Significance
Conventional thought holds that the stable isotopic composition (O and H) of water inside the cells of microorganisms and most aquatic macroorganisms is identical to that of the surrounding water. This assumption is widely applied in studies of environmental reconstruction. Results presented here, based on techniques of multilabeled water isotope probing and measurement of 18O/16O ratios in PO4 moieties of intracellular biomolecules, which allows direct sampling of intracellular environments, demonstrate a significant metabolic water component in intracellular water that is consistent across multiple strains of bacteria when grown under similar conditions. The intracellular water probe presented here, based on PO4 δ18O in DNA/biomass, may be extended to other biomolecules and organisms.
Keywords: metabolic water, isotope probing, phosphate oxygen isotopes
Abstract
Knowledge of the relative contributions of different water sources to intracellular fluids and body water is important for many fields of study, ranging from animal physiology to paleoclimate. The intracellular fluid environment of cells is challenging to study due to the difficulties of accessing and sampling the contents of intact cells. Previous studies of multicelled organisms, mostly mammals, have estimated body water composition—including metabolic water produced as a byproduct of metabolism—based on indirect measurements of fluids averaged over the whole organism (e.g., blood) combined with modeling calculations. In microbial cells and aquatic organisms, metabolic water is not generally considered to be a significant component of intracellular water, due to the assumed unimpeded diffusion of water across cell membranes. Here we show that the 18O/16O ratio of PO4 in intracellular biomolecules (e.g., DNA) directly reflects the O isotopic composition of intracellular water and thus may serve as a probe allowing direct sampling of the intracellular environment. We present two independent lines of evidence showing a significant contribution of metabolic water to the intracellular water of three environmentally diverse strains of bacteria. Our results indicate that ∼30–40% of O in PO4 comprising DNA/biomass in early stationary phase cells is derived from metabolic water, which bolsters previous results and also further suggests a constant metabolic water value for cells grown under similar conditions. These results suggest that previous studies assuming identical isotopic compositions for intracellular/extracellular water may need to be reconsidered.
Metabolic water, more precisely defined as an isotopically distinct flux of O (and H) produced during metabolism (1), has been studied extensively as an alternative water source contributing to body water in animals, such as desert mammals, insects, and migrating birds (2–6), but does not easily lend itself to direct measurement. In recent years, interest in metabolic water has been extended to its oxygen isotopic composition (18O/16O ratio or δ18O value) and contribution to body water because this information is crucial to the interpretation of biomineral [e.g., carbonate—CaCO3—and phosphate—Ca3(PO4)2] oxygen isotopic compositions used heavily in paleoclimate/paleohydrological research (7–14). This includes biomineral shells of aquatic marine organisms that are preserved in the geologic record and used to infer Earth’s climate history (15–19).
A core assumption of applications of biomineral oxygen isotopic compositions to infer environmental conditions is that the 18O:16O ratio is controlled by exchange of oxygen isotopes between oxyanions comprising the biomineral and ambient water in bodily fluids (i.e., body water) (9, 20–24). In multicellular eukaryotic organisms, body water includes all water found in various body compartments (e.g., intravascular/intercellular) and bodily fluids (e.g., blood plasma, urine, breath vapor), including water produced by metabolism, and is averaged over the entire organism (25, 26). Blood is the largest reservoir of body water in mammals. Accordingly, most previous studies have been based on measurements of total body water in blood, in urine, or in breath CO2 that has exchanged and presumably equilibrated with body water, and, thus, not on direct measurement of intracellular water.
A strong linear relationship has been observed between δ18O values of body water determined from mammal blood and δ18O values of extracellular (ingested) water, which is equivalent to local meteoric water (1, 27–30). A contribution of metabolic water to body water can be detected using this linear relation based on deviation of the slope from a value of 1 (7, 8, 28, 30–32). Based on slopes and modeling calculations, the percentage of metabolic water has been determined to vary between 7% and 56% among different mammal species (7, 8, 30–34).
In contrast to macroorganisms and mammals, microorganisms are primarily unicellular (e.g., bacteria and archaea), and, thus, body water is equivalent to intracellular water, which has traditionally been assumed to be identical to water in the surrounding extracellular medium (35), without consideration of a metabolic water component.
Here we present two independent lines of evidence for a significant contribution of metabolic water to the intracellular water pool of bacterial cells, based on a new approach for detecting the contribution of metabolic water by direct sampling of the intracellular environment through measurement of the 18O:16O ratio of PO4 (i.e., δ18OPO4) in intracellular molecules (e.g., DNA) and also PO4 in total biomass. Our findings are consistent with recent results of Kreuzer-Martin et al. (27, 36) indicating a metabolic water contribution to the intracellular water of Escherichia coli cells of as much as 70% during the log phase of growth and up to 27% for cells in the stationary phase. Knowledge of the amount and also the isotopic composition of a metabolic water component of body/intracellular water is important to studies of cell physiology and metabolism in which the 18O:16O ratio of both water and oxyanions such as CO2/CO3 and PO4 are used to track metabolic processes and reaction pathways.
Intracellular Water Isotopic Composition
Water within the cytoplasm of cells is generally assumed to diffuse freely across the cell membrane and thus have an identical 18O:16O ratio to ambient water outside of cells. The same assumption holds for body water of aquatic organisms such that ambient extracellular water oxygen isotopic compositions are used to determine apparent fractionations between body water and biomineral/biomass components that contain oxygen (e.g., refs. 19 and 21–24).
For nonaquatic, terrestrial organisms (e.g., mammals) that are relatively closed systems with respect to inflow/outflow of ambient environmental water (e.g., no free diffusion of water across the outer membrane), a significant component of metabolic water has been observed in the intracellular water pool (e.g., mammals), with the metabolic water contribution increasing with increased metabolic rates and decreased body size (6, 30, 34, 37).
