa Cruz Biotechnology), picrotoxin (30 μM, Tocris Bioscience) or media only and then processed for immunocytochemistry. For BrdU analysis, cells were treated with BrdU 1 hour after plating. After a further 2 days in culture, cells were treated for 30 minutes with 2N HCl, and processed for immunocytochemistry.
Differentiation of mouse embryonic stem cells (mESCs)
R1 mESCs (Nagy’s lab, MSH, Toronto, Canada) were cultured and differentiated on PA6 stromal feeders (RCB1127, Riken BRC Cell Bank, Japan)43. Specifically, mESCs were plated at low density (100 cells/cm2) on a confluent layer of PA6 cells and were grown in Serum Replacement Medium with Noggin (300 ng/ml; R&D Systems) as previously described44. At day 5, 200 ng/ml Shh (R&D Systems) and 25 ng/mL Fgf8b (R&D Systems) were added to the medium. At day 8, the medium was switched to N2 medium containing Shh, Fgf8b, and Fgf2 (10 ng/ml, R&D Systems). At day 10 of differentiation the cells were pulsed with EdU (10 μM, Life Technologies). From day 11 of differentiation, Shh, Fgf8b and Fgf2 were removed from the N2 medium and replaced by BDNF (20 ng/ml, R&D Systems), GDNF (20 ng/ml, R&D Systems), and ascorbic acid (0.2 mM). Between day 8–15, cells were treated daily with either dopamine (10 μM), haloperidol (1 μM), quinpirole (10 μM), sulpiride (10 μM) or media only. At day 15, cells were fixed in 4% PFA and processed for immunocytochemistry as described below.
Immunohistochemistry of mESC and primary cultures and in vitro quantifications
Cells were incubated for 1 h in blocking buffer prior to overnight incubation with the primary antibodies (Supplementary Table 1) at 4 °C. The coverslips were subsequently incubated in secondary antibodies (1:500, Life Technologies or Jackson Immunoresearch) for 1 h, and rinsed in PBS. EdU determinations were done according to Clik-it EdU kit (Life Technologies). DAPI or Hoechst (Sigma-Aldrich) was used for counterstaining. Each condition was analyzed in duplicates for three independent experiments (n = 3). Specifically, for the primary cultures, cells positive for each marker were counted in 8 consecutive fields/well. For the mESC-derived cultures, the number of TH+ and TH+ EdU+ cell in each well were quantified in 15 fields with an Olympus FV1000 confocal microscope.
Statistical analysis
Prism (GraphPad software inc) and Statistica 8.0 (Statsoft) software were used for the statistical analyses. Experimental data was analysed using either student’s t test, 1-way ANOVA or 2-way ANOVA. All data are presented as mean ± SEM.
Additional Information
How to cite this article: Hedlund, E. et al. Dopamine Receptor Antagonists Enhance Proliferation and Neurogenesis of Midbrain Lmx1a-expressing Progenitors. Sci. Rep. 6, 26448; doi: 10.1038/srep26448 (2016).
Supplementary Material
Acknowledgments
We would like to thank D Berg for exploratory experiments in the initial phase of this study. This work was funded by grants from the Swedish Research Council, AFA Insurances, Cancerfonden and the European Research Council to A.S. The European Union, Seventh Framework Programme (mdDANeurodev), Swedish Strategic Research Foundation and Knut and Alice Wallenberg Foundation to T.P. The Swedish Research Council (VR projects: DBRM, 2008–2811, 2011–3116 and 2011–3318), Swedish Strategic Research Foundation (SRL program), European Commission (NeuroStemcell) and Karolinska Institutet (SFO Thematic Center in Stem cells and Regenerative Medicine) to E.A. Swedish Medical Research Council (2011–2651) and NEURO Sweden to E.H. L.B. was supported by a postdoc fellowship from Wenner-Gren Foundation, Sweden. C.P. was supported by a senior medical research fellowship from the Viertel charitable foundation, Australia. C.B. was supported by a Peter Doherty Fellowship from the National Health and Medical Research Council, Australia. Human post mortem tissues were kindly received from the National Disease Research Interchange (NDRI; www.ndriresource.org). We would like to thank Shanzheng Yang for help with the microphotographs of the midbrain cultures (E.A.).
