Abstract
After stone removal, accurate analysis of urinary stone composition is the most crucial laboratory diagnostic procedure for the treatment and recurrence prevention in the stone-forming patient. The most common techniques for routine analysis of stones are infrared spectroscopy, X-ray diffraction and chemical analysis. The aim of the present study was to assess the quality of urinary stone analysis of laboratories in Europe. Nine laboratories from eight European countries participated in six quality control surveys for urinary calculi analyses of the Reference Institute for Bioanalytics, Bonn, Germany, between 2010 and 2014. Each participant received the same blinded test samples for stone analysis. A total of 24 samples, comprising pure substances and mixtures of two or three components, were analysed. The evaluation of the quality of the laboratory in the present study was based on the attainment of 75% of the maximum total points, i.e. 99 points. The methods of stone analysis used were infrared spectroscopy (n = 7), chemical analysis (n = 1) and X-ray diffraction (n = 1). In the present study only 56% of the laboratories, four using infrared spectroscopy and one using X-ray diffraction, fulfilled the quality requirements. According to the current standard, chemical analysis is considered to be insufficient for stone analysis, whereas infrared spectroscopy or X-ray diffraction is mandatory. However, the poor results of infrared spectroscopy highlight the importance of equipment, reference spectra and qualification of the staff for an accurate analysis of stone composition. Regular quality control is essential in carrying out routine stone analysis.
Introduction
Prevalence and incidence of urolithiasis in industrialized countries have markedly increased over the past decades. The prevalence of urinary stone disease in the Unites States significantly increased from 5.2% in 1988 to 1994 to 8.8% in 2007 to 2010 [1,2]. In Japan, a rise in the prevalence from 4.0% to 5.4% was observed within 10 years [3]. In Germany, the prevalence of urolithiasis markedly increased from 4.0% to 4.7% and the incidence from 0.54% to 1.47% between 1979 and 2001 [4].
The recurrence rate of urinary stones is estimated to be up to 42% [4,5,6]. The high incidence of recurrence indicates that metaphylactic measures after stone removal are still inadequate. Patients at high risk of recurrent stone formation are those with infection stones, uric acid, urate (i.e. monoammonium urate, monopotassium urate and monosodium urate monohydrate), brushite and genetically determined stones (i.e. cystine, 2,8-dihydroxyadenine and xanthine stones) [7]. Depending on different risk factors, calcium oxalate stone disease is likewise characterised by a high frequency of recurrence [8]. For effective management of the stone-forming patient, accurate stone analysis is, therefore, an essential component of the diagnostic work-up and a prerequisite of metabolic evaluation [9].
According to the EAU guidelines (2015), all patients should have at least one stone analysed [7]. As the stone composition has significant therapeutic importance in the evaluation of patients, all urinary stones should be analysed [10]. Different techniques have been used for the compositional analysis of urinary stones, including X-ray diffraction (XD), infrared spectroscopy (IR) and chemical analysis (CA). Whereas IR is used for the examination of chemical molecular structures, XD is used for the determination of the crystalline structure of a substance. For the correct analysis of stone composition, IR and XD offer the highest degree of certainty. Due to poor results, wet chemical analysis of urinary stones is considered to be obsolete [10].
Urinary stones are often composed of more than one substance, which presents a difficulty in accurate assessment of the stone composition. A study performed in the United States found that commercial laboratories reliably recognised pure calculi, whereas variability in the reporting of mixtures was observed [11]. Analysis of more than 10.000 human urinary calculi revealed that only 7% of stones consisted of just one component [12], reflecting the clinical relevance of such a test variability.
Data regarding the accuracy of urinary stone analysis in Europe is lacking, stressing the importance of quality assessment of stone analysis in Europe. The application of analytical methods for stone analysis and the quality of the results were evaluated in nine European stone analysis laboratories participating in quality control surveys for urinary calculi analyses.
Materials and Methods
Nine urinary stone analysis laboratories from eight European countries (Denmark, France, Germany, Italy, Portugal, Switzerland, Turkey and United Kingdom) participated in six quality control surveys for urinary calculi analyses by the Reference Institute for Bioanalytics (RfB), Bonn, Germany, between 2010 and 2014. Of the nine laboratories, four had already participated in previous quality control surveys conducted by the RfB. Each participant received the same 24 blinded test samples for stone analysis to allow for direct comparison.
