Abstract
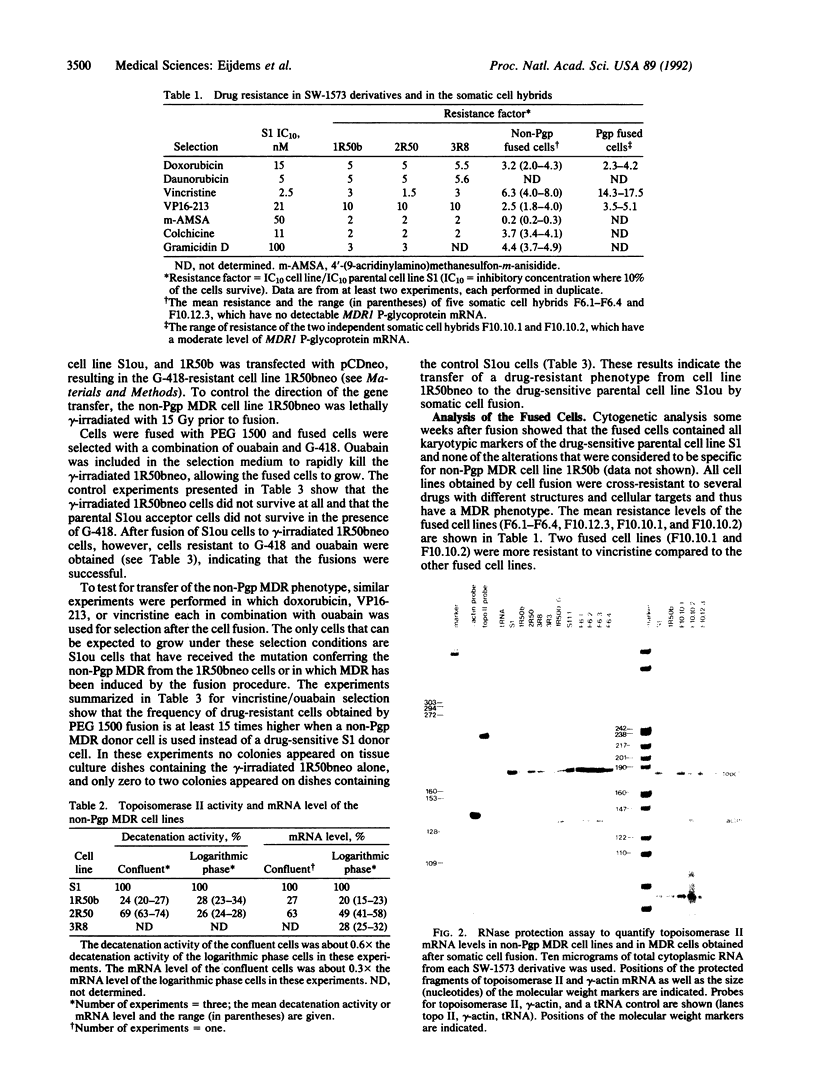
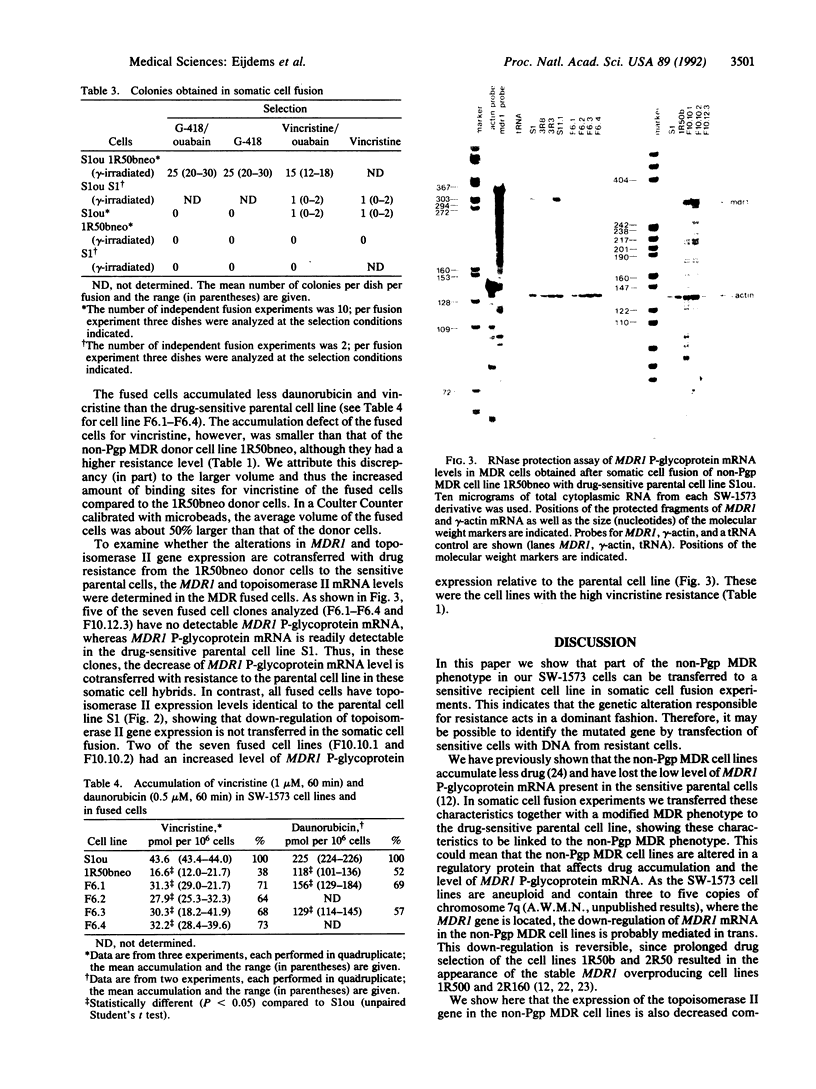
A non-P-glycoprotein-mediated mechanism of multidrug resistance (non-Pgp MDR) has been identified in doxorubicin-selected sublines of the human non-small cell lung carcinoma cell line SW-1573. These sublines are cross-resistant to daunorubicin, VP16-213, Vinca alkaloids, colchicine, gramicidin D, and 4'-(9-acridinylamino)methanesulfon-m-anisidide (m-AMSA). They accumulate less drug than the parental cells and their resistance is not due to the MDR1-encoded P-glycoprotein, as the resistant cell lines have lost the low amount of MDR1 mRNA detectable in parental cells. Here we show that the resistant cell lines also contain less topoisomerase II mRNA and enzyme activity than the parental cells. This might contribute to the resistance of these lines to drugs interacting with topoisomerase II, such as doxorubicin, daunorubicin, and VP16-213, but cannot account for the resistance to the other drugs. We have tested whether all properties of the non-Pgp MDR cell lines cosegregate in somatic cell fusions between lethally gamma-irradiated, resistant donor cells and drug-sensitive acceptor cells. Whereas a MDR phenotype with reduced drug accumulation and the loss of MDR1 P-glycoprotein mRNA were cotransferred to the acceptor cells, the decrease in topoisomerase II gene expression was not. We conclude that the MDR phenotype, the reduced drug accumulation, and the loss of MDR1 P-glycoprotein mRNA are genetically linked. They might be due to a single dominant mutation, which does not cause the alteration in topoisomerase II.
Full text
PDF


Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Baas F., Jongsma A. P., Broxterman H. J., Arceci R. J., Housman D., Scheffer G. L., Riethorst A., van Groenigen M., Nieuwint A. W., Joenje H. Non-P-glycoprotein mediated mechanism for multidrug resistance precedes P-glycoprotein expression during in vitro selection for doxorubicin resistance in a human lung cancer cell line. Cancer Res. 1990 Sep 1;50(17):5392–5398. [PubMed] [Google Scholar]
- Beck W. T., Cirtain M. C., Danks M. K., Felsted R. L., Safa A. R., Wolverton J. S., Suttle D. P., Trent J. M. Pharmacological, molecular, and cytogenetic analysis of "atypical" multidrug-resistant human leukemic cells. Cancer Res. 1987 Oct 15;47(20):5455–5460. [PubMed] [Google Scholar]
- Biedler J. L., Riehm H. Cellular resistance to actinomycin D in Chinese hamster cells in vitro: cross-resistance, radioautographic, and cytogenetic studies. Cancer Res. 1970 Apr;30(4):1174–1184. [PubMed] [Google Scholar]