Footnotes
Author Contributions E.H.: Conceived the study; performed the characterization of the Lmx1a-GFP animals, conducted the D2R expression and TH innervation experiments in mouse and man, analysed all data, and wrote the paper. L.B.: Conceived the study; performed the in vivo pharmacological studies, analysed all data, and wrote the paper. S.T.: Designed, performed, analysed and interpreted the primary midbrain culture experiments. C.S.: Designed, performed, analysed and interpreted the ESCs experiments. C.B. and C.P.: Performed and analysed in vivo D2R KO mouse experiments. Q.D.: Initiated the analysis of Lmx1a-GFP mice and performed some characterization. B.K.: Contributed to Nurr1CKO and DATCreYFP mouse experiments. J.E.: Conceived, analysed and, supervised characterization of the Lmx1a-GFP animals. E.A.: Conceived, analysed and supervised the mESC and midbrain progenitor culture experiments and contributed to writing the paper. T.P.: Conceived the study, analysed all data, supervised characterization of the Lmx1a-GFP animals, and wrote the paper. A.S.: Conceived the study, analysed all data, supervised the in vivo pharmacological studies, and wrote the paper. All authors reviewed and edited the manuscript.
References
- Grealish S. et al. Human ESC-derived dopamine neurons show similar preclinical efficacy and potency to fetal neurons when grafted in a rat model of Parkinson’s disease. Cell stem cell 15, 653–665, doi: 10.1016/j.stem.2014.09.017 (2014). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kriks S. et al. Dopamine neurons derived from human ES cells efficiently engraft in animal models of Parkinson’s disease. Nature 480, 547–551, doi: 10.1038/nature10648 (2011). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hedlund E. et al. Embryonic stem cell-derived Pitx3-enhanced green fluorescent protein midbrain dopamine neurons survive enrichment by fluorescence-activated cell sorting and function in an animal model of Parkinson’s disease. Stem cells 26, 1526–1536, doi: 10.1634/stemcells.2007-0996 (2008). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Steinbeck J. A. & Studer L. Moving stem cells to the clinic: potential and limitations for brain repair. Neuron 86, 187–206, doi: 10.1016/j.neuron.2015.03.002 (2015). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Arenas E., Denham M. & Villaescusa J. C. How to make a midbrain dopaminergic neuron. Development 142, 1918–1936, doi: 10.1242/dev.097394 (2015). [DOI] [PubMed] [Google Scholar]
- Andersson E. et al. Identification of intrinsic determinants of midbrain dopamine neurons. Cell 124, 393–405, doi: 10.1016/j.cell.2005.10.037 (2006). [DOI] [PubMed] [Google Scholar]
- Ono Y. et al. Differences in neurogenic potential in floor plate cells along an anteroposterior location: midbrain dopaminergic neurons originate from mesencephalic floor plate cells. Development 134, 3213–3225, doi: 10.1242/dev.02879 (2007). [DOI] [PubMed] [Google Scholar]
- Deng Q. et al. Specific and integrated roles of Lmx1a, Lmx1b and Phox2a in ventral midbrain development. Development 138, 3399–3408, doi: 10.1242/dev.065482 (2011). [DOI] [PubMed] [Google Scholar]
- Yan C. H., Levesque M., Claxton S., Johnson R. L. & Ang S. L. Lmx1a and lmx1b function cooperatively to regulate proliferation, specification, and differentiation of midbrain dopaminergic progenitors. The Journal of neuroscience: the official journal of the Society for Neuroscience 31, 12413–12425, doi: 10.1523/JNEUROSCI.1077-11.2011 (2011). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vitalis T., Cases O. & Parnavelas J. G. Development of the dopaminergic neurons in the rodent brainstem. Experimental neurology 191 Suppl 1, S104–112, doi: 10.1016/j.expneurol.2004.05.044 (2005). [DOI] [PubMed] [Google Scholar]
- Yang S. et al. Cxcl12/Cxcr4 signaling controls the migration and process orientation of A9-A10 dopaminergic neurons. Development 140, 4554–4564, doi: 10.1242/dev.098145 (2013). [DOI] [PubMed] [Google Scholar]
- Bayer S. A., Wills K. V., Triarhou L. C. & Ghetti B. Time of neuron origin and gradients of neurogenesis in midbrain dopaminergic neurons in the mouse. Experimental brain research 105, 191–199 (1995). [DOI] [PubMed] [Google Scholar]