The 18O:16O ratio in water varies with chemical reaction rates/pathways and also physical processes such as evaporation, precipitation, adsorption, and diffusion (38). Water 18O:16O ratios (henceforth expressed as δ18OWATER values) also vary due to the exchange or replacement of oxygen atoms (i.e., isotopes) in water with oxygen in other molecules. In the case of exchange of oxygen between water and the oxyanions that are precipitated within/by cells as biominerals (e.g., CO32−, PO43−, and SO42−), 18O:16O ratios often vary according to temperature, which has led to construction of a number a of oxyanion−water−temperature equations or “paleothermometers” that are widely used in the study of Earth’s climate (21, 23, 39–41). A primary assumption underlying the use of these oxygen isotope paleothermometers to reconstruct environmental temperatures is that oxyanion 18O:16O ratios reflect equilibrium exchange with ambient environmental water that is further assumed to be identical in oxygen isotopic composition to the intracellular water.
Approach: Oxygen Isotopic Composition of Water Imprinted onto Intracellular PO4
Several recent studies in our group using stable isotope probing to elucidate intracellular phosphoenzyme reaction mechanisms and attendant isotope effects led to the hypothesis underlying the present work, which is as follows: PO4 can undergo complete and rapid exchange of all four O atoms with intracellular water due to the action of the ubiquitous, intracellular enzyme inorganic pyrophosphatase (PPase) (Fig. 1 and Fig. S1) (42, 43). Previous experiments using the purified cell-free PPase enzyme, inorganic PO4, and water firmly demonstrate that the oxygen in dissolved inorganic PO4 (DIP) is rapidly and completely (100%) exchanged with oxygen in ambient water in the medium (Fig. S1), such that a plot of δ18OWATER vs. δ18OPO4 gives a slope of 1 reflecting that all four oxygens in PO4 are derived from ambient water in the medium (42). Thus, cellular compounds and biomass that are synthesized from the intracellular dissolved PO4 pool should also carry the isotopic signature imprinted by PPase (Fig. 1).
Fig. 1.
Schematic sketch of PPase-catalyzed O exchange between intracellular water and the intracellular DIP pool, and its transfer to DNA/biomass.
Fig. S1.
Approach of PO4−water fractionations to steady-state, equilibrium values from opposite directions (i.e., in different isotopically labeled waters) due to PO4−water exchange catalyzed by inorganic pyrophosphatase (PPase) at 27 °C. Reprinted from ref. 43.
In the present study, we tested the above-mentioned hypothesis, shown schematically in Fig. 1, by performing a series of controlled culture experiments to look for the imprinting of water 18O:16O ratios onto the intracellular dissolved PO4 pool due to the action of intracellular PPase or other intracellular enzymes with similar isotope effects, and its subsequent transference into cell biomass (e.g., DNA) that is synthesized from the intracellular PO4 pool. This should permit PO4 in cellular biomass to serve as a proxy for intracellular water isotopic composition, assuming complete exchange of oxygen in PO4 with oxygen in water and minimal subsequent overprinting by other cellular processes.
In contrast to previous studies reporting direct measurement of intracellular water from only one microbial strain, E. coli (27, 36), we tested several different strains of bacteria to look for species-/strain-specific effects and also used the technique of multilabeled water isotope probing (MLWIP), used in previous studies of metabolic water in rats (30) and further developed in our group to probe organophosphorus and phosphoenzyme reaction mechanisms (23, 42–44). Using this technique, multiple (three or more) labeled-water probes having different oxygen isotopic composition are used to interrogate reaction mechanisms and trace oxygen transfers between compounds in reactions. This technique contrasts with traditional isotope probing methods that typically use only a single probing compound that is usually strongly labeled with one (or more) heavy or even radioactive isotope(s). Importantly, MLWIP methods use both heavy and light isotopic probes that also have isotopic compositions close to natural abundances to provide both higher sensitivity and additional insights into the reaction mechanism, such as the fractionation involved during isotope exchange and transfer/replacement reactions (23, 29, 30, 42, 44). Here we used the MLWIP method, combined with graphical analysis, to both identify a metabolic water component in the intracellular water pool and to confirm the transfer of the oxygen isotopic signature of intracellular water to phosphate (PO4) in DNA and cellular biomass.
Results and Discussion
Results from 14 separate 1- to 2-L microbial culture experiments are shown in Table S1 and plotted in Figs. 2–4. Oxygen isotope ratios are reported as delta (δ) values in units of per mil and referenced to the Vienna Standard Mean Ocean Water (VSMOW) international reference standard. The δ18OPO4 values are reported with a precision of ±0.2–0.3‰. Growth experiments were carried out under optimal conditions of temperature, pH, and medium composition for each organism. All strains were grown aerobically. Each data point plotted in Figs. 3 and 4 represents a completely independent 1- to 2-L experimental growth system that underwent extensive processing to extract biomass and to extract/purify DNA [after Blake et al. (45)], which, due to the large volumes of these systems, required conventional chemical methods to extract and purify biomass/DNA with all reagents prepared from scratch and appropriately scaled (see SI Materials and Methods), versus the more typically used premade and ready-to-use small-scale DNA extraction kits used in studies involving PCR and/or sequencing. Added to this was an extensive series of steps to next extract PO4 from biomass/DNA, purify it, and convert it to silver phosphate (Ag3PO4) for stable oxygen isotope analysis. Despite the large number of complex steps involved in processing/analyzing these samples, the δ18OPO4 values of both DNA and biomass are highly consistent for a given set of conditions [e.g., δ18OWATER, temperature (T)], even across different microbial strains (Table S1). For example, the ∼−5.0‰ water culture experiments at 23–24 °C yielded DNA δ18OPO4 values for P.fluorescens, Acinetobacter ADP1, and M. aquaeolei of 17.2‰, 17.9‰, and 17.4‰, respectively, or 17.5 (±0.3)‰, and biomass δ18OPO4 values of 17.2‰, 16.6‰, and 16.8‰, respectively, or 16.9 (±0.3)‰. This high level of consistency attests to the rigor and reproducibility of our experimental and analytical protocols.
Table S1.