All laboratories advertised the use of their method for stone analysis. The certification of the analytical results by the RfB was based primarily on the correct qualitative proof of the substances present in each sample. A correct result required precise qualitative determination in mixtures at 10% gradations. For evaluation, the following scoring system was used:
| Substance present ≥ 80% | +4 points |
| Substance present between 30 and 70% | +3 points |
| Substance present ≤ 20% | +2 points |
| per false analysis (but the score for one sample cannot become negative) | -1 point |
Simplified analysis results (e.g. calcium oxalate instead of calcium oxalate monohydrate or calcium oxalate dihydrate; calcium phosphate instead of carbonate apatite, apatite, brushite or whitlockite; uric acid instead of uric acid dihydrate) scored with half of the possible points.
In order to get the certificate for each survey, at least 75% of the maximum points were needed. If the analysis of the laboratory yielded only simplified analyses results (e.g. calcium oxalate instead of calcium oxalate monohydrate or calcium oxalate dihydrate), then the laboratory would normally fail in achieving the required number of points. A total of 132 points could be maximally achieved by each laboratory after participation in the present six surveys. The evaluation of the quality of a laboratory in the present study was based on the attainment of 75% of the maximum total points, i.e. 99 points.
The samples used as test substances, single components and mixtures are shown in Table 1. The mixtures comprised two or three components of varying percentages by weight. While the majority of the samples were synthetic, the reference material for the remaining samples consisted of native urinary calculi. The use of synthetic products as test substances is necessary, as sufficient amounts of native urinary stone material are rarely available in the high quantities needed for the large number of participants in these surveys. The advantage of synthetic material is that these substances are standardised for purity and crystallinity, whereas urinary stones frequently contain mixtures and differ strongly in their crystallinity [13]. The test substances for the quality control surveys for urinary calculi analyses of the RfB were obtained from Dr. G. Schubert, Vivantes Klinikum im Friedrichshain, Institute of Laboratory Diagnostics, Berlin, Germany.
Table 1. Test substances for analysis.
| Sample | Proportion | Chemical name | Mineralogical name | Reference material |
|---|---|---|---|---|
| Pure | ||||
| 1 | 100% | Calcium carbonate (n = 2) | Calcite | Fluka |
| 2 | 100% | Calcium phosphate | Whitlockite | Fluka |
| 3 | 100% | Cholesterol (n = 2) | - | Aldrich |
| 4 | 100% | Monopotassium urate | - | Sigma |
| 5 | 100% | Monosodium urate monohydrate | - | Sigma |
| 6 | 100% | Sucrose | Cristal sugar | Cristal sugar |
| Two components | ||||
| 7 | 70% | Calcium hydrogen phosphate dihydrate | Brushite | Fluka |
| 30% | Calcium oxalate monohydrate | Whewellite | ||
| 8 | 50% | Calcium hydrogen phosphate dihydrate | Brushite | Fluka |
| 50% | Calcium oxalate monohydrate | Whewellite | ||
| 9 | 60% | Calcium hydrogen phosphate dihydrate | Brushite | Fluka |
| 40% | Apatite | Apatite | ||
| 10 | 70% | Calcium oxalate monohydrate | Whewellite | Native urinary calculus |
| 30% | Apatite | Apatite | ||
| 11 | 60% | Cystine | - | Fluka |
| 40% | Calcium oxalate monohydrate | Whewellite | ||
| 12 | 20% | Cystine | - | Fluka |
| 80% | Calcium oxalate monohydrate | Whewellite | ||
| 13 | 70% | Magnesium ammonium phosphate hexahydrate | Struvite | Native urinary calculus |
| 30% | Apatite (n = 2) | Apatite | ||
| 14 | 30% | Magnesium ammonium phosphate hexahydrate | Struvite | Native urinary calculus |
| 70% | Apatite | Apatite | ||
| 15 | 60% | Uric acid dihydrate | - | Native urinary calculus |
| 40% | Uric acid | Uricite | ||
| 16 | 30% | Uric acid dihydrate | - | Native urinary calculus |
| 70% | Uric acid | Uricite | ||
| 17 | 80% | Uric acid | Uricite | Native urinary calculus |
| 20% | Calcium oxalate monohydrate | Whewellite | ||
| 18 | 70% | Uric acid | Uricite | Fluka |
| 30% | Calcium oxalate monohydrate | Whewellite | ||
| 19 | 50% | Uric acid | Uricite | Native urinary calculus |
| 50% | Calcium oxalate monohydrate | Whewellite | ||
| Three components | ||||
| 20 | 50% | Calcium oxalate dihydrate | Weddellite | Native urinary calculus |
| 30% | Calcium oxalate monohydrate | Whewellite | ||
| 20% | Apatite | Apatite | ||
| 21 | 50% | Magnesium ammonium phosphate hexahydrate | Struvite | Native urinary calculus |
| 30% | Apatite | Apatite | ||
| 20% | Calcium oxalate monohydrate | Whewellite |
Results
The method of analysis most frequently used by participating laboratories was IR (n = 7). One laboratory reported the application of XD and another of CA. The comparison of the analytical results of the nine laboratories and the correct composition of the samples for stone analysis are presented in Table 2. The error rates of qualitative analysis for test substances of single components and components in mixtures are shown in Table 3.