- Aponso P. M., Faull R. L. & Connor B. Increased progenitor cell proliferation and astrogenesis in the partial progressive 6-hydroxydopamine model of Parkinson’s disease. Neuroscience 151, 1142–1153, doi: 10.1016/j.neuroscience.2007.11.036 (2008). [DOI] [PubMed] [Google Scholar]
- Kay J. N. & Blum M. Differential response of ventral midbrain and striatal progenitor cells to lesions of the nigrostriatal dopaminergic projection. Developmental neuroscience 22, 56–67, doi: 17427 (2000). [DOI] [PubMed] [Google Scholar]
- Zhao M. et al. Evidence for neurogenesis in the adult mammalian substantia nigra. Proceedings of the National Academy of Sciences of the United States of America 100, 7925–7930, doi: 10.1073/pnas.1131955100 (2003). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Frielingsdorf H., Schwarz K., Brundin P. & Mohapel P. No evidence for new dopaminergic neurons in the adult mammalian substantia nigra. Proceedings of the National Academy of Sciences of the United States of America 101, 10177–10182, doi: 10.1073/pnas.0401229101 (2004). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Berg D. A., Kirkham M., Wang H., Frisen J. & Simon A. Dopamine controls neurogenesis in the adult salamander midbrain in homeostasis and during regeneration of dopamine neurons. Cell stem cell 8, 426–433, doi: 10.1016/j.stem.2011.02.001 (2011). [DOI] [PubMed] [Google Scholar]
- Parish C. L., Beljajeva A., Arenas E. & Simon A. Midbrain dopaminergic neurogenesis and behavioural recovery in a salamander lesion-induced regeneration model. Development 134, 2881–2887, doi: 10.1242/dev.002329 (2007). [DOI] [PubMed] [Google Scholar]
- Berg D. A. et al. Efficient regeneration by activation of neurogenesis in homeostatically quiescent regions of the adult vertebrate brain. Development 137, 4127–4134, doi: 10.1242/dev.055541 (2010). [DOI] [PubMed] [Google Scholar]
- Kadkhodaei B. et al. Nurr1 is required for maintenance of maturing and adult midbrain dopamine neurons. The Journal of neuroscience: the official journal of the Society for Neuroscience 29, 15923–15932, doi: 10.1523/JNEUROSCI.3910-09.2009 (2009). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Berg D. A., Belnoue L., Song H. & Simon A. Neurotransmitter-mediated control of neurogenesis in the adult vertebrate brain. Development 140, 2548–2561, doi: 10.1242/dev.088005 (2013). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Young S. Z., Taylor M. M. & Bordey A. Neurotransmitters couple brain activity to subventricular zone neurogenesis. The European journal of neuroscience 33, 1123–1132, doi: 10.1111/j.1460-9568.2011.07611.x (2011). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Corns L. F. et al. Cholinergic Enhancement of Cell Proliferation in the Postnatal Neurogenic Niche of the Mammalian Spinal Cord. Stem cells 33, 2864–2876, doi: 10.1002/stem.2077 (2015). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Perez M. R. et al. Relationships between radial glial progenitors and 5-HT neurons in the paraventricular organ of adult zebrafish - potential effects of serotonin on adult neurogenesis. The European journal of neuroscience 38, 3292–3301, doi: 10.1111/ejn.12348 (2013). [DOI] [PubMed] [Google Scholar]
- Freundlieb N. et al. Dopaminergic substantia nigra neurons project topographically organized to the subventricular zone and stimulate precursor cell proliferation in aged primates. The Journal of neuroscience: the official journal of the Society for Neuroscience 26, 2321–2325, doi: 10.1523/JNEUROSCI.4859-05.2006 (2006). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Winner B. et al. Striatal deafferentation increases dopaminergic neurogenesis in the adult olfactory bulb. Experimental neurology 197, 113–121, doi: 10.1016/j.expneurol.2005.08.028 (2006). [DOI] [PubMed] [Google Scholar]
- Sui Y., Horne M. K. & Stanic D. Reduced proliferation in the adult mouse subventricular zone increases survival of olfactory bulb interneurons. Plos one 7, e31549, doi: 10.1371/journal.pone.0031549 (2012). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Suzuki K. et al. Destruction of dopaminergic neurons in the midbrain by 6-hydroxydopamine decreases hippocampal cell proliferation in rats: reversal by fluoxetine. Plos one 5, e9260, doi: 10.1371/journal.pone.0009260 (2010). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hoglinger G. U. et al. Dopamine depletion impairs precursor cell proliferation in Parkinson disease. Nature neuroscience 7, 726–735, doi: 10.1038/nn1265 (2004). [DOI] [PubMed] [Google Scholar]