Results of DNA and biomass PO4 oxygen isotope analyses for 14 1-L batch culture experiments and calculation of the metabolic water component of intracellular water
| Microbial strains | T, °C | δ18OH2O, ‰ | δ18OPO4, ‰ | δ18OH2O,* ‰ | 1,000 ln α (PO4−H2O) |
| DNA | |||||
| P. fluorescens | 23 | −5.05 | 17.2 | −4.6 | 21.6 |
| 30 | −5.01 | 16.8 | −4.5 | 21.2 | |
| 37 | −5.02 | 14.0 | −4.6 | 18.5 | |
| Acinetobacter | 23 | −5.56 | 17.9 | −4.9 | 22.7 |
| 30 | −5.32 | 16.1 | −4.8 | 20.7 | |
| 37 | −5.09 | 15.5 | −4.6 | 20.0 | |
| M. aquaeolei | 24 | −5.57 | 17.4 | −4.9 | 22.2 |
| 37 | −5.54 | 13.9 | −4.9 | 18.7 | |
| 30 | 6.30 | 24.5 | 3.3 | 20.9 | |
| 37 | 6.28 | 22.0 | 3.3 | 18.5 | |
| 45 | 6.34 | 22.8 | 3.3 | 19.3 | |
| 30 | 28.75 | 39.5 | 18.8 | 20.2 | |
| 37 | 28.47 | 38.7 | 18.6 | 19.6 | |
| 45 | 28.20 | 39.3 | 18.4 | 20.3 | |
| Biomass | |||||
| P. fluorescens | 23 | −5.05 | 17.2 | −4.4 | 21.4 |
| 30 | −5.01 | 16.5 | −4.4 | 20.7 | |
| 37 | −5.02 | 15.3 | −4.4 | 19.6 | |
| Acinetobacter | 23 | −5.56 | 16.6 | −4.7 | 21.2 |
| 30 | −5.32 | 16.1 | −4.6 | 20.6 | |
| 37 | −5.09 | 15.9 | −4.4 | 20.2 | |
| M. aquaeolei | 24 | −5.57 | 16.8 | −4.7 | 21.4 |
| 37 | −5.54 | 15.2 | −4.7 | 20.3 | |
| 30 | 6.30 | 23.4 | 2.3 | 20.8 | |
| 37 | 6.28 | 22.4 | 2.3 | 19.9 | |
| 45 | 6.34 | 21.1 | 2.3 | 18.6 | |
| 30 | 28.75 | 37.7 | 15.5 | 21.6 | |
| 37 | 28.47 | 35.0 | 15.4 | 19.2 | |
| 45 | 28.20 | 35.3 | 15.2 | 19.6 |
The δ18OH2O value of intracellular water is recalculated based on the assumption that metabolic water with a δ18OH2O value of −3.5‰ accounts for 31% (for DNA) and 41% (for biomass) of intracellular water.
Fig. 2.
The δ18OPO4 of PO4 in (A) DNA and (B) biomass, versus δ18OWATER of extracellular water for cultures grown in 18O-labeled waters at different temperatures. Positive correlation between δ18Op and δ18Ow and the slope indicates that ∼31% of the O in DNA PO4 and ∼41% of the O in biomass PO4 is not derived from extracellular water.
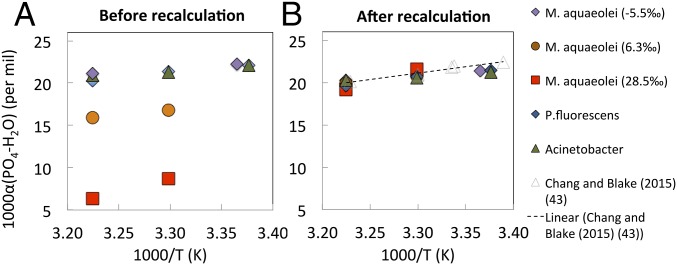
Fig. 4.
(A) Fractionation between biomass PO4 and intracellular water before considering the contribution of metabolic water to intracellular water; (B) recalculated values, based on the assumption that metabolic water with δ18OWATER = −3.5‰ accounts for 41% of intracellular water.
Fig. 3.
(A) Fractionation between DNA PO4 and intracellular water before considering the contribution of metabolic water to intracellular water; (B) recalculated values, based on the assumption that metabolic water with δ18OWATER = −3.5‰ accounts for 31% of intracellular water.
In previous studies (27), direct oxygen isotope analysis of intracellular water was achieved by extracting water from whole cells using a vacuum cryodistillation method. In the present study, we measured the oxygen isotopic composition of PO4 in DNA and total cellular biomass, which should reflect complete exchange of all 4 O atoms in PO4 with O in water due to the action of intracellular PPase (Fig. 1). Thus, PO4 δ18O values serve as a proxy for intracellular water due to the 100% transfer of water O to PO4, and also due to the consistent relationship between PO4 and water δ18O values that has been demonstrated by controlled laboratory experiments on PPase catalyzed PO4−water oxygen isotope exchange (42, 43).
It is also assumed that any PO4 initially present in growth medium components is cycled through the intracellular DIP pool and reset to equilibrium by PPase, and does not bypass this route and enter directly into newly synthesized biomass (Fig. 1). In other words, all PO4 within the intracellular pool, regardless of initial source, is reset to equilibrium with ambient water by intracellular PPase.
Two Lines of Evidence for a Metabolic Water Component in Intracellular Water.
Slope analysis of multilabeled water experiments.
We used MLWIP methods to detect oxygen transfers and probe reaction mechanisms. We first analyzed the δ18OPO4 values of biomass and DNA from experiments with multiple labeled-water probes by evaluating the slopes of plots of δ18OPO4 vs. the δ18O value of labeled water used in the experiment (Fig. 2 A and B). As explained above, such plots should give a slope of 1 in the case of wholesale exchange between PO4 and ambient (i.e., extracellular) water. For this study, plots of δ18OPO4 from DNA/biomass vs. δ18O of extracellular labeled waters (Fig. 2 A and B) yielded slopes of 0.59–0.69 (Fig. 2 A and B). This indicates that ∼30% of the O in DNA PO4, and ∼40% of the O in total microbial biomass PO4 is not derived from extracellular water. Note that the additional 10% nonextracellular water O in biomass compared with DNA, both from the same sample, could be an artifact of sample processing, for example, a reflection of more variable P−O bonding structures and sites for P−O hydrolysis in total biomass than in the pure diester structure of DNA. Also, possible contributions from other O species could be present, such as oxygen radicals that may have formed during the ultraviolet radiation (UVR) treatment step used to process the samples, whose oxygen isotopic compositions could not be determined.