- Broxterman H. J., Kuiper C. M., Schuurhuis G. J., Tsuruo T., Pinedo H. M., Lankelma J. Increase of daunorubicin and vincristine accumulation in multidrug resistant human ovarian carcinoma cells by a monoclonal antibody reacting with P-glycoprotein. Biochem Pharmacol. 1988 Jun 15;37(12):2389–2393. doi: 10.1016/0006-2952(88)90365-6. [DOI] [PubMed] [Google Scholar]
- Broxterman H. J., Pinedo H. M., Kuiper C. M., van der Hoeven J. J., de Lange P., Quak J. J., Scheper R. J., Keizer H. G., Schuurhuis G. J., Lankelma J. Immunohistochemical detection of P-glycoprotein in human tumor cells with a low degree of drug resistance. Int J Cancer. 1989 Feb 15;43(2):340–343. doi: 10.1002/ijc.2910430229. [DOI] [PubMed] [Google Scholar]
- Chen C. J., Chin J. E., Ueda K., Clark D. P., Pastan I., Gottesman M. M., Roninson I. B. Internal duplication and homology with bacterial transport proteins in the mdr1 (P-glycoprotein) gene from multidrug-resistant human cells. Cell. 1986 Nov 7;47(3):381–389. doi: 10.1016/0092-8674(86)90595-7. [DOI] [PubMed] [Google Scholar]
- Chen C., Okayama H. High-efficiency transformation of mammalian cells by plasmid DNA. Mol Cell Biol. 1987 Aug;7(8):2745–2752. doi: 10.1128/mcb.7.8.2745. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Danks M. K., Yalowich J. C., Beck W. T. Atypical multiple drug resistance in a human leukemic cell line selected for resistance to teniposide (VM-26). Cancer Res. 1987 Mar 1;47(5):1297–1301. [PubMed] [Google Scholar]
- Dano K. Cross resistance between vinca alkaloids and anthracyclines in Ehrlich ascites tumor in vivo. Cancer Chemother Rep. 1972 Dec;56(6):701–708. [PubMed] [Google Scholar]
- Deffie A. M., Batra J. K., Goldenberg G. J. Direct correlation between DNA topoisomerase II activity and cytotoxicity in adriamycin-sensitive and -resistant P388 leukemia cell lines. Cancer Res. 1989 Jan 1;49(1):58–62. [PubMed] [Google Scholar]
- Endicott J. A., Ling V. The biochemistry of P-glycoprotein-mediated multidrug resistance. Annu Rev Biochem. 1989;58:137–171. doi: 10.1146/annurev.bi.58.070189.001033. [DOI] [PubMed] [Google Scholar]
- Enoch T., Zinn K., Maniatis T. Activation of the human beta-interferon gene requires an interferon-inducible factor. Mol Cell Biol. 1986 Mar;6(3):801–810. doi: 10.1128/mcb.6.3.801. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ferguson P. J., Fisher M. H., Stephenson J., Li D. H., Zhou B. S., Cheng Y. C. Combined modalities of resistance in etoposide-resistant human KB cell lines. Cancer Res. 1988 Nov 1;48(21):5956–5964. [PubMed] [Google Scholar]