Table 2. Comparison of analytical results of nine laboratories and the correct composition of samples for stone analysis.
| Lab A | Lab B | Lab C | Lab D | Lab E | Lab F | Lab G | Lab H | Lab I | |||
|---|---|---|---|---|---|---|---|---|---|---|---|
| XD | IR | IR | IR | IR | IR | IR | IR | CD | |||
| Sample | Substances | % | % | % | % | % | % | % | % | % | % |
| Survey 1 | |||||||||||
| A | Cholesterol | 100 | 100 | 100 | 100 | 100 | 100 | 100 | 0 | 0 | 0 |
| False analysis | - | - | - | - | - | - | - | 100 MP | 100 AF | 100 NI | |
| B | Uric acid dihydrate | 60 | 50 | 50 | 0 | 60 | 30 | 0 | 0 | 0 | 0 |
| Uricite | 40 | 50 | 50 | 80 | 40 | 60 | 0 | 0 | 100 | 100 | |
| False analysis | - | - | - | 20 WD | - | 10 WH | 100 UA | 100 UA | - | - | |
| C | Brushite | 50 | 60 | 70 | 50 | 60 | 30 | 50 | 100 | 50 | 0 |
| Whewellite | 50 | 40 | 30 | 50 | 40 | 50 | 0 | 0 | 0 | 40 | |
| False analysis | - | - | - | - | - | 20 WD | 50 CaOx | - | 50 WD | 30 WD | |
| False analysis | - | - | - | - | - | - | - | - | - | 30 AP | |
| D | Monopotassium urate | 100 | 100 | 100 | 100 | 100 | 100 | 0 | 0 | 0 | 0 |
| False analysis | - | - | - | - | - | - | 100 UA | 100 MU | 100 MU | 100 NI | |
| Survey 2 | |||||||||||
| A | Calcite | 100 | 100 | 100 | 100 | 100 | 100 | 100 | 100 | 0 | 0 |
| False analysis | - | - | - | - | - | - | - | - | 100 NI | 100 NI | |
| B | Uricite | 50 | 30 | 60 | 50 | 70 | 35 | 0 | 100 | 50 | 0 |
| Whewellite | 50 | 70 | 30 | 50 | 30 | 65 | 0 | 0 | 50 | 0 | |
| False analysis | - | - | 10 UD | - | - | - | 65 CaOx | - | - | 100 NI | |
| False analysis | - | - | - | - | - | - | 35 UA | - | - | - | |
| C | Cystine | 60 | 80 | 60 | 60 | 60 | 55 | 70 | 100 | 90 | 100 |
| Whewellite | 40 | 20 | 40 | 40 | 40 | 45 | 0 | 0 | 10 | 0 | |
| False analysis | - | - | - | - | - | - | 30 CaOx | - | - | - | |
| D | Whitlockite | 100 | 90 | 100 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| False analysis | - | 10 NI | - | 100 NI | 100 NI | 100 AF | 100 CP | 100 NI | 100 NI | 100 NI | |
| Survey 3 | |||||||||||
| A | Whewellite | 70 | 70 | 30 | 40 | 40 | 55 | 0 | 25 | 60 | 0 |
| Apatite | 30 | 30 | 70 | 60 | 60 | 40 | 65 | 75 | 40 | 0 | |
| False analysis | - | - | - | - | - | 5 PR | 35 CaOx | - | - | 100 NI | |
| B | Struvite | 70 | 80 | 80 | 60 | 80 | 50 | 0 | 35 | 40 | 0 |
| Apatite | 30 | 20 | 20 | 40 | 20 | 40 | 25 | 65 | 60 | 0 | |
| False analysis | - | - | - | - | - | 10 PR | 75 MP | - | - | 100 NI | |
| C | Weddellite | 50 | 40 | 40 | 40 | 40 | 65 | 0 | 0 | 85 | 0 |
| Whewellite | 30 | 40 | 30 | 30 | 40 | 20 | 0 | 50 | 0 | 0 | |
| Apatite | 20 | 20 | 30 | 30 | 20 | 15 | 10 | 50 | 15 | 0 | |