This observation of slopes < 1 in δ18OPO4 vs. δ18OWATER space was the first of two lines of evidence for a metabolic water component in intracellular water based on δ18OPO4. We further tested the hypothesis that this 31–41% nonextracellular water component imprinted onto DNA/biomass PO4 could be metabolic water, by performing an equilibrium analysis.
Equilibrium analysis of MLWIP experimental results.
According to previously established relations based on PPase-catalyzed PO4−water exchange (Fig. S1) (42, 43) as well as empirical studies of biological phosphates (bioapatites) (e.g., ref. 23), the difference in PO4 and water oxygen isotopic compositions, δ18OPO4 − δ18OWATER (or 1,000 ln α(p-w)) or fractionation between δ18OPO4 and δ18OWATER, is unchanged at equilibrium and at a given temperature, even when the isotopic composition of either PO4 or water is changed. Thus, under the traditional assumption that intracellular water is identical in isotopic composition to extracellular water, the δ18O value of PO4 in equilibrium with extracellular water having a known δ18O value can be predicted. In the present experiments, however, these predicted biomass/DNA equilibrium δ18OPO4 values and invariant fractionation (1,000 ln α(p-w)) values were not observed (Figs. 3A and 4A).
Because, according to our hypothesis, intracellular dissolved PO4−water fractionations are expected to reflect equilibrium PO4−water exchange catalyzed by intracellular PPase, δ18Op and δ18OWATER data were evaluated by comparing observed PO4−water oxygen isotope fractionations (1,000 ln α(p-w)) to calculated equilibrium fractionations based on our recent calibrations based on PPase-catalyzed equilibrium exchange in the more relevant, dissolved PO4−water system (43). Again, assuming that intracellular water has the same isotopic composition as extracellular water, as shown in Figs. 3A and 4A, these calculated fractionations do not fit onto a single equilibrium line as expected.
Additionally, there is a clear trend in both DNA (Fig. 3A) and biomass (Fig. 4A) data that shows that the heavier the labeled water used to make growth media, the larger the offset of fractionations from expected equilibrium values. This second line of evidence suggests the influence of another factor in the system, a most likely candidate being some contribution to the intracellular water pool having a different isotopic composition than the extracellular water. Again, we posited that metabolic water was the most likely candidate.
Recalculation of Equilibrium PO4−Water Fractionations with a Metabolic Water Component.
To test the hypothesis that a contribution from metabolic water to the intracellular water pool was responsible for both the deviation of slopes in δ18OPO4 vs. δ18OWATER space from 1 (Fig. 2 A and B) and offsets of measured PO4−water fractionations from expected values, we recalculated these fractionations using the oxygen isotopic composition of metabolic water reported by Kreuzer-Martin et al. (27) for E. coli per below.
Assuming that the intracellular water is a mixture of extracellular water (69% for DNA and 59% for biomass) and metabolic water (31% for DNA and 41% for biomass) and using a δ18OWATER value of −3.5‰ for metabolic water (calculated based on direct analysis of intracellular water extracted from E. coli by cryogenic distillation and modeling) (27), we calculated a new oxygen isotopic composition for intracellular water based on these mixtures and then used this “mixed water” value to recalculate the fractionations between DNA/biomass PO4 and mixed water (Figs. 3B and 4B). Although, as stated above, ∼10% of O in biomass PO4 could be an artifact of O species formed during sample processing that cannot be determined, we assumed, for simplicity, that this 10% O had the same isotopic composition as metabolic water. Recalculated data plotted in Figs. 3B and 4B, show close agreement with expected equilibrium fractionations based on dissolved inorganic PO4−water systems (43). We recognize the apparent temperature dependence of PO4−water fractionations seen in Figs. 3B and 4B, which has been thoroughly explored in studies that have been submitted separately (45).
For the three organisms studied, M. aquaeolei, P. fluorescens, and Acinetobater ADP1 harvested at their late log/early stationary phase, metabolic activities result in an ∼31–41% contribution of metabolic water to the intracellular water pool, which is very close to the percentage metabolic water contribution (30%) determined previously by Kreuzer-Martin et al. (27), for only E. coli cells in stationary phase. Close agreement between previous results (27) and our new data sets based on 14 separate culture experiments using three additional strains of microorganisms from wide-ranging, natural habitats (marine water column, soils) and having different growth conditions/behavior than E. coli—an enteric microbe found in the gut/intestines of mammals—provides strong support for the previous results.
Importantly, the constant δ18OWATER value of −3.5‰ for metabolic water reported by Kreuzer-Martin et al. (27) is also supported by our results. Recalculation of DNA and biomass PO4−water fractionations based on the assumption that the δ18O value of metabolic water is −3.5‰, the same as for E. coli, is justifiable because central metabolic pathways in most microorganisms are similar, and thus the metabolic water produced should also be similar, given a similar source of O for this water. Although this O source cannot be further resolved based on results of the present study, its constant value points to a source such as atmospheric O2, which has a constant value of ∼+23.5‰ (46), or metabolic CO2, which exchanges readily with water, and a conserved central metabolic pathway characterized by constant oxygen isotopic fractionations.
There are three O sources contributing to the metabolic flux of O: intake/drinking water, atmospheric O2 (with a constant δ18O value of +23.5‰), and food/nutrient O. Changing the isotopic composition (i.e., food/nutrient/intake water/drinking water with various δ18O values) and/or relative contributions of any of these sources to the metabolic flux of O could potentially lead to metabolic water having variable oxygen isotopic compositions. For example, increased contribution of atmospheric O2 by increased aeration of culture vessels could increase cellular metabolic rates compared with poorly aerated (i.e., static) systems. This also suggests possible differences in metabolic water oxygen isotopic composition for aerobes vs. anaerobes. Organisms with different food/nutrient sources, such as those inhabiting surface vs. bottom waters, or coastal vs. pelagic ocean regions, could also have metabolic water with variable oxygen isotopic compositions.