- Glisson B., Gupta R., Smallwood-Kentro S., Ross W. Characterization of acquired epipodophyllotoxin resistance in a Chinese hamster ovary cell line: loss of drug-stimulated DNA cleavage activity. Cancer Res. 1986 Apr;46(4 Pt 2):1934–1938. [PubMed] [Google Scholar]
- Gottesman M. M., Pastan I. The multidrug transporter, a double-edged sword. J Biol Chem. 1988 Sep 5;263(25):12163–12166. [PubMed] [Google Scholar]
- Keizer H. G., Schuurhuis G. J., Broxterman H. J., Lankelma J., Schoonen W. G., van Rijn J., Pinedo H. M., Joenje H. Correlation of multidrug resistance with decreased drug accumulation, altered subcellular drug distribution, and increased P-glycoprotein expression in cultured SW-1573 human lung tumor cells. Cancer Res. 1989 Jun 1;49(11):2988–2993. [PubMed] [Google Scholar]
- Kent R. B., Emanuel J. R., Ben Neriah Y., Levenson R., Housman D. E. Ouabain resistance conferred by expression of the cDNA for a murine Na+, K+-ATPase alpha subunit. Science. 1987 Aug 21;237(4817):901–903. doi: 10.1126/science.3039660. [DOI] [PubMed] [Google Scholar]
- Lai S. L., Goldstein L. J., Gottesman M. M., Pastan I., Tsai C. M., Johnson B. E., Mulshine J. L., Ihde D. C., Kayser K., Gazdar A. F. MDR1 gene expression in lung cancer. J Natl Cancer Inst. 1989 Aug 2;81(15):1144–1150. doi: 10.1093/jnci/81.15.1144. [DOI] [PubMed] [Google Scholar]
- Marini J. C., Miller K. G., Englund P. T. Decatenation of kinetoplast DNA by topoisomerases. J Biol Chem. 1980 Jun 10;255(11):4976–4979. [PubMed] [Google Scholar]
- McGrath T., Center M. S. Adriamycin resistance in HL60 cells in the absence of detectable P-glycoprotein. Biochem Biophys Res Commun. 1987 Jun 30;145(3):1171–1176. doi: 10.1016/0006-291x(87)91560-9. [DOI] [PubMed] [Google Scholar]
- Merkel D. E., Fuqua S. A., Tandon A. K., Hill S. M., Buzdar A. U., McGuire W. L. Electrophoretic analysis of 248 clinical breast cancer specimens for P-glycoprotein overexpression or gene amplification. J Clin Oncol. 1989 Aug;7(8):1129–1136. doi: 10.1200/JCO.1989.7.8.1129. [DOI] [PubMed] [Google Scholar]
- Mirski S. E., Gerlach J. H., Cole S. P. Multidrug resistance in a human small cell lung cancer cell line selected in adriamycin. Cancer Res. 1987 May 15;47(10):2594–2598. [PubMed] [Google Scholar]
- Pommier Y., Kerrigan D., Schwartz R. E., Swack J. A., McCurdy A. Altered DNA topoisomerase II activity in Chinese hamster cells resistant to topoisomerase II inhibitors. Cancer Res. 1986 Jun;46(6):3075–3081. [PubMed] [Google Scholar]