| False analysis | - | - | - | - | - | - | 90 CaOx | - | - | 100 NI | |
| D | Struvite | 50 | 20 | 20 | 30 | 60 | 20 | 0 | 100 | 10 | 0 |
| Apatite | 30 | 70 | 70 | 70 | 30 | 70 | 45 | 0 | 75 | 0 | |
| Whewellite | 20 | 10 | 10 | 0 | 0 | 10 | 0 | 0 | 10 | 0 | |
| False analysis | - | - | - | - | 10 WD | - | 55 MP | - | 5 WD | 100 NI | |
| Survey 4 | |||||||||||
| A | Whewellite | 80 | 70 | 60 | 80 | 70 | 90 | 0 | 0 | 100 | 0 |
| Cystine | 20 | 30 | 40 | 20 | 30 | 10 | 50 | 95 | 0 | 100 | |
| False analysis | - | - | - | - | - | - | 50 CaOx | 5 BR | - | - | |
| B | Cholesterol | 100 | 100 | 100 | 100 | 100 | 100 | 100 | 0 | 0 | 0 |
| False analysis | - | - | - | - | - | - | - | 100 CY | 100 NI | 100 NI | |
| C | Monosodium urate monohydrate | 100 | 100 | 100 | 100 | 100 | 100 | 100 | 100 | 100 | 0 |
| False analysis | - | - | - | - | - | - | - | - | - | 100 NI | |
| D | Brushite | 70 | 80 | 80 | 80 | 70 | 65 | 80 | 100 | 0 | 0 |
| Whewellite | 30 | 20 | 20 | 20 | 30 | 35 | 0 | 0 | 0 | 0 | |
| False analysis | - | - | - | - | - | - | 20 CaOx | - | 100 WL | 100 NI | |
| Survey 5 | |||||||||||
| A | Apatite | 70 | 60 | 30 | 50 | 40 | 80 | 60 | 100 | 80 | 0 |
| Struvite | 30 | 30 | 70 | 40 | 60 | 15 | 0 | 0 | 15 | 0 | |
| False analysis | - | 10 WH | - | 10 WH | - | 5 WH | 40 MP | - | 5 PR | 100 NI | |
| B | Uricite | 70 | 50 | 80 | 60 | 50 | 60 | 0 | 0 | 85 | 0 |
| Uric acid dihydrate | 30 | 50 | 20 | 40 | 50 | 40 | 0 | 100 | 15 | 0 | |
| False analysis | - | - | - | - | - | - | 100 UA | - | - | 100 NI | |
| C | Calcite | 100 | 100 | 100 | 100 | 100 | 100 | 100 | 100 | 0 | 0 |
| False analysis | - | - | - | - | - | - | - | - | 100 NI | 100 NI | |
| D | Uricite | 80 | 20 | 50 | 40 | 40 | 50 | 0 | 100 | 50 | 0 |
| Whewellite | 20 | 80 | 50 | 60 | 60 | 50 | 0 | 0 | 50 | 0 | |
| False analysis | - | - | - | - | - | - | 55 UA | - | - | 100 NI | |
| False analysis | - | - | - | - | - | - | 45 CaOx | - | - | - | |
| Survey 6 | |||||||||||
| A | Uricite | 70 | 70 | 60 | 70 | 60 | 60 | 0 | 0 | 80 | 0 |
| Whewellite | 30 | 30 | 40 | 30 | 40 | 40 | 0 | 0 | 20 | 0 | |
| False analysis | - | - | - | - | - | - | 45 CaOx | 100 NI | - | 100 NI | |
| False analysis | - | - | - | - | - | - | 55 UA | - | - | - | |
| B | Struvite | 70 | 50 | 70 | 70 | 70 | 55 | 0 | 35 | 40 | 0 |
| Apatite | 30 | 50 | 30 | 30 | 30 | 40 | 30 | 0 | 60 | 0 | |
| False analysis | - | - | - | - | - | 5 PR | 70 MP | 65 WL | - | 100 NI | |
| C | Brushite | 60 | 80 | 60 | 50 | 30 | 45 | 80 | 0 | 0 | 0 |
| Apatite | 40 | 20 | 40 | 50 | 70 | 55 | 20 | 65 | 30 | 0 | |
| False analysis | - | - | - | - | - | - | - | 35 ST | 70 WL | 100 NI | |