Studies of isotopic compositions of metabolic water in small mammals indicate that the metabolic flux of O atoms is 56% from ingested/drinking water, 30% from atmospheric O2, and 15% from food (47). If the same holds true for microbes used in our study, then atmospheric O2 would contribute much more O to metabolic water than would nutrients in the growth medium (e.g., peptone, yeast extract), such that changes in nutrient oxygen isotopic composition may not significantly impact the δ18O value of metabolic water. The δ18O value of water used to prepare growth media in our studies was similar to that used by Kreuzer-Martin et al. (27). This, combined with a minimal contribution from O in nutrients and a constant oxygen isotopic composition of atmospheric O2, may explain the consistency in metabolic water oxygen isotopic composition (−3.5‰) observed between the two studies.
Summary and Conclusions
Results presented here from experiments using different strains of bacteria grown on media prepared with multiple 18O-labeled waters provide two independent lines of evidence for a significant contribution of metabolic water to the intracellular water pool that subsequently becomes imprinted onto PO4 in DNA and total biomass. First is the indication from plots of δ18OPO4 of PO4 in DNA/biomass vs. δ18O of extracellular water that less than 100% of O in PO4 is derived from extracellular water (i.e., slopes < 1), suggesting a possible metabolic water contribution. Second is the observation that calculated equilibrium PO4−water fractionations match expected values when it is assumed that the nonextracellular water component indicated by the slopes < 1 has an isotopic composition of −3.5‰, determined previously for metabolic water in E. coli cells (27). The consistency of this metabolic water value across different experiments, water δ18O compositions, and microbial strains warrants further investigation.
Results presented here strongly support earlier results based on direct measurement of intracellular water in E. coli (27) and have important implications for all other studies where it is assumed that intracellular and extracellular water have identical isotopic composition. For example, if it is found that other aquatic organisms, such as invertebrates (e.g., foraminifera, bivalves, gastropods), which are widely used as paleotemperature proxies in reconstructing Earth’s climate history (22), also have a significant metabolic water component in their intracellular water, then the temperatures calculated from the 18O/16O ratio of oxyanions (e.g., phosphate, carbonate) comprising the shells of these organisms may require reevaluation.
It is generally assumed that isotopic equilibrium exists between these biominerals and intracellular/body water, which is further assumed to have the same isotopic composition as extracellular water (19, 21–24). Sensitivity analysis shows that the inclusion of 30%/50%/70% of metabolic water to intracellular water would lead to an underestimation of temperature by ∼3/5/7 °C for a system where metabolic water is −3.5‰, while the extracellular water is −6‰. This error in paleotemperature estimation is proportional to the isotopic difference between metabolic water and ambient extracellular water. In modern marine systems where the ambient extracellular water (seawater) is ∼0‰, if the oxygen isotope value of metabolic water is still assumed to be −3.5‰, then 30%/50%/70% of intracellular water from metabolism will lead to an overestimation of temperature by ∼4/7/10 °C [1 per mil difference in water equals an ∼4.6–4.8 °C difference in temperature for the carbonate system (22) and an ∼4.2 °C difference in the phosphate system (23)].
Our results provide a new probe for intracellular water based on the oxygen isotopic composition of intracellular dissolved PO4 that is preserved in organophosphate of DNA and cell biomass/soft tissues. The intracellular water proxy presented here based on the δ18O value of PO4 in DNA, present in all cells, can be easily applied to a wide range of studies on cell metabolism and function, water balance, and biosynthetic pathways and has potentially widespread future applications in diverse fields of biology, ecology, plant physiology, biochemistry, geobiology, and beyond.
Materials and Methods
Microbial strains were cultured aerobically with manual shaking twice daily and at large-scale (1−2 L) to provide sufficient DNA for further phosphate O isotope analysis. Biomass was harvested when cells reached late log/early stationary phase. Large-scale DNA extraction was based on the method of Ausubel et al. (48), with minor modifications after Blake et al. (45) (Fig. S2). Following extraction and purification of DNA from total biomass, PO4 moieties in DNA and total biomass were treated using a UV digestion method developed in our laboratory (49, 50) to release free inorganic PO4. Phosphate concentrations were determined by standard colorimetric (molybdate blue) methods (51). Using methods modified from Kolodny et al. (21) and Tudge (52), PO4 was purified by precipitation as ammonium phosphomolybdate (APM) followed by reprecipitation as magnesium ammonium phosphate (MAP). Next, MAP crystals were dissolved and the resulting solution was treated with cation resin to remove magnesium and remaining impurities (42, 49, 50, 53). Phosphate was finally precipitated as silver phosphate for O isotope analysis. Oxygen isotope ratios of PO4 were determined using a Thermo-Chemolysis Elemental Analyzer (TC/EA) coupled to a DeltaplusXP isotope ratio monitoring mass spectrometer operating in continuous flow mode (Thermo-Finnigan). Water δ18O values were determined by standard CO2−water equilibration methods using a Thermo-Finnigan Gas Bench. All work (culture experiments, sample processing, and chemical analyses) was carried out at Yale University and all isotope analyses were performed at the Yale Analytical and Stable Isotope Center (YASIC) of the Yale Institute for Biospheric Studies.
Fig. S2.
Gel electrophoresis results of DNA extraction. The first column was loaded with a lambda HindIII ladder as a reference. Columns outlined in red are (from left to right) DNA extractions from Marinobacter aquaeolei grown at 23 °C in media made with three different waters (∼−5‰, 6‰, and 29‰) and Marinobacter aquaeolei grown at 37 °C in media made with three different waters (∼−5‰, 6‰, and 29‰).
SI Materials and Methods
Microbial Culture and Growth.