- Schinkel A. H., Borst P. Multidrug resistance mediated by P-glycoproteins. Semin Cancer Biol. 1991 Aug;2(4):213–226. [PubMed] [Google Scholar]
- Simpson A. M., Simpson L. Isolation and characterization of kinetoplast DNA networks and minicircles from Crithidia fasciculata. J Protozool. 1974 Nov;21(5):774–781. doi: 10.1111/j.1550-7408.1974.tb03751.x. [DOI] [PubMed] [Google Scholar]
- Slovak M. L., Hoeltge G. A., Dalton W. S., Trent J. M. Pharmacological and biological evidence for differing mechanisms of doxorubicin resistance in two human tumor cell lines. Cancer Res. 1988 May 15;48(10):2793–2797. [PubMed] [Google Scholar]
- Sullivan D. M., Glisson B. S., Hodges P. K., Smallwood-Kentro S., Ross W. E. Proliferation dependence of topoisomerase II mediated drug action. Biochemistry. 1986 Apr 22;25(8):2248–2256. doi: 10.1021/bi00356a060. [DOI] [PubMed] [Google Scholar]
- Tsai-Pflugfelder M., Liu L. F., Liu A. A., Tewey K. M., Whang-Peng J., Knutsen T., Huebner K., Croce C. M., Wang J. C. Cloning and sequencing of cDNA encoding human DNA topoisomerase II and localization of the gene to chromosome region 17q21-22. Proc Natl Acad Sci U S A. 1988 Oct;85(19):7177–7181. doi: 10.1073/pnas.85.19.7177. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Versantvoort C. H., Broxterman H. J., Pinedo H. M., de Vries E. G., Feller N., Kuiper C. M., Lankelma J. Energy-dependent processes involved in reduced drug accumulation in multidrug-resistant human lung cancer cell lines without P-glycoprotein expression. Cancer Res. 1992 Jan 1;52(1):17–23. [PubMed] [Google Scholar]
- Weber J. M., Sircar S., Horvath J., Dion P. Non-p-glycoprotein-mediated multidrug resistance in detransformed rat cells selected for resistance to methylglyoxal bis(guanylhydrazone). Cancer Res. 1989 Nov 1;49(21):5779–5783. [PubMed] [Google Scholar]
- Zinn K., DiMaio D., Maniatis T. Identification of two distinct regulatory regions adjacent to the human beta-interferon gene. Cell. 1983 Oct;34(3):865–879. doi: 10.1016/0092-8674(83)90544-5. [DOI] [PubMed] [Google Scholar]
- de Jong S., Zijlstra J. G., de Vries E. G., Mulder N. H. Reduced DNA topoisomerase II activity and drug-induced DNA cleavage activity in an adriamycin-resistant human small cell lung carcinoma cell line. Cancer Res. 1990 Jan 15;50(2):304–309. [PubMed] [Google Scholar]
- van der Bliek A. M., Borst P. Multidrug resistance. Adv Cancer Res. 1989;52:165–203. doi: 10.1016/s0065-230x(08)60213-4. [DOI] [PubMed] [Google Scholar]