| D | Sucrose | 100 | 100 | 100 | 100 | 100 | 100 | 100 | 0 | 0 | 0 |
| False analysis | - | - | - | - | - | - | - | 100 NI | 100 NI | 100 NI |
AF: artifact; AP: apatite; BR: brushite; CaOx: calcium oxalate (simplified analysis result); CP: calcium phosphate (simplified analysis result); CY: cystine; MP: magnesium ammonium phosphate (simplified analysis result); MU: monoammonium urate; NI: non-identifiable; PR: protein; ST: struvite; UA: uric acid (simplified analysis result); UD: uric acid dihydrate; WD: weddellite; WH: whewellite; WL: whitlockite
Table 3. Error rates of qualitative analysis for test substances.
| Substances | Incorrect qualitative analysis (%) | Total | ||
|---|---|---|---|---|
| IR (n = 7) | CA (n = 1) | XD (n = 1) | ||
| Substances in mixtures | ||||
| Calcium oxalate monohydrate (whewellite) | 32.9% (23/70) | 90% (9/10) | 0% (0/10) | 35.6% (32/90) |
| Calcium oxalate dihydrate (weddellite) | 28.6% (2/7) | 100% (1/1) | 0% (0/1) | 33.3% (3/9) |
| Apatite | 4.1% (2/49) | 100% (7/7) | 0% (0/7) | 14.3% (9/63) |
| Calcium hydrogen phosphate dihydrate (brushite) | 14.3% (3/21) | 100% (3/3) | 0% (0/3) | 22.2% (6/27) |
| Magnesium ammonium phosphate hexahydrate (struvite) | 17.9% (5/28) | 100% (4/4) | 0% (0/4) | 25% (9/36) |
| Uric acid | 22.9% (8/35) | 80% (4/5) | 0% (0/5) | 26.7% (12/45) |
| Uric acid dihydrate | 35.7% (5/14) | 100% (2/2) | 0% (0/2) | 38.9% (7/18) |
| Cystine | 7.1% (1/14) | 0% (0/2) | 0% (0/2) | 6.3% (1/16) |
| Pure substances | ||||
| Calcium carbonate (calcite) | 14.3% (2/14) | 100% (2/2) | 0% (0/2) | 22.2% (4/18) |
| Calcium phosphate (whitlockite) | 85.7% (6/7) | 100% (1/1) | 0% (0/1) | 77.8% (7/9) |
| Cholesterol | 28.6 (4/14) | 100% (2/2) | 0% (0/2) | 33.3% (6/18) |
| Monopotassium urate | 42.9 (3/7) | 100% (1/1) | 0% (0/1) | 44.4% (4/9) |
| Monosodium urate monohydrate | 0% (0/7) | 100% (1/1) | 0% (0/1) | 11.1% (1/9) |
| Sucrose | 28.6% (2/7) | 100% (1/1) | 0% (0/1) | 33.3% (3/9) |
The error rate of up to 100% was extremely high for the laboratory using CA. Regarding the most common stone components (i.e. whewellite, uric acid and apatite), the error rate for CA was between 80 and 100%. IR showed incorrect results in up to 86% of the analyses. Although XD provided correct detection of each substance, the laboratory did not receive the maximum total points of 132 due to 2 false-positive analyses of substances.
Whewellite, one of the most common stone components, was either not found or wrongly identified in 36% (32 of 90) of analyses. The error rates were 33% (23 of 70) for IR, 90% for CA and 0% for XD.