Bacteria representative of widely different natural environments were grown in 14 separate 1- to 2-L batch cultures, Marinobacter aquaeolei (facultative anaerobe), Pseudomonas fluorescens, and Acinetobacter ADP1 (obligate aerobe), using isotopically labeled waters ranging in δ18O value from −5.01‰ to + 28.75‰, which allowed verification of fractionations, probing of reaction mechanisms, and also detection and distinction of three different sources of water: ambient extracellular water, intracellular water, and metabolically produced water. Two groups of experiments using different microbial strains were conducted to replicate experiments and test for possible artifacts. Group 1 experiments included three strains with very different growth behavior and growth rates: Marinobacter aquaeolei, Pseudomonas fluorescens, and Acinetobacter ADP1. Group 2 experiments were conducted using Marinobacter aquaeolei only, but with different 18O-labeled waters ranging from −5.01‰ to +28.75‰. Bacteria were grown aerobically in 1- to 2-L culture flasks with manual shaking twice daily until the late log/early stationary phase was reached. Marinobacter aquaeolei cultures were grown on modified LB broth (40 g/L NaCl, 5 g/L tryptone, 5 g /L peptone, and 5 g/L yeast extract) at pH ∼7.3. Pseudomonas fluorescens and Acinetobacter ADP 1 were grown on Miller LB broth (10 g/L NaCl, 10 g/L tryptone, and 5 g/L yeast extract) at pH ∼7.0.
Microbial Biomass/DNA Collection and Processing.
For the analysis of PO4 in both DNA and total cellular biomass, cultures were harvested at late log to early stationary phase. Following standard methods for DNA extraction (48), cells were lysed with 10% SDS, and resulting suspensions were further treated with RNase, Proteinase K, CTAB/NaCl (10% hexadecyltrimethyl ammonium bromide/0.7 M NaCl) solution and chloroform/isoamyl alcohol (24:1) to purify DNA. The DNA was precipitated using isopropanol and checked for purity/quality by electrophoresis on a 0.7% agarose gel (Fig. S2). Shearing or other degradation of extracted DNA does not compromise the subsequent PO4 oxygen isotope analysis.
Extraction of PO4 in Microbial DNA/Biomass.
Following biomass harvesting and DNA extraction and purification, PO4 was extracted from DNA and biomass for δ18O analysis using methods first developed by Liang and Blake (44, 50, 54). DNA or biomass suspended in deionized water was treated with high-intensity ultraviolet radiation (UVR) to release free PO4 from the organophosphorus compounds—primarily phosphodiesters—making up cell biomass (e.g., nucleic acids, phospholipids). Reaction temperatures were maintained below 40 °C during UVR treatment, a temperature at which no reaction of released inorganic dissolved PO4 with water occurs based on parallel UVR treatments done on KH2PO4 standard solution controls (49, 50). Following UV digestion, the concentration of dissolved inorganic PO4 was determined for each sample using the colorimetric phosphomolybdate blue method (51).
Purification of PO4 and Precipitation as Ag3PO4.
Dissolved PO4 was first precipitated as APM, followed by dissolution and reprecipitation of dissolved PO4 as MAP after methods adapted from Tudge (52) and Kolodny et al. (21) and modified by Liang (49) and Blake et al. (55, 56). Next, MAP was dissolved, and the resulting solution was treated with cation resin to remove magnesium before final precipitation of dissolved PO4 as silver phosphate (Ag3PO4) for oxygen isotope analysis. Silver phosphate samples were thoroughly homogenized and then vacuum-roasted at 550 °C to remove residual water and organic matter. Following this treatment, Ag3PO4 crystals were shiny and yellow.
Mass Spectrometric Analysis.
Oxygen in Ag3PO4 was converted to CO by reacting with graphite at 1,450 °C in a TC/EA. The CO was then introduced into a continuous flow isotope ratio-monitoring mass spectrometer (DELTA PLUSXP) to determine the ratio of 18O/16O, expressed as δ18O values in per mil (VSMOW). The precision of these analyses was ±0.2–0.3‰. Measured δ18OPO4 values were calibrated against conventional fluorination values using a series of Ag3PO4 standards according to Vennemann et al. (24).
Water was sampled from microbial growth media at both the start and end of experiments and analyzed for δ18O using the CO2−water equilibration method (57) and a Themo Finnigan Gas Bench connected to a Finnigan DeltaplusXP stable isotope ratio mass spectrometer. The δ18O values of water samples were normalized to the oxygen isotope compositions of three laboratory water standards (−1.40‰, −8.96‰, and −3.76‰) calibrated against VSMOW. The overall analytical precision of the water δ18O analyses, on the basis of replicate analyses of the laboratory water standards under the same analytical conditions as for the water samples, was better than ±0.22‰ (1 SD). All oxygen isotope analyses were performed at the Yale Analytical and Stable Isotope Center (YASIC) of the Yale Institute for Biospheric Studies.
Acknowledgments
This work was supported primarily by grants awarded to R.E.B. from the US National Science Foundation (OCE 0928247 and OCE 1129499) and NASA (N00199-1073649), as well as: the Innovative Personnel Training Project of the University of Science and Technology Beijing and the International Joint Key Project from the Chinese Ministry of Science and Technology (2010DFB23160) supporting the participation of C.Y. (assigned to J.Y.); and the China Scholarship Council Visiting Post-Doctoral Research Project (awarded to F.W.).
Footnotes
The authors declare no conflict of interest.
This article is a PNAS Direct Submission.
This article contains supporting information online at www.pnas.org/lookup/suppl/doi:10.1073/pnas.1521038113/-/DCSupplemental.
References
- 1.Kreuzer HW, et al. Detection of metabolic fluxes of O and H atoms into intracellular water in mammalian cells. PLoS One. 2012;7(7):e39685. doi: 10.1371/journal.pone.0039685. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Schmidt-Nielsen B, Schmidt-Nielsen K. A complete account of the water metabolism in kangaroo rats and an experimental verification. J Cell Physiol. 1951;38(2):165–181. doi: 10.1002/jcp.1030380204. [DOI] [PubMed] [Google Scholar]
- 3.Schmidt-Nielsen K. Desert Animals: Physiological Problems of Heat and Water. Clarendon; London: 1964. [Google Scholar]
- 4.Frank CL. Diet selection by a heteromyid rodent: Role of net metabolic water production. Ecology. 1988;69(6):1943–1951. [Google Scholar]
- 5.Jindra M, Sehnal F. Linkage between diet humidity, metabolic water production and heat dissipation in the larvae of Galleria Mellonella. Insect Biochem. 1990;20(4):389–395. [Google Scholar]
- 6.Storm-Suke A, Wassenaar LI, Nol E, Norris DR. The influence of metabolic rate on the contribution of stable-hydrogen and oxygen isotopes in drinking water to quail blood plasma and feathers. Funct Ecol. 2012;26(5):1111–1119. [Google Scholar]
- 7.Longinelli A, Peretti AP. 1983. Oxygen isotopic composition of mammal bones as a possible tool for palaeoclimatic studies. First results. Palaeoclimates and palaeowaters: A Collection of Environmental Isotope Studies (Int Atom Agency, Vienna), pp 105–112.