- Baker S. A., Baker K. A. & Hagg T. Dopaminergic nigrostriatal projections regulate neural precursor proliferation in the adult mouse subventricular zone. The European journal of neuroscience 20, 575–579, doi: 10.1111/j.1460-9568.2004.03486.x (2004). [DOI] [PubMed] [Google Scholar]
- Peng J., Xie L., Jin K., Greenberg D. A. & Andersen J. K. Fibroblast growth factor 2 enhances striatal and nigral neurogenesis in the acute 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine model of Parkinson’s disease. Neuroscience 153, 664–670, doi: 10.1016/j.neuroscience.2008.02.063 (2008). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Park J. H. & Enikolopov G. Transient elevation of adult hippocampal neurogenesis after dopamine depletion. Experimental neurology 222, 267–276, doi: 10.1016/j.expneurol.2010.01.004 (2010). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dawirs R. R., Hildebrandt K. & Teuchert-Noodt G. Adult treatment with haloperidol increases dentate granule cell proliferation in the gerbil hippocampus. Journal of neural transmission 105, 317–327 (1998). [DOI] [PubMed] [Google Scholar]
- Kippin T. E., Kapur S. & van der Kooy D. Dopamine specifically inhibits forebrain neural stem cell proliferation, suggesting a novel effect of antipsychotic drugs. The Journal of neuroscience: the official journal of the Society for Neuroscience 25, 5815–5823, doi: 10.1523/JNEUROSCI.1120-05.2005 (2005). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu X., Wang Q., Haydar T. F. & Bordey A. Nonsynaptic GABA signaling in postnatal subventricular zone controls proliferation of GFAP-expressing progenitors. Nature neuroscience 8, 1179–1187, doi: 10.1038/nn1522 (2005). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nguyen L. et al. Autocrine/paracrine activation of the GABA(A) receptor inhibits the proliferation of neurogenic polysialylated neural cell adhesion molecule-positive (PSA-NCAM+) precursor cells from postnatal striatum. The Journal of neuroscience: the official journal of the Society for Neuroscience 23, 3278–3294 (2003). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fernando R. N. et al. Cell cycle restriction by histone H2AX limits proliferation of adult neural stem cells. Proceedings of the National Academy of Sciences of the United States of America 108, 5837–5842, doi: 10.1073/pnas.1014993108 (2011). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Song J. et al. Neuronal circuitry mechanism regulating adult quiescent neural stem-cell fate decision. Nature 489, 150–154, doi: 10.1038/nature11306 (2012). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ekstrand M. I. et al. Progressive parkinsonism in mice with respiratory-chain-deficient dopamine neurons. Proceedings of the National Academy of Sciences of the United States of America 104, 1325–1330, doi: 10.1073/pnas.0605208103 (2007). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kelly M. A. et al. Pituitary lactotroph hyperplasia and chronic hyperprolactinemia in dopamine D2 receptor-deficient mice. Neuron 19, 103–113 (1997). [DOI] [PubMed] [Google Scholar]
- Comley L. et al. Motor neurons with differential vulnerability to degeneration show distinct protein signatures in health and ALS. Neuroscience 291, 216–229, doi: 10.1016/j.neuroscience.2015.02.013 (2015). [DOI] [PubMed] [Google Scholar]
- Anderson T. R., Hedlund E. & Carpenter E. M. Differential Pax6 promoter activity and transcript expression during forebrain development. Mech Dev 114, 171–175 (2002). [DOI] [PubMed] [Google Scholar]
- Perrier A. L. et al. Derivation of midbrain dopamine neurons from human embryonic stem cells. Proceedings of the National Academy of Sciences of the United States of America 101, 12543–12548, doi: 10.1073/pnas.0404700101 (2004). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Andersson E. R. et al. Wnt5a cooperates with canonical Wnts to generate midbrain dopaminergic neurons in vivo and in stem cells. Proceedings of the National Academy of Sciences of the United States of America 110, E602–610, doi: 10.1073/pnas.1208524110 (2013). [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.