Seventy-three percent of all participants reliably recognised uric acid. However, only 61% (11 of 18) of analyses correctly identified the component uric acid dihydrate. The error rates in the case of uric acid and uric acid dihydrate when using IR were 23% and 36%, respectively. The laboratory using CA did not identify uric acid dihydrate, whereas the laboratory with XD correctly reported uric acid and uric acid dihydrate.
Brushite was reliably recognized in nearly 80% and apatite in 86% of analyses. Whereas the laboratory using CA was unable to identify brushite and apatite, XD correctly provided detection of both substances in mixtures. Error rates regarding analyses of brushite and apatite were 14% and 4%, respectively, for IR.
The single components comprised the biliary calculus substance cholesterol, the rare stone components whitlockite, monopotassium urate, monosodium urate monohydrate and calcium carbonate and the artifact sucrose, which occasionally occurs in stone analysis laboratories. Error rates of up to nearly 80% of all analyses were recorded. Whereas the laboratory using XD correctly determined each of these substances, the participant using CA was unable to identify any of these components. The error rates for IR were between 0% and 86%, depending on the substance.
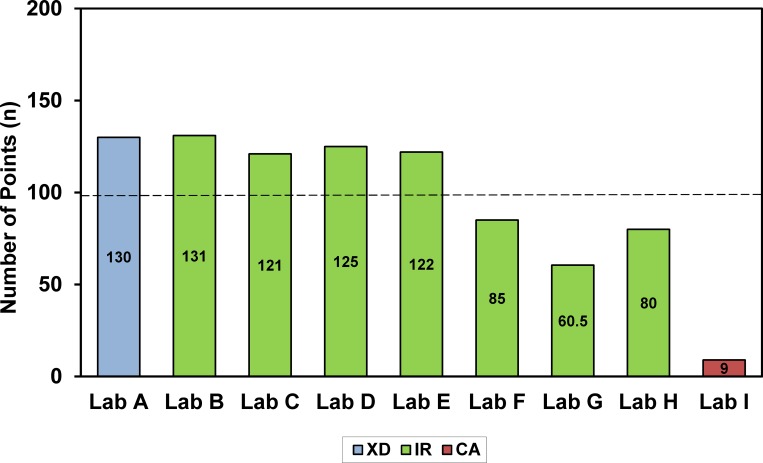
In the current study, only 56% (5 of 9) of the participants fulfilled the quality requirements of at least 99 points (Fig 1).
Fig 1. Evaluation of analytical results of laboratories (maximum: 132 points; minimum: 99 points).
Discussion
Appropriate and thorough analysis of urinary stone composition is the most crucial laboratory diagnostic procedure and the basis for dietary and medical treatment of the stone-forming patient [7,9,14]. Compositional stone analysis should, therefore, be an integral part of the metabolic evaluation of patients with urolithiasis [15]. Incorrect analysis or failure to identify a stone substance may result in inadequate therapy.
Two previous studies based on the analysis of more than 50,000 and 200,000 urinary stones, respectively, revealed a significant increase in the occurrence of brushite stones during recent years [16,17]. Brushite stones are known to grow rapidly with a correspondingly very high recurrence rate. Effective treatment is therefore highly important in brushite stone disease [18]. Additionally, the composition of a stone can be an important factor in its fragility. Due to their hardness, brushite and whewellite (calcium oxalate monohydrate) have been shown to respond poorly to disintegration by Extracorporeal Shock Wave Lithotripsy (ESWL) [7,19]. Whewellite is among the most common urinary stone substances. The correct qualitative determination and differentiation of whewellite and brushite is therefore essential for a selective approach to stone therapy. The participant using CA was unable to identify whewellite in 90% of the analyses, but incorrect results were also found in one third of analyses (33%) using IR. While the laboratory with CA could not detect brushite, IR showed incorrect results in 14% of the analyses. The participant with XD correctly reported brushite and whewellite in each sample.
Compared to brushite, the mixture of uric acid and uric acid dihydrate is relatively common. A recent study based on the analysis of 43,545 stones found that uric acid stone composition increased markedly in both sexes above the age of 50 years [20]. The differentiation between uric acid and uric acid dihydrate can provide valuable information regarding the pathogenesis of the stone, because uric acid dihydrate is formed in very acidic urine [9]. While more than 70% of all analyses reliably recorded uric acid, only 61% of laboratories correctly detected the component uric acid dihydrate. CA was unable to identify uric acid dihydrate; however, 5 of 14 (36%) of analyses using IR also failed.