- 8.Luz B, Kolodny Y. Oxygen isotope variation in bone phosphate. Appl Geochem. 1989;4(3):317–323. [Google Scholar]
- 9.Ayliffe LK, Chivas AR. Oxygen isotope composition of the bone phosphate of Australian kangaroos: Potential as a palaeoenvironmental recorder. Geochim Cosmochim Acta. 1990;54(9):2603–2609. [Google Scholar]
- 10.Fricke HC, O’Neil JR. Inter- and intra-tooth variation in the oxygen isotope composition of mammalian tooth enamel phosphate: Implications for paleoclimatological and palaeobiological research. Palaeogeogr Palaeoclimatol Palaeoecol. 1996;126(1):91–99. [Google Scholar]
- 11.Kohn MJ. Predicting animal δ18O: Accounting for diet and physiological adaptation. Geochim Cosmochim Acta. 1996;60(23):4811–4829. [Google Scholar]
- 12.Surge D, Walker KJ. Oxygen isotope composition of modern and archaeological otoliths from the estuarine hardhead catfish (Ariopsis felis) and their potential to record low-latitude climate change. Palaeogeogr Palaeoclimatol Palaeoecol. 2005;228(1-2):179–191. [Google Scholar]
- 13.Purton L, Brasier M. Gastropod carbonate δ18O and δ13C values record strong seasonal productivity and stratification shifts during the late Eocene in England. Geology. 1997;25(10):871–874. [Google Scholar]
- 14.Rosenmeier MF, et al. A model of the 4000-year paleohydrology (δ18O) record from Lake Salpetén, Guatemala. Global Planet Change. 2016;138:43–55. [Google Scholar]
- 15.Shackleton NJ, Opdyke ND. Oxygen isotope and paleomagnetic stratigraphy of equatorial Pacific core V28-238: Oxygen isotope temperatures and ice volumes on a 105 year and 106 year scale. Quat Res. 1973;3(1):39–55. [Google Scholar]
- 16.Lécuyer C, Grandjean P, Emig CC. Determination of oxygen isotope fractionation between water and phosphate from living lingulids: Potential application to palaeoenvironmental studies. Palaeogeogr Palaeoclimatol Palaeoecol. 1996;126(1-2):101–108. [Google Scholar]
- 17.Zachos J, Pagani M, Sloan L, Thomas E, Billups K. Trends, rhythms, and aberrations in global climate 65 Ma to present. Science. 2001;292(5517):686–693. doi: 10.1126/science.1059412. [DOI] [PubMed] [Google Scholar]
- 18.Zachos JC, Dickens GR, Zeebe RE. An early Cenozoic perspective on greenhouse warming and carbon-cycle dynamics. Nature. 2008;451(7176):279–283. doi: 10.1038/nature06588. [DOI] [PubMed] [Google Scholar]
- 19.Joachimski MM, et al. Devonian climate and reef evolution: Insights from oxygen isotopes in apatite. Earth Planet Sci Lett. 2009;284(3-4):599–609. [Google Scholar]
- 20.Grossman EL. Stable isotopes in modern benthic foraminifera: A study of vital effect. J Foraminiferal Res. 1987;17(1):48–61. [Google Scholar]
- 21.Kolodny Y, Luz B, Navon O. Oxygen isotope variations in phosphate of biogenic apatites, 1. Fish bone apatite-rechecking the rules of the game. Earth Planet Sci Lett. 1983;64(3):398–404. [Google Scholar]
- 22.Bemis BE, Spero HJ. Reevaluation of the oxygen isotopic composition of planktonic foraminifera: Experimental results and revised paleotemperature equations. Paleoceanography. 1998;13(2):150–160. [Google Scholar]
- 23.Pucéat E, et al. Revised phosphate–water fractionation equation reassessing paleotemperatures derived from biogenic apatite. Earth Planet Sci Lett. 2010;298(1-2):135–142. [Google Scholar]
- 24.Vennemann TW, Fricke HC, Blake RE, O’Neil JR, Colman A. Oxygen isotope analysis of phosphates: A comparison of techniques for analysis of Ag3PO4. Chem Geol. 2002;185(3-4):321–336. [Google Scholar]
- 25.Schoeller DA, van Santen E. Measurement of energy expenditure in humans by doubly labeled water method. J Appl Physiol. 1982;53(4):955–959. doi: 10.1152/jappl.1982.53.4.955. [DOI] [PubMed] [Google Scholar]
- 26.Pivarnik JM, Leeds EM, Wilkerson JE. Effects of endurance exercise on metabolic water production and plasma volume. J Appl Physiol. 1984;56(3):613–618. doi: 10.1152/jappl.1984.56.3.613. [DOI] [PubMed] [Google Scholar]
- 27.Kreuzer-Martin HW, Ehleringer JR, Hegg EL. Oxygen isotopes indicate most intracellular water in log-phase Escherichia coli is derived from metabolism. Proc Natl Acad Sci USA. 2005;102(48):17337–17341. doi: 10.1073/pnas.0506531102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Longinelli A, Peretti AP. Oxygen isotopic composition of water from mammal blood: First results. Eur J Mass Spectrom Biochem Med Environ Res. 1980;1:135–139. [Google Scholar]
- 29.Longinelli A. Oxygen isotopes in mammal bone phosphate: A new tool for paleohydrological and paleoclimatological research? Geochim Cosmochim Acta. 1984;48(2):385–390. [Google Scholar]
- 30.Luz B, Kolodny Y. Oxygen isotope variations in phosphate of biogenic apatites, IV. Mammal teeth and bones. Earth Planet Sci Lett. 1985;75(1):29–36. [Google Scholar]