Among the pure substances, only monosodium urate monohydrate, a rare urinary stone component, was correctly determined by IR. On the contrary, laboratories using IR were unable to identify whitlockite (calcium phosphate) in 86% of the analyses. Whereas XD showed correct analyses of all pure substances, the error rate of CA was 100%.
It is not only undetected urinary stone components that may lead to inaccurate diagnosis and subsequent inadequate recurrence prevention, but also false-positive analysis of substances that are not present in the stone. For instance, incorrect analysis specifying the urinary calculus substance cystine or magnesium ammonium phosphate instead of cholesterol is more detrimental to the patient than the statement “not identifiable”.
Several studies revealed that CA exhibits very high error rates of up to 94%, depending on the stone component [10,21,22]. Our study in nine European laboratories confirms the high error rates with the application of CA in the analysis of pure substances as well as mixtures. Regarding the most common stone components (i.e. whewellite, uric acid and apatite), the error rate for CA was between 80 and 100%. According to the current standard, CA is considered to be insufficient for stone analysis, whereas IR or XD is mandatory [7,10]. Among the different methods for urinary stone analysis, chemical analysis has been traditionally used most widely due to its low cost. While XD requires more expensive equipment than IR, the advantage of IR is the moderate cost.
Surprisingly, the evaluation of the results of our study revealed high error rates for both pure substances and mixtures also in laboratories using IR, which is in contrast to previous findings [10,21,22]. Of the 7 laboratories that used IR, just 4 (57%) fulfilled the quality requirements. Except for uric acid dihydrate, which is difficult to identify by IR in a mixture with uric acid, all other pure substances and mixtures would have been expected to be correctly detected. The results of our study showed that high performance can also be achieved by IR, but it requires the use of sophisticated equipment and qualified personnel. Most computer programs for the automated evaluation of IR spectra and XD diagrams are considered to provide unreliable component identification of mixtures [23]. The quality of the library of reference spectra is regarded as the major factor contributing to the reliability of the evaluation software [13]. Evaluation of the analysis carried out using IR and XD should always be checked by qualified personnel [10]. The results underline previous findings that the equipment, reference spectra and trained staff are indispensable preconditions for an accurate application of IR [10,13].
Conclusions
Targeted recurrence prevention requires reliable information on stone composition. For laboratories that are not able to offer sophisticated analysis techniques and interpretative expertise, referral or centralisation of stone analysis seems to be the only recommendable approach.
In order to ensure reliable results, measures for quality control of urinary stone analysis are required. Among the five laboratories in Europe that fulfilled the quality requirements, four of these laboratories had already participated in previous quality control surveys for urinary calculi analyses. It can be concluded that regular quality control is essential in carrying out routine stone analysis
Acknowledgments
The full membership list is available in https://uroweb.org/section/eulis/board/.
Data Availability
All relevant data are within the paper.
Funding Statement
The authors received no specific funding for this work.
References
- 1.Stamatelou KK, Francis ME, Jones CA, Nyberg LM, Curhan GC. Time trends in reported prevalence of kidney stones in the United States: 1976–1994. Kidney Int. 2003; 63:1817–1823. [DOI] [PubMed] [Google Scholar]
- 2.Scales CD, Smith AC, Hanley JM, Saigal CS. Prevalence of kidney stones in the United States. Eur Urol. 2012; 62: 160–165. 10.1016/j.eururo.2012.03.052 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Yoshida O, Okada Y. Epidemiology of urolithiasis in Japan: a chronological and geographical study. Urol Int. 1990; 45: 104–111. [DOI] [PubMed] [Google Scholar]
- 4.Hesse A, Brändle E, Wilbert D, Köhrmann KU, Alken P. Study on the prevalence and incidence of urolithiasis in Germany comparing the years 1979 vs. 2000. Eur Urol. 2003; 44: 709–713. [DOI] [PubMed] [Google Scholar]
- 5.Ahlstrand C, Tiselius HG. Recurrences during a 10-year follow-up after first renal stone episode. Urol Res. 1990; 18: 397–399. [DOI] [PubMed] [Google Scholar]
- 6.Trinchieri A, Ostini F, Nespoli R, Rovera F, Montanari E, Zanetti G. A prospective study of recurrence rate and risk factors for recurrence after a first renal stone. J Urol. 1999; 162: 27–30. [DOI] [PubMed] [Google Scholar]
- 7.Skolarikos A, Straub M, Knoll T, Sarica K, Seitz C, Petrik A, et al. Metabolic evaluation and recurrence prevention for urinary stone patients: EAU Guidelines. Eur Urol. 2015; 67: 750–763. 10.1016/j.eururo.2014.10.029 [DOI] [PubMed] [Google Scholar]
- 8.Siener R, Glatz S, Nicolay C, Hesse A. Prospective study on the efficacy of a selective treatment and risk factors for relapse in recurrent calcium oxalate stone patients. Eur Urol. 2003; 44: 467–474. [DOI] [PubMed] [Google Scholar]
- 9.Hesse A, Tiselius HG, Siener R, Hoppe B. Urinary stones: Diagnosis, treatment, and prevention of recurrence. 3rd revised and enlarged edition, Basel, Karger; 2009.