- 31.Luz B, Cormie AB, Schwarcz HP. Oxygen isotope variations in phosphate of deer bones. Geochim Cosmochim Acta. 1990;54(6):1723–1728. [Google Scholar]
- 32.Longinelli A, Iacumin P, Davanzo S, Nikolaev V. Modern reindeer and mice: Revised phosphate–water isotope equations. Earth Planet Sci Lett. 2003;214(3-4):491–498. [Google Scholar]
- 33.Luz B, Kolodny Y, Kovach J. Oxygen isotope variations in phosphate of biogenic apatites, III. Conodonts. Earth Planet Sci Lett. 1984;69(2):255–262. [Google Scholar]
- 34.Bryant JD, Froelich PN. A model of oxygen isotope fractionation in body water of large mammals. Geochim Cosmochim Acta. 1995;59(21):4523–4537. [Google Scholar]
- 35.Madigan MT, Martinko JM. Brock Biology of Microorganisms. 11th Ed Pearson Prentice Hall; Upper Saddle River, NJ: 2006. [Google Scholar]
- 36.Kreuzer-Martin HW, Lott MJ, Ehleringer JR, Hegg EL. Metabolic processes account for the majority of the intracellular water in log-phase Escherichia coli cells as revealed by hydrogen isotopes. Biochemistry. 2006;45(45):13622–13630. doi: 10.1021/bi0609164. [DOI] [PubMed] [Google Scholar]
- 37.D’Angela D, Longinelli A. Oxygen isotopes in living mammals’ bone phosphate: Further results. Chem Geol. 1990;86(1):75–82. [Google Scholar]
- 38.Faure G. Principles of Isotope Geology. 2nd Ed Wiley; New York: 1986. [Google Scholar]
- 39.Epstein S, Buchsbaum R, Lowenstam HA, Urey HC. Revised carbonate-water isotopic temperature scale. Geol Soc Am Bull. 1953;64(11):1315–1326. [Google Scholar]
- 40.Longinelli A, Nuti S. Revised phosphate-water isotopic temperature scale. Earth Planet Sci Lett. 1973;19(3):373–376. [Google Scholar]
- 41.Kim ST, O’Neil JR. Equilibrium and non-equilibrium oxygen isotope effects in synthetic carbonates. Geochim Cosmochim Acta. 1997;61(16):3461–3475. [Google Scholar]
- 42.Blake RE, O’Neil JR, Surkov AV. Biogeochemical cycling of phosphorus: Insights from oxygen isotope effects of phosphoenzymes. Am J Sci. 2005;305(6-8):596–620. [Google Scholar]
- 43.Chang SJ, Blake RE. Precise calibration of equilibrium oxygen isotope fractionations between dissolved phosphate and water from 3 to 37 °C. Geochim Cosmochim Acta. 2015;150:314–329. [Google Scholar]
- 44.Liang Y, Blake RE. Oxygen isotope signature of P(i) regeneration from organic compounds by phosphomonoesterases and photooxidation. Geochim Cosmochim Acta. 2006a;70(15):3957–3969. [Google Scholar]
- 45.Blake RE, et al. DNA thermometry: New information from ‘the information molecule’ and a universal biothermometer in the PO4 backbone of DNA. Am J Sci. 2016 in press. [Google Scholar]
- 46.Kroopnick P, Craig H. Atmospheric oxygen: Isotopic composition and solubility fractionation. Science. 1972;175(4017):54–55. doi: 10.1126/science.175.4017.54. [DOI] [PubMed] [Google Scholar]
- 47.Podlesak DW, et al. Turnover of oxygen and hydrogen isotopes in the body water, CO2, hair, and enamel of a small mammal. Geochim Cosmochim Acta. 2008;72(1):19–35. [Google Scholar]
- 48.Ausubel FM, et al. Short Protocols in Molecular Biology. 4th Ed Wiley; New York: 1999. [Google Scholar]
- 49.Liang Y. 2005. Oxygen isotope studies of biogeochemical cycling of phosphorus. Ph.D. dissertation (Yale Univ, New Haven, CT)
- 50.Liang Y, Blake RE. Oxygen isotope composition of phosphate in organic compounds: Isotope effects of extraction methods. Org Geochem. 2006b;37(10):1263–1277. [Google Scholar]
- 51.Koroleff F. Determination of nutrients. In: Grasshoff K, Ehrhardt M, Kremling K, editors. Methods of Seawater Analysis. 2nd Ed. Verlag Chemie; New York: 1983. pp. 125–187. [Google Scholar]
- 52.Tudge AP. A method of analysis of oxygen isotopes in orthophosphates—Its use in the measurement of paleotemperatures. Geochim Cosmochim Acta. 1960;18:81–93. [Google Scholar]
- 53.Blake RE, Alt JC, Martini AM. Oxygen isotope ratios of PO4: An inorganic indicator of enzymatic activity and P metabolism and a new biomarker in the search for life. Proc Natl Acad Sci USA. 2001;98(5):2148–2153. doi: 10.1073/pnas.051515898. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Liang Y, Blake RE. Compound- and enzyme-specific phosphodiester hydrolysis mechanisms revealed by δ18O of dissolved inorganic phosphate: Implications for marine P cycling. Geochim Cosmochim Acta. 2009;73(13):3782–3794. [Google Scholar]
- 55.Blake RE, O’Neil JR, Garcia GA. Oxygen isotope systematics of biologically mediated reactions of phosphate. 1. Microbial degradation of organophosphorus compounds. Geochim Cosmochim Acta. 1997;61(20):4411–4422. [Google Scholar]
- 56.Blake RE, O’Neil JR, Garcia GA. Effects of microbial activity on the δ18O of dissolved inorganic phosphate and textural features of synthetic apatites. Am Mineral. 1998;83(11):1516–1531. [Google Scholar]
- 57.Cohn M, Urey HC. Oxygen exchange reactions of organic compounds and water. J Am Chem Soc. 1938;60(3):679–687. [Google Scholar]