- 10.Hesse A, Kruse R, Geilenkeuser WJ, Schmidt M. Quality control in urinary stone analysis: results of 44 ring trials (1980–2001). Clin Chem Lab Med. 2005; 43: 298–303. [DOI] [PubMed] [Google Scholar]
- 11.Krambeck AE, Khan NF, Jackson ME, Lingeman JE, McAteer JA, Williams JC. Inaccurate reporting of mineral composition by commercial stone analysis laboratories: implications for infection and metabolic stones. J Urol. 2010; 184: 1543–1549. 10.1016/j.juro.2010.05.089 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Daudon M, Donsimoni R, Hennequin C, Fellahi S, Le Moel G, Paris M, et al. Sex and age-related composition of 10617 calculi analyzed by infrared spectroscopy. Urol Res. 1995; 23: 319–326. [DOI] [PubMed] [Google Scholar]
- 13.Hesse A, Sanders G. Atlas of infrared spectra for the analysis of urinary concrements. Stuttgart, Georg Thieme; 1988.
- 14.Smith CL. Renal stone analysis: is there any clinical value? Curr Opinion Nephrol Hypertens. 1998; 7: 703–709. [DOI] [PubMed] [Google Scholar]
- 15.Kourambas J, Aslan P, Teh CL, Mathias BJ, Preminger GM. Role of stone analysis in metabolic evaluation and medical treatment of nephrolithiasis. J Endourol. 2001; 15: 181–186. [DOI] [PubMed] [Google Scholar]
- 16.Daudon M, Bouzidi H, Bazin D. Composition and morphology of phosphate stones and their relation with etiology. Urol Res. 2010; 38: 459–467. 10.1007/s00240-010-0320-3 [DOI] [PubMed] [Google Scholar]
- 17.Knoll T, Schubert AB, Fahlenkamp D, Leusmann DB, Wendt-Nordahl G, Schubert G. Urolithiasis through the ages: data on more than 200,000 urinary stone analyses. J Urol. 2011; 185: 1304–1311. 10.1016/j.juro.2010.11.073 [DOI] [PubMed] [Google Scholar]
- 18.Siener R, Netzer L, Hesse A. Determinants of brushite stone formation: A case-control study. PLOS ONE 2013; 8(11): e78996 10.1371/journal.pone.0078996 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Dretler SP. Stone fragility—a new therapeutic distinction. J Urol. 1988; 139: 1124–1127. [DOI] [PubMed] [Google Scholar]
- 20.Lieske JC, Rule AD, Krambeck AE, Williams JC, Bergstralh EJ, Mehta RA, et al. Stone composition as a function of age and sex. Clin J Am Soc Nephrol. 2014; 9: 2141–2146. 10.2215/CJN.05660614 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Kasidas GP, Samuell CT, Weir TB. Renal stone analysis: why and how? Ann Clin Biochem. 2004; 41: 91–97. [DOI] [PubMed] [Google Scholar]
- 22.Schubert G. Stone analysis. Urol Res. 2006; 34: 146–150. [DOI] [PubMed] [Google Scholar]
- 23.Hesse A, Gergeleit M, Schüller P, Möller K. Analysis of urinary stones by computerized infrared spectroscopy. J Clin Chem Clin Biochem. 1989; 27: 639–642. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
All relevant data are within the paper.